Abstract
Background
The long-term post acute pulmonary sequelae of COVID-19 remain unknown.
Purpose
To evaluate lung injury in patients affected by COVID-19 pneumonia at six-month follow-up compared to baseline chest CT.
Methods
From March 19th,2020 to May 24th,2020, patients with moderate to severe COVID-19 pneumonia and baseline Chest CT were prospectively enrolled at six-months follow-up. CT qualitative findings, semi-quantitative Lungs Severity Score (LSS) and well-aerated lung quantitative Chest CT (QCCT) were analyzed. Baseline LSS and QCCT performances in predicting fibrotic-like changes (reticular pattern and/or honeycombing) at six-month follow-up Chest CT were tested with receiver operating characteristic curves. Univariable and multivariable logistic regression analysis were used to test clinical and radiological features predictive of fibrotic-like changes. The multivariable analysis was performed with clinical parameters alone (clinical model), radiological parameters alone (radiological model) and the combination of clinical and radiological parameters (combined model).
Results
One-hundred-eighteen patients, with both baseline and six-month follow-up Chest CT, were included in the study (62 female, mean age 65±12 years). At follow-up Chest CT, 85/118 (72%) patients showed fibrotic-like changes and 49/118 (42%) showed GGOs. Baseline LSS (>14), QCCT (≤3.75L and ≤80%) showed an excellent performance in predicting fibrotic-like changes at Chest CT follow-up. In the multivariable analysis, AUC was .89 (95%CI .77-.96) for the clinical model, .81 (95%CI .68-.9) for the radiological model and .92 (95%CI .81-.98)for the combined model.
Conclusion
At six-month follow-up Chest CT, 72% of patients showed late sequelae, in particular fibrotic-like changes. Baseline LSS and QCCT of well-aerated lung showed an excellent performance in predicting fibrotic-like changes at six-month Chest CT (AUC>.88). Male sex, cough, lymphocytosis and QCCT well-aerated lung were significant predictors of fibrotic-like changes at six-month with an inverse correlation (AUC .92).
See also the editorial by Wells and Devaraj.
Summary
At 6-month follow-up chest CT, COVID-19 postacute sequelae were detected in 72% of patients: fibrotic-like changes were the most common residual findings (72%), followed by ground-glass opacities (42%).
Key Results
Baseline Lung Severity Score (>14) showed an optimal performance in predicting fibrotic-like changes at follow-up (AUC: .91; sensitivity: 88%; specificity: 80%).
Multivariable analysis showed that male sex, cough, lymphocytosis and Quantitative Chest Computed Tomography well-aerated lung volume were significant predictors of fibrotic-like changes at six-month follow-up with an inverse correlation (AUC: .92; sensitivity: 100%; specificity: 73%).
Introduction
Coronavirus disease 2019 (COVID-19) is caused by a novel coronavirus, known as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and has been declared a pandemic by the World Health Organization (WHO) on March 11, 2020 (1).
Chest CT qualitative findings of COVID-2019 pneumonia have been deeply investigated in literature (2), as well as quantitative Chest CT (QCCT) methods (3–5) and semiquantitative lung severity scores (LSS) (6, 7). Typical Chest CT findings of COVID-19 pneumonia include bilateral and multilobe ground glass opacities (GGOs) with posterior and peripheral distribution associated with consolidations, interlobular septal thickening and subsegmental pulmonary vessels enlargement (> 3 mm) (8).
The short- and mid-term Chest CT outcomes in COVID-19 patients (9, 10) have been reported in some studies, with both qualitative and semiquantitative methods (11–13); fibrosis was a common finding at few weeks from the onset of the symptoms (14–16).
A recent study demonstrated that approximately one-third of COVID-19 survivors showed pulmonary fibrotic-like changes at six-month follow-up Chest CT (17); nevertheless, as a novel pathological entity, further long-term investigations are needed for a full comprehensive knowledge of COVID-19 pulmonary sequelae. Indeed, there is great concern that some of these findings will resolve over time, and are therefore not fibrosis (18).
In fact, for previous different severe viral pneumonias, several studies demonstrated how long-term lung impairment is a common sequelae (19); among them the long-term follow-up study on severe acute respiratory syndrome (SARS) by Zhang et al. (20) and Wu et al. (21) demonstrated the persisting pulmonary interstitial damage several months after recovery.
Thus, the aims of our study are: 1) to prospectively investigate Chest CT late sequelae in patients affected by COVID-19 pneumonia at six-month follow-up and comparing CT features with those present at baseline Chest CT; 2) to evaluate the role of Chest CT and clinical parameters in predicting pulmonary fibrotic-like changes at six-month follow-up.
Materials and Methods
Patient Population and Study Design
This prospective study was approved by our local institutional review board and written informed consent was obtained from all study participants.
Patients admitted at Sant'Andrea University Hospital, Rome, Italy, from March 19th, 2020 to May 24th, 2020, with the both following inclusion criteria of (a) selected patients with diagnosis of moderate to severe COVID-19 (22), confirmed by reverse transcription polymerase chain reaction (RT-PCR) who underwent Chest CT and (b) positive baseline Chest CT scans with diagnosis of interstitial pneumonia performed at admission, were prospectively enrolled for a six-month follow-up Chest-CT evaluation. In order to define the severity of COVID-19 pneumonia, the WHO's interim guidance diagnostic criteria for adults with severe COVID-19 pneumonia were used (22).
Exclusion criteria were as follows: (a) patient death during follow-up interval; (b) refusal of the patient to undergo follow-up Chest CT; (c) severe motion artifacts at first Chest CT.
On the day of follow-up CT, all patients filled out a questionnaire about the long-lasting symptoms after COVID-19, the smoking habit; oxygen saturation was also measured and recorded for each patient. Patients' demographic characteristics, clinical findings, laboratory results and eventual ventilation support and corticosteroid therapy during the hospitalization were also retrieved from the internal hospital records.
CT Acquisition Technique
All patients underwent six-month (± 14 days) follow-up unenhanced Chest CT scans. Chest CT acquisitions were obtained with the patients in supine position during end-inspiration without contrast medium injection. Chest CT was performed on a 128-slice CT (GE Revolution EVO CT Scanner, GE Medical Systems, Milwaukee, WI, USA). The following technical parameters were used: tube voltage: 120 kV; tube current modulation 100-250 mAs; spiral pitch factor: 0.98; collimation width: 0.625. Reconstructions were made with convolution kernel BONEPLUS at a slice thickness of 1.25mm.
Qualitative Analysis
DICOM data were transferred into a PACS workstation (Centricity Universal Viewer v.6.0, GE Medical Systems, Milwaukee, WI, USA). Two radiologists in consensus (AC and CDD, with 5 and 20 years in thoracic imaging experience respectively), qualitatively analyzed Chest CT images and evaluated the presence of motion artifacts. The following CT findings were recorded: (a) GGOs, (b) GGOs pattern, (c) GGOs location, (d) multilobe involvement, (e) total lobar involvement, (f) bilateral distribution, (g) location of consolidation or GGOs, (h) consolidation, (i) interlobular septal thickening, (j) fibrotic-like changes (reticular pattern and/or honeycombing) (23), (k) bronchiectasis, (l) air bronchogram, (m) bronchial wall thickening, (n) pulmonary nodules surrounded by GGOs, (o) halo sign or reversed halo sign, (p) pleural and (q) pericardial effusion, (r) lymphadenopathy (defined as lymph node with short axis > 10mm) (s) subsegmental vessel enlargement (≥3mm) and (t) pulmonary trunk diameter (<31mm).
All mentioned Chest CT findings were defined as in the Fleischner Society glossary (24).
Lung Severity Score Analysis
The presence of GGOs, consolidation and fibrotic-like changes were semi-quantitatively analyzed in consensus by the same two radiologists above-mentioned, using the LSS ranging from 0 to 40 points, previously used in literature to quantify COVID-19 pneumonia lung impairment (25). Ten segments for each lung were considered; each segment was evaluated 0-2 points on the basis of the area involved, with score 0 for normal parenchyma, 1 for less than 50% segmental involvement and 2 for up to 50% segmental involvement. The final LSS was obtained from the sum of all lung's segments; furthermore, individual segmental scores were added together in total score to perform the statistical analysis.
Quantification Chest CT Analysis
Other two radiologists in consensus (GG and DC, with 3 and 7 years of experience in thoracic imaging respectively) performed QCCT analysis to quantify well-aerated lung by using a dedicated software (Thoracic VCAR v13.1, GE). Attenuation value < −1000 HU was used to exclude trachea air from the analysis, before segmentation. In order to select well-aerated lung for software segmentation, a range between -950 HU and -700 HU density, was selected (26–28). The software automatically calculated the healthy lung expressed both in percentage and liters; in case of unsatisfactory or incorrect lung segmentation, the radiologists manually adjust the lung contours.
Statistical Analysis
Statistical analysis was performed using SPSS version 21.0 (SPSS Inc. Chicago, IL) and MedCalc Statistical Software version 17.9.7 (MedCalc Software bvba, Ostend, Belgium). P values <.05 were considered statistically significant. All data are expressed as mean ± Standard Deviation (SD). Categorical variables were described as counts and percentage; qualitative CT findings comparison was performed using the Chi-square test. The comparison between baseline and follow-up LSS and well-aerated lung QCCT analysis were tested with Student's t test in case of Gaussian distribution of data, otherwise while Wilcoxon test was applied.
Receiver operating characteristic (ROC) curves and Area under the curve (AUC) were calculated to test the performance of the baseline LSS and QCCT to predict fibrotic-like changes at six-month follow-up Chest CT. Univariable and backward multivariable logistic regression analysis were used to test clinical and radiological features as predictor of fibrotic-like changes at six-month follow-up. At first, univariable analysis was individually performed on clinical variables: age, gender, comorbidities (dichotomizing with presence/absence), smoking habit (dichotomizing with present and former smoker grouped together and non-smokers), and baseline parameters as cough, dyspnea, fever, CRP, d-Dimer, LDH, lymphocytes, saturation, ventilation support; baseline radiological variables as LSS, pulmonary bilateral involvement, consolidations and well-aerated lung QCCT analysis (L and %). The multivariable analysis was performed on three different models: the clinical model included age, gender, comorbidities, smoking habit, cough, dyspnea, fever, CRP, d-Dimer, LDH, lymphocytes, saturation at admission and ventilation support; the radiological model contained LSS, pulmonary bilateral involvement, consolidations and well-aerated lung QCCT analysis (L and %); finally the combined model comprised all these parameters.
Results
Patient population, clinical data and laboratory findings
From an initial population of 288 patients, 98 (34%) died during the follow-up period, 45 (16%) refused the follow-up Chest CT since already performed elsewhere and 27 (9.4%) patients were excluded for the presence of severe motion artifacts on Chest CT at admission; thus, the final population comprised 118 patients, 56 male (48%), mean age 65 ± 12 years (range 37-84 years). The enrollment flowchart of the study is showed in Figure 1.
Figure 1.
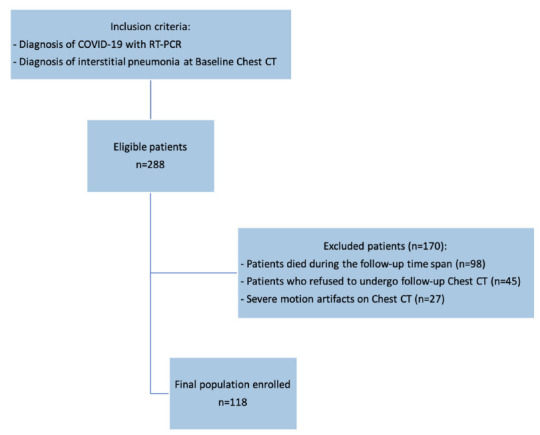
Enrollment flow-chart of the study. From an initial cohort of 288 Patients, 118 patients with both baseline and six-month follow-up Chest CT were enrolled.
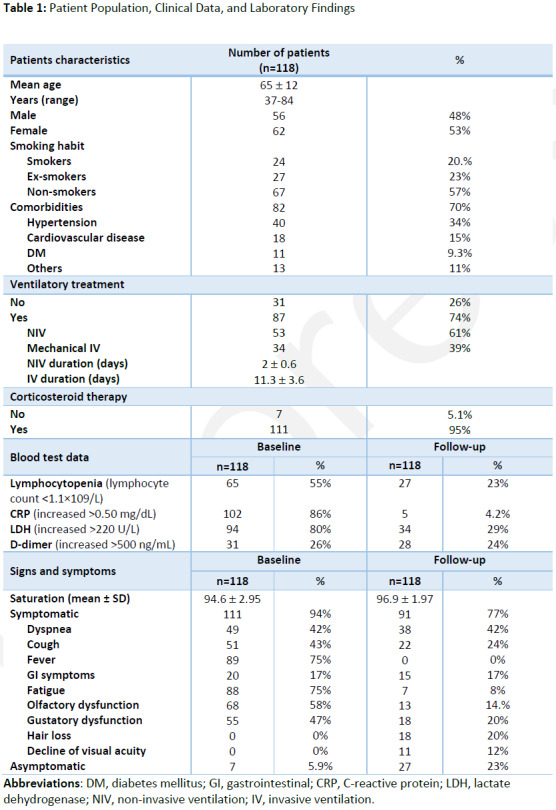
Twenty-four/118 (20%) patients had smoking habit, 27/118 (23%) had quit tobacco and 67/118 (57%) had never smoked. Twenty-four/118 patients (20%) had one underlying comorbidities, while 58/118 patients (49%) had two or more comorbidities; common comorbidities included hypertension (40/118 cases, 34%), cardiovascular diseases (18/118 cases, 15%) and diabetes mellitus (11/118 cases, 9%).
Ninety-one/118 patients (77%) reported persisting or new symptoms, including cough (22/91, 24%), dyspnea (38/91, 42%), fatigue (7/91, 8%), GI symptoms (15/91, 17%), hair loss (18/91, 20%), decline of visual acuity (11/91, 12%), olfactory and gustatory dysfunctions (13/91, 14% and 18/91, 20%, respectively); 27/118 patients (23%) were asymptomatic and mean oxygen saturation was 97% ± 1.97.
Full data about patients' demographics, clinical records, laboratory findings and eventual ventilation support and corticosteroid therapy are reported in Table 1.
Table 1:
Patient Population, Clinical Data, and Laboratory Findings
Qualitative Analysis
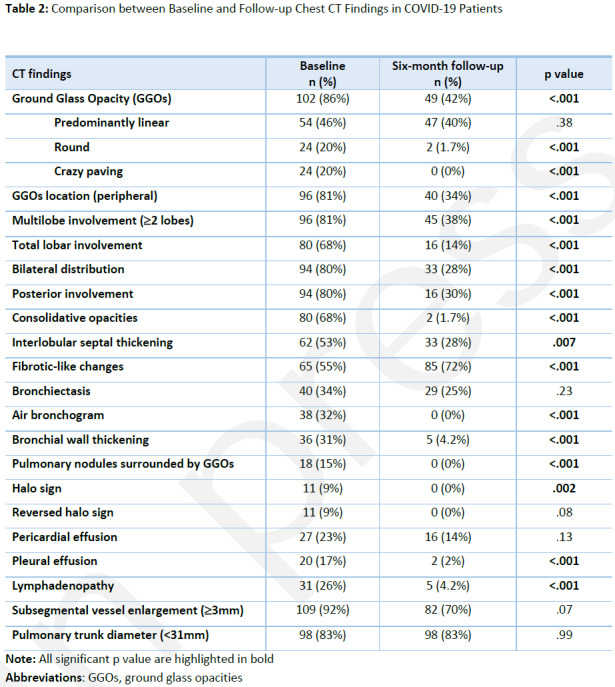
At six-month Chest CT follow-up, 33/118 (28%) presented with normal lungs. Eight-five/118 (72%) patients presented with fibrotic-like changes, 49/118 (42%) with GGOs, peripheral in distribution in 40/118 (34%) cases. Multilobe involvement (≥ 2 lobes) was observed in 45/118 (38%) patients and involvement of all lobes in 16/118 (14%) cases. Interstitial septal thickening was detected in 33/118 (28%) cases and consolidative opacities were observed in 2/118 patient (2%).
All Chest CT findings were reduced at follow-up Chest CT, except for the fibrotic-like changes which was the only feature showing a significant increase (all p<.05). Full results about the follow-up Chest CT findings and the comparison with baseline Chest CT findings are shown in Table 2.
Table 2:
Comparison between Baseline and Follow-up Chest CT Findings in COVID-19 Patients
Chest CT examples of baseline and six-month follow-up are shown in Figures 2 and Figure 3.
Figure 2.
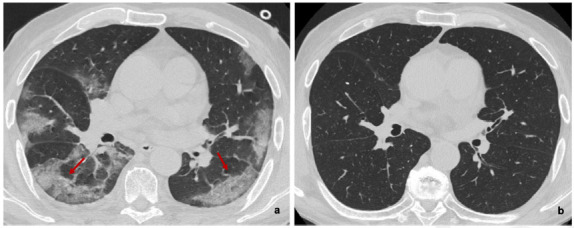
(a) Baseline and (b) six-month follow-up axial thin-section unenhanced Chest CT scans of 83-year-old man, former smokers, who presented fever, cough and worsening dyspnea; COVID-19 was confirmed by reverse transcription polymerase chain reaction (RT-PCR) testing. (a) Baseline scan shows multiple bilateral and confluent ground-glass opacities with predominantly linear pattern and peripheral distribution (red arrows). (b) Six-month follow-up scan shows complete resolution of ground-glass opacities without fibrotic-like changes.
Figure 3.
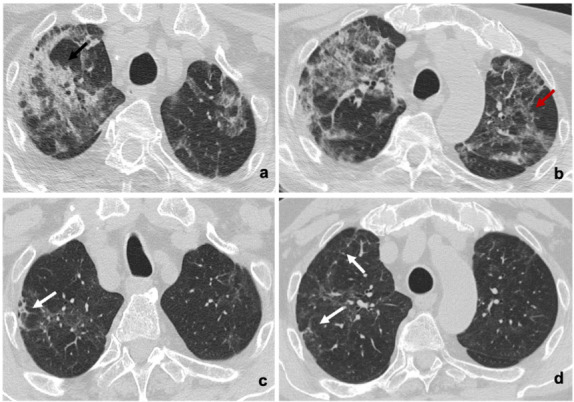
(a,b) Baseline and (c,d) six-month follow-up axial thin-section unenhanced Chest CT scans of 84-year-old man, smoker, admitted to the Emergency Department presenting fever and cough; COVID-19 was confirmed by reverse transcription polymerase chain reaction (RT-PCR) testing. (a,b) Images show bilateral consolidative pulmonary opacities (black arrow) with diffuse ground-glass opacities and interstitial septal thickening (red arrow). (c,d) Six-month follow-up scans show residual ground-glass opacities with decreased density compared to baseline, interstitial septal thickening and peripheral fibrotic-like changes (white arrows).
Lung Severity Score Analysis
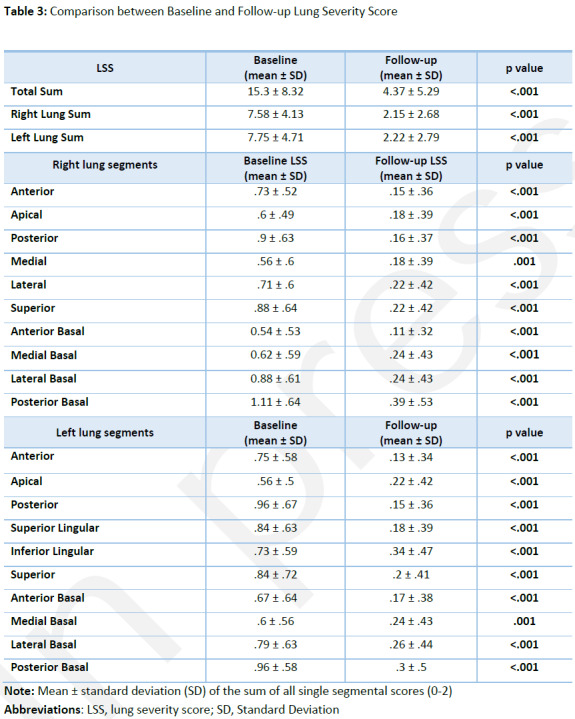
At six-month follow-up total LSS was 4.37 ± 5.29 (range 0-20; LSS in right and left lung 2.15 ± 2.68 and 2.22 ± 2.79 respectively), showing a significant decrease compared with the total sum of baseline LSS 15.34 ± 8.32 (range 0-30; LSS in the right and left lung 7.58 ± 4.13 and 7.75 ± 4.71 respectively), (all p<.001). A per-segment analysis was also performed and fully reported in Table 3.
Table 3:
Comparison between Baseline and Follow-up Lung Severity Score
Quantitative Chest CT Analysis
Six-month follow-up QCCT analysis of well-aerated lung expressed in percentage and liters was 82% ± 12.27 and 3.84 L ± 1.34, respectively, showing significant differences compared to baseline (69% ± 18.2 and 2.99 L ± 1.5, all p<.001). An example of baseline and follow-up comparison QCCT analysis is provided in Figure 4.
Figure 4.
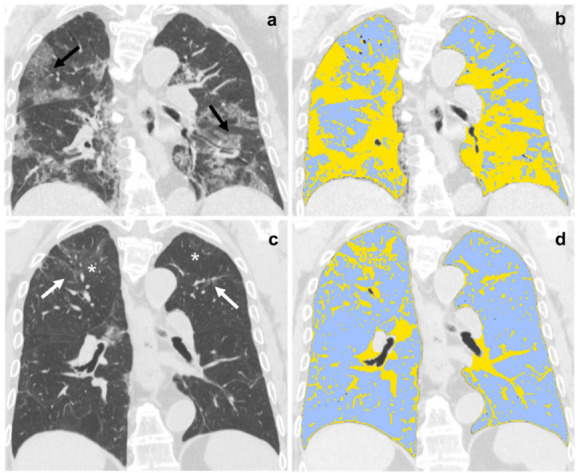
(a,b) Baseline and (c,d) six-month follow-up coronal thin-section unenhanced Chest CT scans of 79-year-old man, admitted to the Emergency Department presenting fever, dyspnea and cough; COVID-19 was confirmed by reverse transcription polymerase chain reaction (RT-PCR) testing. (a) Chest CT scan shows bilateral ground-glass opacities tending to consolidation (black arrow). (b) The same scan after Quantitative Chest CT Analysis highlighted in light-blue well-aerated lung (1.5 liters, 50%) and in yellow pulmonary injury of COVID-19 pneumonia. (c) Six-month follow-up scan shows residual fibrotic-like changes (white arrows) and persisting of low-density ground glass (asterisks). (d) The same scan after Quantitative Chest CT Analysis highlighted in light-blue well-aerated lung (3.5 liters, 82%) and in yellow residual findings of COVID-19 pneumonia at six months follow-up.
ROC curves and logistic regression
Baseline LSS showed an excellent performance in predicting fibrotic-like changes at six-month Chest CT follow-up with AUC of .91, 95%CI .8-.97, sensitivity of 88% and specificity of 80% when the LSS cutoff was >14. Baseline QCCT of well-aerated lung, expressed both in liters and percentage, showed an AUC of .88 (95%CI .77-.96, sensitivity of 86%, specificity of 80%, cutoff of ≤3.75L) and .88 (95%CI .76-.95, sensitivity of 74%, specificity of 100%, cutoff of ≤80%), respectively, as shown in Figure 5.
Figure 5.
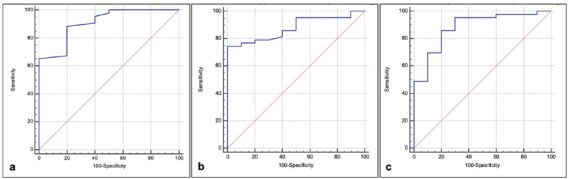
(a) Receiver operating characteristic (ROC) curves tested the performance of baseline lung severity score to predict fibrotic-like changes at six-month follow-up Chest CT, showing an area under the curve (AUC) of .91, 95%CI .8-.97, sensitivity of 88% and specificity of 80% when the cut-off was >14. (b,c) ROC curves tested the performance of baseline quantitative Chest CT (QCCT) analysis of well-aerated lung, expressed in percentage (b) and Liters (c) to predict fibrotic-like changes at six-month follow-up Chest CT: (b) with the cut-off of ≤3.8L an AUC of .88, 95%CI .77-.96, a sensitivity of 86% and a specificity of 80% was found, (c) the cut-off of ≤80% showed an AUC of .88, 95%CI .76-.95, 74% of sensitivity and 100% of specificity.
In the univariable analysis, age >65 (OR, 1.1; 95%CI, 1.03-1.17; p=.004) and baseline LSS (OR, 1.15; 95%CI, 1.05-1.25; p=.003) have proven to be significant positive independent variable for fibrotic-like changes at six-month follow-up Chest CT (all p<.05). On the other side, male gender (OR, .33; 95%CI, .92-1.14; p=.002), saturation=96-97 (OR, .13; 95%CI, .02-.95; p=.04), the absence of the need for ventilation (OR, .16; 95%CI, .04-.63; p=.008), bilateral lung involvement (OR, .28; 95%CI, .06-.92; p=.04), pulmonary consolidations (OR, .27; 95%CI, .08-.96; p=.04) and baseline QCCT well-aerated lung expressed in liters (OR, .4; 95%CI, .22-.71; p=.002) and in percentage (OR, .93; 95%CI, .88-.99; p=.01), showed significant inverse correlation for the presence of fibrotic-like changes at six-month follow-up Chest CT (all p<.05).
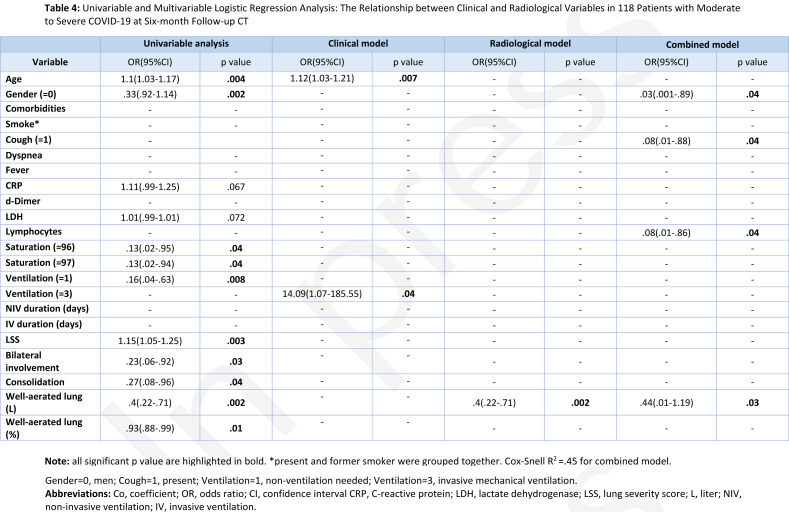
In the multivariable clinical model analysis, the best positive predictors of fibrotic-like changes at six-month follow-up Chest CT were age >65 years (OR, 1.12; 95%CI, 1.03-1.21; p=.007) and the need for mechanical ventilation (OR, 14.02; 95%CI, 1.07-185.55; p=.04). Otherwise, the radiological model showed an inverse correlation between quantified well-aerated lung and fibrotic-like changes (OR, .4; 95%CI, .22-.71; p=.002). Finally, the combined model showed that male gender (OR, .03; 95%CI, .001-.89; p=.04), cough (OR, .08; 95%CI, .01-.88; p=.04), lymphocytosis (OR, .08; 95%CI, .01-.86; p=.04) and QCCT well-aerated lung expressed in liters (OR, .05; 95%CI, .01-1.19; p=.03) were significant predictors of fibrotic-like changes at six-month follow-up with an inverse correlation. Univariable and multivariable logistic regression analysis full results are shown in Table 4.
Table 4:
Univariable and Multivariable Logistic Regression Analysis: The Relationship between Clinical and Radiological Variables in 118 Patients with Moderate to Severe COVID-19 at Six-month Follow-up CT
Diagnostic performance of the three models is reported in Figure 6. AUC was .89 (95%CI .77-.96, sensitivity 82%, specificity 93%) for the clinical model, .81 for the radiological model (95%CI .68-.9, sensitivity 84%, specificity 67%) and .92 for the combined model (95%CI .81-.97, sensitivity 100%, specificity 73%). Even if the clinical model already performed well, combined model was analyzed too, in order to fully investigate possible stronger results with data derived from different fields of investigation.
Figure 6.
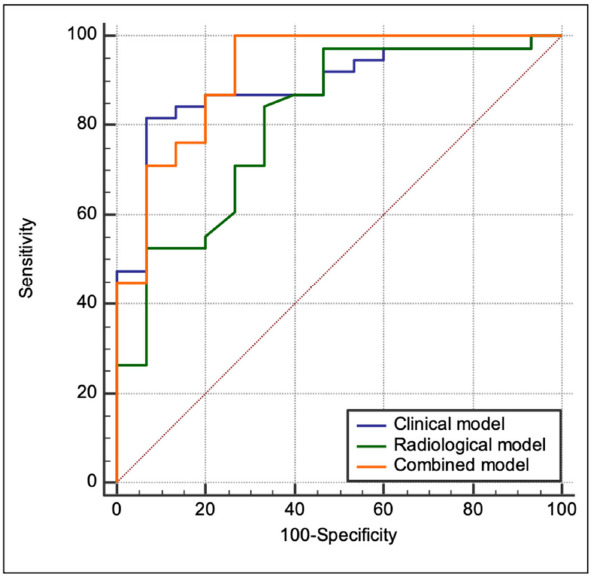
Receiver operating characteristic (ROC) curves tested the performance of clinical (blue line), radiological (green line) and combined model (orange line) in predicting the presence of fibrotic-like changes at six-month follow-up Chest CT. The AUC for the clinical model was .89 (95%CI .77-.96, sensitivity: 82%, specificity: 93%), .81 for radiological model (95%CI .68-.9, sensitivity: 84%, specificity: 67%,) and .92 for combined model (95%CI .81-.97, sensitivity: 100%, specificity: 73%).
Discussion
The results of our study showed that 72% of patients showed fibrotic-like changes at six-month follow-up Chest CT and baseline LSS (>14) was an excellent predictor of fibrotic-like changes (AUC .91). Furthermore, in the multivariable analysis, a model combining clinical and radiological findings reached an excellent diagnostic performance (AUC 0.92). In this model male gender, cough, lymphocytosis and QCCT well-aerated lung expressed in liters showed an inverse correlation with lung fibrotic-like changes. Even if the clinical model already performed well (AUC .89), we decided to analyze also the combined model to fully investigate possible stronger results with data derived from different fields of investigation. The percentage of late sequelae of our population at six-month follow-up Chest CT (72%) is in agreement with the study conducted by Zhao et al. (75%) who found that the most common feature at three-month follow-up was interstitial septal thickening (27 % vs our study 28%) (10). On the other hand, our results differ from those of Tabatabaei et al. who reported residual CT findings in 42% of patients examined at three-month follow-up; among them 55% presented residual GGOs (42%) (9). These different results may be related to the younger age of their population and the lower percentage of patients hospitalized in intensive care unit (ICU), compared to our older population with a higher rate of ventilation support needed during hospitalization. In fact, the older age increased the need for ventilation support and might be related with a more aggressive damage on lung parenchyma, with consequent higher percentage of late sequelae (29). Further studies of long-term effects in COVID-19 patients are mandatory to confirm this hypothesis.
A remarkable six-month follow-up study performed by Han et al. (17) showed approximately one-third (35%) of COVID-19 survivors with pulmonary fibrotic-like changes while 62% of patients presented GGOs. In our population, fibrotic-like changes do not arise de novo, but evolved from GGOs and/or consolidations. Discrepancies with our results might be related with demographics differences such as the prevalence of male and younger patients, even if demographic data on fibrosis development are still lacking and confirmation on Chest CT are needed on larger population sample. In addition, all non-fibrotic-like abnormalities have been reduced at six months, probably because almost all our patients have received steroid therapy during hospitalization.
As a novel disease, it is crucial to perform risk stratification of COVID-19 patients already at baseline stage, in order to identify who will be at high risk to develop residual disease hereafter.
In fact, recent studies have found that semiquantitative Chest CT score is a useful tool to stratify the severity of pneumonia and to predict clinical and radiological outcome in COVID-19 patients at short-term follow-up (9, 10, 30). Our baseline LSS showed an optimal performance in predicting residual findings at follow-up Chest CT (AUC .91); seventy-six patients (64.4%) presented a baseline LSS higher than 14. This result supports a similar analysis conducted at three-month follow-up by Tabatabaei et al. (9) and Zhao et al. (10) who demonstrated that patients with residual lung disease had significantly higher CT severity score compared to the group completely healed. Also the total Chest CT score performed by Han et al. (17) showed correlation in predicting pulmonary fibrotic-like changes at six-month follow-up Chest CT, despite some differences between the two lung score performed.
Likewise, baseline QCCT analysis of well-aerated lung demonstrated a good performance to predict residual findings at follow-up Chest CT, expressed in both liters (AUC .88) and percentage (AUC .88). Our results concerning the role of QCCT well-aerated lung support the previous study conducted by Colombi et al. (27) who showed that low rate of well-aerated lung was a predictor of ICU admission or death, and by Lanza et al. (31) who demonstrated that compromised lung volume was a predictor for oxygen support, need for intubation needed and patient death.
In line with the study conducted at short-term follow-up by Yu et al. (14), we found that older age is a potential predictor of six-month fibrotic-like changes. Moreover, we found that fibrotic-like changes at Chest CT follow-up were more frequent in women. In previous mid-term follow-up studies (9, 10) no significant correlation between gender and follow-up fibrotic-like changes were found (32).
Even if the clinical model already performed well (AUC .89), we decided to analyze also the combined model to fully investigate possible stronger results with data derived from different fields of investigation. Despite the encouraging results, our study had some limitations. First, the relatively small sample size; second, the potential selection bias due to patients' refusal to undergo follow-up CT examinations at our Hospital since already performed in other centers; third, the inter- and intra-reader agreement was not performed.
In conclusion, at six-month follow-up, 72% of patients showed late sequelae, in particular fibrotic-like changes. Baseline LSS and QCCT of well-aerated lung showed an excellent performance in predicting fibrotic-like changes at six-month Chest CT (AUC > .88). Male gender, cough, lymphocytosis and QCCT well-aerated lung (liters) were significant predictors of fibrotic-like changes at six-month with an inverse correlation (AUC .92).
Funding: No founding to declare
Abbreviations:
- SARS-CoV-2
- Severe Acute Respiratory Syndrome Coronavirus 2
- COVID-19
- Coronavirus Disease 2019
- RT-PCR
- Reverse Transcription Polymerase Chain Reaction
- GGO
- Ground Glass Opacity
- LSS
- Lung Severity Score
- QCCT
- Quantitative Chest Computed Tomography
- ROC
- Receiver Operating Characteristic
- AUC
- Area Under the Curve
References
- 1. Naming the coronavirus disease (COVID-19) and the virus that causes it. 2020. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/naming-the-coronavirus-disease-(covid-2019)-and-the-virus-that-causes-it. Published 2020.
- 2. Caruso D, Zerunian M, Polici M, Pucciarelli F, Polidori T, Rucci C, Guido G, Bracci B, De Dominicis C, Laghi A. Chest CT Features of COVID-19 in Rome, Italy. Radiology 2020;296(2):E79–e85. 10.1148/radiol.2020201237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Huang L, Han R, Ai T, Yu P, Kang H, Tao Q, Xia L. Serial quantitative chest ct assessment of covid-19: Deep-learning approach. Radiology: Cardiothoracic Imaging 2020;2(2):e200075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Caruso D, Polici M, Zerunian M, Pucciarelli F, Polidori T, Guido G, Rucci C, Bracci B, Muscogiuri E, De Dominicis C, Laghi A. Quantitative Chest CT analysis in discriminating COVID-19 from non-COVID-19 patients. Radiol Med 2020. 10.1007/s11547-020-01291-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Shen C, Yu N, Cai S, Zhou J, Sheng J, Liu K, Zhou H, Guo Y, Niu G. Quantitative computed tomography analysis for stratifying the severity of Coronavirus Disease 2019. J Pharm Anal 2020;10(2):123–129. 10.1016/j.jpha.2020.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Chung M, Bernheim A, Mei X, Zhang N, Huang M, Zeng X, Cui J, Xu W, Yang Y, Fayad ZA, Jacobi A, Li K, Li S, Shan H. CT Imaging Features of 2019 Novel Coronavirus (2019-nCoV). Radiology 2020;295(1):202–207. 10.1148/radiol.2020200230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Zhan J, Li H, Yu H, Liu X, Zeng X, Peng D, Zhang W. 2019 novel coronavirus (COVID-19) pneumonia: CT manifestations and pattern of evolution in 110 patients in Jiangxi, China. Eur Radiol 2020. 10.1007/s00330-020-07201-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Caruso D, Polidori T, Guido G, Nicolai M, Bracci B, Cremona A, Zerunian M, Polici M, Pucciarelli F, Rucci C, Dominicis C, Girolamo MD, Argento G, Sergi D, Laghi A. Typical and atypical COVID-19 computed tomography findings. World J Clin Cases 2020;8(15):3177–3187. 10.12998/wjcc.v8.i15.3177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Tabatabaei SMH, Rajebi H, Moghaddas F, Ghasemiadl M, Talari H. Chest CT in COVID-19 pneumonia: what are the findings in mid-term follow-up? Emergency Radiology 2020:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Zhao YM, Shang YM, Song WB, Li QQ, Xie H, Xu QF, Jia JL, Li LM, Mao HL, Zhou XM, Luo H, Gao YF, Xu AG. Follow-up study of the pulmonary function and related physiological characteristics of COVID-19 survivors three months after recovery. EClinicalMedicine 2020;25:100463. 10.1016/j.eclinm.2020.100463 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hu Q, Guan H, Sun Z, Huang L, Chen C, Ai T, Pan Y, Xia L. Early CT features and temporal lung changes in COVID-19 pneumonia in Wuhan, China. Eur J Radiol 2020;128:109017. 10.1016/j.ejrad.2020.109017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Huang G, Gong T, Wang G, Wang J, Guo X, Cai E, Li S, Li X, Yu Y, Lin L. Timely Diagnosis and Treatment Shortens the Time to Resolution of Coronavirus Disease (COVID-19) Pneumonia and Lowers the Highest and Last CT Scores From Sequential Chest CT. AJR Am J Roentgenol 2020;215(2):367–373. 10.2214/AJR.20.23078 [DOI] [PubMed] [Google Scholar]
- 13. Liu N, He G, Yang X, Chen J, Wu J, Ma M, Lu W, Li Q, Cheng T, Huang X. Dynamic changes of Chest CT follow-up in Coronavirus Disease-19 (COVID-19) pneumonia: relationship to clinical typing. BMC Med Imaging 2020;20(1):92. 10.1186/s12880-020-00491-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Yu M, Liu Y, Xu D, Zhang R, Lan L, Xu H. Prediction of the Development of Pulmonary Fibrosis Using Serial Thin-Section CT and Clinical Features in Patients Discharged after Treatment for COVID-19 Pneumonia. Korean J Radiol 2020;21(6):746–755. 10.3348/kjr.2020.0215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ojha V, Mani A, Pandey NN, Sharma S, Kumar S. CT in coronavirus disease 2019 (COVID-19): a systematic review of chest CT findings in 4410 adult patients. Eur Radiol 2020;30(11):6129–6138. 10.1007/s00330-020-06975-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Salehi S, Abedi A, Balakrishnan S, Gholamrezanezhad A. Coronavirus Disease 2019 (COVID-19): A Systematic Review of Imaging Findings in 919 Patients. AJR Am J Roentgenol 2020;215(1):87–93. 10.2214/AJR.20.23034 [DOI] [PubMed] [Google Scholar]
- 17. Han X, Fan Y, Alwalid O, Li N, Jia X, Yuan M, Li Y, Cao Y, Gu J, Wu H, Shi H. Six-Month Follow-up Chest CT findings after Severe COVID-19 Pneumonia. Radiology 2021:203153. 10.1148/radiol.2021203153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Wells AU, Devaraj A, Desai SR. Interstitial Lung Disease after COVID-19 Infection: A Catalog of Uncertainties. Radiology 2021;299(1):E216–e218. 10.1148/radiol.2021204482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Salehi S, Reddy S, Gholamrezanezhad A. Long-term Pulmonary Consequences of Coronavirus Disease 2019 (COVID-19): What We Know and What to Expect. J Thorac Imaging 2020;35(4):W87–W89. 10.1097/RTI.0000000000000534 [DOI] [PubMed] [Google Scholar]
- 20. Zhang P, Li J, Liu H, Han N, Ju J, Kou Y, Chen L, Jiang M, Pan F, Zheng Y, Gao Z, Jiang B. Long-term bone and lung consequences associated with hospital-acquired severe acute respiratory syndrome: a 15-year follow-up from a prospective cohort study. Bone Res 2020;8:8. 10.1038/s41413-020-0084-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Wu X, Dong D, Ma D. Thin-Section Computed Tomography Manifestations During Convalescence and Long-Term Follow-Up of Patients with Severe Acute Respiratory Syndrome (SARS). Med Sci Monit 2016;22:2793–2799. 10.12659/msm.896985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Clinical management of severe acute respiratory infection (SARI) when COVID-19 disease is suspected https://www.who.int/docs/default-source/coronaviruse/clinical-management-of-novel-cov.pdf.
- 23. Hansell DM, Goldin JG, King TE, Jr., Lynch DA, Richeldi L, Wells AU. CT staging and monitoring of fibrotic interstitial lung diseases in clinical practice and treatment trials: a position paper from the Fleischner Society. Lancet Respir Med 2015;3(6):483–496. 10.1016/S2213-2600(15)00096-X [DOI] [PubMed] [Google Scholar]
- 24. Hansell DM, Bankier AA, MacMahon H, McLoud TC, Muller NL, Remy J. Fleischner Society: glossary of terms for thoracic imaging. Radiology 2008;246(3):697–722. 10.1148/radiol.2462070712 [DOI] [PubMed] [Google Scholar]
- 25. Yang R, Li X, Liu H, Zhen Y, Zhang X, Xiong Q, Luo Y, Gao C, Zeng W. Chest CT severity score: an imaging tool for assessing severe COVID-19. Radiology: Cardiothoracic Imaging 2020;2(2):e200047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Chen A, Karwoski RA, Gierada DS, Bartholmai BJ, Koo CW. Quantitative CT Analysis of Diffuse Lung Disease. Radiographics 2020;40(1):28–43. 10.1148/rg.2020190099 [DOI] [PubMed] [Google Scholar]
- 27. Colombi D, Bodini FC, Petrini M, Maffi G, Morelli N, Milanese G, Silva M, Sverzellati N, Michieletti E. Well-aerated lung on admitting chest CT to predict adverse outcome in COVID-19 pneumonia. Radiology 2020:201433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Vernuccio F, Giambelluca D, Cannella R, Lombardo FP, Panzuto F, Midiri M, Cabassa P. Radiographic and chest CT imaging presentation and follow-up of COVID-19 pneumonia: a multicenter experience from an endemic area. Emerg Radiol 2020;27(6):623–632. 10.1007/s10140-020-01817-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Chen Y, Klein SL, Garibaldi BT, Li H, Wu C, Osevala NM, Li T, Margolick JB, Pawelec G, Leng SX. Aging in COVID-19: Vulnerability, immunity and intervention. Ageing Res Rev 2021;65:101205. 10.1016/j.arr.2020.101205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lyu P, Liu X, Zhang R, Shi L, Gao J. The Performance of Chest CT in Evaluating the Clinical Severity of COVID-19 Pneumonia: Identifying Critical Cases Based on CT Characteristics. Invest Radiol 2020;55(7):412–421. 10.1097/RLI.0000000000000689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Lanza E, Muglia R, Bolengo I, Santonocito OG, Lisi C, Angelotti G, Morandini P, Savevski V, Politi LS, Balzarini L. Quantitative chest CT analysis in COVID-19 to predict the need for oxygenation support and intubation. Eur Radiol 2020;30(12):6770–6778. 10.1007/s00330-020-07013-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Gebhard C, Regitz-Zagrosek V, Neuhauser HK, Morgan R, Klein SL. Impact of sex and gender on COVID-19 outcomes in Europe. Biol Sex Differ 2020;11(1):29. 10.1186/s13293-020-00304-9 [DOI] [PMC free article] [PubMed] [Google Scholar]