Abstract
While it is known that a substantial proportion of individuals with tuberculosis disease (TB) present subclinically, usually defined as bacteriologically-confirmed but negative on symptom screening, considerable knowledge gaps remain. Our aim was to review data from TB prevalence population surveys and generate a consistent definition and framework for subclinical TB, enabling us to estimate the proportion of TB that is subclinical, explore associations with overall burden and program indicators, and evaluate the performance of screening strategies. We extracted data from all publicly available prevalence surveys conducted since 1990. Between 36.1% and 79.7% (median, 50.4%) of prevalent bacteriologically confirmed TB was subclinical. No association was found between prevalence of subclinical and all bacteriologically confirmed TB, patient diagnostic rate, or country-level HIV prevalence (P values, .32, .4, and .34, respectively). Chest Xray detected 89% (range, 73%–98%) of bacteriologically confirmed TB, highlighting the potential of optimizing current TB case-finding policies.
Keywords: subclinical TB, TB screening, TB prevalence surveys, symptom screening, chest X-ray screening
Our analysis of tuberculosis prevalence surveys showed that a median of 50.4% of prevalent bacteriologically confirmed tuberculosis was subclinical, meaning negative on symptom screening. Chest X ray detected 89% of cases. This could potentially suggest a change in tuberculosis case-finding policies.
Tuberculosis disease (TB) remains the leading cause of death from an infectious disease in the world [1]. Not all individuals with bacteriologically confirmed TB will present with or be aware of (clinical) symptoms [2]. When presenting to TB services, this asymptomatic yet infectious group is usually missed, as access to care mostly relies on positive symptom screening to start the TB diagnostic pathway [3]. Individuals with so-called subclinical TB could therefore continue to contribute to transmission [4], hindering global TB care and prevention efforts [1].
While the importance of the subclinical TB subpopulation is recognized, a clear definition has not been agreed upon. Both “asymptomatic” and “bacteriologically confirmed” are inherently ambiguous. The extent and duration of symptoms used for screening will change the proportion of cases that have a positive symptom screening [5]. Similarly, the extent of bacteriological examination, for example, the number of samples or the technique that is used, will change the proportion that will be bacteriologically confirmed [6, 7].
To enable progress, we propose to define asymptomatic and bacteriologically confirmed TB as defined by TB prevalence surveys, which are population-based surveys that investigate representative samples of the population to estimate the national prevalence of bacteriologically confirmed adult pulmonary TB. Through X ray and symptom screening, individuals become eligible for sputum investigation with Xpert and/or culture (Table 1) [8]. While some variation remains, prevalence surveys can provide comparable measurements for the majority of high-burden countries [9], both between and within countries over time for the proportion of TB that is subclinical, that is, asymptomatic (usually defined as negative on screening for cough of a certain duration) and bacteriologically confirmed (usually defined as positive on at least 1 culture or polymerase chain reaction [PCR]–based test). Through this definition, subclinical TB can be placed in a comprehensive framework that reflects the relevant stages and flows in the spectrum of TB infection and disease.
Table 1.
Prevalence of Tuberculosis and Characteristics of Screening
| Survey | Crude Prevalence of TB (95%CI), n/100 000 Population | Estimated Incidence (95% CI), n/100 000 Population | Symptom Screening Criteria | X-ray Screening Device | X-ray Screening Criteria | Bacteriological Confirmation Test | Criteria for Eligibility for Bacteriological Examination | Total Number of Individuals Screened | Proportion of Individuals Screened That Is S–X– (%) | Proportion of Individuals Screened That Is S+X– (%) | Proportion of Individuals Screened That Is S+X+ (%) | Proportion of Individuals Screened That Is S–X+ (%) | Other (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bangladesh 2015 | 287 (244–330) | 221 (160–290) | Symptom screening score ≥3 | Digital mobile X ray | Any lung abnormality consistent with TB | Culture-positive and/or Xpert-positive | S+ and/or X+ or XNA and symptom score ≥1 | 98 710 | 79.4 | 4.2 | 3.1 | 13.8 | S+XNA 0.04 |
| Cambodia 2002 | 1208 (992–1463) | 600 | Cough ≥3 weeks and/ or hemoptysis in the previous month | Portable X-ray machine | TB-related shadows (active, suspected, and healed TB) or other lung disease, except for those with a single calcification nodule only or a minor pleural adhesion at the costophrenic angle | Smear-positive and/or culture-positive | S+ and/or X+ or XNA | 22 160 | Not reported | 4.6 | 2.6 | 8.2 | Include XNA |
| Cambodia 2011 | 831 (707–977) | Not reported | Cough ≥2 weeks and/or hemoptysis | Portable X-ray machine | Any abnormal shadow in the lung field or mediastinum other than a single small calcification nodule with a size <10 mm or pleural adhesion at the costophrenic angle(s) | Smear-positive and/or culture-positive | S+ and/or X+ or XNA | 37 417 | 87.22 | 3.1 | 1.9 | 7.2 | S–XNA 0.4 S+XNA 0.1 “Other” 0.02 |
| China 2000a | 466 | Not reported | Cough ≥3 weeks and/ or hemoptysis ≥3 weeks | Chest fluoroscopy of all patients, then X ray if they showed abnormal results | Abnormal findings except hilar calcification, a few fibrotic indurated lesions, small area of pleural thickening | Smear positive and/or culture positive | S+ and/or X+ and all known TB cases | 365 097 | Not reported | Not reported | Not reported | Not reported | Not applicable |
| China 2010a | 459 | Not reported | Not reported | Not reported | Not reported | Smear microscopy and culture | S+ and/or X+ and all known TB cases | 25 2940 | Not reported | Not reported | Not reported | Not reported | Not applicable |
| Democratic People’s Republic of Korea 2016 | 567 (510–631) | Not reported | Cough ≥2 weeks and/ or hemoptysis | Portable X-ray machine | Abnormal chest radiograph in the lung field or mediastinum other than a single small calcification nodule with a size <10 mm or pleural adhesion at cost-phrenic angle(s) | Culture-positive | S+ and/or X+ | 60 683 | Not reported | Not reported | 1.7 | 3.1 | S–X– or S–XNA 92 S+X– or S+XNA 3.2 |
| Indonesia 2014 | 759 (589–961) | Not reported | Cough ≥2 weeks and/ or hemoptysis | Digital mobile X ray | Any lung or pleura abnormality | Smear-positive and/or culture-positive and/or Xpert-positive | S+ and/or X+ or XNA | 67 944 | 77.3 | 5.7 | 6.6 | 9.9 | S+XNA 0.37 S–XNA with any symptom of TB 0.2 |
| Lao People’s Democratic Republic 2011 | 595 (457–733) | Not reported | Cough ≥2 weeks and/ or hemoptysis in the previous month | Full-size conventional CXR | Any abnormal lung field shadow | Culture-positive | S+ and/or X+ | 39 212 | 83.8 | Not reported | 3.3 | 7.9 | S+X– or S+XNA 4.9 |
| Mongolia 2015 | 559.6 (454.5–664.7) | 428 (220–703) | Cough ≥2 weeks | Digital mobile X ray | Any abnormal shadow in lung field and mediastinum or pleural effusion | Smear-positive and/or culture-positive | S+ and/or X+ or XNA | 50 309 | 79.3 | 3.4 | 1.6 | 14 | S+XNA 0.08 SNA X+ 0.06 SNA XNA 1.5 |
| Myanmar 2009 | 612.8 (502.2–747.6) | 526 (307–802) | Any symptom | Portable X-ray machine | Any abnormality in the lung field or mediastinum greater than a single small calcification nodule or pleural adhesion at the costophrenic angle | Smear-positive and/or culture-positive | S+ and/or X+ or XNA | 51 367 | 76.2 | 0.8 | 2.5 | 18.3 | S–XNA 2.1 S+XNA 0.1 Suspected false-negative CXR 0.1 |
| Philippines 2016 | 1159 (1016–1301) | 554 (311–866) | Cough ≥2 weeks and/ or hemoptysis in the previous month | Mass miniature radiography | Any abnormality suggestive of TB | Culture-positive and/or Xpert-positive | S+ and/or X+ or XNA | 46 689 | 60.2 | 2.8 | 2.9 | 22.9 | S+XNA 0.3 S–XNA 10.9 |
| Thailand 2012b | 142 (166.3–287.8) | Not obtainable | Cough ≥2 weeks | Not obtainable | Not obtainable | Smear-positive and/or culture-positive | Not obtainable | 62 536 | 90.3 | 2.8 | 0.8 | 6 | Includes XNA |
| Vietnam 2007 | 286 | 171 | Cough ≥2 weeks | Either mass miniature radiography or digital mobile X ray | Any abnormality suggestive of TB | Smear-positive and/or culture-positive | S+ and/or X+ or TB current treatment or history of treatment within 2 years | 94 179 | 92.2 | 0.01 | 0.6 | Not reported | SNA and XNA 0.4 S+XNA 3.7 SNA X+ 2.9 |
| Ethiopia 2011 | 277 (208–347) | 258(191–335) | Cough ≥2 weeks | Portable X-ray machine | Any abnormality in lung field or mediastinum, including cavities, infiltrates, pleural effusion, hilar or mediastinal lymphadenopathy, pulmonary nodules, interstitial abnormalities suggestive or TB or healed TB | Culture-positive | S+ and/or X+ | 46 697 | Not reported | Not reported | 1.7 | 6.4 | S–X– or S–XNA 87.1 S+X– or S+XNA 4.7 |
| Gambia 2012 | 179 (149–231) | 175 (132–215) | Cough ≥2 weeks, or cough ≤2 weeks plus ≥2 symptoms suggestive of TB, or no cough but ≥3 symptoms suggestive of TB | Digital mobile X ray | Any abnormality in lung field or mediastinum, including cavities, infiltrates, pleural effusion, hilar or mediastinal lymphadenopathy, pulmonary nodules, interstitial abnormalities suggestive or TB or healed TB | Culture-positive | S+ and/or X+ | 43 100 | Not reported | 5.5 | 2.4 | 5.5 | S+XNA 0.13 S–XNA or S–X– 86.2 |
| Ghana 2013 | 327 (282–347) | Not reported | Cough ≥2 weeks | Digital mobile X ray | Any abnormalities in lung, pleura, mediastinum | Culture-positive and/or Xpert-positive with X+ | S+ and/or X+ or XNA | 61 726 | 86.6 | 1.8 | 1.2 | 7.1 | S+XNA 0.1 S–XNA 3.1 |
| Kenya 2015 | 558 (455–662) | Not reported | Cough ≥2 weeks | Digital mobile X ray | Any finding suggestive of TB | Culture-positive and/or Xpert-positive | S+ and/or X+ or XNA | 63 050 | 84.6 | 4.5 | 2 | 8.2 | S–XNA 0.6 S+XNA 0.5 |
| Malawi 2013c | 452 (312–593) | Not reported | ≥1 week of cough or sputum or blood in sputum or chest pain or weight loss or night sweats or fatigue or fever or shortness of breath | Conventional radiography (film system), portable X-ray generator | Any lung abnormality (opacities, cavitation, fibrosis, calcification) | Culture-positive and/or Xpert-positive | S+ and/or X+ or XNA | 31 579 | 88.8 | 7.4 | 1.2 | 2.3 | S+XNA 0.2 S–XNA 0.03 missed 0.2 |
| Namibia 2017 | 431 (361.4–514.3) | Not reported | Cough or weight loss or fever or night sweats | Portable X-ray machine | Any abnormality suggestive of TB, read by automatic software and radiologist | Culture-positive and/or Xpert-positive | S+ and/or X+ or XNA | 29 495 | 63.2 | 14 | 5.8 | 11.3 | S+XNA 1.5 S–XNA 4.3 |
| Nigeria 2012 | 524 (378–670) | 108 (50–186) | Cough ≥2 weeks | Mass miniature radiography | Any abnormality suggestive of TB | Smear-positive and/or culture-positive | S+ and/or X+ or XNA | 44 186 | Not reported | Not reported | 1.7 | 5 | S–X– or S–XNA 89.4 S+X– or S+XNA 3.9 |
| Rwanda 2012 | 119.3 (78.8–159.9) | Not reported | Cough any duration | Not reported | Any abnormality suggestive of TB | Culture-positive | S+ and/or X+ or XNA | 43 128 | 88.8 | 4.8 | 1.3 | 4.9 | S+ XNA 0.02 S– XNA 0.1 SNA X– 0.02 |
| Sudan 2014 | 183.4 (129.6–237.2) | Not reported | Cough ≥2 weeks | Digital mobile X ray | Any lung abnormality, including pleura | Culture-positive and/or NAAT-positive | S+ and/or X+ or XNA or TB current treatment | 83 202 | 78.2 | Not reported | 2.2 | Not reported | S–X– or SNA XNA 0.7 SNA XNA 0.13 S+XNA or S+X– 0.8 S–X+ or SNA X+ 11.6 S–XNA 6.3 |
| Tanzania 2012 | 307 (261–360) | Not reported | Cough ≥2 weeks or hemoptysis or fever ≥2 weeks or weight loss or excessive sweating | Digital mobile X ray | Any abnormalities in the lung field or mediastinum | Culture-positive | S+ and/or X+ or XNA | 50 447 | 87.5 | 6.4 | 1.7 | 3.7 | S+XNA 0.6 SNAX+ 0.08 |
| Uganda 2014 | 401 (292–509) | Not reported | Cough ≥2 weeks | Digital mobile X ray | Any abnormalities in lung | Culture-positive and/or Xpert-positive | S+ and/or X+ or XNA | 41 154 | 87.5 | 5.2 | 1.3 | 5.6 | XNA 0.4 |
| Zambia 2014 | 638 (505–774) | Not reported | Cough ≥2 weeks or fever ≥2 weeks or chest pain ≥2 weeks | Digital mobile X ray | Any lung abnormality excluding heart and bone abnormality | Culture-positive and/or Xpert-positive | S+ and/or X+ or XNA | 46 099 | 84.2 | 6.3 | 3.6 | 4.9 | S+XNA 0.09 S–XNA 1.2 |
| Zimbabwe 2014 | 317.1 (250.5–383.8) | Not reported | Any symptom | Digital mobile X ray | Any abnormalities in lung | Culture-positive and/or Xpert-positive | S+ and/or X+ or XNA | 33 736 | 82.7 | 3.4 | 1.9 | 8.3 | S–XNA 3.5 S+XNA 0.1 “other” 0.03 |
A list of references for included prevalence surveys is available in Supplementary Materials Appendix 1.
Abbreviations: CI, confidence interval; CXR, chest X ray; NA, not applicable, NAAT, nucleic acid amplification tests; used when results for symptom (SNA) or X-ray screening (XNA) were not available; S, symptoms; TB, tuberculosis; X, X ray.
aSurveys from China were excluded from the analysis because results active pulmonary cases, of which the proportion of bacteriologically negative clinically diagnosed cases is unknown.
b Some data were not obtainable from Thailand in 2012 because the only version of the survey report was in Thai.
cMalawi 2013: results were excluded from the analysis because the quality of images observed in some clusters was substandard and could not be compared with results from other countries [10].
Our aim was to review data from TB prevalence population surveys and generate a consistent definition and framework for subclinical TB, thereby enabling us to estimate the proportion of TB that is subclinical, as well as explore associations with overall burden and program indicators. Finally, we considered the potential performance of chest X ray–based screening strategies to replace the current symptom-focused TB care and prevention policies.
METHODS
We considered for inclusion population-based TB prevalence surveys completed since 1990, with reports or articles publicly available through August 2019. A literature search for the period from January 1990 to August 2019, restricted to the English language, was conducted by one author (I. L.) in PubMed (August 2019) using the following search terms: “tuberculosis” and “prevalence” in the title and “survey” as text words. Reference lists of identified studies were also examined. Studies that were about a subset of TB cases (eg, drug-resistant TB, women only, healthcare workers), TB infection rather than TB and risk factors for TB (eg, diabetes), and review articles were excluded. Gray literature, such as unpublished survey reports produced by national TB programs, abstracts, and presentations from international meetings and routine progress updates collated by the World Health Organization Global Task Force on TB Impact Measurement on the status of surveys since 2008, was also systematically reviewed.
Subnational TB prevalence surveys were included from the review by Horton et al [11]. Surveys were included if both symptom screening interview and X ray were performed on all eligible participants and if surveys reported the proportion of bacteriologically confirmed cases by screening modality as well as the proportion of bacteriologically confirmed cases that were negative on symptom screening.
We extracted data on the burden of TB (prevalence of bacteriologically confirmed TB), screening and bacteriological confirmation methods, outcomes of screening of the study population, and outcomes of screening of bacteriologically confirmed cases. To explore the impact of program performance, we generated the patient diagnostic rate (PDR) as the case notification rate (number of individuals diagnosed with TB and reported to the National TB Programme per 100 000 population) divided by the prevalence of bacteriologically confirmed TB [11] (inverse of the prevalence to notification ratio).
We defined subclinical TB cases as all participants who were negative on symptom screening, following the criteria established in each survey but confirmed on bacteriological testing. A framework for the natural history of TB was then developed to place subclinical disease in the spectrum of Mycobacterium tuberculosis infection and TB. Bacteriological confirmation generally included at least 1 positive culture or PCR-based test [8]. Participants not eligible for X-ray screening (eg, because of pregnancy) were considered negative at X-ray screening. In settings where TB prevalence surveys were repeated in the same geographical area using similar methodology, we examined longitudinal trends in subclinical TB.
We performed a meta-regression (metareg in STATA v15) analysis for the effect of covariates on the proportion of subclinical TB. To avoid interdependency, 1 survey per country or area was included. We explored the association with TB prevalence in the country, continent, country-level HIV prevalence; definition of symptom screen; the PDR as a metric of program performance; and proportion of cases that was male. We also performed a random-effects meta-analysis using the metaprop command in STATA v15 [12] to quantify between study heterogeneity.
To examine the relative contribution of symptoms compared to X ray as a screening tool, we analyzed the proportion of bacteriologically confirmed cases identified through each method. We also analyzed the proportion of participants who screened positive via symptoms interview, on X ray, or on both methods and were considered eligible for bacteriological examination.
RESULTS
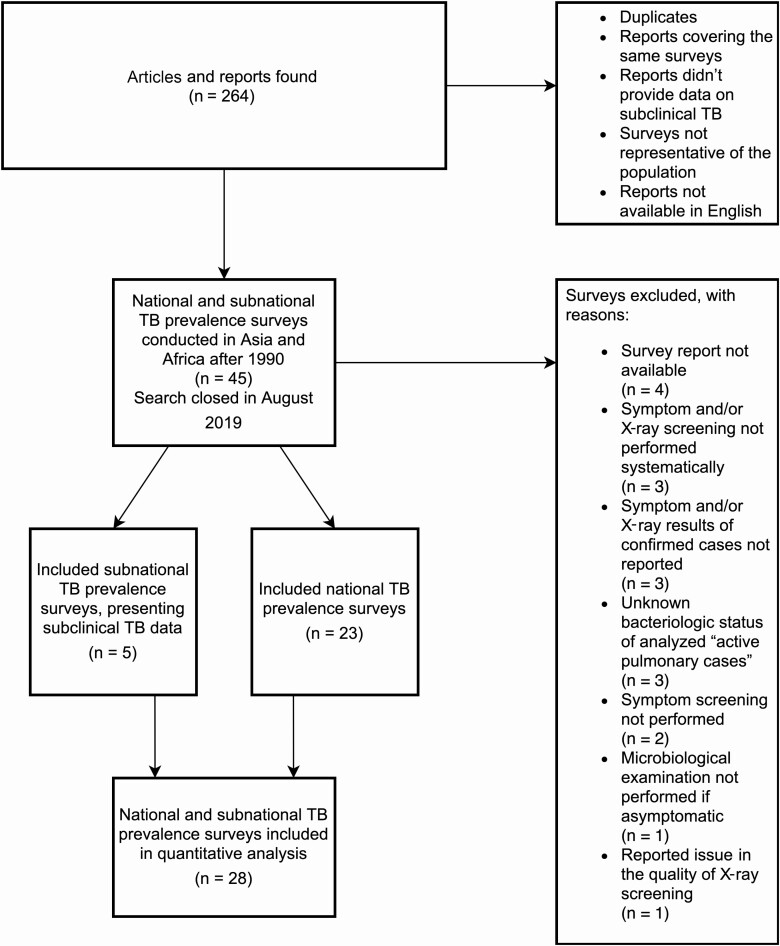
We included 23 national surveys and 5 subnational surveys conducted in 23 countries across Africa and Asia, representing 36% of the global TB burden in 2018 [1] and 57.5% (23/40) of all national-level surveys completed since 1990. (Data available in Tables 1–3, list of references for included surveys available in Supplementary Materials Appendix 1.) The reasons for exclusion of the remaining prevalence surveys are shown in Figure 1.
Table 2.
Subnational Surveys in India
| Survey | Prevalence of TB (95% Confidence Interval)/100 000 Population | Bacteriological Confirmation Test | Criteria for Eligibility for Bacteriological Examination | S–X+ Cases (%) | S– Cases (%) |
|---|---|---|---|---|---|
| Tamil Nadu (India) 1999 | 605 | One culture-positive sample | S+ and/or X+ | 46.3 | 46.3 |
| Tamil Nadu (India) 2001 | 454 | Culture-positive | S+ and/or X+ and all known TB cases | 33.7 | 36 |
| Tamil Nadu (India) 2004 | 309 | Culture-positive | S+ and/or X+ and all known TB cases | 36.4 | 39.1 |
| Tamil Nadu (India) 2006 | 388 | Culture-positive | S+ and/or X+ and all known TB cases | 34.9 | 39.2 |
| Tamil Nadu (India) 2010 | 259 | Culture-positive | S+ and/or X+ and all known TB cases | 32.9 | 55 |
A list of references for included prevalence surveys is available in Supplementary Materials Appendix 1.
Abbreviations: S, symptoms; TB, tuberculosis; X, X ray.
Table 3.
Characteristics of Bacteriologically Confirmed Cases
| Survey | S–X+ Cases (%) | S− Cases (%) | S+ Cases (%) | X+ Cases (%) | S+X− Cases (%) | S+X+ Cases (%) | Proportion Negative on any Symptom Among Cases (%) | Proportion of Males Among All Bacteriologically Confirmed Cases (%) | HIV Prevalence Among All Bacteriologically Confirmed Cases (%) | Percentage of Cases Found Already in TB Care (%) | Bacteriologically Confirmed Notification Rate (n/100 000) | Prevalence to Notification Ratio |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bangladesh 2015 | 61.9 | 61.9 | 38.1 | 90.3 | 9.7 | 36 | Not reported | 72.3 | Not measured | 1.8 | 101.7 | 2.8 |
| Cambodia 2002 | 60.9 | 60.9 | 39.1 | 95.6 | 4.4 | 34.7 | 15.9 | 60 | Not measured | 4.2 | 222.9 | 2.0 |
| Cambodia 2011 | 69.4 | 70.4 | 29.1 | 95.6 | 3.5 | 25.6 | 10.2 | 59.9 | Not measured | 2 | 161.4 | 1.7 |
| China 2000a | Not reported | 12.1 | 87.9 | 49.5 | Not reported | Not reported | Not reported | 70.4 | Not reported | Not reported | Not reported | Not available |
| China 2010a | Not reported | 43.1 | 56.9 | Not reported | Not reported | Not reported | Not reported | 69.9 | Not reported | Not reported | 38.7 | 1.7 |
| Democratic People’s Republic of Korea 2016 | 42.9 | 42.9 | 57 | 97.9 | 0.7 | 55 | Not reported | 69.7 | Not measured | 31.2 | 482.1 | 1.2 |
| Indonesia 2014 | 42.5 | 42.5 | 57.5 | 94.1 | 4.9 | 51.6 | Not reported | 65.5 | Not measured | 4.5 | 113.3 | 2.3 |
| Lao People’s Democratic Republic 2011 | 50.2 | 50.2 | 49.8 | 97 | 2.9 | 46.8 | Not reported | 66.2 | Not measured | 2.5 | 80.4 | 3.5 |
| Mongolia 2015 | 77.8 | 79.4 | 20.6 | 96 | 2.5 | 18.1 | 42.7 | 64.5 | Not measured | 4.4 | 83.2 | 2.5 |
| Myanmar 2009 | Not reported | 78.8 | 19.7 | 95.2 | Not reported | Not reported | 38.2 | 66.2 | Not measured | 3.5 | 114.4 | 2.1 |
| Philippines 2016 | 63.9 | 67.8 | 32.2 | 92.2 | 1.7 | 28.3 | 26 | 69 | Not measured | 6.4 | 142.2 | 3.1 |
| Thailand 2012b | 66.2 | 66.2 | 33.8 | 95.8 | 4.2 | 29.6 | Not obtainable | Not obtainable | Not obtainable | Not obtainable | 56.4 | 1.8 |
| Vietnam 2007 | 67.3 | 73.6 | 26.4 | 85.1 | 8.5 | 17.8 | Not reported | 78.8 | Not measured | 0.07 | 85.2 | 2.3 |
| Ethiopia 2011 | 48.2 | 48.2 | 51.8 | 89 | 10.9 | 40.9 | Not reported | 55.3 | 8.00 | 2.7 | 91.0 | 1.2 |
| Gambia 2012 | 36.6 | 38. | 62 | 81.7 | 15.5 | 45 | Not reported | 62 | Not measured | 5 | 145.3 | 0.6 |
| Ghana 2013 | Not reported | 59 | 41 | 75.2 | not reported | not reported | Not reported | 50 | Not reported | 5 | 45.2 | 2.5 |
| Kenya 2015 | 50.5 | 51.8 | 40.2 | 88.2 | 10.5 | 38 | Not reported | 62 | 13.4 | 4.9 | 158.2 | 3.5 |
| Malawi 2013c | 30.3 | 30.3 | 69.7 | 49.2 | 50.76 | 18.9 | Not reported | 47.7 | 16.7 | 4.5 | 86.8 | 2.5 |
| Namibia 2017 | Not reported | 51.3 | 48.7 | 95 | not reported | not reported | Not reported | 60 | 15.1 | 4.2 | 551.9 | 0.8 |
| Nigeria 2012 | Not reported | 36.1 | 63.9 | 89 | not reported | not reported | 22.9 | 67.7 | Not measured | 0.2 | 55 | 5.8 |
| Rwanda 2012 | 50 | 50 | 50 | 79.6 | 20.4 | 27.8 | Not reported | 73.7 | 3.7 | 5.3 | 56.1 | 1.3 |
| Sudan 2014 | 40 | 40 | 45.1 | 78 | 7.1 | 38 | Not reported | Not reported | Not measured | 7.1 | 25 | 3.5 |
| Tanzania 2012 | not reported | 36.7 | 63.2 | 73.5 | not reported | not reported | Not reported | 60 | 5.9 | Not reported | 92.8 | 3 |
| Uganda 2014 | 50.6 | 50.6 | 49.4 | 88.7 | 10 | 38.1 | Not reported | 75 | 26.9 | 10 | 141.8 | 2.8 |
| Zambia 2014 | 39 | 39 | 61 | 83 | 17 | 44 | Not reported | 66.7 | 13.2 | 2.6 | 159.2 | 2.0 |
| Zimbabwe 2014 | Not reported | 63.55 | 36 | 86 | not reported | not reported | Not reported | 54.2 | Not reported | Not reported | 137.9 | 2.5 |
A list of references for included prevalence surveys is available in Supplementary Materials Appendix 1.
Abbreviations: S, symptoms; X, X ray.
aSurveys from China were excluded from the analysis because results include active pulmonary cases, of which the proportion of bacteriologically negative clinically diagnosed cases is unknown.
bSome data were not obtainable from Thailand in 2012 because the only version of the survey report was in Thai.
cMalawi 2013: results were excluded from the analysis because the quality of images observed in some clusters was substandard and could not be compared with results from other countries [10].
Figure 1.
Selection flow chart for tuberculosis prevalence surveys.
The 2013 Malawi survey was excluded because of reported issues in the quality of X ray in many clusters [10]. Surveys from China were excluded because results were only reported for smear-positive or “active pulmonary cases,” the latter including an unknown proportion of bacteriologically negative, clinically diagnosed cases, which did not match our criteria [13]. Data from these surveys are included in Tables 1–3.
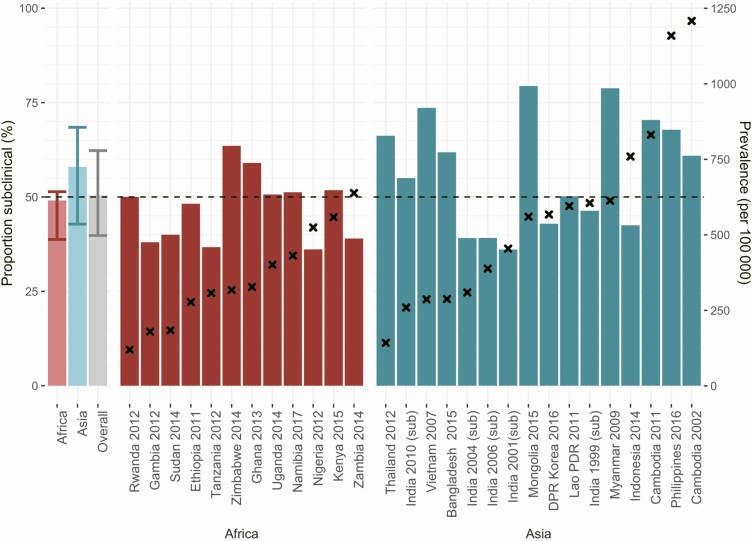
Across included surveys, the median percentage of subclinical TB cases was 50.4% (interquartile range [IQR], 39.8%–62.3%; range, 36.1%–79.7%), which was 49.4% (IQR, 38.8%–52.4%) in African countries. In the Asian countries, the median was 56.4% (IQR, 42.8%–68.5%), with no discernable trend by TB prevalence (Figure 2) in either continent.
Figure 2.
Proportion of subclinical tuberculosis disease (TB) in prevalence surveys. The proportion of all prevalent TB cases that were subclinical (bars: left side y-axis) by the adult crude prevalence of bacteriologically confirmed TB found in that survey (crosses: right side y-axis). The first 3 bars show the median (bar) and interquartile range (error bars) for values found in surveys in Africa, Asia, and overall. Abbreviations: DPR, Democratic People’s Republic; PDR, People’s Democratic Republic; sub, subnational surveys.
Data on repeated surveys were available from Cambodia and Tamil Nadu state in India. Although no clear trend is present, they seemed to suggest that the proportion of subclinical TB increased as TB prevalence declined (Tables 2–3). An indication for this trend was also seen among smear-positive TB in surveys repeated in China from 2000 and 2010 (Table 3).
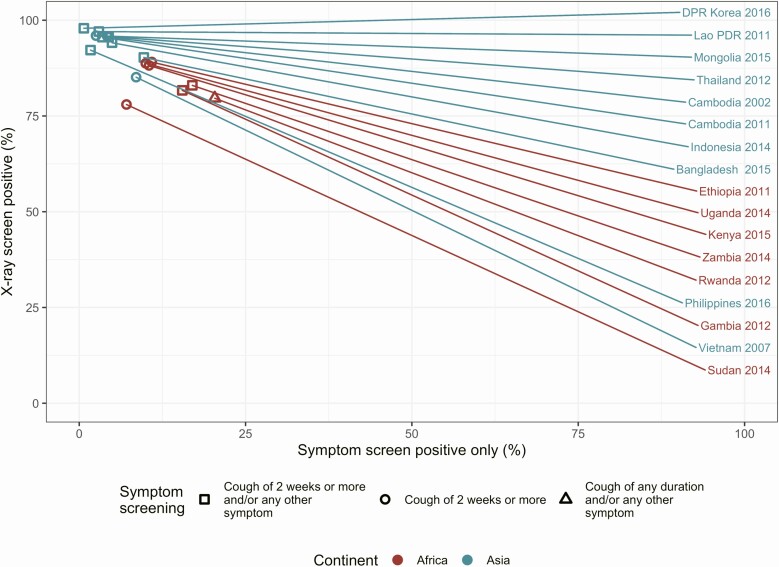
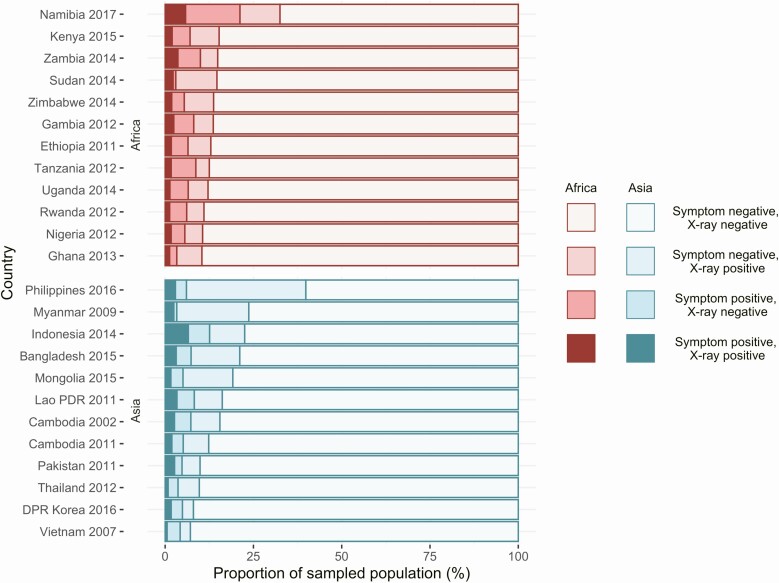
As Figure 3 shows, X-ray screening identified the vast majority of bacteriologically confirmed cases in all countries (median, 89%; range, 73%–98%). In contrast, the percentage of bacteriologically confirmed TB cases that were negative on X ray but positive on symptom was below 25% (median, 7%; range, 0.7%– 22%) in all surveys, with between 0.01% and 15% of bacteriologically confirmed cases diagnosed through direct bacteriological examination (see Figure 3 and Table 1). In the sampled population, surveys found that 8.8% of individuals screened positive on X ray (range, 4.8%–26%), whereas 6.3% (range, 3%–21%) were positive on symptoms (Figure 4).
Figure 3.
Screening modality for bacteriologically confirmed tuberculosis disease (TB) cases. The proportion of bacteriologically confirmed cases in prevalence surveys that screened positive on X ray (y-axis) or on symptom screen only (x-axis). Raw data are available in Table 3. Note: The Vietnam 2007 and Sudan 2014 surveys did not report symptom screening and X-ray results for TB cases who were under treatment or had a history of treatment within 2 years but did receive bacteriological examination. In the Philippines 2016 survey, 5% of bacteriologically confirmed cases were exempted from X ray (see Table 1). Abbreviations: DPR, Democratic People’s Republic; PDR, People’s Democratic Republic.
Figure 4.
Population screening results. The proportion of population included in prevalence surveys that screened positive on X ray, symptom screen, both, or neither. Abbreviations: DPR, Democratic People’s Republic; PDR, People’s Democratic Republic.
We frame subclinical pulmonary TB in the wider context of TB natural history in Figure 5. Here, subclinical TB is a distinct intermediary disease state, which follows after a minimal disease state with initial pathological changes (eg, visible on imaging) but not bacteriologically confirmed (at least within the limits of sampling undertaken) and unlikely to be contributing to transmission. Crucially, individuals can progress and regress from each stage, although how fast or how frequently individuals move between stages will vary widely [14, 15].
Figure 5.
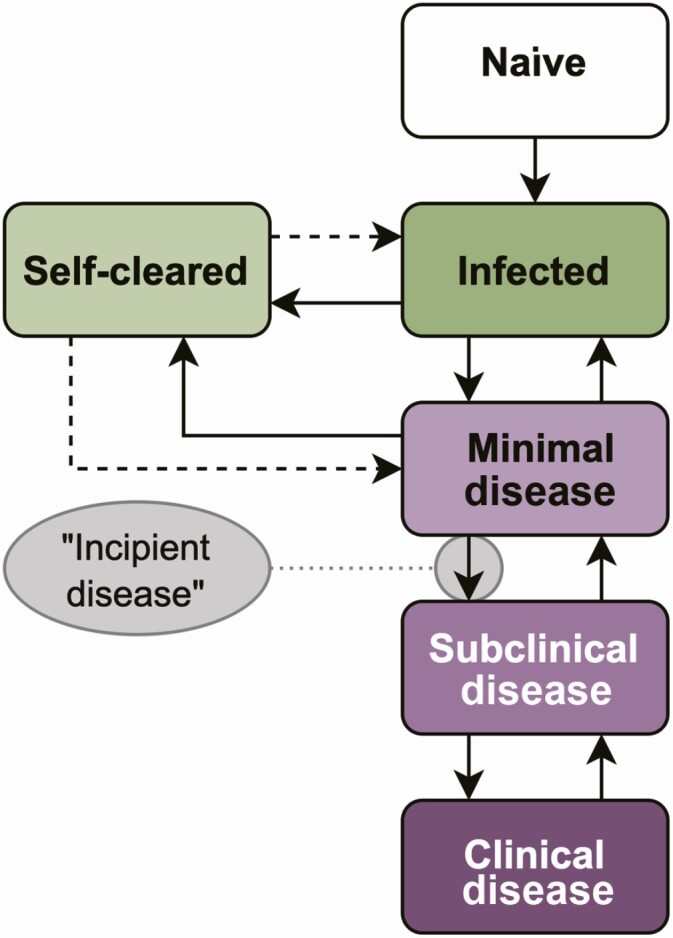
Model representation of the natural history of Mycobacterium tuberculosis (Mtb) infection and tuberculosis disease. Different states of Mtb infection (green) and tuberculosis disease are shown (purple). Infected individuals can progress and regress across the spectrum. Clinical disease: bacteriologically confirmed and symptomatic; incipient disease, transition from minimal to subclinical disease; infected, viable Mtb infection with potential to progress to disease; minimal disease, pathological changes caused by Mtb, but bacteriologically negative; naive-infected-minimal-incipient-subclnical-clinical-self-cleared, individual has cleared the Mtb infection and cannot progress to disease without reinfection (dashed arrows); subclinical disease, bacteriologically confirmed, negative at symptom screening.
Table 4 shows the results from the meta-regression, which provided evidence that in our sample the proportion of subclinical TB cases was higher in surveys from Asia compared with those from Africa (15.2%; 95% confidence interval, 5.6–24.8). There was no evidence for an association with any of the other variables, including country-level TB or HIV prevalence, symptom-screen algorithm, or PDR. Results from the meta-analysis showed very high heterogeneity (I2 = 96%; P < .001). The forest plot is shown in Supplementary Materials Appendix 2.
Table 4.
Survey Level Associations With the Proportion of Prevalent Tuberculosis That Is Subclinical
| Variable (n Observations) | Change in Proportion of Subclinical TB (95% Confidence Interval) | P Value |
|---|---|---|
| Continent (24) | .003 | |
| Africa | Reference | |
| Asia | 15.2% (5.6 to 24.8) | |
| HIV prevalence in country (24) | .34 | |
| Continuous variable | −.07% (−2.0 to .7) | |
| HIV prevalence in country (24) | ||
| <1% | Reference | |
| 1%–2% | −5.4% (−18.9 to 8.1) | .41 |
| ≥2% | −10.9% (−24.4 to 2.7) | .11 |
| Symptom screening (24) | ||
| Any symptom | Reference | |
| Cough ≥2 weeks | −5.0% (−22.1 to 12.1) | .55 |
| Cough ≥2 weeks and/or other symptoms | −10.1% (−26.8 to 6.5) | .22 |
| TB prevalence (23) | .01% (−.01 to .03) | .32 |
| Patient diagnostic rate, average in the previous 5 years (22) | −8.7% (−29.8 to 12.4) | .4 |
| Proportion of males among the cases (21) | .01% (−.8 to 1.0) | .79 |
Results from univariate meta-regression.
Abbreviations: HIV, human immunodeficiency virus; TB, tuberculosis.
DISCUSSION
Where measured, around half of the prevalent infectious TB burden is subclinical, making it likely that ignoring this burden will diminish the impact of TB care and prevention efforts.
Our results show that cough, the cornerstone of symptom-based screening policies, was only self-reported by around half of bacteriologically confirmed cases in populations across Asia and Africa. Expecting extensive population-level impact on transmission from such policies seems misplaced. Similar to historical observations that a large bacillary load is not required for transmission [16, 17], cough is unlikely to be required for transmission [18].
We found that 9 out of 10 individuals with bacteriologically confirmed TB, including those with subclinical disease, were positive on X ray–based screening, which is based on a single posterior–anterior image. We would therefore argue that X ray as a clinical screening tool needs to be reevaluated as part of the End TB Strategy [19]. Aside from its ability to detect the majority of infectious TB, rapid advancements in digitalization, portability of X-ray screening, and computer-aided X-ray reading now enable clear and consistent choices, which can be adjusted to fit the context of each country to further enhance performance [20]. It is now possible to strike a reproducible balance between the need to increase the proportion of all infectious TB found (sensitivity) and the proportion of screened individuals who are referred for bacteriological testing (positivity rate) [20], the latter of which varied between 7.1% and 24% in surveys included in our analysis. As such, X-ray screening can be optimized depending on the population screened, whether these are clinic attendees or community-based.
Prevalence surveys do not capture individuals with symptom-negative, X ray–negative, bacteriologically confirmed TB. While the data are limited, they suggest that another 0%–5% of all bacteriologically confirmed TB would be classified as subclinical [21], which means our estimates for subclinical TB would be conservative. In addition, pediatric and extrapulmonary TB are not measured in prevalence surveys.
Our results are limited to 36% of the global TB burden; therefore, key gaps remain, including China (where surveys have not reported details for bacteriologically confirmed TB cases), India, and South Africa (surveys underway). We strongly argue that surveys should report results separately by screening and bacteriological confirmation, and data could be enriched, for example, with further subdivisions by gender, urban or rural strata, and HIV status to help inform strategies to address this burden. In addition, our data reflect the proportion that is subclinical among the prevalent burden of the infectious disease, not incident disease. Finally, our study does not include data from settings with low TB incidence.
In particular, increased trends over time in the size and composition of the subclinical TB population as the overall TB prevalence changes would improve our understanding of population dynamics. Maximizing the number of repeat data points within countries would enable a within-country analysis of the impact of program performance, including the (limited) ability to address subclinical TB. Our ecological analysis found no association between program performance and subclinical TB, likely due to unmeasured confounding factors specific to each setting. Improved reporting would also provide more data points, which may increase power for more subtle analyses, such as the proportion of subclinical TB by duration of cough, sex, or differences between continents.
We caution for overinterpretation of the evidence for a difference by continent from meta-regression (Table 4) and meta-analysis (Supplementary Materials Appendix 2), especially given that only a subset of countries for each continent is included in our study. Unmeasured confounding factors include differences in the host genetics and bacillary strains that could affect the natural history of the disease [22]. In addition, not all surveys followed the exact same protocol, not all of which was captured in our analysis. Other possible factors are related to cultural differences regarding awareness of symptoms and bacteriological confirmation criteria and techniques. Further studies are necessary to explore the causes and consequences of this result.
Despite the limitations described above, prevalence surveys offer clear advantages as a framework for analysis. First, they represent the most consistent, valid, and extensive effort for TB burden estimation of the past 3 decades [1] and aim to reflect in-country clinical practice and case definitions. As a consequence, we could address the persistent ambiguity of the definitions for subclinical TB, in particular, the precise interpretation of “asymptomatic” and “bacteriologically confirmed.”
Our framework places subclinical TB as a distinct intermediary disease state, which precedes clinical (ie, symptomatic) disease and follows after a minimal disease state. Moreover, incipient disease is not a stage but, as indicated in the name, represents the flow from minimal to subclinical disease. It must be noted that the prevalence of the minimal disease state might be influenced by the limitations of X ray, and more sensitive imaging techniques, such as computed tomography scan, would be more sensitive for initial pathological changes. Progression and regression across the TB natural history spectrum has been postulated and is supported by historical and recent data [23]. The term “incipient TB” has been widely used to refer to a group of individuals who will soon progress to subclinical disease. While this makes it an attractive diagnostic target for predictive tests [24, 25], the word and concept of “incipient” implies both a transition and direction that is a flow, not be a disease state.
Our analysis and conceptual framework should enable scientific discourse and policy progress on the unaddressed burden of subclinical TB. A key consideration is how subclinical TB contributes to transmission, given that individuals do not report (prolonged) cough. However, people may not recognize cough as a symptom, and cough may not be required for effective transmission [4]. A comparison of health-seeking behavior- between individuals with subclinical (asymptomatic) and clinical (symptomatic) disease could shed more light on the impact of recognizing symptoms on accessing care, but unfortunately prevalence surveys did not report the required stratified data. Another advantage is that these disease stages could help distinguish a subpopulation of patients for whom shorter treatment is both beneficial and safe [26].
A significant proportion of the global TB burden is asymptomatic and not detectable by current symptom-based screening efforts, fueling the TB epidemic through continued M. tuberculosis transmission [4]. Detecting subclinical TB provides an opportunity to provide care early in the disease history, which should benefit individuals by preventing extensive lung damage and the risk of post-TB sequelae [27] and benefit society by interrupting transmission. There are both historical and recent precedents to support this thesis, showing that symptom-agnostic screening through X ray [28] or Xpert [29] has near immediate impact on disease burden in high-incidence settings. The TB community needs to recognize both the challenge and opportunities of subclinical TB and develop strategies to address it. If we do so, we should have a much better chance of ending TB in our lifetime.
Supplementary Data
Supplementary materials are available at Clinical Infectious Diseases online. Consisting of data provided by the authors to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the authors, so questions or comments should be addressed to the corresponding author.
Notes
Financial support. This work was supported by European Research Council starting grant funding (TBornotTB, 757699) to R. M. G. J. H., A. S. R., J. C. E., and B. F.
Potential conflicts of interest. The authors: No reported conflicts of interest. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest.
References
- 1. World Health Organization. Global tuberculosis report 2019. Available at: http://www.who.int/tb/publications/global_report/en/. Accessed 13 November 2019.
- 2. Barry CE 3rd, Boshoff HI, Dartois V, et al. The spectrum of latent tuberculosis: rethinking the biology and intervention strategies. Nat Rev Microbiol 2009; 7:845–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. World Health Organization. Systematic screening for active tuberculosis: principles and recommendations. Geneva, Switzerland: World Health Organization, 2013. [PubMed] [Google Scholar]
- 4. Esmail H, Dodd PJ, Houben RMGJ. Tuberculosis transmission during the subclinical period: could unrelated cough play a part? Lancet Respir Med 2018; 6:244–6. [DOI] [PubMed] [Google Scholar]
- 5. Hoa NB, Sy DN, Nhung NV, Tiemersma EW, Borgdorff MW, Cobelens FG. National survey of tuberculosis prevalence in Viet Nam. Bull World Health Organ 2010; 88:273–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Hong Kong Chest Service, Tuberculosis Research Centre, Madras, British Medical Research Council. A study of the characteristics and course of sputum smear-negative pulmonary tuberculosis. Tubercle 1981; 62:155–67. [DOI] [PubMed] [Google Scholar]
- 7. Dorman SE, Schumacher SG, Alland D, et al. ; Study Team . Xpert MTB/RIF Ultra for detection of Mycobacterium tuberculosis and rifampicin resistance: a prospective multicentre diagnostic accuracy study. Lancet Infect Dis 2018; 18:76–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. World Health Organization. Tuberculosis prevalence surveys: a handbook. Available at: http://www.who.int/tb/advisory_bodies/impact_measurement_taskforce/resources_documents/thelimebook/en/. Accessed 13 November 2019.
- 9. Onozaki I, Law I, Sismanidis C, Zignol M, Glaziou P, Floyd K. National tuberculosis prevalence surveys in Asia, 1990–2012: an overview of results and lessons learned. Trop Med Int Health 2015; 20:1128–1145. [DOI] [PubMed] [Google Scholar]
- 10. Center for Social Research, University of Malawi, Centers for Disease Control and Prevention, National Tuberculosis Control Programme (Malawi), World Health Organization. Malawi tuberculosis prevalence survey 2013–2014.
- 11. Horton KC, MacPherson P, Houben RMGJ, White RG, Corbett EL. Sex differences in tuberculosis burden and notifications in low- and middle-income countries: A systematic review and meta-analysis. PLoS Med 2016; 13:e1002119. doi: 10.1371/journal.pmed.1002119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Nyaga VN, Arbyn M, Aerts M. Metaprop: a Stata command to perform meta-analysis of binomial data. Arch Public Health 2014; 72. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4373114/. Accessed 28 August 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Wang L, Zhang H, Ruan Y, et al. Tuberculosis prevalence in China, 1990–2010; a longitudinal analysis of national survey data. Lancet 2014: 383:2057–64. [DOI] [PubMed] [Google Scholar]
- 14. Gothi GD. Natural history of tuberculosis. Indian J Tuberc 1978; 25:13. [Google Scholar]
- 15. Cardona PJ. Revisiting the natural history of tuberculosis. The inclusion of constant reinfection, host tolerance, and damage-response frameworks leads to a better understanding of latent infection and its evolution towards active disease. Arch Immunol Ther Exp (Warsz) 2010; 58:7–14. [DOI] [PubMed] [Google Scholar]
- 16. Behr MA, Warren SA, Salamon H, et al. Transmission of Mycobacterium tuberculosis from patients smear-negative for acid-fast bacilli. Lancet 1999; 353:444–9. [DOI] [PubMed] [Google Scholar]
- 17. Xie YL, Cronin WA, Proschan M, et al. Transmission of Mycobacterium tuberculosis from patients who are nucleic acid amplification test negative. Clin Infect Dis 2018; 67:1653–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Patterson B, Wood R. Is cough really necessary for TB transmission? Tuberculosis (Edinb) 2019; 117:31–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. World Health Organization. Implementing the end TB strategy: the essentials. 2015. Available at: https://www.who.int/tb/publications/2015/end_tb_essential.pdf?ua=1
- 20. Qin ZZ, Sander MS, Rai B, et al. Using artificial intelligence to read chest radiographs for tuberculosis detection: a multi-site evaluation of the diagnostic accuracy of three deep learning systems. Sci Rep 2019; 9:15000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. den Boon S, White NW, van Lill SW, et al. An evaluation of symptom and chest radiographic screening in tuberculosis prevalence surveys. Int J Tuberc Lung Dis 2006; 10:876–82. [PubMed] [Google Scholar]
- 22. Coussens AK, Wilkinson RJ, Nikolayevskyy V, et al. Ethnic variation in inflammatory profile in tuberculosis. PLoS Pathog 2013; 9:e1003468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Houben R, Esmail H, Emery JC, et al. Spotting the old foe—revisiting the case definition for TB. Lancet Respir Med 2019. Available at: https://researchonline.lshtm.ac.uk/id/eprint/4651175/. Accessed 1 December 2019. [DOI] [PubMed] [Google Scholar]
- 24. Consensus meeting report: development of a target product profile (TPP) and a framework for evaluation for a test for predicting progression from tuberculosis infection to active disease. Geneva, Switzerland: World Health Organization; 2017. (WHO/HTM/TB/2017.18). Licence: CC BY-NC-SA 3.0 IGO.pdf. Available at: https://apps.who.int/iris/bitstream/handle/10665/259176/WHO-HTM-TB-2017.18-eng.pdf?sequence=1. Accessed 1 May 2020. [Google Scholar]
- 25. Haas MK, Belknap RW. Diagnostic tests for latent tuberculosis infection. Clin Chest Med 2019; 40:829–37. [DOI] [PubMed] [Google Scholar]
- 26. Imperial MZ, Nahid P, Phillips PPJ, et al. A patient-level pooled analysis of treatment-shortening regimens for drug-susceptible pulmonary tuberculosis. Nat Med 2018; 24:1708–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Allwood B, van der Zalm M, Makanda G, Mortimer K; Steering Committee of the First International Post-Tuberculosis Symposium . The long shadow post-tuberculosis. Lancet Infect Dis 2019; 19:1170–1. [DOI] [PubMed] [Google Scholar]
- 28. Stýblo K, Danková D, Drápela J, et al. Epidemiological and clinical study of tuberculosis in the district of Kolin, Czechoslovakia. Report for the first 4 years of the study (1961–64). Bull World Health Organ 1967; 37:819–74. [PMC free article] [PubMed] [Google Scholar]
- 29. Marks GB, Nguyen NV, Nguyen PTB, et al. Community-wide screening for tuberculosis in a high-prevalence setting. N Engl J Med 2019; 381:1347–57. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.