Abstract
Hereditary Angioedema (HAE) is a rare genetic disease generally caused by deficiency or mutations in the C1-inhibitor gene, SERPING1, a member of the Serpin family. HAE results in acute attacks of edema, vasodilation, GI pain and hypotension. C1INH is a key inhibitor of enzymes controlling complement activation, fibrinolysis and the contact system. In HAE patients, contact system activation leads to uncontrolled production of bradykinin, the vasodilator responsible for the characteristic symptoms of HAE. In this study, we present the first physiological in vivo model to mimic acute HAE attacks. We evaluate hypotension, one of the many hallmark symptoms of acute HAE attacks using Serping1 deficient mice (serping1−/−) and implanted telemetry. Attacks were induced by IV injection of a silica nanoparticle (SiNP) suspension. Blood pressure was measured in real time, in conscious and untethered mice using implanted telemetry. SiNP injection induced a rapid, reversible decrease in blood pressure, in the presence of angiotensin converting enzyme (ACE) inhibition. We also demonstrate that an HAE therapeutic, ecallantide, can prevent HAE attacks in this model. The in vivo murine model described here can facilitate the understanding of acute HAE attacks, support drug development and ultimately contribute to improved patient care.
Subject terms: Experimental models of disease, Complement cascade
Introduction
The intricate coordination between coagulation and inflammation is the basis for a diverse set of disorders in humans. Hereditary angioedema (HAE[OMIM:106100]) was first described in the early 1800s as a familial form of angioedema, now known to have an incidence of 1:10,000–1:150,000 in the general population. Reports of HAE reflect that for more common forms, prevalence of disease is not affected by ethnicity or sex1–3. Patients with HAE typically have intermittent episodes of angioedema of the extremities, gastrointestinal tract, face, larynx or external genitalia, and hypotension2,4–9, due to vasodilation and increased vascular permeability. For the vast majority, the physiologic basis of HAE is low-functioning C1-inhibitor protein. A member of the SERPIN (serine protease inhibitor) superfamily, C1-inhibitor plays a crucial role in regulating contact system activation10–24. Unlike histamine-mediated acute allergic reactions, which are usually resolved within 24 h, HAE attacks are histamine-independent and can last for over 72 h25,26. HAE can be of multiple types including; type I (85% of cases) and type II HAE (14% of cases), which are generally associated with mutations in the C1INH gene, while HAE with normal C1INH (~ 1% of cases) is idiopathic and not associated with deficiency in levels or function of C1INH, however may be associated with mutations in the FXII gene27–30.
Serping1 deficient mice (serping−/−) have been used as a model to better understand the pathophysiology of HAE31–33. The readout in these studies was vascular permeability assessed with Evans Blue dye. The role of bradykinin in this model was verified by the dependence on expression of a bradykinin receptor. This model has been used to assess potential therapies; however, increased vascular permeability is a chronic change and the model does not reflect the acute attacks seen in HAE. An HAE animal model that better reflects acute systemic attacks like hypotension, would be useful in assessing both HAE prophylaxis and treatment.
To develop such a model, we considered stimuli of the contact system.
The contact system involves interaction between coagulation and the kallikrein-kinin cascades. In the physiological setting, activation of factor XII and the subsequent conversion of prekallikrein (PK) to kallikrein, stimulates cleavage of high molecular weight kininogen (HK), leading to generation of bradykinin (BK)34–41. BK has a short half-life (s) and is catabolized rapidly by carboxypeptidases including angiotensin-converting enzyme (ACE). As ACE plays a pivotal role in degrading bradykinin, ACE inhibitors (ACEi) have been implicated in angioedema, mainly for causing uncontrollable bradykinin generation42. While natural biological agents, like RNA43, misfolded proteins44, collagen45 and platelet polyphosphate46,47 can cause autoactivation of FXII48,49, a diverse array of biomaterial surfaces like glass50, dextran sulphate51 and silica nanoparticles (SiNPs)52 have also been shown to activate FXII53–58.
In addition to the selection of a driver of acute activation, an objective real time physiologic readout of an attack, such as hypotension, is also needed for a model of acute HAE attacks. Telemetry devices can be implanted in mice to allow for real time measurement of blood pressure59.
In this article, we present the first in vivo murine model to mimic acute HAE attacks. Attacks were induced by IV injection of a silica nanoparticle (SiNP) suspension60–64. Blood pressure was measured in real time, in conscious and untethered mice using implanted telemetry. SiNP injection induced a rapid, reversible decrease in blood pressure, in the presence of angiotensin converting enzyme (ACE) inhibition. The robust and reproducible murine model reported here will facilitate understanding and development of interventions to prevent and treat acute HAE attacks.
Methods
Animals
Male and female serping1−/− mice as well as wild-type C57BL6J mice (Age 4–6 months) were used. The serping1-deficient mouse was generated by Deltagen Inc. (San Mateo, CA), applying homologous recombination using ES cells. A Lac-Z/neo target vector was introduced to disrupt the serping1 (Gene#763, Serpin 1, Genbank Accession #Y10386 gi:1772997), using mouse ES cells derived from 129/Olahsd mouse sub strain (line #5961). Southern Blot was used to confirm correct targeting. serping1−/− and serping1+/+ genotypes were verified by PCR amplification by genomic DNA using primers spanning the LacZ/Neo insertion site (Supplemental Fig. S1) The resulting serping1−/+ males and females were backcrossed to C57BL6J to generate congenic strains (Charles River Laboratories). In the FDA vivarium, the serping1−/+ animals were interbred for more than 20 generations to obtain serping1−/− animals.
We backcrossed the Serping1−/− (129/Olahsd /C57Bl6J background) congenic mouse strain to wild type C57BL6/J (Stock#000664; Jax Laboratories) for 12 generations to generate the serping1−/− animals on a pure C57Bl6 background. The genotype and pedigree of the backcrossed mice were tested using the GigaMUGA platform (University of North Carolina at Chapel Hill). A western blot was used to verify the knockout animals lack expression of an intact C1 inhibitor protein (Supplemental Fig. S2). All experimental animals were maintained in accordance with the Institutional Animal Care and Use Committee (IACUC) and White Oak Consolidated Animal Program. The study was carried out in compliance with the ARRIVE guidelines.
Surgical implantation using wireless telemetry
Implantable transmitter HD-X11 (Data Sciences International, St. Paul, MN) is designed to simultaneously measure mean arterial pressure (MAP), temperature, locomotor activity and ECG at 10 s intervals allowing for accurate time-resolution of treatment-related effects. Animals were anesthetized with Isoflurane (by vaporizer—3–5% for induction, 1–3% for maintenance, nose cone). Buprenorphine SR (sustained release) Lab at 1 mg/kg was used subcutaneously on the day of the surgery. Because of its sustained release formulation, it can provide analgesic effect for ~ 48–72 h. post-surgery, thereby reducing handling of post-operative animals. The HD-X11 transmitters were implanted in the intraperitoneal cavity, with the fluid filled catheter inserted into the left carotid artery, according to manufacturer’s instructions. After securing the ECG leads on the body muscle, the skin was sutured using non-absorbable 5-O sutures. Post-operative animals were administered warmed Lactate Ringer solution (LRS). Mice were placed in individual cages, on heating pads, until the animals regained independent mobility. All procedures conducted on mice were approved by the IACUC of Division of Veterinary Services (DVS) at the Food and Drug Administration, Silver Spring, Maryland. Raw data collection and analysis were implemented using Dataquest ART (v. 4.3). A 7-day period of post-surgery data collection was performed to record baseline data. Later during experimental dosing, BP data were collected at 10 s intervals for 1 h preceding the intravenous injection and compared to MAP data collected during a 1-h post injection period (Supplemental data S3–S5).
Efficacy studies in mice
After 1 week of post-surgical recovery serping1−/− mice (3–4 animals/group) received 100 µl of 0.25 mg of SiNPs (Silica Nanospheres, mean diameter 50 nm, nanoComposix, San Diego, CA) or saline via bolus intravenous tail injections. The animals were pre-treated with angiotensin converting enzyme inhibitor (ACEi), Captopril (12.5 mg; Mylan, WV) in their drinking water (0.01 mg/ml), 24 h prior to injection65. Based on established values for mean daily water consumption66 each animal received an approximate captopril dose of 2.5 mg/kg/day. Individual animals were subjected to up to three separate interventions under different experimental conditions (e.g. SiNP, Captopril, saline, SiNP+ antagonist). Each intervention was separated by a 1-week recovery period.
A control group of C57BL6J females (Jax# 00064) was used to compare the effect of SiNPs and ecallantide (10 mg/ml, Dyax, Burlington,MA) exposing the wild-type (WT) females to same conditions as their serping1−/− female counterparts.
Effect size calculations
MAP was continuously monitored (every 10 s) via Dataquest ARTv4.3 for each animal from 30 min prior to experimental intervention (injection of SiNPs) until 60 min after injection. The first post-injection blood pressure reading was defined as t = 0. Baseline MAP was defined as the mean blood pressure from t = − 15 to t = − 10 min. For each animal, all post-intervention MAP values were expressed relative to the mean baseline MAP value. The relative MAP at time point n (RMAPn) was calculated as: MAPn/MAPbaseline.
The effect of SiNP on MAP was calculated at each time point by subtracting the MAP at t = 0 from the measured MAP. The net RMAP at time point n (Net RMAPn) was expressed as: Net RMAPn = RMAPn − RMAPt=0. The total effect of SiNP on blood pressure was calculated as the sum of Net RMAP values from t = 0 to 12 min. MAP values generally returned to baseline values by t = 12 min. Only those time points in which RMAPn was ≥ 10% below RMAPt=0 were included in this calculation in order to capture physiologically relevant effects.
Statistical analysis
Statistical analyses were conducted using GraphPad PRISM (8.0 GraphPad Software, San Diego, CA). All data were expressed as the Mean ± SD. Assessment of the differences between groups of treatments were performed using 2-way ANOVA (Analysis of Variance) and Bonferroni’s post-hoc tests comparing all group means. A P-value of < 0.05 was considered statistically significant. At least nine animals were evaluated for each experimental group; However, fewer animals were evaluated for some controls. Animal numbers are included in figure legends.
Results
SiNP injection models acute HAE attack in serping1−/− mice
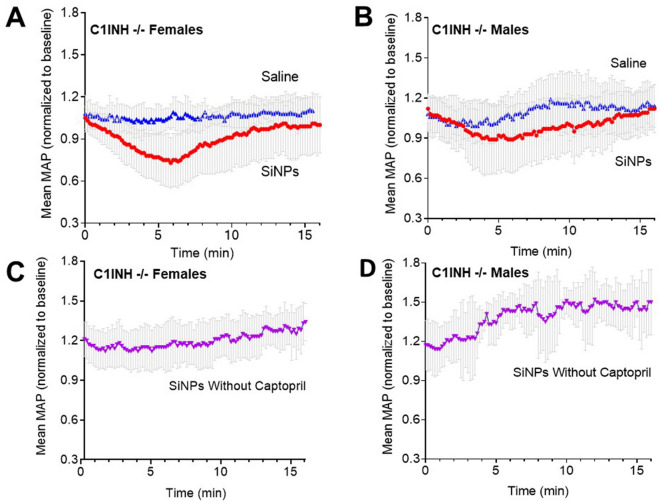
We chose silica nanoparticles (SiNPs) to simulate acute HAE attacks in our in vivo murine model as silica nanoparticles (SiNPs)52 have been shown to activate FXII53–58. A dose of 0.25 mg/100 µl of SiNPs was selected based upon published toxicity data in mice67,68. With ACEi captopril pretreatment69, intravenous SiNP injection induced a reversible decrease in blood pressure in serping1−/− females and males (Fig. 1A,B). When the same set of mice with captopril pretreatment were intravenously injected with saline, they failed to demonstrate any drop-in MAP (Fig. 1A,B). SiNP injections, in the absence of captopril pretreatment, did not lead to blood pressure drops in serping1−/− males or females (Fig. 1C,D); in fact, a blood pressure increase post-injection was observed. Examples of individual animal data are in the “Supplementary Materials S1” (Supplementary Figs. S3 and S4). In addition to decreased blood pressure, observation of captopril and SiNP treated animals revealed markedly decreased locomotor activity relative to captopril and saline treated controls (Supplementary Fig. S5). These observations reversed on the same time course as the measured blood pressure changes.
Figure 1.
In Vivo stimulation of the contact pathway with SiNPs in serping1−/− mice evokes acute HAE-like attacks in the presence of Captopril. Bolus IV injection of SiNPs (0.25 mg/100 µl) at t = 0 in captopril treated serping1−/− females; n = 18 (A) and males; n = 12 (B) evoked a rapid and reversible decrease in MAP. When serping1−/− females (n = 9) and males (n = 6) were injected with a single bolus IV injection of saline (vehicle), in the presence of captopril, no change in MAP was reported (A, B). Bolus IV injection of SiNPs (0.25 mg/100 µl) in the absence of captopril had no such effect on MAP in serping1−/− females; n = 6 (C) or males; n = 3 (D). Data points represent mean ± SD of normalized MAP.
The plasma kallikrein inhibitor ecallantide inhibits SiNP-induced blood pressure decrease
We used ecallantide (Kalbitor), an FDA-approved HAE therapy, to evaluate the model’s ability to respond to HAE therapeutics. Ecallantide (100 µg/100 μl) was injected immediately prior to SiNP injection in captopril-treated mice. Ecallantide blocked the SiNP induced MAP decrease in both captopril pretreated male and female serping1−/− mice (Fig. 2A,B) Silica vs Saline P = 0.002; Silica + Captopril vs Silica Without Captopril P = 0.007, and Silica vs Kalbitor, P = 0.002. The results of all the interventions were integrated over time as per the “Materials and methods” in Fig. 2C. Even though the results indicated a potential trend towards sex-based difference in response to SiNP, post-hoc testing did not indicate significant differences between the sexes (P = 0.87).
Figure 2.
Ecallantide (Kalbitor) ameliorates the SiNP-induced MAP decrease in captopril-treated Serping1−/− mice. (A, B) Kalbitor (100 µg), injected immediately prior to a bolus IV injection of SiNPs (0.25 mg/100 µl) at t = 0, inhibits the SiNP-induced decrease in mean MAP in both (A) serping1−/− females (n = 9) and (B) serping1−/− males (n = 9). (C) The histogram compares the total decrease in MAP (t = 0—12 min) in serping1−/− female and serping1−/− male mice as per the Materials and Methods, with four different interventions. Data are expressed as mean ± SD. Statistical analysis was performed using 2-way ANOVA with Bonferroni’s post-hoc tests SiNPs in the presence of captopril vs. saline in the presence of captopril *P = 0.002; SiNPs in the presence of captopril vs. SiNPs in the absence of Captopril **P = 0.007; SiNPs in the presence of Captopril vs SiNPs + Ecallantide in the presence of captopril **P = 0.002.
Wild-type females failed to show a significant effect of ecallantide treatment on captopril and SiNP-induced in blood pressure drops
Ecallantide inhibited the SiNP-induced MAP decrease in serping1−/− females (P = 0.01) but not in C57Bl6J females (P = 0.86) as shown in Fig. 3. Assessment of the differences between groups of treatments was performed using two-way Analysis of Variance (ANOVA) with Bonferroni’s post-hoc test. SiNP-induced blood pressure decreases were nominally smaller in WT C57Bl6J mice relative to serping1−/− animals. These data support the conclusion that serping1−/− mice pretreated with captopril and injected with SiNP are a more sensitive model for HAE-like attacks and treatments compared with WT mice under the same conditions.
Figure 3.

The Serping1−/− murine model is more sensitive than C57BL6 WT mice for assessing treatment of acute HAE-like attacks. Comparison of the integrated effect size, as per the Materials and Methods, of ecallantide treatment of SiNP-induced blood pressure drops in Serping1−/− (n = 18) and WTC57Bl6/J female mice (n = 9). There is significant amelioration of SiNP-induced MAP decrease with ecallantide treatment in Serping1−/− females (P = 0.01) as compared to the C57Bl6 females (NS, P = 0.86). Assessment of the differences between groups of treatments were performed using 2-way Analysis of Variance (ANOVA) with Bonferroni’s post-hoc test (used to determine the significance of mean MAP change of all groups against each other) with *P < 0.02; NS = non-significant. Data are expressed as mean ± SD.
Discussion
Hereditary angioedema is a rare genetic disease with significant mortality that is generally caused by mutations in the serping1 gene leading to either reduced levels (Type I HAE) or abnormal function (Type II HAE) of the C1-esterase inhibitor. Although there are murine models for HAE, there has not been an HAE animal model for acute attacks of angioedema. In this study, we describe a reliable and reproducible in vivo murine model that mimics acute HAE attacks. In this model, HAE-like attacks are successfully mitigated by the FDA-approved HAE therapeutic, ecallantide.
The role of bradykinin generation in HAE murine models has been demonstrated in earlier studies32. The importance of captopril, an ACE inhibitor, in this model enhances the role of bradykinin as ACE catabolizes bradykinin rapidly and ACE inhibitors can increase the levels and duration of kallikrein generated bradykinin70–72.
In humans, HAE attacks occur in the absence of ACE inhibitors69. The need for ACE inhibition in this model may reflect differences in bradykinin regulation between mice and humans. Human HAE occurs as an autosomal dominant genetic inheritance pattern and affected patients have residual levels of functional serping1. There does not seem to be a known syndrome with complete absence of serping1 in humans. Mice with complete loss of serping1 are viable and do not seem to have spontaneous attacks32. These mice may have additional regulation of kallikrein-mediated bradykinin generation and/or bradykinin degradation (Fig. 4), for example, non-ACE pathways of bradykinin clearance. There may also be other compensatory mechanisms. Of note, in the absence of ACE inhibition, the post-SiNP injection trend of increased blood pressure may reflect other regulatory systems in mice. Such differences may explain the need for ACE inhibition in this model.
Figure 4.
Contact system pathways with the kallikrein-bradykinin pathway and Serping1. Serping1 acts as the primary plasma inhibitor of the contact-kinin system cascades. In the absence of Serping1, FXIIa converts prekallikrein (PK) to kallikrein, which in turn cleaves high molecular weight kininogen (HK) and releases bradykinin (BK). Bradykinin is degraded by ACE and other plasma enzymes. The levels and duration of bradykinin in plasma impacts vascular permeability and vasodilation, leading to decreases in blood pressure and swelling; a hallmark for HAE. Ecallantide acts as a plasma kallikrein inhibitor and limits the production of bradykinin from HK.
A murine model of C1 inhibitor deficiency with a heterozygous genotype and increased vascular permeability despite has been developed73. This model used CRISPR‐Cas9 to generate a truncated protein rather than inserting another construct into the Serping1 gene as we and others32 have done. Our model targets exon 4 and allows for antigen expression but deletes an ~ 75 KDa C1 inhibitor reactive band compatible with glycosylated C1 inhibitor (Supplemental Fig. S2). We cannot rule out alternative splice variants and there is some MAP impact of our approach in WT mice; However, we do observe a difference between the KO mice and WT mice (Fig. 3).
The heterozygote model better mimics heterozygote human disease; However, the model did not exhibit spontaneous attacks. A rat model of HAE did have some spontaneous intestinal swelling; however, this model was based on bradykinin overexpression rather than Serping1 mutations. Directly mimicking all features of human disease is challenging as human disease phenotypes may be dependent on specific mutations74. as well as species-specific characteristics of the contact system. Our approach of using real-time MAP monitoring and an inducer of acute attacks, such as SiNP, could be applied across a range of HAE animal models and increase the utility of such models.
This model can also further explore sex-related effects in HAE with additional studies in larger numbers of mice, in both serping1−/− and WT animals. The evaluation of sex differences is important because female sex hormones, mainly estrogen, may impact bradykinin generation75–77. This robust in vivo model may also facilitate further study of the mechanisms that can induce acute HAE attacks. In summary, the serping1−/− murine model is the first model to evoke acute HAE-like attacks and can also be used to study approved HAE drugs (i.e. danacrine, icatibant, cinryze, lanadelumab)78–82, and support development of future targeted novel drug therapies for HAE prophylaxis.
Supplementary Information
Acknowledgements
We would like to gratefully acknowledge internal peer reviewers Marjorie Shapiro and Melanie Blank for their critical reading of the manuscript. We would like to thank Ashwinkumar Bhirde, Mate Tolnay, Christie Jane Fennell, Odile Engel Tzanko Stantchev and Carole Sourbier for their help and support. The authors thank John Dennis, Eric Nimako and DVS technical staff (Division of Veterinary Sciences, Silver Spring, FDA) for outstanding support with animal protocol and study.
Author contributions
S.B., M.W., and S.K. conceived the study. S.B., M.W., M.L., S.K., J.P., J.D. and Z.Z. designed experiments. S.B. and J.P. performed experiments. S.B. performed in vivo surgical manipulation. S.B. and M.W. implemented data extraction and normalization. M.L. advised and assisted with statistical interpretations. S.B., M.W., M.L. and S.K. wrote the manuscript. S.B., M.W., M.L., S.K., J.P., J.D. and Z.Z. reviewed the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-95125-0.
References
- 1.Osler W. Landmark publication from The American Journal of the Medical Sciences: Hereditary angio-neurotic oedema. 1888. Am. J. Med. Sci. 2010;339(2):175–178. doi: 10.1097/MAJ.0b013e3181b2803f. [DOI] [PubMed] [Google Scholar]
- 2.Nzeako UC, Frigas E, Tremaine WJ. Hereditary angioedema: A broad review for clinicians. Arch. Intern. Med. 2001;161(20):2417–2429. doi: 10.1001/archinte.161.20.2417. [DOI] [PubMed] [Google Scholar]
- 3.Cicardi M, Agostoni A. Hereditary angioedema. N. Engl. J. Med. 1996;334(25):1666–1667. doi: 10.1056/NEJM199606203342510. [DOI] [PubMed] [Google Scholar]
- 4.Ebo DG, Stevens WJ, Bosmans JL. An adverse reaction to angiotensin-converting enzyme inhibitors in a patient with neglected C1 esterase inhibitor deficiency. J. Allergy Clin. Immunol. 1997;99(3):425–426. doi: 10.1016/S0091-6749(97)70063-0. [DOI] [PubMed] [Google Scholar]
- 5.Hoyer C, Hill MR, Kaminski ER. Angio-oedema: An overview of differential diagnosis and clinical management. Continuing Educ. Anaesthesia Crit. Care Pain. 2012;12(6):307–311. doi: 10.1093/bjaceaccp/mks036. [DOI] [Google Scholar]
- 6.Johnston DT. Diagnosis and management of hereditary angioedema. J. Am. Osteopathic Assoc. 2011;111(1):28–36. [PubMed] [Google Scholar]
- 7.Longhurst HJ, Bork K. Hereditary angioedema: An update on causes, manifestations and treatment. Br. J. Hosp. Med. (Lond.). 2019;80(7):391–398. doi: 10.12968/hmed.2019.80.7.391. [DOI] [PubMed] [Google Scholar]
- 8.Zuraw BL, Bernstein JA, Lang DM, Craig T, Dreyfus D, Hsieh F, et al. A focused parameter update: Hereditary angioedema, acquired C1 inhibitor deficiency, and angiotensin-converting enzyme inhibitor-associated angioedema. J. Allergy Clin. Immunol. 2013;131(6):1491–1493. doi: 10.1016/j.jaci.2013.03.034. [DOI] [PubMed] [Google Scholar]
- 9.Kaplan AP. Angioedema. World Allergy Org. J. 2008;1(6):103–113. doi: 10.1097/WOX.0b013e31817aecbe. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Donaldson VH, Evans RR. A biochemical abnormality in hereditary angioneurotic edema. Am. J. Med. 1963;35(1):37–44. doi: 10.1016/0002-9343(63)90162-1. [DOI] [PubMed] [Google Scholar]
- 11.Kaplan AP, Joseph K. The bradykinin-forming cascade and its role in hereditary angioedema. Ann. Allergy Asthma Immunol. Off. Publ. Am. Coll. Allergy Asthma Immunol. 2010;104(3):193–204. doi: 10.1016/j.anai.2010.01.007. [DOI] [PubMed] [Google Scholar]
- 12.de Maat S, Bjorkqvist J, Suffritti C, Wiesenekker CP, Nagtegaal W, Koekman A, et al. Plasmin is a natural trigger for bradykinin production in patients with hereditary angioedema with factor XII mutations. J. Allergy Clin. Immunol. 2016;138(5):1414–1423.e9. doi: 10.1016/j.jaci.2016.02.021. [DOI] [PubMed] [Google Scholar]
- 13.Dewald G, Bork K. Missense mutations in the coagulation factor XII (Hageman factor) gene in hereditary angioedema with normal C1 inhibitor. Biochem. Biophys. Res. Commun. 2006;343(4):1286–1289. doi: 10.1016/j.bbrc.2006.03.092. [DOI] [PubMed] [Google Scholar]
- 14.Lucas A, Yaron JR, Zhang L, Ambadapadi S. Overview of serpins and their roles in biological systems. Methods Mol. Biol. (Clifton, NJ). 2018;1826:1–7. doi: 10.1007/978-1-4939-8645-3_1. [DOI] [PubMed] [Google Scholar]
- 15.Hofman ZL, Relan A, Zeerleder S, Drouet C, Zuraw B, Hack CE. Angioedema attacks in patients with hereditary angioedema: Local manifestations of a systemic activation process. J. Allergy Clin. Immunol. 2016;138(2):359–366. doi: 10.1016/j.jaci.2016.02.041. [DOI] [PubMed] [Google Scholar]
- 16.Bouillet L, Boccon-Gibod I, Massot C. Bradykinin mediated angioedema. Rev. Med. Interne. 2011;32(4):225–231. doi: 10.1016/j.revmed.2009.11.021. [DOI] [PubMed] [Google Scholar]
- 17.Agostoni A, Cicardi M. Hereditary and acquired C1-inhibitor deficiency: Biological and clinical characteristics in 235 patients. Medicine. 1992;71(4):206–215. doi: 10.1097/00005792-199207000-00003. [DOI] [PubMed] [Google Scholar]
- 18.Asmis LM, Sulzer I, Furlan M, Lammle B. Prekallikrein deficiency: The characteristic normalization of the severely prolonged aPTT following increased preincubation time is due to autoactivation of factor XII. Thromb. Res. 2002;105(6):463–470. doi: 10.1016/S0049-3848(02)00045-2. [DOI] [PubMed] [Google Scholar]
- 19.Cugno M, Nussberger J, Cicardi M, Agostoni A. Bradykinin and the pathophysiology of angioedema. Int. Immunopharmacol. 2003;3(3):311–317. doi: 10.1016/S1567-5769(02)00162-5. [DOI] [PubMed] [Google Scholar]
- 20.Nussberger J, Cugno M, Cicardi M. Bradykinin-mediated angioedema. N. Engl. J. Med. 2002;347(8):621–622. doi: 10.1056/NEJM200208223470820. [DOI] [PubMed] [Google Scholar]
- 21.Schmaier AH. Plasma prekallikrein: Its role in hereditary angioedema and health and disease. Front. Med. (Lausanne). 2018;5:3. doi: 10.3389/fmed.2018.00003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schmaier AH. The hereditary angioedema syndromes. J. Clin. Investig. 2019;129(1):66–68. doi: 10.1172/JCI125378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.De Maat S, Hofman ZLM, Maas C. Hereditary angioedema: The plasma contact system out of control. J. Thromb. Haemostasis JTH. 2018;16(9):1674–1685. doi: 10.1111/jth.14209. [DOI] [PubMed] [Google Scholar]
- 24.Nussberger J, Cugno M, Amstutz C, Cicardi M, Pellacani A, Agostoni A. Plasma bradykinin in angio-oedema. Lancet (London, England). 1998;351(9117):1693–1697. doi: 10.1016/S0140-6736(97)09137-X. [DOI] [PubMed] [Google Scholar]
- 25.Zuraw BL. The pathophysiology of hereditary angioedema. World Allergy Org. J. 2010;3(9 Suppl):S25–S28. doi: 10.1097/1939-4551-3-S3-S25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lara-Marquez ML, Christiansen SC, Riedl MA, Herschbach J, Zuraw BL. Threshold-stimulated kallikrein activity distinguishes bradykinin- from histamine-mediated angioedema. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2018;48(11):1429–1438. doi: 10.1111/cea.13219. [DOI] [PubMed] [Google Scholar]
- 27.Zuraw BL. Hereditary angioedema with normal C1 inhibitor: Four types and counting. J. Allergy Clin. Immunol. 2018;141(3):884–885. doi: 10.1016/j.jaci.2018.01.015. [DOI] [PubMed] [Google Scholar]
- 28.Ghannam A, Defendi F, Charignon D, Csopaki F, Favier B, Habib M, et al. Contact system activation in patients with HAE and normal C1 inhibitor function. Immunol. Allergy Clin. N. Am. 2013;33(4):513–533. doi: 10.1016/j.iac.2013.07.007. [DOI] [PubMed] [Google Scholar]
- 29.Magerl M, Germenis AE, Maas C, Maurer M. Hereditary angioedema with normal C1 inhibitor: Update on evaluation and treatment. Immunol. Allergy Clin. N. Am. 2017;37(3):571–584. doi: 10.1016/j.iac.2017.04.004. [DOI] [PubMed] [Google Scholar]
- 30.Bjorkqvist J, de Maat S, Lewandrowski U, Di Gennaro A, Oschatz C, Schonig K, et al. Defective glycosylation of coagulation factor XII underlies hereditary angioedema type III. J. Clin. Investig. 2015;125(8):3132–3146. doi: 10.1172/JCI77139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Davis AE., 3rd New treatments addressing the pathophysiology of hereditary angioedema. Clin. Mol. Allergy CMA. 2008;6:2. doi: 10.1186/1476-7961-6-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Han ED, MacFarlane RC, Mulligan AN, Scafidi J, Davis AE., 3rd Increased vascular permeability in C1 inhibitor-deficient mice mediated by the bradykinin type 2 receptor. J. Clin. Investig. 2002;109(8):1057–1063. doi: 10.1172/JCI200214211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shoemaker LR, Schurman SJ, Donaldson VH, Davis AE., 3rd Hereditary angioneurotic oedema: Characterization of plasma kinin and vascular permeability-enhancing activities. Clin. Exp. Immunol. 1994;95(1):22–28. doi: 10.1111/j.1365-2249.1994.tb06009.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Boulanger C, Schini VB, Moncada S, Vanhoutte PM. Stimulation of cyclic GMP production in cultured endothelial cells of the pig by bradykinin, adenosine diphosphate, calcium ionophore A23187 and nitric oxide. Br. J. Pharmacol. 1990;101(1):152–156. doi: 10.1111/j.1476-5381.1990.tb12105.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Colman RW, Schmaier AH. Contact system: A vascular biology modulator with anticoagulant, profibrinolytic, antiadhesive, and proinflammatory attributes. Blood. 1997;90(10):3819–3843. doi: 10.1182/blood.V90.10.3819. [DOI] [PubMed] [Google Scholar]
- 36.Dixon BS, Breckon R, Fortune J, Vavrek RJ, Stewart JM, Marzec-Calvert R, et al. Effects of kinins on cultured arterial smooth muscle. Am. J. Physiol. 1990;258(2 Pt 1):C299–308. doi: 10.1152/ajpcell.1990.258.2.C299. [DOI] [PubMed] [Google Scholar]
- 37.Parratt JR. Cardioprotection by angiotensin converting enzyme inhibitors—The experimental evidence. Cardiovasc. Res. 1994;28(2):183–189. doi: 10.1093/cvr/28.2.183. [DOI] [PubMed] [Google Scholar]
- 38.Schini VB, Boulanger C, Regoli D, Vanhoutte PM. Bradykinin stimulates the production of cyclic GMP via activation of B2 kinin receptors in cultured porcine aortic endothelial cells. J. Pharmacol. Exp. Therapeutics. 1990;252(2):581–585. [PubMed] [Google Scholar]
- 39.Wang J, Xiong W, Yang Z, Davis T, Dewey MJ, Chao J, et al. Human tissue kallikrein induces hypotension in transgenic mice. Hypertension (Dallas, Tex: 1979). 1994;23(2):236–243. doi: 10.1161/01.HYP.23.2.236. [DOI] [PubMed] [Google Scholar]
- 40.Wiemer G, Scholkens BA, Linz W. Endothelial protection by converting enzyme inhibitors. Cardiovasc. Res. 1994;28(2):166–172. doi: 10.1093/cvr/28.2.166. [DOI] [PubMed] [Google Scholar]
- 41.Schapira M, Scott CF, Colman RW. Protection of human plasma kallikrein from inactivation by C1 inhibitor and other protease inhibitors. The role of high molecular weight kininogen. Biochemistry. 1981;20(10):2738–2743. doi: 10.1021/bi00513a006. [DOI] [PubMed] [Google Scholar]
- 42.Bhoola KD, Figueroa CD, Worthy K. Bioregulation of kinins: Kallikreins, kininogens, and kininases. Pharmacol. Rev. 1992;44(1):1–80. [PubMed] [Google Scholar]
- 43.Kannemeier C, Shibamiya A, Nakazawa F, Trusheim H, Ruppert C, Markart P, et al. Extracellular RNA constitutes a natural procoagulant cofactor in blood coagulation. Proc. Natl. Acad. Sci. USA. 2007;104(15):6388–6393. doi: 10.1073/pnas.0608647104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Maas C, Govers-Riemslag JW, Bouma B, Schiks B, Hazenberg BP, Lokhorst HM, et al. Misfolded proteins activate factor XII in humans, leading to kallikrein formation without initiating coagulation. J. Clin. Investig. 2008;118(9):3208–3218. doi: 10.1172/JCI35424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.van der Meijden PE, Munnix IC, Auger JM, Govers-Riemslag JW, Cosemans JM, Kuijpers MJ, et al. Dual role of collagen in factor XII-dependent thrombus formation. Blood. 2009;114(4):881–890. doi: 10.1182/blood-2008-07-171066. [DOI] [PubMed] [Google Scholar]
- 46.Smith SA, Choi SH, Davis-Harrison R, Huyck J, Boettcher J, Rienstra CM, et al. Polyphosphate exerts differential effects on blood clotting, depending on polymer size. Blood. 2010;116(20):4353–4359. doi: 10.1182/blood-2010-01-266791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Smith SA, Mutch NJ, Baskar D, Rohloff P, Docampo R, Morrissey JH. Polyphosphate modulates blood coagulation and fibrinolysis. Proc. Natl. Acad. Sci. USA. 2006;103(4):903–908. doi: 10.1073/pnas.0507195103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Schmaier AH. The elusive physiologic role of Factor XII. J. Clin. Investig. 2008;118(9):3006–3009. doi: 10.1172/JCI36617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kaplan AP, Joseph K, Silverberg M. Pathways for bradykinin formation and inflammatory disease. J. Allergy Clin. Immunol. 2002;109(2):195–209. doi: 10.1067/mai.2002.121316. [DOI] [PubMed] [Google Scholar]
- 50.Rapaport SI, Aas K, Owren PA. The effect of glass upon the activity of the various plasma clotting factors. J. Clin. Investig. 1955;34(1):9–19. doi: 10.1172/JCI103067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Siebeck M, Cheronis JC, Fink E, Kohl J, Spies B, Spannagl M, et al. Dextran sulfate activates contact system and mediates arterial hypotension via B2 kinin receptors. J. Appl. Physiol. (Bethesda, Md: 1985). 1994;77(6):2675–2680. doi: 10.1152/jappl.1994.77.6.2675. [DOI] [PubMed] [Google Scholar]
- 52.Murugadoss S, Lison D, Godderis L, Van Den Brule S, Mast J, Brassinne F, et al. Toxicology of silica nanoparticles: An update. Arch. Toxicol. 2017;91(9):2967–3010. doi: 10.1007/s00204-017-1993-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Maas C, Renne T. Coagulation factor XII in thrombosis and inflammation. Blood. 2018;131(17):1903–1909. doi: 10.1182/blood-2017-04-569111. [DOI] [PubMed] [Google Scholar]
- 54.Wu Y. Contact pathway of coagulation and inflammation. Thromb. J. 2015;13:17. doi: 10.1186/s12959-015-0048-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gryshchuk V, Galagan N. Silica nanoparticles effects on blood coagulation proteins and platelets. J. Biochem. Res. Int. 2016;2016:6. doi: 10.1155/2016/2959414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kushida T, Saha K, Subramani C, Nandwana V, Rotello VM. Effect of nano-scale curvature on the intrinsic blood coagulation system. Nanoscale. 2014;6(23):14484–14487. doi: 10.1039/C4NR04128C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Maas C, Oschatz C, Renne T. The plasma contact system 2.0. Semin. Thromb. Hemost. 2011;37(4):375–381. doi: 10.1055/s-0031-1276586. [DOI] [PubMed] [Google Scholar]
- 58.Long AT, Kenne E, Jung R, Fuchs TA, Renne T. Contact system revisited: An interface between inflammation, coagulation, and innate immunity. J. Thromb. Haemostasis JTH. 2016;14(3):427–437. doi: 10.1111/jth.13235. [DOI] [PubMed] [Google Scholar]
- 59.Zhao X, Ho D, Gao S, Hong C, Vatner DE, Vatner SF. Arterial pressure monitoring in mice. Curr. Protocols Mouse Biol. 2011;1:105–122. doi: 10.1002/9780470942390.mo100149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Levy JH, O'Donnell PS. The therapeutic potential of a kallikrein inhibitor for treating hereditary angioedema. Expert Opin. Investig. Drugs. 2006;15(9):1077–1090. doi: 10.1517/13543784.15.9.1077. [DOI] [PubMed] [Google Scholar]
- 61.Schneider L, Lumry W, Vegh A, Williams AH, Schmalbach T. Critical role of kallikrein in hereditary angioedema pathogenesis: A clinical trial of ecallantide, a novel kallikrein inhibitor. J. Allergy Clin. Immunol. 2007;120(2):416–422. doi: 10.1016/j.jaci.2007.04.028. [DOI] [PubMed] [Google Scholar]
- 62.Williams A, Baird LG. DX-88 and HAE: A developmental perspective. Transfusion Apheresis Sci. Off. J. World Apheresis Assoc. Off. J. Eur. Soc. Haemapheresis. 2003;29(3):255–258. doi: 10.1016/S1473-0502(03)00170-8. [DOI] [PubMed] [Google Scholar]
- 63.Riedl M. Hereditary angioedema therapy: Kallikrein inhibition and bradykinin receptor antagonism. World Allergy Org. J. 2010;3(9 Suppl):S34–S38. doi: 10.1186/1939-4551-3-S3-S34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lewis LM, Graffeo C, Crosley P, Klausner HA, Clark CL, Frank A, et al. Ecallantide for the acute treatment of angiotensin-converting enzyme inhibitor-induced angioedema: A multicenter, randomized, controlled trial. Ann. Emerg. Med. 2015;65(2):204–213. doi: 10.1016/j.annemergmed.2014.07.014. [DOI] [PubMed] [Google Scholar]
- 65.Emanueli C, Angioni GR, Anania V, Spissu A, Madeddu P. Blood pressure responses to acute or chronic captopril in mice with disruption of bradykinin B2-receptor gene. J. Hypertens. 1997;15(12 Pt 2):1701–1706. doi: 10.1097/00004872-199715120-00075. [DOI] [PubMed] [Google Scholar]
- 66.Sweet DV. Registry of toxic effects of chemical substances (RTECS): Comprehensive guide to the RTECS: US Department of Health and Human Services, Public Health Service, Centers (1997). https://www.cdc.gov/niosh/docs/97-119/pdfs/97-119.pdf. Accessed July 26, 2021.
- 67.Chan WT, Liu CC, Chiang Chiau JS, Tsai ST, Liang CK, Cheng ML, et al. In vivo toxicologic study of larger silica nanoparticles in mice. Int. J. Nanomed. 2017;12:3421–3432. doi: 10.2147/IJN.S126823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Cho M, Cho WS, Choi M, Kim SJ, Han BS, Kim SH, et al. The impact of size on tissue distribution and elimination by single intravenous injection of silica nanoparticles. Toxicol. Lett. 2009;189(3):177–183. doi: 10.1016/j.toxlet.2009.04.017. [DOI] [PubMed] [Google Scholar]
- 69.Agostoni A, Cicardi M. Contraindications to the use of ace inhibitors in patients with C1 esterase inhibitor deficiency. Am. J. Med. 1991;90(2):278. doi: 10.1016/0002-9343(91)80179-P. [DOI] [PubMed] [Google Scholar]
- 70.Gainer JV, Morrow JD, Loveland A, King DJ, Brown NJ. Effect of bradykinin-receptor blockade on the response to angiotensin-converting-enzyme inhibitor in normotensive and hypertensive subjects. N. Engl. J. Med. 1998;339(18):1285–1292. doi: 10.1056/NEJM199810293391804. [DOI] [PubMed] [Google Scholar]
- 71.Pretorius M, Rosenbaum D, Vaughan DE, Brown NJ. Angiotensin-converting enzyme inhibition increases human vascular tissue-type plasminogen activator release through endogenous bradykinin. Circulation. 2003;107(4):579–585. doi: 10.1161/01.CIR.0000046268.59922.A4. [DOI] [PubMed] [Google Scholar]
- 72.Straka BT, Ramirez CE, Byrd JB, Stone E, Woodard-Grice A, Nian H, et al. Effect of bradykinin receptor antagonism on ACE inhibitor-associated angioedema. J. Allergy Clin. Immunol. 2017;140(1):242–248.e2. doi: 10.1016/j.jaci.2016.09.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Qiu T, Chiuchiolo MJ, Whaley AS, Russo AR, Sondhi D, Kaminsky SM, et al. Gene therapy for C1 esterase inhibitor deficiency in a Murine Model of Hereditary angioedema. Allergy. 2019;74(6):1081–1089. doi: 10.1111/all.13582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Caccia S, Suffritti C, Carzaniga T, Berardelli R, Berra S, Martorana V, et al. Intermittent C1-inhibitor deficiency associated with recessive inheritance: functional and structural insight. Sci. Rep. 2018;8(1):977. doi: 10.1038/s41598-017-16667-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Banerji A, Riedl M. Managing the female patient with hereditary angioedema. Womens Health (Lond. Engl.) 2016;12(3):351–361. doi: 10.2217/whe.16.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Bouillet L. Hereditary angioedema in women. Allergy Asthma Clin. Immunol. 2010;6(1):17. doi: 10.1186/1710-1492-6-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Bouillet L, Gompel A. Hereditary angioedema in women: Specific challenges. Immunol. Allergy Clin. N. Am. 2013;33(4):505–511. doi: 10.1016/j.iac.2013.07.006. [DOI] [PubMed] [Google Scholar]
- 78.Banerji A, Riedl MA, Bernstein JA, Cicardi M, Longhurst HJ, Zuraw BL, et al. Effect of lanadelumab compared with placebo on prevention of hereditary angioedema attacks: A randomized clinical trial. JAMA. 2018;320(20):2108–2121. doi: 10.1001/jama.2018.16773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Bernstein JA, Manning ME, Li H, White MV, Baker J, Lumry WR, et al. Escalating doses of C1 esterase inhibitor (CINRYZE) for prophylaxis in patients with hereditary angioedema. J. Allergy Clin. Immunol. Pract. 2014;2(1):77–84. doi: 10.1016/j.jaip.2013.09.008. [DOI] [PubMed] [Google Scholar]
- 80.Bork K, Bygum A, Hardt J. Benefits and risks of danazol in hereditary angioedema: A long-term survey of 118 patients. Ann. Allergy Asthma Immunol. Off. Publ. Am. Coll. Allergy Asthma Immunol. 2008;100(2):153–161. doi: 10.1016/S1081-1206(10)60424-3. [DOI] [PubMed] [Google Scholar]
- 81.Bouillet L. Icatibant in hereditary angioedema: News and challenges. Expert Rev. Clin. Immunol. 2011;7(3):267–272. doi: 10.1586/eci.11.16. [DOI] [PubMed] [Google Scholar]
- 82.Sexton DJ, Kenniston JA, Faucette RR, Nixon AE, TenHoor C, Chyung Y, et al. Comparison of plasma kallikrein inhibition by the endogenous C1-inhibitor versus DX-2930, a monoclonal antibody inhibitor. Blood. 2013;122(21):1066. doi: 10.1182/blood.V122.21.1066.1066. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.