Abstract
Introduction
Treatments of interstitial lung diseases (ILDs) mainly focus on disease stabilization and relief of symptoms by managing inflammation or suppressing fibrosis by (in part costly) drugs. To highlight economic burden of drug treatment in different ILD-subtypes we assessed cost trends and therewith-associated drivers.
Methods
Using data from the German, observational HILDA study we estimated adjusted mean medication costs over 36-month intervals using one- and two-part Generalized Estimating Equation (GEE) regression models with a gamma distribution and log link. Next, we determined factors associated with costs.
Results
In Idiopathic pulmonary fibrosis (IPF) mean per capita medication costs increased from €1442 before to €11,000€ at the end of study. In non-IPF subtypes, the increase took place at much lower level. Mean per capita ILD-specific medication costs at the end of the study ranged between €487 (other ILD) and €9142 (IPF). At baseline, higher FVC %predicted values were associated with lower medication costs in IPF (−9%) and sarcoidosis (−1%). During follow up higher comorbidity burden escalated costs in progressive fibrosing ILD (PF-ILD) (+52%), sarcoidosis (+60%) and other ILDs (+24%). The effect of disease duration was not uniform, with cost savings in PF-ILD (−8%) and sarcoidosis (−6%), but increased spending in IPF (+11%).
Conclusion
Pharmacological management of ILD, in particular of IPF imposes a substantial economic burden on the healthcare system. Strategies to reduce comorbidity burden and early treatment may reduce the impact of ILDs on the healthcare system.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12931-021-01807-8.
Keywords: Diffuse parenchymal lung disease, Healthcare expenditure, Direct costs, ILD management, Healthcare spending
Introduction
The rare group of interstitial Lung Diseases (ILDs) comprises of over 200 subtypes that are heterogeneous regarding etiology, patterns and prognosis, but have similar pathophysiological pathways regarding inflammation and/or fibrosis of the lung parenchyma [1]. ILDs are classified as idiopathic, granulomatous (e.g. sarcoidosis) or associated with known causes [2, 3].
Many ILDs are triggered by environmental, occupational, or medication-related exposures [4] such as e.g. hypersensitivity pneumonitis (HP) [5], and drug-induced ILD [6]. Other ILDs are pulmonary manifestations from systemic autoimmune diseases, [7] such as rheumatic arthritis and connective tissue disease (R-CTD) [4]. Idiopathic ILDs, also referred to as idiopathic interstitial pneumonias (IIPs)—which include idiopathic pulmonary fibrosis (IPF) as their most prominent form—represent an important subset which is associated with a substantial loss of quality of life [8, 9] and with a detrimental survival [10].
Within the various non-IPF ILDs, a proportion of patients can develop a progressive, fibrosing phenotype. This phenotype is characterized by declining lung function and high mortality [11, 12]. In contrast to IPF however, progressive fibrosing ILDs (PF-ILD) are often accompanied by inflammation. Around 20–30% of ILDs may develop into a PF-ILD [12]. ILDs that are associated with this phenotype include sarcoidosis, non-specific interstitial pneumonia (NSIP), CTD-ILD, HP and unclassifiable ILD [11].
ILD treatments mainly focus on disease stabilization and on relief of symptoms by managing inflammation and/or suppressing fibrosis [4]. In this regard, most evidence—particularly on pharmaceutical interventions—is based on treatment of IPF patients with two licensed anti-fibrotic treatments, whereas in non-IPF ILDs evidence-based treatment approaches and thus guidelines are mainly lacking.
Given their mostly chronic, often progressive course combined with the often non-standardized treatment regimens, ILDs might be associated with high healthcare utilization and a significant strain and burden on health services, and healthcare systems [13–15]. Most evidence corresponding to the economic burden of ILDs is however restricted to distinct service providers (e.g. hospital costs) [16], focused on pre-selected ILD subtypes [17, 18] or took a cross-sectional perspective on costs of care [14]. A comprehensive assessment on costs associated with drug treatment that accounts for potentially different trends in various ILDs is lacking so far. In addition, as these chronic diseases are often accompanied by diverse comorbidities [19] an in-depth study to determine the overall cost of drug treatment in ILD patients is crucial.
To close this knowledge gap, our analysis of the HILDA-cohort study portrays the course of medication costs over an 18-months interval and delineates essential cost drivers in pharmacological treatment. The anticipated results are expected to aid German policy makers in planning and allocating healthcare services.
Methods
Study design and population
The German longitudinal HILDA (Health Care in ILD Outpatient Visitors) cohort is a prospective, observational study [9, 20]. Recruitment took place between November 2016 and April 2017 with patient-individual follow-up intervals after 6 months (t1) and 12 months (t2). The study collected information on pharmaceutical treatment retrospectively at each follow up visit. Pharmaceutical data at baseline therefore corresponds to the time before the study began, t1 corresponds to the first 6 months after the recruitment and t2 corresponds to the interval from six to 12 months after recruitment.
Inclusion criteria were an ILD-diagnosis confirmed by the multidisciplinary team board meeting of the recruiting center, a minimum age of 18 years, proficiency in the German language and the provision of written informed consent. In total, 271 patients enrolled into the study.
Medication costs
We calculated drug costs based on information on the Pharmaceutical Central Number of the distinct drug and dosage of drug intake per day during the study period by applying drug-specific prices stemming from the medication database of Scientific Institute of the AOK Statutory Health Insurance funds (WIdO 2016). Apart from nutritional supplements, we considered all pharmaceuticals taken by the patient. Each drug was classified as an ILD-related medication (immunosuppressants, steroids, pirfenidone, nintedanib) or other drug. Subsequently, we calculated medication costs for the three study intervals, 6 months before baseline (t0), baseline to 6 months after baseline (t1) and 6 to 12 months after baseline (t2).
Covariates
We considered the baseline covariates age (in years), sex, duration of disease (in years), smoking status (current or former and never-smoker), study center, comorbidity burden and physician-reported information on forced vital capacity (FVC) % predicted. In addition, we considered four different ILD subtypes: (1) Sarcoidosis, (2) IPF, (3) PF-ILD (determined by a decrease of at least 10% in either FVC %predicted or DLCO %predicted values after 12 months [12]), as well as (4) other ILD subtypes. Other ILD subtypes comprised of idiopathic interstitial pneumonias, hypersensitivity pneumonitis, rheumatic and connective tissue diseases with pulmonary involvement, drug-related ILD, combined pulmonary fibrosis and emphysema, non-classifiable ILD and other forms. A detailed list of this subgroup is available in Additional file 1: Appendix Table S1.
The pre-determined list of comorbidities included pulmonary hypertension, arterial hypertension, coronary heart disease, congestive heart failure, other cardiovascular disease, diabetes mellitus, emphysema/COPD, lung cancer, depression, gastroesophageal reflux disease, renal failure, obstructive sleep apnea, thromboembolism, and malignant tumors excluding lung cancer, which were identified to be of either epidemiological or clinical relevance in ILDs [14]. Physicians had the possibility to include three other comorbidities not present in the list and the highest possible number of comorbidities was hence 17. As the number of patients with each distinct comorbid condition was small, we refrained from a separate analysis of the several conditions. To reflect comorbidity burden we instead calculated a summative index from the documented comorbid conditions which hat a possible range from 0 to 17. A comprehensive list of comorbidities and their frequencies in the sample is illustrated in Additional file 1: Appendix Table S5.
We disregarded information on diffusing capacity of carbon monoxide (DLCO) % predicted in our analyses because this led to multicollinearity problems [12].
Statistical analysis
We disregarded participants with missing information on any variable of interest, which however only applied to disease duration (N = 12) and comorbidity sum score (N = 2). Furthermore, analyses were restricted to complete cases at the distinct assessment points. The respective samples comprised of 257 patients at t0, 229 patients at t1 and 204 patients at t2. We stratified all analyses by ILD subtype and clustered by center to account for correlation between patients from the same treatment center.
To identify structural differences between the distinct ILD subtypes, we compared baseline characteristics by Pearson’s chi-square tests (categorical variables), and Kruskal Wallis tests (continuous variables). Next, we determined the proportion of patients receiving either immunosuppressant medication, steroids or established treatment for IPF at each assessment point and assessed unadjusted mean medication costs.
The outcome of interest was the cost of all medications in ILD patients. The primary analysis estimated covariate-adjusted mean medication costs throughout the study period by ILD subtype. To address intra-subject correlation in context of repeated measures, we applied Generalized Estimating Equation (GEE) regression models with first order auto-regression [21]. To account for the right-skewed distribution of cost data, we assumed a gamma-distribution with log-link in our GEEs. As more than 10% of patients incurred zero costs, we performed two-part GEEs. Two-part models consist of a logistic regression model as part 1 and a gamma model as part 2. The logistic regression model predicts the probability of positive costs, while the gamma model estimates the costs for the subsample with positive costs. The probabilities from part 1 are multiplied by the calculated costs per user from part 2 to determine adjusted per capita costs [22, 23].
In our secondary analyses, we first assessed factors influencing baseline costs using simple gamma models, and subsequently we estimated the factors associated with costs during the study period, using gamma-distributed GEEs. We interpreted the exponentials of the regression coefficients in each of these two analyses as surcharge factors. We adjusted for the same covariates investigated at baseline and additionally accounted for the time point in the GEE models.
As a sensitivity analysis, we re-ran all analyses for those patients who participated at all three assessment points (study completers).
All statistical analyses were performed using the SAS software package (SAS Institute Inc., Cary, NC, USA, version 9.4) and we considered a p-value less than 0.05 statistically significant.
Results
Comparison of patient characteristics in different ILD subgroups at baseline
Table 1 describes the sociodemographic characteristics of the study sample stratified by ILD subtype. Across all ILD subtypes, most patients were male and never smokers. The ILD subtypes differed in terms of disease duration, age and comorbidity burden, with the difference being most pronounced between IPF and sarcoidosis. FVC % and DLCO% predicted differed between the distinct subtypes (p = 0.0016, p < 0.001, respectively), with PF-ILD-patients presenting the highest values at baseline. Sarcoidosis patients had longest disease duration (8.1 ± 10.1 years), were the youngest (52.0 ± 12.1) and presented with the lowest comorbidity burden (2.4 ± 1.3), whereas IPF patients had the shortest disease duration (2.4 ± 1.9 years), were the oldest (71.3 ± 6.5) and presented with the highest comorbidity burden (3.8 ± 1.7). Regarding these baseline characteristics, patients with PF-ILDs were similar to sarcoidosis patients (Table 1).
Table 1.
Differences in patient characteristics stratified by ILD subtypes
| IPF N = 72 | PF-ILD (N = 32) | Sarcoidosis N = 45 | Other ILD N = 122 | p-value | |
|---|---|---|---|---|---|
| Mean FVC % at t0 | 72.9 (21.3) | 83.7 (21.3) | 74.6 (16.9) | 67.8 (20.2) | 0.0016* |
| Mean DLCO % at t0 | 38.9 (12.0) | 57.3 (19.2) | 55.7 (18.0) | 39.0 (13.1) | 0.0010* |
| Mean age, years (SD) | 71.3 (6.5) | 57.9 (13.3) | 52.0 (12.1) | 62.2 (12.4) | < 0.0001* |
| disease duration, years (SD) | 2.4 (1.9) | 5.4 (6.3) | 8.1 (10.1) | 3.1 (4.6) | < 0.0001* |
| Mean number of comorbidities (SD) | 3.8 (1.7) | 2.5 (1.3) | 2.4 (1.3) | 2.8 (1.5) | < 0.0001* |
| Male (%) | 57 (79.2) | 23 (71.2) | 33 (73.3) | 66 (54.1) | 0.0020* |
| Never smoker (%) | 19 (26.4) | 16 (50.0) | 19 (42.2) | 46 (37.7) | 0.0955 |
*p-value < 0.05
Proportion of patients receiving different therapies
The majority of non-IPF patients were administered steroids for the treatment of ILD, 52.5% (N = 135/257), 57.6% (N = 132/229) and 50.0% (N = 102/204) at t0, t1 and t2 respectively. Immunosuppressant medication was used by 33.1% (N = 85) at t0, 28.4% at t1 (N = 65) and 29.9% (N = 61) at t2. A few patients received the combination therapy of steroids and immunosuppressant medication 19.5% (N = 50), 21.4% (N = 49) and 22.1% (N = 45) during the respective time points. At t0 and t1, respectively, about one in five IPF patients received either pirfenidone or nintedanib (t0: N = 15; 20.8%|t1: N = 12; 19.0%), whereas this treatment quota at t2 was 100% (N = 55). Furthermore, a small subset of patients who started on steroid therapy switched to either pirfenidone or nintedanib during the 6-month interval after receiving an official IPF diagnosis, (N = 3 (1.2%), N = 7 (3.1%) and N = 9 (4.4%) at t0, t1 and t2 respectively).
Unadjusted mean medication costs in different ILD subtypes over time
At t0, unadjusted medication costs ranged between €132 (Fibrosing ILD) and €332 (IPF). For sarcoidosis and IPF, costs had multiplied by t1 already and for all subtypes, there was a steep increase of costs between t1 and t2.
Adjusted mean medication costs in different ILD subtypes over time
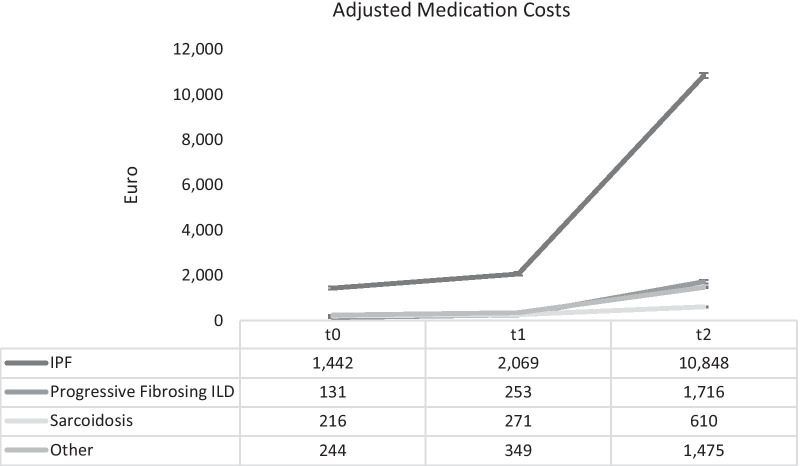
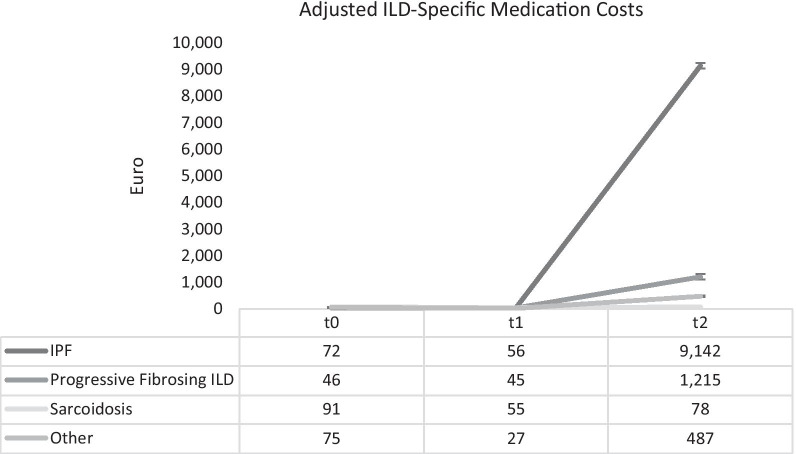
Figures 1 and 2 display the adjusted medication costs for all treatments and ILD-specific treatments respectively. At t0, before study began medication costs for the distinct subtypes varied between about €216 (sarcoidosis) and about €1442 (IPF). During the study, costs for IPF patients increased from ~ €2000 to ~ €11,000. In the non-IPF subtypes, there was also a substantial increase, taking place at much lower level. Spending on ILD-specific medication ranged from €46 (PF-ILD) to €91 (sarcoidosis) before at t0. This amount decreased slightly during the first 6 months of the study but increased again sharply after t1 in all subtypes except sarcoidosis. The ILD-specific medication costs at the end of the study ranged from €487 (other ILD) to €9,142 (IPF).
Fig. 1.
Adjusted mean overall medication costs from baseline to 12 months across different ILD subtypes
Fig. 2.
Adjusted ILD-specific mean medication costs from baseline to 12 months across different ILD subtypes
Factors influencing medication costs at baseline and over time
Table 2 illustrates drivers of baseline costs. Across all ILD subtypes increase of comorbidity burden was associated with a significant increase in medication costs that ranged between + 23% (sarcoidosis) and + 89% (other ILDs). Disease duration was associated with significantly lower expenditures in non-IPF patients. Older age had a cost driving effect in IPF (+ 10%) and PF-ILD (+ 4%) and other ILDs (+ 3%). Smoking was associated with a 247% increase in costs in IPF. Higher FVC % predicted values at baseline were associated with lower costs in IPF and sarcoidosis patients only, with an impact of −9% and −1% respectively.
Table 2.
Influencing baseline factors on medical costs across ILD subtypes
| IPF | PF-ILD | Sarcoidosis | Other ILD | |
|---|---|---|---|---|
| surcharge factor [95% CI] | surcharge factor [95% CI] | surcharge factor [95% CI] | surcharge factor [95% CI] | |
| Male | 0.06 [0.00–0.22] | 1.05 [0.77–1.34] | 0.93 [0.84–1.03] | 1.67 [1.59–1.76]* |
| Former/Current smoker | 2.47 [2.40–2.55]* | 0.41 [0.00–0.83] | 0.91 [0.57—–1.25] | 0.82 [0.54–1.11] |
| Age | 1.10 [1.09–1.10]* | 1.04 [1.03–1.06]* | 1.00 [0.99–1.00] | 1.03 [1.02–1.04]* |
| FVC % pred. At baseline | 0.91 [0.90–0.92]* | 0.99 [0.98–1.00] | 0.99 [0.99–0.99]* | 0.99 [0.99–1.00] |
| Disease duration | 1.02 [0.89–1.15] | 0.90 [0.90–0.91]* | 0.95 [0.93–0.97]* | 0.91 [0.91–0.91]* |
| Comorbidity sum score | 1.59 [1.49–1.69]* | 1.50 [1.41–1.59]* | 1.23 [1.19–1.26]* | 1.89 [1.80–1.97]* |
*p-value < 0.05
Table 3 displays costs drivers of the development of medication costs over the study period. A high comorbidity burden was linked to higher medication costs in PF-ILD (+ 52%), sarcoidosis (+ 60%) and other ILDs (+ 24%). A longer disease duration had a cost-saving impact in PF-ILD (−8%) and sarcoidosis (−6%), and in contrast had a cost-driving effect in IPF (+ 11%). Older age had a cost-driving impact on PF-ILD while smoking had a cost-driving impact in sarcoidosis. Male gender was associated with higher costs in PF-ILD and sarcoidosis. Medication costs at t2 were significantly higher in all subtypes than at t0, while costs at t1 were significantly higher than costs at t0 in PF-ILD only.
Table 3.
Influencing factors on longitudinal medication costs across ILD subtypes
| IPF | PF-ILD | Sarcoidosis | Other ILD | |
|---|---|---|---|---|
| OR [95% CI] | OR [95% CI] | OR [95% CI] | OR [95% CI] | |
| Male | 1.03 [0.65–1.41] | 1.73 [1.27–2.20]* | 2.56 [1.58–3.54]* | 1.02 [0.64–1.40] |
| Former/current smoker | 1.25 [0.92–1.59] | 0.57 [0.13–1.00] | 2.62 [1.79–3.46]* | 1.00 [0.59–1.41] |
| Age | 0.99 [0.97–1.01] | 1.05 [1.03–1.07]* | 1.03 [0.99–1.07] | 0.99 [0.97–1.01] |
| FVC % pred. At baseline | 1.00 [0.99–1.00] | 1.00 [0.99–1.01] | 1.00 [0.97–1.03] | 0.99 [0.98–1.00] |
| Disease duration | 1.11 [1.03–1.19]* | 0.92 [0.88–0.96]* | 0.94 [0.89–0.99]* | 0.93 [0.89–0.97]* |
| Comorbidity sum score | 1.04 [0.96–1.13] | 1.52 [1.31–1.74]* | 1.60 [1.23–1.97]* | 1.24 [1.11–1.38]* |
| T1 | 1.24 [0.87–1.61] | 2.22 [1.72–2.71]* | 0.77 [0.42–1.12] | 1.05 [0.67–1.42] |
| T2 | 5.95 [5.59–6.30]* | 11.8 [11.3–12.3]* | 2.09 [1.64–2.54]* | 6.46 [6.05–6.86]* |
*p-value < 0.05
Sensitivity analysis
The results for the influencing factors on baseline costs and cost development for complete cases are illustrated in Additional file 1: Appendix Tables S2 and S3. For the most part, the results were similar to the main analyses. When comparing cost drivers at baseline, we found that the direction of the effect of male sex and age was reversed in IPF patients. When observing medication costs throughout the study, the direction of the effects of all covariates were the same as in the main analyses.
Discussion
In this study, we illustrated the pharmaceutical treatment costs in patients diagnosed with various ILDs and determined the associated cost drivers in Germany. Our results showed that IPF patients had the highest medication costs of all ILD groups within the study period, which rose sharply after baseline. The reason beyond this observation is the high cost of the two approved anti-fibrotic medications for IPF, pirfenidone and nintedanib [2]. Indeed 12 months after baseline, when all IPF patients had received either pirfenidone or nintedanib, corresponding expenditures explained almost 85% of total medication costs. The steep rise in medication costs suggests that there was a rollout of these two medications until all eligible IPF patients were on these medications by the second half of the study, as shown in our results.
For non-IPF ILDs, treatment mainly relies on relatively inexpensive steroids and immunosuppressant medication, as illustrated by our results. Nevertheless, we observed a general increase in total medication costs in non-IPF patients during the study, especially in PF-ILD patients. This suggests intensified pharmaceutical treatment of comorbidities, mainly in the first six months of the study period, as depicted by the corresponding reduction of ILD-specific medication costs at t1. Regardless, 12 months after baseline, ILD-related costs had increased again, indicating intensified pharmaceutical treatment of ILD in non-IPF patients. The steep increase in medication costs in PF-ILD patients was due to three patients that were administered pirfenidone or nintedanib in t2 for compassionate use. It is to note however, that at the time of the HILDA study, these medications were not yet approved for the treatment of PF-ILD in Germany as nintedanib was approved in 2020 [24]. In future, we assume that the cost of medication in this subgroup of patients will increase and mirror IPF, considering that the costs of anti-fibrotic medication can presumably not be modified except if the patency is removed and generic medication becomes available. Although we did not evaluate cost of entire healthcare resource utilization in our study, other researchers have demonstrated that both IPF and PF-ILD patients are hospitalized more often than patients without a fibrosing phenotype [18, 25, 26], and therefore incur higher costs. For this reason, we expect anti-fibrotic medication will moderate the clinical course of the disease and, if necessary, cost-intensive hospitalizations will be reduced. Therefore, the increase in medication costs should not necessarily be viewed critically.
Cost drivers
Comorbidity burden was a cost-driving factor in all subtypes either at baseline or throughout the study period. In addition to pharmaceutical management of the index disease, a sensitive pharmaceutical management of comorbidities is required in treatment of ILD. In IPF, a study found that comorbidities influence the clinical course of the disease and survival [27]. Accordingly, a systematic review on cost triggers in IPF identified comorbidities as a substantial factor in the increasing cost of IPF management [15] and similar findings have also been established in sarcoidosis [14].
Lower FVC % predicted values were a determinant for higher medication costs in IPF and sarcoidosis. Studies have found that declining values of FVC % predicted are linked to disease progression [28]. Patients with worsening symptoms of the disease may require higher doses or higher frequencies of pharmaceutical treatment. Moreover, sarcoidosis patients with normal lung function values do not require pharmaceutical treatment [7, 29] and this explains the cost-saving effect of higher lung function values.
In our study, a longer disease duration was associated with higher medication costs in IPF but lower medication costs in non-IPF subtypes. With anti-fibrotic treatment one only achieves slowing down the inevitable lung function decline in IPF [30, 31]. Thus, most patients need continuous medication over the entire course of their disease to alleviate symptoms and reduce lung deterioration [32]. In contrast in PF-ILDs it the rapid lung function decline itself that renders drug treatment necessary. Here anti-fibrotic treatment represents a timely-defined intervention to stabilize further lung and to avoid its further deterioration [33]. After this goal has been achieved medication can in some cases be dispensable. In the case of sarcoidosis, this disease may be acute or chronic. Some patients with acute sarcoidosis may recover spontaneously [34]. Chronic sarcoidosis, however, has a subtle onset and is slowly progressing. Thus, in many patients with chronic sarcoidosis, treatment is not required [7, 29]. However, as patients within the HILDA registry were recruited in expert centers, a significant higher treatment indication has to be expected.
Age was a cost-driver in both IPF and PF-ILD. Age has been identified as a risk factor for ILD progression [35]. Furthermore, older age is particularly known in to increase the likelihood of progression and mortality in progressive fibrosing phenotypes [12].
Male sex was associated with higher costs in PF-ILD and sarcoidosis. Risk prediction in ILD can be determined by the Gender, Age and Physiology (GAP) index model, which places men at a higher risk of mortality than women [35]. Increased mortality indicates worse outcomes for men than women, which would require more pharmaceutical treatment and therefore incur higher costs.
We demonstrated that current or former smokers had higher costs than never-smokers in IPF at baseline and in sarcoidosis patients during the study. Smoking is linked to increased inflammation in the lung, which may lead to worse outcomes [36]. Over time, however, smoking status had no cost-driving effect in IPF patients. This could be due to the mechanism of pirfenidone and nintedanib that were accessible to all IPF patients in the study. The drugs have been shown to relieve inflammation [30, 31].
Sensitivity analysis
Factors influencing medication costs were similar in both the main and sensitivity analyses. The differences found in IPF can be explained by dropout analysis (Additional file 1: Appendix Table S4). The dropouts were significantly older and male. The remaining sample favored healthier, younger patients. Male sex therefore had a cost-saving impact in the sensitivity analysis although usually, IPF is a disease that mostly affects men, and being male is also a risk factor for the regression of disease [12].
Strengths and limitations
So far, data on the pharmacological management of ILD in Germany are sparse. One strength of our study is that we simultaneously analyzed different ILD subtypes and compared their burden on the healthcare system as opposed to the previous study conducted in Germany that only included two subtypes of ILD and did not include clinical parameters [14]. Moreover, we were able to assess cost drivers for the most common ILD subtypes. As our study was over a longer period, we could also make concrete deductions on factors influencing medication costs.
There are several limitations to our study. An important limitation is that we did not adjust for lung transplantations, as the study did not collect the information. Several studies have found that lung transplantation and associated medications is one of the most costly component of ILD management [25]. However, generally, lung transplantations are rarely performed [37] and only a third of all lung transplantations are due to ILD [38]. Another limitation is the heterogeneous group of "other ILDs". We are not able to make concrete deductions on this subgroup due to a large variance. In addition, owing to the study design we were not able to differentiate between pulmonary sarcoidosis and systemic sarcoidosis. Hence, obtained costs reflect costs for a heterogeneous sample of sarcoidosis patients, within which those with systemic manifestations presumably incur higher costs than those with pulmonary manifestations only. Lastly, the summative consideration of comorbidity burden does not allow any conclusions to be drawn as to which combinations are particularly costly, therefore—if sample sizes allow—an analysis of individual comorbid conditions could shed further light on this important issue.
Conclusion
The results of our study suggest that the pharmacological management of ILD, in particular that of IPF imposes a substantial economic burden on the healthcare system in Germany. Strategies to reduce comorbidity burden and lung function decline could essentially reduce the burden of all ILD on both caregivers and the healthcare system.
Supplementary Information
Additional file 1: Table S1. List of ‘Other’ ILD subtypes. Table S2. Influencing factors on medical costs at baseline across ILD subtypes in complete cases. Table S3. Influencing factors on longitudinal medication costs across ILD subtypes in complete cases. Table S4. Characteristics of the complete and incomplete cases. Table S5. Frequency of comorbid conditions present at baseline by ILD subtype.
Acknowledgements
The authors would like to acknowledge our study nurses Britta Markworth and Beate Schaufler for their assistance in recruiting subjects for this study, and Boglárka Szentes for her contribution in designing the study.
Abbreviations
- CPFE
Combined pulmonary fibrosis with emphysema
- CTD-ILD
Connective tissue diseases
- DLCO
Diffusing capacity of carbon monoxide
- FVC
Forced vital capacity
- GEE
Generalized Estimating Equations
- GLM
Generalized Linear Models
- HILDA
Health Care in ILD Ambulance Visitors
- HP
Hypersensitivity pneumonitis
- HRQL
Health-related quality of life
- IIPs
Idiopathic interstitial pneumonias
- ILD
Interstitial lung diseases
- IPF
Idiopathic pulmonary fibrosis
- NSIP
Non-specific interstitial pneumonia
- PF-ILD
Progressive fibrosing ILD
- R-CTD
Rheumatic and connective tissue diseases with pulmonary involvement
- WIdO
Scientific Institute of the AOK
Authors' contributions
MK, MC and LS initiated the project. PM and LS conceived and designed the analyses. PM performed the statistical analyses and drafted the manuscript. LS and RH aided in statistical interpretation of the data while MK, MC, TB and NK contributed to the clinical interpretation of the data. All authors read and approved the manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. This research did not receive any particular funding. It was conducted under the unrestricted institutional grants of the German Center for Lung Research (DZL).
Availability of data and materials
The data that support the findings of this study are available on reasonable request. The data are not publicly available due to their containing information that could compromise the privacy of research participants.
Ethics approval and consent to participate
The local Ethics Committees of Heidelberg and Luebeck approved the study (Reference Numbers S-200/2013, and AZ: 16-192, respectively). Patients provided written, informed consent.
Competing interests
All other authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Disayabutr S, Calfee CS, Collard HR, Wolters PJ. Interstitial lung diseases in the hospitalized patient. BMC Med. 2015;13:245. doi: 10.1186/s12916-015-0487-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jakubczyc A. Neurohr C [diagnosis and treatment of interstitial lung diseases] Dtsch Med Wochenschr. 2018;143:1774–1777. doi: 10.1055/a-0622-9299. [DOI] [PubMed] [Google Scholar]
- 3.Ryerson CJ, Collard HR. Update on the diagnosis and classification of ILD. Curr Opin Pulm Med. 2013;19:453–459. doi: 10.1097/MCP.0b013e328363f48d. [DOI] [PubMed] [Google Scholar]
- 4.Kalchiem-Dekel O, Galvin J, Burke A, Atamas S, Todd N. Interstitial lung disease and pulmonary fibrosis: a practical approach for general medicine physicians with focus on the medical history. J Clin Med. 2018;7:476. doi: 10.3390/jcm7120476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Spagnolo P, Rossi G, Cavazza A, Paladini I, Paladini I, Bonella F, et al. Hypersensitivity pneumonitis: a comprehensive review. J Investig Allergol Clin Immunol. 2015;25:237–50. [PubMed] [Google Scholar]
- 6.Schwaiblmair M, Behr W, Haeckel T, Märkl B, Foerg W, Berghaus T. Drug induced interstitial lung disease. Open Respir Med J. 2012;6:63–74. doi: 10.2174/1874306401206010063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Raghu G, Wilson KC, Bargagli E, Bendstrup E, Chami HA, Chua AT, et al. Diagnosis of hypersensitivity pneumonitis in adults: an official ATS/JRS/ALAT clinical practice guideline. Am J Respir Crit Care Med. 2020 doi: 10.1164/rccm.202005-2032ST. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kreuter M, Swigris J, Pittrow D, Geier S, Klotsche J, Prasse A, et al. Health related quality of life in patients with idiopathic pulmonary fibrosis in clinical practice: insights-IPF registry. Respir Res. 2017;18:139. doi: 10.1186/s12931-017-0621-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Maqhuzu PN, Szentes BL, Kreuter M, Bahmer T, Kahn N, Claussen M, et al. Determinants of health-related quality of life decline in interstitial lung disease. Health Qual Life Outcomes. 2020;18:334. doi: 10.1186/s12955-020-01570-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wells AU, Brown KK, Flaherty KR, Kolb M, Thannickal VJ. What’s in a name? That which we call IPF, by any other name would act the same. Eur Respir J. 2018;51:1–12. doi: 10.1183/13993003.00692-2018. [DOI] [PubMed] [Google Scholar]
- 11.Kolb M, Vašáková M. The natural history of progressive fibrosing interstitial lung diseases. Respir Res. 2019;20:57. doi: 10.1186/s12931-019-1022-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wong AW, Ryerson CJ, Guler SA. Progression of fibrosing interstitial lung disease. Respir Res. 2020 doi: 10.1186/s12931-020-1296-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Holtze C, Flaherty K, Kreuter M, Luppi F, Moua T, Vancheri C, et al. Healthcare utilisation and costs in the diagnosis and treatment of progressive-fibrosing interstitial lung diseases. Eur Respir Rev. 2018;27:180078. doi: 10.1183/16000617.0078-2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Frank AL, Kreuter M, Schwarzkopf L. Economic burden of incident interstitial lung disease (ILD) and the impact of comorbidity on costs of care. Respir Med. 2019;152:25–31. doi: 10.1016/j.rmed.2019.04.009. [DOI] [PubMed] [Google Scholar]
- 15.Vaidya S, Hibbert CL, Kinter E, Boes S. Identification of key cost generating events for idiopathic pulmonary fibrosis: a systematic review. Lung. 2017 doi: 10.1007/s00408-016-9960-6. [DOI] [PubMed] [Google Scholar]
- 16.Baughman RP, Field S, Costabel U, Crystal RG, Culver DA, Drent M, et al. Sarcoidosis in America: analysis based on health care use. Ann Am Thorac Soc. 2016;13:1244–1252. doi: 10.1513/AnnalsATS.201511-760OC. [DOI] [PubMed] [Google Scholar]
- 17.Gade Sikjær M, Hilberg O, Ibsen R, Bock K, Løkke A. Direct and indirect economic and health consequences related to sarcoidosis in Denmark: a national register-based study. Respir Med Elsevier. 2019;152:7–13. doi: 10.1016/j.rmed.2019.04.004. [DOI] [PubMed] [Google Scholar]
- 18.Collard HR, Ward AJ, Lanes S, Cortney Hayflinger D, Rosenberg DM, Hunsche E. Burden of illness in idiopathic pulmonary fibrosis. J Med Econ. 2012;15:829–835. doi: 10.3111/13696998.2012.680553. [DOI] [PubMed] [Google Scholar]
- 19.Margaritopoulos GA, Antoniou KM, Wells AU. Comorbidities in interstitial lung diseases. Eur Respir Rev. 2017;26:160027. doi: 10.1183/16000617.0027-2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Szentes BL, Kreuter M, Bahmer T, Birring SS, Claussen M, Waelscher J, et al. Quality of life assessment in interstitial lung diseases:a comparison of the disease-specific K-BILD with the generic EQ-5D-5L. Respir Res. 2018;19:101. doi: 10.1186/s12931-018-0808-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Laird NM, Ware JH. Random-effects models for longitudinal data. Biometrics. 1982;38:963. doi: 10.2307/2529876. [DOI] [PubMed] [Google Scholar]
- 22.Buntin MB, Zaslavsky AM. Too much ado about two-part models and transformation? Comparing methods of modeling Medicare expenditures. J Health Econ North-Holland. 2004;23:525–542. doi: 10.1016/j.jhealeco.2003.10.005. [DOI] [PubMed] [Google Scholar]
- 23.Afifi AA, Kotlerman JB, Ettner SL, Cowan M. Methods for improving regression analysis for skewed continuous or counted responses. Annu Rev Public Health. 2007;28:95–111. doi: 10.1146/annurev.publhealth.28.082206.094100. [DOI] [PubMed] [Google Scholar]
- 24.Wells AU, Flaherty KR, Brown KK, Inoue Y, Devaraj A, Richeldi L, et al. Nintedanib in patients with progressive fibrosing interstitial lung diseases—subgroup analyses by interstitial lung disease diagnosis in the INBUILD trial: a randomised, double-blind, placebo-controlled, parallel-group trial. Lancet Respir Med. 2020;8:453–460. doi: 10.1016/S2213-2600(20)30036-9. [DOI] [PubMed] [Google Scholar]
- 25.Morell F, Esser D, Lim J, Stowasser S, Villacampa A, Nieves D, et al. Treatment patterns, resource use and costs of idiopathic pulmonary fibrosis in Spain—results of a Delphi Panel. BMC Pulm Med. 2016;16:7. doi: 10.1186/s12890-016-0168-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Olson AL, Maher TM, Acciai V, Mounir B, Quaresma M, Zouad-Lejour L, et al. Healthcare resources utilization and costs of patients with non-IPF progressive fibrosing interstitial lung disease based on insurance claims in the USA. Adv Ther Adis. 2020;37:3292–3298. doi: 10.6084/m9.figshare.12249695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kreuter M, Ehlers-Tenenbaum S, Palmowski K, Bruhwyler J, Oltmanns U, Muley T, et al. Impact of comorbidities on mortality in patients with idiopathic pulmonary fibrosis. PLoS One. 2016;11:e0151425. doi: 10.1371/journal.pone.0151425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ley B, Collard HR, King TE. Clinical course and prediction of survival in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2011 doi: 10.1164/rccm.201006-0894CI. [DOI] [PubMed] [Google Scholar]
- 29.Bargagli E, Prasse A. Sarcoidosis: a review for the internist. Intern. Emerg. Med. Springer-Verlag Italia s.r.l.; 2018. p. 325–31. [DOI] [PubMed]
- 30.Richeldi L, Cottin V, du Bois RM, Selman M, Kimura T, Bailes Z, et al. Nintedanib in patients with idiopathic pulmonary fibrosis: Combined evidence from the TOMORROW and INPULSIS® trials. Respir Med. 2016;113:74–9. doi: 10.1016/j.rmed.2016.02.001. [DOI] [PubMed] [Google Scholar]
- 31.King TE, Bradford WZ, Castro-Bernardini S, Fagan EA, Glaspole I, Glassberg MK, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med. 2014;370:2083–92. doi: 10.1056/NEJMoa1402582. [DOI] [PubMed] [Google Scholar]
- 32.Somogyi V, Chaudhuri N, Torrisi SE, Kahn N, Müller V, Kreuter M. The therapy of idiopathic pulmonary fibrosis: what is next? Eur. Respir. Rev. 2019;28(153):190021. doi: 10.1183/16000617.0021-2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.George PM, Spagnolo P, Kreuter M, Altinisik G, Bonifazi M, Martinez FJ, et al. Progressive fibrosing interstitial lung disease: clinical uncertainties, consensus recommendations, and research priorities. Lancet Respir Med. 2020;8:925–34. doi: 10.1016/S2213-2600(20)30355-6. [DOI] [PubMed] [Google Scholar]
- 34.Caruana LB, Redwine GD, Rohde RE, Russian CJ. A prospective study of patients diagnosed with sarcoidosis: factors–environmental exposure, health assessment, and genetic outlooks. Sarcoidosis, Vasc Diffus lung Dis Off J WASOG. Mattioli 1885; 2019;36:228–42. [DOI] [PMC free article] [PubMed]
- 35.Ryerson CJ, Vittinghoff E, Ley B, Lee JS, Mooney JJ, Jones KD, et al. Predicting survival across chronic interstitial lung disease. Chest. 2014;145:723–728. doi: 10.1378/chest.13-1474. [DOI] [PubMed] [Google Scholar]
- 36.Margaritopoulos GA, Vasarmidi E, Jacob J, Wells AU, Antoniou KM. Smoking and interstitial lung diseases. Eur Respir Rev. 2015;23:428–35. doi: 10.1183/16000617.0050-2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Aigner C. Current developments in lung transplantation. Pathologe. 2019 doi: 10.1007/s00292-019-00690-x. [DOI] [PubMed] [Google Scholar]
- 38.Vogl M, Warnecke G, Haverich A, Gottlieb J, Welte T, Hatz R, et al. Lung transplantation in the spotlight: reasons for high-cost procedures. J Hear Lung Transplant. 2016;35:1227–1236. doi: 10.1016/j.healun.2016.05.024. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. List of ‘Other’ ILD subtypes. Table S2. Influencing factors on medical costs at baseline across ILD subtypes in complete cases. Table S3. Influencing factors on longitudinal medication costs across ILD subtypes in complete cases. Table S4. Characteristics of the complete and incomplete cases. Table S5. Frequency of comorbid conditions present at baseline by ILD subtype.
Data Availability Statement
The data that support the findings of this study are available on reasonable request. The data are not publicly available due to their containing information that could compromise the privacy of research participants.