Abstract
Background
Two-dimensional (2D) specimen radiography (SR) and tomosynthesis (DBT) for breast cancer yield data that lack high-depth resolution. A volumetric specimen imager (VSI) was developed to provide full-3D and thin-slice cross-sectional visualization at a 360° view angle. The purpose of this prospective trial was to compare VSI, 2D SR, and DBT interpretation of lumpectomy margin status with the final pathologic margin status of breast lumpectomy specimens.
Methods
The study enrolled 200 cases from two institutions. After standard imaging and interpretation was performed, the main lumpectomy specimen was imaged with the VSI device. Image interpretation was performed by three radiologists after surgery based on VSI, 2D SR, and DBT. A receiver operating characteristic (ROC) curve was created for each method. The area under the curve (AUC) was computed to characterize the performance of the imaging method interpreted by each user.
Results
From 200 lesions, 1200 margins were interpreted. The AUC values of VSI for the three radiologists were respectively 0.91, 0.90, and 0.94, showing relative improvement over the AUCs of 2D SR by 54%, 13%, and 40% and DBT by 32% and 11%, respectively. The VSI has sensitivity ranging from 91 to 94%, specificity ranging from 81 to 85%, a positive predictive value ranging from 25 to 30%, and a negative predicative value of 99%.
Conclusions
The ROC curves of the VSI were higher than those of the other specimen imaging methods. Full-3D specimen imaging can improve the correlation between the main lumpectomy specimen margin status and surgical pathology. The findings from this study suggest that using the VSI device for intraoperative margin assessment could further reduce the re-excision rates for women with malignant disease.
When a lumpectomy is performed for invasive breast cancer (IBC) or ductal carcinoma in situ (DCIS), it is important to obtain negative margins to reduce the risk of local recurrence.1–4 The surgeon must balance removing the tumor completely and excising enough normal tissue to achieve a good oncologic and cosmetic result. A re-excision lumpectomy may be recommended if positive margins are identified on the final surgical pathology. A second procedure increases the risk of complications, poor cosmetic outcome, conversion to mastectomy, and increased cost.5–7
Intraoperative margin assessment has been shown to reduce re-excision rates.8,9 Specimen radiography (SR) is one of the most common methods used by surgeons to assess intraoperative margin status.10 Significant progress has been made since 2014 in reducing re-excision rates.11 However, re-excision rates remain above the 10% goal set by the American Society of Breast Surgeons.12 Further innovations in intraoperative margin assessment are needed to achieve this goal.
Intraoperative specimen radiography is routinely performed using a standard two-dimensional (2D) mammography unit or a digital breast tomosynthesis unit (DBT). The DBT generates multiple projection images by rotating the x-ray source and detector over an arc (e.g., 30°), which then are reconstructed into a series of parallel image slices, typically 1 mm thick.8,13
Portable 2D SR and DBT specimen imaging units also are available for use in the operating room (OR), which allow the surgeon to maintain specimen orientation and interpret the specimen radiograph with or without the input of a radiologist.14,15 However, both 2D SR and DBT imaging lack true 3D resolution, so even with meticulous orientation by the surgeon, these imaging methods are limited in their ability to identify close or positive margins. A recent meta-analysis found that 2D SR has a sensitivity of 53% and a specificity of 86% , and that DBT has a sensitivity of 74% and a specificity of 91%.8,9,16
To address these limitations, volumetric specimen imaging (VSI) was developed to provide intraoperative full-3D imaging of surgical specimens. The VSI method builds on micro-computed tomography (MCT) technology, developed previously for non-medical use.17
More recently, the feasibility of using MCT to image breast specimens and evaluate margin status has been studied.18–20 However, MCT systems have a number of technical hurdles that limit their clinical applicability.17–20 In contrast, VSI uses a novel scanning configuration and an accelerated iterative reconstruction algorithm to synthesize a 3D volumetric image data set of isotropic resolution digitally.21 The 3D data set can be digitally sliced for detailed visualization of tumor anatomy along three orthogonal (axial, sagittal, and coronal) planes, or the entire volume can be rendered for visualization of the entire specimen at any angle for precise tracking of specimen orientation in 3D.
To determine whether VSI could be used for intraoperative margin asssessment in breast cancer, this study aimed to compare the accuracy of 2D SR, DBT, and VSI in identifying positive margins in breast lumpectomy specimens.
Materials and Methods
IRB, Sites, and Inclusion/Exclusion Criteria
Specimen imaging using VSI was performed between 2016 and 2020 at site 1 of Northwestern University (NU) and site 2 of the University of Chicago (UC). Institutional review board (IRB) approval was obtained at both institutions.
We collected VSI images from 379 female patients ages 18–90 years who were undergoing localized lumpetomy (wire, radioactive seed, and SAVI SCOUT) for both benign and malignant lesions. For the patients with a known maligancy at the time of lumpectomy, full cavity-shave margins were taken by all four surgeons at NU. The surgeons at UC took directed cavity-shave margins. Pathologic margin assessment of the lumpectomy specimen and the shave margins was performed on formalin fixed paraffin-embedded tissue at both sites. At NU, an additional consent was required for image-resected breast tissue and for collection of de-identified clinical data.
At NU, 100 patients consented to participate in the study. Three patients decided not to participate after signing the consent, and three patients had their surgery rescheduled due to the COVID-19 pandemic. Consequently, 94 patients had their main breast lumpectomy specimens imaged. Of these 94 patients, 12 had benign disease (fibroadenomas, atypical ductal hyperplasia, radial scar, and papilloma). Under the UC IRB for surgical specimen imaging, no consent was required to image the specimen after standard-of-care (SOC) surgery and imaging.
At UC, 279 women had their main lumpectomy specimen imaged. The patients undergoing neoadjuvant chemotherapy and those with previous breast surgery also were eligible to participate.
Between the two insitutions, seven breast surgeons and two breast pathologists participated. Three fellowship-trained breast radiologists (2 at NU and 1 at UC) served as readers for the study. All the radiologists had at least 6 years of experience. The study analyzed 200 cases of invasive breast cancer, DCIS, and pleomorphic lobular carcinoma in situ (pLCIS) (82 of 94 cases at NU and the most recent consecutive 118 of 279 cases at UC).
Standard of Care, 2D SR and DBT Image Acquisition
At both sites, SOC images were acquired during surgery. The SOC images were interpreted immediately by the radiologists at both sites. At NU, after resection of the main lumpectomy specimen and placement of orientation markings by the surgeon, SOC images were acquired using the mammography mode of a DBT unit (Dimensions; Hologic Inc, Marlborough, MA, USA). Two orthogonal x-ray images were acquired by manual rotation of the main lumpectomy specimen approximately 90°.
A DBT scan also was performed immediately after the 2D imaging, but the DBT image was not used for clinical decision-making. Both the 2D and DBT images were obtained with the specimen compressed according to the device’s standard imaging protocol. At UC, the SOC image was acquired using a portable 2D SR system (Biovision; Faxitron Bioptics LLC, Tucson, AZ, USA) by the surgeon intraoperatively. Two orthogonal x-ray images were acquired by manual rotation of the specimen approximately 90°. The specimen was not compressed during the imaging according to the portable device’s standard imaging protocol.
VSI Image Acquisition
A prototype VSI system was installed in the Anatomic Pathology Gross Room at both sites. The main lumpectomy specimen was imaged without compression using the VSI system. Cavity-shave margins were not imaged by VSI. The VSI imaging of the main lumpectomy specimen did not have an impact on the SOC management of the patients. The specimen then was handed off to pathology for inking, gross evaluation, and placement in formalin.
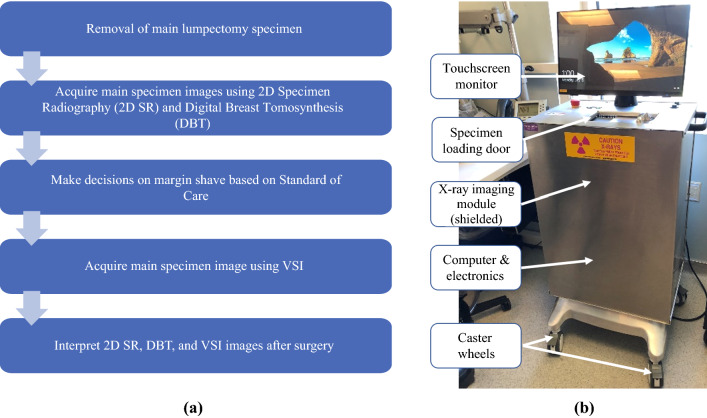
The total specimen scanning and imaging processing time with VSI was 5 to 7 min (Fig. 1). A two-step approach was used to achieve VSI orientation. When the VSI image was generated, it was saved without the actual specimen orientation (default orientation). The radiologists performed their margin assessment using this default image orientation. The default orientation then was converted manually to the actual specimen orientation based on the orientation marking system (sutures, clips, or staples) used by each of the surgeons who participated in the study.
Fig. 1.
a Workflow of data acquisition and b the volumetric specimen imager (VSI) prototype device
Image Interpretation After Surgery
At both sites, interpretation of the main lumpectomy specimen images for this study were performed after surgery. The breast imagers had preoperative images available for reivew. The intraoperative images were re-interpreted for comparison.
At NU, two breast radiologists independently reviewed the 2D SR, DBT, and VSI images acquired at NU. At UC, one breast radiologist independently reviewed the 2D SR and VSI images acquired at UC. Before reading the VSI images, the three radiologists were trained on three cases with known pathology results.
Reading of the 2D SR images required about 30 s, whereas reading of the DBT and VSI images required 1 to 2 min. No cross-reading occurred between the data sets from the two sites. After reviewing each image, the radiologist assigned a categorized percentage value (0%, 25%, 50%, 75%, or 100%) to each of the six (i.e., superior, inferior, anterior, posterior, medial, and lateral) margins based on the location of the lesion, clip, calcifications, and major spiculations. The percentage value corresponded to the likelihood of tumor or microcalcifications extending to the end of the specimen for each particular margin, as selected from the following: 100% (definitely positive), 75% (likely positive), 50% (neutral/equally likely), 25% (likely negative), or 0% (definitely negative). All the interpretations were performed with the radiologist blinded to the pathology results.
Data Analysis
The margin status data of the main lumpectomy specimen reported in the surgical pathology report were retrieved to serve as the gold standard for this study. The criteria for classifying a margin as positive follow the SSO-ASTRO society guidelines (i.e., ink on tumor for invasive carcinomas and less than 2 mm for in situ carcinomas).1,2 For quantitative measurement of the performance of each imaging method, receiver operating characteristic (ROC) analysis was performed for interpretation of the results by each reader. We also computed the sensitivity, specificity, positive predicting power, and negative predicting power.
In the ROC analysis, by comparing the reading with the pathology result for each margin, we computed the true-positive rate (TPR) and the false-positive rate (FPR) for each of the five thresholds (0%, 25%, 50%, 75%, and 100%). An ROC curve was obtained by plotting the TPR-FPR data pairs in a 2D chart, which corresponded to the given method and reader. The ROC-KIT software was used to perform curve-fitting with a semi-parametric, proper binormal model,22,23 and to compute the standard error and area under the curve (AUC).
Although ROC curves characterize the performance of imaging methods over the entire range (0–100%) of the decision threshold, we selected the “optimal” threshold that maximizes the Youden Index24 (i.e., the sum of sensitivity and specificity), and used the data corresponding to that threshold to compute the figures of merit on a per margin basis, including sensitivity (TPR), specificity (1-FPR), positive predicative value (PPV), and negative predictive value (NPV). Finally, at the VSI’s optimal threshold, we estimated its positive margin rate (PMR), defined as the percentage of cases that would have unaddressed positive margins on the main lumpectomy specimen had VSI been used intraoperatively for directing shave margins.
Results
Demographics and Tumor Characteristics
The analysis included 200 cases of IBC, DCIS, and pLCIS (82 cases from site 1 and in 118 cases from site 2). Invasive ductal carcinoma (IDC) was found in 133 patients (67%) including 13 patients (7%) with invasive lobular carcinoma (ILC), 52 patients (26%) with DCIS, and 2 patients (1%) with pLCIS. The distributions of cases at each insitution were similar. The median age of the patients participating in the study was 62 years (range, 30–87 years).
Visual Comparison of 2D SR, DBT, and VSI Images
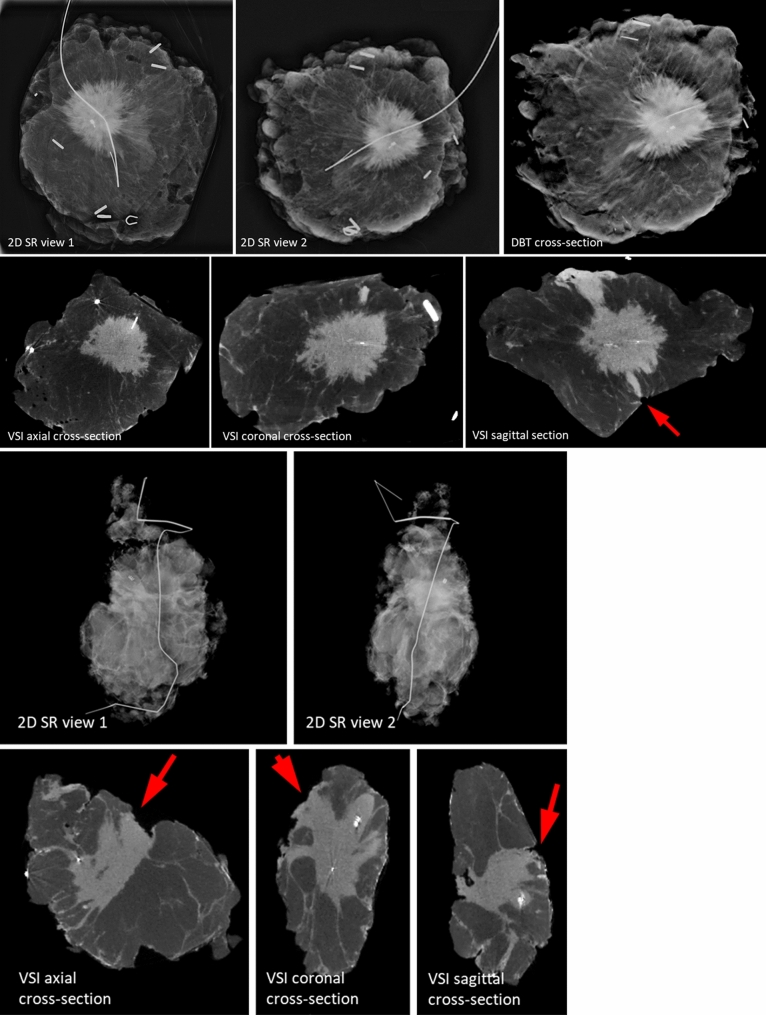
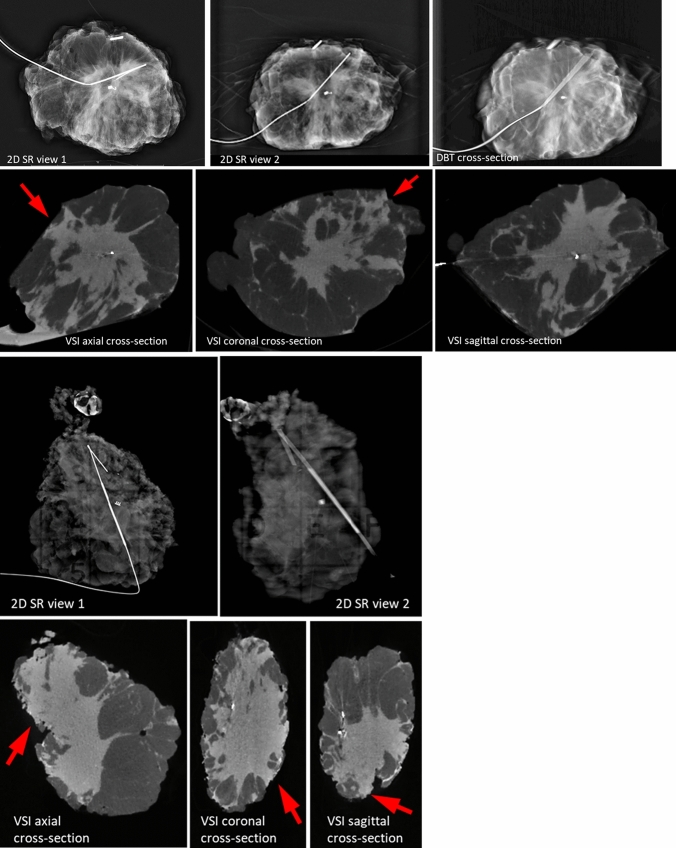
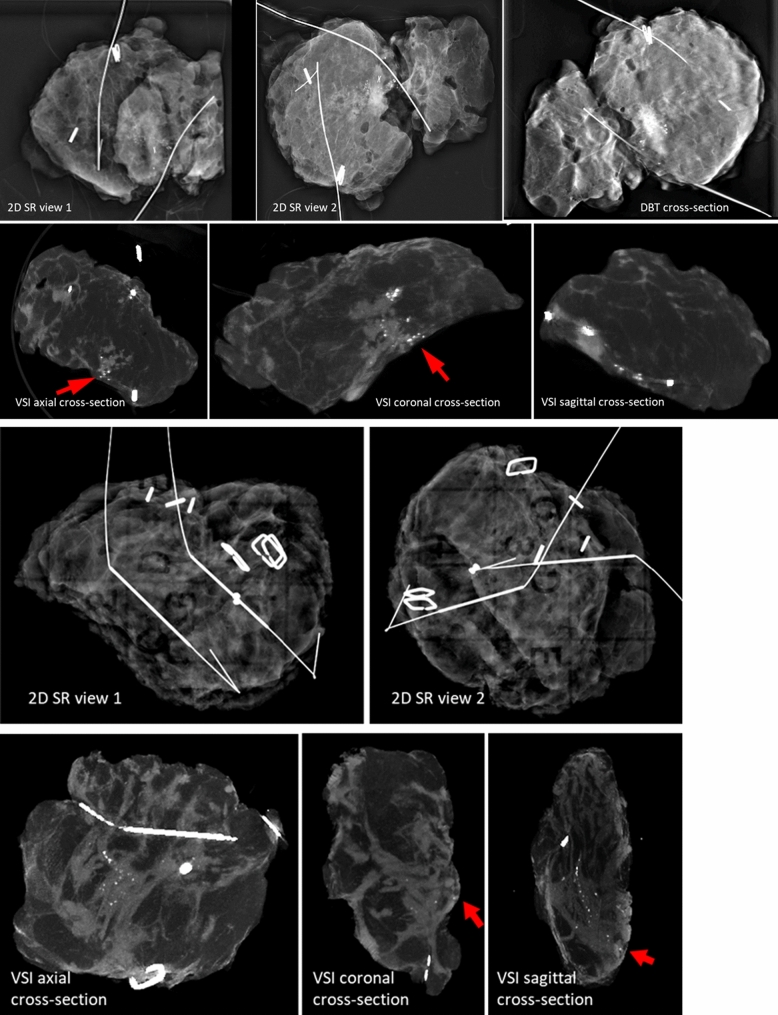
The images collected from the different histologies showed distinct features. Figures 2, 3 and 4display the VSI images from six patients (2 from each site) including two patients with IDC, two patients with ILC, and two patients with pure DCIS. To represent the 3D dataset, three representative cross-sections (axial, sagittal, and coronal) are displayed, showing the tumor or micro-calcifications at or close to the margin. As a reference, we display 2D SR (2 orthogonal views) and DBT (1 representative slice showing tumor/micro-calcifications) images for the cases collected at site 1, and two orthogonal 2D SR images for the cases collected at site 2.
Fig. 2.
Images of two invasive ductal carcinoma cases. Arrows indicate positive margins identified in volumetric specimen imager (VSI) images
Fig. 3.
Images of two invasive lobular carcinoma cases. Arrows indicate positive margins identified in volumetric specimen imager (VSI) images.
Fig. 4.
Images of two ductal carcinoma in situ (DCIS) cases. Arrows indicate positive margins identified in volumetric specimen imager (VSI) images
Invasive Ductal Carcinoma
Case 1 (Fig. 2a) involved a 71-year-old white female with a 2.8-cm IDC grade 2 tumor. Although the posterior margin did not appear to be concerning on the 2D SR and DBT images, VSI identified a positive posterior margin, which was consistent with the patient’s final surgical pathology margin status.
Case 2 (Fig. 2b) involved a 73-year-old black female with a 2.2-cm grade 2 IDC. The 2D SR images were interpreted correctly as showing a positive inferior margin. The inferior margin and an additional positive posterior margin were identifed byVSI, consistent with the final pathology.
Invasive Lobular Carcinoma
Case 1 (Fig. 3a) involved a 55-year-old white female with a 5.9-cm grade 2 ILC. It was challenging to identify the extension of the tumor from the 2D SR and DBT images, but VSI was able to demonstrate the ILC extending to the lateral, anterior, and posterior margins, consistent with the final pathology.
Case 2 (Fig. 3b) involved a 63-year-old black female with a 3.5-cm grade 2 ILC. Evaluation of the 2D SR images found the posterior margin to be positive. Additional positive superior, inferior, lateral, and anterior margins were identified by VSI, consistent with the final pathology.
DCIS
Case 1 (Fig. 4a) involved a 72-year-old white female with a 3.6-cm grade 3 DCIS. This case illustrated the challenges of overlapping dense tissue and an irregular specimen with two wires that obscure calcifications at the margins. However, VSI clearly identified calcifications at the posterior margin, which was confirmed on the final pathology.
Case 2 (Fig. 4b) involved a 43-year-old black female with a 3.5-cm grade 3 DCIS. The 2D SR reading missed the positive superior margin identified on surgical pathology. This is visible in the VSI cross-sectional images.
Comparison Between 2D SR, DBT, and VSI for Identifying Positive Margins
Among the 82 cases at site 1, 24 (29.3%) had positive pathologic margins on the main lumpectomy specimen, and these 24 cases included 32 positive margins. Among the 118 cases at site 2, 33 (28.0%) had positive pathologic margins on the main lumpectomy specimen, and there were 47 positive margins. Using the Youden index, 50% of the margins were indentified as having the optimal threshold value of VSI for all three readers. In other words, a margin is deemed to be positive when the likelihood is greater than or equal to 50%. Similarly, the optimal threshold value was 25% for 2D SR and DBT (readers 1 and 2) and 50% for SR (reader 3).
Table 2 shows the comparison between 2D SR, DBT, and VSI for identifying the status of the 1200 margins from the 200 main lumpectomy specimens at both sites. The comparison was made per margin. At site 1, the sensitivity and specificity of reader 1 were respectively 34% and 78% for 2D SR, 53% and 71% for DBT, and 94% and 81% for VSI, whereas the sensitivity and specificity of reader 2 were respectively 34% and 88% for 2D SR, 38% and 88% for DBT, and 91% and 83% for VSI. At site 2, the sensitivity and specificity of reader 3 were respectively 49% and 81% for 2D SR and 92% and 85% for VSI. The PPV and NPV values are listed in Table 2.
Table 1.
Demographic data
| Site 1 n (%) |
Site 2 n (%) |
||
|---|---|---|---|
|
Patients Casesa |
81 82 |
117 118 |
|
| Mean age (years) | 61.8 ± 13.5 | 61.0 ± 13.0 | |
| Race |
White Black Hispanic Asian Not reported/refused |
52 (63.4) 9 (11.0) 0 (0.0) 7 (8.5) 14 (17.0) |
54 (46.8) 46 (40.0) 1(.8) 2 (1.7) 15 (12.7) |
| Mammographic density |
Entirely fatty Scattered fibroglandular Heterogeneously dense Extremely dense |
1 (1.2) 34 (41.4) 44 (53.7) 3 (3.7) |
3 (16.7) 65 (55.1) 46 (40.0) 4 (3) |
| Initial imaging finding |
Mass Mass and calcifications Calcifications Architectural distortion Asymmetry Other |
27 (33) 26 (32) 20 (24) 1 (1) 7 (9) 1 (1) |
61 (52) 4 (3) 39 (33) 3 (3) 11 (9) 0 (0) |
| Receipt of NAC |
Yes No |
4 (5.0) 78 (95.0) |
6 (5.0) 112 (95.0) |
| Histology |
IDC ILC DCIS pLCIS |
61 (74.4) 3 (3.6) 18 (22.0) 0 (0.0) |
72 (61) 10 (8.4) 34 (28.8) 2 (1.7) |
| Pathologic Margin status (%) |
Main lumpectomy specimen Final margin status (including cavity shaves) |
29.3 4.9 |
28.0 12.7 |
NAC neoadjuvant chemotherapy; IDC invasive ductal carcinoma; ILC invasive lobular carcinoma; DCIS ductal carcinoma in situ; pLCIS pleomorphic lobular carcinoma in situ
aEach site had a patient with a bilateral breast cancer.
Table 2.
Comparison of 2D SR, DBT, and VSI on a per margin basis
| Site 1 (reader 1/reader 2) | Site 2 (reader 3) | |||||||
|---|---|---|---|---|---|---|---|---|
| Path+Image+ | Path–Image– | Path–Image+ | Path+Image– | Path+Image+ | Path–Image– | Path–Image+ | Path+Image– | |
| 2D SR | 11/11 | 358/406 | 102/54 | 21/21 | 23 | 538 | 123 | 24 |
| DBT | 17/12 | 327/403 | 133/57 | 15/20 | N/A | N/A | N/A | N/A |
| VSI | 30/29 | 372/382 | 88/78 | 2/3 | 43 | 559 | 102 | 4 |
| Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | |
|---|---|---|---|---|---|---|---|---|
| 2D SR | 34/34 | 78/88 | 10/17 | 95/95 | 49 | 81 | 16 | 96 |
| DBT | 53/38 | 71/88 | 11/17 | 96/96 | N/A | N/A | N/A | N/A |
| VSI | 94/91 | 81/83 | 25/27 | 99/99 | 91 | 85 | 30 | 99 |
2D SR, two-dimentional specimen radiography (2D mammography for site 1 and portable 2D specimen x-ray for site 2) taken at two orthogonal views; DBT, digital breast tomosynthesis; VSI, volumetric specimen imaging; Path+, final pathology margin status positive; Path–, final pathology margin status negative; Image+, reader-interpreted margin status postive; Image–, reader-interpreted margin status negative; N/A,; PPV, positive predictive value; NPV, negative predictive value
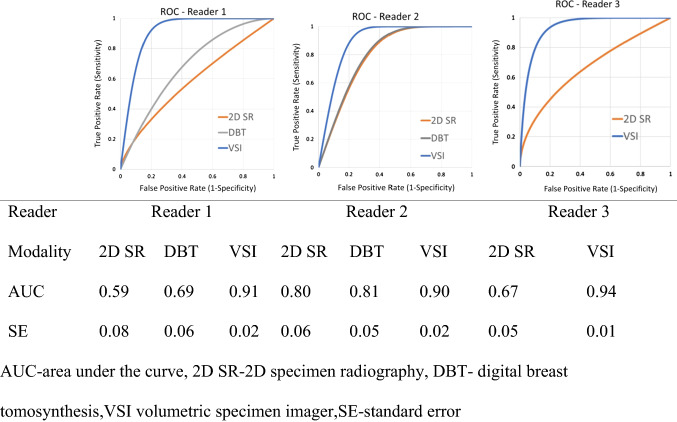
ROC Characterization of the Readers’ Interpretations
Table 3 shows the ROC curves characterizing the interpretation by the three readers from 2D SR, DBT and VSI. For reader 1, the AUC values were 0.59 for 2D SR (standard error [SE], 0.08), 0.69 for DBT (SE, 0.06), and 0.91 for VSI (SE, 0.02). For reader 2, the AUC values were 0.80 for 2D SR (SE, 0.06), 0.81 for DBT (SE, 0.05), and 0.90 for VSI (SE, 0.02). For reader 3, the AUC values were 0.67 for 2D SR (SE, 0.05) and 0.94 for VSI (SE, 0.01).
Table 3.
ROC curves, AUC, and standard error (SE) of the three readers
ROC receiver operating characteristic; AUC area under the curve; 2D SR two-dimensional (2D) specimen radiography; DBT digital breast tomosynthesis; VSI volumetric specimen imager
Positive Margin Rate (PMR) Per Case
Operating at the optimal threshold, VSI missed two positive-margin cases by reader 1 and three positive margins by reader 2, leading to estimated PMR values of 2.4% and 3.7%, respectively. Similarly, at site 2, VSI missed four positive-margin cases, leading to an estimated PMR of 3.4%.
Discussion
This multicenter study of 200 cases showed that VSI is superior to both 2D SR and DBT in identifying positive pathologic margins on lumpectomy specimens. Compared with 2D SR and DBT, VSI images are able to show more detailed anatomic features of the tumors to facilitate determining the extent of the tumor from the image data.
In IDC, the extension of the tumor mass is clearly visualized because the breast tissues in the overlying image slices are digitally removed by VSI, revealing the fine details in the selected cross-sectional slice. In addition, because VSI allows 360° orientation of the volume, the positive or close margin can be precisely located on the surface of the 3D rendering of the specimen.
For patients with ILC, VSI technology has the potential to be particularly useful based on the 13 cases we reviewed. Because of its diffuse nature, ILC is difficult to visualize on 2D SR and DBT. Using VSI, we were able to distinctly visualize the extent of the tumor and identify multiple additional positive margins. Additional ILC cases will need to be reviewed to determine whether our experience can be generalized to all ILC cases.
With respect to DCIS, which has a higher re-excision rate than invasive cancer, micro-calcifications are more easily visualized using VSI because the digital removal of overlying slices can improve image contrast.25 We compared the sensitivity, specificity, and AUC of VSI in detecting pure DCIS and IBC cases and found them to be similar (data not shown). However, because the readers missed only a small number of postive margin cases, we could not draw any difinitive conclusions based on these results.
We also included 10 patients who underwent neoadjuvant chemotherapy as part of our study, with variable residual radiographic findings. However, similarly, due to the limited number of cases in our data, we could not assertain the utility of VSI for this patient population.
Overall, the sensitivity and specificity for identifying positive margins were consistantly high among our three readers. With a sensitivity of 91–94% and a specificity of 81–85% for all three readers, the ROC curves corresponding to VSI were significantly higher than for the 2D SR and DBT methods, with the AUC values above 0.9. This also suggests that VSI can help identify more true-positive margins while not increasing false-positives.
Ultimately, the goal is to use VSI intraoperatively to identify close or postive margins that could be resected during the initial operation, thereby reducing re-excision rates. Our PMR rate was lower than 4%, below the goal of the American Society of Breast Surgeons. However, this is just an estimate, and the actual false-negative rate will be reported in our prospective phase 2/3 trial, which currently is under development.
The 2D SR method, currently the most widely used procedure for intraoperative specimen imaging, originally was used to confirm the targeted lesion in the specimen. But because it is a 2D image, 2D SR is not ideally suited to assess margin status, and the addition of orthoginal views to improve specificity has not been demonstrated consistently.10 The DBT method, orginally developed for radiologic screening and diagnosis of breast diseases, does provide additional information along the depth dimension over 2D imaging.16 However, it lacks true 3D visualization because its depth resolution (~1 mm) is significantly inferior to the in-plane resolution.10
In contrast, VSI has a number of advantages over currently available intraoperative imaging methods. It requires no manual rotation of the specimen. It provides true 3D image visualization of isotropic resolution at any angle or plane, and it has the ability to digitally remove the breast tissues within overlying image slices to reveal details of the tumor mass and micro-calcifications.6,26 Furthermore, VSI allows for orientation of the specimen in 3D, which is crucial for locating the exact positive/close margins.
The VSI technology is based on previously described MCT systems. These devices were orginally designed for material science and pre-clinical research purposes. Their clinical applicability is limited by technical hurdles including a small field of view, slow image acquisition, prominent image artifacts from metal objects such as clips and wires, and prolonged image analysis time. The hardware and software of VSI are optimized specifically for intraoperative application and workflow, which include a number of important advantages over conventional MCT such as a large field of view, rapid scanning, minimized image artifacts, and intuitive image visualization.
In addition to the x-ray imaging methods investigated, radiofrequency spectroscopy, magnetic resonance, optical fluorescence imaging, and optical coherence tomography (OCT) have been developed to assess intraoperative margin status. Overall, to date, these methods offer a modest increase in sensitivity but have a lower specificity.27–30 Intraoperative pathology analysis in the form of gross specimen analysis or frozen section is used as routine practice at a small number of high-volume institutions to assess margin status.8,31,32 Studies have shown that intraoperative frozen section pathology assessment yields highly accurate results comparable with those of permanent pathology and significantly reduces re-excision rates.31,32 However, it requires dedicated pathology resources and potentially prolonged operating room time and may not be feasible at many insititutions.
The main limitation of this study was that we did not measure the impact of VSI on surgical decision-making, and therefore could not measure its impact on re-excision rate, which is the true intended benefit of intraoperative margin assessment. Another limitation was that each institution used different intraoperative imaging techniques and workflows for intraoperative specimen imaging. However, this can be viewed positively from the perspective of demonstrating that VSI compared favorably with more than imaging technique and method. Finally, our scanning time was 5 to 7 min using the prototype, which is longer than the time required for current intraoperative specimen technology. The next-generation VSI devices in development have a shorter scanning time for better integration into the operating room environment.
Conclusions
Full-3D specimen imaging can improve correlation between the main lumpectomy specimen margin status and the final pathology results. The ROC curves of VSI are considerably higher than those of other specimen imaging methods under comparison. The findings from this study suggest that using the VSI device to perform intraoperative margin assessment in conjunction with shave-margin techniques could further reduce the re-excision rates for women undergoing lumpectomy for breast cancer.
Disclosure
Christian Wietholt, Dimple Modgil, David Lester, Bidur Bohara, and Xiao Han are full-time employees of Clarix Imaging Corporation. Li Lan is a part-time employee of Clarix Imaging Corporation. Xiaochuan Pan is a founder of Clarix Imaging Corporation.
Footnotes
Selected and presented as an oral abstract at the American Society of Breast Surgeons Annual Meeting, 2 May 2021.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Moran MS, Schnitt SJ, Giuliano AE, et al. Society of Surgical Oncology-American Society for Radiation Oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in stages I and II invasive breast cancer. Int J Radiat Oncol Biol Phys. 2014;88:553–564. doi: 10.1016/j.ijrobp.2013.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Morrow M, Van Zee KJ, Solin LJ, et al. Society of Surgical Oncology-American Society for Radiation Oncology-American Society of Clinical Oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in ductal carcinoma in situ. Ann Surg Oncol. 2016;23:3801–3810. doi: 10.1245/s10434-016-5449-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Marinovich ML, Azizi L, Macaskill P, et al. The association of surgical margins and local recurrence in women with ductal carcinoma in situ treated with breast-conserving therapy: a meta-analysis. Ann Surg Oncol. 2016;23:3811–3821. doi: 10.1245/s10434-016-5446-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Houssami N, Macaskill P, Marinovich ML, Morrow M. The association of surgical margins and local recurrence in women with early-stage invasive breast cancer treated with breast-conserving therapy: a meta-analysis. Ann Surg Oncol. 2014;21:717–730. doi: 10.1245/s10434-014-3480-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wazer DE, DiPetrillo T, Schmidt-Ullrich R, et al. Factors influencing cosmetic outcome and complication risk after conservative surgery and radiotherapy for early-stage breast carcinoma. J Clin Oncol. 1992;10:356–363. doi: 10.1200/JCO.1992.10.3.356. [DOI] [PubMed] [Google Scholar]
- 6.Cabioglu N, Hunt KK, Sahin AA, et al. Role for intraoperative margin assessment in patients undergoing breast-conserving surgery. Ann Surg Oncol. 2007;14:1458–1471. doi: 10.1245/s10434-006-9236-0. [DOI] [PubMed] [Google Scholar]
- 7.Olsen MA, Nickel KB, Margenthaler JA, et al. Increased risk of surgical-site infection among breast-conserving surgery re-excisions. Ann Surg Oncol. 2015;22:2003–2009. doi: 10.1245/s10434-014-4200-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Park KU, Kuerer HM, Rauch GM, et al. Digital breast tomosynthesis for intraoperative margin assessment during breast-conserving surgery. Ann Surg Oncol. 2019;26:1720–1728. doi: 10.1245/s10434-019-07226-w. [DOI] [PubMed] [Google Scholar]
- 9.St John ER, Al-Khudairi R, Ashrafian H, et al. Diagnostic accuracy of intraoperative techniques for margin assessment in breast cancer surgery: a meta-analysis. Ann Surg. 2017;265:300–310. doi: 10.1097/SLA.0000000000001897. [DOI] [PubMed] [Google Scholar]
- 10.Reyna C, DeSnyder SM. Intraoperative margin assessment in breast cancer management. Surg Oncol Clin North Am. 2018;27:155–165. doi: 10.1016/j.soc.2017.08.006. [DOI] [PubMed] [Google Scholar]
- 11.McEvoy MP, Landercasper J, Naik HR, Feldman S. Update of the American Society of Breast Surgeons Toolbox to address the lumpectomy reoperation epidemic. Gland Surg. 2018;7:536–553. doi: 10.21037/gs.2018.11.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Havel L, Naik H, Ramirez L, et al. Impact of the SSO-ASTRO margin guideline on rates of re-excision after lumpectomy for breast cancer: a meta-analysis. Ann Surg Oncol. 2019;26:1238–1244. doi: 10.1245/s10434-019-07247-5. [DOI] [PubMed] [Google Scholar]
- 13.Eghtedari M, Tsai C, Robles J, et al. Tomosynthesis in breast cancer imaging: how does it fit into preoperative evaluation and surveillance? Surg Oncol Clin North Am. 2018;27:33–49. doi: 10.1016/j.soc.2017.07.004. [DOI] [PubMed] [Google Scholar]
- 14.Maloney BW, McClatchy DM, Pogue BW, et al. Review of methods for intraoperative margin detection for breast-conserving surgery. J Biomed Opt. 2018;23:1–19. doi: 10.1117/1.JBO.23.10.100901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kornfeld HW, Mulder LL, Spivey TL, et al. The temporal and financial benefit of intraoperative breast specimen imaging: a pilot study of the Kubtec MOZART. Breast J. 2019;25:766–768. doi: 10.1111/tbj.13337. [DOI] [PubMed] [Google Scholar]
- 16.Partain N, Calvo C, Mokdad A, et al. Differences in re-excision rates for breast-conserving surgery using intraoperative 2D versus 3D tomosynthesis specimen radiograph. Ann Surg Oncol. 2020;27:4767–4776. doi: 10.1245/s10434-020-08877-w. [DOI] [PubMed] [Google Scholar]
- 17.Ritman EL. Current status of developments and applications of micro-CT. Annu Rev Biomed Eng. 2011;13:531–552. doi: 10.1146/annurev-bioeng-071910-124717. [DOI] [PubMed] [Google Scholar]
- 18.DiCorpo D, Tiwari A, Tang R, et al. The role of micro-CT in imaging breast cancer specimens. Breast Cancer Res Treat. 2020;180:343–357. doi: 10.1007/s10549-020-05547-z. [DOI] [PubMed] [Google Scholar]
- 19.Tang R, Buckley JM, Fernandez L, et al. Micro-computed tomography (micro-CT): a novel approach for intraoperative breast cancer specimen imaging. Breast Cancer Res Treat. 2013;139:311–316. doi: 10.1007/s10549-013-2554-6. [DOI] [PubMed] [Google Scholar]
- 20.Tang R, Saksena M, Coopey SB, et al. Intraoperative micro-computed tomography (micro-CT): a novel method for determination of primary tumour dimensions in breast cancer specimens. Br J Radiol. 2016;89:20150581. doi: 10.1259/bjr.20150581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sidky EY, Pan X. Image reconstruction in circular cone-beam computed tomography by constrained, total-variation minimization. Phys Med Biol. 2008;53:4777–4807. doi: 10.1088/0031-9155/53/17/021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Metz CE. Basic principles of ROC analysis. Semin Nucl Med. 1978;8:283–298. doi: 10.1016/S0001-2998(78)80014-2. [DOI] [PubMed] [Google Scholar]
- 23.Pan X, Metz CE. The “proper” binormal model: parametric receiver operating characteristic curve estimation with degenerate data. Acad Radiol. 1997;4:380–389. doi: 10.1016/S1076-6332(97)80121-3. [DOI] [PubMed] [Google Scholar]
- 24.Fluss R, Faraggi D, Reiser B. Estimation of the Youden Index and its associated cutoff point. Biom J. 2005;47:458–472. doi: 10.1002/bimj.200410135. [DOI] [PubMed] [Google Scholar]
- 25.Howard-McNatt M, Dupont E, Tsangaris T, et al. Impact of cavity-shave margins on margin status in patients with pure ductal carcinoma in situ. J Am Coll Surg. 2021;232:373–378. doi: 10.1016/j.jamcollsurg.2020.11.019. [DOI] [PubMed] [Google Scholar]
- 26.Chagpar A, Yen T, Sahin A, et al. Intraoperative margin assessment reduces reexcision rates in patients with ductal carcinoma in situ treated with breast-conserving surgery. Am J Surg. 2003;186:371–377. doi: 10.1016/S0002-9610(03)00264-2. [DOI] [PubMed] [Google Scholar]
- 27.Zysk AM, Chen K, Gabrielson E, et al. Intraoperative assessment of final margins with a handheld optical imaging probe during breast-conserving surgery may reduce the reoperation rate: results of a multicenter study. Ann Surg Oncol. 2015;22:3356–3362. doi: 10.1245/s10434-015-4665-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schnabel F, Boolbol SK, Gittleman M, et al. A randomized prospective study of lumpectomy margin assessment with use of MarginProbe in patients with nonpalpable breast malignancies. Ann Surg Oncol. 2014;21:1589–1595. doi: 10.1245/s10434-014-3602-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Smith BL, Gadd MA, Lanahan CR, et al. Real-time, intraoperative detection of residual breast cancer in lumpectomy cavity walls using a novel cathepsin-activated fluorescent imaging system. Breast Cancer Res Treat. 2018;171:413–420. doi: 10.1007/s10549-018-4845-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Papa M, Allweis T, Karni T, et al. An intraoperative MRI system for margin assessment in breast-conserving surgery: initial results from a novel technique. J Surg Oncol. 2016;114:22–26. doi: 10.1002/jso.24246. [DOI] [PubMed] [Google Scholar]
- 31.Racz JM, Glasgow AE, Keeney GL, et al. Intraoperative pathologic margin analysis and re-excision to minimize reoperation for patients undergoing breast-conserving surgery. Ann Surg Oncol. 2020;27:5303–5311. doi: 10.1245/s10434-020-08785-z. [DOI] [PubMed] [Google Scholar]
- 32.Boughey JC, Keeney GL, Radensky P, et al. Economic implications of widespread expansion of frozen section margin analysis to guide surgical resection in women with breast cancer undergoing breast-conserving surgery. J Oncol Pract. 2016;12:e413–e422. doi: 10.1200/JOP.2015.005652. [DOI] [PubMed] [Google Scholar]