Abstract
The microbial populations in the gut microbiome have recently been associated with COVID-19 disease severity. However, a causal impact of the gut microbiome on COVID-19 patient health has not been established. Here we provide evidence that gut microbiome dysbiosis is associated with translocation of bacteria into the blood during COVID-19, causing life-threatening secondary infections. Antibiotics and other treatments during COVID-19 can potentially confound microbiome associations. We therefore first demonstrate that the gut microbiome is directly affected by SARS-CoV-2 infection in a dose-dependent manner in a mouse model, causally linking viral infection and gut microbiome dysbiosis. Comparison with stool samples collected from 101 COVID-19 patients at two different clinical sites also revealed substantial gut microbiome dysbiosis, paralleling our observations in the animal model. Specifically, we observed blooms of opportunistic pathogenic bacterial genera known to include antimicrobial-resistant species in hospitalized COVID-19 patients. Analysis of blood culture results testing for secondary microbial bloodstream infections with paired microbiome data obtained from these patients suggest that bacteria translocate from the gut into the systemic circulation of COVID-19 patients. These results are consistent with a direct role for gut microbiome dysbiosis in enabling dangerous secondary infections during COVID-19.
Keywords: microbes, COVID-19, secondary infectionsgut microbiome
A better understanding of factors contributing to the pathology of coronavirus disease 2019 (COVID-19) is an urgent global priority. Infections by SARS-CoV-2 are frequently asymptomatic or mild in nature, but may also cause a broad range of severe and life-threatening symptoms. Previous reports have demonstrated that severe COVID-19 is frequently associated with specific inflammatory immune phenotypes, lymphopenia, and a generally disproportionate immune response leading to systemic organ failure1,2. Even in mild cases, gastrointestinal symptoms are reported frequently, and recent studies reported that COVID-19 patients lose commensal taxa of the gut microbiome during hospitalization3,4. Differences in gut bacterial populations relative to healthy controls were observed in all COVID-19 patients, but most strongly in patients who were treated with antibiotics during their hospitalization4. Most recently, COVID-19 patients treated with broad spectrum antibiotics at admission were shown to have increased susceptibility to multi-drug resistant infections and nearly double the mortality rate from septic shock5,6, and a recent meta-analysis found that over 14% of 3,338 COVID-19 patients acquired a secondary bacterial infection7. However, the causal direction of the relationship between disease symptoms and gut bacterial populations is not yet clear.
Complex gut microbiota ecosystems can prevent the invasion of potentially pathogenic bacteria8,9. Conversely, when the gut microbiota incurs damage, such as through antibiotics treatment, competitive exclusion of pathogens is weakened10 and potentially dangerous blooms of antibiotic resistant bacterial strains can occur 11,12. In immunocompromised cancer patients, blooms of Enterococcaceae and Gram-negative proteobacteria can lead to gut dominations by few or single species13–16. Gut domination events are dangerous to these patients because antibiotic resistant bacteria may translocate from the gut into the blood stream. Consequently, enterococcal dominations have been associated with 9-fold increased risk of bloodstream infections (BSIs) with vancomycin-resistant Enterococcus (VRE), and domination by Gram-negative proteobacteria with 5-fold increased risk of Gram-negative rod BSIs13. Bacterial co-infection can also cause life-threatening complications in patients with severe viral infections6,17; therefore, antibacterial agents were administered empirically to nearly all critically ill suspected COVID-19 patients since the incidence of bacterial superinfection was unknown early during the pandemic4,18. However, it is now known that nosocomial infection during prolonged hospitalization is the primary threat to patients with COVID-1919, rather than bacterial co-infection upon hospital admission7,20–22. Evidence from immunocompromised cancer patients suggests that indiscriminate administration of broad-spectrum antibiotics may, counter-intuitively, increase nosocomial BSI rates by causing gut dominations of resistant microbes that can translocate into the blood13,23. Thus, empiric antimicrobial use, i.e. without direct evidence for a bacterial infection, in patients with severe COVID-19 may be especially pernicious because it may select for antimicrobial resistance and could promote gut translocation-associated BSI.
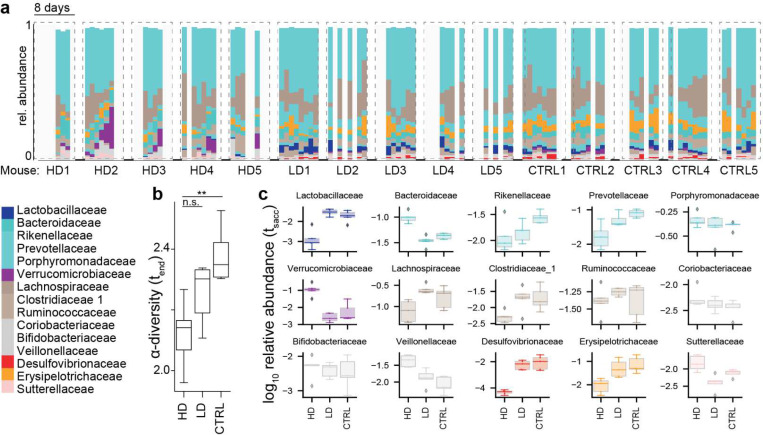
The role of the gut microbiome in respiratory viral infections in general24, and in COVID-19 patients in particular, is only beginning to be understood. Animal models of influenza virus infection have uncovered mechanisms by which the microbiome influences antiviral immunity25–27, and in turn, the viral infection was shown to disrupt the intestinal barrier of mice by damaging the gut microbiota28,29. Hence, we hypothesized that gut dysbiosis during COVID-19 may be associated with BSIs. To test this, we first determined whether SARS-CoV-2 infection could directly cause gut dysbiosis independently of hospitalization and treatment. Daily changes in fecal bacterial populations were monitored following intranasal inoculation of transgenic mice expressing human ACE2 driven by the cytokeratin-18 promoter (K18-ACE2tg mice) with either a high dose (HD,104PFU) or low dose (LD, 10PFU) of SARS-CoV-2 (Figure 1a). Although disease was not as evident in LD mice, we confirmed the presence of infectious virus in the lung by plaque assay at sacrifice (Supplementary Figure S1). Among the HD mice, we observed significant microbiome changes (Figure 1b), with a repeatedly observed community trajectory corresponding to a loss in relative abundances of obligate anaerobe species such as members of the Clostridiales order (Figure 1c), concurrent with an expansion of Verrucomicrobiales (Figure 1a,c). During this shift in the microbiome, α-diversity in the gut bacterial ecosystem was decreasing, a trend also observed in the LD mice, albeit to a lesser extent, but not in the control mice (Figure 1b). After less than one week of viral infection, α-diversity was reduced in infected mice (Figure 1b, 95%HDI<0 Bayesian estimation of differences in group means BEST, methods). Alongside the progressive increase in microbiota compositional dysbiosis, we also observed systemic signs of severe infection, including weight loss (Supplementary Figure S2), as well as ruffled fur, heavy breathing, reduced activity and hunched posture (Supplementary Table S1). These results demonstrate that SARS-CoV2 infection directly causes gut microbiome dysbiosis in a mouse model.
Figure 1. SARS-CoV-2 infection causes gut microbiome alterations in mice.
a Timelines of fecal microbiota composition measured by 16S rRNA gene sequencing in mice infected with high (HD, 104PFU), or low doses (LD, 10PFU) and in uninfected control mice (CTRL); time of infection=Day 1. Bars represent the composition of the 30 most abundant bacterial families per sample, blocks of samples correspond to an individual mouse’s time course. b α-diversity (Shannon) in the final samples per infection group; **: HDI95<0 BEST. c log10-relative family abundances at the final time point. heavy breathing, reduced activity and hunched posture (Supplementary Table S1).
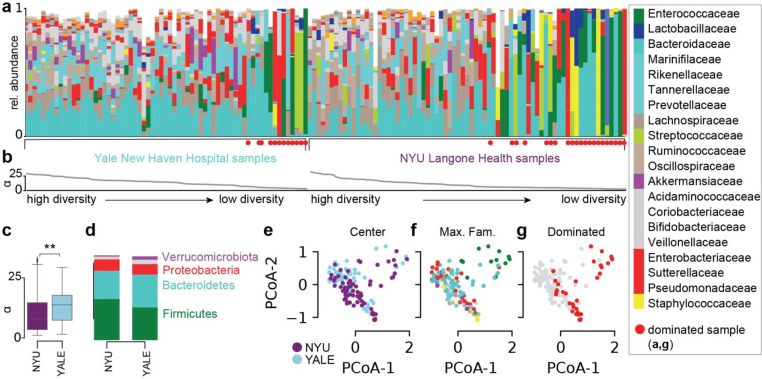
We next profiled the bacterial composition of the fecal microbiome in 138 samples (Figure 2a) obtained from SARS-CoV-2 infected patients treated at NYU Langone Health (NYU, 73 samples, Supplementary Table S2) and Yale New Haven Hospital (YALE, 65 samples, Supplementary Table S3). Analysis of metagenomic data obtained from sequencing of the 16S rRNA genes revealed a wide range of bacterial community diversities, as measured by the inverse Simpson index, in samples from both centers (NYU: [1.0, 32.2], YALE: [1.5, 29.3], Figure 2b); on average, samples from NYU were less diverse (−2.5, p<0.01, Figure 2c). However, the composition in samples between the two centers did not show systematic compositional differences (Figure 2d,e,f). On average, in both centers, members of the phyla Firmicutes and Bacteroidetes represented the most abundant bacteria, followed by Proteobacteria (Figure 2d). The wide range of bacterial diversities was reflected in the high variability of bacterial compositions across samples (Figure 2e,f). In samples from both centers, microbiome dominations, defined as a community where a single genus reached more than 50% of the population, were observed frequently (NYU: 21 samples, YALE: 12 samples), representing states of severe microbiome injury in COVID-19 patients (Figure 2g).
Figure 2. Gut microbiome bacterial compositions during COVID-19 in patients from NYU Langone Health and Yale New Haven Hospital.
a Bacterial family composition in stool samples identified by 16S rRNA gene sequencing; bars represent the relative abundances of bacterial families; red circles indicate samples with single taxa >50%. Samples are sorted by the bacterial α-diversity (inverse Simpson index, b). c α-diversity in samples from NYU Langone Health and Yale New Haven Hospital. d Average phylum level composition per center. e-g Principal coordinate plots of all samples shown in a, labeled by center (e), most abundant bacterial family (f) and domination status of the sample (g).
In agreement with a recent study associating gut microbial compositions with disease severity4, we found that samples from patients who were treated in the ICU had reduced bacterial diversity (Supplementary Figure S3). In 22 cases, gastrointestinal symptoms were recorded, but only two of those patients required ICU treatment and corresponding stool samples had higher average diversity than other samples (Supplementary Figure S3). Strikingly, however, samples associated with a BSI had strongly reduced bacterial α-diversities (mean difference: −7.7, CIBEST[−10.2, −5.2], Supplementary Figure S3).
The lower diversity associated with samples from 21 patients with BSIs led us to investigate their bacterial taxon compositions and the potential that gut dysbiosis was associated with BSI events. All BSI patients had received antibiotic treatments during hospitalization, which could exacerbate COVID-19 induced shifts in microbiota populations11,12,15, but may indeed be administered in response to a suspected or confirmed BSI. However, we noted that most BSI patients (80%) also received antibiotics prior to their BSI. Principal coordinate analysis of all stool samples indicated that the BSI-associated samples spanned a broad range of compositions (Figure 3a). To identify bacterial abundance patterns that consistently distinguished BSI from non-BSI-associated samples, we next performed a Bayesian logistic regression. This analysis estimated the association of the 10 most abundant bacterial genera with BSI cases, i.e. it identified enrichment or depletion of bacterial genera in BSI associated samples (Figure 3b). This analysis revealed that the genus Faecalibacterium was negatively associated with BSI (OR: −1.49, CI:[−2.82, −0.18]). Faecalibacterium is an immunosupportive Clostridiales genus that is a prominent member of the human gut microbiome30–32, and its reduction is associated with disruption to intestinal barrier function33,34, perhaps via ecological network effects34.
Figure 3. Microbiome composition is associated with secondary bloodstream infections.
a Principal coordinate plot of all samples, BSI associated samples in orange. b Posterior coefficient estimates from a Bayesian logistic regression regressing log10 relative abundances of the top 10 most abundant bacterial genera on BSI status. c Posterior prediction of BSI risk based on bacterial composition contrasting the predicted risk of the average composition across all samples (red) with the risk estimated for the same composition changed such that Faecalibacterium was increased by 10% (blue). d Log10 relative abundances of Faecalibacterium correlated with α-diversity, shaded region: 95%CI. e Sample compositions with BSIs indicated; left: Staphylococcus BSI associated samples; right: other BSI associated samples, the BSI causing microbial genus annotated in colors corresponding to the colors in stool microbiome compositions. f Rank analysis of abundance patterns in stool samples from different BSI categories; a filled circle indicates the calculated rank of the focal BSI category (row) in terms of the corresponding taxon stool abundance relative to samples from other BSI categories (only 5 out of 7 BSI categories are shown because fungal BSIs and the uninfected category have no corresponding bacterial stool abundances). g Posterior coefficients of the statistical association between bacterial order log10 relative abundances of BSI causing bacteria and BSI events from a hierarchical Bayesian categorical regression; **: 95%HDI>0, *: 90%HDI>0, .:85% HDI>0.
To evaluate the effect size of the association between Faecalibacterium and BSIs, we performed a posterior predictive check. Using the average genus composition found across all samples, we first computed the distribution of predicted BSI risks (Figure 3c), and compared this risk distribution with a hypothetical bacterial composition which increased Faecalibacterium by 10% points. The predicted risk distributions associated with these two compositions differed strongly (mean difference 26%, CI: [−9%, 67%], Figure 3c). Domination states of the microbiome increase the risk for BSIs in immunocompromised cancer patients 13; such dominations imply high relative abundances of single taxa, and therefore a low diversity. Consistent with this, Faecalibacterium abundance was positively correlated with diversity (R: 0.55, p<10−10, Figure 3d) in our data set and as reported previously 30.
We therefore next investigated a direct association between the bacteria populating the gut microbiome and the organisms identified in the blood of patients. Visualizing the bacterial composition in stool samples from patients alongside the BSI microorganism (Supplementary Figure S4, Figure 3e) suggested a correspondence with the respective taxa identified in the blood: high abundances of the BSI-causing microbes were found in corresponding stool samples (Figure 3e). To analyze this, we first assigned stool samples associated with each BSI event into 5 categories defined by the taxonomic order of the causative bacterial organisms, as well as one singleton group of a fungal infection case as a sixth category; stool samples from uninfected patients were assigned a seventh, “uninfected” category. For samples of each BSI category, we first calculated their median abundances of bacterial predictors in the stool. We then ranked these stool taxon median abundances across BSI categories. As expected from the visualization of sample compositions (Figure 3e), we found that BSI category sample sets were generally enriched in their respective taxa in the stool. For example, samples associated with Klebsiella, Escherichia or Serratia BSIs (Enbct category, Figure 3f) had the highest rank of Enterobacterales abundances across the BSI category sample sets (Figure 3f). We tested this observation statistically using the log10-relative bacterial abundances in stool samples as independent predictors of identified BSI pathogens, i.e. the BSI category, in a Bayesian categorical regression model where the uninfected class was used as a pivot (see methods). In addition to taxon abundances, the model included the bacterial α-diversity as a predictor. As expected, a strong statistical association between diversity and BSIs in general was detected (Supplementary Figure S5). The rank analysis had suggested that Staphylococcales are not enriched in BSIs by Staphylococcus (Figure 3f); this was supported by the Bayesian model which showed that log10-abundances of Staphylococcales in the stool were not detectably predictive of a Staphylococcus BSI (Figure 3g). By contrast, our analysis demonstrated that the bacterial abundances of all other BSI-causing organisms in the stool were predictive of corresponding BSIs.
Collectively, these results reveal an unappreciated link between SARS-CoV-2 infection, gut microbiome dysbiosis, and a major complication of COVID-19, BSIs. The loss of diversity and immunosupportive Faecalibacterium in patients with BSIs mirrored a similar loss of diversity and Clostridiales in the mice receiving high doses of SARS-CoV-2, suggesting that this virus causally affects the microbiome, either through direct infection35–39 or through a systemic inflammatory response 2,4. However, the dysbiosis in patients with COVID-19 exceeded the microbiota shifts observed in the mouse experiments, including microbiome dominations by single taxa, which was not seen in the mouse experiments. It is possible that in our experiment, mice were sacrificed before perturbations to the gut microbial populations reached a maximum. However, it is also plausible that the frequently administered antibiotic treatments that hospitalized COVID-19 patients receive exacerbated SARS-CoV-2 induced microbiome perturbations. Additionally, unlike the controlled environment experienced by laboratory mice, hospitalized patients are uniquely exposed to antimicrobial-resistant infectious agents present on surfaces and shed by other patients. Indeed, domination events where the gut is populated by only a few taxa have been described in hospitalized, immunocompromised cancer patients treated with broad spectrum antibiotics15. We frequently observed such dominations in our COVID-19 cohorts treated at two hospitals.
Our observation that the type of bacteria that entered the bloodstream was disproportionately enriched in the associated stool samples is a well characterized phenomenon in cancer patients13, especially during chemotherapy induced leukocytopenia when patients are severely immunocompromised11,30. COVID-19 patients are also immunocompromised and frequently incur lymphopenia, rendering them susceptible to secondary infections40. Our data suggests dynamics in COVID-19 patients may be similar to those observed in cancer patients: BSI-causing organisms may translocate from the gut into the blood, potentially due to loss of gut barrier integrity, through virus-induced tissue damage rather than chemotherapy. Consistent with this possibility, soluble immune mediators such as TNFa and interferons produced during viral infections, including SARS-CoV-2, damage the intestinal epithelium to disrupt the gut barrier, especially when the inflammatory response is sustained as observed in patient with severe COVID-1941–43.
One limitation of our data is temporal ordering of samples. Occasionally stool samples were collected after observation of BSI, and this mismatch in temporal ordering is counter intuitive for gut-to-blood translocation and a causal interpretation of our associations. However, the reverse direction, that blood infection populates and changes the gut community, is unlikely for the organisms identified in the blood, and if our associations were not causal, we would expect no match between BSI organisms and stool compositions.
Taken together, our findings support a scenario in which gut-to-blood translocation of microorganisms following microbiome dysbiosis, a known issue for chronic conditions such as cancer, leads to dangerous BSIs during COVID-19. We suggest that investigating the underlying mechanism behind our observations will inform the judicious application of antibiotics and immunosuppressives in patients with respiratory viral infections and increase our resilience to pandemics.
Materials and Methods
Bioethics statement
The collection of COVID-19 human biospecimens for research has been approved by the NYUSOM Institutional Review Board under il8–01121 Inflammatory Bowel Disease and Enteric Infection at NYU Langone Health. The data presented in this study were also approved by Yale Human Research Protection Program Institutional Review Boards (FWA00002571, protocol ID 2000027690). Informed consent was obtained from all enrolled patients.
Mouse experiments
Cells & virus
Vero E6 (CRL-1586; American Type Culture Collection) were cultured Dulbecco’s Modified Eagle’s Medium (DMEM,Corning) supplemented with 10% fetal bovine serum (FBS, Atlanta Biologics) and 1% nonessential amino acids (NEAA,Corning). SARS-CoV-2, isolate USA-WA1/2020 19 (BEI resources #NR52281), a gift from Dr. Mark Mulligan at the NYU Langone Vaccine Center was amplified once in Vero E6cells. All experiments with SARS-CoV-2 were conducted in the NYU Grossman School of Medicine ABSL3 facility by personnel equipped with powered air-purifying respirators.
Mice
Heterozygous K18-hACE2 C57BL/6J mice (strain: 2B6.Cg-Tg(K18-ACE2)2Prlmn/J) were obtained from The Jackson Laboratory. Animals were housed in groups and fed standard chow diets. All animal studies were performed according to protocols approved by the NYU School of Medicine Institutional Animal Care and Use Committee (IACUC n°170209). 24-week-old K18-hACE2 males were administered either 10PFU SARS-CoV-2 (low dose, LD), 104PFU SARS-CoV-2 (high dose, HD) diluted in 50μL PBS (Corning) or 50μL PBS (non-infected, CTRL) via intranasal administration under xylazine-ketamine anesthesia (AnaSedR AKORN Animal Health, KetathesiaTM Henry Schein Inc). Viral titer in the inoculum was verified by plaque assay in Vero E6 cells. Following infection, mice were monitored daily for weight loss and signs of disease. Stool samples were collected and stored at −80°C.
Measurement of viral load by plaque assay
Six or seven days after infection, mice were sacrificed. For some mice lungs were collected in Eppendorf tubes containing 500μl of PBS and a 5mm stainless steel bead (Qiagen) and homogenized using with the Qiagen TissueLyser II. Homogenates were cleared for 5 min at 5,000 × g, and viral supernatant was frozen at −80°C for titration through plaque assay. In brief, Vero E6 cells were seeded at a density of 2.2 * 105 cells per well in flat-bottom 24-well tissue culture plates. The following day, media was removed and replaced with 100μL of tenfold serial dilutions of the virus stock, diluted in infection medium. Plates were incubated for 1h at 37°C. Following incubation, cells were overlaid with 0.8% agarose in DMEM containing 2% FBS and incubated at 37°C for 72hrs. Cells were then fixed with formalin buffered 10% (Fisher Chemical) for 1h. Agarose plugs were then removed and cells were stained for 20 min with crystal violet and then washed with tap water.
Human study population and data collection
This study involved 101 patients with laboratory-confirmed SARS-CoV-2 infection. SARS-CoV-2 infection was confirmed by a positive result of real-time reverse transcriptase-polymerase chain reaction assay on a nasopharyngeal swab. 64 patients were seen at NYU Langone Health, New York, for routine medical procedures, outpatient care, or admitted through the Emergency Department at NYU Langone Health’s Tisch Hospital, New York City, between January 29, 2020 – July 2, 2020 and were followed until discharge. In order to be eligible for inclusion in the study, stool specimens needed to be from individuals >18 years of age. Data including demographic information, clinical outcomes, and laboratory results were extracted from the electronic medical records in the NYU Langone Health clinical management system. Blood and stool samples were collected by hospital staff. OmnigeneGut kits were used on collected stool. In parallel, 37 patients were admitted to YNHH with COVID-19 between 18 March 2020 and 27 May 2020 as part of the YALE IMPACT cohort described at length elsewhere2. Briefly, participants were enrolled after providing informed consent and paired blood and stool samples were collected longitudinally where feasible for duration of hospital admission. No statistical methods were used to predetermine sample size for this cohort. Demographic information of patients was aggregated through a systematic and retrospective review of the EHR and was used to construct Supplementary Table 3. Symptom onset and aetiology were recorded through standardized interviews with patients or patient surrogates upon enrolment in our study, or alternatively through manual EHR review if no interview was possible owing to clinical status at enrolment. The clinical data were collected using EPIC EHR and REDCap 9.3.6 software. At the time of sample acquisition and processing, investigators were blinded to patient clinical status.
DNA extraction and bacterial 16S rRNA sequencing
For bacterial DNA extraction 700μL of SL1 lysis buffer (NucleoSpin Soil kit, Macherey-Nagel) was added to the stool samples and tubes were heated at 95°C for 2h to inactivate SARS-CoV-2. Samples were then homogenized using the FastPrep-24TM instrument (MP Biomedicals) and extraction was pursued using the NucleoSpin Soil kit according to the manufacturer’s instructions. DNA concentration was assessed using a NanoDrop spectrophotometer. Samples with too low DNA concentration were excluded. DNA from human samples was extracted with PowerSoil Pro (Qiagen) on the QiaCube HT (Qiagen), using Powerbead Pro (Qiagen) plates with 0.5mm and 0.1mm ceramic beads. For mouse samples, the variable region 4 (V4) of the 16S rRNA gene was amplified by PCR using primers containing adapters for MiSeq sequencing and single-index barcodes. All PCR products were analyzed with the Agilent TapeStation for quality control and then pooled equimolar and sequenced directly in the Illumina MiSeq platform using the 2×250 bp protocol. Human samples were prepared with a protocol derived from 44, using KAPA HiFi Polymerase to amplify the V4 region of the 16S rRNA gene. Libraries were sequenced on an Illumina MiSeq using paired-end 2×250 reads and the MiSeq Reagent Kitv2.
Bioinformatic processing and taxonomic assignment
Amplicon sequence variants (ASVs) were generated via dada2 v1.16.0 using post-QC FASTQ files. Within the workflow, the paired FASTQ reads were trimmed, and then filtered to remove reads containing Ns, or with maximum expected errors >= 2. The dada2 learn error rate model was used to estimate the error profile prior to using the core dada2 algorithm for inferring the sample composition. Forward and reverse reads were merged by overlapping sequence, and chimeras were removed before taxonomic assignment. ASV taxonomy was assigned up to genus level using the SILVAv.138 database with the method described in 45 and a minimum boostrapping support of 50%. Species-level taxonomy was assigned to ASVs only with 100% identity and unambiguous matching to the reference.
Compositional analyses
α-Diversity
We calculated the inverse Simpson (IVS) index from relative ASV abundances (p) with N ASVs in a given sample, .
Principal Coordinate Analyses
Bray-Curtis distances were calculated from the filtered ASV table using QIIME 1.9.1 and principal components of the resulting distance matrix were calculated using the scikit-learn package for the Python programming language, used to embed sample compositions in the first two principal coordinates (see published code for the implementation in the Python programming language).
Average compositions and manipulation of compositions
To describe the average composition of a set of samples we calculated the central tendency of a compositional sample 46. For counter factual statistical analyses that require changes to a composition, e.g. an increase in a specific taxon, we deployed the perturbation operation (⊕), which is the compositional analogue to addition in Euclidean space46. A sample x containing the original relative taxon abundances is perturbed by a vector y,
where SD represents the D-part simplex.
Statistical analyses
Computer code alongside processed data tables are made available and can be used to reproduce the statistical analysis and regenerate the figures (Supplementary File 1).
Bayesian t-test
To compare diversity measurements between different sample groups, e.g. different clinical status, we performed a Bayesian estimation of group differences (BEST, 47) , implemented using the pymc3 package for the Python programming language; with priors (~) and deterministic calculations (=) to assess differences in estimated group means as follows:
Bayesian inference was performed using “No U-turn sampling”48. Highest density intervals (HDI) of the posterior estimation of group differences (Δ) were used to determine statistical certainty (***: 99% HDI >0 or <0, **: 95%HDI, *:90% HDI). The BEST code is provided in the Supplementary and implemented following the pymc3 documentation (Supplementary File 1).
Bayesian logistic regression
We performed a Bayesian logistic regression to distinguish compositional differences between infection-associated samples and samples from patients without secondary infections. We modeled the infection state of patient sample i, yi with a Binomial likelihood:
Where prior distributions are indicated by ~; α is the intercept of the generalized linear model, β is the coefficient vector for the log10-relative taxon abundances Xi in sample i.
Bayesian categorical regression
To interrogate a correspondence between the taxon abundances in stool samples and the microorganisms causing BSIs, we performed a Bayesian categorical regression. Briefly, we chose to investigate an association between stool taxon abundances (independent predictor variable) and the microbe identified in the blood (categorical outcome variable with seven unordered values) using a multiclass regression (categorical regression). We estimated for each sample a probability of being associated with one of the 6 BSI types (i.e. BSI by: Bacteroidaceae, Enterobacteriaceae, Lactobacillaceae, Pseudomonadaceae, Staphylococcaceae, Saccharomycetaceae), and we used a seventh class, uninfected, as a pivot. This means we are estimating a seven component simplical vector (s) containing the probabilities of a sample to be associated with one of the seven categories (6 BSI types and uninfected). For each category, we set up a linear model (sg, where g indicates the category). Each linear model includes log10-relative stool taxon abundances of the taxa corresponding to the BSI category. Furthermore, we had shown that alpha diversity (IVS) was globally associated with BSI; thus, diversity was a predictor in each linear model. We set up a model using varying intercept and varying slope terms such that the linear models used partially pooled coefficients for baseline risks (β[1]) and slopes corresponding to the stool sample predictors (β[2]). The multiclass probabilities in s were then obtained by applying the softmax function. The following model and priors were used:
Where p corresponds to the probabilities of each category, β is a two-component variable for the intercept and slope terms, representing the global baseline probability of bacterial infections with Bacteroidaceae, Enterobacteriaceae, Lactobacillaceae, and Pseudomonadaceae as well as the global slope coefficient for the effect of stool log10-relative abundances, which are partially pooling information across bacterial infection categories. To achieve partial pooling and account for correlations between varying intercepts and slopes (z), we jointly inferred the Choleski-factorized covariance matrix (σp, Lp, zp), using the Lewandowski-Kurowicka-Joe (LKJ) distribution as a prior (LKJ_corr_choleski). βdiversity is the coefficient for the effect of the IVS diversity. Of note, 16S rRNA sequencing does not provide abundances for the fungal infection by Saccharomycetaceae; therefore, we used only a baseline risk for this infection type (αSaccharomycetaceae) and IVS as predictors. To ensure equal prior probabilities for this category relative to the other categories, which have an additional predictor term and thus wider prior probabilities, we compensated the otherwise reduced prior uncertainty by widening the prior for αSaccharomycetaceae. Also, we assumed that infections by Staphylococcaceae could sometimes include contaminations from the skin of the patient or staff; therefore, we did not pool estimates for BSIs by Staphylococcaceae with other coefficients. The model was implemented in the STAN programming language and compiled using cmdstan. Code, the compiled STAN model, R notebooks to obtain and process the posterior chains, and data tables are provided in the supplement (Supplementary File 2).
Data Availability
All data is made available as Supplementary. We provide code and data to reproduce the main analyses in the form of jupyter notebooks and R notebooks, alongside processed “tidy” data tables, compiled STAN programs and code to regenerate the figures. The raw sequencing data have been deposited on the Sequencing Reads Archive (SRA), and SRA accession numbers are available for two bioprojects corresponding to the mouse sequencing data (Supplementary File 3) and the human stool samples (Supplementary File 4).
Supplementary Material
Acknowledgments
We thank René Niehus for helpful discussions on the implementation of the various Bayesian analyses. We thank the NYU Langone’s Genome Technology Center supported in part by NYU Langone Health’s Laura and Isaac Perlmutter Cancer Center Support (grant P30CA016087) from the National Cancer Institute Langone for the mouse gut microbiota 16S rRNA sequencing.
Funding
This work was in part funded by NYU Grossman School of Medicine startup research funds to JS, and the Yale School of Public Health and the Beatrice Kleinberg Neuwirth Fund, as well as NIH grants to KC (DK093668, AI121244, HL123340, AI130945, AI140754, DK124336), a Faculty Scholar grant from the Howard Hughes Medical Institute (KC), Crohn’s & Colitis Foundation (KC), Kenneth Rainin Foundation (KC), Judith & Stewart Colton Center of Autoimmunity (KC). Further funding was provided by grants from the NIH/NIAID to MD (R01AI143639 and R21AI139374), by Jan Vilcek/David Goldfarb Fellowship Endowment Funds to AMVJ, by The G. Harold and Leila Y. Mathers Charitable Foundation to MD, and by NYU Grossman School of Medicine Startup funds to MD and KAS, and the NYU Grossman School of Medicine COVID-19 seed research funds to VJT. MN was supported by the American Heart Association Postdoctoral Fellowship 19-A0–00-1003686. IMPACT received support from the Yale COVID-19 Research Resource Fund. AI and DRL are Investigators of the Howard Hughes Medical Institute. AIK received support from the Beatrice Kleinberg Neuwirth Fund, Bristol Meyers Squibb Foundation and COVID-19 research funds from the Yale Schools of Public Health and Medicine.
Conflicts
KC has received research support from Pfizer, Takeda, Pacific Biosciences, Genentech, and Abbvie; consulted for or received an honoraria from Puretech Health, Genentech, and Abbvie; and holds U.S. patent 10,722,600 and provisional patents 62/935,035 and 63/157,225. JS is cofounder of Postbiotics Plus Research LLC.
Yale IMPACT Team
Abeer Obaid, Alice Lu-Culligan, Allison Nelson, Anderson Brito, Angela Nunez, Anjelica Martin, Annie Watkins, Bertie Geng, Chaney Kalinich, Christina Harden, Codruta Todeasa, Cole Jensen, Daniel Kim, David McDonald, Denise Shepard, Edward Courchaine, Elizabeth B. White, Eric Song, Erin Silva, Eriko Kudo, Giuseppe DeIuliis, Harold Rahming, Hong-Jai Park, Irene Matos, Jessica Nouws, Jordan Valdez, Joseph Fauver, Joseph Lim, Kadi-Ann Rose, Kelly Anastasio, Kristina Brower, Laura Glick, Lokesh Sharma, Lorenzo Sewanan, Lynda Knaggs, Maksym Minasyan, Maria Batsu, Mary Petrone, Maxine Kuang, Maura Nakahata, Melissa Campbell, Melissa Linehan, Michael H. Askenase, Michael Simonov, Mikhail Smolgovsky, Nicole Sonnert, Nida Naushad, Pavithra Vijayakumar, Rick Martinello, Rupak Datta, Ryan Handoko, Santos Bermejo, Sarah Prophet, Sean Bickerton, Sofia Velazquez, Tara Alpert, Tyler Rice, William Khoury-Hanold, Xiaohua Peng, Yexin Yang, Yiyun Cao & Yvette Strong
Bibliography
- 1.Fajgenbaum D. C. & June C. H. Cytokine Storm. N. Engl. J. Med. 383, 2255–2273 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lucas C. et al. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature 584, 463–469 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zuo T. et al. Alterations in Gut Microbiota of Patients With COVID-19 During Time of Hospitalization. Gastroenterology 159, 944–955.e8 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yeoh Y. K. et al. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut 70, 698–706 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nori P. et al. Bacterial and fungal coinfections in COVID-19 patients hospitalized during the New York City pandemic surge. Infect. Control Hosp. Epidemiol. 42, 84–88 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Grasselli G. et al. Hospital-Acquired Infections in Critically Ill Patients With COVID-19. Chest (2021). doi: 10.1016/j.chest.2021.04.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Langford B. J. et al. Bacterial co-infection and secondary infection in patients with COVID-19: a living rapid review and meta-analysis. Clin. Microbiol. Infect. 26, 1622–1629 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Buffie C. G. et al. Precision microbiome reconstitution restores bile acid mediated resistance to Clostridium difficile. Nature 517, 205–208 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Buffie C. G. & Pamer E. G. Microbiota-mediated colonization resistance against intestinal pathogens. Nat. Rev. Immunol. 13, 790–801 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Modi S. R., Collins J. J. & Relman D. A. Antibiotics and the gut microbiota. J. Clin. Invest. 124, 4212–4218 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Morjaria S. et al. Antibiotic-Induced Shifts in Fecal Microbiota Density and Composition during Hematopoietic Stem Cell Transplantation. Infect. Immun. 87, (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Niehus R. et al. Quantifying antibiotic impact on within-patient dynamics of extended-spectrum beta-lactamase resistance. Elife 9, (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Taur Y. et al. Intestinal domination and the risk of bacteremia in patients undergoing allogeneic hematopoietic stem cell transplantation. Clin. Infect. Dis. 55, 905–914 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Taur Y. et al. Reconstitution of the gut microbiota of antibiotic-treated patients by autologous fecal microbiota transplant. Sci. Transl. Med. 10, (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liao C. et al. Compilation of longitudinal microbiota data and hospitalome from hematopoietic cell transplantation patients. Sci. Data 8, 71 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Peled J. U. et al. Microbiota as Predictor of Mortality in Allogeneic Hematopoietic-Cell Transplantation. N. Engl. J. Med. 382, 822–834 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yu D. et al. Low prevalence of bloodstream infection and high blood culture contamination rates in patients with COVID-19. PLoS One 15, e0242533 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang D. et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA 323, 1061–1069 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Westblade L. F., Simon M. S. & Satlin M. J. Bacterial coinfections in coronavirus disease 2019. Trends Microbiol. (2021). doi: 10.1016/j.tim.2021.03.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sepulveda J. et al. Bacteremia and Blood Culture Utilization during COVID-19 Surge in New York City. J. Clin. Microbiol. 58, (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lansbury L., Lim B., Baskaran V. & Lim W. S. Co-infections in people with COVID-19: a systematic review and meta-analysis. J. Infect. 81, 266–275 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sieswerda E. et al. Recommendations for antibacterial therapy in adults with COVID-19 - an evidence based guideline. Clin. Microbiol. Infect. 27, 61–66 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhai B. et al. High-resolution mycobiota analysis reveals dynamic intestinal translocation preceding invasive candidiasis. Nat. Med. 26, 59–64 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Haak B. W. et al. Impact of gut colonization with butyrate-producing microbiota on respiratory viral infection following allo-HCT. Blood 131, 2978–2986 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Steed A. L. et al. The microbial metabolite desaminotyrosine protects from influenza through type I interferon. Science 357, 498–502 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Abt M. C. et al. Commensal bacteria calibrate the activation threshold of innate antiviral immunity. Immunity 37, 158–170 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ichinohe T. et al. Microbiota regulates immune defense against respiratory tract influenza A virus infection. Proc. Natl. Acad. Sci. USA 108, 5354–5359 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sencio V. et al. Influenza infection impairs the gut’s barrier properties and favors secondary enteric bacterial infection through reduced production of short-chain fatty acids. Infect. Immun. (2021). doi: 10.1128/IAI.00734-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang J. et al. Respiratory influenza virus infection induces intestinal immune injury via microbiota-mediated Th17 cell-dependent inflammation. J. Exp. Med. 211, 2397–2410 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schluter J. et al. The gut microbiota is associated with immune cell dynamics in humans. Nature 588, 303–307 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gopalakrishnan V. et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 359, 97–103 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Diefenbach C. S. et al. Microbial dysbiosis is associated with aggressive histology and adverse clinical outcome in B-cell non-Hodgkin lymphoma. Blood Adv. 5, 1194–1198 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sokol H. et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl. Acad. Sci. USA 105, 16731–16736 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wrzosek L. et al. Bacteroides thetaiotaomicron and Faecalibacterium prausnitzii influence the production of mucus glycans and the development of goblet cells in the colonic epithelium of a gnotobiotic model rodent. BMC Biol. 11, 61 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gaebler C. et al. Evolution of antibody immunity to SARS-CoV-2. Nature 591, 639–644 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Park S.-K. et al. Detection of SARS-CoV-2 in Fecal Samples From Patients With Asymptomatic and Mild COVID-19 in Korea. Clin. Gastroenterol. Hepatol. 19, 1387–1394.e2 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xiao F. et al. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology 158, 1831–1833.e3 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cheung K. S. et al. Gastrointestinal Manifestations of SARS-CoV-2 Infection and Virus Load in Fecal Samples From a Hong Kong Cohort: Systematic Review and Meta-analysis. Gastroenterology 159, 81–95 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lamers M. M. et al. SARS-CoV-2 productively infects human gut enterocytes. Science 369, 50–54 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang L. et al. An observational cohort study of bacterial co-infection and implications for empirical antibiotic therapy in patients presenting with COVID-19 to hospitals in North West London. J. Antimicrob. Chemother. 76, 796–803 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Matsuzawa-Ishimoto Y. et al. Autophagy protein ATG16L1 prevents necroptosis in the intestinal epithelium. J. Exp. Med. 214, 3687–3705 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Labarta-Bajo L. et al. Type I IFNs and CD8 T cells increase intestinal barrier permeability after chronic viral infection. J. Exp. Med. 217, (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Karki R. et al. Synergism of TNF-α and IFN-γ Triggers Inflammatory Cell Death, Tissue Damage, and Mortality in SARS-CoV-2 Infection and Cytokine Shock Syndromes. Cell 184, 149–168.e17 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gohl D. M. et al. Systematic improvement of amplicon marker gene methods for increased accuracy in microbiome studies. Nat. Biotechnol. 34, 942–949 (2016). [DOI] [PubMed] [Google Scholar]
- 45.Wang Q., Garrity G. M., Tiedje J. M. & Cole J. R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 73, 5261–5267 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Pawlowsky-Glahn V., Egozcue J. J. & Tolosana-Delgado R. Modelling and analysis of compositional data. (John Wiley & Sons, Ltd, 2015). doi: 10.1002/9781119003144 [DOI] [Google Scholar]
- 47.Kruschke J. K. Bayesian estimation supersedes the t test. J. Exp. Psychol. Gen. 142, 573–603 (2013). [DOI] [PubMed] [Google Scholar]
- 48.Homan M. D. & Gelman A. The No-U-Turn Sampler: Adaptively Setting Path Lengths in Hamiltonian Monte Carlo. J. Mach. Learn. Res. 15, 1593–1623 (2014). [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data is made available as Supplementary. We provide code and data to reproduce the main analyses in the form of jupyter notebooks and R notebooks, alongside processed “tidy” data tables, compiled STAN programs and code to regenerate the figures. The raw sequencing data have been deposited on the Sequencing Reads Archive (SRA), and SRA accession numbers are available for two bioprojects corresponding to the mouse sequencing data (Supplementary File 3) and the human stool samples (Supplementary File 4).