Abstract
The alteration of the mucociliary clearance is a major hallmark of respiratory diseases related to structural and functional cilia abnormalities such as chronic obstructive pulmonary diseases (COPD), asthma and cystic fibrosis. Primary cilia and motile cilia are the two principal organelles involved in the control of cell fate in the airways. We tested the effect of primary cilia removal in the establishment of a fully differentiated respiratory epithelium. Epithelial barrier integrity was not altered while multiciliated cells were decreased and mucous‐secreting cells were increased. Primary cilia homeostasis is therefore paramount for airway epithelial cell differentiation. Primary cilia‐associated pathophysiologic implications require further investigations in the context of respiratory diseases.
Keywords: airway epithelium, cell differentiation, cilia
1. INTRODUCTION
Most primary cilia (PC) studies in respiratory research are restricted to development or repair processes. These solitary non‐mobile organelles act as sensors and molecular signalling hubs while motile cilia (MC) in differentiated epithelial cells are paramount for mucociliary clearance. 1 We recently demonstrated the presence of PC in human airway epithelial undifferentiated cells during homeostasis and in chronic obstructive pulmonary disease (COPD). 2 In addition, recent studies reported a decrease in multiciliated cells (MCC) in COPD patient‐derived bronchial and bronchiolar air‐liquid interface (ALI) cultures, 3 , 4 and we highlighted an abnormal cilia‐associated genomic signature in COPD patients (CiliOPD 5 ). As it was suggested that multiciliated cells (MCC) originated from primary ciliated cells (PCC), 6 we investigated the consequences of PC removal on human airway epithelial cell (AEC) differentiation.
2. MATERIALS AND METHODS
Detailed materials and methods are provided in the Appendix S1 section.
3. RESULTS
First, we confirmed the deciliation induced by CH on ALI AEC. As it was shown that deciliation occurred in less than 24 hours post‐treatment, 7 we maintained PC loss with daily treatment of CH. Considering that multiple PC per cell is a very rare event, we observed a sixfold decrease in PCC at ALI‐7 (3607 ± 778.5 PC/mm² in control (CTL) versus 582.3 ± 283.8 PC/mm² in CH‐treated cells) and a threefold decrease at ALI‐14 (5947 ± 891 PC/mm² in CTL versus 1745 ± 528.8 PC/mm² in CH‐treated cells) (Figure 1A and B). Key ciliogenesis‐associated genes such as FOXJ1, MCIDAS and HEATR2 8 were significantly down‐regulated upon CH treatment as early as 2 days after the initiation of differentiation (Figure 1C).
FIGURE 1.
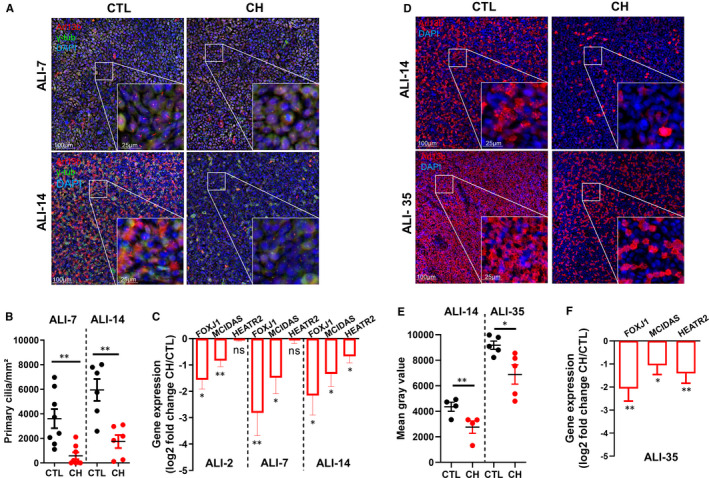
Loss of PC induces an alteration of motile ciliogenesis during AEC differentiation. A, Examples of micrographs taken from AEC cultures at ALI‐7 (upper panel) and ALI‐14 (bottom panel) showing PC (Arl13b, red; γ‐tubulin, green). Nuclei are stained in blue (DAPI). B, Dot plot (mean ±SEM) showing PC number per mm² at ALI‐7 (n = 8) and ALI‐14 (n = 6) in control (CTL) condition (black) and CH (red)‐treated cells. **P < 0.01 CH vs CTL. C, Histograms representing the assessment of fold‐change (log2) in the normalized expression to GAPDH during ALI cultures by RT‐qPCR (n = 11) for ciliogenesis markers FOXJ1, MCIDAS and HEATR2 at ALI‐2; ALI‐7 and ALI‐14. Results show mean ±SEM, **P < 0.005, *P < 0.05 CH vs CTL. D, Examples of micrographs taken from AEC cultures at ALI‐14 (left panel) and ALI‐35 (right panel) showing MC (Arl13b, red). Nuclei are stained in blue (DAPI). E, Dot plot (mean ±SEM) represents the mean grey values of MC‐associated fluorescence in CTL condition (black; n = 4) and CH‐treated cells (red; n = 5) at ALI‐14 and ALI‐35. **P < 0.01 CH vs CTL and *P < 0.05 CH vs CTL. F, Histograms representing the assessment of fold‐change CH/CTL (log2) in the normalized expression to GAPDH during ALI cultures by RT‐qPCR (n = 10) for ciliogenesis markers FOXJ1, MCIDAS and HEATR2 at ALI‐35. Results show mean ±SEM, **P < 0.005, *P < 0.05 CH vs CTL
We next investigated the impact of PC removal on MC establishment. As daily treatment with CH ultimately leads to cell death after 14 days of culture, we maintained PC inhibition with CH treatment every other day instead. In this condition, non‐differentiated AEC entered a 48‐hour cycle of ciliogenesis/deciliation for the duration of ALI cell culture. 7 Chloral hydrate‐treated ALI cell cultures presented significantly less MCC than control ALI cell cultures with a 40% reduction at ALI‐14 and a 25% reduction at ALI‐35 (Figure 1D and E). The down‐regulation of key ciliogenesis‐associated genes persisted at ALI‐35 (Figure 1F). These findings suggest that PC are essential to establish a fully differentiated epithelium with MCC.
To further address the role of PC in AEC differentiation, we next analysed epithelial barrier integrity. Transepithelial electrical resistance (TEER) was significantly reduced during differentiation with a sixfold decrease in ALI‐35 in CH‐treated cells (Figure 2A). Therefore, we evaluated tight junction assembly and apoptosis. We did not find any differences in the junctional network whereas apoptosis was slightly increased (Figure 2B and C). This suggests that the loss of TEER is likely caused by a reduction in epithelial height and a lack of differentiation rather than a permeability issue. To further investigate the global epithelial differentiation programming, we assessed the transcript levels of markers for non‐differentiated cells (CK5, SOX2, SOX9) and secretory cells (SPDEF, SCGB1A1, MUC5AC, MUC5B). We observed a significant decrease in non‐differentiated cell markers, and a trend towards the decreased expression of all the others (Figure 2D). Considering that an increase in mucous‐secreting cells is a common feature of several lung diseases, we also evaluated the production of the two main mucins in the lung: Muc5ac and Muc5b. Interestingly, Muc5b‐secreting cells but not Muc5ac‐secreting cells showed a threefold increase in CH‐treated ALI cultures (Figure 2E and F). These findings suggest that PC are essential to warrant epithelial cell integrity and orientate cell fate determination during AEC differentiation (Figure 2G).
FIGURE 2.
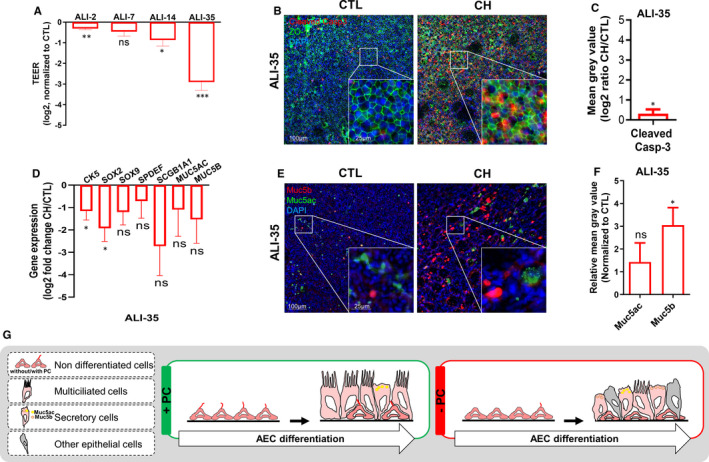
Loss of PC induces global epithelial remodelling during AEC differentiation. A, Histograms representing the TEER (log2, normalized to CTL, n = 6) of CH‐treated ALI cultures (n = 6). Results show mean ±SEM, ***P < 0.001, **P < 0.01, *P < 0.05 CH vs CTL. B, Examples of micrographs taken from AEC cultures at ALI‐35 showing cleaved caspase‐3 (red) and Zonula occludens‐1 (ZO1, green). Nuclei are stained in blue (DAPI). C, Histogram representing mean grey values of the cleaved caspase‐3‐associated fluorescence of CH‐treated AEC (n = 3) in log 2 ratio CTL/CH at ALI‐35. *P < 0.05 CH vs CTL. D, Histograms representing the assessment of fold‐change (log2) in the normalized expression to GAPDH during ALI cultures by RT‐qPCR (n = 11) for non‐differentiated cell markers (CK5, SOX2, SOX9), and secretory cell markers (SPDEF, SCGB1A1, MUC5AC, MUC5B) at ALI‐35. Results show mean ±SEM, *P < 0.05 CH vs CTL. E, Examples of micrographs taken from AEC cultures at ALI‐35 showing mucins (Muc5b, red; Muc5ac, green). Nuclei are stained in blue (DAPI). F, Histogram representing the relative mean grey values of the mucins‐associated fluorescence normalized to CTL at ALI‐35 of CH‐treated cells for Muc5b and Muc5ac (n = 9). *P < 0.05 CH vs CTL. G, Illustration summarizing in vitro AEC remodelling upon PC inhibition
4. DISCUSSION
To our knowledge, we provided the first experimental demonstration that PC directly orchestrate MCC differentiation and are crucial for AEC homeostasis. We confirmed previous in vitro observations of transient PC 6 but in contrast, we detected non‐transient PC in human adult non‐differentiated AEC during differentiation. This was consistent with our previous report on human lung adult tissues. 2
Deciliation has been indicted in epithelial remodelling, 7 and the cell populations obtained after 35 days of culture upon primary ciliogenesis inhibition described two major features of respiratory diseases characterized by remodelled epithelium including a decrease in MCC and an increase in goblet cells. 9 Our findings highlight the role of PC in the process of AEC differentiation. This is particularly important as a recent study demonstrated the presence of a hybrid cilium in MCC with PC features, 10 and PC functions are discussed in several respiratory diseases including COPD, asthma and idiopathic pulmonary fibrosis. 2 , 11 , 12
Given that PC are involved in many signalling pathways, a better understanding of their involvement in lung physiology and pathologies may help identify crucial molecular actors to expand diagnosis, prognosis and therapeutics. Hedgehog pathway appears as the first candidate that has been connected to PC during lung development, 13 and has been associated with AEC differentiation and lung diseases. 14 , 15 Two other crucial developmental signalling pathways, the canonical Wnt and Notch, have also been highlighted in human AEC remodelling and were further associated with COPD features. 16 , 17 Therefore, integrating the key players of organogenesis in the context of the global cilia‐associated alterations in lung diseases may pave the way towards the identification of promising biomarkers and targeted therapies.
There are two main limitations to our study. First, experimental approaches to reach deciliation are limited to gene silencing 18 or pharmacological treatments with their associated caveats. 19 Although CH targets highly stabilized microtubules (ie PC), non‐stabilized microtubules may also be impacted. Second, we used AEC isolated from nasal polyps as a surrogate of respiratory epithelia. It will be important to confirm our results on bronchial and bronchiolar epithelial cells.
In conclusion, we have shown that primary ciliogenesis is a crucial step for MCC determinism in AEC differentiation. We propose to include PC as important organelles to investigate in the context of respiratory research. In addition, the exploration of signalling pathways related to PC in the adult lung may provide novel perspectives to understand lung diseases associated with cilia alterations.
CONFLICT OF INTEREST
Dr Ruaux reports grants and personal fees from Sanofi‐Aventis outside the submitted work. Dr Deslée reports personal fees from Nuvaira, personal fees from BTG/PneumRx, personal fees from Chiesi, personal fees from Boehringer and personal fees from Astra Zeneca, outside the submitted work. Dr Dormoy reports personal fees from Chiesi outside the submitted work.
AUTHOR CONTRIBUTION
Randa Belgacemi: Conceptualization (equal); Formal analysis (equal); Investigation (equal); Writing‐original draft (equal); Writing‐review & editing (equal). Zania Diabasana: Formal analysis (supporting); Investigation (supporting); Writing‐review & editing (supporting). Antony Hoarau: Formal analysis (equal); Investigation (equal); Writing‐review & editing (supporting). Xavier Dubernard: Formal analysis (equal); Resources (equal); Writing‐review & editing (supporting). Jean‐Claude Mérol: Formal analysis (equal); Resources (equal); Writing‐review & editing (supporting). Christophe Ruaux: Formal analysis (equal); Resources (equal); Writing‐review & editing (supporting). Myriam Polette: Formal analysis (equal); Investigation (equal); Writing‐review & editing (equal). Jeanne‐Marie Perotin: Formal analysis (equal); Investigation (equal); Methodology (equal); Resources (equal); Writing‐original draft (equal); Writing‐review & editing (equal). Gaëtan Deslée: Formal analysis (equal); Investigation (equal); Resources (lead); Supervision (equal); Writing‐original draft (equal); Writing‐review & editing (equal). Valérian Dormoy: Conceptualization (lead); Formal analysis (lead); Funding acquisition (lead); Investigation (lead); Methodology (lead); Project administration (lead); Supervision (lead); Writing‐original draft (lead); Writing‐review & editing (lead).
5. ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the institutional review board of the University Hospital of Reims, France (IRB Reims‐CHU 20 110 612), and was conducted in accordance with the ethical guidelines of the Declaration of Helsinki. All patients gave their written informed consent prior to inclusion in the study.
Supporting information
Appendix S1
ACKNOWLEDGEMENTS
We thank the members of the Inserm UMR‐S 1250 and our collaborators for their helpful comments and insights. We thank the Clinic “La Sagesse” for their collaboration on sample collection.
Belgacemi R, Diabasana Z, Hoarau A, et al. Primary ciliogenesis is a crucial step for multiciliated cell determinism in the respiratory epithelium. J Cell Mol Med. 2021;25:7575–7579. 10.1111/jcmm.16729
Funding information
This work was supported by University of Reims Champagne‐Ardenne and the French National Institute of Health and Medical Research (Inserm).
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1. Bustamante‐Marin XM, Ostrowski LE. Cilia and mucociliary clearance. Cold Spring Harb. Perspect. Biol. 2017;9(4):a028241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Perotin J‐M, Coraux C, Lagonotte E, et al. Alteration of primary cilia in COPD. Eur Respir J. 2018;52. [DOI] [PubMed] [Google Scholar]
- 3. Gohy S, Carlier FM, Fregimilicka C, et al. Altered generation of ciliated cells in chronic obstructive pulmonary disease. Sci Rep. 2019;9:17963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Gindele JA, Kiechle T, Benediktus K, et al. Intermittent exposure to whole cigarette smoke alters the differentiation of primary small airway epithelial cells in the air‐liquid interface culture. Sci Rep. 2020;10:6257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Perotin J‐M, Polette M, Deslée G, Dormoy V. CiliOPD: A ciliopathy‐associated COPD endotype. Respir Res. 2021;22:74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Jain R, Pan J, Driscoll JA, et al. Temporal relationship between primary and motile ciliogenesis in airway epithelial cells. Am J Respir Cell Mol Biol. 2010;43:731‐739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Overgaard CE, Sanzone KM, Spiczka KS, Sheff DR, Sandra A, Yeaman C. Deciliation is associated with dramatic remodeling of epithelial cell junctions and surface domains. Mol Biol Cell. 2009;20:102‐113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Horani A, Ustione A, Huang T, et al. Establishment of the early cilia preassembly protein complex during motile ciliogenesis. Proc. Natl. Acad. Sci. U. S. A. 2018;115:E1221‐E1228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kesimer M, Ford AA, Ceppe A, et al. Airway mucin concentration as a marker of chronic bronchitis. N Engl J Med. 2017;377:911‐922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Liu Z, Nguyen QPH, Nanjundappa R, et al. Super‐resolution microscopy and FIB‐SEM imaging reveal parental centriole‐derived, hybrid cilium in mammalian multiciliated cells. Dev Cell. 2020;55:224‐236.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Trempus CS, Song W, Lazrak A, et al. A novel role for primary cilia in airway remodeling. Am J Physiol Lung Cell Mol Physiol. 2017;313:L328‐L338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Tilley AE, Walters MS, Shaykhiev R, Crystal RG. Cilia dysfunction in lung disease. Annu Rev Physiol. 2015;77:379‐406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Kugler MC, Joyner AL, Loomis CA, Munger JS. Sonic hedgehog signaling in the lung. From development to disease. Am J Respir Cell Mol Biol. 2015;52:1‐13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Belgacemi R, Luczka E, Ancel J, et al. Airway epithelial cell differentiation relies on deficient Hedgehog signalling in COPD. EBioMedicine. 2020;51:102572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ancel J, Belgacemi R, Perotin J‐M, et al. Sonic hedgehog signalling as a potential endobronchial biomarker in COPD. Respir Res. 2020;21:207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Carlier FM, Dupasquier S, Ambroise J, et al. Canonical WNT pathway is activated in the airway epithelium in chronic obstructive pulmonary disease. EBioMedicine. 2020;61:103034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bodas M, Moore AR, Subramaniyan B, et al. Cigarette smoke activates NOTCH3 to promote goblet cell differentiation in human airway epithelial cells. Am J Respir Cell Mol Biol. 2021;64:426‐440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Wheway G, Schmidts M, Mans DA, et al. An siRNA‐based functional genomics screen for the identification of regulators of ciliogenesis and ciliopathy genes. Nat Cell Biol. 2015;17:1074‐1087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Khan NA, Willemarck N, Talebi A, et al. Identification of drugs that restore primary cilium expression in cancer cells. Oncotarget. 2016;7:9975‐9992. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix S1
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


