The monocarboxypeptidase angiotensin-converting enzyme 2 (ACE2) is a major player in the the renin–angiotensin system (RAS) as it converts the decapeptide angiotensin (Ang) I to Ang-(1–9) and Ang II to Ang-(1–7) (figure 1a) [1]. ACE2 is also a target for the new human coronavirus SARS-CoV-2, which is responsible for the dramatic ongoing COVID-19 pandemic [2]. It has been suggested that following SARS-CoV-2/ACE2 internalisation, Ang II level increases [3] in parallel to a decrease of Ang-(1–7) level [4]. These changes would be expected both at tissue and circulatory levels. Considering that Ang-(1–7) has many beneficial effects, including anti-inflammatory, antithrombogenic and antifibrotic activities [1], it has been hypothesised that Ang-(1–7) administration would improve the clinical outcome of COVID-19 patients. Aiming to test this hypothesis, a phase I/II clinical trial (www.clinicaltrials.gov identifier NCT04633772) has been initiated with a planned phase III clinical trial (NCT04332666).
Short abstract
This letter reports an unexpected increase of the ACE2 product angiotensin-(1–7) and a parallel decrease of its substrate angiotensin II, suggesting a dysregulation of the renin–angiotensin system towards angiotensin-(1–7) formation in #COVID19 patients https://bit.ly/3xFXuTU
To the Editor:
The monocarboxypeptidase angiotensin-converting enzyme 2 (ACE2) is a major player in the the renin–angiotensin system (RAS) as it converts the decapeptide angiotensin (Ang) I to Ang-(1–9) and Ang II to Ang-(1–7) (figure 1a) [1]. ACE2 is also a target for the new human coronavirus SARS-CoV-2, which is responsible for the dramatic ongoing COVID-19 pandemic [2]. It has been suggested that following SARS-CoV-2/ACE2 internalisation, Ang II level increases [3] in parallel to a decrease of Ang-(1–7) level [4]. These changes would be expected both at tissue and circulatory levels. Considering that Ang-(1–7) has many beneficial effects, including anti-inflammatory, antithrombogenic and antifibrotic activities [1], it has been hypothesised that Ang-(1–7) administration would improve the clinical outcome of COVID-19 patients. Aiming to test this hypothesis, a phase I/II clinical trial (www.clinicaltrials.gov identifier NCT04633772) has been initiated with a planned phase III clinical trial (NCT04332666).
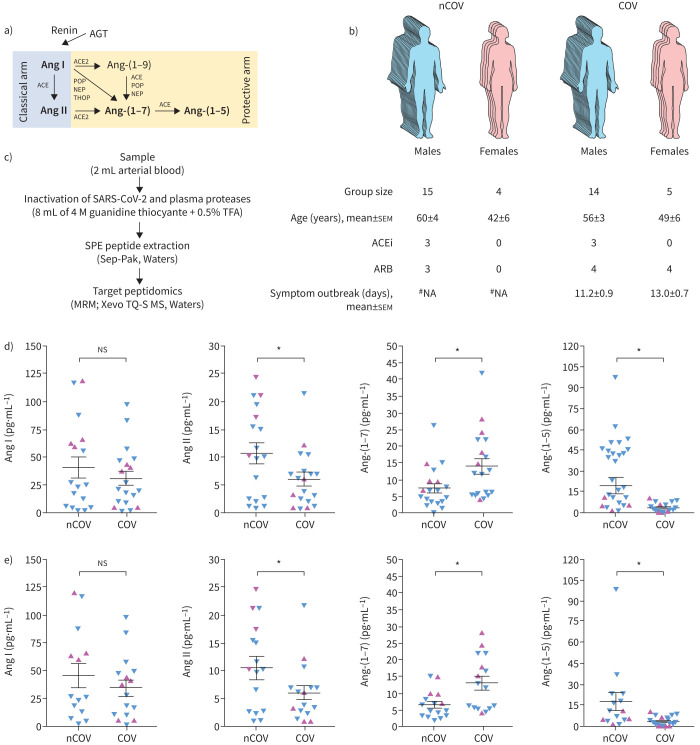
FIGURE 1.
Circulating renin–angiotensin peptides in SARS-CoV-2 infection. a) A noncomprehensive view of the formation of the peptides from the renin–angiotensin system (RAS). Peptides measured in this study are in bold. b) Epidemiological parameters of the subjects included in this study (COVID-19 patients (COV) and non-COVID-19 volunteers (nCOV)). c) A simplified schematic view of the methodology employed to quantify the selected peptides from the RAS. d) Arterial blood concentration of the selected RAS peptides (angiotensin (Ang) I, Ang II, Ang-(1–7) and Ang-(1–5)) from COV and nCOV. e) Arterial blood concentration of the RAS peptides excluding individuals under angiotensin-converting enzyme inhibitor (ACEi) treatment. Blue symbols: men; pink symbols: women. Parametric t-test was used for the statistical analyses. AGT: angiotensinogen; ACE: angiotensin-converting enzyme; POP: prolyloligopeptidase; NEP: neutral endopeptidase; THOP: thimet oligopeptidase; ARB: angiotensin II receptor antagonist; #NA: not applicable; TFA: trifluoroacetic acid; SPE: solid-phase extraction; MRM: multiple reaction monitoring. *: p<0.05.
This report aims to answer the key question of whether the circulating levels of Ang peptides are indeed altered in COVID-19 patients using liquid chromatography–tandem mass spectrometry (LC-MS/MS) (figure 1b). This is important since there are no data in the literature on the concentration of circulating Ang peptides using direct measurements in COVID-19 patients. Others reported that Ang II is higher in the plasma of COVID-19 patients using an ELISA method to quantify Ang II [3]. However, this method has been recently criticised by Chappell et al. [5] due to its poor specificity for the measurement of Ang II and Ang-(1–7) in human plasma. Kintscher et al. [6] have also recently reported “plasma angiotensin peptide profiling” in COVID-19 patients but, even though they used a mass spectrometry-based strategy, they collected the blood using neither protease inhibitors nor under denaturing conditions to “quench” the activity of plasma proteases. They even incubated the heparinised plasma samples at 37°C to increase proteases activities and thus measure what they call “peptides’ equilibrium concentrations” (named the “equilibrium method” here). This method allows only the indirect measurement of RAS peptides as it does not account for the important role of enzymes on the endothelium; for example, in the production or hydrolysis of RAS peptides in the circulation. Here, we are reporting the direct measurement of the RAS peptides Ang I, Ang II, Ang-(1–7) and Ang-(1–5) in arterial blood samples from 19 patients with severe COVID-19 and 19 non-COVID-19 volunteers. The study protocol has been approved by the Ethics Committee of the Federal University of Minas Gerais, Belo Horizonte, Brazil (CAAE 34080720.0.1001.5149). Written consent was obtained from the patients or their relatives for blood sampling. The COVID-19 patients were recruited from two hospitals (Mater Dei Hospital and Eduardo de Menezes Hospital, Belo Horizonte, Minas Gerais, Brazil). Arterial blood samples were obtained from patients admitted at the hospitals’ intensive care unit (ICU) upon admission and before starting any additional treatment (medication, oxygen therapy, etc.) in the ICU. The arterial samples from non-COVID-19 subjects were collected from healthy volunteers (n=6) or before cardiac catheterisation (n=13). Importantly, only the arterial blood samples from patients with no diagnostic for cardiovascular diseases, as per the catheterisation results, were included in the non-COVID-19 group. Three COVID-19 patients and three non-COVID-19 individuals were previously using angiotensin-converting enzyme inhibitors (ACEi) (figure 1b) and the treatment was not interrupted at any time.
A mass spectrometry-based approach was used to quantify the RAS peptides. Inactivation of plasma proteases and potential SARS-CoV-2 in the blood samples was achieved by mixing 2 mL arterial blood sample with 8 mL denaturation solution (4 M guanidine thiocyanate in 0.5% TFA). Samples (1200 µL) were cleaned up using C18 solid-phase extraction (Sep-Pak, Waters), reconstituted in 60 µL 0.1% formic acid (concentration factor 20×) and analysed by LC-MS/MS (Xevo TQ-S, Waters) using the multiple reaction monitoring mode (figure 1c). The calibration curve was obtained using a stock solution containing synthetic peptides (Bachem) of all RAS peptides used in this study. The applied calibration curve model (y=ax+b) proved accurate over the concentration range from 10 to 1000 pg·mL−1 (r=0.997). The limit of quantitation, and inter- and intravariability of this method have been previously reported [7]. Data are presented as mean±sem and the parametric t-test was used for the statistical analyses.
Arterial concentrations of the Ang peptides are shown in figure 1d. In COVID-19 patients, the arterial concentration of Ang II (6.03±1.18 versus 10.7±1.87 pg·mL−1; p=0.0381) and Ang-(1–5) (3.43±0.75 versus 19.3±5.80 pg·mL−1; p=0.0084) were significantly lower than in the non-COVID-19 volunteers. Surprisingly, the blood levels of Ang-(1–7) were significantly higher in COVID-19 patients (14.0±2.32 versus 7.49±1.42 pg·mL−1; p=0.0214). No significant difference was observed for Ang I (31.2±6.23 versus 40.8±9.54 pg·mL−1; p=0.3959). ACEi therapy did not significantly change the observed results, as shown by the calculated values excluding the data from individuals under ACEi treatment (figure 1e): Ang I 34.5±7.07 versus 45.2±10.8 pg·mL−1, p=0.4023; Ang II 6.00±1.33 versus 11.2±2.07, p=0.0407; Ang-(1–7) 12.9±2.02 versus 6.47±1.03 pg·mL−1, p=0.0080; Ang-(1–5) 3.75±0.86 versus 17.5±6.29 pg·mL−1, p=0.0330.
Although we did not measure the tissular levels of RAS peptides, our findings contrast with the initial hypothesis that the interaction of SARS-CoV-2 with ACE2 would result in higher Ang II and lower Ang-(1–7) levels compared to non-COVID-19 subjects [4]. Recent studies using the equilibrium method to measure the ACE2 activity [8, 9] are in line with the results presented here, as they reported higher Ang-(1–7) and lower Ang II plasma levels in severe COVID-19. For general clinical studies including nonsevere COVID-19 patients, it seems that all circulating RAS peptides are reduced due to decreased activity of renin [10]. The role of ACE2 in the observed results is questionable as previous studies suggested that one of the main routes to produce Ang-(1–7) in the circulation is by ACE2-independent pathways [11, 12]. The observed significant decrease of Ang II and increase of Ang-(1–7) arterial levels in severe COVID-19 patients (figure 1d) is probably due to a direct dysregulation of RAS pathways in COVID-19 rather than a direct consequence of ACEi usage (figure 1e). The Ang-(1–7)/Ang II ratio, which is an estimation of Ang II→Ang-(1–7) conversion, was three-fold higher in COVID-19 patients (2.79±0.682 versus 0.878±0.201; p=0.0141), which may suggest an increased ACE2 or other Ang-(1–7)-forming activity in COVID-19. Increased soluble ACE2 in severe COVID-19 patients have been recently reported [9, 13, 14]. Reindl-Schwaighofer et al. [9] reported that ACE2 level increased over the course of the disease, reaching its maximum peak after ∼10 days of hospitalisation. Increased ACE2 correlated with increased Ang-(1–7) and decreased Ang II levels [9], suggesting its important role in controlling the circulating RAS peptides in severe COVID-19.
In contrast to a previous report [3], we observed a significant reduction of Ang II concentration in COVID-19 patients, which may add more data against the reliability of ELISA to measure Ang II in human plasma [5]. The Ang II/Ang I ratio was not significantly altered in the COVID-19 patients (0.205±0.0322 versus 0.292±0.0517; p=0.1518), which may suggest that ACE activity is not altered in COVID-19. Unfortunately, we were unable to perform direct measurements of enzymatic activities in this study due to the denaturating conditions that we used to collect the samples, but our estimation of increased ACE2 activity in severe COVID-19 blood samples is in line with previous reports [9, 14]. The remaining question to be answered, though, is whether the observed increased Ang-(1–7) and decreased Ang II levels are a direct effect of SARS-CoV-2 or a consequence of inflammation due to the infection that triggers Ang-(1–7) synthesis via Ang II metabolisation.
Limitations of our study includes the small cohort included (n=19 for each group) and the lack of direct activity measurements of the RAS-related enzymes. Nonetheless, this is the first report of the direct measurement of RAS peptides in the arterial circulation of severe COVID-19 patients. Although future studies are obviously necessary to better understand the effects of this disease on RAS pathways, our data provide new insights for the interpretation and planning of future therapies to modulate the RAS in the context of COVID-19.
Footnotes
Ethics statement: The study protocol has been approved by the Ethics Committee of the Federal University of Minas Gerais, Belo Horizonte, Brazil (CAAE 34080720.0.1001.5149). Written consent was obtained from the patients or their relatives for blood sampling.
Conflict of interest: A.L. Valle Martins reports a patent pending (BR1020210009950).
Conflict of interest: F.A. da Silva has nothing to disclose.
Conflict of interest: L. Bolais-Ramos has nothing to disclose.
Conflict of interest: G.C. de Oliveira has nothing to disclose.
Conflict of interest: R.C. Ribeiro has nothing to disclose.
Conflict of interest: D.A.A. Pereira has nothing to disclose.
Conflict of interest: F. Annoni has nothing to disclose.
Conflict of interest: M.M.L. Diniz has nothing to disclose.
Conflict of interest: T.G.F. Silva has nothing to disclose.
Conflict of interest: B. Ziviani has nothing to disclose.
Conflict of interest: A.C. Cardoso has nothing to disclose.
Conflict of interest: J.C. Martins has nothing to disclose.
Conflict of interest: D. Motta-Santos has nothing to disclose.
Conflict of interest: M.J. Campagnole-Santos has nothing to disclose.
Conflict of interest: F.S. Taccone has nothing to disclose.
Conflict of interest: T. Verano-Braga has nothing to disclose.
Conflict of interest: R.A.S. Santos reports patents planned.
Support statement: This work was supported by the Research Support Foundation of the State of Minas Gerais (FAPEMIG), grant number APQ-00325-20, and Angitec. M.J. Campagnole-Santos, T. Verano-Braga and R.A. Souza Santos also acknowledge the National Council for Scientific and Technological Development (CNPq) for the personal support (#306962/2019-5, # 309122/2019-8 and #310515/2015-7, respectively).
References
- 1.Santos RAS, Sampaio WO, Alzamora AC, et al. The ACE2/angiotensin-(1-7)/MAS axis of the renin-angiotensin system: focus on angiotensin-(1–7). Physiol Rev 2018: 98: 505–553. doi: 10.1152/physrev.00023.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020: 181: 271–280. doi: 10.1016/j.cell.2020.02.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Liu Y, Yang Y, Zhang C, et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci 2020: 63: 364-374. doi: 10.1007/s11427-020-1643-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Peiro C, Moncada S. Substituting angiotensin-(1–7) to prevent lung damage in SARS-CoV-2 infection? Circulation 2020: 141: 1665–1666. doi: 10.1161/CIRCULATIONAHA.120.047297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chappell MC, Pirro NT, South AM, et al. Concerns on the specificity of commercial ELISAs for the measurement of angiotensin-(1–7) and angiotensin II in human plasma. Hypertension 2021; 77: e29–e31. doi: 10.1161/HYPERTENSIONAHA.120.16724 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kintscher U, Slagman A, Domenig O, et al. Plasma angiotensin peptide profiling and ACE (angiotensin-converting enzyme)-2 activity in COVID-19 patients treated with pharmacological blockers of the renin–angiotensin system. Hypertension 2020: 76: e34–e36. doi: 10.1161/HYPERTENSIONAHA.120.15841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Paquette K, Fernandes RO, Xie LF, et al. Kidney size, renal function, Ang (angiotensin) peptides, and blood pressure in young adults born preterm. Hypertension 2018: 72: 918–928. doi: 10.1161/HYPERTENSIONAHA.118.11397 [DOI] [PubMed] [Google Scholar]
- 8.van Lier D, Kox M, Santos K, et al. Increased blood angiotensin converting enzyme 2 activity in critically ill COVID-19 patients. ERJ Open Res 2021; 7: 00848-2020. doi: 10.1183/23120541.00848-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Reindl-Schwaighofer R, Hodlmoser S, Eskandary F, et al. Angiotensin-converting enzyme 2 (ACE2) elevation in severe COVID-19. Am J Respir Crit Care Med 2021; 203: 1191–1196. doi: 10.1164/rccm.202101-0142LE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kutz A, Conen A, Gregoriano C, et al. Renin–angiotensin–aldosterone system peptide profiles in patients with COVID-19. Eur J Endocrinol 2021: 184: 543–552. doi: 10.1530/EJE-20-1445 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Serfozo P, Wysocki J, Gulua G, et al. Ang II (angiotensin II) conversion to angiotensin-(1–7) in the circulation is POP (prolyloligopeptidase)-dependent and ACE2 (angiotensin-converting enzyme 2)-independent. Hypertension 2020: 75: 173–182. doi: 10.1161/HYPERTENSIONAHA.119.14071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Santos RA, Brosnihan KB, Jacobsen DW, et al. Production of angiotensin-(1–7) by human vascular endothelium. Hypertension 1992: 19: Suppl. 2, II56–II61. [DOI] [PubMed] [Google Scholar]
- 13.Burns K, Cheng M, Lee T, et al. Sustained dysregulation of the plasma renin–angiotensin system in acute COVID-19. Res Square 2021; pre-print [ 10.21203/rs.3.rs-125380/v1]. [DOI] [Google Scholar]
- 14.Patel SK, Juno JA, Lee WS, et al. Plasma ACE2 activity is persistently elevated following SARS-CoV-2 infection: implications for COVID-19 pathogenesis and consequences. Eur Respir J 2021; 57: 2003730. doi: 10.1183/13993003.03730-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]