Abstract
Herpes zoster results from latent varicella zoster virus reactivation in the dorsal root ganglia, causing blistering rash along the dermatomal distribution and post-herpetic neuralgia. Increasing studies indicated that there may be a correlation between herpes zoster and COVID-19. Nevertheless, the detailed pathophysiological mechanism is still unclear. We used bioinformatic analyses to study the potential genetic crosstalk between herpes zoster and COVID-19. COVID-19 and herpes zoster were associated with a similar subset of genes involved in “cytokine-cytokine receptor interaction,” “Jak-STAT signaling pathway,” and “IL-17 signaling pathway,” including TNF, IL10, ESR1, INFG, HLA-A, CRP, STAT3, IL6, IL7, and IL17A. Protein-protein interaction network assay showed that the combined gene set indicated a raised connectivity as compared to herpes zoster or COVID-19 alone, particularly the potentiated interactions with APOE, ARSA, CCR2, CCR5, CXCL13, EGFR, GAL, GP2, HLA-B, HLA-DRB1, IL5, TECTA, and THBS1, and these genes are related to “cytokine-cytokine receptor interaction”. Augmented Th17 cell differentiation and the resulting enhanced IL-17 signaling were identified in both COVID-19 and herpes zoster. Our data suggested aberrant interleukin-17 signaling as one possible mechanism through which COVID-19 could raise the risk of herpes zoster.
Keywords: SARS-CoV-2, Varicella zoster virus, Cytokine, Jak-STAT
Introduction
The coronavirus disease 2019 (COVID-19) pandemic caused by the SARS-CoV-2, a positive-sense RNA virus containing a large genome of 30 kb in size likely originated from bats, has resulted in a huge death toll and an unprecedented economic loss worldwide (Beck da Silva Etges et al. 2021, Davillas and Jones 2021, Dunne et al. 2021, Kumari 2021, Sood et al. 2020). As with 17 April 2020, over 139 million people globally have been diagnosed of COVID-19, causing nearly 3 million deaths (Lin et al. 2017). Although 40 to 50% of patients tested positive for COVID-19 are asymptomatic, some COVID-19 patients could develop severe symptoms, such as high fever, severe cough, and shortness of breath, and progress to acute respiratory distress syndrome and multiple organ failure (Harmon et al. 2021, Li et al. 2021, Yanes-Lane et al. 2020). The respiratory system is the primary site of attack by SARS-CoV-2, but growing number of evidences indicated that COVID-19 might influence other organ systems, including the skin and the nervous system (Birlutiu et al. 2021, Kyriakoudi et al. 2021, McMahon et al. 2021, Terzi et al. 2021). In this regard, COVID-19 may lead to cutaneous manifestations including widespread or localized urticaria, erythematous rash, or herpes zoster (Andina-Martinez et al. 2021, Pagali and Parikh 2021, Sachdeva et al. 2020). SARS-CoV-2 may also spread to the brain via both hematogenous and neural routes (El-Sayed et al. 2021b). Endeavors have been put forth to understand the pathogenic mechanism of SARS-CoV-2, especially the underlying factors triggering respiratory failure (El-Sayed et al. 2021a).
Herpes zoster (commonly known as shingles), common in middle age groups, is caused by the reactivation varicella zoster virus (VZV) and manifests as the painful vesicular rash along a dermatomal distribution (Nagel and Gilden 2013). VZV is also the causative agent of varicella (commonly known as chickenpox) that predominantly affects children under the age of 12 (Bérar et al. 2021, Shiraki et al. 2021). Following the initial infection, this virus remains latent in the ganglion cells of dorsal root or cranial nerve (Fukuyasu et al. 2021, Osman 2020, Zhu et al. 2020). Increasing age and immunosuppression are two major risk factors for VZV reactivation (Amaral et al. 2021). The most frequent complication of herpes zoster is post-herpetic neuralgia (Andrei and Snoeck 2021, Rahmatpour Rokni et al. 2020). Other complications include cutaneous (secondary bacterial infection, scarring), neurologic (motor neuropathy, meningoencephalitis), ocular (keratitis, iritis, retinitis), and visceral (pneumonitis, hepatitis) aspects (Nagel and Gilden 2013).
Recently, a growing number of studies showed that herpes zoster might be associated with COVID-19. Recalcati reported for the first time that varicella-like vesicles could be a cutaneous manifestation of COVID-19 (Recalcati 2020). In a later multicenter case series, Marzano et al. reported that 22 patients with a COVID-19–positive nasopharyngeal swab developed varicella-like papulovesicular exanthem with a predominant trunk involvement (Marzano et al. 2020). Fernandez-Nieto et al. also reported that among 53 COVID-19 patients who developed vesicular rashes, 15 patients presented typical clinical lesions and symptoms of herpes simplex/zoster (Fernandez-Nieto et al. 2020). Ertugrul and Aktas also reported that the number of patients with herpes zoster seeking medical attention in Turkey was remarkably increased in May and June 2020 during the COVID-19 outbreak as compared to the same period in 2019 (Cao et al. 2020). By searching the diagnostic code from the hospital automation system, Maia et al. also reported an increased number of herpes zoster cases in Brazil during the pandemic period (Maia et al. 2021). As the COVID-19 pandemic continues, these clinical characteristic of COVID-19-dependent herpes zoster may continue to be expound. However, the detailed pathological mechanisms about the disease entity remain undefined. In our research, we utilized bioinformatic method to clarify the potential crosstalk between herpes zoster- and COVID-19-associated genes.
Materials and methods
Identification of herpes zoster- and COVID-19-associated genes
The genes information related to these two disease entities (COVID-19, disease id: C000657245, version 5, N.PMIDs ≥ 10; herpes zoster, disease id: C0019360, Gene-Disease Association Score ≥ 0.01) was searched from DisGeNET, a platform about disease genomics, on 17 April 2021.
Protein-protein interaction networks of herpes zoster- and COVID-19-associated genes
The abovementioned gene sets, alone or in combination, were chosen to functional protein correlation networks assay by STRING. Protein-protein interactions were visualized with minimum interaction score of 0.900 (and the highest confidence) and disconnected nodes hidden in this network. Only “databases,” “experiments,” “co-occurrence,” and “co-expression” were used as the active interaction sources. The MLC (Markov Cluster) algorithm with the inflation parameter of 3 was then utilized for the network clustering.
Pathway enrichment analysis
KEGG (Kyoto Encyclopedia of Genes and Genomes) pathway enrichment assay was utilized with the STRING built-in function to clarify the biological functions of COVID-19- and herpes zoster-correlated gene networks.
Results
Protein-protein interaction networks of COVID-19- and herpes zoster-associated genes indicated common enrichment in cytokine-cytokine receptor interaction
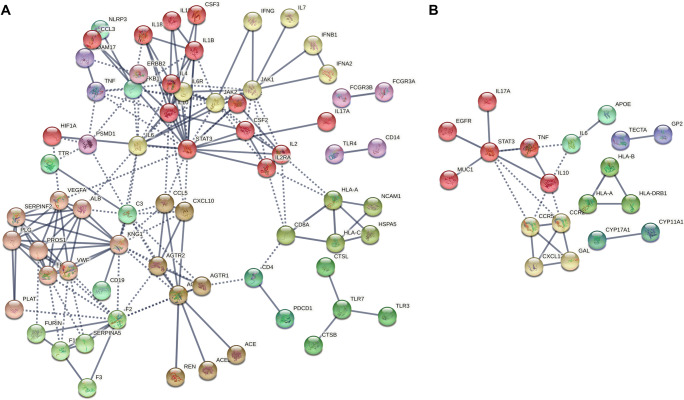
We first defined the list of genes involved in the pathogenesis of COVID-19 and herpes zoster using a disease-based pathway analysis. A total of 114 and 51 genes correlated with COVID-19 (disease id: C000657245, version 5; N.PMIDs ≥ 10) and herpes zoster (disease id: C0019360; Gene-Disease Association Score ≥ 0.01), respectively, were choose from DisGeNET (Table 1). Disease-related genes overlapped between COVID-19 and herpes zoster are TNF, IL10, ESR1, INFG, HLA-A, CRP, STAT3, IL6, IL7, and IL17A, which are enriched in the KEGG pathways “cytokine-cytokine receptor interaction,” “Jak-STAT signaling pathway,” and “IL-17 signaling pathway.” We reconstructed the protein-protein correlation networks among genes using STRING and identified connecting genes (Figure 1A for COVID-19; Figure 1B for herpes zoster). Notably, STAT3 indicated a high connectivity in the both herpes zoster- and COVID-19-associated gene networks with number of interacting partners of 21 and 8, respectively. The functional prediction further showed that “cytokine-cytokine receptor interaction,” “Jak-STAT signaling pathway,” “hematopoietic cell lineage,” “antigen processing and presentation,” “IL-17 signaling pathway,” “T cell receptor signaling pathway,” “intestinal immune network for IgA production,” and “Th17 cell differentiation” were then enriched in both these gene networks (Table 2).
Table 1.
Disease-associated genes retrieved from DisGeNET. Genes associated with both COVID-19 and herpes zoster are bolded. COVID-19, coronavirus disease 2019
| Diseases | Disease-associated genes |
|---|---|
| COVID-19 | ACE2, CRP, IL6, ACE, REN, TNF, TMPRSS2, CD4, CD8A, AGT, ALB, IL1B, GPT, F2, IL10, LOC102724971, LOC102723407, INS, IFNG, IFNA1, FURIN, CSF2, DPP4, TNNI3, F3, IL2, GOT1, CRX, IFNB1, NFKB1, AGTR1, RPGR, IL6R, PLAT, CALCA, IL17A, SERPINA13P, HLA-C, HBA1, CENPJ, IL2RA, NELFCD, MB, CXCL10, VWF, CTSL, NLRP3, CTRL, CTSB, IL4, SPECC1, MAS1, ZFYVE9, MS4A1, PDCD1, RTN1, RTN4, BSG, MTOR, VEGFA, PLG, KRT20, F8, JAK1, SERPINA5, CD19, IFNA2, PSMD1, STAT3, TTR, KNG1, NPPB, EMSLR, AGTR2, CSF3, IL7, IL18, PROS1, LINC01672, ESR1, NFE2L2, EEF1A2, ERBB2, G6PD, GGT1, NCAM1, ADAM17, TLR4, GGTLC5P, GGRLC3, GGT2, GGTLC4P, FCGR3A, FCGR3B, HSPA5, IL1RN, INSRR, JAK2, CCL3, CCL5, TLR3, KLK4, SH2D3C, WDTC1, PGR-AS1, C3, CD14, F10, HIF1A, HLA-A, SERPINF2, SLC5A2, ADAMTS2, TLR7 |
| Herpes zoster | HLA-B, LRRC32, LINC02785, HCP5, FAM110B, TMC7, TNF, IL10, CPS1, HLA-DRB1, ESR1, APOE, IFNG, HLA-A, ARSA, CRP, RBM45, PART1, GAL, ENOSF1, STAT3, TECTA, THBS1, CCR2, TRPV1, CCR5, TMPRSS13, LMLN, LINC02605, IL33, BOD1, RECQL4, MUC1, MTX1, MST1, GP2, ETFA, CXCL13, EGFR, CYP17A1, CYP11A1, IL5, IL6, IL7, CTLA4, IL17A, MBL2, MECP2, MIP, MSMB, CD34 |
Figure 1.
Functional protein-protein correlation networks of the COVID-19 (A) and herpes zoster-related (B) genes were determined using STRING. Genes correlated with these 2 disease entities (herpes zoster, disease id: C0019360, Gene-Disease Association Score ≥ 0.01; COVID-19, id: C000657245, version 5, N.PMIDs ≥ 10) were retrieved from the DisGeNET, a platform for the disease genomics. Only highly confident interactions with minimum required interaction score of 0.900, derived from “databases,” “co-occurrence,” “co-expression,” and “experiments,” were counted. Network clustering was done with the MLC (Markov cluster) algorithm with an inflation parameter of 3
Table 2.
Significantly enriched “KEGG Pathways” in COVID-19- and herpes zoster-associated gene networks. KEGG pathways are ranked according to the false discovery rate (all < 0.0001) in an ascending order. Pathways under the category of “human diseases” were omitted. Pathways associated with both COVID-19 and herpes zoster are bolded. COVID-19, coronavirus disease 2019
| Diseases | KEGG pathways of disease-associated gene networks |
|---|---|
| COVID-19 | Cytokine-cytokine receptor interaction, hematopoietic cell lineage, Jak-STAT signaling pathway, Th17 cell differentiation, PI3K-Akt signaling pathway, Toll-like receptor signaling pathway, complement and coagulation cascades, NOD-like receptor signaling pathway, HIF-1 signaling pathway, necroptosis, cytosolic DNA-sensing pathway, natural killer cell mediated cytotoxicity, IL-17 signaling pathway, renin-angiotensin system, antigen processing and presentation, T cell receptor signaling pathway, AGE-RAGE signaling pathway in diabetic complications, EGFR tyrosine kinase inhibitor resistance, Th1 and Th2 cell differentiation, phagosome, osteoclast differentiation, TNF signaling pathway, RIG-I-like receptor signaling pathway, intestinal immune network for IgA production, NF-kappa B signaling pathway, renin secretion, Insulin resistance |
| Herpes zoster | Cytokine-cytokine receptor interaction, Jak-STAT signaling pathway, hematopoietic cell lineage, antigen processing and presentation, IL-17 signaling pathway, T cell receptor signaling pathway, Th17 cell differentiation, intestinal immune network for IgA production |
Potentiating and additive interactions of COVID-19- and herpes zoster-correlative gene networks
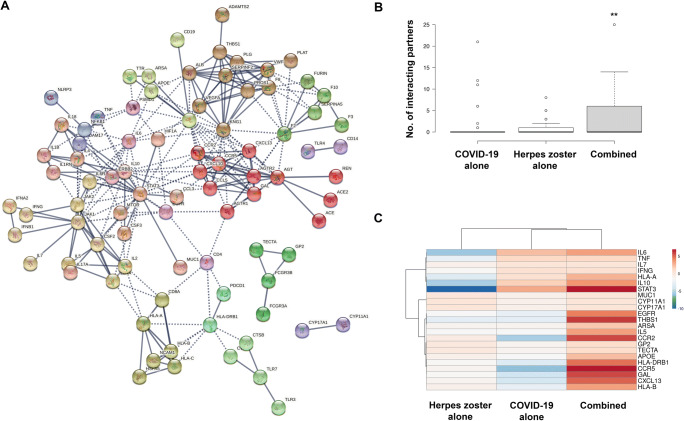
To study the potential interactions between the herpes zoster- and COVID-19-correlated gene networks, two genes sets were subject and combined to the STRING assay. The network of combined genes indicated a better integration than that of herpes zoster- and COVID-19-correlated genes alone, with many clusters previously divorced from the major network now rejoined (e.g., CTSB/ TLR7-CTSL/TLR3, TLR4-CD14, FCGR3B-FCGR3A clusters in COVID-19; HLA-A-HLA-B-HLA-DRB1 cluster in herpes zoster) or expanded (TECTA-GP2 cluster in herpes zoster) (Figure 2A). To quantitatively study the impact of the COVID-19 on herpes zoster-correlated gene networks, the amount of these genes connecting with 51 herpes zoster-correlated genes were calculated in herpes zoster-alone, COVID-19-alone, and COVID-19-herpes zoster-correlated networks. The 51 GBS-correlated genes were connected to significant genes in combined network as compared to 2 disease entities alone (Figure 2B). Clustering and visualization of this connectivity with the heap map indicated two distinct patterns of interactions, namely additive (e.g., HLA-A, IL10, IL6, IL7, IFNG, MUC1, STAT3, TNF) and synergistic (e.g., APOE, ARSA, CCR2, CCR5, CXCL13, EGFR, GAL, GP2, HLA-B, HLA-DRB1, IL5, TECTA, THBS1) interactions (Figure 2C). The latter are significantly enriched in “cytokine-cytokine receptor interaction.”
Figure 2.
STRING analysis of combined COVID-19/herpes zoster gene sets. A Interaction map shows better integration of COVID-19- and herpes zoster-associated clusters. B Herpes zoster-associated genes showed higher connectivity with other genes in the combined network as compared to COVID-19- or herpes zoster-alone network. C Heap map visualizes the additive and synergistic effects of incorporating COVID-19-associated genes into of herpes zoster-associated gene interacting network on the network connectivity. **, P < .01, significantly different by non-parametric paired assay of the mount of connecting genes of herpes zoster-associated genes in the protein-protein interaction networks using Wilcoxon matched-pairs signed rank test
Discussion
Emerging evidence supports that herpes zoster might be a cutaneous manifestation of COVID-19 (Brancaccio et al. 2021, Jamshidi et al. 2021). In this regard, it has been postulated that COVID-19-associated immunosuppression (e.g., suppressed T cell-mediated immunity) or the psychological stress inflicted by the pandemic might promote VZV reactivation and thus account for the escalating number of herpes zoster cases (Pona et al. 2020). In this study, by bioinformatic analysis of COVID-19- and herpes zoster-associated genes, we found that both diseases are associated with “cytokine-cytokine receptor interaction,” “Jak-STAT signaling pathway,” and “IL-17 signaling pathway,” in which STAT3 acts as a central hub in the protein-protein interaction networks of both diseases. To this end, STAT3 is known to mediate the differentiation of naive CD4+ T cells into T helper 17 (Th17) cells, which secretes IL-17A, IL-21, and IL-22 to promote inflammatory processes, such as recruitment of neutrophils (Egwuagu 2009).
IL-17 signaling is important for the clearance of bacterial and fungal infections but its role in viral infection remains poorly defined (Lin et al. 2021, Xue et al. 2021). In COVID-19, lymphocytes isolated from these cases were demonstrated to secret more IL-17 (De Biasi et al. 2020). In herpes zoster, a recent study showed that patients had a higher number of circulating Th17 cells without alteration of the number of regulatory T cells (Kim et al. 2017). Concordantly, the serum level of IL-17 was reported to be higher in patients with herpes zoster (Zajkowska et al. 2016). These findings collectively suggest that deregulated cytokine responses, particularly the differentiation of Th17 cells and IL-17 signaling, were linked to pathogenesis of COVID-19-correlated herpes zoster. Nevertheless, it is still unclear if IL-17 elevation is the cause or consequence of VZV reactivation. In this connection, a recent study showed that IL-17A could directly support reactivation and de novo lytic infection of γ-herpesvirus (whereas VZV belongs to the α-herpesvirus family) (Jondle et al. 2021). Our present study thus suggests that COVID-19 might induce excessive Th17 differentiation to increase the circulating level of IL-17A, which in turn triggers VZV reactivation to increase the risk of herpes zoster.
By defining the genes involved in COVID-19 and herpes zoster, we delineated the protein-protein interaction networks of these two diseases, in which Th17 cell differentiation and IL-17 signaling were found to play a common role. Although zoster vaccine and antiviral therapy (e.g., acyclovir started within 72 h after the onset of the rash) have been proven effective for preventing and treating herpes zoster, respectively (Gershon et al. 2015, Rosamilia 2020), the best strategies to clinically manage COVID-19-associated herpes zoster remained undefined. To this end, our data suggested that the augmented Th17 cell differentiation and IL-17 signaling might underlie the increased risk of herpes zoster among patients with COVID-19. It is therefore worthwhile to investigate in the future if modulation of IL-17 signaling could be of any prophylactic and therapeutic value to this disease entity.
Data availability statement
Research data will be shared upon request.
Author contribution
XY and LL conceptualized the study and analyzed the data. WKKW performed the bioinformatic analysis. XY, MTVC, and WKKW revised the manuscript.
Funding
This study was supported by the National Natural Science Foundation of P.R. China (Grant Number: 81903194).
Declarations
Ethical approval
This study was approved by the Clinical Ethics Committee of Beijing Friendship Hospital.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Linfeng Li, Email: zoonli@sina.com.
William Ka Kei Wu, Email: wukakei@cuhk.edu.hk.
References
- Amaral V, Shi J, Tsang A, Chiu S. Primary varicella zoster infection compared to varicella vaccine reactivation associated meningitis in immunocompetent children. J Paediatr Child Health. 2021;57:19–25. doi: 10.1111/jpc.15303. [DOI] [PubMed] [Google Scholar]
- Andina-Martinez D, Nieto-Moro M, Alonso-Cadenas J, Añon-Hidalgo J, Hernandez-Martin A, Perez-Suarez E, Colmenero-Blanco I, Iglesias-Bouza M, Cano-Fernandez J, Mateos-Mayo A, Torrelo A. Mucocutaneous manifestations in hospitalized children with COVID-19. J Am Acad Dermatol. 2021;85:88–94. doi: 10.1016/j.jaad.2021.03.083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrei G, Snoeck R (2021) Advances and Perspectives in the Management of Varicella-Zoster Virus Infections. Molecules:26 [DOI] [PMC free article] [PubMed]
- Beck da Silva Etges A, et al. The economic impact of COVID-19 treatment at a hospital-level: investment and financial registers of Brazilian hospitals. J Health Econ Outcomes Res. 2021;8:36–41. doi: 10.36469/jheor.2021.22066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bérar A, Ardois S, Walter-Moraux P, Jegonday M, Henriot B. Primary varicella-zoster virus infection of the immunocompromised associated with acute pancreatitis and hemophagocytic lymphohistiocytosis: a case report. Medicine. 2021;100:e25351. doi: 10.1097/MD.0000000000025351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birlutiu V, Feiereisz A, Oprinca G, Dobritoiu S, Rotaru M, Birlutiu R, Iancu G (2021) Cutaneous manifestations associated with anosmia, ageusia and enteritis in SARS-CoV-2 infection - a possible pattern? Observational study and review of the literature. Int J Inf Dis Off Pub Int Soc Inf Dis [DOI] [PMC free article] [PubMed]
- Brancaccio G, Gussetti N, Sasset L, Alaibac M, Tarantello M, Salmaso R, Trevenzoli M, Cattelan A (2021) Cutaneous manifestations in a series of 417 patients with SARS-CoV-2 infection: epidemiological and clinical correlates of chilblain like lesions. Pathogens and Global Health:1–4 [DOI] [PMC free article] [PubMed]
- Cao X, Zhang X, Meng W, Zheng H. Herpes zoster and postherpetic neuralgia in an elderly patient with critical COVID-19: a case report. J Pain Res. 2020;13:2361–2365. doi: 10.2147/JPR.S274199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davillas A, Jones A. Unmet health care need and income-related horizontal equity in use of health care during the COVID-19 pandemic. Health Econ. 2021;30:1711–1716. doi: 10.1002/hec.4282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Biasi S, et al. Marked T cell activation, senescence, exhaustion and skewing towards TH17 in patients with COVID-19 pneumonia. Nat Commun. 2020;11:3434. doi: 10.1038/s41467-020-17292-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunne E, Morgan E, Wells-Moore B, Pierson S, Zakroff S, Haskell L, Link K, Powell J, Holland I, Elgethun K, Ball C, Haugen R, Hahn C, Carter K, Starr C. COVID-19 outbreaks in correctional facilities with work-release programs - Idaho, July-November 2020. Morb Mortal Wkly Rep. 2021;70:589–594. doi: 10.15585/mmwr.mm7016a3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Egwuagu C. STAT3 in CD4+ T helper cell differentiation and inflammatory diseases. Cytokine. 2009;47:149–156. doi: 10.1016/j.cyto.2009.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Sayed A, Abdel-Daim MM, Kamel M. Causes of respiratory failure in COVID-19 patients. Environ Sci Pollut Res Int. 2021;28:28825–28830. doi: 10.1007/s11356-021-14200-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Sayed A, Aleya L, Kamel M (2021b) COVID-19: a new emerging respiratory disease from the neurological perspective. Environ Sci Pollut Res Int [DOI] [PMC free article] [PubMed]
- Fernandez-Nieto D, Ortega-Quijano D, Suarez-Valle A, Burgos-Blasco P, Jimenez-Cauhe J, Fernandez-Guarino M. Comment on: “To consider varicella-like exanthem associated with COVID-19, virus varicella zoster and virus herpes simplex must be ruled out. Characterization of herpetic lesions in hospitalized COVID-19 patients”. J Am Academy Dermatol. 2020;83:e257–e259. doi: 10.1016/j.jaad.2020.06.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuyasu A, Kamata M, Sy Hau C, Nagata M, Fukaya S, Hayashi K, Tanaka T, Ishikawa T, Ohnishi T, Tada Y, Kanda N. Serum interleukin-10 level increases in patients with severe signs or symptoms of herpes zoster and predicts the duration of neuralgia. J Dermatol. 2021;48:511–518. doi: 10.1111/1346-8138.15818. [DOI] [PubMed] [Google Scholar]
- Gershon AA, Breuer J, Cohen JI, Cohrs RJ, Gershon MD, Gilden D, Grose C, Hambleton S, Kennedy PG, Oxman MN, Seward JF, Yamanishi K. Varicella zoster virus infection. Nat Rev Dis Prim. 2015;1:15016. doi: 10.1038/nrdp.2015.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harmon K, Fliss M, Marshall S, Peticolas K, Proescholdbell S, Waller A. The impact of the COVID-19 pandemic on the utilization of emergency department services for the treatment of injuries. Am J Emerg Med. 2021;47:187–191. doi: 10.1016/j.ajem.2021.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jamshidi P, Hajikhani B, Mirsaeidi M, Vahidnezhad H, Dadashi M, Nasiri M. Skin manifestations in COVID-19 patients: are they indicators for disease severity? A systematic review. Front Med. 2021;8:634208. doi: 10.3389/fmed.2021.634208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jondle C, Johnson K, Aurubin C, Sylvester P, Xin G, Cui W, Huppler A, Tarakanova V (2021) Gammaherpesvirus usurps host IL-17 signaling to support the establishment of chronic infection. mBio:12 [DOI] [PMC free article] [PubMed]
- Kim M, Kim D, Na C, Shin B. A study of the changes of T helper 17 cells and regulatory T cells in herpes zoster. Ann Dermatol. 2017;29:578–585. doi: 10.5021/ad.2017.29.5.578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumari S (2021) Understanding of Stigmatization and Death Amid COVID-19 in India. A Sociological Exploration Omega 302228211008753 [DOI] [PMC free article] [PubMed]
- Kyriakoudi A, Pontikis K, Tsaraklis A, Soura E, Vourlakou C, Kossyvakis A, Potamianou E, Kaniaris E, Ioannidou I, Mentis A, Kloukina I, Daganou M, Koutsoukou A. Cutaneous vasculopathy in a COVID-19 critically ill patient: a histologic, immunohistochemical, and electron microscopy study. Case Rep Crit Care. 2021;2021:6644853. doi: 10.1155/2021/6644853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z, Huang Z, Li X, Huang C, Shen J, Li S, Zhang L, Wong S, Chan M, Wu W (2021) Bioinformatic analyses hinted at augmented T helper 17 cell differentiation and cytokine response as the central mechanism of COVID-19-associated Guillain-Barré syndrome. Cell Prolif:e13024 [DOI] [PMC free article] [PubMed]
- Lin H, Qiu S, Xie L, Liu C, Sun S. Nimbolide suppresses non-small cell lung cancer cell invasion and migration via manipulation of DUSP4 expression and ERK1/2 signaling. Biomed Pharmacother. 2017;92:340–346. doi: 10.1016/j.biopha.2017.05.072. [DOI] [PubMed] [Google Scholar]
- Lin X, Fu B, Yin S, Li Z, Liu H, Zhang H, Xing N, Wang Y, Xue W, Xiong Y, Zhang S, Zhao Q, Xu S, Zhang J, Wang P, Nian W, Wang X, Wu H. ORF8 contributes to cytokine storm during SARS-CoV-2 infection by activating IL-17 pathway. Iscience. 2021;24:102293. doi: 10.1016/j.isci.2021.102293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maia C, Marques N, de Lucena E, de Rezende L, Martelli D, Martelli-Júnior H. Increased number of Herpes Zoster cases in Brazil related to the COVID-19 pandemic. Int J Inf Dis Off Pub Int Soc Inf Dis. 2021;104:732–733. doi: 10.1016/j.ijid.2021.02.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marzano A, Genovese G, Fabbrocini G, Pigatto P, Monfrecola G, Piraccini B, Veraldi S, Rubegni P, Cusini M, Caputo V, Rongioletti F, Berti E, Calzavara-Pinton P. Varicella-like exanthem as a specific COVID-19-associated skin manifestation: multicenter case series of 22 patients. J Am Acad Dermatol. 2020;83:280–285. doi: 10.1016/j.jaad.2020.04.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMahon D, Amerson E, Rosenbach M, Lipoff J, Moustafa D, Tyagi A, Desai S, French L, Lim H, Thiers B, Hruza G, Blumenthal K, Fox L, Freeman E. Cutaneous reactions reported after Moderna and Pfizer COVID-19 vaccination: a registry-based study of 414 cases. J Am Acad Dermatol. 2021;85:46–55. doi: 10.1016/j.jaad.2021.03.092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagel M, Gilden D. Complications of varicella zoster virus reactivation. Curr Treat Options Neurol. 2013;15:439–453. doi: 10.1007/s11940-013-0246-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osman S. Successful oral treatment of third cranial nerve palsy and optic neuritis from neglected herpes zoster in an immunocompetent patient. Am J Ophthal Case Rep. 2020;20:100953. doi: 10.1016/j.ajoc.2020.100953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pagali S, Parikh R. Severe urticarial rash as the initial symptom of COVID-19 infection. BMJ Case Rep. 2021;14:e241793. doi: 10.1136/bcr-2021-241793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pona A, Jiwani R, Afriyie F, Labbe J, Cook P, Mao Y. Herpes zoster as a potential complication of coronavirus disease 2019. Dermatol Ther. 2020;33:e13930. doi: 10.1111/dth.13930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahmatpour Rokni G, Rathod D, Tabarestani M, Mirabi A, Jha A, de Vita V, Mashhadi Kholerdi H, Feliciani C, Jafferany M, Wollina U, Lotti T, Goldust M. Postherpetic neuralgia and recalcitrant cystic lesions following herpes zoster: a case report and review of treatment options. Dermatol Ther. 2020;33:e13793. doi: 10.1111/dth.13793. [DOI] [PubMed] [Google Scholar]
- Recalcati S. Cutaneous manifestations in COVID-19: a first perspective. J Europ Academy Dermatol Venereol. 2020;34:e212–e213. doi: 10.1111/jdv.16387. [DOI] [PubMed] [Google Scholar]
- Rosamilia LL. Herpes Zoster presentation, management, and prevention: a modern case-based review. Am J Clin Dermatol. 2020;21:97–107. doi: 10.1007/s40257-019-00483-1. [DOI] [PubMed] [Google Scholar]
- Sachdeva M, Gianotti R, Shah M, Bradanini L, Tosi D, Veraldi S, Ziv M, Leshem E, Dodiuk-Gad R. Cutaneous manifestations of COVID-19: Report of three cases and a review of literature. J Dermatol Sci. 2020;98:75–81. doi: 10.1016/j.jdermsci.2020.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shiraki K, Takemoto M, Daikoku T (2021) Emergence of varicella-zoster virus resistance to acyclovir: epidemiology, prevention, and treatment. Expert Rev Anti-Infect Ther:1–11 [DOI] [PubMed]
- Sood S, Aggarwal V, Aggarwal D, Upadhyay SK, Sak K, Tuli HS, Kumar M, Kumar J, Talwar S (2020) COVID-19 pandemic: from molecular biology, pathogenesis, detection, and treatment to global societal impact. Cur Pharmacol Rep:1–16 [DOI] [PMC free article] [PubMed]
- Terzi K, Kesici S, Özsürekci Y, Bayrakci B (2021) Periorbital erythema is a common cutaneous manifestation in COVID-19. Clin Exp Dermatol [DOI] [PMC free article] [PubMed]
- Xue T, Liu Y, Cao M, Zhang X, Fu Q, Yang N, Li C. Genome-wide identification of interleukin-17 (IL-17)/interleukin-17 receptor (IL- 17R) in turbot (Scophthalmus maximus) and expression pattern analysis after Vibrio anguillarum infection. Dev Comp Immunol. 2021;121:104070. doi: 10.1016/j.dci.2021.104070. [DOI] [PubMed] [Google Scholar]
- Yanes-Lane M, Winters N, Fregonese F, Bastos M, Perlman-Arrow S, Campbell J, Menzies D. Proportion of asymptomatic infection among COVID-19 positive persons and their transmission potential: a systematic review and meta-analysis. PLoS One. 2020;15:e0241536. doi: 10.1371/journal.pone.0241536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zajkowska A, Garkowski A, Świerzbińska R, Kułakowska A, Król M, Ptaszyńska-Sarosiek I, Nowicka-Ciełuszecka A, Pancewicz S, Czupryna P, Moniuszko A, Zajkowska J. Evaluation of chosen cytokine levels among patients with herpes zoster as ability to provide immune response. PLoS One. 2016;11:e0150301. doi: 10.1371/journal.pone.0150301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu J, Fei Y, Deng J, Huang B, Yao M. Application and therapeutic effect of puncturing of the costal transverse process for pulsed radiofrequency treated T1-T3 herpes zoster neuralgia. J Pain Res. 2020;13:2519–2527. doi: 10.2147/JPR.S266481. [DOI] [PMC free article] [PubMed] [Google Scholar]