Abstract
Objective
The primary objectives of this prospective cross-sectional study were to estimate the prevalence of drug-related long QT syndrome (LQTS) and the prevalence of use of QT-prolonging drugs in older patients admitted to an internal medicine unit.
Methods
We screened consecutive patients hospitalized in an internal medicine unit over a 2-year period. A 12-lead electrocardiogram using an electrocardiograph with automated measurement of QT interval was recorded. Patient characteristics (age, sex, body mass index), drug treatments, and variables associated with QT interval prolongation, including hypothyroidism, type 2 diabetes mellitus, and cardiac disease, were also recorded. In addition, we also measured serum levels of potassium, calcium, magnesium, and creatinine at admission. The list of medications known to cause or to contribute to LQTS was obtained from CredibleMeds®.
Results
A total of 243 patients were enrolled: mean ± standard deviation age, 79.65 ± 8.27 years; males, n = 121 (40.8%); mean corrected QT (QTc) interval, 453.70 ± 43.77 ms. Overall, 89/243 (36.6%) patients had a prolonged QTc interval, with 29/243 (11.9%) having QTc interval prolongation > 500 ms (11.9%). A vast majority were prescribed at least one QT-prolonging drug (218/243 [89.7%]), whereas 74/218 (30.5%) were receiving at least one medication with a known risk of Torsades des Pointes (TdP). Proton pump inhibitors were the second most commonly prescribed class of drugs. After logistic regression, male sex was independently associated with LQTS (odds ratio 2.85; 95% confidence interval 1.56–5.22; p = 0.001).
Conclusions
The prevalence of LQTS with QTc interval > 500 ms in geriatric inpatients was > 10%, and QT-prolonging drugs were frequently used on admission (more than 30% of patients were receiving drugs with a known risk of TdP).
Supplementary Information
The online version contains supplementary material available at 10.1007/s40801-021-00234-x.
Key Points
| The prevalence of drug-related corrected QT (QTc) interval prolongation > 500 ms in geriatric medical inpatients may be higher than expected. |
| Proton pump inhibitors are largely prescribed in older inpatients with QTc interval prolongation. |
| Male sex is associated with long QT syndrome in older hospitalized patients. |
Introduction
Long QT syndrome (LQTS) is a multifactorial form of heart rate (HR)-corrected QT (QTc) interval prolongation associated with an increased risk of torsade de pointes (TdP), a polymorphic ventricular tachycardia that can lead to ventricular fibrillation and sudden cardiac death [1]. A QTc interval > 500 ms is associated in the literature with a two- to threefold higher risk of TdP [1]. Recently, in a Norwegian study [2], patients with a QTc interval ≥ 500 ms had twice as high 30-day all-cause mortality as patients with a QTc interval < 500 ms, matched for age and sex and independent of comorbidity.
LQTS can be classified as either congenital or acquired. The acquired form of LQTS is far more prevalent than the congenital form and is predominantly related to the use of drugs blocking the human ether-a-go–go-related gene (hERG) potassium channel, thereby reducing the rapid component of the delayed rectifier potassium current IKr [3]. Other well-known risk factors of acquired LQTS include advanced age, hypokalemia, hypocalcemia, hypomagnesemia, bradycardia, organic heart disease, subarachnoid hemorrhage, starvation, and female sex [4]. However, two recent studies [5, 6] reported male sex as a risk factor of QTc interval prolongation in older inpatients. Moreover, specific autoantibody-mediated mechanisms occurring in patients with autoimmune diseases (anti-Ro/SSA antibodies-52kD subtype) [7] and systemic inflammation [8] have been identified as additional risk factors for TdP.
Aging is usually associated with polypharmacy, as older patients often need multiple medications, including QT-prolonging drugs, to treat comorbidities. A large German study reported that, at discharge from geriatric units, 58.7 and 22.1% of patients aged > 65 years received at least one QT-prolonging drug and two or more of such medications, respectively [9]. Moreover, a vast majority of geriatric patients are likely to cumulate other risk factors of TdP, such as heart failure, left ventricular hypertrophy, and electrolyte disturbances [10].
The prevalence of LQTS in patients admitted to different hospital settings ranges from 22 to 35% [4, 11–15]. Medical older inpatients are typically polymorbid and frequently receive QT-prolonging medications, so are at particular risk of TdP. A recent study utilizing data from a collaborative and independent prospective registry of the Italian Society of Internal Medicine (REPOSI), collecting data from 87 internal medicine and geriatric wards, found a prevalence of QT-prolonging drug use of 55.2% in geriatric inpatients on admission [16]. However, in this large retrospective analysis, Franchi et al. [16] did not collect electrocardiogram (ECG) data, electrolyte values, and other common risk factors of TdP with QT-prolonging drugs on admission or at discharge.
Despite the potentially high risk of drug-associated LQTS in geriatric inpatients, data in the literature are scarce. The primary objectives of this cross-sectional study were to estimate the prevalence of drug-related LQTS and the prevalence of use of QT-prolonging drugs in unselected older patients admitted to an internal medicine unit. Other aims of the study were to identify clinical and drug-associated risk factors of LQTS and to evaluate drug–drug interactions (DDIs) with a potential risk of QT prolongation (QT-DDIs) in this special population.
Materials and Methods
Patient Eligibility, Data Collection, and Measurement of the QTc Interval
One trained physician screened consecutive patients hospitalized in the Internal Medicine 2 Unit, University Hospital of Siena, from 13 April 2017 to 13 April 2019. Our institution is a public hospital with approximately 700 beds: our unit has 38 beds, with 86 hospitalizations per month recorded in 2017, 88 in 2018, and 92 in 2019. We excluded patients with atrial fibrillation, bigeminy, wide QRS complex (> 120 ms), or unidentifiable T waves on the baseline ECG as the QT interval cannot be reliably measured under such conditions. All patients aged > 65 years prescribed at least one drug on admission who signed the informed consent were enrolled in the study. At the time of enrollment, the physician collected information from patients on drugs prescribed by their general practitioner. Details of prescriptions during hospitalization were extracted from electronic medical records. Characteristics associated with LQTS in the medical literature [4] were also recorded in each case report form, including patient age, sex, body mass index (BMI), serum potassium, calcium, magnesium, and creatinine levels (all obtained the same day of the ECG) and diseases, including hypertension, hypothyroidism, type 2 diabetes mellitus, and cardiac diseases (acute coronary syndrome, dilated cardiomyopathy/heart failure, chronic ischemic cardiopathy, left ventricular hypertrophy). We defined hypokalemia as a potassium level < 3.5 mEq/L, hypocalcemia as calcium level < 8.0 mg/dL, hypomagnesemia as magnesium level < 1.50 mg/dL, and elevated serum creatinine as a creatinine level of > 1.10 mg/dL. Only levels obtained before the administration of intravenous magnesium sulfate and/or replacement therapy with potassium or calcium were considered appropriate for inclusion in the study. Within 24 h of patient admission, a trained physician performed a 12-lead ECG using an electrocardiograph with automated measurement of QT interval (Philips PageWriter TC50®). LQTS was defined as a QTc interval of > 450 ms in men and > 470 ms in women using Bazett’s formula (QTc = QT/√RR) [17]. Although the Bazett correction (QTcB) tends to produce overlong QTc interval values at faster heart rates [1], it was chosen because it is the most widely used in clinical practice, and QTc interval lengthening based on this correction formula has been associated with an increased risk of TdP and sudden death in the geriatric population [18].
As in most similar studies, we obtained the list of QT-prolonging medications from CredibleMeds® (formerly the Arizona Center for Education and Research on Therapeutics [AZCERT]) [19]. The AZCERT team rigorously evaluates every drug and constantly updates these evaluations using information available from basic science and clinical evidence from the medical literature, the US FDA summary basis of drug approvals, and labels. For this study, we included drugs from all three risk group lists: Drugs with a known risk of TdP (list 1) are generally accepted as causing TdP; drugs with a possible risk of TdP (list 2) have been associated with TdP in some reports, but compelling evidence for causing this arrhythmia is lacking; drugs with a conditional risk of TdP (list 3) carry a risk of TdP under certain conditions, such as in patients with congenital LQTS, drug overdose, or coadministration of interacting drugs. Our analysis was based on data extracted from the CredibleMeds® website on 23 August 2017. Drugs considered to have no risk of TdP by AZCERT were not considered in the analysis. Since hospitalization in the acute medical units (AMU) in our institution can last ≤ 3 days before admission to internal medicine units, we define chronic use as treatment with a QT-prolonging agent for ≥ 4 days. The IBM Micromedex Drugdex® [20] drug interactions tool was used to evaluate the medicines prescribed for each patient to identify the presence of QT-DDIs. The mechanism of each QT-DDI was classified as either pharmacokinetic or pharmacodynamic. Pharmacokinetic QT-DDIs were defined as attributable to alterations in the metabolism of QT-prolonging medications. Pharmacodynamic QT-DDIs were defined as attributable to the effects of two drugs identified as directly causing prolongation of the QTc interval (mainly blocking the hERG potassium channel).
Statistical Analysis
Continuous variables were described as mean and standard deviation. Possible statistical associations between QTc interval and continuous variables were analyzed using the bivariate Pearson correlation analysis. Categorical variables were expressed as number and percentage. Patients were divided into two subgroups according to the presence of LQTS, and a case–control analysis was performed. For continuous variables, the two-tailed independent t test or the Mann–Whitney test (in case of non-normal distribution) were used. To explore statistical correlation between LQTS and dichotomous variables, the two-sided χ2 test or the Fisher’s exact test (if n < 5) were used. Variables that were significant in the univariate analysis were then tested in a multivariate model using logistic regression. Statistical significance was set at p < 0.05 for all comparisons. All statistical analyses were performed using SPSS v.24.0 (IBM Corp., Armonk, NY, USA).
Results
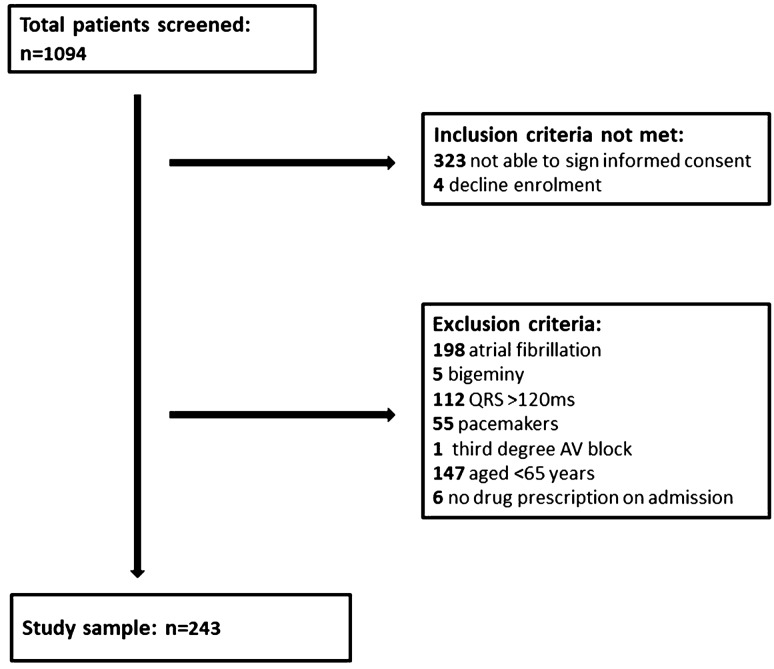
A total of 243 patients were enrolled in the study: mean age, 79.65 ± 8.27 years, males, n = 121 (40.8%), mean QTc interval, 453.70 ± 43.77 ms. Figure 1 shows the flow chart of the full screening process. Table 1 displays demographic and clinical characteristics and a comparison of variables in patients with and without LQTS on admission. Overall, 89/243 (36.6%) patients had LQTS. A QTc interval prolongation > 500 ms was present in 29/243 (11.9%) patients. A vast majority were prescribed at least one QT-prolonging drug (218/243 [89.7%]), whereas 74/218 (30.5%) were on at least one medication with known risk of TdP (list 1). Notably, more than one-third of our cohort was receiving treatment with more than two QT-prolonging agents at the time of the ECG.
Fig. 1.
Selection of study sample AV atrioventricular
Table 1.
Demographic, clinical, and laboratory characteristics of patients and comparison of variables in subjects with and without long QT syndrome on admission
| Variable | Patients (n = 243) | Patients with LQTSa (n = 89) | Patients without LQTSa (n = 154) | p valueb |
|---|---|---|---|---|
| Age, years | 79.65 ± 8.27 | 81.79 ± 8.40 | 78.42 ± 7.96 | 0.002 |
| Males | 121 (49.8) | 55 (61.8) | 66 (27.8) | 0.004 |
| Body mass index | 25.32 ± 4.79 | 24.28 ± 3.22 | 25.92 ± 5.41 | 0.005 |
| Total number of drugs | 7.10 ± 3.04 | 7.62 ± 2.92 | 6.80 ± 3.07 | 0.043 |
| QT-prolonging drug usersc | 218 (89.7) | 84 (94.4) | 134 (87.0) | 0.069 |
| Number of QT-prolonging drugs | 2.04 ± 1.27 | 2.35 ± 1.25 | 1.86 ± 1.25 | 0.004 |
| > 1 QT-prolonging drug usersc | 156 (64.2) | 65 (73.0) | 91 (59.1) | 0.029 |
| > 2 QT-prolonging drugs usersc | 82 (33.7) | 40 (44.9) | 42 (27.3) | 0.005 |
| Chronic QT-prolonging drugs usersc | 165 (67.9) | 70 (78.6) | 95 (61.7) | 0.006 |
| List 1c drug users | 74 (30.5) | 32 (35.9) | 42 (27.3) | 0.157 |
| List 2c drug users | 25 (10.3) | 13 (14.6) | 12 (7.8) | 0.092 |
| List 3c drug users | 206 (84.8) | 79 (88.8) | 127 (82.5) | 0.188 |
| Serum potassium (mEq/L)d | 4.11 ± 0.58 | 3.96 ± 0.56 | 4.20 ± 0.58 | 0.002 |
| Serum calcium (mg/dL)d | 8.30 ± 0.81 | 8.13 ± 0.97 | 8.40 ± 0.69 | 0.013 |
| Serum magnesium (mg/dL)d | 1.91 ± 0.32 | 1.90 ± 0.38 | 1.92 ± 0.28 | 0.550 |
| Creatinine (mg/dL), median (IQR) | 1.21 ± 0.84 | 1.10 (0.75–1.61) | 0.88 (0.67–1.29) | 0.009 |
| Bradycardia (heart rate < 60 bpm) | 28 (11.5) | 10 (11.2) | 18 (11.3) | 0.915 |
| Hypokalemiad (< 3.5 mEq/L) | 23 (9.7) | 10 (11.2) | 13 (8.4) | 0.450 |
| Hypocalcemiad(< 8.0 mg/dL) | 63 (26.8) | 31 (34.8) | 32 (20.8) | 0.015 |
| Hypomagnesemiad (< 1.5 mg/dL) | 18 (7.9) | 9 (10.1) | 9 (5.8) | 0.222 |
| Hypercreatininemia (> 1.2 mg/dL) | 99 (40.7) | 43 (48.3) | 56 (36.4) | 0.068 |
| Hypertension | 152 (63.0) | 59 (66.3) | 94 (61.0) | 0.414 |
| Hypothyroidism | 18 (7.4) | 12 (13.5) | 6 (3.9) | 0.006 |
| Type 2 diabetes mellitus | 60 (24.7) | 24 (27.0) | 36 (22.6) | 0.532 |
| Acute coronary syndrome | 3 (1.2) | 2 (2.2) | 1 (0.6) | 0.556 |
| Dilated cardiomyopathy/heart failure | 34 (14.0) | 21 (21.8) | 13 (8.9) | 0.001 |
| Chronic ischemic cardiopathy | 43 (17.7) | 20 (22.5) | 23 (14.5) | 0.138 |
| Left ventricular hypertrophy | 52 (21.4) | 24 (27.0) | 28 (17.6) | 0.108 |
Data are presented as mean ± standard deviation or n (%) unless otherwise indicated
IQR interquartile range, LQTS long QT syndrome, QTc corrected QT interval
aDefined as QTc interval QTc ≥ 450 ms in males and ≥ 470 ms in females using Bazett’s correction formula
bThe two-tailed independent t-test or the Mann–Whitney test (in case of non-normal distribution) were used for continuous variables, whereas the χ2 test or the Fisher exact test (in case of n < 5) were used for categorical variables
cDrugs on admission, from https://www.crediblemeds.org
dSerum potassium, calcium, and magnesium levels available in 237, 235, and 229 patients, respectively. Outcomes: LQTS,b n = 89 (36.6%); QTc > 500 ms, n = 29 (11.9%); uncorrected QT, 406.92 ± 49.09 ms; heart rate, 76.02 ± 13.28 beats/minute; QTc, 453.70 ± 43.77 ms
Serum potassium, calcium, and magnesium levels were available in 237 (97.5%), 235 (96.7%), and 229 (94.2%) patients, respectively. The most common electrolyte imbalance observed was hypocalcemia (63 [26.8%]). Moreover, a significant proportion of patients had underlying cardiac diseases, more frequently left ventricular hypertrophy (52 [21.4%]), dilated cardiomyopathy/heart failure (43 [17.7%]), and chronic ischemic cardiopathy (34 [14.0%]).
Table 2 shows the number and TdP risk lists of QT-prolonging drugs prescribed in 218 patients. A total of 498 QT-prolonging agents were used at the time of ECG: the most prescribed were furosemide (121/498 prescriptions [24.3%]), pantoprazole (91/498 [18.3%]), and lansoprazole (35/498 [7.0%]). Clarithromycin (19/498 prescriptions [3.8%]), amiodarone (13/498 [2.6%]), and citalopram (11/498 [2.2%]) were the most used medications with a known risk of TdP (list 1).
Table 2.
Prescription of QT-prolonging drugs (more than two prescriptions) stratified by risk category and therapeutic classes
| QT-prolonging drugs with more than two prescriptions | Number of prescriptions (n = 498) | TdP risk listsa |
|---|---|---|
| Antimicrobials | ||
| Piperacillin/tazobactam | 34 (6.8) | 3 |
| Clarithromycin | 19 (3.8) | 1 |
| Metronidazole | 11 (2.2) | 3 |
| Levofloxacin | 10 (2.0) | 1 |
| Ciprofloxacin | 6 (1.2) | 1 |
| Antidepressants | ||
| Citalopram | 11 (2.2) | 1 |
| Sertraline | 11 (2.2) | 3 |
| Paroxetine | 9 (1.8) | 3 |
| Venlafaxine | 7 (1.4) | 2 |
| Escitalopram | 4 (0.8) | 1 |
| Mirtazapine | 4 (0.8) | 2 |
| Trazodone | 3 (0.6) | 3 |
| Cardiovascular | ||
| Furosemide | 121 (24.3) | 3 |
| Amiodarone | 13 (2.6) | 1 |
| Flecainide | 4 (0.8) | 1 |
| Ranolazine | 4 (0.8) | 2 |
| Valsartan/hydrochlorothiazide | 4 (0.8) | 3 |
| Ivabradine | 3 (0.6) | 3 |
| Ramipril/hydrochlorothiazide | 3 (0.6) | 3 |
| Perindopril/indapamide | 3 (0.6) | 3 |
| Proton pump inhibitors | ||
| Pantoprazole | 91 (18.3) | 3 |
| Lansoprazole | 35 (7.0) | 3 |
| Omeprazole | 22 (4.4) | 3 |
| Esomeprazole | 6 (1.2) | 3 |
| Others | ||
| Metoclopramide | 6 (1.2) | 3 |
| Quetiapine | 5 (1.0) | 3 |
| Alfuzosin | 5 (1.0) | 2 |
Data are presented as n (%) unless otherwise indicated
aClassification from https://www.crediblemeds.org. List 1 = drugs with known risk of torsade de pointes; list 2 = drugs with possible risk of torsade de pointes; list 3 = drugs with conditional risk of torsade de pointes
A total of 31/243 (12.8%) patients presenting with 44 QT-DDIs were identified (Table 3). Multiple QT-DDIs were identified in 9/31 (29.0%) subjects: 6/31 (19.3%) presented with two QT-DDIs, 1/31 (3.2%) with three QT-DDIs, and 2/31 (6.4%) with four QT-DDIs. The most frequent (n > 1) drug interacting pairs were citalopram–omeprazole (4/44 [9.1%]), citalopram–pantoprazole (3/44 [6.8%]), quetiapine–trazodone, citalopram–clarithromycin, clarithromycin–amiodarone, and clarithromycin–paroxetine (2/44 [4.5%]). According to CredibleMeds® classifications, drugs with a known risk of TdP (list 1), a possible risk of TdP (list 2), and a conditional risk of TdP (list 3) were involved in 32/44 (72.7%), 7/44 (15.9%), and 33/44 (75.0%) QT-DDIs, respectively. The most common medications (n > 4) implicated in QT-DDIs were citalopram (n = 8), quetiapine (n = 6), metronidazole (n = 6), and clarithromycin (n = 5). According to Micromedex®, the majority of the QT-DDIs were pharmacodynamic (n = 26), but the two most frequent QT-DDIs (citalopram–omeprazole [n = 4] and citalopram–pantoprazole [n = 3]) were ascribable to a pharmacokinetic mechanism.
Table 3.
Two or more QT-prolonging drug–drug interactions according to CredibleMeds® risk category and mechanism
| QT-DDIs | n (N = 15) | List 1a | List 2a | List 3a | QT-DDI classification |
|---|---|---|---|---|---|
| Citalopram–omeprazole | 4 | 1 | 0 | 1 | PK |
| Citalopram–pantoprazole | 3 | 1 | 0 | 1 | PK |
| Quetiapine–trazodone | 2 | 0 | 0 | 2 | PD |
| Citalopram–clarithromycin | 2 | 2 | 0 | 0 | PD |
| Clarithromycin–amiodarone | 2 | 2 | 0 | 0 | PK, PD |
| Clarithromycin–paroxetine | 2 | 1 | 0 | 1 | PD |
DDI drug–drug interaction, PD pharmacodynamics, PK pharmacokinetics, QT-DDIs DDIs with a potential risk of QT interval prolongation identified using the IBM Micromedex Drugdex® drug interactions tool
aClassification from https://www.crediblemeds.org. List 1 = drugs with known risk of torsade de pointes; list 2 = drugs with possible risk of torsade de pointes; list 3 = drugs with conditional risk of torsade de pointes
Table 4 presents the results of the Pearson correlation for continuous variables. Significant positive associations were present between QTc and age (r = 0.159, p = 0.013) and number of QT interval-prolonging drugs used (r = 0.200, p = 0.002), whereas significant negative associations existed between QTc interval and potassium levels (r = − 0.280, p < 0.0001), calcium levels (r = − 0.166, p = 0.011), and magnesium levels (r = − 0.133, p = 0.044).
Table 4.
Pearson correlation coefficients of quantitative determinants of corrected QT interval prolongation
| Variable | Pearson r | p value |
|---|---|---|
| Age | 0.159 | 0.013 |
| Body mass index | − 0.091 | 0.172 |
| Serum potassium | − 0.280 | < 0.0001 |
| Serum calcium | − 0.166 | 0.011 |
| Serum magnesium | − 0.133 | 0.044 |
| Creatinine | 0.066 | 0.304 |
| Total number of drugs | 0.119 | 0.064 |
| Number of QT-prolonging drugs | 0.200 | 0.002 |
Results of the univariate analysis of the dichotomous variables are shown in Table 5. A significant correlation was identified between LQTS and the use of more than one QT interval-prolonging drug (odds ratio [OR] 1.87; 95% confidence interval [CI] 1.06–3.31; p = 0.029), use of more than two QT interval-prolonging drugs (OR 2.18; 95% CI 1.26–3.76; p = 0.005), chronic use of QT interval-prolonging drugs (OR 2.29; 95% CI 1.25–4.18; p = 0.006), male sex (OR 2.16; 95% CI 1.26–3.68; p = 0.004), hypocalcemia (OR 2.06; 95% CI 1.14–3.71; p = 0.015), hypothyroidism (OR 3.84; 95% CI 1.39–10.64; p = 0.006), and dilated cardiomyopathy/heart failure (OR 3.35; 95% CI 1.58–7.09; p = 0.001). Multivariate analysis of the seven significant variables determined by univariate analysis identified five factors significantly associated with LQTS (Table 6): male sex (OR 2.85; 95% CI 1.56–5.22; p = 0.001), hypocalcemia (OR 2.18; 95% CI 1.13–4.20; p = 0.019), hypothyroidism (OR 3.74; 95% CI 1.25–11.25; p = 0.019), dilated cardiomyopathy/heart failure (OR 3.67; 95% CI 1.58–8.51; p = 0.002), and chronic use of QT-prolonging drugs (OR 2.37; 95% CI 1.13–4.99; p = 0.023).
Table 5.
Univariate analysis of dichotomy risk factors of long QT syndrome
| Variable | OR (95% CI) for LQTS | p value |
|---|---|---|
| QT-prolonging drugs users | 2.51 (0.91–6.93) | 0.069 |
| > 1 QT-prolonging drug users | 1.87 (1.06–3.31) | 0.029 |
| > 2 QT-prolonging drugs users | 2.18 (1.26–3.76) | 0.005 |
| Chronic QT-prolonging drugs users | 2.29 (1.25–4.18) | 0.006 |
| List 1 drugs users | 1.50 (0.85–2.62) | 0.157 |
| List 2 drugs users | 2.02 (0.88–4.65) | 0.092 |
| List 3 drugs users | 1.68 (0.77–3.66) | 0.188 |
| Male sex | 2.16 (1.26–3.68) | 0.004 |
| Bradycardia | 0.96 (0.42–2.17) | 0.915 |
| Hypokalemia | 1.40 (0.59–3.34) | 0.450 |
| Hypocalcemia | 2.06 (1.14–3.71) | 0.015 |
| Hypomagnesemia | 1.81 (0.69–4.77) | 0.222 |
| Hypercreatininemia | 1.64 (0.96–2.78) | 0.068 |
| Hypertension | 1.25 (0.73–2.17) | 0.414 |
| Hypothyroidism | 3.84 (1.39–10.64) | 0.006 |
| Type 2 diabetes mellitus | 1.21 (0.66–2.20) | 0.532 |
| Acute coronary syndrome | 3.52 (0.31–39.35) | 0.556 |
| Dilated cardiomyopathy/heart failure | 3.35 (1.58–7.09) | 0.001 |
| Chronic ischemic cardiopathy | 1.65 (0.85–3.21) | 0.138 |
| Left ventricular hypertrophy | 1.66 (0.89–3.09) | 0.108 |
CI confidence interval, LQTS long QT syndrome, OR odds ratio
Table 6.
Multivariate model by logistic regression of dichotomy risk factors of long QT syndrome
| Variable | Adjusted OR (95% CI) for LQTS |
p value |
|---|---|---|
| > 1 QT-prolonging drug user | 0.94 (0.43–2.08) | 0.885 |
| > 2 QT-prolonging drug users | 1.60 (0.78–3.30) | 0.200 |
| Chronic QT-prolonging drugs users | 2.35 (1.11–4.95) | 0.025 |
| Male sex | 2.83 (1.55–5.19) | 0.001 |
| Hypocalcemia | 2.17 (1.13–4.18) | 0.020 |
| Hypothyroidism | 3.74 (1.25–11.22) | 0.019 |
| Dilated cardiomyopathy/heart failure | 3.65 (1.58–8.46) | 0.003 |
CI confidence interval, LQTS long QT syndrome, OR odds ratio
Discussion
In this study, we observed a high proportion (89/243 [36.6%]) of LQTS in a cohort of consecutive older medical inpatients and, notably, 11.9% (29/243) of QTc interval prolongation > 500 ms. Our prevalence rates were significantly greater than those described in two previous retrospective studies in geriatric inpatients. Using the same definition of LQTS, Lubart et al. [12] reported QTc interval prolongation in 27% (115/422) of patients admitted to an acute geriatric ward (7.15% with QTc interval > 500 ms). In a 6-month study performed in a French university hospital, Maison et al. [5] found LQTS in 22% (33/152) of patients (5% with QTc interval > 500 ms). In studies applying Bazett’s correction, a comparable prevalence of QTc interval > 500 ms of 10.7% was found in 412 patients aged > 18 years consecutively admitted to an intensive care unit [21], and a considerably higher prevalence of 18.2% was reported by Tisdale et al. [22] in patients (n = 900) admitted to cardiac care units. Although it seems plausible that the use of Bazett’s formula with thresholds of 450 ms in men and 470 ms in women may have resulted in an overestimation of overall patients with LQTS in this study, the prevalence of QTc > 500 ms may not be affected by the use of this correction formula, given that the application of Fridericia’s formula to QTc interval in our cohort led to a similar prevalence of 10.3%.
Another relevant finding of this study is that drugs classified in the CredibleMeds® lists were used in the majority of older patients on admission to our internal medicine unit: nearly 90% (218/243) of subjects were receiving at least one QT interval-prolonging drug at the time of ECG. In addition, more than 30% (74/243) of patients were under treatment with agents in CredibleMeds® list 1 (known risk of TdP). We postulated that this finding was because of a large use of QT interval-prolonging medications during the hospitalization. Indeed, 53/243 (21.8%) patients may have had prescriptions of QT interval-prolonging agents during their stay in the emergency department and/or in the AMU. This hypothesis is consistent with several studies reporting a trend towards adding QT interval-prolonging drugs in different hospital settings, either during hospitalization or at discharge. Pasquier et al. [11] found that half of patients with prolonged QT interval at the time of admission received at least one additional QT interval-prolonging drug during the hospital stay in an internal medicine unit. Tisdale et al. [22] also reported that 87/251 (34.7%) patients admitted to cardiac care units with QTc interval prolongation were subsequently administered QT interval-prolonging drugs. Franchi et al. [16] observed the use of QT interval-prolonging drugs in 55.2% of geriatric inpatients on admission that increased to 64.2% at discharge, a finding consistent with that of Schächtele et al. [9], who found that 58.7% of patients were prescribed QT interval-prolonging drugs at discharge from geriatric units. In our cohort, 165/243 (67.9%) patients were chronic users of QT interval-prolonging agents, a finding consistent with that of Maison et al. [5], who reported the use of QT interval-prolonging medications in 61.8% (94/152) of patients at the time of ECG. As we defined chronic use as treatment with a QT interval-prolonging agent for ≥ 4 days, it seems plausible that these data highlight prescriptions by general practitioners, suggesting that drug-induced LQTS may be underestimated in this setting.
Frequent co-prescription of QT interval-prolonging drugs has been reported in the literature for the last two decades. A retrospective cohort study using an outpatient database of one of the largest pharmacy benefit managers in the USA identified a prevalence of QT-DDIs of 9.4% (103,119/1.1 million individuals) in 1999 [23]. Despite this, it appears that software to help prescribers detect QT-DDIs is not commonly adopted and a retrospective database study [24] found that, even with the support of an electronic system alert, ECG monitoring was performed in only 33% of patients prescribed two or more medications associated with LQTS. In the present study, we found that Micromedex®, one of the most reliable and sensitive software programs for the analysis of DDIs [25], may be insufficient to quantify the cumulative pharmacological QT interval-lengthening risk in internal medical units. The systematic use of this tool in our cohort identified QT-DDIs in only 31/243 (12.8%) patients, largely underestimating pharmacodynamic QT-DDIs, given that almost two-thirds of our patients received two or more medications that prolonged the QT interval according to CredibleMeds® lists at the time of ECG.
In line with previously published papers showing an increased risk of LQTS with the use of QT interval-prolonging drug combinations compared with single drugs, we observed that QTc interval correlated with the number of QT interval-prolonging agents at Pearson correlation. In animal studies, the simultaneous administration of two potassium channel blockers markedly potentiated the increase of the QTc interval [26, 27]. Moreover, most clinical studies have highlighted an enhanced TdP risk in patients prescribed multiple QT interval-prolonging drugs. A retrospective case–control study that quantified the risk of cardiac arrest related to the use of non-arrhythmic LQTS inducers in 140 patients resuscitated from cardiac arrest at a Dutch university hospital found an increased risk of arrest associated with the number of prescribed LQTS-associated drugs (OR 4.8; 95% CI 1.6–14) [28]. Drug interactions also represented a high proportion of cases of drug-induced TdP within a Belgium study using the EudraVigilance database (18/31 cases) [29]. However, some evidence in the literature is conflicting. A retrospective study based on a US hospital database that quantified the risk of LQTS in patients receiving medicines classified in CredibleMeds® lists did find QTc interval prolongation (p < 0.001) in patients receiving a single medication with a known risk of TdP compared with those receiving medications with no risk of TdP but found no statistically significant difference in the QTc interval among patients prescribed multiple QT interval-prolonging drugs [30].
As highlighted in several studies, a limited number of agents account for the majority of prescriptions of QT interval-prolonging drugs in geriatric inpatients, namely furosemide, citalopram, and amiodarone. Indeed, in the previously cited German study [9], citalopram and amiodarone were identified as the first and third most commonly used drugs with a known risk of TdP, and only 20 QT-DDIs accounted for more than 90% of co-prescriptions of QT interval-prolonging agents. Thus, it seems plausible that an accurate analysis of a restricted number of drugs could be very relevant in the clinical management of the risk of TdP in older inpatients.
Our finding that furosemide was the most used QT interval-prolonging drug in geriatric inpatients is in agreement with findings of other recent studies [5, 16]. The association between diuretics and in-hospital TdP may be explained by its correlation with congestive heart failure, hypokalemia, and hypomagnesemia [1]. Although the use of loop diuretics was an independent risk factor for QTc interval prolongation [6, 31], the risk of associated TdP is generally considered low. However, as furosemide is the mainstay of treatment of severe cardiovascular diseases, greater attention should be paid to frequently co-prescribed QT interval-prolonging medications.
In our cohort, proton pump inhibitors (PPIs) were the second most commonly prescribed class of QT interval-prolonging drugs, a finding consistent with the Italian REPOSI study [16], which found pantoprazole to be the second most frequently prescribed QT interval-prolonging agent on admission (340/2156 [11.8%]). We found a total of 154/498 (25.1%) PPI prescriptions, and PPIs were frequently co-administered with furosemide (n = 151) and amiodarone (n = 19). Despite accumulating evidence, PPIs do not currently receive attention as a factor potentially contributing to QTc interval prolongation in clinical practice. In a very recent Swedish prospective register-based cohort study on the occurrence of TdP in relation to drugs listed in the CredibleMeds® classification, 131/410 (32%) patients with TdP were also receiving PPIs [32]. Although rare, PPI-induced hypomagnesemia can be associated with TdP [33] according to experimental studies showing that cytosolic magnesium promotes repolarization of myocardial cells via its modulating effects on several potassium channels, including IKr and the transient outward current [34]. Nevertheless, PPIs may also significantly influence the QTc, regardless of hypomagnesemia. In this study, we identified five cases of LQTS (three with QTc interval > 500 ms) in eight patients co-administered ceftriaxone and lansoprazole. This finding is in agreement with recent evidence showing that lansoprazole in combination with ceftriaxone carries a risk of QTc interval prolongation, as it has been demonstrated that ceftriaxone + lansoprazole can significantly block the hERG channel and inhibit IKr (− 57.6%) [35, 36]. According to these data, and given that PPIs are often prescribed inappropriately [37, 38], the use of this class of drugs in older people who present with a cumulative risk of TdP should be accurately evaluated either during hospitalization or at hospital discharge.
Citalopram and sertraline were the most prescribed antidepressants in our cohort. Citalopram was implicated in more QT-DDIs (n = 12; 4.94% of the study sample), notably eight pharmacokinetic QT-DDIs (including in combination with omeprazole in four patients, who might therefore have been exposed to significantly higher citalopram concentrations (+ 35.3%) because of omeprazole’s inhibition of cytochrome P450-2C19 [39]) and four pharmacodynamic QT-DDIs (see the electronic supplementary material). Despite general agreement in the literature, including a recent meta-analysis [40], that citalopram is the selective serotonin reuptake inhibitor more consistently associated with QTc prolongation, and recommendations from the FDA [41] and other regulatory authorities, awareness of this issue in older patients seems to be lacking.
The multivariate analysis identified non-pharmacological risk factors associated with LQTS in the literature, including hypocalcemia, hypothyroidism, and dilated cardiomyopathy/heart failure. However, in our opinion, the most relevant finding was the independent association between male sex and LQTS (OR 2.83; 95% CI 1.55−5.19; p = 0.001). Although females are generally considered at greater risk for drug-induced TdP than males [1], this surprising correlation has also been observed in three previous studies in older inpatients [5]. In a cross-sectional study in 178 residents of long-term care wards in a geriatric hospital, Lubart et al. [42] found a statistically significant prevalence of prolonged QTc interval in males versus females, whereas the previously cited paper by Maison et al. [5] reported an association between male sex and QTc interval prolongation after multivariate analysis (OR 3.25; 95% CI 1.43–7.41; p = 0.004). More recently, in an Italian study in 432 patients (mean age 83.3 years), Bo et al. [6] observed a significant association between male sex and QTc interval prolongation (OR 2.09; 95% CI 1.34–3.26). Physiological evidence could at least partly explain this finding. The mean QTc interval is longer in women than in men from adolescence until the sixth decade, but this difference tends to disappear in very old patients. Recently, Rautaharju et al. [43] demonstrated that, while the QTc interval still increases with increasing age in the elderly, the slope appears to be greater in men, and the men’s curve almost crosses the women’s curve in patients aged over 80 years, highlighting the fact that the sex difference in QTc may arise from a shortened QT interval in adolescent males, with no shortening in adolescent females, as documented in a previous study of the same group [44]. The shorter QTc interval in men is likely due to the testosterone-associated altered dynamics of cardiac ion channels [45, 46]: preclinical studies showed that testosterone can both increase the repolarizing potassium currents IKr and IKs and decrease the depolarizing L-type calcium current, ICaL [45]. Furthermore, Bidoggia et al. [47] found that the QT interval was prolonged in castrated compared with non-castrated men and that women with hyperandrogenism had shorter QT intervals than controls. Zhang et al. [48] compared multivariate-adjusted QT intervals in the highest and lowest quartiles of total and free testosterone levels in men in the Third National Health and Nutrition Examination Survey and in men and postmenopausal women in the Multi-Ethnic Study of Atherosclerosis. They identified significant differences in the adjusted QT interval between the highest and lowest quartiles of testosterone in men but not in women [48]. In addition, in recent years, evidence has supported the hypothesis that androgen-deprivation therapy (ADT) can increase the risk of TdP [49]. Clinical interventional studies in men with prostate cancer demonstrated that ADT is associated with QTc prolongation [50–53]. In a prospective cohort of 58 consecutive unselected patients experiencing TdP, ADT unexpectedly represented the second most frequently administered QT interval-prolonging drug in males (3/19 [16%]), after amiodarone [54]. Moreover, Salem et al. [55] reported that the androgen receptor antagonist enzalutamide significantly prolonged action potential duration in induced pluripotent stem cell-derived cardiomyocytes. These effects were associated with IKr inhibition and INa enhancement and were reversed by dihydrotestosterone. However, on the basis of the available evidence, a similar risk of LQTS should be identified in very old men and women, so further research is needed to clarify this issue.
Study Limitations
The present study has several limitations. First, this study enrolled patients admitted to a single medical unit. Second, this was a cross-sectional design without follow-up of possible arrhythmic events in patients with LQTS on admission. Other limitations are related to the use of Bazett’s formula, as it tends to overcorrect QT intervals at a faster HR; moreover, we used the clinical standards of 450 ms in men and 470 ms in women, which may have overestimated the number of patients with prolonged QT interval when using QTcB. In addition, the high proportion of patients who did not provide informed consent and so were excluded from the study, may have affected the true prevalence of LQTS in our cohort. Recall bias is also possible as participants were asked to remember information about their prescriptions from their general practitioners on admission.
Conclusions
The prevalence of LQTS with QTc interval > 500 ms in geriatric inpatients was > 10%, and QT interval-prolonging drugs were frequently used on admission (more than 30% of patients were treated with drugs with a known risk of TdP). Male sex was independently associated with LQTS in our cohort. PPIs were the second most frequently prescribed class of drugs, after diuretics. Given the increasing evidence on the risk of LQTS from PPIs, education on the appropriate prescription of this class of drugs in older patients, who are at particular risk for life-threatening arrhythmias, is needed. Further larger population-based studies are warranted to better define the role of PPIs and sex as a risk factor of LQTS in very old patients.
Supplementary Information
Below is the link to the electronic supplementary material.
Declarations
Funding
This study was supported by a grant from the Italian Medicines Agency, AIFA (no. H56D15000370001).
Conflicts of Interest
Marco Rossi, Federico Marzi, Mariarita Natale, Aristotele Porceddu, Marco Tuccori, Pietro Enea Lazzerini, Franco Laghi-Pasini, and Pier Leopoldo Capecchi have no conflicts of interest that are directly relevant to the content of this article.
Ethics Approval
The study has been approved by our institutional ethic committee on December 19, 2016 (reference number 10451/2016) and it has been performed in accordance with the ethical standards as laid down in the Declaration of Helsinki. Informed consent, including consent for publication, was obtained from all individual participants.
Consent to Participate
All patients included in the database were anonymized by the use of alphanumeric codes assigned by the system.
Consent for Publication
Consent for publication was obtained from all individual participants.
Availability of Data and Material
The dataset generated and analyzed during the study is available from the corresponding author.
Code Availability
All patients included in the database were anonymized by the use of alphanumeric codes assigned by the system.
Author Contributions
MR: conceptualization, methodology, software, formal analysis, writing—original draft. FM: investigation, validation. AP: investigation. MN: data curation, validation. MT: software. PEL: supervision, methodology, formal analysis. FLP: conceptualization, methodology, resources, funding acquisition. PLC: supervision, visualization, writing—review and editing. All authors read and approved the final manuscript.
Footnotes
Franco Laghi-Pasini: Retired.
References
- 1.Drew BJ, Ackerman MJ, Funk M, Gibler WB, Kligfield P, Menon V, et al. Prevention of torsade de pointes in hospital settings: a scientific statement from the American Heart Association and the American College of Cardiology Foundation. J Am Coll Cardiol. 2010;55(9):934–947. doi: 10.1016/j.jacc.2010.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gibbs C, Thalamus J, Kristoffersen D, Svendsen M, Holla O, Heldal K, et al. QT prolongation predicts short-term mortality independent of comorbidity. Europace. 2019;21(8):1254-60. [DOI] [PMC free article] [PubMed]
- 3.Schwartz PJ, Woosley RL. Predicting the unpredictable: drug-induced QT prolongation and torsades de pointes. J Am Coll Cardiol. 2016;67(13):1639–1650. doi: 10.1016/j.jacc.2015.12.063. [DOI] [PubMed] [Google Scholar]
- 4.Roden DM. Drug-induced prolongation of the QT interval. N Engl J Med. 2004;350(10):1013–1022. doi: 10.1056/NEJMra032426. [DOI] [PubMed] [Google Scholar]
- 5.Maison O, de la Gastine B, Dayot L, Goutelle S. Prevalence and risk factors of drug-associated corrected QT prolongation in elderly hospitalized patients: results of a retrospective analysis of data obtained over 6 months. Drugs Aging. 2017;34(7):545–53. [DOI] [PubMed]
- 6.Bo M, Ceccofiglio A, Mussi C, Bellelli G, Nicosia F, Riccio D. Prevalence, predictors and clinical implications of prolonged corrected QT in elderly patients with dementia and suspected syncope. Eur J Intern Med. 2019;61:34–39. doi: 10.1016/j.ejim.2018.10.011. [DOI] [PubMed] [Google Scholar]
- 7.Lazzerini PE, Yue Y, Srivastava U, Fabris F, Capecchi PL, Bertolozzi I, et al. Arrhythmogenicity of Anti-Ro/SSA Antibodies in Patients With Torsades de Pointes. Circ Arrhythm Electrophysiol. 2016;9(4):e003419. doi: 10.1161/CIRCEP.115.003419. [DOI] [PubMed] [Google Scholar]
- 8.Lazzerini PE, Capecchi PL, El-Sherif N, Laghi-Pasini F, Boutjdir M. Emerging arrhythmic risk of autoimmune and inflammatory cardiac channelopathies. J Am Heart Assoc. 2018;7(22):e010595. doi: 10.1161/JAHA.118.010595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schächtele S, Tümena T, Gaßmann KG, Fromm MF, Maas R. Co-Prescription of QT-interval prolonging drugs: an analysis in a large cohort of geriatric patients. PLoS One. 2016;11(5):e0155649. doi: 10.1371/journal.pone.0155649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Moreno-Gutiérrez PA, Gaviria-Mendoza A, Cañón MM, Machado-Alba JE. High prevalence of risk factors in elderly patients using drugs associated with acquired torsades de pointes chronically in Colombia. Br J Clin Pharmacol. 2016;82(2):504-11. [DOI] [PMC free article] [PubMed]
- 11.Pasquier M, Pantet O, Hugli O, Pruvot E, Buclin T, Waeber G, et al. Prevalence and determinants of QT interval prolongation in medical inpatients. Intern Med J. 2012;42(8):933–940. doi: 10.1111/j.1445-5994.2011.02447.x. [DOI] [PubMed] [Google Scholar]
- 12.Lubart E, Segal R, Yearovoi A, Fridenson A, Baumoehl Y, Leibovitz A. QT interval disturbances in hospitalized elderly patients. Isr Med Assoc J. 2009;11(3):147–150. [PubMed] [Google Scholar]
- 13.Seftchick MW, Adler PH, Hsieh M, Wolfson AB, Chan ST, Webster BW, et al. The prevalence and factors associated with QTc prolongation among emergency department patients. Ann Emerg Med. 2009;54(6):763–768. doi: 10.1016/j.annemergmed.2009.03.021. [DOI] [PubMed] [Google Scholar]
- 14.Dumontet J, Malyuk R, Kiang G, Procyshyn RM. Corrected QT intervals in newly admitted geriatric psychiatric patients: an examination of risk factors. Can J Psychiatry. 2006;51(6):371–376. doi: 10.1177/070674370605100606. [DOI] [PubMed] [Google Scholar]
- 15.Golzari H, Dawson NV, Speroff T, Thomas C. Prolonged QTc intervals on admission electrocardiogra ms: prevalence and correspondence with admission electrolyte abnormalities. Conn Med. 2007;71(7):389–397. [PubMed] [Google Scholar]
- 16.Franchi C, Ardoino I, Rossio R, Nobili A, Biganzoli EM, Marengoni A, et al. Prevalence and risk factors associated with use of qt-prolonging drugs in hospitalized older people. Drugs Aging. 2016;33(1):53–61. doi: 10.1007/s40266-015-0337-y. [DOI] [PubMed] [Google Scholar]
- 17.Bazett HC. An analysis of the time-relations of electrocardiogra ms. Heart. 1920;7:35–70. [Google Scholar]
- 18.Straus SM, Kors JA, De Bruin ML, van der Hooft CS, Hofman A, Heeringa J, et al. Prolonged QTc interval and risk of sudden cardiac death in a population of older adults. J Am Coll Cardiol. 2006;47(2):362–367. doi: 10.1016/j.jacc.2005.08.067. [DOI] [PubMed] [Google Scholar]
- 19.Woosley R, Romero K. QTdrugs List, AZCERT, Inc. 1822 Innovation Park Dr., Oro Valley, AZ 85755. Available at http://www.Crediblemeds.org. [cited 2017; QTdrugs List, AZCERT, Inc. 1822 Innovation Park Dr., Oro Valley, AZ 85755].
- 20.Micromedex,® D. IBM Micromedex Drugdex®. Greenwood Village, CO: Truven Health Analytics. http://www.micromedexsolutions.com/. 2019.
- 21.Fernandes F, Silva E, Martins R, Oliveira A. QTc interval prolongation in critically ill patients: Prevalence, risk factors and associated medications. Plos One. 2018;13(6). [DOI] [PMC free article] [PubMed]
- 22.Tisdale JE, Wroblewski HA, Overholser BR, Kingery JR, Trujillo TN, Kovacs RJ. Prevalence of QT interval prolongation in patients admitted to cardiac care units and frequency of subsequent administration of QT interval-prolonging drugs: a prospective, observational study in a large urban academic medical center in the US. Drug Saf. 2012;35(6):459–470. doi: 10.2165/11598160-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 23.Curtis L, Ostbye T, Sendersky V, Hutchison S, Lapointe N, Al-Khatib S, et al. Prescription of QT-prolonging drugs in a cohort of about 5 million outpatients. Am J Med. 2003;114(2):135-41. [DOI] [PubMed]
- 24.van der Sijs H, Kowlesar R, Klootwijk A, Nelwan S, Vulto A, van Gelder T. Clinically relevant QTc prolongation due to overridden drug-drug interaction alerts: a retrospective cohort study. Br J Clin Pharmacol. 2009;67(3):347–54. [DOI] [PMC free article] [PubMed]
- 25.Roblek T, Vaupotic T, Mrhar A, Lainscak M. Drug-drug interaction software in clinical practice: a systematic review. Eur J Clin Pharmacol. 2015;71(2):131–42. [DOI] [PubMed]
- 26.Lengyel C, Varro A, Tabori K, Papp J, Baczko I. Combined pharmacological block of I-Kr and I-Ks increases short-term QT interval variability and provokes torsades de pointes. Br J Pharmacol. 2007;151(7):941–51. [DOI] [PMC free article] [PubMed]
- 27.Frommeyer G, Fischer C, Ellermann C, Dechering D, Kochhauser S, Lange P, et al. Additive proarrhythmic effect of combined treatment with QT-prolonging agents. Cardiovasc Toxicol. 2018;18(1):84–90. [DOI] [PubMed]
- 28.De Bruin M, Langendijk P, Koopmans R, Wilde A, Leufkens H, Hoes A. In-hospital cardiac arrest is associated with use of non-antiarrhythmic QTc-prolonging drugs. Br J Clin Pharmacol. 2007;63(2):216–23. [DOI] [PMC free article] [PubMed]
- 29.Vandael E, Vandenberk B, Vandenberghe J, Wille ms R, Foulon V. Cases of drug-induced Torsade de Pointes: a review of Belgian cases in the EudraVigilance database. Acta Clinica Belgica. 2017;72(6):385–90. [DOI] [PubMed]
- 30.Riad FS, Davis AM, Moranville MP, Beshai JF. Drug-induced QTc prolongation. Am J Cardiol. 2017;119(2):280–283. doi: 10.1016/j.amjcard.2016.09.041. [DOI] [PubMed] [Google Scholar]
- 31.Tisdale JE, Jaynes HA, Kingery JR, Mourad NA, Trujillo TN, Overholser BR, et al. Development and validation of a risk score to predict QT interval prolongation in hospitalized patients. Circ Cardiovasc Qual Outcomes. 2013;6(4):479–487. doi: 10.1161/CIRCOUTCOMES.113.000152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Danielsson B, Collin J, Nyman A, Bergendal A, Borg N, State M, et al. Drug use and torsade de pointes cardiac arrhythmias in Sweden: a nationwide register-based cohort study. BMJ Open. 2020;10:e034560. doi: 10.1136/bmjopen-2019-034560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lazzerini PE, Bertolozzi I, Finizola F, Acampa M, Natale M, Vanni F, et al. Proton pump inhibitors and serum magnesium levels in patients with torsades de pointes. Front Pharmacol. 2018;9:363. doi: 10.3389/fphar.2018.00363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chrysant S. Proton pump inhibitor-induced hypomagnesemia complicated with serious cardiac arrhythmias. Expert Rev Cardiovasc Ther. 2019;17(5):345–51. [DOI] [PubMed]
- 35.Lorberbaum T, Sampson KJ, Chang JB, Iyer V, Woosley RL, Kass RS, et al. Coupling data mining and laboratory experiments to discover drug interactions causing QT prolongation. J Am Coll Cardiol. 2016;68(16):1756–1764. doi: 10.1016/j.jacc.2016.07.761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lazzerini P, Bertolozzi I, Rossi M, Capecchi P, Laghi-Pasini F. Combination therapy with ceftriaxone and lansoprazole, acquired long QT syndrome, and torsades de pointes risk. J Am Coll Cardiol. 2017;69(14):1876–7. [DOI] [PubMed]
- 37.Batuwitage B, Kingham J, Morgan N, Bartlett R. Inappropriate prescribing of proton pump inhibitors in primary care. Postgrad Med J. 2007;83(975):66–8. [DOI] [PMC free article] [PubMed]
- 38.Heidelbaugh J, Metz D, Yang Y. Proton pump inhibitors: are they overutilised in clinical practice and do they pose significant risk? Int J Clin Pract. 2012;66(6):582–91. [DOI] [PubMed]
- 39.Gjestad C, Westin A, Skogvoll E, Spigset O. Effect of proton pump inhibitors on the serum concentrations of the selective serotonin reuptake inhibitors citalopram, escitalopram, and sertraline. Therap Drug Monit. 2015;37(1):90–7. [DOI] [PMC free article] [PubMed]
- 40.Beach S, Kostis W, Celano C, Januzzi J, Ruskin J, Noseworthy P, et al. Meta-analysis of selective serotonin reuptake inhibitor-associated QTc prolongation. J Clin Psychiatry. 2014;75(5):E441–E9. [DOI] [PubMed]
- 41.FDA Drug Safety Communication. Celexa (citalopram hydrobromide): abnormal heart rhyth ms associated with high doses. August 24, 2011.
- 42.Lubart E, Segal R, Megid S, Yarovoy A, Leibovitz A. QT interval disturbances in elderly residents of long-term care facilities. Isr Med Assoc J. 2012;14(4):244–246. [PubMed] [Google Scholar]
- 43.Rautaharju PM, Mason JW, Akiyama T. New age- and sex-specific criteria for QT prolongation based on rate correction formulas that minimize bias at the upper normal limits. Int J Cardiol. 2014;174(3):535–540. doi: 10.1016/j.ijcard.2014.04.133. [DOI] [PubMed] [Google Scholar]
- 44.Rautaharju PM, Zhou S, Wong S, Calhoun H, Berenson G, Prineas R, et al. Sex-differences in the evolution of the electrocardiographic qt interval with age. Can J Cardiol. 1992;8(7):690–5. [PubMed]
- 45.Salem J, Alexandre J, Bachelot A, Funck-Brentano C. Influence of steroid hormones on ventricular repolarization. Pharmacol Therap. 2016;167:38–47. [DOI] [PubMed]
- 46.van Noord C, Dörr M, Sturkenboom MC, Straus SM, Reffelmann T, Felix SB, et al. The association of serum testosterone levels and ventricular repolarization. Eur J Epidemiol. 2010;25(1):21–28. doi: 10.1007/s10654-009-9406-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bidoggia H, Maciel J, Capalozza N, Mosca S, Blaksley E, Valverde E, et al. Sex differences on the electrocardiographic pattern of cardiac repolarization: Possible role of testosterone. Am Heart J. 2000;140(4):678–83. [DOI] [PubMed]
- 48.Zhang Y, Ouyang P, Post W, Dalal D, Vaidya D, Blasco-Colmenares E, et al. Sex-steroid hormones and electrocardiographic QT-interval duration: findings from the third national health and nutrition examination survey and the multi-ethnic study of atherosclerosis. Am J Epidemiol. 2011;174(4):403–11. [DOI] [PMC free article] [PubMed]
- 49.Barber M, Nguyen LS, Wassermann J, Spano JP, Funck-Brentano C, Salem JE. Cardiac arrhythmia considerations of hormone cancer therapies. Cardiovasc Res. 2019;115(5):878–894. doi: 10.1093/cvr/cvz020. [DOI] [PubMed] [Google Scholar]
- 50.Klotz L, Boccon-Gibod L, Shore ND, Andreou C, Persson BE, Cantor P, et al. The efficacy and safety of degarelix: a 12-month, comparative, randomized, open-label, parallel-group phase III study in patients with prostate cancer. BJU Int. 2008;102(11):1531–1538. doi: 10.1111/j.1464-410X.2008.08183.x. [DOI] [PubMed] [Google Scholar]
- 51.Smith MR, Klotz L, Persson BE, Olesen TK, Wilde AA. Cardiovascular safety of degarelix: results from a 12-month, comparative, randomized, open label, parallel group phase III trial in patients with prostate cancer. J Urol. 2010;184(6):2313–2319. doi: 10.1016/j.juro.2010.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tolcher AW, Chi KN, Shore ND, Pili R, Molina A, Acharya M, et al. Effect of abiraterone acetate plus prednisone on the QT interval in patients with metastatic castration-resistant prostate cancer. Cancer Chemother Pharmacol. 2012;70(2):305–313. doi: 10.1007/s00280-012-1916-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Gagliano-Jucá T, Travison TG, Kantoff PW, Nguyen PL, Taplin ME, Kibel AS, et al. Androgen deprivation therapy is associated with prolongation of QTc interval in men with prostate cancer. J Endocr Soc. 2018;2(5):485–496. doi: 10.1210/js.2018-00039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Lazzerini PE, Bertolozzi I, Acampa M, Cantara S, Castagna MG, Pieragnoli L, et al. Androgen deprivation therapy for prostatic cancer in patients with Torsades des Pointes. Front Pharmacol. 2020;11:684. doi: 10.3389/fphar.2020.00684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Salem JE, Yang T, Moslehi JJ, Waintraub X, Gandjbakhch E, Bachelot A, et al. Androgenic effects on ventricular repolarization: a translational study from the international pharmacovigilance database to iPSC-cardiomyocytes. Circulation. 2019;140(13):1070–1080. doi: 10.1161/CIRCULATIONAHA.119.040162. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.