Abstract
Background
Immunosuppressive therapies proposed for Coronavirus disease 2019 (COVID-19) management may predispose to secondary infections. We evaluated the association of immunosuppressive therapies with bloodstream-infections (BSIs) in hospitalized COVID-19 patients.
Methods
This was an institutional review board-approved retrospective, multicenter, cohort study of adults hospitalized with COVID-19 over a 5-month period. We obtained clinical, microbiologic and laboratory data from electronic medical records. Propensity-score-matching helped create balanced exposure groups. Demographic characteristics were compared across outcome groups (BSI/no BSI) using two-sample t-test and Chi-Square test for continuous and categorical variables respectively, while immunosuppressive therapy use was compared using McNemar’s test. Conditional logistic regression helped assess the association between immunosuppressive therapies and BSIs.
Results
13,007 patients were originally included, with propensity-score-matching producing a sample of 6,520 patients. 3.74% and 3.97% were diagnosed with clinically significant BSIs in the original and propensity-score-matched populations respectively. COVID-19 patients with BSIs had significantly longer hospitalizations, higher intensive care unit admission and mortality rates compared to those without BSIs. On univariable analysis, combinations of corticosteroids/anakinra [odds-ratio (OR) 2.00, 95% confidence intervals (C.I.) 1.05-3.80, P value.0342] and corticosteroids/tocilizumab [OR 2.13, 95% C.I. 1.16–3.94, P value .0155] were significantly associated with BSIs. On multivariable analysis (adjusting for confounders), combination corticosteroids/tocilizumab were significantly associated with any BSI [OR 1.97, 95% C.I. 1.04–3.73, P value.0386] and with bacterial BSIs [OR 2.13, 95% C.I. 1.12–4.05, p-value 0.0217].
Conclusions
Combination immunosuppressive therapies were significantly associated with BSI occurrence in COVID-19 patients; their use warrants increased BSI surveillance. Further studies are needed to establish their causative role.
Keywords: bacteremia, COVID-19, fungemia, immunosuppressive therapy, SARS-CoV-2
Immunosuppressive therapies predispose to secondary infections. We retrospectively evaluated the association of immunosuppressive therapies with bloodstream infection (BSI) incidence in hospitalized adults with COVID-19, using propensity score matching and multivariable logistic regression. Combination corticosteroid-tocilizumab therapy was significantly associated with BSIs.
In December 2019, the World Health Organization (WHO) was notified about cases of pneumonia of unknown etiology originating in Wuhan, China [1]. The causative agent was identified as a novel coronavirus, subsequently classified as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1]. The acute respiratory disease, named coronavirus disease 2019 (COVID-19), was declared a pandemic by the WHO on 11 March 2020 [1]. As of 28 April 2021, >148 million cases with approximately 3.1 million deaths have been reported globally due to COVID-19 [2].
Of several therapies investigated for the treatment of COVID-19, only the antiviral remdesivir has been approved by the US Food and Drug Administration [3]. As severe cases of COVID-19 have been associated with a “cytokine storm” (intense immune response) and presumed secondary hemophagocytic lymphohistiocytosis, there is immense interest in the use of immunosuppressive medications including corticosteroids and interleukin (IL) inhibitors [3–6]. Corticosteroids have been used in COVID-19 patients, including those admitted to the intensive care unit (ICU) [5, 7, 8]. Recent prospective studies, particularly the Randomized Evaluation of Covid-19 Therapy (RECOVERY) trial have shown survival benefit with dexamethasone in COVID-19 patients who received supplemental oxygen [9, 10]. Tocilizumab, an IL-6 receptor (IL-6R) antagonist used in rheumatoid arthritis, showed promise in both retrospective and observational studies [11, 12]. Additionally, in recent prospective studies, tocilizumab helped prevent disease progression, reduced need for mechanical ventilation, and improved mortality in intubated COVID-19 patients [13–15]. However, these findings were not replicated in other studies [16, 17]. Anakinra, a recombinant IL-1 receptor (IL-1R) antagonist, is another immunomodulating agent with favorable outcomes reported in limited cohort studies; however, prospective clinical trial data are lacking [18].
However, there is evidence to indicate that immunosuppressive medications may predispose to infections in COVID-19 patients [4, 8, 19]. IL inhibitors have been associated with a wide spectrum of infections [20]. Bloodstream infections (BSIs) have also been reported in COVID-19 patients, though specific risk factors were not elucidated [21, 22].
In this large retrospective multisite cohort study, we evaluated the incidence of hospital-acquired BSIs in COVID-19 patients admitted to several hospitals of a large health care system. We also assessed whether use of immunosuppressive therapies was associated with occurrence of these BSIs.
Our primary aim was to evaluate the association of immunosuppressive therapies used for the treatment of COVID-19 in hospitalized patients with the incidence of subsequent BSIs. Our secondary aims were to compare outcomes (survival/death) and laboratory parameters between COVID-19 patients with BSIs and those without BSIs.
We hypothesized that hospitalized COVID-19 patients receiving immunosuppressive therapies were at greater risk for BSIs, compared to those who did not receive these medications.
MATERIALS AND METHODS
This was a retrospective observational cohort study of hospitalized patients at 5 academic tertiary and 7 community hospitals in a large health system. The institutional review board approved the study as minimal-risk research and waived the requirement for informed consent.
Patients who presented to one of these hospitals between 1 March 2020 and 13 August 2020 were initially evaluated for inclusion in the study. Patients aged ≥18 years, diagnosed with COVID-19 infection (by positive reverse-transcription polymerase chain reaction [PCR] test on nasopharyngeal or bronchoalveolar lavage fluid specimen) and requiring hospitalization were included. We excluded patients discharged from the emergency department, those still admitted at time of data analysis, those enrolled in a randomized double-blind clinical trial involving the IL inhibitor sarilumab, patients diagnosed with BSIs >48 hours prior to COVID-19 diagnosis, and patients who developed BSIs within 48 hours of hospitalization. Clinical outcomes were recorded up to 22 September 2020 (date on which data analysis was initiated).
Only patients with BSIs meeting study definitions (Supplementary Table 1) were included. The “immunosuppressed” group included patients who received any of the following treatments for COVID-19: systemic corticosteroids (prednisone, prednisolone, dexamethasone, methylprednisolone, hydrocortisone), the IL-1R antagonist anakinra, or the IL-6R antagonist tocilizumab. The “non-immunosuppressed” group received none of these.
Data were collected from an electronic health record database (Sunrise Clinical Manager, Allscripts, Chicago, Illinois). We collected the following data: demographic information, comorbidities, and ICU admission; laboratory parameters closest to COVID-19 diagnosis (within 48 hours of diagnosis) and closest to positive blood culture results (within 48 hours of BSIs); details of immunosuppressive therapies received during hospitalization; and patient outcomes (eg, length of stay [LOS], mortality, discharge).
Study Definitions
A BSI (bacteremia and/or fungemia) in an immunosuppressed group patient was considered clinically significant (Definition 1) if growth occurred within 60 days of first dose of immunosuppressive therapy and after at least 2 days of hospital admission (Supplementary Table 1). For the non-immunosuppressed group (Definition 2), a BSI was considered clinically significant if growth occurred after at least 2 days of hospital admission and within 82 days of date of COVID-19 diagnosis (this cutoff was chosen based on the sum of median time to blood culture positivity in the immunosuppressed group [22 days] and the 60-day cutoff followed for the immunosuppressed group). Patients who had BSIs prior to receiving immunosuppressive therapy were included in the non-immunosuppressed group.
Polymicrobial infections (Definition 3) were defined as growth of different isolates within 5 days of initial positive blood culture. For patients with blood cultures growing commensal organisms (Definition 4) (as defined by the National Healthcare Safety Network [NHSN] Patient Safety Component Manual), 2 infectious diseases clinicians performed independent reviews of medical records to distinguish pathogens from contaminants based on clinical relevance [23]. Only BSIs considered pathogens were included. In cases of discordance between them, a third physician performed an independent blinded review.
STATISTICAL ANALYSIS
Propensity score matching was used to improve comparability of the immunosuppressed and non-immunosuppressed groups with respect to potential confounders. Patients were matched across exposure groups on the logit of the propensity score using greedy matching and calipers of 0.2 times the standard deviation of the logit of the propensity score, with matching being done without replacement. The variables included in the propensity score model were age, sex, comorbidities (diabetes mellitus [DM], hypertension, coronary artery disease, congestive heart failure [CHF], chronic obstructive pulmonary disease, chronic kidney disease [CKD] and cancer), need for hemodialysis, need for continuous renal replacement therapy (CRRT), ICU admission, inotrope/vasopressor use, and central venous catheter (CVC) placement.
Descriptive statistics were used to summarize patient characteristics by immunosuppressive therapy status before and after matching. Continuous variables were summarized using mean and standard deviation and categorical variables were summarized using frequency and percentage. The absolute standardized difference was used to evaluate covariate balance before and after matching, with an absolute standardized difference threshold of <0.10 indicating balance [24–26]. The bivariate association between immunosuppressive therapy use and the outcome (BSI occurrence) was assessed using McNemar test. The bivariate association between all other covariates of interest and BSI occurrence was assessed using the 2-sample t test for continuous variables and the χ 2 test for categorical variables.
Conditional logistic regression was used to assess the association between immunosuppressive therapy and BSI occurrence, as well as the association between type of immunosuppressive therapy and any positive culture due to the matched nature of the data. To assess the association between immunosuppressive therapy with positive bacterial culture, patients with fungal culture only and their matches were excluded before performing conditional logistic regression. Additionally, when assessing type of immunosuppressive therapy as an exposure, patients who received combined anakinra with tocilizumab and their matches were excluded from the analysis due to the small sample size of this group. Balance was reassessed after any exclusions were made prior to regression analysis. For multivariable analysis, backwards elimination was applied to all covariates included in the propensity score matching procedure. Backwards elimination sequentially removes variables from the model until all variables significantly contribute to the model. In some instances, the main exposure (immunosuppressive therapy use) was then added back to the model. Variance inflation factor (VIF) was calculated to check for collinearity between variables and all VIF <2, indicating no collinearity.
Laboratory test results were evaluated using descriptive statistics only in the unmatched population due to the large amount of missing data. All analyses were performed using SAS Studio version 3.8 (SAS Institute, Cary, North Carolina), and results were considered statistically significant at P < .05.
RESULTS
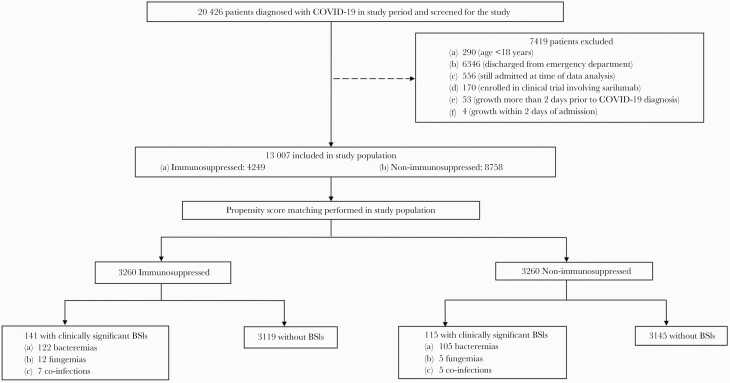
During the 166-day study period, 20 426 patients were diagnosed with COVID-19 in our health system and were evaluated for inclusion in the study. As shown in Figure 1, 13 007 patients met inclusion criteria and 7419 patients were excluded.
Figure 1.
Patient selection and allocation in the study. Abbreviations: BSI, bloodstream infection; COVID-19, coronavirus disease 2019.
Prior to propensity score matching, the immunosuppressed and non-immunosuppressed groups were imbalanced with respect to many potential confounders (variables with absolute standardized difference [ASR] >0.10) (Table 1). Propensity score matching resulted in a sample of 6520 patients (3260 patients in both the immunosuppressed and non-immunosuppressed groups), with no imbalance between the 2 groups with respect to any potential confounders (ASR <0.10) (Table 1).
Table 1.
Demographic Characteristics of Patients Before Matching and After Propensity Score Matching
| Variable | Immunosuppressed (n = 4249) No. (%) | Non-immunosuppressed (n = 8758) No. (%) | Absolute standardized difference |
|---|---|---|---|
| Original unmatched population (N = 13 007) | |||
| Age, Mean ± SD (years) | 65.68 (14.96) | 62.03 (19.30) | 0.21144 |
| Male Gender | 2648 (62.32) | 4536 (51.79) | 0.2139 |
| DM | 1240 (29.18) | 1811 (20.68) | 0.19756 |
| Hypertension | 2357 (55.47) | 2882 (32.91) | 0.24029 |
| CAD | 329 (7.74) | 511 (5.83) | 0.07592 |
| CHF | 233 (5.48) | 410 (4.68) | 0.03653 |
| COPD | 217 (5.11) | 220 (2.51) | 0.13588 |
| CKD | 296 (6.97) | 477 (5.45) | 0.06303 |
| Hemodialysis | 531 (12.50) | 438 (5.00) | 0.26766 |
| Cancer | 229 (5.39) | 318 (3.63) | 0.08481 |
| ICU admission | 1897 (44.65) | 1050 (11.99) | 0.77771 |
| CRRT | 160 (3.77) | 25 (0.29) | 0.24895 |
| Inotrope/ vasopressor use | 209 (4.92) | 155 (1.77) | 0.17582 |
| Presence of CVC | 1517 (35.70) | 542 (6.19) | 0.77828 |
| Variable | Immunosuppressed (n = 3260), No. (%) | Non-immunosuppressed (n = 3260), No. (%) | Absolute standardized difference |
| Propensity score–matched population (n = 6520) | |||
| Age, Mean ± SD (years) | 66.20 (15.59) | 66.94 (16.10) | 0.04326 |
| Male Gender | 1906 (58.47) | 2006 (61.53) | 0.06232 |
| DM | 978 (30.00) | 880 (26.99) | 0.06983 |
| Hypertension | 1457 (44.69) | 1417 (43.47) | 0.02537 |
| CAD | 258 (7.91) | 233 (7.15) | 0.03051 |
| CHF | 182 (5.58) | 164 (5.03) | 0.02514 |
| COPD | 186 (5.71) | 152 (4.66) | 0.05461 |
| CKD | 218 (6.69) | 196 (6.01) | 0.02798 |
| Hemodialysis | 273 (8.37) | 232 (7.12) | 0.04491 |
| Cancer | 180 (5.52) | 163 (5.00) | 0.02515 |
| ICU admission | 911 (27.94) | 859 (26.35) | 0.03799 |
| CRRT | 60 (1.84) | 25 (0.77) | 0.07680 |
| Inotrope/vasopressor use | 139 (4.26) | 91 (2.79) | 0.08221 |
| Presence of CVC | 540 (16.56) | 533 (16.35) | 0.00566 |
Abbreviations: CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CRRT, continuous renal replacement therapy; CVC, central venous catheter; DM, diabetes mellitus; ICU, intensive care unit; SD, standard deviation.
Among the 3260 patients who received immunosuppressive therapy in the propensity score–matched group, 90.74% received corticosteroids, 24.66% received tocilizumab, and 21.17% received anakinra (Table 2). Approximately 65% were on a single immunosuppressive therapy only, while others received combination therapies; 56.07% received corticosteroids alone, 16.96% received corticosteroids and anakinra, and 15.95% received corticosteroids and tocilizumab. The median times to administration of corticosteroids, tocilizumab, and anakinra were similar (Supplementary Table 2).
Table 2.
Immunosuppressive therapy use in propensity score matched population (n = 3260).
| Immunosuppressive Therapy Use (Alone or Combination) | No. (%) |
|---|---|
| Corticosteroids | 2958 (90.74) |
| Tocilizumab | 804 (24.66) |
| Anakinra | 690 (21.17) |
| No. of immunosuppressive therapies used | |
| Single therapy | 2125 (65.18) |
| Two therapies | 1078 (33.07) |
| Three therapies | 57 (1.75) |
| Subcategory of immunosuppressive therapies used | |
| Corticosteroid alone | 1828 (56.07) |
| Tocilizumab alone | 222 (6.81) |
| Anakinra alone | 75 (2.30) |
| Corticosteroid + Tocilizumab | 520 (15.95) |
| Corticosteroid + Anakinra | 553 (16.96) |
| Anakinra + Tocilizumab | 5 (0.15) |
| Corticosteroid + Anakinra + Tocilizumab | 57 (1.75) |
Of the total population of 13 007 patients, 486 patients (3.74%) met our definition of clinically significant BSIs. Of the propensity score–matched population of 6520 patients, 256 patients (3.93%) had clinically significant BSIs—227 patients had bacteremias, 17 fungemias, and 12 combined infections. The most common bacterial isolates were methicillin-susceptible Staphylococcus aureus (20.08%), Enterococcus faecalis (11.30%), Escherichia coli (8.37%), Klebsiella pneumoniae (7.53%), and methicillin-resistant Staphylococcus aureus (6.69%). The most common fungal isolates were Candida albicans (55.17%) and Candida glabrata (20.69%) (Supplementary Table 3).
We compared laboratory parameters between immunosuppressed and non-immunosuppressed groups and between patients with BSIs and without BSIs in the unmatched population (Supplementary Table 4). We noted several differences between the patient groups—however, due to missing data, only descriptive analyses could be performed.
On bivariate analysis (after matching across immunosuppressive therapy use), patients with BSIs were noted to be older, with a higher proportion of DM and CHF (Table 3). They had significantly higher rates of hemodialysis and CRRT, inotrope/vasopressor use, and CVC insertions. Additionally, occurrence of a BSI was associated with longer mean LOS and increased ICU admission and mortality rates.
Table 3.
Summary of Covariates by Positive Culture Status in Propensity Score Matched Population (n = 6520)
| Variable | Occurrence of BSI (n = 256), No. (%) | No BSI (n = 6264), No. (%) | P value |
|---|---|---|---|
| Immunosuppressive therapy | 141 (55.08) | 3119 (49.79) | .0878 |
| Age, Mean ± SD (years) | 69.20 (15.87) | 66.46 (15.84) | .0067 |
| Male Gender | 154 (60.16) | 3758 (59.99) | .9585 |
| DM | 96 (37.50) | 1762 (28.13) | .0011 |
| Hypertension | 117 (45.70) | 2757 (44.01) | .5935 |
| CAD | 22 (8.59) | 469 (7.49) | .5108 |
| CHF | 22 (8.59) | 324 (5.17) | .0167 |
| COPD | 16 (6.25) | 322 (5.14) | .4325 |
| CKD | 20 (7.81) | 394 (6.29) | .3275 |
| Hemodialysis | 41 (16.02) | 464 (7.41) | <.0001 |
| Cancer | 19 (7.42) | 324 (5.17) | .1141 |
| ICU admission | 179 (69.92) | 1591 (25.40) | <.0001 |
| CRRT | 25 (9.77) | 60 (0.96) | <.0001 |
| Inotrope/vasopressor use | 27 (10.55) | 203 (3.24) | <.0001 |
| Presence of CVC | 154 (60.16) | 919 (14.67) | <.0001 |
| Discharge disposition, n (%) | <.0001 | ||
| Discharged | 100 (39.06) | 4640 (74.07) | |
| Deceased | 156 (60.94) | 1624 (25.93) | |
| Length of stay (days), Mean ± SD | 23.77 (23.40) | 9.18 (9.60) | <.0001 |
Bolded P values indicate those P values that are clinically significant (P value < .05).
Abbreviations: CAD, coronary artery disease; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; CKD, chronic kidney disease; CRRT, continuous renal replacement therapy; CVC, central venous catheter; DM, diabetes mellitus; ICU, intensive care unit; SD, standard deviation.
Backwards selection was applied to all the covariates in the included in the propensity score match, with the final multivariable model including immunosuppressive therapy, age, ICU stay, CRRT, inotrope/vasopressor use, and presence of CVC. On univariable and multivariable analysis, immunosuppressive therapy was not found to be significantly associated with any BSI (Table 4).
Table 4.
Association Between any Immunosuppressive Therapy and BSIs in the Propensity Score Matched Population
| Testing of association between any immunosuppressive therapy and BSIs in entire propensity score matched population (n = 6520) | ||||
|---|---|---|---|---|
| Parameter | Univariable OR (95% CI) | P value | Multivariablea OR (95% CI) | P Value |
| Immunosuppressive therapy (yes vs no) | 1.25 (0.97, 1.62) | .0885 | 1.10 (0.83, 1.46) | .5059 |
| Testing of association between immunosuppressive category and BSIs in entire propensity score matched population (n = 6510) b | ||||
| Immunosuppressive category | Univariable OR (95% CI) | P Value | Multivariablea OR (95% CI) | P Value |
| Steroid alone vs none | 0.87 (0.60, 1.26) | .4541 | 0.78 (0.53, 1.15) | .2019 |
| Anakinra alone vs none | 1.00 (0.06, 15.99) | 1.0000 | 0.82 (0.05, 13.31) | .8877 |
| Tocilizumab alone vs none | 1.80 (0.60, 5.37) | .2920 | 1.98 (0.64, 6.10) | .2361 |
| Steroid + Anakinra vs none | 2.00 (1.05, 3.80) | .0342 | 1.54 (0.79, 3.02) | .2092 |
| Steroid + Tocilizumab vs none | 2.13 (1.16, 3.94) | .0155 | 1.97 (1.04, 3.73) | .0386 |
| Steroid + Anakinra + Tocilizumab vs none | 1.00 (0.32, 3.10) | 1.0000 | 0.81 (0.24, 2.70) | .7251 |
Bolded P values indicate those P values that are clinically significant (P value < .05).
Abbreviations: CI: confidence intervals; OR: odds ratio.
aAfter backwards selection was applied, final multivariable model was adjusted for age, ICU, CRRT, inotrope/ vasopressor use, and presence of CVC.
bThe number of study patients who received Anakinra + Tocilizumab were too small (n = 10) to perform meaningful statistical analyses and so they were excluded.
We then evaluated the association of each category of immunosuppressive therapies (alone or in combination) with BSIs in the propensity score–matched population (Table 4). On univariable analysis, the combinations of corticosteroids with anakinra (odds ratio [OR], 2.00 [95% confidence interval {CI}, 1.05–3.80]; P = .0342) and corticosteroids with tocilizumab (OR, 2.13 [95% CI, 1.16–3.94]; P = .0155) were significantly associated with BSIs. On multivariable analysis, the combined use of corticosteroids and tocilizumab was found to be significantly associated with BSIs (OR, 1.97 [95% CI, 1.04–3.73]; P = .0386). We also evaluated the association of these immunosuppressive therapies with bacterial BSIs in the propensity score–matched population (Table 5). We noted similar findings in this subgroup—on univariable analysis, the combinations of corticosteroids with anakinra (OR, 2.00 [95% CI, 1.00–4.00]; P = .0499) and corticosteroids with tocilizumab (OR, 2.07 [95% CI, 1.12–3.83]; P = .0210) were significantly associated with bacterial BSIs. On multivariable analysis, the combination of corticosteroids and tocilizumab was found to be significantly associated with bacterial BSIs (OR, 2.13 [95% CI, 1.12–4.05]; P = .0217). Due to the small number of fungal BSIs (n = 17), we did not evaluate the association of immunosuppressive therapies with these BSIs in the propensity score–matched population.
Table 5.
Association Between Immunosuppressive Category and Positive Bacterial Culture Status in Propensity Score Matched Population (n = 6476)a,b
| Immunosuppressive category | Univariable OR (95% CI) | P value | Multivariablec OR (95% CI) | P value |
|---|---|---|---|---|
| Steroid alone vs none | 0.814 (0.56, 1.19) | .2885 | 0.76 (0.51, 1.13) | .1711 |
| Anakinra alone vs none | 1.00 (0.06, 15.99) | 1.0000 | 0.79 (0.05, 12.95) | .8696 |
| Tocilizumab alone vs none | 1.80 (0.60, 5.37) | .2920 | 2.06 (0.67, 6.38) | .2093 |
| Steroid + Anakinra vs none | 2.00 (1.00, 4.00) | .0499 | 1.81 (0.88, 3.71) | .1053 |
| Steroid + Tocilizumab vs none | 2.07 (1.12, 3.83) | .0210 | 2.13 (1.12, 4.05) | .0217 |
| Steroid + Anakinra + Tocilizumab vs none | 1.00 (0.32, 3.10) | 1.0000 | 0.94 (0.29, 3.09) | .9169 |
Bolded P values indicate those P values that are clinically significant (P value < .05).
Abbreviations: CI, confidence intervals; OR, odds ratio.
aThe number of study patients who received Anakinra + Tocilizumab were too small (n = 10) to perform meaningful statistical analyses and so they were excluded from the population.
bPatients with fungal infection only and their matches (n = 34) were excluded from the population.
cAfter backwards selection was applied, final multivariable model was adjusted for age, ICU, CRRT, inotrope/ vasopressor use, and presence of CVC.
DISCUSSION
In the early months of the COVID-19 pandemic, a low incidence of bacterial coinfections was reported, whereas subsequent studies reported higher rates [21, 22, 27, 28]. Systematic reviews reported bacterial infections occurring in 5.9% to 8.1% of COVID-19 patients and correlated incidence with increasing LOS [29–31]. However, risk factors and patient characteristics contributing to the BSIs were not clearly defined; only a few studies have described the risk factors (including use of immunosuppressive therapies) associated with BSIs in hospitalized and ICU COVID-19 patients [32–35].
Similar to other studies, patients in our study were older and predominantly male [34, 36]. The overall rate of BSIs in both unmatched and propensity score–matched populations (3.74% and 3.93%, respectively) was lower than those recently reported in COVID-19 patients (5.3%–43.64%) [35, 37]. As noted in prior studies, COVID-19 patients with BSIs had significantly higher mortality rates compared to those without BSIs [34, 35]. Patients with BSIs had longer LOS (mean, 23 days), which was higher than that noted in prior studies [32, 34]. We believe the lower BSI rates in our study may be due to the larger population size and strict inclusion/exclusion criteria.
Consistent with prior observations, gram-positive organisms were the most commonly isolated pathogens in BSIs [32–35]. However, in some other studies of BSIs, gram-negative organisms have been more commonly isolated [34]. Previous studies identified gram-positive organism infections more commonly in hospitalized and ICU patients diagnosed with viral infections such as influenza, likely due to respiratory tract colonization by these bacteria [38, 39]. The presence of CVC has been reported as a significant risk factor for BSIs [32, 34]. Approximately 60% of patients with BSIs in our study had a CVC; we were unable to evaluate their potential role as a source of infection.
In univariable analyses, factors associated with BSIs in our study were similar to those seen in prior studies—older age, DM, ICU admission, use of CRRT, hemodialysis, and presence of CVC [33–35]. CKD was noted in a higher number of patients with BSIs, but this was not statistically significant (unlike what has been reported in some studies [34]).
Patients in our study received immunosuppressive therapies at similar rates of administration noted in other studies [32–34]. In the propensity score–matched population in our study, univariable analysis demonstrated significant association of combination corticosteroid-anakinra and combination of corticosteroid-tocilizumab with occurrence of any BSI and bacterial BSIs. On multivariable analysis, this association remained significant for the combination of corticosteroids and tocilizumab. Our findings are in concurrence with other studies that reported increased risk of BSIs in association with immunosuppressive therapy on multivariable analyses [33, 34]. In one multicenter study, patients with BSIs had higher corticosteroid use, longer LOS, and higher ICU admission and in-hospital mortality rates, though the multivariable logistic regression analysis only adjusted for age, sex, and race [34]. In our study, the multivariable model included immunosuppressive therapy, age, ICU stay, CRRT, inotrope/vasopressor use, and presence of CVC.
Prior studies that evaluated the role of corticosteroids in COVID-19 did not elaborate on the risk of secondary infections [9, 10]. With tocilizumab, some studies reported increased risk of infections [12, 15], while others noted either no differences in infection rates [11], or lower infection rates [13, 14, 16, 17]. However, these studies had heterogeneous study populations and interventions and were not primarily investigating the incidence of secondary infections, therefore limiting a direct comparison with our study.
Preliminary results of a recent multicenter study reported improved survival in COVID-19 patients who received tocilizumab or sarilumab in combination with other therapies [40]. Based on this, the National Health Service (England) and National Institutes of Health have recommended that clinicians consider use of tocilizumab or sarilumab for select COVID-19 patients [41, 42]. However, given the significantly increased risk of BSIs with combination corticosteroids and tocilizumab in our study, we believe that this risk should be weighed with any potential benefits of these therapies. We also advocate for enhanced surveillance for BSIs in COVID-19 patients receiving combination immunosuppressive therapies.
As a retrospective study, there were some inherent limitations. Though we attempted to control for the severity of infection through propensity score matching for potential confounding factors, followed by univariable and multivariable analyses and conditional logistic regression in the matched population, only a prospective randomized control study would be able to account for all the possible confounding factors. It was not feasible to perform a time-dependent analysis of some factors (such as ICU admission and CVC insertion) in our patient population, as in some cases the exact timing of some of these characteristics could not be determined accurately. The evolving standards of care during the pandemic likely impacted the adoption and use of immunosuppressive therapies. For example, use of dexamethasone likely increased after release of the RECOVERY trial results, and we could not control for the heterogeneity of these therapies [10]. We were unable to analyze the timing or potential sources of BSIs in our study. Furthermore, we could not determine the durations of mechanical ventilation and/or central line insertion accurately enough to analyze them in our model. Last, as laboratory data points were missing for some of our patients in the propensity score–matched population, we could not perform meaningful statistical analyses on some of these data.
The large ICU and non-ICU COVID-19 patient population in our study enabled us to evaluate multiple risk factors, perform propensity score matching to create uniform patient populations, and test the association of the multiple factors with BSI incidence, while adjusting for multiple confounding variables. In prior studies, the authors noted that either their study design did not enable detection of differences in infection rates secondary to immunosuppressive therapy, or they did not have sufficient patients with BSIs to perform multivariable analyses [32, 35]. Canadian researchers are maintaining a living meta-analysis of bacterial coinfections and super-infections (https://www.tarrn.org/covid), incorporating multiple variables in the meta-regression [29]. At the time of submission for publication of this paper, use of immunosuppressive therapies had not been included as a variable.
CONCLUSIONS
Our study adds to the growing literature describing the risk factors associated with secondary bacterial and fungal infections in COVID-19 patients. Our study found that the combined use of corticosteroids and tocilizumab in COVID-19 patients was associated with the occurrence of bloodstream infections in a balanced population, independent of other contributory risk factors. Further prospective multicenter studies are needed to establish the pathophysiologic role of immunosuppressive therapy in development of infections (including BSIs) in COVID-19 patients. We recommend enhanced surveillance for bloodstream infections (including close clinical monitoring and maintaining a low threshold for blood culture testing in the compatible clinical setting) in COVID-19 patients receiving immunosuppressive therapies.
Supplementary Data
Supplementary materials are available at Open Forum Infectious Diseases online. Consisting of data provided by the authors to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the authors, so questions or comments should be addressed to the corresponding author.
Notes
Acknowledgments. We gratefully acknowledge the invaluable assistance of Angelie Oberoi, Michael Kalina, Abraham Saraya, and their entire team in acquisition of electronic patient medical data. Preliminary data from this study have been accepted as an abstract at the European Congress of Clinical Microbiology and Infectious Diseases (ECCMID) meeting.
Author contributions. A. Kh., P. M., A.Ki., M. O., D. H., M. E., and B. F. conceived the study design. A. Kh., P. M., S. I., and M. O. were involved in initial data collection and data analysis. A. Kh., P. M., A. Ki., M. O., P. G.-G., T. C., T. D., I. B., N. N., S. F., D. H., M. E., and B. F. were involved in review of patient charts to evaluate for commensal organisms. A. Kh., P. M., and S. I. interpreted the results and created the initial draft of the manuscript. All authors were involved in critical appraisal and review of the manuscript. All authors approved the final manuscript.
Patient consent statement. The procedures followed were in accordance with the ethical standards of the Helsinki Declaration (1964, amended in 2008) of the World Medical Association. The Northwell Health institutional review board reviewed and approved the study design. Given the retrospective nature, the study was deemed as minimal-risk research and thus the requirement for informed consent was waived.
Financial support. This work was supported by the Jane and Dayton Brown Research Fund.
Potential conflicts of interest. In the last 12 months, P. M. and M. E. were site investigators for an industry-sponsored COVID-19 clinical trial. All other authors report no potential conflicts.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1. Liu YC, Kuo RL, Shih SR. COVID-19: the first documented coronavirus pandemic in history. Biomed J 2020; 43:328–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. World Health Organization. Coronavirus (COVID-19) dashboard. Available at: https://covid19.who.int/?gclid=CjwKCAjwj6SEBhAOEiwAvFRuKADGthEkWNYRBR5XCNqRkqdXYYoM36syqie0qW39KriGjPsMOLbG-RoCHRgQAvD_BwE. Accessed 28 April 2021.
- 3. National Institutes of Health. COVID-19 treatment guidelines—therapeutic options for COVID-19 currently under investigation. Available at: https://covid19treatmentguidelines.nih.gov/therapeutic-options-under-investigation/. Accessed 24 April 2021.
- 4. Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 2020; 395:507–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020; 395:497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Mehta P, McAuley DF, Brown M, et al. ; HLH Across Speciality Collaboration, UK. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet 2020; 395:1033–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382:1708–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Zhang G, Hu C, Luo L, et al. Clinical features and short-term outcomes of 221 patients with COVID-19 in Wuhan, China. J Clin Virol 2020; 127:104364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Sterne JAC, Murthy S, Diaz JV, et al. ; WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group. Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19: a meta-analysis. JAMA 2020; 324:1330–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med 2021; 384:693–704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Malgie J, Schoones JW, Pijls BG. Decreased mortality in COVID-19 patients treated with tocilizumab: a rapid systematic review and meta-analysis of observational studies. Clin Infect Dis 2021; 72:e742–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Gupta S, Wang W, Hayek SS, et al. ; STOP-COVID Investigators. Association between early treatment with tocilizumab and mortality among critically ill patients with COVID-19. JAMA Intern Med 2021; 181:41–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Salama C, Han J, Yau L, et al. Tocilizumab in patients hospitalized with Covid-19 pneumonia. N Engl J Med 2021; 384:20–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Hermine O, Mariette X, Tharaux PL, et al. ; CORIMUNO-19 Collaborative Group. Effect of tocilizumab vs usual care in adults hospitalized with COVID-19 and moderate or severe pneumonia: a randomized clinical trial. JAMA Intern Med 2021; 181:32–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Somers EC, Eschenauer GA, Troost JP, et al. Tocilizumab for treatment of mechanically ventilated patients with COVID-19 [manuscript published online ahead of print 11 July 2020]. Clin Infect Dis 2020. doi:10.1093/cid/ciaa954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Salvarani C, Dolci G, Massari M, et al. ; RCT-TCZ-COVID-19 Study Group. Effect of tocilizumab vs standard care on clinical worsening in patients hospitalized with COVID-19 pneumonia: a randomized clinical trial. JAMA Intern Med 2021; 181:24–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Stone JH, Frigault MJ, Serling-Boyd NJ, et al. ; BACC Bay Tocilizumab Trial Investigators. Efficacy of tocilizumab in patients hospitalized with Covid-19. N Engl J Med 2020; 383:2333–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Huet T, Beaussier H, Voisin O, et al. Anakinra for severe forms of COVID-19: a cohort study. Lancet Rheumatol 2020; 2:e393–400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med 2020; 8:475–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Winthrop KL, Mariette X, Silva JT, et al. ESCMID study group for infections in compromised hosts (ESGICH) consensus document on the safety of targeted and biological therapies: an infectious diseases perspective (soluble immune effector molecules [II]: agents targeting interleukins, immunoglobulins and complement factors). Clin Microbiol Infect 2018; 24(Suppl 2):21–40. [DOI] [PubMed] [Google Scholar]
- 21. Zhan M, Qin Y, Xue X, Zhu S. Death from Covid-19 of 23 health care workers in China. N Engl J Med 2020; 382:2267–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Goyal P, Choi JJ, Pinheiro LC, et al. Clinical characteristics of Covid-19 in New York City. N Engl J Med 2020; 382:2372–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Centers for Disease Control and Prevention, National Healthcare and Safety Network. Patient safety component manual. Available at: https://www.cdc.gov/nhsn/pdfs/pscmanual/pcsmanual_current.pdf. Accessed 19 January 2021.
- 24. Normand ST, Landrum MB, Guadagnoli E, et al. Validating recommendations for coronary angiography following acute myocardial infarction in the elderly: a matched analysis using propensity scores. J Clin Epidemiol 2001; 54:387–98. [DOI] [PubMed] [Google Scholar]
- 25. Mamdani M, Sykora K, Li P, et al. Reader’s guide to critical appraisal of cohort studies: 2. Assessing potential for confounding. BMJ 2005; 330:960–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Austin PC. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat Med 2009; 28:3083–107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Sepulveda J, Westblade LF, Whittier S, et al. Bacteremia and blood culture utilization during COVID-19 surge in New York City. J Clin Microbiol 2020; 58:e00875-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Rawson TM, Moore LSP, Zhu N, et al. Bacterial and fungal coinfection in individuals with coronavirus: a rapid review to support COVID-19 antimicrobial prescribing. Clin Infect Dis 2020; 71:2459–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Langford BJ, So M, Raybardhan S, et al. Bacterial co-infection and secondary infection in patients with COVID-19: a living rapid review and meta-analysis. Clin Microbiol Infect 2020; 26:1622–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lansbury L, Lim B, Baskaran V, Lim WS. Co-infections in people with COVID-19: a systematic review and meta-analysis. J Infect 2020; 81:266–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Hughes S, Troise O, Donaldson H, et al. Bacterial and fungal coinfection among hospitalized patients with COVID-19: a retrospective cohort study in a UK secondary-care setting. Clin Microbiol Infect 2020; 26:1395–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Nori P, Cowman K, Chen V, et al. Bacterial and fungal coinfections in COVID-19 patients hospitalized during the New York City pandemic surge. Infect Control Hosp Epidemiol 2021; 42:84–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Giacobbe DR, Battaglini D, Ball L, et al. Bloodstream infections in critically ill patients with COVID-19. Eur J Clin Invest 2020; 50:e13319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Bhatt PJ, Shiau S, Brunetti L, et al. Risk factors and outcomes of hospitalized patients with severe COVID-19 and secondary bloodstream infections: a multicenter, case-control study. Clin Infect Dis 2021; 72:e995–1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Engsbro AL, Israelsen SB, Pedersen M, et al. Predominance of hospital-acquired bloodstream infection in patients with Covid-19 pneumonia. Infect Dis (Lond) 2020; 52:919–22. [DOI] [PubMed] [Google Scholar]
- 36. Goncalves Mendes Neto A, Lo KB, Wattoo A, et al. Bacterial infections and patterns of antibiotic use in patients with COVID-19. J Med Virol 2021; 93:0489–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Ceccarelli G, Alessandri F, Oliva A, et al. Superinfections in patients treated with teicoplanin as anti-SARS-CoV-2 agent. Eur J Clin Invest 2021; 51:e13418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Chertow DS, Memoli MJ. Bacterial coinfection in influenza: a grand rounds review. JAMA 2013; 309:275–82. [DOI] [PubMed] [Google Scholar]
- 39. Klein EY, Monteforte B, Gupta A, et al. The frequency of influenza and bacterial coinfection: a systematic review and meta-analysis. Influenza Other Respir Viruses 2016; 10:394–403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Gordon AC, Mouncey PR, Al-Beidh F, et al. Interleukin-6 receptor antagonists in critically ill patients with Covid-19. N Engl J Med 2021; 384:1491–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. National Health Service. Interim position statement: interleukin-6 inhibitors (tocilizumab or sarilumab) for patients admitted to ICU with COVID-19 pneumonia (adults). Available at: https://www.england.nhs.uk/coronavirus/wp-content/uploads/sites/52/2020/11/IL6_Inhibitors_Position_Statement-1.pdf. Accessed 19 January 2021.
- 42. National Institutes of Health. COVID-19 treatment guidelines. Available at: https://www.covid19treatmentguidelines.nih.gov/immunomodulators/interleukin-6-inhibitors/. Accessed 26 April 2021. [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.