Abstract
Purpose
In primary prostate cancer (PCa) patients, accurate staging and histologic grading are crucial to guide treatment decisions. 18F-DCFPyL (PSMA)-PET/CT has been successfully introduced for (re)staging PCa, showing high accuracy to localise PCa in lymph nodes and/or osseous structures. The diagnostic performance of 18F-DCFPyL-PET/CT in localizing primary PCa within the prostate gland was assessed, allowing for PSMA-guided targeted-prostate biopsy.
Methods
Thirty patients with intermediate-/high-risk primary PCa were prospectively enrolled between May 2018 and May 2019 and underwent 18F-DCFPyL-PET/CT prior to robot-assisted radical prostatectomy (RARP). Two experienced and blinded nuclear medicine physicians assessed tumour localisation within the prostate gland on PET/CT, using a 12-segment mapping model of the prostate. The same model was used by a uro-pathologist for the RARP specimens. Based on PET/CT imaging, a potential biopsy recommendation was given per patient, based on the size and PET-intensity of the suspected PCa localisations. The biopsy recommendation was correlated to final histopathology in the RARP specimen. Sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) for clinically significant PCa (csPCa, Gleason score ≥ 3 + 4 = 7) were assessed.
Results
The segments recommended for potential targeted biopsy harboured csPCA in 28/30 patients (93%), and covered the highest Gleason score PCa segment in 26/30 patient (87%). Overall, 122 of 420 segments (29.0%) contained csPCa at final histopathological examination. Sensitivity, specificity, PPV and NPV for csPCa per segment using 18F-DCFPyL-PET/CT were 61.4%, 88.3%, 68.1% and 84.8%, respectively.
Conclusions
When comparing the PCa-localisation on 18F-DCFPyL-PET/CT with the RARP specimens, an accurate per-patient detection (93%) and localisation of csPCa was found. Thus, 18F-DCFPyL-PET/CT potentially allows for accurate PSMA-targeted biopsy.
Electronic supplementary material
The online version of this article (10.1007/s00345-020-03490-8) contains supplementary material, which is available to authorized users.
Keywords: 18F-DCFPyL PET/CT, Primary detection, Prostate cancer, PSMA, Targeted biopsy
Introduction
Prostate cancer (PCa) is the most common cancer in men in the Western world [1, 2]. Histopathological verification is required to confirm the diagnosis and is standardly attained through ultra-sound-guided systematic prostate biopsies [3]. These random biopsies are subject to sampling error, however, resulting in false-negative outcomes and imprecise tumour-risk assessment [4, 5]. To overcome this, multi-parametric magnetic resonance imaging (mpMRI) prior to systematic biopsy has been implemented in clinical guidelines, enabling targeted biopsies of radiologically suspected lesions (MRI-TBx) [3, 6–8].
Besides conventional imaging modalities such as mpMRI, novel imaging techniques including prostate-specific membrane antigen-positron emission tomography/computed tomography (PSMA-PET/CT) have been introduced. PSMA is significantly overexpressed in malignant prostate cells, correlates with higher tumour grades and represents a marker of tumour aggressiveness [9, 10]. PSMA-PET/CT-imaging has been shown to accurately identify the primary prostate tumours, with detection rates of 98–100% [11, 12]. PSMA-PET/CT could thus be used to localize and guide targeted prostate biopsy in patients with clinically suspected PCa. Furthermore, PSMA-PET/CT would simultaneously provide screening for bone and lymph-node metastases, as it is repeatedly found to be more sensitive than conventional imaging (i.e., MRI, bone scintigraphy and CT) in the initial staging setting [13, 14].
This is the first prospective study on the accuracy of 18F-DCFPyL (PSMA) PET/CT imaging for the primary detection of PCa. The primary aim was to assess the accuracy of 18F-DCFPyL-PET/CT to localise primary PCa within the prostate gland, by comparing imaging results from 18F-DCFPyL-PET/CT to final histopathology of the robot-assisted radical prostatectomy (RARP) specimen. The secondary objectives were to investigate the ability of 18F-DCFPyL-PET/CT to provide a recommendation for potential targeted biopsy and to assess the diagnostic accuracy of determining local tumour stage (pT).
Methods
Study design and patient population
This was a prospective, non-randomised study in patients with diagnosed primary PCa. Pre-operative imaging results were compared to histopathology following RARP. All subjects signed informed-consent for the collection of their clinical data. The study has been approved by the ethical review board of the Amsterdam University Medical Centre (AUMC) (review number 2017.543). Patients were enrolled consecutively between May 2018-May 2019 in Amsterdam UMC, location VUmc.
Patients had histologically proven, intermediate or high-risk, PCa, for which they underwent RARP [3, 15]. Of all included patients, age, prostate volume, initial prostate-specific antigen (PSA)-level, clinical T-stage, pathological biopsy features (histopathological grade, number of cores with cancer) and European Association of Urology (EAU)-risk category were collected [3, 15]. A 12-segment anatomic mapping model of the prostate was used to localise and characterise the prostatic tumours, with 2 additional segments representing the seminal vesicles (pT3b) (Appendix-1 in ESM) [16].
Imaging protocol
Patients were staged with 18F-DCFPyL which was synthesised under Good Manufacturing Practices conditions, as described by Jansen et al. [17, 18]. PET images were acquired at a median of 118 min after injection of the radiotracer (interquartile range [IQR] 113–122 min) with a median dose of 313 MBq 18F-DCFPyL (IQR 299–324 MBq), and a median of 5.4 weeks (IQR 3.0–7.2) prior to surgery. Image-acquisitions were performed using a Philips Ingenuity TF (Philips Healthcare®, NL/USA)-PET/CT system. No diuretics were administered prior to the scan. The scan trajectory included mid-thighs to skull-base, with 4 min per bed position. All PET scans were combined with a diagnostic CT scan (110 mAs, 120 kV), without contrast-enhancement. Images were corrected for decay, scatter, random coincidences, and photon attenuation.
Images were reconstructed with a BLOB-based Ordered-Subsets Expectations Maximization algorithm (Philips, 3 iterations; 33 subsets) [19]). The default Ordered-Subsets Expectations Maximization with Time-of-Flight reconstruction was used. Reconstructions included both 4 mm for semi-quantification purposes, and 2 mm slices for visual interpretation (matrix size 144 × 144, slice thickness 4 mm; matrix size 288 × 288, slice thickness 2 mm).
Image interpretation and 18F-DCFPyL-based potential biopsy recommendation
Scan interpretation was performed blinded for the pathology results and other imaging by two nuclear medicine physicians (DO,GZ) with ample experience in 18F-DCFPyL-PET/CT reading (> 300 scans), in consensus. The readers used the 12-segment mapping model to demarcate the image-detected tumour extent (Appendix-1 in ESM) [20]. For all positive segments, the readers’ diagnostic confidence was evaluated using a five-point scale, alike the PSMA-RADS classification [21] (score 1–2 ‘benign’; 3 ‘equivocal’; 4–5 ‘likely PCa’). PSMA-RADS 4–5 were defined as suspicious for PCa (‘positive’), and were used for the final diagnostic accuracy analysis. Based on PET/CT-imaging, two segments per patient were indicated to be potentially targeted by prostate biopsy. These segments were selected based on both visual interpretation (location, size) and semi-quantification by determining the highest standardised uptake values (SUVmax) of the suspected lesions. Finally, the readers indicated if radiological extra-capsular extension (ECE, rT-stage 3a) or invasion into the seminal vesicles (rT3b) was suspected.
Pathology analysis
RARP specimens were processed according to clinical routine and the International Society of Urological Pathology (ISUP) guidelines [22]. All specimens were fixated in formaldehyde (10%) directly after surgery. The surface of the specimens was inked and the apex and base (bladder neck) were removed. The mid part of the specimen was cut perpendicular to the urethra in 4 mm slices. The apex and base parts were cut in a sagittal fashion. Histologic slices were produced after sectioning in quadrants. Blinded by PET/CT results, an experienced uro-pathologist (PV) reviewed all slices and delineated all tumour depositions on the 12-segment mapping model of the prostate and the 2 segments of the seminal vesicles (Appendix-1 in ESM). For all segments that contained tumour, a Gleason score and ISUP grade was provided and the presence of ECE (pT3a). Clinically significant PCa (csPCa) was defined as PCa with Gleason score ≥ 3 + 4 = 7 (ISUP grade ≥ 2) or any tumour with ≥ pT3a. The index lesion was defined as the largest lesion with the highest ISUP grade or stage.
Statistical analysis
The localisation of the detected prostate tumour by 18F-DCFPyL-PET/CT was matched to the histopathology results and the sensitivity, specificity, positive predicting value (PPV), and negative predicting value (NPV) were calculated on a segment basis. Correlation of 18F-DCFPyL-PET/CT with histopathology was considered if exactly the same segment was demarcated (total agreement). Since there are no anatomical landmarks to delineate the segments, artificial segmentation can occur, causing a mismatch between the PET/CT- and histopathological findings while both correspond with the same lesion. Therefore, a second analysis of diagnostic accuracy was performed, in which PET correlation was also considered if there was a discrepancy of up to 1 region in the coronal or sagittal plane (near-total agreement) [23, 24]. Reciever‐operating characteristic curves (ROC) and area under the curve (AUC) analysis were performed to explore the accuracy of PSMA-PET/CT in the detection of segments containing csPCa based on the 5-point scale. For the assessment of pathological tumour stage (pT), we investigated the accuracy of 18F-DCFPyL-PET/CT to differentiate locally advanced disease (> rT3a) from prostate-confined disease (rT2). Numerical variables were summarised with median values and interquartile ranges (IQR); categorical variables with proportions (%). To compare medians of non-parametric data, the Mann–Whitney-Wilcoxon test and the Kruskal-Wallis test were used (significance set at p < 0.05). Statistical analysis was performed with IBM® SPSS® Statistics for Windows®, version 26.
Results
Patient characteristics
A total of 30 patients was included in this study, having a median initial PSA-level of 11.1 ng/ml (IQR 5.8–22.4). According to EAU guidelines, 10/30 (33.3%) patients had intermediate-risk PCa and 20/30 (66.6%) had high-risk PCa [15]. Pre-operative and post-operative characteristics of included patients are listed in Table 1.
Table 1.
Pre- and postoperative characteristics of patients undergoing 18F-DCFPyL-PET/CT before robot-assisted radical prostatectomy
| Baseline (pre-operative) characteristics | ||
|---|---|---|
| Median | IQR | |
| Age (years) | 68.5 | 64.3–69.8 |
| Prostate volume (ml) | 44.0 | 32.2–69.0 |
| Initial PSA (ng/ml) | 11.1 | 5.8–22.4 |
| Positive biopsy cores (% of total cores) | 50.0 | 35.0–71.0 |
| MSKCC risk of lymph-node metastases (%) | 13.4 | 10.6–28.0 |
| n | % | |
| Biopsy ISUP gradea | ||
| 1 | 0 | 0.0 |
| 2 | 9 | 30.0 |
| 3 | 7 | 23.3 |
| 4 | 11 | 36.7 |
| 5 | 3 | 10.0 |
| Total | 30 | 100.0 |
| Clinical T stage | ||
| 1c | 5 | 16.6 |
| 2a/b | 18 | 60.0 |
| 2c | 6 | 20.0 |
| 3a | 1 | 3.3 |
| Total | 30 | 100.0 |
| EAU risk category | ||
| Intermediate | 10 | 33.3 |
| High | 20 | 66.6 |
| Total | 30 | 100.0 |
| Pathology (post-operative) results | ||
|---|---|---|
| ISUP categorya | ||
| 1 | 0 | 0.0 |
| 2 | 13 | 43.3 |
| 3 | 6 | 20.0 |
| 4 | 2 | 6.7 |
| 5 | 6 | 20.0 |
| Total | 30 | 100.0 |
| Pathological tumour (pT) stage | ||
| pT2 | 16 | 53.3 |
| pT3a | 10 | 33.3 |
| pT3b | 4 | 13.3 |
| Total | 30 | 100.0 |
| Pathological Lymph-node (N) stage | ||
| 0 | 29 | 96.7 |
| 1 | 1 | 3.3 |
| Total | 30 | 100.0 |
IQR interquartile range, PSA prostate-specific antigen, MSKCC Memorial Sloan Kettering Cancer Centre, ISUP International Society of Urological Pathology, EAU European Association of Urology
aISUP definition
ISUP 1 = Gleason score 3 + 3 = 6
ISUP 2 = Gleason score 3 + 4 = 7
ISUP 3 = Gleason score 4 + 3 = 7
ISUP 4 = Gleason score 4 + 4 = 8/Gleason score 3 + 5 = 8/Gleason score 5 + 3 = 8
ISUP 5 = Gleason score 4 + 5 = 9/Gleason score 5 + 4 = 9/Gleason score 5 + 5 = 10
Accuracy of 18F-DCFPyL-PET/CT to detect local prostate cancer on a segmental level
All patients showed PSMA expression in the prostate. In 30 evaluated patients, 420 segments (12 prostate segments + 2 seminal vesicle segments per patient) could be used both for PET/CT and histopathological mapping evaluation. PCa was present in 129 of the 420 (30.7%) segments on histopathological examination, and csPCa was found in 122 of the 420 segments (29.0%) (median 3 segments per patient, IQR 2–5). The sensitivity, specificity, PPV and NPV of 18F-DCFPyL-PET/CT to detect csPCa per segment with total agreement was 61.4% (95%CI 52.2–70.0%), 88.3% (95%CI 83.9–91.6%), 68.1% (95%CI 58.5–76.6%), and 84.8% (95%CI 80.2–88.5%), respectively (Appendix-2 in ESM). For near-total agreement, the sensitivity, specificity, PPV and NPV of 18F-DCFPyL PET/CT to detect csPCa per segment was 84.4% (95%CI 76.5–90.1%), 97.0% (95%CI 94.1–98.5%), 92.0% (95%CI 84.9–96.0%), and 93.8% (95%CI 90.3–96.1%), respectively. The area under the curve (AUC) of 18F-DCFPyL-PET/CT was 0.78 (95%CI 0.73–0.84) for the total agreement scores, and 0.85 (95%CI 0.80–0.90) for the near-total agreement scores (Appendix-3 in ESM). True positive segments had a median SUVmax of 8.26 (IQR 5.25–11.40), which was significantly higher than the median SUVmax of false-positive segments of 4.06 (IQR 3.56–5.10) (p = 0.02). The median SUVmax of true positive segments did not correlate with ISUP grade groups (p = 0.95) (Appendix-4 in ESM).
Potential targeted biopsy recommendation on a patient level
The primary potential biopsy recommendation by the nuclear medicine physician harboured csPCa in 24/30 (80.0%) patients and detected the index PCa lesion in 23/30 (76.6%) patients. When both the primary and secondary recommended segments would potentially be targeted, 18F-DCFPyL-PET/CT revealed csPCa in 28/30 (93.3%) patients. Moreover, it pinned the index PCa lesion in 26/30 (86.7%) patients. An example of potential 18F-DCFPyL-guided biopsy recommendation and concurrent histopathological examination of the RARP specimen is shown in Fig. 1. Potential biopsy recommendation that matched the index PCa lesion had a median SUVmax 8.62 (IQR 6.41–12.62). The recommendation for potential biopsy from the nuclear physicians that matched csPCA had a median SUVmax of 8.55 (IQR 6.34–13.79), and was significantly higher than the recommended potential biopsy segments that did not contain csPCa (median SUVmax of 3.10 (IQR 2.86–3.87) (p = 0.02).
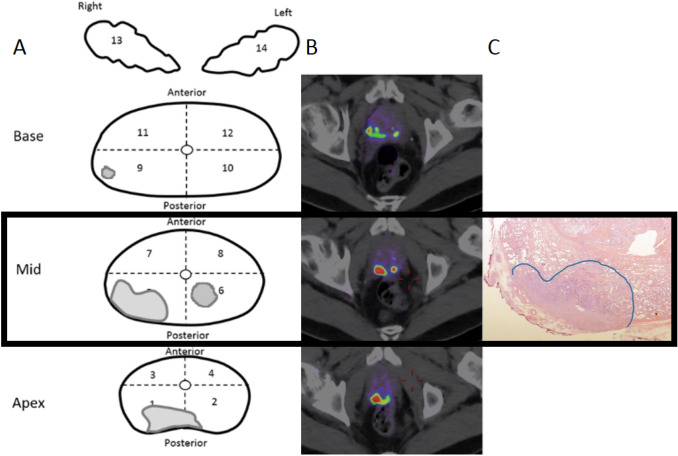
Fig. 1.
67-Year-old man with cT3a, Gleason score 3 + 4 = 7 prostate cancer and an initial PSA 27 ng/ml considered candidate for radical prostatectomy with extended pelvic lymph-node dissection. a Schematic drawing of the 12 prostate segments included in the study. b Transversal fused 18F-DCFPyL-PET and CT show intense focal uptake in the right posterior midgland and apex segments, with extracapsular extension. Based on the highest SUVmax of 6.09 and tumour size, segment 5 is recommended for potential targeted biopsy. c histopathology (hematoxylin and eosin stain, original magnification × 10) shows a tumour focus, in both segment 5 and 1 with a Gleason score 3 + 4 = 7 prostate cancer with extraprostatic extension (pT3a), hereby confirming the index lesion localisation by PET
Local staging
Final histopathological analysis revealed pT3a in 10/30 (33.3%) patients, and pT3b in 4/30 (13.3%) patients. The sensitivity, specificity, PPV and NPV of 18F-DCFPyL-PET/CT to detect locally advanced tumour growth (≥ pT3a) was 35.7% (95%CI 14.0–64.3%), 93.8% (95%CI 67.7–99.7%), 83.3% (95%CI 36.5–99.1%), and 62.5% (95%CI 40.8–80.4%), respectively (Appendix-5 in ESM). For the detection of pT3a sub-stage, the sensitivity, specificity, PPV and NPV of 18F-DCFPyL-PET/CT were 20.0% (95%CI 3.5–55.8), 100% (95%CI 80.0–100), 100.0% (95%CI 19.7–100), and 71.4% (95%CI 51.1–86.0), respectively. For the detection of pT3b sub-stage, the sensitivity, specificity, PPV and NPV of 18F-DCFPyL-PET/CT were 75.0% (95%CI 21.9–98.7), 92.3% (95%CI 73.4–98.7), 60.0% (95%CI 17.0–92.7), and 96.0% (95%CI 77.7–99.8), respectively.
Discussion
This is the first prospective study in which 18F-DCFPyL-PET/CT imaging was used to locate primary PCa within the prostate gland, exploring the diagnostic potential of PSMA-based targeted biopsies. A total of 30 patients diagnosed with intermediate and high-risk PCa that underwent 18F-DCFPyL-PET/CT prior to RARP was analysed. When using a prostate-mapping model, the potential 18F-DCFPyL-PET/CT-based targeted biopsy recommendation detected csPCa in 28/30 (93.3%) patients. Moreover, it detected the index PCa lesion in 26/30 (86.7%) patients. Potentially, this makes PSMA-targeted biopsy a diagnostic tool that may adequately guide precision prostate biopsy. In biopsy-naive patients at increased risk of metastatic spread, and in whom staging imaging is mandatory (e.g. PSA ≥ 20), 18F-DCFPyL-PET/CT could potentially be used simultaneously to stage patients and to target PSMA-avid prostatic lesions suspicious for PCa.
18F-DCFPyL-PET/CT imaging demonstrated a moderate per segment-based sensitivity for the detection of csPCa of 61.4%, at a 88.3% specificity. The moderate sensitivity indicates that 18F-DCFPyL-PET/CT was not able to detect all localised csPCa. Segmentation of the prostate gland is problematic, as no clear anatomical landmarks are available to delineate the different segments within the prostate (Appendix-1 in ESM). A tumour located on the border of the apex and middle part of the prostate could be classified in different segments by the nuclear medicine physician and uro-pathologist, while in fact, they detected the same lesion. Therefore, the near-agreement score was introduced to approximate clinical reality. The sensitivity of the near-agreement score of 18F-DCFPyL-PET/CT imaging for the detection of csPCa was higher at 84.4% with a specificity of 97.0%.
MpMRI has found a prominent place in the identification and localisation of PCa [8]. Few studies have directly compared the outcome of mpMRI to that of PSMA-PET/CT for the detection of localised PCa. Scheltema et al. analysed 56 patients with intermediate-risk PCa who underwent 68Ga-PSMA-PET/CT and mpMRI prior to RARP [20]. This study used the same 12-segment-based model of the prostate, and a leniency method similar to our near-agreement score. The patient-based sensitivity for detecting ISUP grade 2–3 PCa was 100% for PSMA-PET/CT vs 98% for mpMRI (PI-RADS 3–5). The segment-based sensitivity and specificity for PSMA-PET/CT was 88% (95% CI 83–92) and 93% (95% CI 91–95) compared 68% (95% CI 61–75) and 91% (95% CI 87–93) for mpMRI (PI-RADS 3–5).
In another study, Kesch et al. [23] studied 10 patients with primary high-risk PCa who underwent 18F- PSMA-1007-PET/CT and mpMRI with subsequent RARP. Nine of the men were diagnosed with MRI-TBx and 1 with systematic biopsy. Using a 36-segment mapping model, a similar assessment of agreement and near-total agreement was used. In 10 patients, 18F-PSMA-1007 PET/CT showed a high sensitivity (71% for total and 93% for near-total agreement), specificity (81% for total and 92% for near-total agreement), and accuracy (75% for total and 93% for near-total agreement) for the detection of csPCa. Although the specificity was similar, this study did however show lower sensitivity for PSMA-PET/CT compared to mpMRI (86% for total and 92% for near-total agreement). Above mentioned studies implicate that PSMA-PET/CT imaging performs at least equal to mpMRI to locate primary PCa. The rates of mpMRI might have been overestimated as at least a part of included patients in previously mentioned studies were diagnosed by MRI-TBx. Thus, selection bias may have been introduced.
There is therapeutic importance to distinguish between T2 and T3 disease (i.e. for planning nerve-sparing surgery, to opt for active surveillance) [3]. A moderate sensitivity for the detection of pT3a-b of 35.7% was observed using 18F-DCFPyL-PET/CT. So, similar to mpMRI, a substantial number of patients with ≥ pT3a was understaged by PSMA-based imaging [8]. However, in a majority of patients, a PSMA-PET/CT rT3a-b finding was confirmed after histopathological examination (PPV for pT3a-b disease of 83.3%). Moreover, the promising specificity for pT3a-b using 18F-DCFPyL-PET/CT of 93.8% is congruent with similar 68 Ga-PSMA studies (specificity > 90% for T3b disease) [12, 25, 26]. Therefore, we recommend nuclear medicine physicians to report on the presence of rT3a-b specifically.
Our study has inherent limitations. Since the PET/CT resolution is confined at 5 mm, limited diagnostic accuracy for small PCa-foci is to be expected. A selection bias has been introduced due to the selection of patients with biopsy-confirmed csPCa. It is thus unclear how 18F-DCFPyL-PET/CT performs in a truly biopsy naïve cohort of patients. The present study was set up to evaluate the capability of PSMA-PET/CT to guide targeted prostate biopsy for the detection of csPCa, not with the goal to discriminate between those who should be or should not be biopsied. Unfortunately, not all patients received a pre-operative mpMRI, limiting direct comparison to 18F-DCFPyL-PET/CT. Moreover, some of the patients who received a mpMRI had a longer interval between the mpMRI and the PET/CT scans due to the mpMRI being performed at the referring centre. Finally, since the RARP-specimen will always change shape (due to organ slicing and shrinking artefacts) when it is removed from the body, no truly exact anatomical correlation is possible. Therefore, the partial agreement score was used to correct for this pitfall.
Conclusions
When comparing the localisation of PCa on 18F-DCFPyL-PET/CT with the RARP specimen using anatomical mapping, an accurate per-patient localization (93%) of csPCa was found within the prostate. 18F-DCFPyL-PET/CT proves promising for PSMA-targeted biopsy and provides a moderate local staging ability.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
We gratefully acknowledge the patients for their participation in this study.
Author contributions
YJLB: protocol/project development, data collection or management, data analysis and manuscript writing/editing. BHEJ: protocol/project development, data collection or management, data analysis and manuscript writing/editing. JPV: data collection or management and manuscript writing/editing. GJCZ: data collection or management and manuscript writing/editing. DM: data analysis and manuscript writing/editing. JAN: data collection or management and manuscript writing/editing. RB: data analysis and manuscript writing/editing. NHH: data analysis and manuscript writing/editing. OSH: protocol/project development, data analysis and manuscript writing/editing. RJAM: data analysis and manuscript writing/editing. DEO-L: protocol/project development, data collection or management, data analysis and manuscript writing/editing. ANV: protocol/project development, data collection or management, data analysis and manuscript writing/editing.
Funding
No funding was received for conducting this research.
Availability of data and materials
Data are available on request to the corresponding author.
Compliance with ethical standards
Conflict of interest
Prof. Dr. Boellaard reports the receiving of a grant from Philips Healthcare, outside the submitted work. The other authors declare that they have no competing interests.
Ethical approval
This study was approved by the institutional research board of the Amsterdam UMC (location VUmc), and each participant provided informed consent to participate.
Consent for publication
Each participant provided consent for data publication.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ferlay J, Colombet M, Soerjomataram I, Dyba T, Randi G, Bettio M, et al. Cancer incidence and mortality patterns in Europe: estimates for 40 countries and 25 major cancers in 2018. Eur J Cancer. 2018;103:356–387. doi: 10.1016/j.ejca.2018.07.005. [DOI] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7–34. doi: 10.3322/caac.21551. [DOI] [PubMed] [Google Scholar]
- 3.Mottet N, Bellmunt J, Briers E, Bolla M, Bourke L, Cornford P et al (2020) EAU – ESTRO – ESUR – SIOG Guidelines on Prostate Cancer. Arnhem, The Netherlands. EAU Guidelines Office
- 4.Bjurlin MA, Carter HB, Schellhammer P, Cookson MS, Gomella LG, Troyer D, et al. Optimization of initial prostate biopsy in clinical practice: sampling, labeling and specimen processing. J Urol. 2013;189:2039–2046. doi: 10.1016/j.juro.2013.02.072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lane BR, Zippe CD, Abouassaly R, Schoenfield L, Magi-Galluzzi C, Jones JS. Saturation technique does not decrease cancer detection during follow-up after initial prostate biopsy. J Urol. 2008;179:1746–1750. doi: 10.1016/j.juro.2008.01.049. [DOI] [PubMed] [Google Scholar]
- 6.Kasivisvanathan V, Rannikko AS, Borghi M, Panebianco V, Mynderse LA, Vaarala MH, et al. MRI-targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med. 2018;378:1767–1777. doi: 10.1056/NEJMoa1801993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rouviere O, Puech P, Renard-Penna R, Claudon M, Roy C, Mege-Lechevallier F, et al. Use of prostate systematic and targeted biopsy on the basis of multiparametric MRI in biopsy-naive patients (MRI-FIRST): a prospective, multicentre, paired diagnostic study. Lancet Oncol. 2019;20:100–109. doi: 10.1016/s1470-2045(18)30569-2. [DOI] [PubMed] [Google Scholar]
- 8.Drost FH, Osses DF, Nieboer D, Steyerberg EW, Bangma CH, Roobol MJ, et al. Prostate MRI, with or without MRI-targeted biopsy, and systematic biopsy for detecting prostate cancer. Cochrane Datab Syst Rev. 2019;4:012663. doi: 10.1002/14651858.CD012663.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Perner S, Hofer MD, Kim R, Shah RB, Li H, Moller P, et al. Prostate-specific membrane antigen expression as a predictor of prostate cancer progression. Hum Pathol. 2007;38:696–701. doi: 10.1016/j.humpath.2006.11.012. [DOI] [PubMed] [Google Scholar]
- 10.Silver DA, Pellicer I, Fair WR, Heston WD, Cordon-Cardo C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin Cancer Res. 1997;3:81–85. [PubMed] [Google Scholar]
- 11.Wondergem M, van der Zant FM, Roeleveld TA, Srbljin S, Kartachova MS, van Dongen A, et al. 18F-DCFPyL PET/CT in primary staging of prostate cancer. Eur J Hybrid Imaging. 2018;2:26. doi: 10.1186/s41824-018-0044-0. [DOI] [Google Scholar]
- 12.van Leeuwen PJ, Donswijk M, Nandurkar R, Stricker P, Ho B, Heijmink S, et al. 68Ga PSMA PET/CT predicts complete biochemical response from radical prostatectomy and lymph node dissection in intermediate and high-risk prostate cancer. BJU Int. 2018 doi: 10.1111/bju.14506. [DOI] [PubMed] [Google Scholar]
- 13.Wu H, Xu T, Wang X, Yu YB, Fan ZY, Li DX, et al. Diagnostic performance of (6)(8)gallium labelled prostate-specific membrane antigen positron emission tomography/computed tomography and magnetic resonance imaging for staging the prostate cancer with intermediate or high risk prior to radical prostatectomy: a systematic review and meta-analysis. World J Men's Health. 2019 doi: 10.5534/wjmh.180124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hofman MS, Lawrentschuk N, Francis RJ, Tang C, Vela I, Thomas P, et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multi-centre study. Lancet. 2020 doi: 10.1016/s0140-6736(20)30314-7. [DOI] [PubMed] [Google Scholar]
- 15.D'Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280:969–974. doi: 10.1001/jama.280.11.969. [DOI] [PubMed] [Google Scholar]
- 16.Rowe SP, Gage KL, Faraj SF, Macura KJ, Cornish TC, Gonzalez-Roibon N, et al. (1)(8)F-DCFBC PET/CT for PSMA-based detection and characterization of primary prostate cancer. J Nucl Med. 2015;56:1003–1010. doi: 10.2967/jnumed.115.154336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jansen BHE, Kramer GM, Cysouw MCF, Yaqub MM, de Keizer B, Lavalaye J, et al. Healthy tissue uptake of (68)Ga-prostate specific membrane antigen (PSMA), (18)F-DCFPyL, (18)F-Fluoromethylcholine (FCH) and (18)F-Dihydrotestosterone (FDHT) J Nucl Med. 2019 doi: 10.2967/jnumed.118.222505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jansen BHE, Yaqub M, Voortman J, Cysouw MCF, Windhorst AD, Schuit RC, et al. Simplified methods for quantification of (18)F-DCFPyL uptake in patients with prostate cancer. J Nucl Med. 2019 doi: 10.2967/jnumed.119.227520. [DOI] [PubMed] [Google Scholar]
- 19.Popescu LM, Matej S, Lewitt RM (2004) Iterative image reconstruction using geometrically ordered subsets with list-mode data. In: Nuclear science symposium conference record, 2004, pp 3536–3540
- 20.Scheltema MJ, Chang JI, Stricker PD, van Leeuwen PJ, Nguyen QA, Ho B, et al. Diagnostic accuracy of (68) Ga-prostate-specific membrane antigen (PSMA) positron-emission tomography (PET) and multiparametric (mp)MRI to detect intermediate-grade intra-prostatic prostate cancer using whole-mount pathology: impact of the addition of (68) Ga-PSMA PET to mpMRI. BJU Int. 2019;124(Suppl 1):42–49. doi: 10.1111/bju.14794. [DOI] [PubMed] [Google Scholar]
- 21.Rowe SP, Pienta KJ, Pomper MG, Gorin MA. Proposal for a structured reporting system for prostate-specific membrane antigen-targeted PET imaging: PSMA-RADS version 1.0. J Nucl Med. 2018;59:479–485. doi: 10.2967/jnumed.117.195255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cornford P, Bellmunt J, Bolla M, Briers E, De Santis M, Gross T, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part II: treatment of relapsing, metastatic, and castration-resistant prostate cancer. Eur Urol. 2016 doi: 10.1016/j.eururo.2016.08.002. [DOI] [PubMed] [Google Scholar]
- 23.Kesch C, Vinsensia M, Radtke JP, Schlemmer HP, Heller M, Ellert E, et al. Intra-individual comparison of 18F-PSMA-1007-PET/CT, multi-parametric MRI and radical prostatectomy specimen in patients with primary prostate cancer—a retrospective, proof of concept study. J Nucl Med. 2017 doi: 10.2967/jnumed.116.189233. [DOI] [PubMed] [Google Scholar]
- 24.Giesel FL, Sterzing F, Schlemmer HP, Holland-Letz T, Mier W, Rius M, et al. Intra-individual comparison of (68)Ga-PSMA-11-PET/CT and multi-parametric MR for imaging of primary prostate cancer. Eur J Nucl Med Mol Imaging. 2016;43:1400–1406. doi: 10.1007/s00259-016-3346-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dekalo S, Kuten J, Mabjeesh NJ, Beri A, Even-Sapir E, Yossepowitch O. 68Ga-PSMA PET/CT: does it predict adverse pathology findings at radical prostatectomy? Urol Oncol. 2019;37:574e1–e24. doi: 10.1016/j.urolonc.2019.05.015. [DOI] [PubMed] [Google Scholar]
- 26.von Klot CJ, Merseburger AS, Boker A, Schmuck S, Ross TL, Bengel FM, et al. (68)Ga-PSMA PET/CT Imaging predicting intraprostatic tumor extent, extracapsular extension and seminal vesicle invasion prior to radical prostatectomy in patients with prostate cancer. Nucl Med Mol Imaging. 2017;51:314–322. doi: 10.1007/s13139-017-0476-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data are available on request to the corresponding author.