Abstract
Background
Family history (FH) is one of important risk factors for cardiovascular disease (CVD). However, little is known about its impact on dyslipidemia prevalence and management status. Thus, we aimed to investigate the impact of FH of CVD on dyslipidemia prevalence, awareness, treatment, control, and healthy behaviors in Korean adults.
Methods
We conducted a cross-sectional study using representative data from the Korea National Health and Nutrition Examination Survey (KNHANES) 2014–2018. A total of 22,024 participants aged ≥ 19 years without histories of CVDs were classified into two groups according to the presence of FH of CVD (with FH, n = 3,778; without FH, n = 18,246). FH of CVD was defined as having a first-degree relative with ischemic heart disease or stroke. Multivariate logistic regression analyses were performed to evaluate the association between FH of CVD and dyslipidemia prevalence, awareness, treatment, control, and healthy behaviors (weight control, non-smoking, non-risky drinking, sufficient physical activity, and undergoing health screening).
Results
FH of CVD was significantly associated with a higher dyslipidemia prevalence (adjusted odds ratio [aOR] 1.34, 95% confidence interval [CI] 1.18–1.51), better awareness (aOR 1.54, 95%CI 1.19–2.00), and treatment rates (aOR 1.34, 95%CI 1.12–1.60), but not control. Having an FH of CVD was not predictive of any healthy behaviors in dyslipidemia patients. For non-dyslipidemia patients, FH of CVD even showed significant association with smoking (aOR 1.18, 95%CI 1.02–1.36), and risky drinking (aOR 1.20, 95%CI 1.03–1.40) while it was predictive of receiving health screening (aOR 1.14, 95% CI 1.02–1.27).
Conclusions
Having an FH of CVD might positively trigger dyslipidemia patients to start pharmacological intervention, but not non-pharmacological interventions. Therefore, physicians should make more efforts to educate and promote the importance of non-pharmacological behavioral modification in dyslipidemia patients with an FH of CVD.
Introduction
Globally, cardiovascular disease (CVD) is the leading cause of mortality [1,2] and management of dyslipidemia is an essential factor for the primary prevention of CVD [3,4]. In addition, family history (FH) is known to be an independent risk factor for CVDs, such as ischemic heart disease and stroke [5–8]. Thus, those with an FH of CVD would need extra attention when managing dyslipidemia for the primary prevention of CVD. In fact, various guidelines on the management of dyslipidemia including 2018 American College of Cardiology/American Heart Association (ACC/AHA) guidelines, 2019 European Society of Cardiology/European Atherosclerosis Society (ESC/EAS) guidelines and the Korean Society of Lipid and Atherosclerosis (KSoLA)’s 2018 guideline, the FH of premature coronary heart disease (CHD) is one of the major risk factors of CVD that modify treatment goals in patients with dyslipidemia [9–11]. Furthermore, all three guidelines mentioned above and 2019 ACC/AHA guidelines on the primary prevention of CVD emphasize not only pharmacological therapy for dyslipidemia, but also non-pharmacological behavioral modifications (e.g., weight control, increasing physical activity, smoking cessation, etc.) for the control of dyslipidemia and the primary prevention of CVD [3,9,10]. There have been a number of previous studies conducted in various countries regarding the prevalence, awareness, treatment, and control of dyslipidemia [12–15]. However, studies that investigated the rates of dyslipidemia in patients with an FH of CVD are relatively rare. Furthermore, the results of studies that examined the healthy behaviors of patients with an FH of CVD compared to those without an FH have been inconsistent. Some studies reported that an FH of CVD did not predict healthy behaviors [16–18] whereas other studies did [19,20]. In addition, few studies have analyzed the association between healthy behaviors and FH of CVD among patients with dyslipidemia. Thus, using nationally representative sample data from the Korea National Health and Nutrition Survey (KNHANES), the purpose of this study was to investigate the impact of an FH of CVD on the prevalence, awareness, treatment, control of dyslipidemia, and the practice of healthy behaviors for the primary prevention of CVD.
Methods
Study population
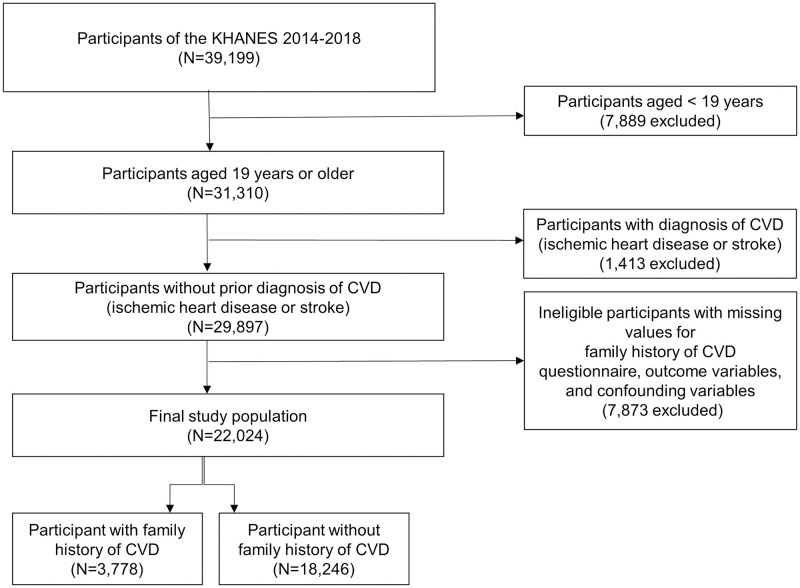
KNHANES is a cross-sectional nationwide survey, representing the non-institutionalized civilian population of Korea, that has been conducted on an annual basis since 1998 by the Korea Centers for Disease Control and Prevention (KCDC). The KNHANES is composed of three parts: a health interview survey, health examination, and a nutrition survey. The selection of participants for KNHANES are performed using a complex, multi-stage, and probability sampling to gather unbiased nationally representative data. The details and representativeness of the KHNANES have been discussed in previous studies [21,22]. This study analyzed data from the KNHANES between 2014 and 2018. Among the 39,199 participants from the KNHANES 2014–2018, those who were younger than 19 years (N = 7,889) were excluded in order to include only the adults. Furthermore, to determine the association between an FH of CVD and prevalence, awareness, treatment, and control status of dyslipidemia, and the practice of healthy behaviors for primary prevention of CVD, those with a prior diagnostic history of CVD (N = 1,413) were excluded. Diagnosis of CVD was defined as a self-report of a prior diagnosis of ischemic heart disease (myocardial infarction or angina) or stroke that was confirmed by a doctor. Ineligible participants with missing values for an FH of CVD, outcome variables, and confounders (N = 7,873) were also excluded, leading to a final study population for analysis of 22,024. We then divided the study participants into two groups according to the presence of an FH of CVD. Participants with an FH of CVD were defined as participants who responded positively to the questions from the health interview survey of KNHANES that were probing if any of their first-degree relatives (parents or siblings) were ever diagnosed with ischemic heart disease (myocardial infarction or angina) or stroke. Subsequently, 3,778 participants with and 18,246 without an FH of CVD were included in the final analysis (Fig 1). This study was approved by the institutional review board (IRB) of the Seoul St. Mary’s Hospital, Catholic University of Korea (IRB approval number: KC20ZASI0909). The requirement for written informed consent was waived.
Fig 1. Flow chart of selection process of study population.
KNHANES, Korea National Health and Nutrition Examination Survey; CVD, cardiovascular disease.
Definitions of variables
Using the risk stratification suggested by 2018 KSoLA guidelines for the management of dyslipidemia, the prevalence of dyslipidemia was defined as the proportion of participants with low-density lipoprotein cholesterol (LDL-C) higher than each individual’s desirable levels according to their categorized risk groups (LDL-C ≥160 mg/dL for the low risk group, ≥130 mg/dL for the moderate risk group, and ≥100 mg/dL for the high risk group) [10], a previous diagnosis of dyslipidemia by a physician or those who were currently treated with lipid lowering agents. Awareness of dyslipidemia was defined as a positive response to prior diagnosis of dyslipidemia by a physician among those with dyslipidemia. Treatment of dyslipidemia was defined as those taking lipid-lowering agents among participants with dyslipidemia. Control of dyslipidemia was defined as the proportion of participants who achieved LDL-C goals for each risk group among those treated for dyslipidemia. Both the risk stratification and LDL-C goals for the treatment of dyslipidemia for each risk group suggested by 2018 KSoLA guidelines were generally based on the National Cholesterol Education Program-Adult Treatment Panel III [10,23]. The prevalence of hypertension was defined as the proportion of participants with systolic blood pressure ≥140 mmHg or diastolic blood pressure ≥90 mmHg, a previous diagnosis of hypertension by a physician or those who were currently treated with antihypertensive agent. Prevalence of diabetes mellitus (DM) was defined as the proportion of patients with fasting blood glucose (FBS) ≥126 mg/dl or glycated hemoglobin (HbA1c) ≥6.5%, a previous diagnosis of DM by a physician or those who were currently treated with oral hypoglycemic agent (OHA) or insulin. Overweight and obese participants were defined using body mass index (BMI) values according to the Korean obesity standards (23–24.9 kg/m2 for overweight and ≥25.0 kg/m2 for obese) [24]. Variables regarding healthy behaviors were derived from the results of a health behavior survey, which is a subcategory of the health interview. The overweight and obese participants who responded positively to making efforts to lose weight were considered as the “weight control” group. As for the smoking status, participants were categorized into either “non-smoker” or “current smoker” groups, with the non-smokers consisting of never-smokers and former-smokers. Risky drinkers were defined as those who drank twice or more a week with an average of 7 or more standard drinks per occasion for men or 5 or more standard drinks per occasion for women. Non-drinkers and persons who drink less alcohol than risky drinkers were considered non-risky drinkers. The degree of physical activity was defined based on the Physical Activity Guidelines for Americans, 2nd edition (PAG) [25]. The PAG states that adults are required to have at least 150 minutes of moderate-intensity aerobic physical activity or at least 75 minutes of vigorous-intensity aerobic physical activity per week or its equivalent combination of moderate and vigorous-intensity activity [25]. Thus, participants who fulfilled the criteria were classified into the “sufficient physical activity” group. To evaluate if participants were actively receiving health screening tests regularly for the prevention of CVD, we also examined the proportion of participants who responded positively to a survey question that asked whether they underwent health screening within the last 2 years.
Anthropometric and laboratory measurements
Each participant underwent blood work during the survey after fasting for at least 8 hours (12 hours for triglyceride (TG)). All obtained blood samples were immediately processed, refrigerated, and delivered in cold storage to the central laboratory. The transported samples were all analyzed within 24 h. Total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), triglycerides (TG), and FBS were evaluated with a Hitachi Autonomic Analyzer 7600–210 (Hitachi, Japan), while HbA1c was measured using a Tosoh G8 (Tosoh, Japan). The LDL-C level was obtained using the Friedewald formula (LDL-C = TC—HDL-C +TG/5) when the TG level was <200 mg/dL, while it was directly measured when the TG level was ≥200 mg/dL [14,26]. We also calculated the LDL-C using the Friedewald formula for participants with values of TC, TG, and HDL-C, but had missing LDL-C values. Blood pressure (BP) was obtained by applying the standard protocol according to the AHA’s determination of BP by sphygmomanometry [27]. It was measured 3 times on the right arm of the participant in a sitting position using a mercury sphygmomanometer (Baumanometer; WA Baum Co Inc., Coptague, NY, USA) after resting for at least 5 min. The final BP was determined by averaging the second and third BP readings [14].
Confounders
Confounding variables reflected in the present study were age, sex, marital status, educational status, employment status, household income, residential area, type of health insurance, level of self-rated health, prevalence of hypertension, prevalence of DM, and BMI. The definitions of the prevalence of hypertension and DM and calculation of BMI are mentioned above. Other confounders were derived from the self-administered questionnaire of the health interview survey of KNHANES.
Statistical analysis
Sampling weights were applied to all statistical analyses, as designated by the KCDC to reflect the entire Korean general population without bias [21,22]. The sampling weights for each sample participant were constructed based on three factors: the inverse of selection probabilities (primary sampling unit; household); an adjustment for nonresponse (household, person); and a post-stratification to make the calculated survey estimates approximately equivalent to the total Korean population [21]. The characteristics and outcome variables of patients with and without an FH of CVD were compared by means and standard deviations or percentages and standard errors. We performed adjusted Wald test for means and chi-square test for proportions to evaluate the statistical differences of the variables between those with and without an FH of CVD. Moreover, multivariate logistic regression analysis was performed to assess the association between the presence of an FH of CVD and prevalence, awareness, treatment, control of dyslipidemia, and healthy behaviors, with adjustments being made for all of the confounding factors mentioned above. For the outcomes regarding healthy behaviors, subgroup analyses were additionally performed, according to the morbidity status of dyslipidemia. F-adjusted mean residual tests were performed to assess goodness of fit for each multivariate logistic regression model. All analyses were conducted using STATA 14.1 (Stata Corp., College Station, TX, USA). P-values <0.05 were regarded as statistically significant.
Results
Baseline characteristics
The baseline characteristics of all study participants, those with and without FH of CVD are summarized in Table 1.
Table 1. Baseline characteristics of participants according to the family history of cardiovascular diseasea.
| Characteristics | All (n = 22,024) % (SE) or mean±SD | No family history (n = 18,246) % (SE) or mean±SD | Family history (n = 3,778) % (SE) or mean±SD | P value |
|---|---|---|---|---|
| Socio-demographic factors | ||||
| Sex | 0.001 | |||
| Male | 49.5 (0.3) | 50.0 (0.4) | 46.4 (1.0) | |
| Female | 50.5 (0.3) | 50.0 (0.4) | 53.6 (1.0) | |
| Age (years) | 44.7±17.8 | 43.6±18.9 | 50.4±27.2 | <0.0001 |
| Marital status | <0.0001 | |||
| Married | 66.3 (0.5) | 35.9 (0.6) | 21.8 (0.9) | |
| Single/divorced/separated/widowed | 33.7 (0.5) | 64.1 (0.6) | 78.2 (0.9) | |
| Educational status | <0.0001 | |||
| Middle school or lower | 17.4 (0.4) | 16.7 (0.4) | 21.2 (0.8) | |
| High school | 28.0 (0.5) | 27.0 (0.5) | 33.1 (0.9) | |
| College or higher | 54.6 (0.6) | 56.3 (0.7) | 45.7 (1.1) | |
| Employment status | 0.238 | |||
| Manual | 22.4 (0.5) | 22.2 (0.5) | 23.6 (0.9) | |
| Non-manual | 44.3 (0.5) | 44.5 (0.5) | 43.4 (1.0) | |
| Others (students or housewives) | 33.3 (0.4) | 33.3 (0.4) | 33.0 (0.9) | |
| Income | 0.267 | |||
| Low | 12.4 (0.4) | 12.5 (0.4) | 12.0 (0.7) | |
| Lower middle | 23.2 (0.5) | 23.1 (0.5) | 23.7 (0.9) | |
| Upper middle | 31.2 (0.6) | 31.5 (0.6) | 30.0 (1.0) | |
| High | 33.1 (0.7) | 32.9 (0.7) | 34.3 (1.1) | |
| Residential area | 0.565 | |||
| Urban | 64.6 (0.9) | 64.7 (1.0) | 64.1 (1.2) | |
| Rural | 35.4 (0.9) | 35.3 (1.0) | 35.9 (1.2) | |
| Health insurance | 0.915 | |||
| Medicare | 97.0 (0.2) | 97.0 (0.2) | 97.0 (0.4) | |
| Medical aid | 3.0 (0.2) | 3.0 (0.2) | 3.0 (0.4) | |
| Health status | ||||
| Body mass index (kg/m2) | 23.8±3.1 | 23.7±3.4 | 24.1±6.5 | <0.0001 |
| Overweight or obesity | 55.7 (0.4) | 55.0 (0.5) | 59.5 (0.9) | <0.0001 |
| Self-rated health | <0.0001 | |||
| Very poor/poor | 15.3 (0.3) | 14.6 (0.3) | 19.1 (0.7) | |
| Fair | 52.4 (0.4) | 52.2 (0.4) | 53.2 (1.0) | |
| Good/Excellent | 32.3 (0.4) | 33.2 (0.4) | 27.7 (0.9) | |
| Prevalence of chronic diseases | ||||
| Hypertension | 22.8 (0.4) | 20.1 (0.4) | 32.5 (0.9) | <0.0001 |
| Diabetes mellitus | 9.0 (0.2) | 8.6 (0.2) | 11.3 (0.6) | <0.0001 |
| Dyslipidemia | 15.5 (0.3) | 14.1 (0.3) | 23.1 (0.7) | <0.0001 |
| Healthy behaviors | ||||
| Weight controlb | 70.8 (0.5) | 70.1 (0.5) | 70.2 (1.2) | 0.585 |
| No smoking | 78.2 (0.4) | 78.0 (0.4) | 79.3 (0.9) | 0.162 |
| Non-risky drinking | 86.8 (0.3) | 86.8 (0.3) | 86.3 (0.7) | 0.485 |
| Sufficient physical activity | 50.1 (0.5) | 51.4 (0.5) | 48.4 (1.0) | 0.003 |
| Health screening within the last 2 years | 64.6 (0.4) | 63.2 (0.5) | 72.1 (0.9) | <0.0001 |
All data were weighted to the standard Korean population.
aAt least one first-degree relative with ischemic heart disease or stroke.
bThe analysis included only those who are overweight or obese (n = 12,373).
P values were obtained by adjusted Wald test for means or chi-square test for proportions.
Abbreviation: SE, standard error; SD, standard deviation.
Association between FH of CVD and prevalence, awareness, treatment, and control rates of dyslipidemia
The awareness and treatment rates of dyslipidemia were 76.9% and 46.7% respectively. In addition, the control rate among those treated for dyslipidemia was 23.4%. Dyslipidemia patients with an FH of CVD had significantly higher awareness (84.1% vs. 74.7%) and treatment (54.8% vs. 44.2%) rates than those without an FH of CVD. In terms of control of dyslipidemia, the FH of CVD group had significantly lower rate than the non-FH group (19.6% vs 24.8%) (Table 2). The results of the multivariate logistic regression analysis for the prevalence, awareness, treatment, and control of dyslipidemia between those with and without an FH of CVD are also shown in Table 2. The presence of an FH of CVD was significantly associated with an increased risk of dyslipidemia prevalence (adjusted odds ratio [aOR] 1.34, 95% confidence interval [CI] 1.18–1.51), awareness (aOR 1.54, 95% CI 1.19–2.00), and treatment (aOR 1.34, 95% CI 1.12–1.60). However, an FH of CVD was not significantly associated with the control of dyslipidemia. All multivariate logistic regression models showed good fit (F-adjusted mean residual tests, P values: <0.0001).
Table 2. Association between family history of cardiovascular diseasea and the prevalence, awareness, treatment and control of dyslipidemia.
| Variables | All (n = 22,024) | No family history (n = 18,246) | Family history (n = 3,778) | P value |
|---|---|---|---|---|
| All (n = 22,024) | ||||
| Dyslipidemia prevalence | ||||
| Proportion [% (SE)] | 15.5 (0.3) | 14.1 (0.3) | 23.1 (0.7) | <0.0001 |
| Adjusted ORb (95% CI) | 1 (reference) | 1.34 (1.18–1.51) | <0.0001 | |
| Participants with dyslipidemia (n = 4,153) | ||||
| Awareness | ||||
| Proportion [% (SE)] | 76.9 (0.8) | 74.7 (0.9) | 84.1 (1.3) | <0.0001 |
| Adjusted ORb (95% CI) | 1 (reference) | 1.54 (1.19–2.00) | 0.001 | |
| Treatment | ||||
| Proportion [% (SE)] | 46.7 (0.9) | 44.2 (1.0) | 54.8 (1.8) | <0.0001 |
| Adjusted ORb (95% CI) | 1 (reference) | 1.34 (1.12–1.60) | 0.001 | |
| Participants treated for dyslipidemia (n = 2,087) | ||||
| Control | ||||
| Proportion [% (SE)] | 23.4 (1.1) | 24.8 (1.3) | 19.6 (1.9) | 0.030 |
| Adjusted ORb (95% CI) | 1 (reference) | 0.76 (0.49–1.17) | 0.214 |
All data were weighted to the standard Korean population.
aAt least one first-degree relative with ischemic heart disease or stroke.
bAdjusted for age, sex, marital status, educational status, employment status, income, residential area, type of health insurance, body mass index, level of self-related health, prevalence of hypertension, and prevalence of diabetes mellitus.
P values were obtained by chi-square test for proportions.
Adjusted odds ratios and their corresponding P values were obtained by multivariate logistic regression analyses.
Abbreviation: SE, standard error; OR, odds ratio; CI, confidence interval.
Association between FH of CVD and healthy behaviors
According to the multivariate analysis for FH of CVD and the healthy behaviors of all the participants, undergoing health screening within the last 2 years (aOR 1.16, 95% CI 1.05–1.28) was significantly associated with the presence of an FH of CVD. However, having an FH of CVD was rather predictive of smoking (aOR 1.14, 95% CI 1.01–1.30) and risky drinking (aOR 1.15, 95% CI 1.00–1.32) while no significant correlation was found between an FH of CVD and other healthy behaviors. This trend was consistently found in the subgroup analysis conducted with participants without dyslipidemia (aOR 1.14, 95% CI 1.02–1.27 for undergoing health screening; aOR 1.18, 95% CI 1.02–1.36 for smoking; and aOR 1.20, 95% CI 1.03–1.40 for risky drinking). In terms of those with dyslipidemia, none of the healthy behaviors significantly correlated with FH of CVD. All multivariate logistic regression models showed good fit (F-adjusted mean residual tests, P values: <0.0001) (Table 3).
Table 3. Association between family history of cardiovascular diseasea and healthy behaviors according to subgroups of dyslipidemia prevalence.
| Variables | No family history (reference) | Family history adjusted ORb (95% CI) | P value |
|---|---|---|---|
| All (n = 22,024) | |||
| Weight controlc | 1 (reference) | 1.02 (0.90–1.16) | 0.776 |
| Smoking | 1 (reference) | 1.14 (1.01–1.30) | 0.038 |
| Risky drinking | 1 (reference) | 1.15 (1.00–1.32) | 0.046 |
| Sufficient physical activity | 1 (reference) | 1.04 (0.96–1.13) | 0.323 |
| Health screening within the last 2 years | 1 (reference) | 1.16 (1.05–1.28) | 0.003 |
| Participants with dyslipidemia (n = 4153) | |||
| Weight controld | 1 (reference) | 1.13 (0.90–1.41) | 0.293 |
| Smoking | 1 (reference) | 0.92 (0.69–1.21) | 0.542 |
| Risky drinking | 1 (reference) | 0.96 (0.71–1.29) | 0.768 |
| Sufficient physical activity | 1 (reference) | 1.15 (0.96–1.38) | 0.118 |
| Health screening within the last 2 years | 1 (reference) | 1.11 (0.90–1.38) | 0.332 |
| Participants without dyslipidemia (n = 17,871) | |||
| Weight controle | 1 (reference) | 0.97 (0.83–1.13) | 0.674 |
| Smoking | 1 (reference) | 1.18 (1.02–1.36) | 0.022 |
| Risky drinking | 1 (reference) | 1.20 (1.03–1.40) | 0.018 |
| Sufficient physical activity | 1 (reference) | 1.01 (0.92–1.11) | 0.849 |
| Health screening within the last 2 years | 1 (reference) | 1.14 (1.02–1.27) | 0.021 |
All data were weighted to the standard Korean population.
aAt least one first-degree relative with ischemic heart disease or stroke.
bAdjusted for age, sex, marital status, education status, employment status, income, residential area, type of health insurance, body mass index, level of self-related health, prevalence of hypertension and prevalence of diabetes mellitus.
cThe analysis included only those who are overweight or obese (n = 12,373).
dThe analysis included only those who are overweight or obese (n = 3,005).
eThe analysis included only those who are overweight or obese (n = 9,368).
Analyses were performed by multivariate logistic regression model.
Abbreviations: OR, odds ratio; CI, confidence interval.
Discussion
In this nationwide cross-sectional study, individuals with an FH of CVD had a significantly higher prevalence of dyslipidemia compared to those without an FH. This suggests that individuals with an FH of CVD are at a higher risk of developing dyslipidemia. In fact, these results are expected because both the FH of CVD and dyslipidemia are well-known risk factors for CVD occurrence [5,28,29]. The results of previous studies were also consistent with ours, as the FH of CVD group displayed significantly higher dyslipidemia prevalence [12,15,30].
In terms of the management of dyslipidemia, our results revealed that the awareness and treatment rates were also significantly higher in the FH of CVD group than in the non-FH group. Furthermore, a positive FH of CVD was independently associated with a better awareness and treatment for dyslipidemia. This tendency could be explained by the Health Belief Model [31,32] as those who believe they are more vulnerable to developing a certain disease are likely be more alert to their health status and make more efforts to reduce the threat of that disease. Thus, patients with an FH of CVD would have likely recognized that their risk for CVD was higher than those without, which may have led to better awareness and higher treatment rates. However, despite these results, control rate of dyslipidemia was found to be significantly lower in FH of CVD group than the non-FH group and having an FH of CVD was not significantly associated with control of dyslipidemia in the multivariate analysis. This may imply that when patients’ lipid levels worsen and reach a point where they would require medical treatment, patients without an FH of CVD would also become conscious on the better control of their potential disease. In addition, once patients with dyslipidemia start taking lipid-lowering agents, the LDL-C level would decrease to a certain point regardless of a positive FH of CVD, resulting in the impact of FH of CVD being less significant on control rates compared to other rates. There have been very few studies that examined the relationship between an FH of CVD and awareness, treatment, and control rates of dyslipidemia. One Chinese study also reported that the awareness and treatment rates were significantly higher in the patients with an FH of CHD compared to those without an FH, but the difference in the control rates between the two groups was insignificant [15]. Further studies investigating the medication-taking behavior, such as adherence to lipid-lowering medications between the FH of CVD and non-FH groups, would be needed to verify our results that an FH of CVD does not predict better control rates of dyslipidemia.
In terms of healthy behaviors for the prevention of CVD, there have been various studies that investigated the relationship between individuals with a positive FH of CVD and risk-reducing behaviors. Among these, the results were inconsistent as positive [19,20], negative [17,18] and no correlation [16,33] between the two have been reported previously. In addition, Imes et al. performed a systematic review of 23 articles and concluded that an individual’s awareness of a positive FH of CVD was not a sufficient predictor of health-related behavioral changes [34]. This conclusion was mostly consistent with our overall results for all participants as having an FH of CVD was positively associated with only undergoing health check-ups while it was either negatively associated (non-smoking and non-risky drinking) or had no significant correlation (weight control and sufficient physical activity) with other healthy behaviors. This trend was also consistently seen in individuals without dyslipidemia. Thus, given the result that those with an FH of CVD had a higher risk of developing dyslipidemia, it is crucial to encourage them to adopt healthier behaviors.
Regarding the healthy behaviors of patients with dyslipidemia, a positive FH of CVD was not associated with any of healthy behaviors for dyslipidemia patients. Given that a positive FH of CVD predicted better awareness and treatment rates for dyslipidemia patients but did not predict any of risk-reducing behaviors, we can assume that having an FH of CVD might positively trigger dyslipidemia patients to start pharmacological intervention, but not non-pharmacological behavioral change. In addition, considering that healthy behaviors such as weight control and sufficient physical activity are one of the most important non-pharmacological interventions that are recommended to dyslipidemia patients for the control of disease [9,10], lack of association between a positive FH of CVD and these behaviors could be another reason for an FH of CVD not predicting the better control of dyslipidemia despite its positive association with better awareness and treatment rates. Hence, physicians should pay more attention to motivating and educating patients with dyslipidemia with an FH of CVD to engage in healthier lifestyles for better control of dyslipidemia and prevention of CVD.
The strength of this study is that we used nationally representative survey data for the analyses. To the best of our knowledge, our study is the first to evaluate not only the effect of an FH of CVD on the prevalence, awareness, treatment, and control of dyslipidemia, but also healthy behaviors in patients without a history of CVD. Moreover, various confounding variables, including sociodemographic factors and health status, were adjusted in the analyses. Thus, we believe that our findings offer a meaningful perspective for the control of dyslipidemia and primary prevention of CVD in individuals with an FH of CVD.
However, our study also had some limitations that need to be addressed. First, due to the nature of the cross-sectional study, the causal relationship between an FH of CVD and outcome variables could not be guaranteed. Second, since a large portion of data used in this study were based on self-reported questionnaires, reporting bias could not be ruled out. Third, we could not fully reflect the KSoLA standard for risk stratification of each participant and its corresponding LDL-C goals since some of the risk factors were not available in the KNHANES. For instance, KSoLA actually includes an FH of premature CHD (male < 55 years, female < 65 years) as a risk factor [10]; however, we included an FH of CHD in general since there was no information regarding the age of the diagnosed family members. In addition, KSoLA also considers patients with carotid artery disease and abdominal aneurysm as a high-risk group [10] but the participants’ diagnosis status of these two conditions could not be reflected because such information was also unavailable in the KNHANES. Thus, theoretically, the prevalence and control of dyslipidemia could have been partially misestimated, but we believe that this would not affect the overall tendency of our results considering the well-established representativeness of KHNANES data. Lastly, due to the lack of information in KNHANES, we could not reflect other potential factors that could affect the awareness, treatment, control of dyslipidemia, and healthy behaviors, such as the participants’ knowledge of higher risk of CVD for those with an FH of CVD, their attitudes toward dyslipidemia and CVD, and rapport between the healthcare provider.
Conclusions
In this representative nationwide study, a positive FH of CVD was associated with higher prevalence and better awareness and treatment rates of dyslipidemia, but not control. In addition, having an FH of CVD was not predictive of any of risk-reducing behaviors for dyslipidemia patients. Therefore, physicians should make more efforts to educate and promote the importance of non-pharmacological behavioral modification among patients with dyslipidemia who have an FH of CVD.
Supporting information
(XLSX)
Data Availability
All relevant data are within the manuscript.
Funding Statement
The author(s) received no specific funding for this work.
References
- 1.Mahase E. Cancer overtakes CVD to become leading cause of death in high income countries. BMJ: British Medical Journal (Online). 2019;366. doi: 10.1136/bmj.l5368 [DOI] [PubMed] [Google Scholar]
- 2.Roth GA, Abate D, Abate KH, Abay SM, Abbafati C, Abbasi N, et al. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: a systematic analysis for the Global Burden of Disease Study 2017. The Lancet. 2018;392(10159):1736–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Arnett DK, Blumenthal RS, Albert MA, Buroker AB, Goldberger ZD, Hahn EJ, et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Journal of the American College of Cardiology. 2019;74(10):1376–414. doi: 10.1016/j.jacc.2019.03.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.LaRosa JC, He J, Vupputuri S. Effect of statins on risk of coronary disease: a meta-analysis of randomized controlled trials. Jama. 1999;282(24):2340–6. doi: 10.1001/jama.282.24.2340 [DOI] [PubMed] [Google Scholar]
- 5.Grech E, Ramsdale D, Bray C, Faragher E. Family history as an independent risk factor of coronary artery disease. European heart journal. 1992;13(10):1311–5. doi: 10.1093/oxfordjournals.eurheartj.a060059 [DOI] [PubMed] [Google Scholar]
- 6.Roncaglioni MC, Santoro L, D’Avanzo B, Negri E, Nobili A, Ledda A, et al. Role of family history in patients with myocardial infarction. An Italian case-control study. GISSI-EFRIM Investigators. Circulation. 1992;85(6):2065–72. doi: 10.1161/01.cir.85.6.2065 [DOI] [PubMed] [Google Scholar]
- 7.Liao D, Myers R, Hunt S, Shahar E, Paton C, Burke G, et al. Familial history of stroke and stroke risk: the Family Heart Study. Stroke. 1997;28(10):1908–12. doi: 10.1161/01.str.28.10.1908 [DOI] [PubMed] [Google Scholar]
- 8.Choi JC, Lee JS, Kang S-Y, Kang J-H, Bae J-M. Family history and risk for ischemic stroke: sibling history is more strongly correlated with the disease than parental history. Journal of the neurological sciences. 2009;284(1–2):29–32. doi: 10.1016/j.jns.2009.03.015 [DOI] [PubMed] [Google Scholar]
- 9.Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, Blumenthal RS, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Journal of the American College of Cardiology. 2019;73(24):e285–e350. doi: 10.1016/j.jacc.2018.11.003 [DOI] [PubMed] [Google Scholar]
- 10.Rhee E-J, Kim HC, Kim JH, Lee EY, Kim BJ, Kim EM, et al. 2018 Guidelines for the Management of Dyslipidemia in Korea. Journal of Lipid and Atherosclerosis. 2019;8(2):78–131. doi: 10.12997/jla.2019.8.2.78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mach F, Baigent C, Catapano AL, Koskinas KC, Casula M, Badimon L, et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. European heart journal. 2020;41(1):111–88. doi: 10.1093/eurheartj/ehz455 [DOI] [PubMed] [Google Scholar]
- 12.Pan L, Yang Z, Wu Y, Yin R-X, Liao Y, Wang J, et al. The prevalence, awareness, treatment and control of dyslipidemia among adults in China. Atherosclerosis. 2016;248:2–9. doi: 10.1016/j.atherosclerosis.2016.02.006 [DOI] [PubMed] [Google Scholar]
- 13.McDonald M, Hertz RP, Unger AN, Lustik MB. Prevalence, awareness, and management of hypertension, dyslipidemia, and diabetes among United States adults aged 65 and older. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences. 2009;64(2):256–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lee Yh, Lee SG, Lee MH, Kim JH, Lee BW, Kang ES, et al. Serum cholesterol concentration and prevalence, awareness, treatment, and control of high low‐density lipoprotein cholesterol in the Korea National Health and Nutrition Examination Surveys 2008–2010: beyond the Tip of the Iceberg. Journal of the American Heart Association. 2014;3(1):e000650. doi: 10.1161/JAHA.113.000650 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhang F-L, Xing Y-Q, Wu Y-H, Liu H-Y, Luo Y, Sun M-S, et al. The prevalence, awareness, treatment, and control of dyslipidemia in northeast China: a population-based cross-sectional survey. Lipids in health and disease. 2017;16(1):61. doi: 10.1186/s12944-017-0453-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Elis A, Pereg D, Tirosh A, Shochat T, Tekes-Manova D, Lishner M. Family history of cardiovascular disease does not predict risk-reducing behavior. European Journal of Cardiovascular Prevention & Rehabilitation. 2008;15(3):325–8. [DOI] [PubMed] [Google Scholar]
- 17.Andersson P, Sjöberg RL, Öhrvik J, Leppert J. Effects of family history and personal experience of illness on inclination to change health related behavior. Central European journal of public health. 2009;17(1):3–7. doi: 10.21101/cejph.a3506 [DOI] [PubMed] [Google Scholar]
- 18.Slattery ML, Murtaugh MA, Lanier AP, Ma K-N, Ferucci ED, Etzel RA, et al. Family health history and health behaviors in Alaska Native and American Indian people. Journal of health care for the poor and underserved. 2009;20(3):678. doi: 10.1353/hpu.0.0191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Thanavaro JL, Moore SM, Anthony M, Narsavage G, Delicath T. Applied Nursing Research. 2006;19(3):149–55. doi: 10.1016/j.apnr.2005.07.006 [DOI] [PubMed] [Google Scholar]
- 20.Allen JK, Blumenthal RS. Risk factors in the offspring of women with premature coronary heart disease. American Heart Journal. 1998;135(3):428–34. doi: 10.1016/s0002-8703(98)70318-7 [DOI] [PubMed] [Google Scholar]
- 21.Kim Y. The Korea National Health and nutrition examination survey (KNHANES): current status and challenges. Epidemiology and health. 2014;36. doi: 10.4178/epih/e2014002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kweon S, Kim Y, Jang M-j, Kim Y, Kim K, Choi S, et al. Data resource profile: the Korea national health and nutrition examination survey (KNHANES). International journal of epidemiology. 2014;43(1):69–77. doi: 10.1093/ije/dyt228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Health NIo. ATP III guidelines at-a-glance quick desk reference. NIH publication. 2001:01–3305.
- 24.Oh SW. Obesity and metabolic syndrome in Korea. Diabetes & metabolism journal. 2011;35(6):561–6. doi: 10.4093/dmj.2011.35.6.561 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Piercy KL, Troiano RP, Ballard RM, Carlson SA, Fulton JE, Galuska DA, et al. The physical activity guidelines for Americans. Jama. 2018;320(19):2020–8. doi: 10.1001/jama.2018.14854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shin S, Wook Shin D, Young Cho I, Jeong S-M, Jung H. Status of dyslipidemia management and statin undertreatment in Korean cancer survivors: A Korean National Health and Nutrition Examination Survey study. European Journal of Preventive Cardiology. 2020:2047487320905722. doi: 10.1177/2047487320905722 [DOI] [PubMed] [Google Scholar]
- 27.Perloff D, Grim C, Flack J, Frohlich ED, Hill M, McDonald M, et al. Human blood pressure determination by sphygmomanometry. Circulation. 1993;88(5):2460–70. doi: 10.1161/01.cir.88.5.2460 [DOI] [PubMed] [Google Scholar]
- 28.Stamler J, Daviglus ML, Garside DB, Dyer AR, Greenland P, Neaton JD. Relationship of baseline serum cholesterol levels in 3 large cohorts of younger men to long-term coronary, cardiovascular, and all-cause mortality and to longevity. Jama. 2000;284(3):311–8. doi: 10.1001/jama.284.3.311 [DOI] [PubMed] [Google Scholar]
- 29.Chen Z, Peto R, Collins R, MacMahon S, Lu J, Li W. Serum cholesterol concentration and coronary heart disease in population with low cholesterol concentrations. British Medical Journal. 1991;303(6797):276–82. doi: 10.1136/bmj.303.6797.276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Valerio L, Peters RJ, Zwinderman AH, Pinto‐Sietsma SJ. Association of family history with cardiovascular disease in hypertensive individuals in a multiethnic population. Journal of the American Heart Association. 2016;5(12):e004260. doi: 10.1161/JAHA.116.004260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rosenstock IM, Strecher VJ, Becker MH. Social learning theory and the health belief model. Health education quarterly. 1988;15(2):175–83. doi: 10.1177/109019818801500203 [DOI] [PubMed] [Google Scholar]
- 32.Rosenstock IM. Why people use health services. The Milbank Quarterly. 2005;83(4). [Google Scholar]
- 33.Kip KE, McCreath HE, Roseman JM, Hulley SB, Schreiner PJ. Absence of risk factor change in young adults after family heart attack or stroke: the CARDIA Study. American journal of preventive medicine. 2002;22(4):258–66. doi: 10.1016/s0749-3797(02)00416-6 [DOI] [PubMed] [Google Scholar]
- 34.Imes CC, Lewis FM. Family history of cardiovascular disease (CVD), perceived CVD risk, and health-related behavior: A review of the literature. The Journal of cardiovascular nursing. 2014;29(2):108. doi: 10.1097/JCN.0b013e31827db5eb [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(XLSX)
Data Availability Statement
All relevant data are within the manuscript.