Abstract
DHA (docosahexaenoic acid) is an essential fatty acid that is required for the normal development and function of the brain. Because of its inability to synthesize adequate amounts of DHA from the precursors, the brain has to acquire DHA from plasma through the blood brain barrier (BBB). Recent studies demonstrated the presence of a transporter at the BBB that specifically transports DHA into the brain in the form of lysophosphatidylcholine (LPC-DHA). However, the mechanism by which LPC-DHA is generated in the plasma is not known. Our previous studies showed that there are at least three different enzymes - lecithin cholesterol acyltransferase (LCAT), endothelial lipase (EL), and hepatic lipase (HL), which can generate LPC-DHA from sn-2 DHA phosphatidylcholine. Here we determined the relative contributions of these enzymes in the delivery of DHA to the brain by measuring the brain DHA levels in the mice deficient in each of these enzymes. The results show that the brain DHA levels of LCAT-deficient mice or EL-deficient mice were not significantly lower than those of their littermates. However, brain DHA was significantly decreased in HL deficient mice (13.5% of total fatty acids) compared to their littermates (17.1%) (p<0.002), and further decreased to 8.3% of total fatty acids in mice deficient in both HL and EL. These results suggest that HL activity may be the major source for the generation of LPC-DHA in the plasma necessary for transport into the brain, and EL might contribute to this process in the absence of HL.
Keywords: DHA, brain, LPC-DHA, Hepatic lipase, endothelial lipase, LCAT
1. Introduction
Docosahexaenoic acid (DHA), which is highly concentrated in the brain and retina, is an essential omega-3 fatty acid that is required for the normal development and function of the brain and retina. The presence of DHA in the membranes helps maintain proper membrane fluidity in neuronal cells, which is important for the optimal functioning of membrane receptors and transporters, signal transduction and neurotransmission [1–3]. Furthermore, DHA counteracts the pro-inflammatory effects of arachidonic acid, and the metabolic products of DHA (resolvins, protectins, maresins) play critical roles in the resolution of inflammation [4]. Deficiency of DHA is associated with several neurodegenerative diseases such as Alzheimer’s disease, Parkinson’s disease, dementia, schizophrenia, amyotrophic lateral sclerosis and Huntington disease [1, 3, 5]. Because of its inability to synthesize adequate amounts of DHA from the precursors, the brain has to acquire DHA from the plasma through the blood brain barrier (BBB). Plasma DHA occurs in several molecular forms including phospholipids, triacylglycerols (TAG), and cholesteryl esters (CE) all of which are carried by various lipoproteins, and free (unesterified) DHA and lysophosphatidylcholine (LPC)-DHA which are primarily bound to albumin. Among these, only the albumin-bound forms (free DHA and LPC-DHA) [6, 7] are known to be transported through the blood brain barrier. Although previous studies suggested that free DHA is the predominant source of brain DHA [6], more recent studies support the view that LPC is the major carrier of DHA responsible for brain enrichment [8], because of the presence of a specific transporter in the BBB, Mfsd2a (major facilitator superfamily domain-containing protein a) which transports LPC-DHA but not free DHA [9]. Furthermore, the deficiency of this transporter results in defective brain development and impaired brain function in mice as well as in humans [10]. Our laboratory has demonstrated that dietary LPC-DHA enriches brain DHA efficiently, whereas dietary free DHA and triacylglycerol DHA at equivalent dose have virtually no effect on brain DHA content [8, 11]. Although the presence of LPC-DHA in the plasma has been reported by several studies [12–15], the mechanism by which it is formed is not known. Since LPC is not stored in the tissues, it is reasonable to assume that LPC-DHA is formed by the action of a phospholipase A1 (PLA1) in the tissues or plasma, because DHA is present exclusively at the sn-2 position of phospholipids. Our previous studies showed that there are three potential enzymes in plasma which can generate LPC-DHA. The first is LCAT, which normally transfers a fatty acid from the sn-2 position of PC to cholesterol, but alters its positional specificity in presence of 16:0-22:6 PC, and transfers 16:0 to cholesterol, generating LPC-DHA as byproduct [16, 17]. This enzyme is essential for the maturation of HDL in the plasma, and its deficiency results in the absence of mature HDL, increased free cholesterol/cholesteryl ester ratio, and impaired reverse cholesterol transport [18]. The second enzyme is endothelial lipase (EL) which exhibits strong PLA1 activity towards polyunsaturated PCs, and generates LPC-DHA [19, 20]. Unlike LCAT, EL activity is negatively correlated with plasma HDL levels, and its deficiency leads to increased HDL [21]. The third enzyme is hepatic lipase (HL) which hydrolyzes both TAG and phospholipids of the lipoproteins, preferably the intermediate density lipoproteins (IDL) and the HDL, and therefore HL deficiency leads to increased levels of HDL and IDL in the plasma [22]. The substrate preference of HL is intermediate between that of lipoprotein lipase (LPL) which is more specific for TAG, and of endothelial lipase which is more specific for phospholipids. It also generates LPC-DHA in plasma lipoproteins [23]., and probably in the liver. Based on these studies, we reasoned that if one of the three enzymes is essential for the generation of LPC-DHA and subsequent transport of DHA into the brain, its deficiency should result in a decrease of brain DHA levels. The results presented here show that hepatic lipase may be the most important enzyme for the generation of LPC-DHA required for the enrichment of brain DHA, although endothelial lipase may also contribute to a smaller extent.
2. Materials and Methods
2.1. Animals:
All animal procedures were carried out in accordance with the guidelines of NIH Guide for the care and use of laboratory animals, and were approved by the Institutional Animal Care and Use committee (Protocol numbers 11-049 and 14-046). The generation and characterization of LCAT-deficient animals has been described previously [24]. Plasma (EDTA) and tissue samples from male LCAT-deficient mice and their wild type littermates (4-month old) were obtained in the fasting state. The samples were kept frozen at −80 °C until the analysis.
C57BL/6 WT mice and HL KO mice were obtained from Jackson laboratories, and the EL KO and HL,EL DKO (double knockout) mice were generated as described previously [25] [26]. Mice were maintained in a monitored small animal facility at the University of Pennsylvania under IACUC-approved protocols. Mice were fed ad libitum with standard chow diet and were maintained with a 12h on/12h off light cycle with light off from 7:00 p.m. to 7:00 a.m., daily. Blood was collected via retro-orbital bleeding, under isoflurane anesthesia and using EDTA-coated tubes. Plasma was obtained by ultracentrifugation (10000 rpm, 7min at 4°C). Plasma and tissues were obtained from male EL KO (19-week old) and their WT littermates, male HL KO mice (25-week old), and male HL-EL-DKO mice (16-week old), after the animals were fasted for 4h.
2.2. Lipid Extraction and analysis:
Total lipids were extracted from plasma and various tissues by a modified method of Bligh and Dyer procedure. The tissue (100 mg) was homogenized at 4 °C in a glass homogenizer three times with 800 μl each of 50% methanol in water containing 0.01N HCl. A mixture of internal standards of tri-15:0 TAG, di-17:0 PC, di-15:0 PE and 17:0 LPC (10 μg each) was included in the methanolic HCl. Chloroform (2 ml) was added and the sample vortexed for 30s, followed by 1ml water and vortexing for 30s. The sample was centrifuged and the chloroform layer was transferred to another glass tube, and the lipids were concentrated under nitrogen and re-dissolved in chloroform before further analysis.
2.3. Analysis of fatty acids by GC/MS
The fatty acid composition of lipids in all tissues was analyzed by GC/MS after conversion to methyl esters. Briefly, the lipids were evaporated under N2 and dissolved in 0.5 ml toluene containing 25 μg each of 17:0 and 22:3 free fatty acids and 250 μg butylated hydroxytoluene. Methanolic HCl (0.3 ml of 8% HCl in methanol) was then added and the reaction mixture was heated under nitrogen at 100 °C for 1 h. The acid was neutralized by adding 1.0 ml of 0.33N NaOH and the fatty acid methyl esters were extracted twice with 3 ml of hexane. The pooled hexane extracts were evaporated under nitrogen and re-dissolved in 30ul of hexane and 1 ul was injected into GC/MS. The analysis was carried out using a Shimadzu QP2010SE GC/MS equipped with a Supelco Omega wax column (30m×0.25mm×0.25μ) as described previously. Total ion current in the range of 50-400 m/z was used to quantify the fatty acids, using 17:0 as the internal standard.
2.4. LC/MS analysis of DHA molecular species of LPC, PC, PE and TAG
LC/MS analysis of molecular species was performed on an AB Sciex 6500 QTRAP mass spectrometer coupled with Agilent 2600 UPLC system, as described previously [8]. Quantification of DHA-containing molecular species PC, LPC, PE and TAG was performed from the relative intensities of the various species and corresponding internal standards (17:0-17:0 PC, 17:0 LPC, 15:0-15:0 PE, and 15:0-15:0-15:0 TAG respectively). The data processing was carried out using Analyst 1.62 (AB Sciex, USA).
2.3. Statistical analyses:
Statistical analyses were performed using GraphPad Prism 9.0 software (GraphPad Software, La Jolla California USA). Significance of difference between WT and KO groups was determined by unpaired t test (2-tailed).
3. Results
3.1. Brain DHA levels
The total DHA levels in the brains of mice deficient in various enzymes and their corresponding WT littermates are shown in Fig. 1. The DHA content was not significantly different in the brains of LCAT-deficient and EL-deficient animals relative to the corresponding WT littermates. However, there was a significant decrease of DHA (−19%, p<0.0001) in HL-deficient brains compared to their WT controls. The DHA level was further decreased (−55%, p<0.0001) by the absence of both HL and EL (HL EL DKO mice). These results suggest that HL activity may be the major contributor of DHA to the brain, although EL may also contribute under conditions of HL-deficiency.
Fig. 1. Brain DHA levels.

The fatty acid composition of brains from LCAT KO, EL KO, HL KO and HL EL DKO mice, and their corresponding WT littermates was determined by GC/MS as described in the text. The values shown are % of DHA in the total fatty acids. Each KO group is compared with the corresponding WT littermates by unpaired t test. LCAT KO mice and EL KO mice did not show significant differences from their WT littermates, but the HL KO mice and HL EL DKO mice showed significantly lower DHA levels in the brain.
*** p<0.0001.
3.2. Brain ARA levels and DHA/ARA ratios
In general, there is a reciprocal relationship between the tissue levels of DHA and ARA after dietary interventions, since these two essential fatty acids compete with each other for enzymatic pathways. However, since all the PLA1 activities studied here also generate sn-2 ARA LPC in addition to sn-2 DHA LPC, and since both these LPCs are transported by Mfsd2a pathway [9] it is of interest to determine whether the deficiency of the enzymes also affects the ARA content of the brain in the absence of dietary intervention. The ARA content of the brains of various knockout mice and their corresponding controls is shown in Fig. 2, and the DHA/ARA ratios are shown in Fig. 3. There was a significant increase in the ARA content of the brain in LCAT-deficient (+35%, p<0.02), and HL EL double knockout (+29%, p<0.0001) animals, but it was not significantly different in the EL or HL knockout mice, relative to their corresponding WT littermates. The DHA/ARA ratio was also significantly lower in LCAT-deficient (− 19%, p<0.05) and the double knockout mice (−71%, p<0.0001) (Fig. 3). These results suggest that HL deficiency affects the DHA content of the brain more severely than the ARA content. It may be noted that our previous studies showed that HL is relatively more active on DHA-containing PC species, compared to the ARA-containing PC species [23], supporting this conclusion.
Fig. 2. Brain ARA levels.
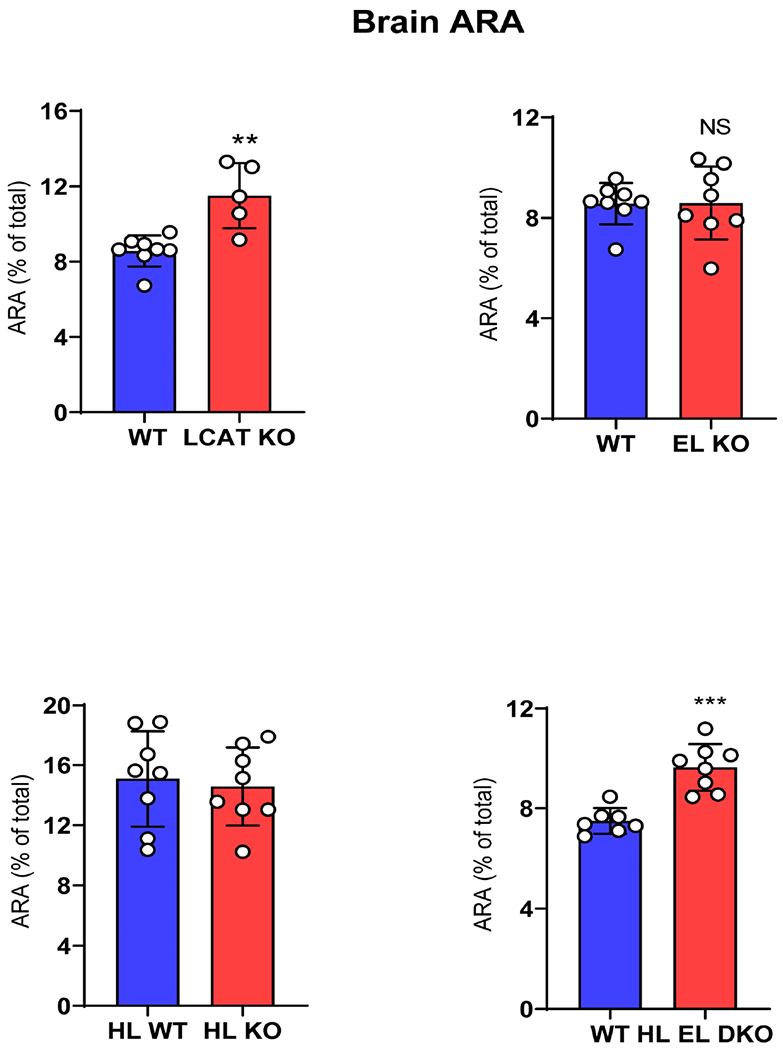
Fatty acid composition was determined by GC/MS, and the percentage of ARA in the total fatty acids of the brains of various genotypes and the corresponding WT littermates is presented. Statistical significance between the KO mice and the WT littermates was determined by unpaired t test. A significant increase brain ARA was observed in LCAT KO mice and HL EL DKO mice.
** p<0.02; *** p<0.0001.
Fig. 3. DHA/ARA ratios in the brain.
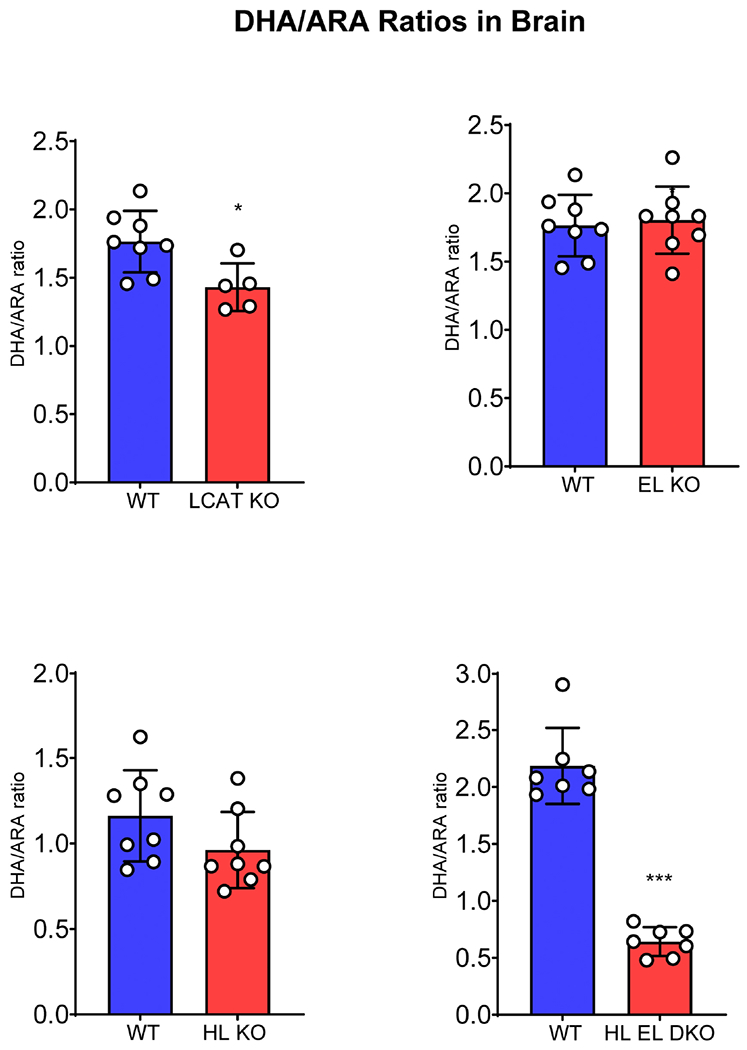
The ratio of DHA/ARA was significantly decreased only in LCAT KO and HL EL DKO mice.
P<0.05; *** p<0.0001, WT vs KO (unpaired t test)
3.3. Molecular species of DHA PC, and DHA PE in the brain of HL KO mice.
In order to determine whether HL deficiency affects the molecular species composition of the brain, we analyzed the DHA-containing molecular species of PC and PE by LC/MS/MS in HL KO mice, HL EL DKO mice and the corresponding WT littermates using selected ion monitoring. In the HL KO mice all major DHA species, except for 20:4-22:6 PC and PE, 18:2-22:6 PC and PE were significantly decreased compared to the brains of WT littermates (Fig. 4). In the HL EL double KO mice all major PC species except for 20:4-22:6 PC were decreased (Fig. 5). Among the PE species containing DHA, there was actually a slight increase in 20:4-22:6 PE and 18:2-22:6 PE, although the other major PE species decreased significantly. The increase in 20:4-22:6 PE is consistent with the relative increase in brain ARA in the absence of HL and EL (Fig. 2).
Fig. 4. Molecular species of DHA-containing PC and PE species in the brains of HL KO and WT mice.
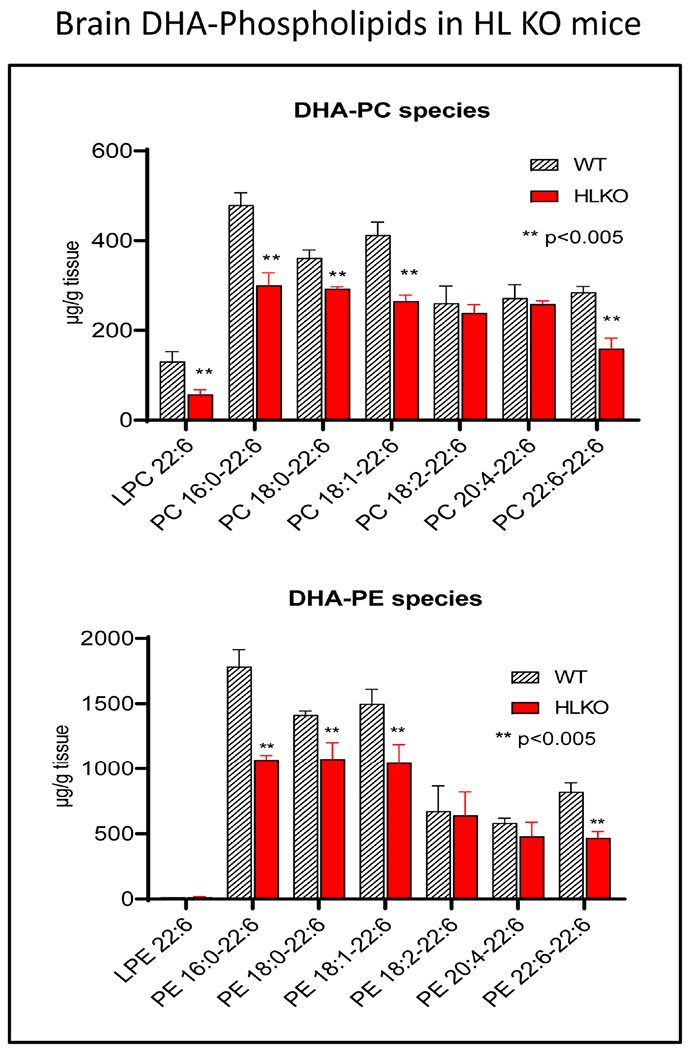
The molecular species composition was determined by selected ion monitoring LC/MS/MS, as described in the text. Statistical significance between HL KO and WT littermates was determined by unpaired t test. N=6 per group. All major species except those containing 18:2 and 20:4 showed a significant decrease in both PC and PE.
Fig. 5. Molecular species of DHA-containing PC and PE species in the brains of HL EL DKO and WT mice.
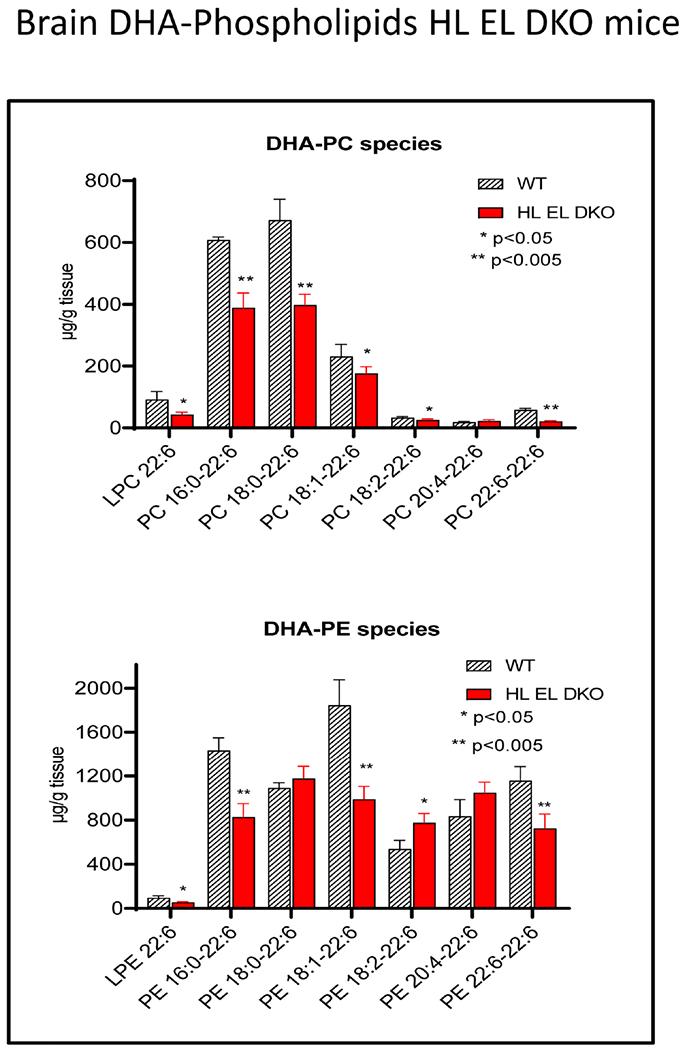
The molecular species composition was determined by selected ion monitoring in LC/MS/MS, as described in the text. Statistical significance between HL EL DKO mice and their WT littermates was determined by unpaired t test. N=4 per group. In the PC fraction, all major species except 20:4-22:6 PC showed a significant decrease in the DKO mice. In the PE fraction, 18:2-22:6, 18:0-22:6, and 20:4-22:6 species showed an increase in DKO mice, whereas all other specs showed a decrease compared to the WT littermates.
3.4. Total DHA and LPC-DHA in plasma
The total DHA content in the plasma was not significantly different in LCAT-deficient or EL-deficient animals, compared to their WT littermates. However, the DHA levels were significantly lower in the HL KO and HL EL DKO mice, relative to their WT littermates (Fig. 6, top). Since the WT and the corresponding KO animals were on identical diets, this indicates a decreased secretion of DHA from the liver or increased catabolism in the absence of HL and EL. The LPC-DHA content of the plasma, determined by LC/MS/MS, was significantly decreased in EL, HL, and HL EL DKO mice (Fig. 6, bottom), the decrease being greater in the double KO mice compared to the single KO mice. This is in agreement with the relative decrease in brain DHA in the HL KO vs HL EL DKO mice.
Fig. 6. DHA levels in the liver.

Fatty acid composition of livers from various KO mice and their WT littermates was determined by GC/MS. Unlike in the brain, the DHA concentration in the liver was not significantly decreased in HL KO or HL EL DKO mice, but the LCAT KO mice showed a significant decrease.
** p<0.005 compared to WT (unpaired t test)
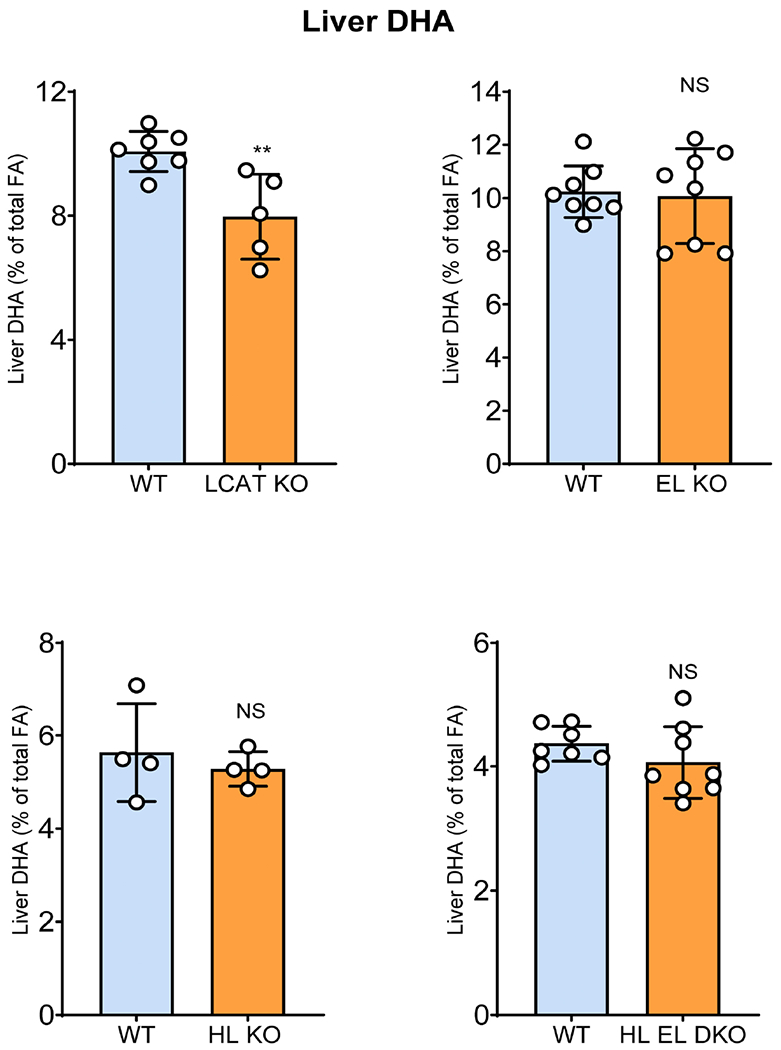
3.5. Liver DHA levels
Since the source of LPC-DHA in plasma and the brain appears to be the liver [14], we have also determined the DHA content of the liver in various mouse genotypes. In contrast to the brain DHA levels, the LCAT deficient mice showed a significant decrease in the liver DHA levels. On the other hand, the HL-deficient, EL-deficient, and HL EL double knockout mice showed no significant difference from their normal littermate (Fig. 7).
Fig. 7. Total DHA and LPC-DHA in plasma.
Top panel: Total lipid DHA in the plasma was determined by GC/MS as described in the text. The values shown are mean ± SD of 4-6 samples for each cohort (% of total fatty acids). A significant decrease was observed only in HL KO and HL EL KO mice (unpaired t test).
Bottom panel: LPC-DHA was determined by LC/MS/MS, as described in the text, employing 17:0 LPC as internal standard, and the values shown (nmol/ml) are corrected for the differential response factors of 17:0 LPC and 22:6 LPC. A significant decrease in LPCHA was observed in EL KO, HL KO, and HL EL DKO mice, but not in LCAT KO mice.
4. Discussion
These studies show a novel physiological function of HL in the development and function of the brain. The major known functions of HL are the hydrolysis of chylomicron remnants and IDL, the formation of LDL, and the hydrolysis of TAG-rich HDL to form small HDL. While these functions of HL are considered to be pro-atherogenic, it has also been reported that low HL activity is associated with increased cardiovascular risk, possibly because of its role in facilitating the reverse cholesterol transport [27]. Its potential role in the generation of LPC-DHA, as shown here, represents a novel beneficial function because LPC-DHA is the preferred molecular carrier of DHA into the brain [9]. We have previously shown that HL acts preferentially on phospholipids containing DHA at the sn-2 position, thus generating LPC-DHA [23]. Although the enzyme is secreted into the plasma in humans after heparin injection, its major site of activity is in the lipid metabolism of liver [28–30]. This enzyme is activated by apo E, but the apo E4 isoform is less effective than the apo E2 isoform [31]. It is well established that the carriers of apo E4 are at higher risk of developing Alzheimer’s, although the exact mechanisms by which apo E4 promotes Alzheimer’s are not yet clear. Previous studies suggested that apo E4 may affect blood brain barrier, impair amyloid clearance from the brain, increase inflammation, and accelerate DHA catabolism [32, 33]. Notably, the carriers of apo E4 have been reported to not benefit from dietary treatment with TAG-DHA, whereas the non-carriers show improvement in dementia under the same conditions [34]. Uptake of DHA by the brain was shown to be significantly lower in apo E4-carrying mice compared to apo E2- mice [35]. Furthermore, it has been reported that plasma LPC-DHA levels were not increased by fish oil supplementation in apo E4 carriers, whereas they were increased in the non-carriers [36]. Since the TAG-DHA in the fish oil needs to be converted to LPC-DHA for efficient uptake of DHA by the brain, it is possible that one mechanism for the inability of fish oil supplements to benefit the apo E4 carriers is because of the inefficient generation of LPC-DHA through the HL reaction. In support of this hypothesis, our recent studies showed that mice carrying human apo E4 isoform have decreased plasma LPC-DHA content, and also lower DHA levels in the brain compared to the mice carrying human apo E3. Furthermore we showed that dietary LPC-DHA (in the form of lipase-treated krill oil) increased the brain DHA efficiently in the apo E4 mice as well as apo E3 mice, whereas untreated krill oil, which has little LPC-DHA, was ineffective [37].
In addition to HL, our studies indicate a potential role for EL in the supply of LPC-DHA to the brain. Although the deficiency of EL did not decrease brain DHA, it decreased the plasma LPC-DHA levels compared to the WT controls (Fig. 7). Furthermore, the deficiency of both HL and EL resulted in a greater decrease of brain DHA compared to the deficiency of HL alone. While EL and EL have similar specificity towards HDL phospholipids, HL plays a greater role in the hepatic metabolism of lipoproteins, and in the hydrolysis of PC-DHA in the liver. EL is anchored to the plasma membrane of endothelial cells at the blood brain barrier, and is not released into the circulation. Therefore, its capacity to hydrolyze plasma PC may be limited, whereas HL may act on lipoprotein remnants and HDL in the hepatic capillaries as well as in the circulation. The expression of HL and EL is regulated in reciprocal manner during inflammation. Whereas HL is down-regulated during inflammation [38], EL is upregulated [39, 40]. Therefore, it is possible that EL plays a significant role in the generation of LPC-DHA under inflammatory conditions, and helps maintain the brain DHA levels at normal levels when the activity of HL is decreased (Fig. 8). It would be of interest to determine whether plasma LPC-DHA or brain DHA are decreased significantly in EL KO animals under inflammatory conditions.
Fig. 8. Potential roles of HL and EL in supplying DHA to the brain.

Dietary DHA is absorbed mostly as TAG in the chylomicrons (CM), which pass through adipose tissue, heart, and muscle, before entering the liver as CM remnants. Free DHA released from CM remnants is incorporated into sn-2 position of PC by the Kennedy pathway. Part of the PC-DHA is hydrolyzed by HL, to generate LPC-DHA, which is secreted into plasma, bound to serum albumin, and taken by the brain through the Mfsd2a pathway. In addition to HL, EL may also generate LPC-DHA to some extent in the plasma, especially at the blood brain barrier. Under inflammatory conditions, the relative contribution of EL may be increased because HL is down-regulated during inflammation [38], whereas EL is up-regulated [39, 40]. HL activity may also be decreased in carriers of apo E4, leading to decreased transport of DHA into the brain. This might contribute to the increased risk of Alzheimer’s disease in apo E4 carriers.
Although our studies show a significant decrease in brain DHA levels in HL deficiency, the possible effects on brain function in HL deficient animals or patients is not known. Gene polymorphism studies in human populations showed inconsistent results. Thus while Xiao et al [41] reported that LIPC G/A variant, which decreases HL expression, influences the memory scores and suggested potential susceptibility to Alzheimer’s disease, the results of Laws et al [42] showed no association with the disease. Further studies using HL-deficient animals, especially under chronic inflammatory conditions are warranted to resolve this question.
Highlights.
Deficiency of either LCAT or endothelial lipase (EL) did not affect brain DHA level
Deficiency of hepatic lipase (HL) decreased brain DHA content by 19%
Deficiency of both HL and EL decreased brain DHA by 55%
HL may be primary source of lysophosphatidylcholine (LPC)-DHA for the brain uptake
EL may provide LPC-DHA for the brain in the absence of HL
Acknowledgements:
This research was supported by U.S. Department of Veterans Affairs Merit Review Award I01 BX004315 (PVS), Office of the NIH Director Grant S10 OD010660 (LC/MS equipment) (PVS.), a grant from Alzheimer’s Association (AARG-19-616614) (DS), and a grant from National Institutes of Health (R37HL055323) (DJR). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or Veterans Administration or Alzheimer’s Association.
Abbreviations used
- ARA
Arachidonic acid
- BBB
Blood brain barrier
- DHA
Docosahexaenoic acid
- DKO
Double gene knockout
- EL
Endothelial lipase
- HL
hepatic lipase
- KO
Gene knockout
- LCAT
Lecithin-cholesterol acyltransferase
- LPC
Lysophosphatidylcholine
- PC
Phosphatidylcholine
- PE
Phosphatidylethanolamine
- PLA1 or 2
Phospholipase A1 or A2
- TAG
Triacylglycerol
- WT
Wild type
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of Interest: All authors declare no conflict of interest.
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- [1].Sun GY, Simonyi A, Fritsche KL, Chuang DY, Hannink M, Gu Z, Greenlief CM, Yao JK, Lee JC, Beversdorf DQ, Docosahexaenoic acid (DHA): An essential nutrient and a nutraceutical for brain health and diseases, Prostaglandins Leukotrienes & Essential Fatty Acids, 136 (2018) 3–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Weiser MJ, Butt CM, Mohajeri MH, Docosahexaenoic Acid and Cognition throughout the Lifespan, Nutrients, 8 (2016) 99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Hashimoto M, Hossain S, Al Mamun A, Matsuzaki K, Arai H, Docosahexaenoic acid: one molecule diverse functions, Critical Reviews in Biotechnology, (2016) 1–19. [DOI] [PubMed] [Google Scholar]
- [4].Bazan NG, Docosanoids and elovanoids from omega-3 fatty acids are pro-homeostatic modulators of inflammatory responses, cell damage and neuroprotection, Molecular Aspects of Medicine, 64 (2018) 18–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Cunnane SC, Chouinard-Watkins R, Castellano CA, Barberger-Gateau P, Docosahexaenoic acid homeostasis, brain aging and Alzheimer’s disease: Can we reconcile the evidence?, Prostaglandins Leukotrienes & Essential Fatty Acids, 88 (2013) 61–70. [DOI] [PubMed] [Google Scholar]
- [6].Chen CT, Kitson AP, Hopperton KE, Domenichiello AF, Trepanier MO, Lin LE, Ermini L, Post M, Thies F, Bazinet RP, Plasma non-esterified docosahexaenoic acid is the major pool supplying the brain, Sci Rep, 5 (2015) 15791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Lagarde M, Bernoud N, Brossard N, Lemaitre-Delaunay D, Thies F, Croset M, Lecerf J, Lysophosphatidylcholine as a preferred carrier form of docosahexaenoic acid to the brain, Journal of Molecular Neuroscience, 16 (2001) 201–204. [DOI] [PubMed] [Google Scholar]
- [8].Sugasini D, Thomas R, Yalagala PCR, Tai LM, Subbaiah PV, Dietary docosahexaenoic acid (DHA) as lysophosphatidylcholine, but not as free acid, enriches brain DHA and improves memory in adult mice, Scientific Reports, 7 (2017) 11263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Nguyen LN, Ma D, Shui G, Wong P, Cazenave-Gassiot A, Zhang X, Wenk MR, Goh ELK, Silver DL, Mfsd2a is a transporter for the essential omega-3 fatty acid docosahexaenoic acid, Nature, 509 (2014) 503–506. [DOI] [PubMed] [Google Scholar]
- [10].Wong BH, Silver DL, Mfsd2a: A Physiologically Important Lysolipid Transporter in the Brain and Eye, Adv Exp Med Biol, 1276 (2020) 223–234. [DOI] [PubMed] [Google Scholar]
- [11].Sugasini D, Yalagala PCR, Goggin A, Tai LM, Subbaiah PV, Enrichment of brain docosahexaenoic acid (DHA) is highly dependent upon the molecular carrier of dietary DHA: Lysophosphatidylcholine is more efficient than either phosphatidylcholine or triacylglycerol, The Journal of Nutritional Biochemistry, 74 (2019) 108231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Croset M, Brossard N, Polette A, Lagarde M, Characterization of plasma unsaturated lysophosphatidylcholines in human and rat, Biochemical Journal, 345 (2000) 61–67. [PMC free article] [PubMed] [Google Scholar]
- [13].Okudaira M, Inoue A, Shuto A, Nakanaga K, Kano K, Makide K, Saigusa D, Tomioka Y, Aoki J, Separation and quantification of 2-acyl-1-lysophospholipids and 1-acyl-2-lysophospholipids in biological samples by LC-MS/MS, J Lipid Res, 55 (2014) 2178–2192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Brindley DN, Hepatic secretion of lysophosphatidylcholine: A novel transport system for polyunsaturated fatty acids and choline, J. Nutr. Biochem, 4 (1993) 442–449. [Google Scholar]
- [15].Sekas G, Patton GM, Lincoln EC, Robins SJ, Origin of plasma lysophosphatidylcholine: evidence for direct hepatic secretion in the rat, J Lab Clin Med, 105 (1985) 190–194. [PubMed] [Google Scholar]
- [16].Subbaiah PV, Liu M, Bolan PJ, Paltauf F, Altered positional specificity of human plasma lecithin-cholesterol acyltransferase in the presence of sn-2 arachidonoyl phosphatidyl cholines. Mechanism of formation of saturated cholesteryl esters., Biochimica et Biophysica Acta, 1128 (1992) 83–92. [DOI] [PubMed] [Google Scholar]
- [17].Subbaiah PV, Sowa JM, Davidson MH, Evidence for altered positional specificity of LCAT in vivo: studies with docosahexaenoic acid feeding in humans, Journal of Lipid Research, 45 (2004) 2245–2251. [DOI] [PubMed] [Google Scholar]
- [18].Dobiasova M, Frohlich JJ, Advances in understanding of the role of lecithin cholesterol acyltransferase (LCAT) in cholesterol transport, Clin Chim Acta, 286 (1999) 257–271. [DOI] [PubMed] [Google Scholar]
- [19].Chen S, Subbaiah PV, Phospholipid and fatty acid specificity of endothelial lipase: Potential role of the enzyme in the delivery of docosahexaenoic acid (DHA) to tissues, Biochim. Biophys. Acta, 1771 (2007) 1319–1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Chen S, Subbaiah PV, Regioisomers of Phosphatidylcholine Containing DHA and Their Potential to Deliver DHA to the Brain: Role of Phospholipase Specificities, Lipids, 48 (2013) 675–686. [DOI] [PubMed] [Google Scholar]
- [21].Ishida T, Choi S, Kundu RK, Hirata K, Rubin EM, Cooper AD, Quertermous T, Endothelial lipase is a major determinant of HDL level, J Clin Invest, 111 (2003) 347–355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Kobayashi J, Miyashita K, Nakajima K, Mabuchi H, Hepatic Lipase: a Comprehensive View of its Role on Plasma Lipid and Lipoprotein Metabolism, J Atheroscler Thromb, 22 (2015) 1001–1011. [DOI] [PubMed] [Google Scholar]
- [23].Yang P, Subbaiah PV, Regulation of hepatic lipase activity by sphingomyelin in plasma lipoproteins, Biochim Biophys Acta, 1851 (2015) 1327–1336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Ng DS, Francone OL, Forte TM, Zhang JL, Haghpassand M, Rubin EM, Disruption of the murine lecithin:cholesterol acyltransferase gene causes impairment of adrenal lipid delivery and up- regulation of scavenger receptor class b type i, Journal of Biological Chemistry, 272 (1997) 15777-15781: 15777. [DOI] [PubMed] [Google Scholar]
- [25].Yang Y, Kuwano T, Lagor WR, Albert CJ, Brenton S, Rader DJ, Ford DA, Brown RJ, Lipidomic analyses of female mice lacking hepatic lipase and endothelial lipase indicate selective modulation of plasma lipid species, Lipids, 49 (2014) 505–515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Brown RJ, Lagor WR, Sankaranaravanan S, Yasuda T, Quertermous T, Rothblat GH, Rader DJ, Impact of combined deficiency of hepatic lipase and endothelial lipase on the metabolism of both high-density lipoproteins and apolipoprotein B-containing lipoproteins, Circ. Res, 107 (2010) 357–364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Dugi KA, Brandauer K, Schmidt N, Nau B, Schneider JG, Mentz S, Keiper T, Schaefer JR, Meissner C, Kather H, Bahner ML, Fiehn W, Kreuzer J, Low Hepatic Lipase Activity Is a Novel Risk Factor for Coronary Artery Disease, Circulation, 104 (2001) 3057–3062. [DOI] [PubMed] [Google Scholar]
- [28].Shafi S, Brady SE, Bensadoun A, Havel RJ, Role of hepatic lipase in the uptake and processing of chylomicron remnants in rat liver, Journal of Lipid Research, 35 (1994) 709–720. [PubMed] [Google Scholar]
- [29].Perret B, Mabile L, Martinez L, Tercé F.o., Barbaras R, Collet X, Hepatic lipase: structure/function relationship, synthesis, and regulation, Journal of Lipid Research, 43 (2002) 1163–1169. [PubMed] [Google Scholar]
- [30].Connelly PW, The role of hepatic lipase in lipoprotein metabolism, Clin Chim Acta, 286 (1999) 243–255. [DOI] [PubMed] [Google Scholar]
- [31].Hime NJ, Drew KJ, Hahn C, Barter PJ, Rye KA, Apolipoprotein E Enhances Hepatic Lipase-Mediated Hydrolysis of Reconstituted High-Density Lipoprotein Phospholipid and Triacylglycerol in an Isoform-Dependent Manner, Biochemistry, 43 (2004) 12306–12314. [DOI] [PubMed] [Google Scholar]
- [32].Yassine HN, Braskie MN, Mack WJ, Castor KJ, Fonteh AN, Schneider LS, Harrington MG, Chui HC, Association of Docosahexaenoic Acid Supplementation With Alzheimer Disease Stage in Apolipoprotein E ε4 Carriers: A Review, JAMA Neurology, 74 (2017) 339–347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Nock TG, Chouinard-Watkins R, Plourde M, Carriers of an apolipoprotein E epsilon 4 allele are more vulnerable to a dietary deficiency in omega-3 fatty acids and cognitive decline, Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids, 1862 (2017) 1068–1078. [DOI] [PubMed] [Google Scholar]
- [34].Huang TL, Zandi PP, Tucker KL, Fitzpatrick AL, Kuller LH, Fried LP, Burke GL, Carlson MC, Benefits of fatty fish on dementia risk are stronger for those without APOE ε4, Neurology, 65 (2005) 1409–1414. [DOI] [PubMed] [Google Scholar]
- [35].Vandal M, Alata W, Tremblay C, Rioux-Perreault C, Salem N, Calon F, Plourde M, Reduction in DHA transport to the brain of mice expressing human APOE4 compared to APOE2, Journal of Neurochemistry, 129 (2014) 516–526. [DOI] [PubMed] [Google Scholar]
- [36].Abdullah L, Evans JE, Emmerich T, Crynen G, Shackleton B, Keegan AP, Luis C, Tai L, LaDu MJ, Mullan M, Crawford F, Bachmeier C, APOE ε4 specific imbalance of arachidonic acid and docosahexaenoic acid in serum phospholipids identifies individuals with preclinical Mild Cognitive Impairment/Alzheimer’s Disease, Aging, 9 (2017) 964–985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Scheinman SB, Zayed M, Yalagala PC, Marottoli FM, Dhavamani S, Subbaiah PV and Tai LM, LPC-DHA/EPA-enriched diets increase brain DHA and modulate behavior in mice that express human APOE4, Frontiers in neuroscience, 10.3389/fnins.2021.690410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Feingold KR, Memon RA, Moser AH, Shigenaga JK, Grunfeld C, Endotoxin and interleukin-1 decrease hepatic lipase mRNA levels, Atherosclerosis, 142 (1999) 379–387. [DOI] [PubMed] [Google Scholar]
- [39].Yasuda T, Hirata K, Ishida T, Kojima Y, Tanaka H, Okada T, Quertermous T, Yokoyama M, Endothelial lipase is increased by inflammation and promotes LDL uptake in macrophages, J Atheroscler Thromb, 14 (2007) 192–201. [DOI] [PubMed] [Google Scholar]
- [40].Jin WJ, Sun GS, Marchadier D, Octtaviani E, Glick JM, Rader DJ, Endothelial cells secrete triglyceride lipase and phospholipase activities in response to cytokines as a result of endothelial lipase, Circulation Research, 92 (2003) 644–650. [DOI] [PubMed] [Google Scholar]
- [41].Xiao Z, Wang J, Chen W, Wang P, Zeng H, Chen W, Association studies of several cholesterol-related genes (ABCA1, CETP and LIPC) with serum lipids and risk of Alzheimer’s disease, Lipids Health Dis, 11 (2012) 163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Laws SM, Eckart K, Friedrich P, Eisele T, Kurz A, Förstl H, Riemenschneider M, No association of lipase C polymorphisms with Alzheimer’s disease, Neurobiol Aging, 31 (2010) 2192–2193. [DOI] [PubMed] [Google Scholar]


