Abstract
Native Hawaiian and Pacific Islander (NHPI) populations suffer from disproportionately higher rates of chronic conditions, such as type 2 diabetes, that arises from metabolic dysfunction and are often associated with obesity and inflammation. In addition, the global coronavirus disease 2019 pandemic has further compounded the effect of health inequities observed in Indigenous populations, including NHPI communities. Reversible lifestyle habits, such as diet, may either be protective of or contribute to the increasing prevalence of health inequities in these populations via the immunoepigenetic-microbiome axis. This axis offers insight into the connection between diet, epigenetics, the microbiome composition, immune function, and response to viral infection. Epigenetic mechanisms that regulate inflammatory states associated with metabolic diseases, including diabetes, are impacted by diet. Furthermore, diet may modulate the gut microbiome by influencing microbial diversity and richness; dysbiosis of the microbiome is associated with chronic disease. A high fiber diet facilitates a favorable microbiome composition and in turn increases production of intermediate metabolites named short-chain fatty acids (SCFAs) that act on metabolic and immune pathways. In contrast, low fiber diets typically associated with a westernized lifestyle decreases the abundance of microbial derived SCFAs. This decreased abundance is characteristic of metabolic syndromes and activation of chronic inflammatory states, having larger implications in disease pathogenesis of both communicable and non-communicable diseases. Native Hawaiians and Pacific Islanders that once thrived on healthy traditional diets may be more sensitive than non-indigenous peoples to the metabolic perturbation of westernized diets that impinge on the immunoepigenetic-gut microbiome axis. Recent studies conducted in the Maunakea lab at the University of Hawai‘i at Mānoa John A. Burns School of Medicine have helped elucidate the connections between diet, microbiome composition, metabolic syndrome, and epigenetic regulation of immune function to better understand disease pathogenesis. Potentially, this research could point to ways to prevent pre-disease conditions through novel biomarker discovery using community-based approaches.
Keywords: ethnic health disparities, immunoepigenetic-microbiome axis, intestinal microbiome, metabolic syndrome, SARS-CoV-2, COVID-19 severity, short-chain fatty acid, type II diabetes risk
Introduction
Health disparities among minority populations across the US include higher rates of metabolic syndrome, a condition that can arise from persistent low-grade systemic inflammation. Common characteristics of this syndrome include obesity, insulin resistance, hypertension, and dyslipidemia.1 Native Hawaiians and Pacific Islanders suffer from the fastest rising prevalence rate of type 2 diabetes globally, in part due to socioeconomic constraints that restrict their access to healthy dietary options and lead to metabolic and immune dysregulation.2–4 Diet contributes to the development of both metabolic and immune function via the immunoepigenetic-microbiome axis that functions to establish and maintain metabolic homeostasis. Unhealthy diets alter gut microbiome composition leading to dysbiosis and aberrant regulation associated with increased local and systemic inflammation and metabolic dysfunction.5
The Maunakea lab located at John A. Burns School of Medicine is part of the Institute for Biogenesis Research in the Department of Anatomy, Biochemistry and Physiology. The lab focuses on the biological mechanisms underlying health disparities and studies diet as both an epigenetic modulator and as a potential intervention to reverse health inequities. Community-based participatory research studies that have engaged NHPI individuals and their social networks have offered new insight into the connection between diet, epigenetics, the microbiome composition, immune function, and response to viral infection. The lab refers to these interactions as the immunoepigenetic-microbiome axis. This article summarizes each component of this network and highlights the importance of understanding the relationships in the context of metabolic function and disease outcomes.
The Intestinal Microbiome
The gut microbiome plays a key role in metabolic syndrome and overall human health. Because microbiota facilitate functions in metabolic and immune responses, dysregulation of the gut microbiota that results in changes in the abundance and diversity of protective microbial species (ie, dysbiosis) is implicated in adverse health outcomes and disease progression.5 Dietary choices can influence microbiome composition and health outcomes, and there are differences observed between individuals who consume a westernized diet that is high in processed foods, animal protein, and low in dietary fiber, versus a non-westernized traditional diet that is plant-based and high in fiber.5
For example, fiber consumption has positive influences on microbiome composition and is usually associated with non-westernized diets.6 Non-digestible carbohydrates, also known as insoluble fiber or dietary fiber, are found in whole grains and some fruits and vegetables. The human gastrointestinal tract lacks digestive enzymes to process these sources of dietary fiber, but the gut microbiota utilizes insoluble fiber to synthesize short chain fatty acids (SCFAs) such as butyrate, acetate, and propionate via bacterial fermentation. SCFAs represent epigenetic regulators of the microbiome-immune axis and are mediators of immune activation. Locally, SCFAs such as butyrate are the main source of cellular energy of the colonic epithelium and contribute to strengthening the gut barrier and reducing intestinal inflammation. Systematically, SCFAs promote cellular metabolism and activate signaling cascades to regulate immune functions in colonic tissue and circulating immune cells.7
Immunoepigenetic-Microbiome Axis and Immunomodulatory Metabolites
The interactions between microbial derived metabolites and epigenetic regulation of immune cell function, part of the immunoepigenetic-microbiome axis, influence overall immune function and play major roles in disease. The innate immune system is shaped by epigenetic processes as seen in inflammatory states precursing type 2 diabetes pathogenesis. Microbial derivatives stimulate the innate immune system to cause cascading events that initiate transcriptional responses in specific cell types. Epigenetic modulators such as SCFAs recruit transcription factors, prevent expression of mediators, and repress or activate secondary gene programs responsible for metabolic functions such as histone modification and DNA methylation. Additionally, SCFA metabolites regulate immune responses resulting in either pro-inflammatory or anti-inflammatory cascades.8
SCFAs are important mediators between the intestinal microbiome and immune system. In the local gut environment, SCFAs strengthen gap junctions and epithelial barrier integrity.9 In doing so, they reduce the opportunity for pathogenic bacteria to elicit inflammatory cascades in macrophage and monocyte immune cells. Additionally, SCFAs are actively transported through the epithelial barrier and up taken by lymphocytes to further mediate immune activation. SCFAs have 2 major signaling mechanisms: inhibition of histone deacetylases (HDAC) and activation of G protein-coupled receptors (GPCRs). HDACs epigenetically regulate gene expression and are implicated in anti-inflammatory immune programming. SCFAs are ligands of GPCRs such as GPR41, GPR43, and GPR109A that are expressed on a wide variety of cells, including hematopoietic tissues and their derivative immune cells indicating modulatory activity of immune cell development and differentiation to produce anti-inflammatory states (Figure).9
Figure.
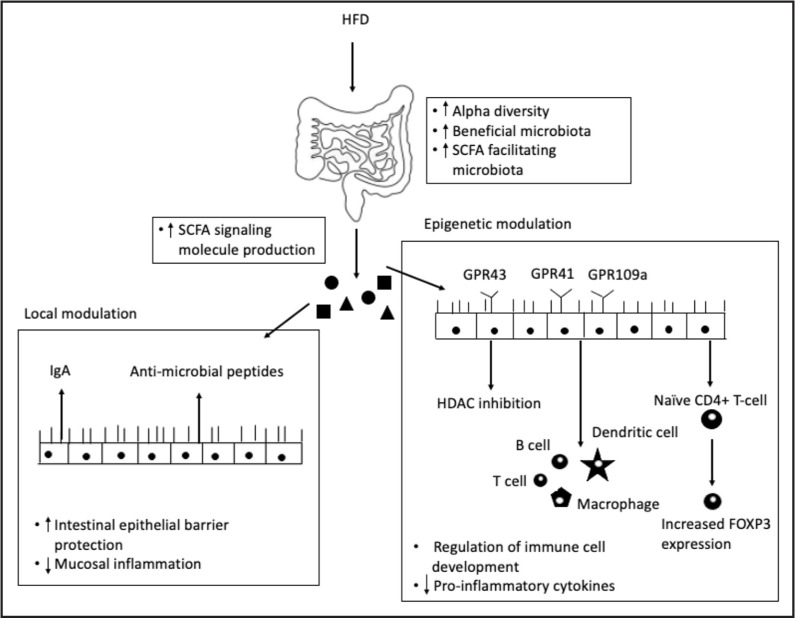
Diagram of the Immunoepigenetic-Microbiome Axis. Consumption of a high fiber diet enhances gut microbiome health and metabolic output to decrease systemic inflammation. Abbreviations: HFD, high fiber diet; SCFA, short chain fatty acid; HDAC, histone deacetylase
Epigenetic Modulation to Reduce Type 2 Diabetes Risk
The Mauli Ola study is an ongoing community-based research project of the Maunakea lab that includes NHPI participants interning at MA‘O Organic Farms in Wai‘anae, O‘ahu. This study provides an example of how diet-mediated changes in the gut microbiome can impact epigenetic regulation of the inflammation linked to glycemic control and risk for type 2 diabetes. Data from this study have revealed that individuals with higher fiber intake modulate their risk for type 2 diabetes as measured by changes in levels of hemoglobin A1c, a biomarker of glycemic control. The changes in A1c appeared to be mediated by increased butyrate-producing gut bacteria and associated with decreased systemic inflammation, which is consistent with other studies.10 Additionally, butyrate reduces systemic inflammatory cytokines IL-epigenetically regulated. This in turn causes a systemic cascade to decrease inflammation in the pancreas and adipose tissue, ultimately eliciti1b, TNF-a, IFN-g, IL-6 and MCP-1; the genes encoding these cytokines are ng homeostatic regulation of glucose uptake and insulin production. Furthermore, SCFA microbiota are associated with increased production of gut hormones PYY and GLP-1 which act through molecular pathways to further reduce type 2 diabetes by initiating insulin release, glucose uptake, and fatty acid oxidation.10
Diet, Microbiome and Type 2 diabetes
The results from the Mauli Ola study offers insight into the mechanisms in which SCFAs influence metabolic functions and reduce type 2 diabetes risk. Participants whose gut microbiomes exhibited increased alpha diversity scores, attributed to consuming high fiber diets (HFD), had greater capacity to produce SCFAs. This is because greater diversity was associated with increased abundance of SCFA-producing microbiota. Furthermore, participants with greater microbial diversity had increased presence of the butyrate kinase (Buk) gene and subsequent production of the butyrate kinase enzyme in the gut microbiome. Presence of the Buk gene enables microbiota to ferment non-digestible fiber into butyrate and this metabolic capacity was significantly associated with decreased type 2 diabetes risk. Notably, participants with confirmed type 2 diabetes suffered from a characteristically dysbiotic microbiome with constrained host-symbiotic interactions, elucidating possible dietary interventions to minimize or reverse pathogenesis of type II diabetes. Further research in the Maunakea lab is currently underway to investigate how the youth-targeted, land-based programs of MA‘O Organic Farms may offer an innovative intervention to ameriorate type II diabetes risk in the NHPI community.
Microbiome Composition and SARS-CoV-2
In another study of the Maunakea lab, data from participants recovering from severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection offered insights into how the immunoepigenetic-microbiome axis impact the development of coronavirus disease (COVID-19). Initial results of this study indicate that the gut microbiome appears to play a major role in the host-viral response and in some cases was protective against severe pathogenesis. Specifically, in comparison to those who harbored low gut microbiome diversity as measured by DNA sequencing technologies, participant plasma samples who exhibited a more diverse microbiome were able to neutralize the SARS-CoV-2 virus with better efficacy in ex vivo assessment; these individuals also experienced less severe symptoms of COVID-19. In addition, those with pre-existing conditions, such as obesity, tended to experience more severe symptoms of COVID-19 and had an attenuated immune response during recovery, which was associated with dysbiosis of the gut microbiome beyond what is characteristic of people with obesity. Although preliminary, these results reinforce the concept of a relationship between immune function and gut microbial composition. Given the protective role of a more diverse microbiome that is characteristic of a HFD, the work of the Maunakea lab implicates that interventions that include diet to reduce diabetes and/or obesity risk may be potentially useful to prevent and/or mitigate severe COVID-19 progression.
Conclusion
Systematically imposed socioeconomic constructs on Indigenous communities are partially responsible for unhealthy lifestyle choices that include poor nutrition. This has fostered the manifestation of gut microbiome dysbiosis that contributes to epigenetically altered immune responses and leads to the onset of inflammation underlying the progression of non-communicable diseases. Dysregulation of the immunoepigenetic-gut microbiome axis acts as a preface for many inflammatory related diseases because gut dysbiosis leads to increased intestinal permeability and subsequent mucosal inflammation. Prolonged perturbation of the immunoepigenetic-gut microbiome may be responsible for inducing systemic inflammation via epigenetic reprogramming of immune cells to influence lineage differentiation to pro-inflammatory states.
Thus, it is essential to study the immunoepigenetic-gut microbiome axis in diverse populations where access to healthy traditional diets is limited in order to better understand the degree such diets might serve to restore health in populations with high risk for metabolic diseases. Modifying diet offers interventional opportunity to enhance gut microbiome resilience and re-establish healthy metabolic and immune function. Understanding the impact of dietary modifications on the composition and function of the microbiome for favorable characteristics may allow for manipulation of the immunoepigenetic-microbiome axis to reduce metabolic dysregulation and deter onset of inflammation implicated in obesity and other co-morbidities associated with type 2 diabetes. Incorporating diet interventions can improve SCFA metabolite production, which could combat systemic inflammation through the immunoepigenetic-gut microbiome axis and may even protect against severe disease from viral infections such as COVID-19. As the microbiome is labile over the course of a lifetime and can rapidly respond to modifications, dietary interventions that consider the immunoepigenetic-microbiome axis may reveal new therapeutic and preventative strategies for populations afflicted by diseases of health disparities.
Acknowledgements
The authors wish to express sincere gratitude to the various agencies that in part supported some of the work described, including by grant numbers R03-HL146886 and R56-MD014630 from the National Institutes of Health (NIH), 20HCF-101573 from the Hawaii Community Foundation (HCF), and 010399-02 from Kamehameha Schools (KS). The contents of this work are solely the responsibility of the authors and do not represent the official views of the NIH, HCF, and KS.
Abbreviations and Acronyms
- COVID-9
coronavirus disease 2019
- HDAC
histone deacetylase
- HFD
high fiber diet
- GCPR
G protein-coupled receptor
- NHPI
Native Hawaiians and Pacific Islanders
- SCFA
short chain fatty acid
- SARS-CoV-2
severe acute respiratory syndrome coronavirus 2
References
- 1.Esposito K, Giugliano D. The metabolic syndrome and inflammation: association or causation? Nutr Metab Cardiovasc Dis NMCD. 2004;14((5)):228–232. doi: 10.1016/s0939-4753(04)80048-6. [DOI] [PubMed] [Google Scholar]
- 2.Gelaye B, Foster S, Bhasin M, Tawakol A, Fricchione G. SARS-CoV-2 morbidity and mortality in racial/ethnic minority populations: A window into the stress related inflammatory basis of health disparities? Brain Behav Immun - Health. 2020;9:100158. doi: 10.1016/j.bbih.2020.100158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McElfish PA, Purvis RS, Esquivel MK, et al. Diabetes disparities and promising interventions to address diabetes in Native Hawaiian and Pacific Islander populations. Curr Diab Rep. 2019;19((5)):19. doi: 10.1007/s11892-019-1138-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kaholokula JK, Samoa RA, Miyamoto RES, Palafox N, Daniels S-A. COVID-19 Special column: COVID-19 hits Native Hawaiian and Pacific Islander communities the hardest. Hawaii J Health Soc Welf. 2020;79((5)):144–146. [PMC free article] [PubMed] [Google Scholar]
- 5.Anand S, Mande SS. Diet, Microbiota and gut-lung connection. Front Microbiol. 2018;9 doi: 10.3389/fmicb.2018.02147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hills RD, Pontefract BA, Mishcon HR, Black CA, Sutton SC, Theberge CR. Gut microbiome: Profound implications for diet and disease. Nutrients. 2019;11((7)):1613. doi: 10.3390/nu11071613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Parada Venegas D, De la Fuente MK, Landskron G, et al. Short chain fatty acids (SCFAs)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front Immunol. 2019;10 doi: 10.3389/fimmu.2019.00277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Corrêa-Oliveira R, Fachi JL, Vieira A, Sato FT, Vinolo MAR. Regulation of immune cell function by short-chain fatty acids. Clin Transl Immunol. 2016;5((4)):e73. doi: 10.1038/cti.2016.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Alt FW. Advances in Immunology. Elsevier Science & Technology; 2014. Accessed June 3, 2021. http://ebookcentral.proquest.com/lib/uhm/detail.action?docID=1594323. [Google Scholar]
- 10.Bach Knudsen KE, Lærke HN, Hedemann MS, et al. Impact of diet-modulated butyrate production on intestinal barrier function and inflammation. Nutrients. 2018;10((10)) doi: 10.3390/nu10101499. [DOI] [PMC free article] [PubMed] [Google Scholar]


