Abstract
Wnt signaling is an ancient and evolutionarily conserved pathway with fundamental roles in the development of adipose tissues. Roles of this pathway in mesenchymal stem cell fate determination and differentiation have been extensively studied. Indeed, canonical Wnt signaling is a significant endogenous inhibitor of adipogenesis and promoter of other cell fates, including osteogenesis, chondrogenesis, and myogenesis. However, emerging genetic evidence in both humans and mice suggests central roles for Wnt signaling in body fat distribution, obesity, and metabolic dysfunction. Herein, we highlight recent studies that have begun to unravel the contributions of various Wnt pathway members to critical adipocyte functions, including carbohydrate and lipid metabolism. We further explore compelling evidence of complex and coordinated interactions between adipocytes and other cell types within adipose tissues, including stromal, immune, and endothelial cells. Given the evolutionary conservation and ubiquitous cellular distribution of this pathway, uncovering the contributions of Wnt signaling to cell metabolism has exciting implications for therapeutic intervention in widespread pathologic states, including obesity, diabetes, and cancers.
Introduction
Historically, adipose tissue was considered to be an inert organ primarily responsible for storage of excess energy. Ability to store nutrients for mobilization during periods when demands exceed caloric intake is an evolutionarily preserved function; indeed, energy storage as cytoplasmic triacylglycerol is conserved from Saccharomyces cerevisiae to Drosophila melanogaster to Homo sapiens (1). With the discovery of leptin (2), an adipocyte-derived hormone that acts in the brain to regulate energy balance, a global effort was initiated to explore the molecular underpinnings of adipocyte development and physiology. We now know that adipose tissues have important roles within their particular niches and throughout the body, not only in whole-body energy homeostasis, but also in thermoregulation, mechanical support, and secretion of adipokines to regulate insulin sensitivity, metabolism, and immune responses (3,4) (Fig. 1).
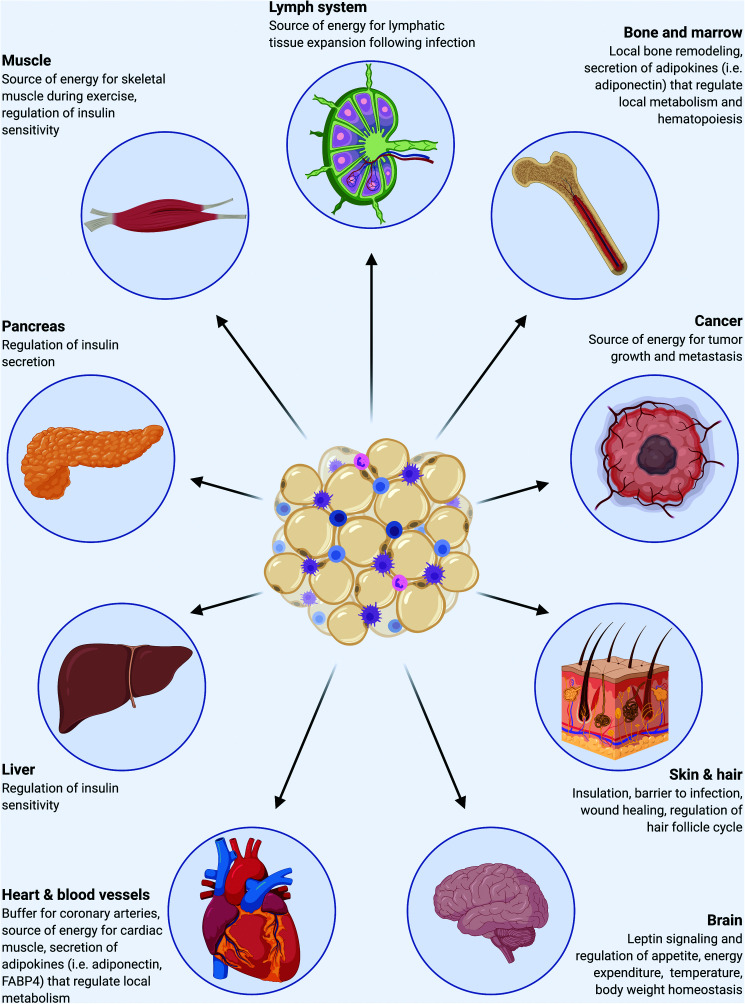
Figure 1.
Adipose tissues interact with myriad organs to maintain global homeostasis. Adipocytes are located in discrete depots and niches throughout the body and have fundamental and complex roles in storage and release of energy in response to local and global needs, thermoregulation, mechanical support, and secretion of adipokines to regulate energy balance, metabolism, and immune responses.
Wnt signaling is an ancient pathway used by multicellular animals from the earliest metazoans to mice to humans, with central roles in development and maintenance of diverse cell types and tissues (5). Mammalian Wnt signaling is complex: 19 Wnts, 10 frizzled receptors, and two Lrp coreceptors on the cell surface are accompanied by an intricate network of intracellular components and innumerable downstream molecular responses. To date, studies have established the Wnt/β-catenin pathway as a critical endogenous inhibitor of adipocyte differentiation (6). In addition, compelling evidence has recently emerged linking this pathway to the function of mature adipocytes, particularly under obesogenic conditions. Herein, we highlight key contributions of Wnt signaling pathway members to adipose tissue development and metabolism and share perspectives on avenues worthy of future inquiry.
A Brief Overview of Wnt Signaling
The first mammalian Wnt gene, originally named integration 1 (Int1) and now known as Wnt1, was discovered in 1982 as a gene activated by integration of mouse mammary tumor virus proviral DNA into mammary carcinomas (7). During this time, independent studies in Drosophila larval development identified the wingless (Wg) gene as a pivotal regulator of segmental polarity and wing formation. In 1987, Int1 and Wg were found to share homology, and the Wnt nomenclature was derived from the combination of both gene names to yield a consistent classification system across species (8). Since then, interest in Wnt signaling and its fundamental functions in organismal biology has grown steadily. In 1991, mutation in the Apc gene, which encodes adenomatous polyposis coli (APC), was identified as the genetic cause of familial adenomatous polyposis, a hereditary cancer syndrome. Shortly thereafter, APC was found to interact directly with the central Wnt signaling protein, β-catenin, providing a direct connection between Wnt signaling and human disease (9). To date, aberrant Wnt signaling has been implicated in diverse pathologic processes, including development disorders, cancers, and degenerative diseases (10,11).
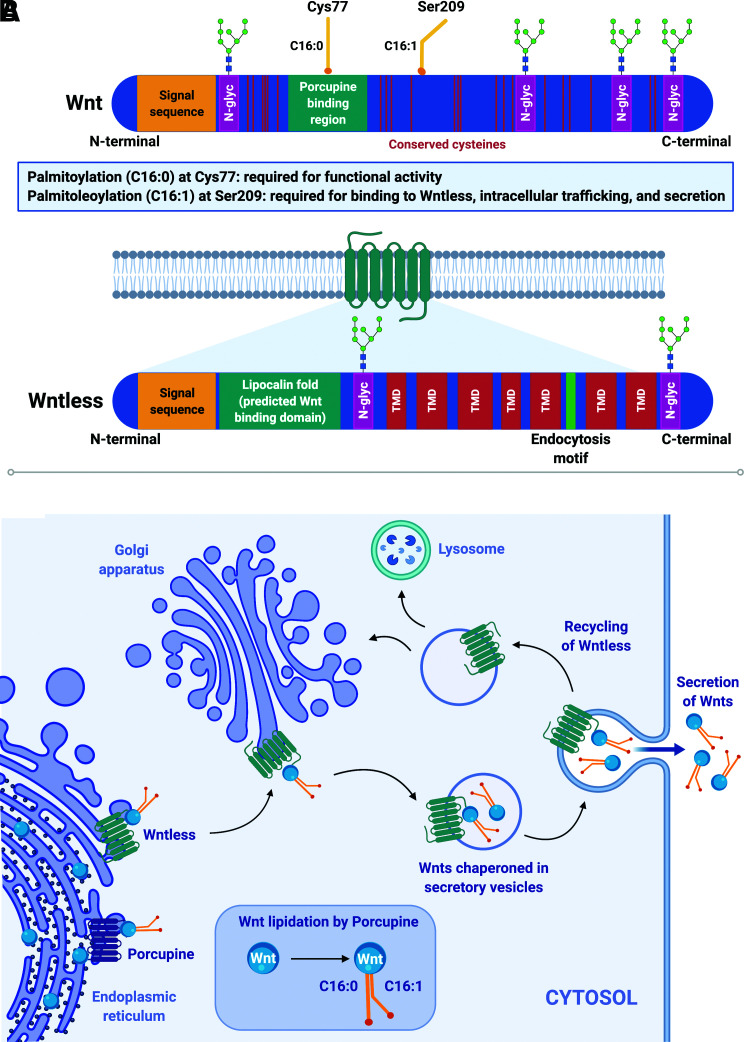
Wnt ligands are evolutionarily conserved, secreted, lipid-modified glycoproteins with well-established and diverse roles in cell proliferation, fate determination, and differentiation. Analyses of primary amino acid sequences reveal that Wnts share a conserved pattern of 23 cysteine residues and an N-terminal signal sequence for endoplasmic reticulum (ER) targeting and subsequent secretion. During synthesis, Wnt proteins, which are ∼30–40 kDa in size, undergo extensive posttranslational processing, including N-glycosylation, palmitoylation, and palmitoleoylation, to ensure proper secretion and function (12) (Fig. 2A). Whereas the function of N-glycosylation remains unclear, lipid modifications are critically coupled to proper secretion and function of Wnts: Ser209 acylation is required for intracellular targeting and secretion, whereas Cys77 acylation is required for extracellular signaling (13) (Fig. 2B).
Figure 2.
Wnts play fundamental and diverse roles in tissue development and maintenance. A: Wnts are lipid-modified secreted glycoproteins (∼30–40 kDa) characterized by a signal peptide sequence, 23 conserved cysteine residues, varying numbers of N-glycosylation sites (N-glyc), and two conserved lipid modifications: palmitic acid (C16:0) at Cys77 and palmitoleic acid (C16:1) at Ser209. Palmitoleoylation (C16:1) at Ser209 is required for the interaction between Wnts and their dedicated chaperone protein, Wntless; palmitoylation (C16:0) at Cys77 is required for functionality of secreted Wnts. Wntless is an evolutionarily conserved transmembrane protein required for intracellular trafficking and secretion of lipidated Wnts. Wntless (∼62 kDa) is predicted to have seven transmembrane domains (TMD), a signal peptide sequence, an endocytosis motif, and a hydrophobic lipocalin domain thought to be the site of interaction with Wnts. B: After Wnt proteins are synthesized in the ER, they undergo significant posttranslational modifications, including N-glycosylation and lipidation. The ER acyltransferase Porcupine catalyzes addition of palmitic (C16:0) and palmitoleic (C16:1) acid moieties at conserved Cys77 and Ser209 residues, respectively. Wntless binds to and chaperones Wnt proteins from the ER through the trans-Golgi network and to the plasma membrane in secretory vesicles. Once Wnts are secreted, Wntless is transported back via a retromer complex to the Golgi for reuse or to lysosomes for degradation.
Once secreted, hydrophobic Wnt proteins have limited abilities to diffuse in the extracellular environment and thus act mainly through autocrine/paracrine mechanisms. Several pathways can be activated by binding of Wnts to their receptors, including the canonical Wnt/β-catenin, noncanonical Wnt/calcium, and planar cell polarity pathways. Of these, the best studied is the canonical pathway, which regulates activation of Wnt target genes through stabilization of the central signaling protein β-catenin (14) (Fig. 3A).
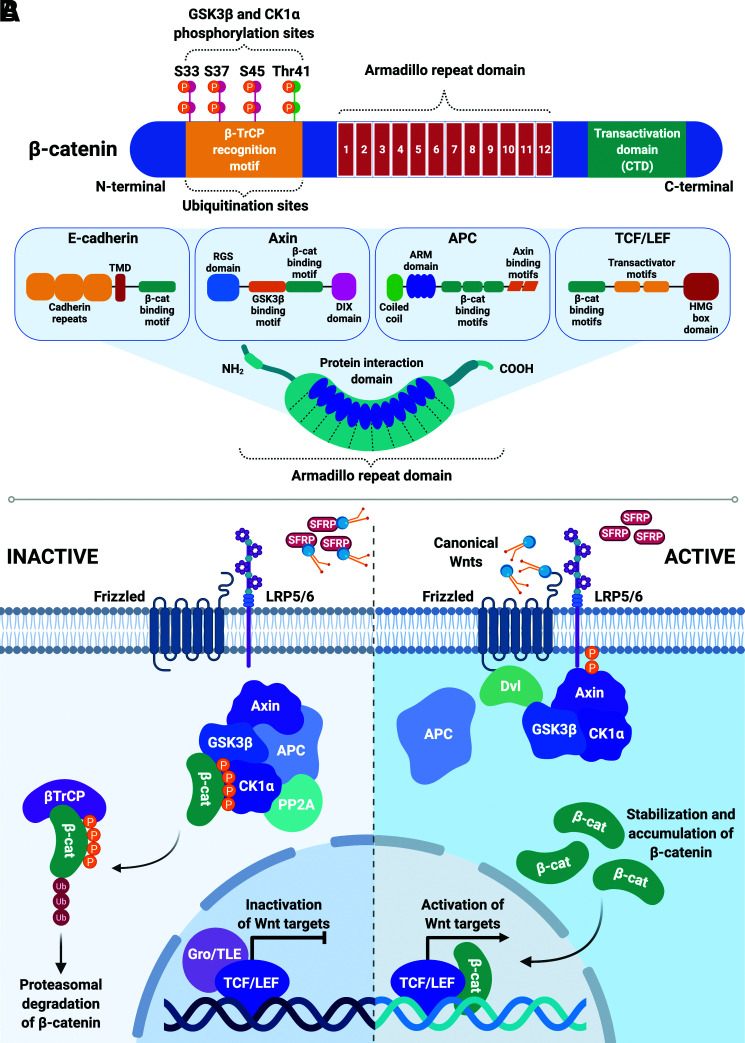
Figure 3.
β-catenin is the central player in the canonical Wnt pathway. A: β-catenin is comprised of a central 12-unit Armadillo repeat domain (ARM) flanked on either side by distinct N-terminal domain and C-terminal domain (CTD). The N-terminal region consists of conserved serine (S33, S47, S45) and threonine (Thr41) residues that are sequentially phosphorylated by CK1α and GSK3β; phosphorylation of these sites promotes binding to β-TrCP, subsequent ubiquitination, and proteasomal degradation. The ARM region forms a superhelix featuring a positively charged groove that serves as a platform for interactions with various β-catenin binding partners, including E-cadherin, Axin, APC, and TCF/LEF proteins. These partners share overlapping binding sites in the ARM groove and thus typically cannot bind simultaneously. DIX domain, Dishevelled/Axin domain; HMG box domain: high-mobility group box domain; RGS domain: regulator of G-protein signaling domain; TMD, transmembrane domain. B: In the absence of extracellular Wnts (inactive [left panel]), β-catenin is bound by a destruction complex comprised of Axin, APC, GSK3β, CK1α, and PP2A. CK1α and GSK3β sequentially phosphorylate (P) free cytosolic β-catenin, targeting it for ubiquitination (Ub) by β-TrCP and subsequent proteasomal degradation. When canonical Wnts bind to their frizzled receptors and LRP coreceptors (active [right panel]), they trigger an intracellular signaling cascade that leads to hypophosphorylation, stabilization, and accumulation of cytosolic β-catenin. β-catenin can then translocate to the nucleus, displace Groucho/TLE repressors, and coactivate TCF/LEF transcription factors to mediate Wnt target gene expression.
The majority of endogenous intracellular β-catenin is bound to E-cadherin and α-catenin at adherens junctions, whereas a small pool exists as free cytosolic β-catenin. Association of β-catenin with E-cadherin protects it from binding to a multiunit destruction complex comprised of Axin, APC, protein phosphatase 2A (PP2A), protein kinases casein kinase 1α (CK1α) and glycogen synthase 3β (GSK3β), and the E3 ligase β-TrCP. In the absence of Wnt signaling, this destruction complex facilitates CK1α- and GSK3β-mediated phosphorylation of N-terminal serine and threonine residues on free cytosolic β-catenin, thereby promoting its binding to and ubiquitination by β-TrCP (15). Phosphorylated and ubiquitinated β-catenin is subsequently targeted for proteasomal degradation to prevent activation of Wnt target genes (Fig. 3B). The half-life of free cytosolic β-catenin is on the order of minutes, whereas the pool associated with adherens junctions turns over slowly; thus, free cytosolic β-catenin is the Wnt-responsive signaling pool.
When Wnts bind to their membrane-spanning frizzled (Fzd) receptors and LDL receptor–related protein (LRP) coreceptors, they trigger an intracellular signaling cascade that disrupts the destruction complex and prevents phosphorylation of free cytosolic β-catenin, thereby promoting its stabilization. Accumulated β-catenin then translocates to the nucleus to associate with DNA-binding TCF/LEF proteins, which act as transcriptional switches (16). In the absence of Wnt signals, TCF/LEF factors form complexes with groucho/transducin-like enhancer of split (TLE) to act as transcriptional repressors. Binding of β-catenin physically displaces groucho/TLE, forming bipartite TCF/β-catenin coactivators that then mediate transcription of downstream Wnt target genes (Fig. 3B).
Endogenous Canonical Wnt Signaling Inhibits Adipogenesis and Promotes Osteoblastogenesis
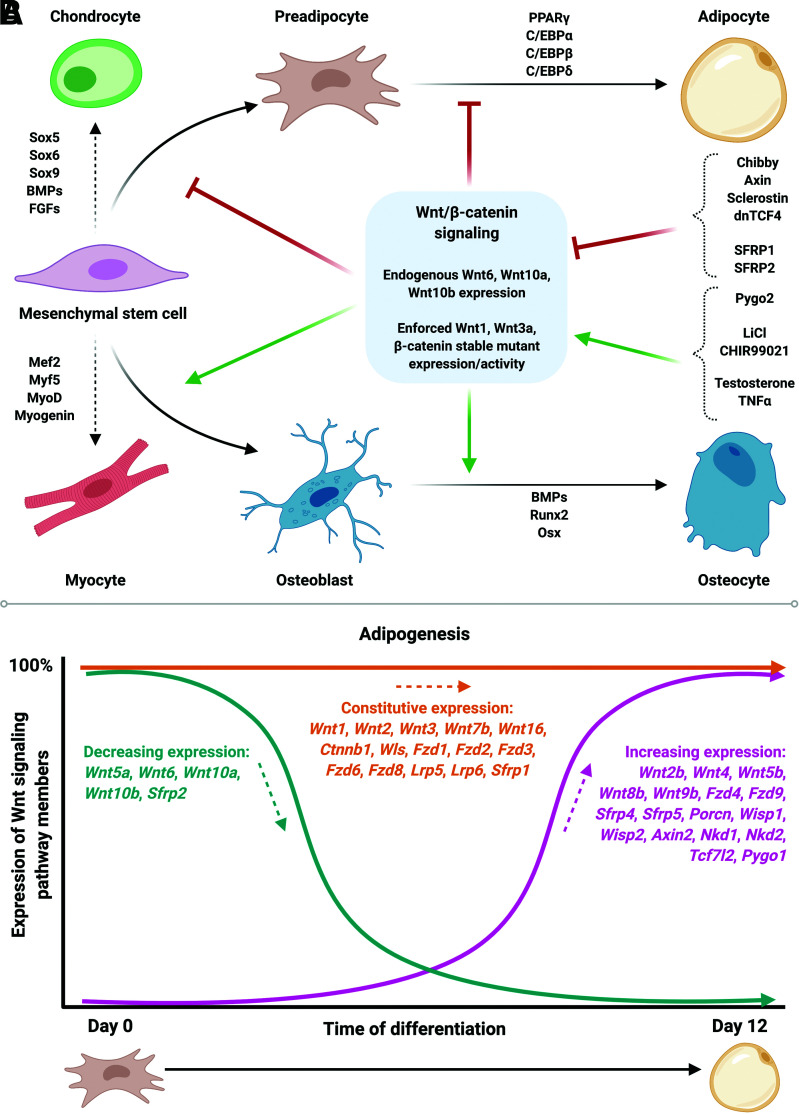
Mesenchymal stem cells (MSCs) arising from the mesoderm are multipotent and have the ability to differentiate into adipocytes, osteocytes, chondrocytes, or myocytes (17); adipogenesis is the process by which MSCs differentiate into mature lipid-laden adipocytes. Wnt signaling through β-catenin has profound effects on determination of MSC fate (6). Although this pathway is also involved in myogenesis and chondrogenesis, its differential regulatory roles in adipogenesis versus osteogenesis have been particularly well characterized (18) (Fig. 4A). Indeed, stabilization of β-catenin by endogenous expression of canonical Wnt6, Wnt10a, or Wnt10b, or enforced expression of Wnt1, Wnt3a, or a β-catenin stable mutant, in multipotent progenitor cells inhibits adipogenesis and promotes osteoblastogenesis (19).
Figure 4.
Canonical Wnt signaling regulates MSC fate determination and differentiation. A: Activation of canonical Wnt/β-catenin signaling in MSCs suppresses adipogenesis and promotes osteoblastogenesis. Wnt signaling suppresses adipocyte differentiation by inhibiting expression of PPARγ and C/EBPα, the central regulators of adipogenesis. B: The complexities of Wnt signaling are evident during adipogenesis: although canonical Wnt signaling inhibits adipocyte differentiation, many Wnts, frizzled receptors, and central regulatory components of the pathway are constitutively expressed or induced in mature mouse adipocytes derived from MSCs. Thus, the canonical Wnt signaling machinery is also highly operative in terminally differentiated adipocytes.
Further, activation of canonical Wnt signaling in committed preadipocytes by enforced expression of Wnt1, Wnt6, Wnt10a, Wnt10b, or a dominant-stable form of β-catenin, or pharmacological inhibition of GSK3 with lithium or CHIR99021, blocks adipogenesis by suppressing induction of key transcription factors PPARγ and C/EBPα (6,20). Similarly, overexpression in preadipocytes of Pygo2, a nuclear promoter of β-catenin/TCF/LEF transcription, mediates β-catenin signaling to inhibit adipogenesis, whereas Pygo2 deletion induces spontaneous differentiation (21). Although β-catenin is often considered to function exclusively as a canonical Wnt effector, it can also signal through unrelated pathways in certain contexts (6). In response to testosterone, for example, β-catenin binds to the androgen receptor and is subsequently shuttled to the nucleus to coactivate TCF/LEFs and inhibit adipocyte differentiation (22). Additionally, tumor necrosis factor α (TNFα) treatment blocks differentiation of preadipocytes by stabilizing β-catenin and enhancing TCF7L2-dependent transcription, suggesting that β-catenin mediates effects of inflammatory cytokines on adipogenesis (23).
Conversely, inhibition of Wnt signaling in precursor cells promotes adipogenic differentiation. Indeed, treatment of preadipocytes with soluble Wnt inhibitors such as secreted frizzled-related protein 1 (SFRP1) or SFRP2, or overexpression of negative pathway regulators, such as axin, dickkopf 1, or dominant-negative TCF4, results in spontaneous differentiation into adipocytes (24). Enforced expression of chibby, a nuclear β-catenin antagonist, induces spontaneous adipogenesis by binding to β-catenin and blocking activation of TCF/LEF, thus repressing β-catenin–mediated transcription (25). In contrast, loss of chibby upregulates β-catenin transcriptional activity and blocks differentiation. Further, enforced expression of PPARγ coactivator TLE3 in preadipocytes displaces β-catenin and directly represses TCF7L2 to induce expression of adipogenic genes during differentiation (26). In contrast, suppression of TLE3 results in reduced PPARγ target gene expression and impaired adipogenesis.
In mice, conditional deletion of β-catenin in uterine mesenchymal cells impairs smooth muscle myogenesis and stimulates conversion of these progenitors into adipocytes (27). This report provided the first in vivo evidence linking Wnt signaling and MSC fate determination. Since then, studies have shown that adipose-specific overexpression of canonical Wnt10b results in dramatically reduced white adipose tissue (WAT) mass and resistance to genetic and diet-induced obesity, absence of brown adipose tissue development, and increased trabecular bone thickness and strength, whereas loss of Wnt10b causes reduction of trabecular bone mass (28). Additionally, deletion of Wnt signaling activator Pygo2 in progenitor cells promotes accumulation of visceral WAT, glucose intolerance, and insulin insensitivity in diet-induced obese mice (21).
A robust literature has also emerged to highlight the role of canonical Wnt/β-catenin signaling in bone development and homeostasis in vivo (29,30). Interest in Wnt signaling within bone was first sparked by identification of LRP5 loss-of-function mutations in human patients diagnosed with osteoporosis pseudoglioma (31). Shortly thereafter, LRP5 gain-of-function mutations were found to be associated with high bone mass in humans (32). Since then, mutations in other Wnt signaling proteins, including LRP4, LRP6, LGR4, WNT1, WNT16, SOST, CTNNB1, and WLS, have also been associated with altered bone mass in humans (33).
These human data have been supported by many mouse models with altered Wnt signaling showing effects on bone development and function (33). Indeed, deletion of either Wntless (Wls) or Ctnnb1 in progenitor cells blocks osteoblast commitment and differentiation, resulting in severe osteopenia, whereas overexpression of β-catenin promotes bone formation. Osteoblast-specific Wls knockout mice are characterized by severe impairment in bone formation, dramatic reductions in both cortical and trabecular bone mass, increased fracture risk, and premature lethality. Further, Ctnnb1 deletion in osteoblasts results in severe osteopenia due to increased osteoclastogenesis; in contrast, constitutive activation of β-catenin in osteoblasts impairs formation of osteoclasts. Indeed, FABP4- and OCN-Wnt10b mice exhibit increased osteoblastogenesis and extensive trabeculation throughout the entire endocortical compartment, whereas Wnt10b−/− mice are characterized by decreased trabecular bone volume and bone mineral density (34).
Finally, investigations into the role of sclerostin, a glycoprotein secreted predominantly by osteocytes, have opened a new avenue of inquiry into endocrine-mediated communication between WAT and bone. Sclerostin, encoded by Sost, interacts with LRP5 and LRP6 coreceptors, thereby preventing downstream intracellular propagation of Wnt signaling. In humans, elevated serum sclerostin levels are associated with increased BMI, adiposity, fasting glucose concentrations, and insulin resistance. Global deletion of Sost in mice profoundly influences both WAT and bone: Sost−/− mice exhibit dramatically increased bone formation, decreased adiposity, and improved insulin sensitivity, whereas overproduction of sclerostin causes adipocyte hypertrophy and insulin insensitivity (35). Taken together, these studies and many others illustrate the fundamental roles of Wnt signaling in directing MSC fate determination and the development of adipose tissues and bone.
Noncanonical Wnt Signaling Has Proposed Roles in Adipocyte Function
To date, the complex and detailed mechanisms underlying noncanonical Wnt pathways, which encompass Wnt/calcium and Wnt/planar cell polarity signaling, remain elusive (36). In planar cell polarity signaling, Wnt activation results in the downstream activation of c-Jun N-terminal kinase (JNK) and regulation of cytoskeletal organization. In the Wnt/calcium pathway, binding of Wnts to Fzd receptors activates a heterotrimeric G-protein–mediated signaling cascade involving phospholipase C that ultimately leads to elevated intracellular calcium. The Wnt/calcium pathway has proposed roles in ventral patterning, cell adhesion and migration, and inhibition of Wnt/β-catenin signaling. Indeed, Wnt5a and Wnt5b have been reported to promote adipogenesis through upregulation of PPARγ and FABP4 (37). Adenoviral overexpression of Wnt5b in 3T3-L1 preadipocytes results in impaired translocation of β-catenin to the nucleus and increased adipogenic differentiation (38). However, further studies are required to more fully understand the contribution of noncanonical pathways to adipose tissue development and metabolism.
Wnt Signaling Is Associated With Metabolic Disease in Humans
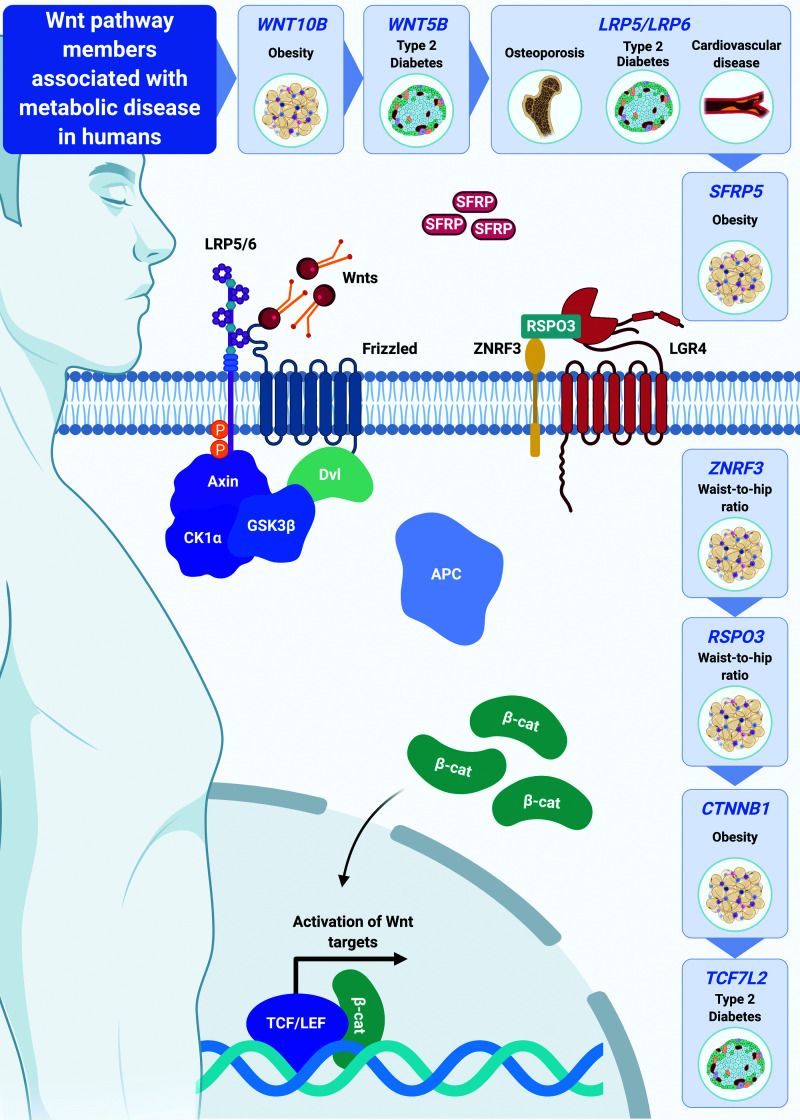
Whereas the critical role of Wnt signaling in regulation of adipogenesis and osteoblastogenesis has been well studied, its function in terminally differentiated adipocytes is less clear. However, increasing genetic evidence in humans has begun to link various Wnt signaling pathway members to body fat distribution, obesity, and metabolic dysfunction (Fig. 5). For example, missense WNT10B variants are associated with increased risk of obesity (39), and although only observed in smaller cohorts, polymorphisms in Wnt5b and the Wnt inhibitor SFRP5 locus are linked to type 2 diabetes and decreased adiposity in men, respectively (40,41). Further, common variants in Wnt signaling inhibitor ZNRF3 and Wnt signaling activator RSPO3 are associated with increased waist-to-hip ratio and insulin resistance (42–45). Indeed, these variants are also associated with altered ZNRF3 and RSPO3 expression in adipose tissues (46). Patients harboring loss-of-function mutations in Wnt coreceptors LRP5 and LRP6 are predisposed to development of impaired glucose homeostasis, coronary disease, and osteoporosis (47,48), whereas gain-of-function LRP5 mutations are correlated with altered body fat distribution, increased adiposity, and osteosclerosis (49). Nonsense mutations in LGR4, which encodes a protein that stabilizes Wnt receptors, are associated with reduced adiposity and impaired bone formation and remodeling (50); in contrast, gain-of-function mutations in LGR4 are correlated with increased visceral adiposity (51). Strikingly, genome-wide association studies across a broad range of human populations have found that common genetic variation at the TCF7L2 gene locus is the strongest genetic determinant of type 2 diabetes risk (52). Finally, rare gain-of-function mutations in CTNNB1 (β-catenin) have recently been linked to altered body fat distribution and increased obesity risk (53), as have common single nucleotide polymorphisms (https://t2d.hugeamp.org). Together, these studies provide compelling genetic evidence for critical roles of Wnt/β-catenin signaling in regulation of adiposity, body composition, and metabolic health.
Figure 5.
Wnt signaling is associated with metabolic disease in humans. Emerging genetic evidence in humans links specific members of the Wnt signaling pathway to body fat distribution, obesity, cardiovascular disease, and type 2 diabetes.
Genetic Studies in Mice Reveal Various Functional Roles of Wnt Signaling in Mature Adipocyte Function and Systemic Metabolism
Current research using novel cultured cell and mouse models is focused on uncovering the underlying mechanisms by which Wnt pathway members contribute to adipocyte and systemic metabolism. Of note, several Wnts and their frizzled receptors are induced during adipogenesis, including Wnt2b, Wnt4, Wnt5b, Wnt8b, Wnt9b, Fzd4, and Fzd9. Further, many central regulatory components of the signaling pathway are constitutively expressed or induced in mature mouse adipocytes, including Wnt2, Wnt7b, Wnt16, Ctnnb1, Lrp5, Lrp6, Axin2, Wisp1, Wisp2, and Tcf7l2. Thus, the canonical Wnt signaling machinery is highly operative in terminally differentiated adipocytes (54) (Fig. 4B). Perhaps unsurprisingly, given the breadth and complexity of the pathway and differences in experimental models and approaches, genetic studies in mice have found that specific Wnt pathway members have unique and distinct contributions to adipocyte function, including regulation of carbohydrate and lipid metabolism.
Endogenous Wnt Inhibitors
Given the pivotal role of Wnt signaling in tissue development and maintenance, it is perhaps unsurprising that this pathway is tightly regulated by a number of evolutionarily conserved modulators. Secreted Wnt inhibitors include SFRPs, dickkopfs, sclerostin, and Wnt inhibitory factor 1 (Wif1); of these, SFRPs are the most well studied in adipose tissues. Due to their structural similarity to frizzled receptors, SFRPs bind to and sequester extracellular Wnt proteins, thus blocking binding and downstream signaling. Of the five SFRP family members, SFRP1 and SFRP5 are upregulated during adipogenesis. Further, high Sfrp5 expression is a strong predictor of increased adiposity in C57BL/6J mice fed a high-fat diet (HFD). Consistent with this, SFRP5 expression is highly upregulated specifically in adipocytes by diet-induced obesity. Global deletion of Sfrp5 in mice stabilizes Wnt signaling and confers resistance to genetic or diet-induced obesity (55). Adipocytes of Sfrp5 mutant mice are smaller in size secondary to upregulated mitochondrial biogenesis and function; thus, knockout mice exhibit reduced WAT mass and improved glucose handling and insulin sensitivity. Consistent with these genetic models, treatment of obese mice with an anti-SFRP5 monoclonal antibody results in lower fasting blood glucose and improved glucose tolerance (56). In contrast, adeno-associated viral (AAV) overexpression of SFRP5 predisposes mice to increased adiposity, hyperglycemia, and insulin resistance with HFD feeding.
Circulating sclerostin, which is primarily secreted by osteocytes, binds to LRP5 and LRP6 coreceptors, thus inhibiting downstream propagation of Wnt signaling. Chow-fed mice with global Sost deletion exhibit reduced total WAT mass, smaller adipocyte size, improved glucose tolerance and insulin sensitivity, and increased bone mass (35). Further, Sost−/− mice are protected against diet-induced obesity, WAT inflammation, and metabolic dysfunction, in part due to elevated fatty acid oxidation within WAT depots. HFD-fed wild-type mice treated with sclerostin-neutralizing antibody accumulate significantly less WAT mass and hepatosteatosis and have improved glucose handling compared with control counterparts. Indeed, adipocytes of antibody-treated mice maintained on chow diet or HFD are smaller in size and exhibit increased fatty acid oxidation. In contrast, AAV-mediated sclerostin overexpression in mice results in suppression of Wnt signaling markers and increased WAT mass, adipocyte hypertrophy, and impaired glucose tolerance. Increased adiposity may be secondary to upregulation of adipocyte differentiation and lipid synthesis, as indicated by increased expression of adipogenic and lipogenic genes including Pparg, Cebpa, Fabp4, Acaca, Fasn, and Mogat1.
These studies indicate that sclerostin has profound Wnt-mediated endocrine effects that influence adipose tissue and whole-body metabolism. Within bone, sclerostin interacts directly with LRP4 to exert its effects; until recently, whether LRP4 plays a role within adipose tissues was unknown. However, a recent study found that mice harboring adipocyte-specific Lrp4 deletion have elevated Wnt signaling within WAT depots, greater number of adipocytes that are smaller in size, and improved glucose handling and insulin sensitivity, without any effect on bone mass (57). Thus, sclerostin may inhibit Wnt signaling in WAT in part through interactions with LRP4. Together, these models using loss of Wnt inhibition as a surrogate for gain of Wnt function suggest that Wnt signaling in adipose tissues inhibits the accumulation of adiposity and systemic metabolic dysfunction.
Adipocyte-Derived Wnt Proteins
Although it is clear that Wnt signaling is active within adipose tissues, an outstanding question has been the cellular source of physiologically relevant Wnts. Previous studies have shown that ablation of Wntless (Wls) in various vertebrate and invertebrate animal models and tissues results in phenotypes consistent with loss of Wnt function, supporting the notion that Wls regulates signaling at the level of Wnt transport and secretion in signal-producing cells. Thus, adipocyte-specific deletion of Wls has facilitated investigation of the functional roles of both canonical and noncanonical Wnts secreted from adipocytes.
Wls is an evolutionarily conserved, multipass, transmembrane protein dedicated to intracellular trafficking of Wnts to the plasma membrane for secretion (58) (Fig. 2A). After Wnts are produced, the covalent addition of acyl moieties at the ER renders Wnt proteins hydrophobic and limits their mobility through the secretory pathway. Wls is localized throughout the Wnt secretory route, including the ER, Golgi apparatus, intracellular vesicles, and plasma membrane. Thus, Wls binds modified Wnts at the ER and Golgi apparatus and facilitates their movement throughout the cell to the plasma membrane for extracellular release (Fig. 2B). Importantly, palmitoleoylation at conserved Ser209 on Wnt proteins is thought to be required for binding to Wls; mutants lacking this lipid modification are unable to interact with Wls and are thus retained at the ER.
Adipocyte-specific deletion of Wls results in downregulation of Wnt target genes and coordinated repression of a network of lipogenic genes, including Srebf1, Mlxipl, Acly, Acaca, Fasn, and Scd1 (59). Consistent with this, Wls−/− adipocytes have impaired de novo lipogenesis and lipid monounsaturation. Thus, endogenous Wnt signaling within adipocytes appears to be an important regulator of palmitoleic acid formation. Interestingly, the reciprocal relationship has also been shown in the cancer field: Wnt secretion and subsequent functionality depend on lipid modification with SCD1-derived palmitoleic acid (60). These findings suggest the existence of Wnt-mediated cross talk between adipose tissues and cancers.
Importantly, Wls−/− mice maintained on chow diet do not exhibit an overt phenotype. Further investigation reveals that this is due to a compensatory mechanism by which surrounding stromal vascular cells sense the loss of adipocyte-derived Wnt secretion and respond by upregulating various Wnt mRNAs. Thus, stromal vascular cells of Wls−/− mice have elevated Wnt target gene expression compared with those of control mice, and Wnt signaling is maintained within Wls-deficient adipocytes. Intriguingly, this compensatory mechanism is overcome by HFD feeding, revealing that Wls−/− mice have decreased WAT mass, smaller adipocytes, and improved glucose tolerance and insulin sensitivity (59).
β-Catenin: The Central Player in Canonical Wnt Signaling
Canonical Wnts signal through the central protein β-catenin to mediate downstream target gene expression in widespread tissues (Fig. 3). Recently, genome-wide association studies in humans have linked CTNNB1 gain-of-function mutations to increased obesity risk (53). Further, two independent investigations targeted adipocyte-specific deletion of β-catenin in mice to uncover the contribution of canonical Wnt signaling to mature adipocyte function (53,54). These studies revealed that β-catenin deletion suppresses expression of canonical Wnt target and lipogenic genes, in part through regulation of key transcription factors Srebf1 and Mlxipl. Thus, similar to Wls-deficient adipocytes, β-cat−/− cells are characterized by impaired fatty acid desaturation and lipogenesis.
In vivo, β-catenin deletion does not appear to affect systemic metabolism in chow-fed mice. However, Bagchi et al. (54) found sustained expression of β-catenin mRNA and protein within adipocytes despite complete ablation at the genomic level. Consistent with this, Chen et al. (53) also reported substantial expression of Ctnnb1 mRNA in adipocytes isolated from WAT of knockout mice. Similar to observations in chow-fed Wls knockout mice, loss of adipocyte β-catenin appears to be sensed and compensated for by surrounding CD45−/CD31− stromal cell populations. Further, consistent with Ctnnb1 expression patterns, downstream Wnt targets are also increased in this cell population. Thus, these data suggest that a subset of CD45−/CD31− stromal cells are able to sense and respond, directly or indirectly, to the loss of adipocyte β-catenin, such that chow-fed mice maintain normal metabolic function.
Interestingly, HFD feeding overrides this compensatory mechanism and β-catenin expression is ablated in adipocytes isolated from knockout mice. Both Chen et al. and Bagchi et al. reported remarkably consistent data showing that β-catenin knockout mice challenged with HFD are resistant to diet-induced obesity. Indeed, β-cat−/− mice have decreased weight gain and adiposity, improved glucose handling and insulin sensitivity, and protection from hepatosteatosis.
Wnt Target Gene Tcf7l2
Wnt/β-catenin mediates downstream gene expression primarily through coactivation of TCF/LEF transcription factors. To date, TCF/LEF family member Tcf7l2 is one of the strongest genetic risk factors associated with development of type 2 diabetes in humans. Several groups have recently sought to explore mechanisms by which Tcf7l2 regulates metabolism by generating three distinct models of adipocyte-specific Tcf7l2 ablation (61–63). Chen et al. (62) ablated exon 11, which is common to all splice variants, and found that mice with adipocyte-specific deletion of Tcf7l2 exhibit subcutaneous adipocyte hypertrophy, glucose intolerance, and insulin resistance when fed either chow diet or HFD. Geoghegan et al. (61) excised exon 5 of Tcf7l2 and found that whereas knockout mice challenged with HFD demonstrate increased adiposity, subcutaneous adipocyte hypertrophy, impaired glucose handling, and greater insulin resistance, mice maintained on chow diet do not exhibit an overt meta-bolic phenotype. Interestingly, Geoghegan et al. also observed elevated expression of a subset of lipogenic genes, including Acly and Scd1, and decreased levels of lipolytic genes in WAT of knockout mice. Finally, Nguyen-Tu et al. (63) generated a model in which exon 1 is excised and found that loss of adipocyte Tcf7l2 leads to impaired glucose tolerance in male but not female mice fed chow diet or HFD. Possible bases for differences in results include varied genetic strategies or alternative gene splicing. However, the overarching results suggest that Tcf7l2 regulates adipocyte hypertrophy and systemic metabolism.
Discussion and Future Perspectives
The role of Wnt signaling in MSC fate determination and differentiation is well established (6). Indeed, the preponderance of data within the field of adipose biology to date has characterized Wnt inhibition of adipogenesis and promotion of other cell fates, including osteoblastogenesis, chondrogenesis, and myogenesis. However, increasing genetic evidence in humans has linked various Wnt pathway members to body fat distribution, obesity, and metabolic dysfunction. In recent years, a flurry of global and adipose-targeted studies in mice has provided compelling, albeit in some cases contradictory, evidence that Wnt signaling plays important roles in accumulation of adipose tissues and in whole-body glucose and lipid metabolism, particularly under obesogenic conditions (53–55,59,61–63). Indeed, adipocyte-specific deletion of various Wnt pathway members, including Ctnnb1, Wls, and Tcf7l2, has been shown to influence lipogenic gene expression, underscoring the critical importance of Wnt signaling in adipocyte lipogenesis.
These studies also point toward Wnt signaling being required for other adipocyte functions. Indeed, gene set enrichment analyses of differentially expressed genes have identified several metabolic pathways as being downregulated with β-catenin deletion, including those involved in glycolysis, oxidative phosphorylation, and fatty acid, cholesterol, and bile acid metabolism (54). Further, RNA-sequencing analysis revealed that Tcf7l2 regulates many metabolic pathways in WAT, including cholesterol, fatty acyl-CoA and triacylglycerol biosynthesis, and amino acid, carbohydrate, and peroxisomal lipid metabolism (61). Thus, it is conceivable that various Wnt signaling members influence unique networks of metabolic pathways in adipocytes. Indeed, interaction between effects on distinct pathways may explain, in part, differences observed between reported phenotypes of impaired Wnt signaling in adipocytes. These works also reveal that intercellular Wnt signaling forms a nuanced and flexible system for monitoring and regulating the state of adipose tissue metabolism and other functions. Thus, disruption of the Wnt pathway is compensated for on a tissue level, and it is only when these compensatory mechanisms are overridden that effects on global metabolism are observed.
In addition to their fundamental contributions to whole-body homeostasis and widespread physiologic processes (Fig. 1), adipose tissues also influence growth and progression of many malignancies (64). Tumors require large sources of energy to facilitate rapid cell division, which may explain, in part, why many invasive cancers occur in close proximity to adipose depots. Indeed, a distinctive feature of oncogenic transformation is metabolic reprogramming, with increased production and use of monounsaturated fatty acids. Upregulation of SCD1 has been observed in many cancers and is correlated with increased aggression and worsened outcomes (65). Interestingly, Wnt/β-catenin signaling in mature adipocytes regulates a wide network of lipogenic genes, including Fasn and Scd1, thus promoting de novo lipogenesis and production of monounsaturated fatty acids. One intriguing possibility is that Wnt-mediated fatty acid production in adipocytes contributes to cancer pathogenesis. Indeed, aberrant upregulation of Wnt signaling is tightly associated with many malignancies, including colorectal, pancreatic, breast, skin, and blood cancers (66). Thus, further studies exploring the contribution of adipocyte Wnt signaling and fatty acid production to cancer pathogenesis may uncover novel therapeutic targets to limit tumor progression and improve patient outcomes.
Article Information
Funding. This study was funded by the National Institutes of Health (AG069795, DK092759, DK121759, DK125513, DK62876, GM007863, HD007505).
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
References
- 1. Kadereit B, Kumar P, Wang WJ, et al. Evolutionarily conserved gene family important for fat storage. Proc Natl Acad Sci USA 2008;105:94–99 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature 1994;372:425–432 [DOI] [PubMed] [Google Scholar]
- 3. Bagchi DP, Forss I, Mandrup S, MacDougald OA. SnapShot: niche determines adipocyte character I. Cell Metab 2018;27:264–264.e1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Rosen ED, Spiegelman BM. What we talk about when we talk about fat. Cell 2014;156:20–44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Loh KM, van Amerongen R, Nusse R. Generating cellular diversity and spatial form: Wnt signaling and the evolution of multicellular animals. Dev Cell 2016;38:643–655 [DOI] [PubMed] [Google Scholar]
- 6. Prestwich TC, Macdougald OA. Wnt/beta-catenin signaling in adipogenesis and metabolism. Curr Opin Cell Biol 2007;19:612–617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Nusse R, Varmus HE. Many tumors induced by the mouse mammary tumor virus contain a provirus integrated in the same region of the host genome. Cell 1982;31:99–109 [DOI] [PubMed] [Google Scholar]
- 8. Cadigan KM, Nusse R. Wnt signaling: a common theme in animal development. Genes Dev 1997;11:3286–3305 [DOI] [PubMed] [Google Scholar]
- 9. Rubinfeld B, Souza B, Albert I, et al. Association of the APC gene product with beta-catenin. Science 1993;262:1731–1734 [DOI] [PubMed] [Google Scholar]
- 10. Clevers H, Nusse R. Wnt/β-catenin signaling and disease. Cell 2012;149:1192–1205 [DOI] [PubMed] [Google Scholar]
- 11. Polakis P. The many ways of Wnt in cancer. Curr Opin Genet Dev 2007;17:45–51 [DOI] [PubMed] [Google Scholar]
- 12. Janda CY, Garcia KC. Wnt acylation and its functional implication in Wnt signalling regulation. Biochem Soc Trans 2015;43:211–216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Tang X, Wu Y, Belenkaya TY, et al. Roles of N-glycosylation and lipidation in Wg secretion and signaling. Dev Biol 2012;364:32–41 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 2009;17:9–26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Valenta T, Hausmann G, Basler K. The many faces and functions of β-catenin. EMBO J 2012;31:2714–2736 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Willert K, Jones KA. Wnt signaling: is the party in the nucleus? Genes Dev 2006;20:1394–1404 [DOI] [PubMed] [Google Scholar]
- 17. Andrzejewska A, Lukomska B, Janowski M. Concise review: mesenchymal stem cells: from roots to boost. Stem Cells 2019;37:855–864 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Christodoulides C, Lagathu C, Sethi JK, Vidal-Puig A. Adipogenesis and WNT signalling. Trends Endocrinol Metab 2009;20:16–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Kang S, Bennett CN, Gerin I, Rapp LA, Hankenson KD, Macdougald OA. Wnt signaling stimulates osteoblastogenesis of mesenchymal precursors by suppressing CCAAT/enhancer-binding protein alpha and peroxisome proliferator-activated receptor gamma. J Biol Chem 2007;282:14515–14524 [DOI] [PubMed] [Google Scholar]
- 20. Cawthorn WP, Bree AJ, Yao Y, et al. Wnt6, Wnt10a and Wnt10b inhibit adipogenesis and stimulate osteoblastogenesis through a β-catenin-dependent mechanism. Bone 2012;50:477–489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Xie YY, Mo CL, Cai YH, et al. Pygo2 regulates adiposity and glucose homeostasis via β-catenin–Axin2–GSK3β signaling pathway. Diabetes 2018;67:2569–2584 [DOI] [PubMed] [Google Scholar]
- 22. Singh R, Artaza JN, Taylor WE, et al. Testosterone inhibits adipogenic differentiation in 3T3-L1 cells: nuclear translocation of androgen receptor complex with beta-catenin and T-cell factor 4 may bypass canonical Wnt signaling to down-regulate adipogenic transcription factors. Endocrinology 2006;147:141–154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Cawthorn WP, Heyd F, Hegyi K, Sethi JK. Tumour necrosis factor-alpha inhibits adipogenesis via a beta-catenin/TCF4(TCF7L2)-dependent pathway. Cell Death Differ 2007;14:1361–1373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Christodoulides C, Laudes M, Cawthorn WP, et al. The Wnt antagonist Dickkopf-1 and its receptors are coordinately regulated during early human adipogenesis. J Cell Sci 2006;119:2613–2620 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Takemaru K, Yamaguchi S, Lee YS, Zhang Y, Carthew RW, Moon RT. Chibby, a nuclear beta-catenin-associated antagonist of the Wnt/Wingless pathway. Nature 2003;422:905–909 [DOI] [PubMed] [Google Scholar]
- 26. Villanueva CJ, Waki H, Godio C, et al. TLE3 is a dual-function transcriptional coregulator of adipogenesis. Cell Metab 2011;13:413–427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Arango NA, Szotek PP, Manganaro TF, Oliva E, Donahoe PK, Teixeira J. Conditional deletion of beta-catenin in the mesenchyme of the developing mouse uterus results in a switch to adipogenesis in the myometrium. Dev Biol 2005;288:276–283 [DOI] [PubMed] [Google Scholar]
- 28. Longo KA, Wright WS, Kang S, et al. Wnt10b inhibits development of white and brown adipose tissues. J Biol Chem 2004;279:35503–35509 [DOI] [PubMed] [Google Scholar]
- 29. Baron R, Kneissel M. WNT signaling in bone homeostasis and disease: from human mutations to treatments. Nat Med 2013;19:179–192 [DOI] [PubMed] [Google Scholar]
- 30. Krishnan V, Bryant HU, Macdougald OA. Regulation of bone mass by Wnt signaling. J Clin Invest 2006;116:1202–1209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Boyden LM, Mao J, Belsky J, et al. High bone density due to a mutation in LDL-receptor-related protein 5. N Engl J Med 2002;346:1513–1521 [DOI] [PubMed] [Google Scholar]
- 32. Little RD, Carulli JP, Del Mastro RG, et al. A mutation in the LDL receptor-related protein 5 gene results in the autosomal dominant high-bone-mass trait. Am J Hum Genet 2002;70:11–19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Maupin KA, Droscha CJ, Williams BO. A comprehensive overview of skeletal phenotypes associated with alterations in Wnt/β-catenin signaling in humans and mice. Bone Res 2013;1:27–71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Bennett CN, Longo KA, Wright WS, et al. Regulation of osteoblastogenesis and bone mass by Wnt10b. Proc Natl Acad Sci USA 2005;102:3324–3329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Kim SP, Frey JL, Li Z, et al. Sclerostin influences body composition by regulating catabolic and anabolic metabolism in adipocytes. Proc Natl Acad Sci USA 2017;114:E11238–E11247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. van Amerongen R. Alternative Wnt pathways and receptors. Cold Spring Harb Perspect Biol 2012;4:a007914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. van Tienen FH, Laeremans H, van der Kallen CJ, Smeets HJ. Wnt5b stimulates adipogenesis by activating PPARgamma, and inhibiting the beta-catenin dependent Wnt signaling pathway together with Wnt5a. Biochem Biophys Res Commun 2009;387:207–211 [DOI] [PubMed] [Google Scholar]
- 38. Kanazawa A, Tsukada S, Kamiyama M, Yanagimoto T, Nakajima M, Maeda S. Wnt5b partially inhibits canonical Wnt/beta-catenin signaling pathway and promotes adipogenesis in 3T3-L1 preadipocytes. Biochem Biophys Res Commun 2005;330:505–510 [DOI] [PubMed] [Google Scholar]
- 39. Christodoulides C, Scarda A, Granzotto M, et al. WNT10B mutations in human obesity. Diabetologia 2006;49:678–684 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Kanazawa A, Tsukada S, Sekine A, et al. Association of the gene encoding wingless-type mammary tumor virus integration-site family member 5B (WNT5B) with type 2 diabetes. Am J Hum Genet 2004;75:832–843 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Van Camp JK, Beckers S, Zegers D, Verrijken A, Van Gaal LF, Van Hul W. Common genetic variation in sFRP5 is associated with fat distribution in men. Endocrine 2014;46:477–484 [DOI] [PubMed] [Google Scholar]
- 42. Shungin D, Winkler TW, Croteau-Chonka DC, et al. New genetic loci link adipose and insulin biology to body fat distribution. Nature 2015;518:187–196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Heid IM, Jackson AU, Randall JC, et al. Meta-analysis identifies 13 new loci associated with waist-hip ratio and reveals sexual dimorphism in the genetic basis of fat distribution. Nat Genet 2010;42:949–960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Hao HX, Xie Y, Zhang Y, et al. ZNRF3 promotes Wnt receptor turnover in an R-spondin-sensitive manner. Nature 2012;485:195–200 [DOI] [PubMed] [Google Scholar]
- 45. Loh NY, Minchin JEN, Pinnick KE, et al. RSPO3 impacts body fat distribution and regulates adipose cell biology in vitro. Nat Commun 2020;11:2797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Wu Y, Broadaway KA, Raulerson CK, et al. Colocalization of GWAS and eQTL signals at loci with multiple signals identifies additional candidate genes for body fat distribution. Hum Mol Genet 2019;28:4161–4172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Saarinen A, Saukkonen T, Kivela T, et al. Low density lipoprotein receptor-related protein 5 (LRP5) mutations and osteoporosis, impaired glucose metabolism and hypercholesterolaemia. Clin Endocrinol (Oxf) 2010;72:481–488 [DOI] [PubMed] [Google Scholar]
- 48. Singh R, Smith E, Fathzadeh M, et al. Rare nonconservative LRP6 mutations are associated with metabolic syndrome. Hum Mutat 2013;34:1221–1225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Loh NY, Neville MJ, Marinou K, et al. LRP5 regulates human body fat distribution by modulating adipose progenitor biology in a dose- and depot-specific fashion. Cell Metab 2015;21:262–273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Styrkarsdottir U, Thorleifsson G, Sulem P, et al. Nonsense mutation in the LGR4 gene is associated with several human diseases and other traits. Nature 2013;497:517–520 [DOI] [PubMed] [Google Scholar]
- 51. Zou Y, Ning T, Shi J, et al. Association of a gain-of-function variant in LGR4 with central obesity. Obesity (Silver Spring) 2017;25:252–260 [DOI] [PubMed] [Google Scholar]
- 52. Jin T. Current understanding on role of the Wnt signaling pathway effector TCF7L2 in glucose homeostasis. Endocr Rev 2016;37:254–277 [DOI] [PubMed] [Google Scholar]
- 53. Chen M, Lu P, Ma Q, et al. CTNNB1/β-catenin dysfunction contributes to adiposity by regulating the cross-talk of mature adipocytes and preadipocytes. Sci Adv 2020;6:eaax9605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Bagchi DP, Nishii A, Li Z, et al. Wnt/β-catenin signaling regulates adipose tissue lipogenesis and adipocyte-specific loss is rigorously defended by neighboring stromal-vascular cells. Mol Metab 2020;42:101078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Mori H, Prestwich TC, Reid MA, et al. Secreted frizzled-related protein 5 suppresses adipocyte mitochondrial metabolism through WNT inhibition. J Clin Invest 2012;122:2405–2416 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Rulifson IC, Majeti JZ, Xiong Y, et al. Inhibition of secreted frizzled-related protein 5 improves glucose metabolism. Am J Physiol Endocrinol Metab 2014;307:E1144–E1152 [DOI] [PubMed] [Google Scholar]
- 57. Kim SP, Da H, Li Z, et al. Lrp4 expression by adipocytes and osteoblasts differentially impacts sclerostin’s endocrine effects on body composition and glucose metabolism. J Biol Chem 2019;294:6899–6911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Nygaard R, Yu J, Kim J, et al. Structural basis of WLS/Evi-mediated Wnt transport and secretion. Cell 2021;184:194–206.e114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Bagchi DP, Li Z, Corsa CA, et al. Wntless regulates lipogenic gene expression in adipocytes and protects against diet-induced metabolic dysfunction. Mol Metab 2020;39:100992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Yu Y, Kim H, Choi S, et al. Targeting a lipid desaturation enzyme, SCD1, selectively eliminates colon cancer stem cells through the suppression of Wnt and NOTCH signaling. Cells 2021;10:106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Geoghegan G, Simcox J, Seldin MM, et al. Targeted deletion of Tcf7l2 in adipocytes promotes adipocyte hypertrophy and impaired glucose metabolism. Mol Metab 2019;24:44–63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Chen X, Ayala I, Shannon C, et al. The diabetes gene and Wnt pathway effector TCF7L2 regulates adipocyte development and function. Diabetes 2018;67:554–568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Nguyen-Tu MS, Martinez-Sanchez A, Leclerc I, Rutter GA, da Silva Xavier G. Adipocyte-specific deletion of Tcf7l2 induces dysregulated lipid metabolism and impairs glucose tolerance in mice. Diabetologia 2021;64:129–141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Lengyel E, Makowski L, DiGiovanni J, Kolonin MG. Cancer as a matter of fat: the crosstalk between adipose tissue and tumors. Trends Cancer 2018;4:374–384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Igal RA. Stearoyl CoA desaturase-1: new insights into a central regulator of cancer metabolism. Biochim Biophys Acta 2016;1861:1865–1880 [DOI] [PubMed] [Google Scholar]
- 66. Zhan T, Rindtorff N, Boutros M. Wnt signaling in cancer. Oncogene 2017;36:1461–1473 [DOI] [PMC free article] [PubMed] [Google Scholar]