Abstract
Berberine (BBR) is an isoquinoline alkaloid from plants known to improve cardiac mitochondrial function in gestational diabetes mellitus (GDM) offspring but the mechanism is poorly understood. We examined the role of the mitochondrial phospholipid cardiolipin (CL) in mediating this cardiac improvement. C57BL/6 female mice were fed either a Lean-inducing low-fat diet or a GDM-inducing high-fat diet for 6 weeks prior to breeding. Lean and GDM-exposed male offspring were randomly assigned a low-fat, high-fat, or high-fat diet containing BBR at weaning for 12 weeks. The content of CL was elevated in the heart of GDM offspring fed a high fat diet containing BBR. The increase in total cardiac CL was due to significant increases in the most abundant and functionally important CL species, tetralinoleoyl-CL and this correlated with an increase in the expression of the CL remodeling enzyme tafazzin. Additionally, BBR treatment increased expression of cardiac enzymes involved in fatty acid uptake and oxidation and electron transport chain subunits in high fat diet fed GDM offspring. Thus, dietary BBR protection from cardiac dysfunction in GDM exposed offspring involves improvement in mitochondrial function mediated through increased synthesis of CL.
Subject terms: Biochemistry, Biological techniques, Cell biology, Molecular biology, Cardiology, Diseases, Medical research
Introduction
Maternal obesity or overweight before and throughout pregnancy is a major risk factor for development of gestational diabetes mellitus (GDM)1,2. Diagnosis of GDM has increased dramatically over the past two decades due in part to the obesity epidemic3–5. For example, 60% of all women are classified as being overweight at the beginning of pregnancy and approximately 30% of all women are classified as being obese at the beginning of pregnancy6. Maternal obesity and diabetes during pregnancy predispose offspring to the development of cardiometabolic diseases, including obesity and diabetes7,8. Moreover, we have shown that diet-induced GDM exposure accelerates postnatal high fat diet-induced obesity and cardiometabolic dysfunction in the offspring12,14,15. The neonatal period represents a key intervention window for reducing the lifetime disease risk of the effects of GDM9. Earlier interventions in vulnerable populations from GDM pregnancies could prevent the development of cardiometabolic disease, but presently few evidence-based strategies are known.
Berberine (BBR) is an isoquinoline alkaloid from plants that improves insulin sensitivity and reduces blood glucose levels10,11. The actions of BBR are also known to promote improved mitochondrial function11. We recently demonstrated that supplemental BBR in a high fat diet reduced adiposity and improved cardiac dysfunction in offspring of mouse dams with GDM12. In GDM offspring there were significant reductions in ejection fraction and fractional shortening. BBR improved the contractility of the left ventricle as reflected by a normalized myocardial performance index. In addition, BBR improved both diastolic and systolic function in GDM offspring. Thus, BBR treatment of high fat fed offspring improved overall metabolic health and cardiac mitochondrial function; however, the mechanism for the cardioprotective effect was unknown. The liver plays a key role in dietary lipid handling and metabolism. In addition, fatty acids derived from triacylglycerol are a major oxidative fuel for the heart and skeletal muscle. Optimal mitochondrial function in these tissues is paramount for maintaining healthy body weight, insulin sensitivity and cardiovascular function. Since cardiolipin (CL) is a key mitochondrial membrane phospholipid known to regulate cardiac mitochondrial bioenergetics13, we examined if changes in CL were, in part, responsible for the cardioprotective effects of BBR in the heart of high fat fed GDM offspring.
Materials and methods
Animal care and food preparation
The study was conducted with approval of the University of Manitoba Animal Policy and Welfare Committee. All methods were performed in accordance with the Canadian Council on Animal Care guidelines and regulations. This study is reported in accordance with ARRIVE guidelines. Animals were maintained in an environmentally controlled facility (12 h light/dark cycle) with ad libitium access to food and water. A diet-induced GDM rodent model used was previously described12,14,15. 6 week old female C57BL/6 mice were fed either a low-fat (10% kcal, Research Diets D12450B) or a high-fat/ sucrose diet (45% kcal, Research Diets D12451) for 6 weeks prior to mating for 4 days with 6–10 week old low-fat fed male C57BL/6 mouse. The diet assigned to the dams was continued during mating, pregnancy and suckling periods. Resulting litters were culled at a maximum of eight to prevent nutritional deprivation of offspring. Male offspring were weaned at 3 weeks and randomly assigned to one of three diets (1) low fat (10% kcal) (LF), (2) high-fat/sucrose (45% kcal) (HF), or (3) high fat/sucrose (45% kcal) diet supplemented with BBR (160 mg/kg/day) (HFB) for a 12 week period. Rodent food was prepared with BBR by generating agar blocks using powered rodent diet purchased commercially from Research Diets (Low fat—D12450B, or high fat D12451). Agar 14.5 g/kg (Sigma) and the indicated LF or HF diet 435 g/kg were dissolved in heated water (1 kg) with or without BBR 1.45 g/kg (Sigma). The mixture was cooled to 4 °C, cut into blocks and were placed in animal hoppers every 2–3 days12. All animals were 15 weeks of age when experiments were conducted. At 15 weeks of age, offspring underwent a 16 h fast before being anesthetized with 1–1.5% isoflurane and then euthanized by cervical dislocation.
Blood measurements
Non-esterified fatty acids (NEFA), triglycerides (TG), ketones and alanine amino transferase (ALT) were determined after 12 weeks of experimental diet. Plasma was collected from blood in tubes coated with EDTA (Sarstedt, Numbrecht, Germany) followed by centrifugation at 6000 RPM at 4 °C for 20 min. Commercially available kits were used to measure plasma non-esterified fatty acids, ketones, triglyceride (Wako Diagnositic), and alanine aminotransferase (Biotron Diagnostic). All assays were analyzed using an Epoch Biotek microplate spectrophotometer (Agilent Technologies, ON, Canada).
Tissue lipid measurements
A commercially available kit was used to measure tissue triglyceride (Wako Diagnositics). Phospholipids from tissue homogenates including phosphatidylethanolamine (PE), phosphatidylserine (PS), phosphatidylcholine (PC) and phosphatidylinositol (PI) were quantitated by lipid phosphorus assay16. Molecular species of CL were quantitated from tissue homogenates by HPLC coupled to electrospray ionization mass spectrometry17. The total CL was calculated from the sum of the eight most prominent CL species.
RNA isolation and quantitative RT-PCR analysis
Total RNA was isolated from cardiac, skeletal muscle (soleus) or liver tissue using an RNeasy® kit (Qiagen) and first-strand cDNA synthesis performed (2 µg total RNA) with SuperScript II (Invitrogen). PCR was performed using Eppendorf Realplex2 instrument with the gene specific primers (IDT) as indicated (Table 1). All mRNA levels were quantitated using a standard curve followed by normalized to the geometric mean (geomean) of Tata Box binding protein (TBP) and Transcription factor IIB (TFIIB)18.
Table 1.
PCR primers.
| Gene | Forward primer | Reverse primer |
|---|---|---|
| TBP | ACCCTTCACCAATGACTCCTATG | TGACTGCAGCAAATCGCTTGG |
| TFIIB | TGGAGATTTGTCCACCATGA | GAATTGCCAAACTCATCAAAACT |
| TAZ | GACCCTCATCTCTGGGGGAT | CAGCTCCTTGGTGAAGCAGA |
| PTPMT | GCAACACCTCGAAGGAATGG | GAGATTGGCCAAGGTTGGGA |
| PGS | ACACAGGTTCCAGTGGATCAG | TTTATCTGCCCCTTCATGAGC |
| PGC1 | ATACCGCAAAGAGCACGAGAAG | CTCAAGAGCAGCGAAAGCGTCACAG |
| SIRT1 | GAAACCTTTGCCTCATCTACA | CACCTAGCCTATGACACAACT |
| CPT1 | ATGGCGGAAGCACACCAGGC | CGCCCAGACTCCGGTGGAGA |
|
SREBP1 CD36 |
ATCCAAGGGCATCTGAGAACTC TGGCTAAATGAGACTGGGACC |
GGCTATTCCGTGAACATCTCCTA ACATCACCACTCCAATCCCAAG |
| FABP1 | CTGGCACTGGTGGACGCCAG | TTGCCTGGGGCTTGTGCCAG |
| FABP4 | TGGAAGCTTGTCtCCAGTGA | AATCCCCATTTACGCTGATG |
|
NDUFS3 SDHD |
CTGACTTGACGGCAGTGGAT GATCCCTGCTTGGGTACTTGA |
CATACCAATTGGCCGCGATG AAGTAGCAAAGCCCAGCAA |
| COX5β | CGTCCATCAGCAACAAGAGA | AGATAACACAGGGGCTCAGT |
| ATP5i | CCCCTGCTGAAATCCCTACA | TAAAACCACATCCGCACCTC |
TBP tata box binding protein, TFIIB transcription factor IIB, TAZ tafazzin, PGS phosphatidylglycerolphosphate synthase, PTPMT protein tyrosine phosphatase mitochondrial 1, PGC1α peroxisome proliferator-activated receptor gamma coactivator 1α, SIRT1 sirtuin 1, CPT1β carnitine palmitoyltransferase 1, SREBP1 sterol regulatory element-binding protein 1, ACOX1 acyl-CoA oxidase, FABP fatty acid binding protein, NDUF NADH dehydrogenase oxidoreductase core subunit S3, SDHD succinate dehydrogenase complex subunit D, COX5β cytochrome c oxidase subunit 5β, ATP5i ATP synthase subunit 5i.
Statistical analysis
Data are expressed either as means ± standard error of the mean (SEM) or as means with individual data points representing each individual animal. Comparisons between Lean and GDM Dams were performed by unpaired two-tailed Student’s t-test. Comparisons between Lean and GDM exposed HF, HFB and LF fed offspring were determined separately for male and female animals using two-way analysis of variance using Tukey post-hoc analysis. When appropriate adjustments were made for multiple comparisons using Bonferroni correction. Analysis of covariance was used to determine whether body weight was a confounding factor within indirect calorimetry measurements. For each measurement, the offspring were derived from multiple litters. A probability value of < 0.05 was considered significant.
Results
Berberine treatment reduces triacylglycerol accumulation in the heart, skeletal muscle and liver of GDM exposed offspring
As previously reported12, BBR treatment attenuated body weight gain in HF fed offspring from both lean and GDM dams (Table 2). Next, we examined plasma lipids levels to determine how BBR affected lipid homeostasis in GDM offspring. Plasma levels of NEFA did not differ between lean and GDM offspring fed a LF diet. Plasma levels of NEFA were elevated in HF fed offspring from lean dams and this was reduced by treatment with BBR. In addition, BBR treatment reduced plasma levels of TG in lean offspring fed the HF diet. Plasma levels of ketones were elevated in GDM offspring compared to lean offspring and BBR treatment significantly reduced plasma ketone levels in the HF fed GDM offspring. Thus, BBR treatment attenuated the HF diet induced increase in plasma lipid levels in lean offspring but not GDM offspring.
Table 2.
Characteristics of offspring. All measurements were obtained following 12-weeks of the indicated diet (LF, HF, HFB).
| LEAN | LEAN | LEAN | GDM | GDM | GDM | |
|---|---|---|---|---|---|---|
| LF | HF | HFB | LF | HF | HFB | |
|
Body weight (g) |
26.3 ± 0.529c | 27.1 ± 0.808c | 22.5 ± 0.431e | 29.1 ± 0.522b | 31.3 ± 0.895a | 24.7 ± 0.500d |
| NEFA (µmol/L) | 402 ± 35.9a,b | 455 ± 31.7b | 298 ± 34.7a | 393 ± 29.3a,b | 364 ± 38.8a,b | 338 ± 29.3a,b |
| TG (mg/dL) | 20.0 ± 1.33a,b | 28.0 ± 3.22a | 17.9 ± 2.44b | 22.6 ± 2.48a | 29.6 ± 3.55a | 29.7 ± 4.01a |
| Ketones (mmol/L) | 0.440 ± 0.0961b | 0.514 ± 0.0751b | 0.689 ± 0.0884a,b | 0.822 ± 0.0961a | 0.770 ± 0.101a | 0.524 ± 0.0651b |
| ALT (IU/L) | 35.8 ± 7.00b | 28.9 ± 5.92b | 30.9 ± 6.39b | 40.5 ± 4.72a,b | 46.7 ± 5.54a | 37.7 ± 4.72a,b |
Values are means ± SEMs (n = 5–15), means without a common letter differ, p < 0.05.
NEFA non-esterified fatty acids, ALT alanine aminotransferase.
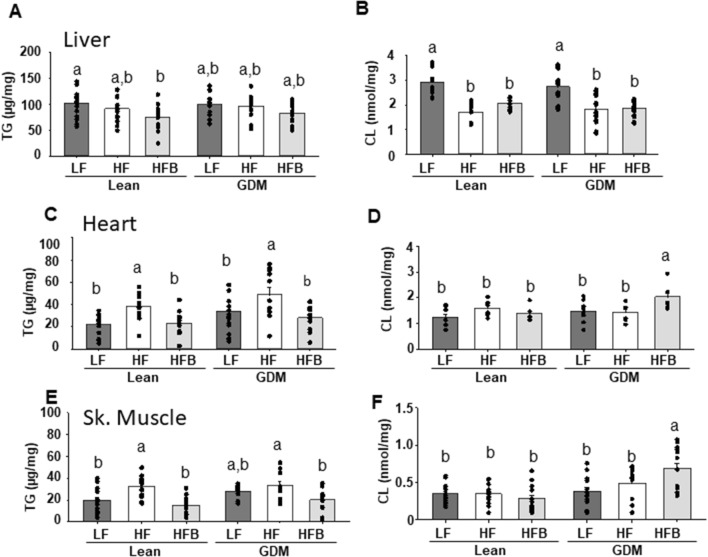
We determined that BBR broadly reduced TG accumulation in the liver, heart and skeletal muscle (Fig. 1A,C,E). Specifically, an elevation in TG (~ 50%, p < 0.05) which occurred in the heart of both the HF fed Lean and GDM exposed offspring, was normalized in both groups of HFB offspring (Fig. 1C). A similar BBR mediated reduction in TG accumulation was observed in skeletal muscle (Fig. 1E). In the livers of lean offspring, BBR induced a more modest, but significant (~ 25%, p < 0.05) reduction in TG (Fig. 1A).
Figure 1.
Triacylglycerol (TG) and total cardiolipin (CL) levels in liver, heart and skeletal muscle. All measurements were obtained following 12 weeks of the indicated diet (LF, HF, HFB). Values are means ± SEMs (n = 5–15). Means without a common letter differ, p < 0.05. GDM gestational diabetes mellitus.
While significant modification of hepatic gene expression occurred in response to HF diet, including increased fatty acid (FA) uptake genes (Fatbp1, Fatbp4) and reduced fatty acid oxidation (FAO) genes (Pgc1a, Sirt1), these levels were not further augmented by GDM exposure (Table 3). When BBR was supplemented in the diet, the significant reduction in hepatic TG levels was limited to the lean gestational group (HFB Lean, Fig. 1A). This result was mirrored by significant alterations in hepatic gene expression (increased FAO and reduced FA uptake) in the HFB Lean offspring (Table 3)19,20. In the HF fed offspring, GDM-exposure promoted a robust increase in the gene expression of several mitochondrial respiratory complexes (Nduf, Cox5b). The increased mitochondrial respiratory complex gene expression was attenuated in HFB mice. The elevation in the gene expression of respiratory complexes may be a compensatory mechanism in the liver of GDM HF fed offspring to maintain mitochondrial function.
Table 3.
PCR analysis of liver.
| Gene | LEAN | LEAN | LEAN | GDM | GDM | GDM |
|---|---|---|---|---|---|---|
| LF | HF | HFB | LF | HF | HFB | |
| CL synthesis | ||||||
| TAZ | 1.00 ± 0.11a,b | 0.84 ± 0.12b | 1.21 ± 0.20a.b | 1.00 ± 0.18a,b | 1.67 ± 0.61a | 0.82 ± 0.10b |
| PGS | 1.00 ± 0.18 | 1.17 ± 0.35 | 0.97 ± 0.26 | 1.32 ± 0.34 | 1.34 ± 0.26 | 0.97 ± 0.26 |
| PTPMTI | 1.00 ± 0.14 | 0.82 ± 0.16 | 0.70 ± 0.18 | 1.19 ± 0.27 | 1.11 ± 17 | 0.85 ± 0.13 |
| FAO | ||||||
| PGC1α | 1.00 ± 0.14a,b | 0.67 ± 0.11b | 1.29 ± 0.11a | 1.31 ± 0.25a | 0.74 ± 0.11b | 0.57 ± 0.04b |
| SIRT1 | 1.00 ± 0.12a | 0.60 ± 0.10b | 0.83 ± 0.08a,b | 0.90 ± 0.10a,b | 0.66 ± 0.15b | 0.77 ± 0.16a,b |
| CPT1β | 1.00 ± 0.27b | 1.13 ± 0.24b | 3.32 ± 1.09a | 0.40 ± 0.11c | 0.88 ± 0.14b | 0.73 ± 0.24b |
| SREBP1 | 1.00 ± 0.28 | 1.80 ± 0.37 | 1.11 ± 0.18 | 1.07 ± 0.22 | 2.10 ± 0.68 | 1.24 ± 0.11 |
| ACOX1 | 1.00 ± 0.06a | 0.84 ± 0.11a | 0.39 ± 0.12b | 0.84 ± 0.35a | 0.55 ± 0.09a,b | 0.57 ± 0.18a,b |
| FA uptake | ||||||
| CD36 | 1.00 ± 0.10a | 0.59 ± 0.08b | 0.53 ± 0.11b | 0.97 ± 0.14a | 0.98 ± 0.19a | 0.73 ± 0.09a,b |
| FABP1 | 1.00 ± 0.14a,b | 1.26 ± 0.21a | 0.94 ± 0.12b | 0.80 ± 0.06b | 1.01 ± 0.09a | 0.81 ± 0.03b |
| FABP4 | 1.00 ± 0.14a,b | 1.62 ± 0.27a | 1.20 ± 0.51a,b | 0.49 ± 0.10b | 2.03 ± 0.12a | 0.87 ± 0.14b |
| ETC | ||||||
| NDUFS3 (I) | 1.00 ± 0.12a,b | 1.10 ± 0.31a,b | 1.25 ± 0.18a | 0.55 ± 0.07b | 1.51 ± 0.17a | 0.78 ± 0.07b |
| SDHD (II) | 1.00 ± 0.08b | 1.08 ± 0.21b | 2.82 ± 0.58a | 1.25 ± 0.22a,b | 1.96 ± 0.31a | 1.44 ± 0.22a,b |
| COX5β (IV) | 1.00 ± 0.25b | 0.82 ± 0.36b | 0.97 ± 0.27b | 2.18 ± 0.50a | 2.34 ± 0.65a | 0.88 ± 0.43b |
| ATP5i (V) | 1.00 ± 0.20 | 0.77 ± 0.09 | 0.86 ± 0.19 | 0.88 ± 0.23 | 0.98 ± 0.18 | 0.86 ± 0.19 |
mRNAs were measured in the liver of offspring following 12-weeks of the indicated diet (LF, HF, HFB). Values are means ± SEMs (n = 7), means without a common letter differ, p < 0.05.
TAZ tafazzin, PGS phosphatidylglycerolphosphate synthase, PTPMT protein tyrosine phosphatase mitochondrial 1, PGC1α peroxisome proliferator-activated receptor gamma coactivator 1α, SIRT1 sirtuin 1, CPT1β carnitine palmitoyltransferase 1, SREBP1 sterol regulatory element-binding protein 1, ACOX1 acyl-CoA oxidase, FABP1 fatty acid binding protein 1, NDUF NADH dehydrogenase oxidoreductase core subunit S3, SDHD succinate dehydrogenase complex subunit D, COX5β cytochrome c oxidase subunit 5β, ATP5i ATP synthase subunit 5i.
We next assessed hepatic cardiolipin (CL) content since it is well established that this phospholipid is required for normal mitochondrial bioenergetics function13. HF diet significantly reduced hepatic CL in both lean and GDM offspring (Fig. 1B) (Table 4). Consistent with the lack of BBR mediated protection against mitochondrial dysfunction in the liver12, BBR treatment did not restore hepatic CL content (Fig. 1B). Minimal alterations in other phospholipids were observed by BBR treatment of GDM offspring (Table 4). In addition, plasma levels of ALT were not appreciably altered with BBR treatment of HF diet fed GDM offspring, suggesting normal liver function (Table 2). Together these data indicate that BBR treatment of GDM offspring had a minimal effect on hepatic mitochondrial parameters.
Table 4.
Quantitation of phospholipids and CL molecular species in the liver. Quantitation of phospholipids and molecular species of CL from the liver of offspring following 12-weeks of the indicated diet (LF, HF, HFB).
| Phospholipid species | LEAN | LEAN | LEAN | GDM | GDM | GDM |
|---|---|---|---|---|---|---|
| LF (nmol/mg) | HF (nmol/mg) | HFB (nmol/mg) | LF (nmol/mg) | HF (nmol/mg) | HFB (nmol/mg) | |
| CL species | ||||||
| 1422 | 0.337 ± 0.027a | 0.100 ± 0.012c | 0.083 ± 0.012c | 0.269 ± 0.021b | 0.119 ± 0.018c | 0.075 ± 0.005c |
| 1424 | 0.753 ± 0.050a | 0.153 ± 0.059b | 0.117 ± 0.016b | 0.683 ± 0.047a | 0.207 ± 0.035b | 0.124 ± 0.010b |
| 1448 | 0.609 ± 0.033b | 0.720 ± 0.059b | 0.959 ± 0.042a | 0.554 ± 0.065b | 0.696 ± 0.069a,b | 0.814 ± 0.047a |
| 1450 | 0.792 ± 0.063a | 0.480 ± 0.044b | 0.604 ± 0.024b | 0.725 ± 0.067a | 0.508 ± 0.054b | 0.546 ± 0.027b |
| 1472 | 0.118 ± 0.013a | 0.075 ± 0.006b | 0.090 ± 0.003a,b | 0.129 ± 0.011a | 0.086 ± 0.008b | 0.087 ± 0.004b |
| 1474 | 0.238 ± 0.021a | 0.087 ± 0.089b | 0.117 ± 0.011b | 0.221 ± 0.019a | 0.128 ± 0.021b | 0.109 ± 0.006b |
| 1496 | 0.058 ± 0.005 | 0.050 ± 0.004 | 0.060 ± 0.005 | 0.059 ± 0.005 | 0.052 ± 0.006 | 0.049 ± 0.003 |
| 1498 | 0.064 ± 0.014a | 0.017 ± 0.002b | 0.030 ± 0.003b | 0.050 ± 0.004a | 0.023 ± 0.005b | 0.028 ± 0.001b |
| PC | 59.6 ± 2.13a | 49.6 ± 2.06b | 51.1 ± 1.51b | 57.2 ± 2.69a | 51.8 ± 3.84c | 52.0 ± 2.16c |
| PE | 33.6 ± 1.26a | 21.9 ± 1.06c | 28.1 ± 1.24b | 33.0 ± 1.47a | 24.9 ± 2.43c | 23.8 ± 1.30c |
| PS | 4.1 ± 0.17 | 3.1 ± 0.29 | 4.1 ± 0.45 | 3.7 ± 0.21 | 3.7 ± 0.42 | 3.5 ± 0.19 |
| PI | 11.7 ± 0.67a | 9.9 ± 0.68a,b | 13.4 ± 1.42a | 11.4 ± 1.30a | 9.1 ± 1.33a,b | 6.6 ± 0.78b |
Values are means ± SEMs (n = 7–10), means without a common letter differ, p < 0.05.
Berberine treatment increases cardiolipin in the heart and skeletal muscle of GDM exposed offspring
In the heart and skeletal muscles, CL content was similar among the Lean and GDM offspring LF and HF-fed groups. Interestingly HFB did not affect CL levels in the Lean offspring, but increased CL (~ 42%, p < 0.05) in the GDM offspring (Fig. 1D,F). Interestingly, in the heart this was due to significant increases (~ 32%, p < 0.05) in the most abundant and functionally important CL species, tetralinoleoylcardiolipin (L4CL) (1448) (Table 5). A BBR-mediated increase (47%, p < 0.05) in mRNA expression of tafazzin, the major CL remodeling enzyme13, may account for this increase in cardiac CL content in GDM HF offspring compared to lean HF offspring (Table 6). In addition, further PCR analysis of the heart indicated elevations in expression of enzymes involved in FA uptake, FAO (PGC1α, CD36, FABP1, CTP1β) and ETC subunits (NDUF(I), SDHD(II)) of the GDM exposed groups compared to lean animals (Table 6). This additionally would account for the previous observation of a BBR mediated improvement of cardiac mitochondrial function in HF diet fed GDM offspring12. Minimal alteration in other cardiac phospholipids was observed by BBR treatment of GDM offspring. Together, these results indicate that the BBR mediated protection from cardiac dysfunction in GDM exposed offspring, in part, involves its effects on improvements in mitochondrial function through increased CL synthesis.
Table 5.
PCR analysis of the heart. mRNAs were measured from the heart of offspring following 12-weeks of the indicated diet (LF, HF, HFB).
| Gene | LEAN | LEAN | LEAN | GDM | GDM | GDM |
|---|---|---|---|---|---|---|
| LF | HF | HFB | LF | HF | HFB | |
| CL synthesis | ||||||
| TAZ | 1.00 ± 0.09b | 1.38 ± 0.11a | 0.87 ± 0.05b | 1.16 ± 0.08a,b | 1.19 ± 0.10a | 1.28 ± 0.05a |
| PGS | 1.00 ± 0.21b | 1.38 ± 0.05a | 0.90 ± 0.10b | 1.13 ± 0.13a,b | 1.40 ± 0.15a | 1.06 ± 0.08b |
| PTPMTI | 1.00 ± 0.10 | 1.00 ± 0.09 | 0.92 ± 0.04 | 1.00 ± 0.22 | 1.15 ± 16 | 1.10 ± 0.24 |
| FAO | ||||||
| PGC1α | 1.00 ± 0.14b | 1.12 ± 0.09b | 1.02 ± 0.12b | 1.79 ± 0.21a | 1.62 ± 0.18a | 1.46 ± 0.15a |
| SIRT1 | 1.00 ± 0.15 | 0.91 ± 0.07 | 0.79 ± 0.04 | 1.05 ± 0.22 | 0.82 ± 0.06 | 1.14 ± 0.10 |
| CPT1β | 1.00 ± 0.16a,b | 1.16 ± 0.19a,b | 0.83 ± 0.15b | 1.06 ± 0.22a,b | 1.51 ± 0.17a | 1.52 ± 0.22a |
| ACOX1 | 1.00 ± 0.23a,b | 0.37 ± 0.10b | 1.45 ± 0.28a | 1.10 ± 0.32a,b | 1.13 ± 0.28a,b | 1.40 ± 0.31a |
| FA uptake | ||||||
| CD36 | 1.00 ± 0.10a | 0.50 ± 0.06b | 0.54 ± 0.09b | 1.37 ± 0.14a | 0.65 ± 0.07b | 1.02 ± 0.14a |
| FABP1 | 1.00 ± 0.14a | 0.52 ± 0.05b | 0.50 ± 0.08b | 0.63 ± 0.10b | 0.64 ± 0.04b | 1.00 ± 0.19a |
| ETC | ||||||
| NDUF (I) | 1.00 ± 0.10a | 0.67 ± 0.04b | 0.70 ± 0.09b | 1.15 ± 0.18a | 0.84 ± 0.08a,b | 1.01 ± 0.17a |
| SDHD (II) | 1.00 ± 0.20a | 0.78 ± 0.06a,b | 0.54 ± 0.09b | 0.90 ± 0.24a,b | 0.61 ± 0.09b | 1.24 ± 0.18a |
| COX5β (IV) | 1.00 ± 0.08 | 1.27 ± 0.06 | 1.02 ± 0.10 | 1.20 ± 0.14 | 1.30 ± 0.15 | 1.26 ± 0.16 |
| ATP5i (V) | 1.00 ± 0.12 | 1.11 ± 0.09 | 0.96 ± 0.13 | 1.05 ± 0.22 | 1.16 ± 0.12 | 1.15 ± 0.27 |
Values are means ± SEMs (n = 7), means without a common letter differ, p < 0.05.
TAZ tafazzin, PGS phosphatidylglycerolphosphate synthase, PTPMT protein tyrosine phosphatase mitochondrial 1, PGC1α peroxisome proliferator-activated receptor gamma coactivator 1α, SIRT1 sirtuin 1, CPT1β carnitine palmitoyltransferase 1, SREBP1 sterol regulatory element-binding protein 1, ACOX1 acyl-CoA oxidase, FABP fatty acid binding protein, NDUF NADH dehydrogenase oxidoreductase core subunit S3, SDHD succinate dehydrogenase complex subunit D, COX5β cytochrome c oxidase subunit 5β, ATP5i ATP synthase subunit 5i.
Table 6.
Quantitation of phospholipids and CL molecular species in the heart.
| Phospholipid species | LEAN | LEAN | LEAN | GDM | GDM | GDM |
|---|---|---|---|---|---|---|
| LF (nmol/mg) | HF (nmol/mg) | HFB (nmol/mg) | LF (nmol/mg) | HF (nmol/mg) | HFB (nmol/mg) | |
| CL species | ||||||
| 1422 | 0.098 ± 0.010a | 0.036 ± 0.001b | 0.028 ± 0.004b | 0.115 ± 0.014a | 0.046 ± 0.008b | 0.030 ± 0.003b |
| 1424 | 0.055 ± 0.008a | 0.020 ± 0.004b | 0.023 ± 0.003b | 0.057 ± 0.010a | 0.023 ± 0.005b | 0.030 ± 0.005b |
| 1448 | 0.569 ± 0.077b | 0.901 ± 0.059a | 0.718 ± 0.073b | 0.639 ± 0.081b | 0.715 ± 0.07b | 0.943 ± 0.072a |
| 1450 | 0.180 ± 0.022b | 0.222 ± 0.016a,b | 0.232 ± 0.026a | 0.232 ± 0.018a,b | 0.249 ± 0.037a | 0.283 ± 0.03a |
| 1472 | 0.086 ± 0.03 | 0.100 ± 0.015 | 0.119 ± 0.021 | 0.115 ± 0.016 | 0.113 ± 0.012 | 0.151 ± 0.011 |
| 1474 | 0.050 ± 0.005 | 0.064 ± 0.007 | 0.063 ± 0.006 | 0.052 ± 0.007 | 0.065 ± 0.014 | 0.082 ± 0.011 |
| 1496 | 0.153 ± 0.011b | 0.205 ± 0.024a,b | 0.233 ± 0.018a | 0.201 ± 0.030a,b | 0.228 ± 0.035a | 0.287 ± 0.025a |
| 1498 | 0.044 ± 0.007b | 0.044 ± 0.007b | 0.086 ± 0.020a | 0.061 ± 0.006b | 0.057 ± 0.01b | 0.107 ± 0.023a |
| PC | 44.06 ± 1.88 | 48.91 ± 2.34 | 40.68 ± 2.00 | 43.51 ± 2.67 | 51.40 ± 3.74 | 45.70 ± 3.09 |
| PE | 24.30 ± 1.44 | 27.61 ± 2.18 | 22.69 ± 1.22 | 23.57 ± 2.00 | 28.81 ± 2.63 | 25.83 ± 1.87 |
| PS | 5.92 ± 0.36a | 6.44 ± 0.54a | 6.75 ± 0.33a | 6.12 ± 0.45a | 5.18 ± 0.57a,b | 3.60 ± 0.67b |
| PI | 4.12 ± 0.33b | 5.57 ± 0.73a,b | 5.46 ± 0.55a,b | 6.24 ± 0.63a | 6.17 ± 0.46a | 4.45 ± 0.25b |
Quantitation of phospholipids and molecular species of CL from the heart of offspring following 12-weeks of the indicated diet (LF, HF, HFB). Values are means ± SEMs (n = 8), means without a common letter differ, p < 0.05.
Discussion
The major findings of this study are, (1) supplemental BBR in a HF diet attenuates tissue TG accumulation in offspring from lean and GDM dams, (2) The cardiometabolic protective effects of BBR in GDM offspring are minimally impacted by alterations in liver mitochondrial metabolism, (3) BBR treatment increased total CL in heart and skeletal muscle and L4CL levels in the heart of GDM offspring, (4) The increase in cardiac L4CL was likely due to an increase in expression of the CL remodeling enzyme tafazzin, and (5) Elevation in expression of cardiac enzymes involved in FA uptake, FAO and ETC subunits coupled with the increase in L4CL may account for the improved cardiac mitochondrial function in BBR treated GDM offspring.
Previously we demonstrated that that supplemental BBR in a high fat diet reduced adiposity and improved cardiac mitochondrial dysfunction in offspring of mouse dams with GDM fed a HF diet12. However, the mechanism for the improvement in cardiac function in these GDM offspring was unknown. Since abnormal lipid accumulation is associated with the development of cardiac and mitochondrial dysfunction, and the liver, heart and skeletal muscle play major roles in handling lipids21, we assessed whether cardiac, skeletal muscle and liver lipid homeostasis was altered by BBR treatment in GDM offspring fed a HF diet. Consistent with the role of BBR as a lipid-lowering agent11, we determined that the cardiovascular protective effect of BBR was mirrored by reduced TG accumulation in the liver, heart and skeletal muscle.
There are numerous studies on the beneficial effects of BBR on the prevention of hepatic steatosis19,20,22. Since GDM has previously been shown to promote hepatic steatosis14,15 and BBR attenuated TG accumulation in liver (Fig. 1), we investigated whether BBR altered gene expression of hepatic bioenergetics enzymes. While significant modification of hepatic gene expression occurred in response to HF diet for both FA uptake and FAO, these levels were not further augmented by GDM exposure. As previously reported, BBR treatment did not significantly improve hepatic mitochondrial CI or CII mediated oxygen consumption rate for any respiratory state measured12.
Based on evidence that BBR protected against HF diet induced mitochondrial dysfunction in muscle23 and our previous observation that dietary BBR prevented the HF diet mediated reductions in cardiac state I and state III respiration and normalized spare capacity in GDM offspring12, we assessed whether these protective effects accompanied changes in CL, the signature phospholipid that regulates mitochondrial bioenergetics13. BBR was effective at elevating CL content in both the heart and skeletal muscle of the GDM exposed groups. Interestingly, in the heart this was due to an increase in L4CL, the most abundant and functionally important CL species13. PCR analysis indicated an increase in mRNA expression of tafazzin, a CL remodeling enzyme, which likely accounted for this increase in cardiac L4CL content. Studies in mice have indicated that tafazzin deficiency in heart accompanies reduced cardiac L4CL and mitochondrial bioenergetic function24–27. This is underscored by the X-linked genetic disease Barth Syndrome in which mutations in tafazzin result in reduced cardiac CL and L4CL levels in patients28.
Moreover, mRNA gene expression analysis demonstrated that increased expression of enzymes involved in FA uptake, FAO and ETC subunits in GDM HF exposed groups compared to lean animals could contribute to our previous observation of a BBR mediated improvement of cardiac function in HF diet fed GDM offspring12. Together, our results indicate that the BBR mediated protection from cardiac dysfunction in GDM HF exposed offspring involve its effects on improvements to mitochondrial function which are mediated, in part, through increased CL synthesis.
Acknowledgements
LKC was the recipient of a CIHR/HSFC IMPACT Fellowship. GMH is a Canada Research Chair in Molecular Cardiolipin Metabolism. VWD is the Allen Rouse-Manitoba Medical Services Foundation Basic Scientist. This research was supported by an Environments, Genes and Chronic Disease Canadian Institutes for Health Research (CIHR) Team Grant #144626, the Heart and Stroke Foundation of Canada, the Natural Sciences and Engineering Research Council (NSERC), Children’s Hospital Research Institute of Manitoba (CHRIM) and the University of Manitoba Research Grants Program (URGP).
Author contributions
L.K.C., V.W.D. & G.M.H. were responsible for conceptual design and writing and editing of the manuscript. L.K.C., M.V., B.X., G.C.S. performed experiments. All authors reviewed the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Catalano PM, Vargo KM, Bernstein IM, Amini SB. Incidence and risk factors associated with abnormal postpartum glucose tolerance in women with gestational diabetes. Am. J. Obst. Gynecol. 1991;165:914–919. doi: 10.1016/0002-9378(91)90438-W. [DOI] [PubMed] [Google Scholar]
- 2.Catalano PM, Huston L, Amini SB, Kalhan SC. Longitudinal changes in glucose metabolism during pregnancy in obese women with normal glucose tolerance and gestational diabetes mellitus. Am. J. Obst. Gynecol. 1999;180:903–916. doi: 10.1016/S0002-9378(99)70662-9. [DOI] [PubMed] [Google Scholar]
- 3.Ferrara A. Increasing prevalence of gestational diabetes mellitus: A public health perspective. Diab. Care. 2007;30(Suppl 2):S141–146. doi: 10.2337/dc07-s206. [DOI] [PubMed] [Google Scholar]
- 4.Reece EA, Leguizamon G, Wiznitzer A. Gestational diabetes: The need for a common ground. Lancet. 2009;373:1789–1797. doi: 10.1016/S0140-6736(09)60515-8. [DOI] [PubMed] [Google Scholar]
- 5.Guariguata L, Linnenkamp U, Beagley J, Whiting DR, Cho NH. Global estimates of the prevalence of hyperglycaemia in pregnancy. Diab. Res. Clin. Pract. 2014;103:176–185. doi: 10.1016/j.diabres.2013.11.003. [DOI] [PubMed] [Google Scholar]
- 6.Hinkle SN, et al. Prepregnancy obesity trends among low-income women, United States, 1999–2008. Matern. Child Health J. 2012;16:1339–1348. doi: 10.1007/s10995-011-0898-2. [DOI] [PubMed] [Google Scholar]
- 7.Clausen TD, et al. Overweight and the metabolic syndrome in adult offspring of women with diet-treated gestational diabetes mellitus or type 1 diabetes. J. Clin. Endocrinol. Metab. 2009;94:2464–2470. doi: 10.1210/jc.2009-0305. [DOI] [PubMed] [Google Scholar]
- 8.Franks PW, et al. Gestational glucose tolerance and risk of type 2 diabetes in young Pima Indian offspring. Diabetes. 2006;55:460–465. doi: 10.2337/diabetes.55.02.06.db05-0823. [DOI] [PubMed] [Google Scholar]
- 9.Chan LL, et al. Effect of maternal nutrient restriction from early to midgestation on cardiac function and metabolism after adolescent-onset obesity. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009;296:R1455–1463. doi: 10.1152/ajpregu.91019.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wei S, et al. Berberine attenuates development of the hepatic gluconeogenesis and lipid metabolism disorder in type 2 diabetic mice and in palmitate-incubated HepG2 cells through suppression of the HNF-4alpha miR122 pathway. PLoS ONE. 2016;11:e0152097. doi: 10.1371/journal.pone.0152097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chang W, Chen L, Hatch GM. Berberine as a therapy for type 2 diabetes and its complications: From mechanism of action to clinical studies. Biochem. Cell Biol. 2015;93:479–486. doi: 10.1139/bcb-2014-0107. [DOI] [PubMed] [Google Scholar]
- 12.Cole LK, et al. Supplemental berberine in a high-fat diet reduces adiposity and cardiac dysfunction in offspring of mouse dams with gestational diabetes mellitus. J. Nutr. 2021;20:nxaa408. doi: 10.1093/jn/nxaa408. [DOI] [PubMed] [Google Scholar]
- 13.Mejia EM, Hatch GM. Mitochondrial phospholipids: Role in mitochondrial function. J. Bioenerg. Biomembr. 2016;48:99–112. doi: 10.1007/s10863-015-9601-4. [DOI] [PubMed] [Google Scholar]
- 14.Pereira TJ, et al. Maternal obesity characterized by gestational diabetes increases the susceptibility of rat offspring to hepatic steatosis via a disrupted liver metabolome. J. Physiol. 2015;593:3181–3197. doi: 10.1113/JP270429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Brawerman GM, et al. Maternal resveratrol administration protects against gestational diabetes-induced glucose intolerance and islet dysfunction in the rat offspring. J. Physiol. 2019;597:4175–4192. doi: 10.1113/JP278082. [DOI] [PubMed] [Google Scholar]
- 16.Rouser G, Siakotos AN, Fleischer S. Quantitative analysis of phospholipids by thin-layer chromatography and phosphorus analysis of spots. Lipids. 1966;1:85–86. doi: 10.1007/BF02668129. [DOI] [PubMed] [Google Scholar]
- 17.Sparagna GC, Johnson CA, McCune SA, Moore RL, Murphy RC. Quantitation of cardiolipin molecular species in spontaneously hypertensive heart failure rats using electrospray ionization mass spectrometry. J. Lipid Res. 2005;46:1196–1204. doi: 10.1194/jlr.M500031-JLR200. [DOI] [PubMed] [Google Scholar]
- 18.Vandesompele J, et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002;3:RESEARCH034. doi: 10.1186/gb-2002-3-7-research0034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yuan X, et al. Berberine ameliorates nonalcoholic fatty liver disease by a global modulation of hepatic mRNA and lncRNA expression profiles. J. Transl. Med. 2015;13:24. doi: 10.1186/s12967-015-0383-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhao L, et al. Berberine improves glucogenesis and lipid metabolism in nonalcoholic fatty liver disease. BMC Endocr. Disord. 2017;17:13. doi: 10.1186/s12902-017-0165-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.D'Souza K, Nzirorera C, Kienesberger PC. Lipid metabolism and signaling in cardiac lipotoxicity. Biochim. Biophys. Acta. 2016;1861:1513–1524. doi: 10.1016/j.bbalip.2016.02.016. [DOI] [PubMed] [Google Scholar]
- 22.Sun Y, et al. Restoration of GLP-1 secretion by Berberine is associated with protection of colon enterocytes from mitochondrial overheating in diet-induced obese mice. Nutr. Diabetes. 2018;8:53. doi: 10.1038/s41387-018-0061-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gomes AP, et al. Berberine protects against high fat diet-induced dysfunction in muscle mitochondria by inducing SIRT1-dependent mitochondrial biogenesis. Biochim. Biophys. Acta. 2012;1822:185–195. doi: 10.1016/j.bbadis.2011.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Acehan D, et al. Cardiac and skeletal muscle defects in a mouse model of human Barth syndrome. J. Biol. Chem. 2011;286:899–908. doi: 10.1074/jbc.M110.171439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Soustek MS, et al. Characterization of a transgenic shRNA induced murine model of tafazzin deficiency. Hum. Gene Ther. 2010;22:865–871. doi: 10.1089/hum.2010.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kiebish MA, et al. Dysfunctional cardiac mitochondrial bioenergetic, lipidomic, and signaling in a murine model of Barth syndrome. J. Lipid Res. 2013;54:1312–1325. doi: 10.1194/jlr.M034728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cole LK, et al. Cardiolipin deficiency elevates susceptibility to a lipotoxic hypertrophic cardiomyopathy. J. Mol. Cell. Cardiol. 2020;144:24–34. doi: 10.1016/j.yjmcc.2020.05.001. [DOI] [PubMed] [Google Scholar]
- 28.Schlame M, et al. Phospholipid abnormalities in children with Barth syndrome. J. Am. Coll. Cardiol. 2003;42:1994–1999. doi: 10.1016/j.jacc.2003.06.015. [DOI] [PubMed] [Google Scholar]