Abstract
Aims
Coronavirus disease 2019 (COVID-19) is a novel viral infection threatening worldwide health as currently there exists no effective treatment strategy and vaccination programs are not publicly available yet. T lymphocytes play an important role in antiviral defenses. However, T cell frequency and functionality may be affected during the disease.
Material and methods
Total blood samples were collected from patients with mild and severe COVID-19, and the total lymphocyte number, as well as CD4+ and CD8 + T cells were assessed using flowcytometry. Besides, the expression of exhausted T cell markers was evaluated. The levels of proinflammatory cytokines were also investigated in the serum of all patients using enzyme-linked immunesorbent assay (ELISA). Finally, the obtained results were analyzed along with laboratory serological reports.
Results
COVID-19 patients showed lymphopenia and reduced CD4+ and CD8 + T cells, as well as high percentage of PD-1 expression by T cells, especially in severe cases. Serum secretion of TNF-α, IL-1β, and IL-2 receptor (IL-2R) were remarkably increased in patients with severe symptoms, as compared with healthy controls. Moreover, high levels of triglyceride (TG) and low density lipoprotein cholesterol (LDL-C), were correlated with the severity of the disease.
Conclusion
Reduced number and function of T cells were observed in COVID-19 patients, especially in severe patients. Meanwhile, the secretion of proinflammatory cytokines was increased as the disease developed. High level of serum IL-2R was also considered as a sign of lymphopenia. Additionally, hypercholesterolemia and hyperlipidemia could be important prognostic factors in determining the severity of the infection.
Keywords: COVID-19, T cells, Exhaustion, Inflammation, Cytokine
1. Introduction
Coronavirus disease 2019 (COVID-19) was officially described as pandemic since it distributed all over the world from December 2019. The virus has also been known as SARS-COV-2 due to possessing similar characteristics as SARS-COV, and belongs to β-lineage coronaviruses (Bordoni et al., 2020; Soltani-Zangbar et al., 2021). COVID-19 manifestation varies from asymptomatic to serious clinical complications, such as acute respiratory distress syndrome (ARDS) or even death. So far, there exists no effective treatment strategy; therefore, the infected areas are unable to manage the outbreak effectively (Etemadi et al., 2021; Liao et al., 2020).
Regarding efficacious immune responses against viral infections which mainly depend on T cell activation, improving the function and number of T cells is vital for a successful recovery in COVID-19 patients (Braun et al., 2020; Chen et al., 2020). CD8+ cytotoxic T cells (CTLs) secret antiviral mediators, including perforin and granzymes, to kill virus-infected cells (Mescher et al., 2006). CD4+ helper T (Th) cells assist CTLs and B cells through secreting cytokines in order to improve their ability for pathogen clearance (Mullen et al., 2002). However, in prolonged viral infections, T cells experience a exhaustion state by the over expression of inhibitory signaling molecules, such as programmed death 1(PD-1), preventing the function of virus-specific T-cell effectors and the subsequent viral clearance (Fenwick et al., 2019). On the other side, from an immunological and pathogenic perspective cytokines interleukin-1 (IL-1) and IL-6 play a substantial role in initiating and maintaining the inflammatory response (Bhimraj et al., 2020; Shen et al., 2020). It has been shown that virally-induced cytokine secretion of tumor necrosis factor (TNF), IL-6, IL-1 and IL-2 can eventuate in adverse outcomes including interrupted air exchange, pulmonary edema and cardiac injury leading to death, unless treatment in appropriate time of infection (Mehra and Ruschitzka, 2020; Xu et al., 2020b). However, TNF-α enjoys the capability of promoting T cell apoptosis through possessing interactions with its receptor, TNFR1, which shows increased expression in aged T cells (Aggarwal et al., 1999; Gupta et al., 2005). IL-2 is also essential for the function, differentiation, and proliferation of T cells; however, soluble IL-2 receptor (sIL-2R), namely CD25, is also capable of inhibiting the function and proliferation of T cells (Ross and Cantrell, 2018; Zhang et al., 2020).
Despite the fact that numerous studies have focused on T cell changes during COVID-19 and have highlighted the differences of T cell characteristics in mild and severe patients, our knowledge of the factors contributing to the progression of the disease is limited. Besides, the serological features, including hyperglycerolemia and hyperlipidemia, may provide valuable information which could serve as prognostic factors. Therefore, the aim of this study was to compare T cell traits in patients with mild and severe COVID-19, as well as finding immunological prognostic factors related to the serological features.
2. Materials and methods
2.1. Patients
One hundred adult patients with approved COVID-19 disease, who were admitted into the general hospital of Imam Reza at Tabriz University of Medical Sciences, Tabriz, Iran, from May until August 2020, were enrolled in this study. Fifty healthy individuals (aged 52.8 ± 15.32) referring to the hospital for routine physical examination were also considered as controls. Total patients involved in the experiment were classified into two groups based on their symptoms, which included mild and severe (aged 51.1 ± 15.2 for severe cases and 52.2 ± 12.46 for the mild ones). The number of participants was roughly equal in gender. Patients were categorized according to their severe and mild condition based on their general condition, hospitalization time, and severity of lung involvement. Patients who had severe lung involvement and were hospitalized more than one week were included in the group of SARS-CoV-2 patients with severe condition. Demographic data, medical history and main laboratory findings from patients and healthy individuals were recorded. Unfortunately, the ethnicity of the participants is not available. Patient hospitalization was performed between 4–7 days after symptom onset, and peripheral blood samples were taken on the first day of hospitalization. These samples were used for both clinical and immunological assays. Patients received routine treatments employed for COVID-19 patients, such as anti-viral drugs and corticosteroids. All subjects were analyzed by routine serological tests, and COVID-19 infection was confirmed by the positive result of throat swab samples assessed through SARS-COV2 real-time polymerase chain reaction (RT-PCR) and chest CT scan. The approval to this study was provided by Ethics committee of Tabriz University of Medical Sciences (IR.TBZMED.REC.1399.017). Written informed consent was obtained by the Ethics Commission of the designated hospital for emerging infectious diseases.
2.2. Sample collection and flowcytometric analysis
Peripheral blood sample was collected from both patients and healthy volunteers, which was then supplemented with anticoagulants (EDTA-K2) and stained with fluorescent labeled antibodies to analyze the number and frequency of total lymphocytes, namely CD4+ and CD8 + T cells. The detection of T cells was performed through employing human CD4-FITC (eBioscience, San Diego, CA, U.S.A), CD8-PE (eBioscience, San Diego, CA, U.S.A), and PD-1-APC (eBioscience, San Diego, CA, U.S.A) antibodies. Following staining, the cells were evaluated by FACS via flow cytometry Analyzer (BD Biosciences), where the data represented the mean of duplicate measurements. All the procedures of the experiments were performed under the biosafety level II plus condition.
2.3. Quantification of plasma cytokine levels
Plasma levels of IL-1β, TNF-α, and soluble IL-2R were quantified using ELISA kits (Mybiosource, San Diego, USA), according to the manufacturer’s instruction. Coating the wells of micro-titer ELISA plate (Maxisorp, Nunc, Roskilde, Denmark) took place at 4 °C overnight with anti-IL-1β, anti-TNF-α, and anti-sIL-2R capture antibodies (in 100 mM Na2HPO4 buffer, pH 9.0). The blockade of these was then performed by phosphate buffered-saline (PBS, pH 7.4) which contained 0.05 % Tween-20 and 10 % FBS. Next, the plasma of patients and healthy controls were added in triplicates. The process of washing of the wells was performed with wash buffer (PBS + 0.05 % [v/v] Tween-20) after 2 h of incubation at 25 °C. Then, biotinylated mouse anti-hIL-1β mAb, anti-hTNF-α mAb, or anti-hsIL-2R mAb (each 1:10 dilution in 1% BSA/PBS) were added to specified wells and the plate was incubated at room temperature for 1 h. Thereafter, they were gently washed with wash buffer, and the existence of anti-cytokine antibody was examined through the addition of a streptavidin alkaline phosphatase conjugated anti-mouse IgG Ab (Sigma) (diluted 1:300 in Tris-Buffered Saline (TBS, pH 7.6). After 20 min of incubation at 25 °C and a gentle rinsing with wash buffer, p-nitrophenyl phosphate (4 mg/mL) was added to each well as substrate and incubated for 30 min at 25 °C. Finally, the evaluation of absorbance was performed for each well at 405 nm in an ELISA plate reader (LabSystems, Helsinki, Finland). Wells with serial amounts of each cytokine, as kit-provided standards, were also run in parallel, which enabled quantifying the level of each cytokine in a given sample by the standard curves and regression analysis of mean absorbance. Data in scatter plots depicted the mean of two technical replicates.
2.4. Statistical analysis
Inter-group comparisons were done using Brown-Forsythe and welch ANOVA Dunnett's T3 multiple comparisons test for quantitative data, and Chi-square test for qualitative data. All data are reported as mean ± SD, for which P < 0.05 was considered as statistically significant. GraphPad Prism version 8.0 (GraphPad Software, Inc., San Diego, CA, USA) was utilized to perform the statistical analyses.
3. Results
3.1. Patient demographics and laboratory findings
A total of 100 (50 milds and 50 severe) patients with SARS-CoV-2, and 50 healthy controls, with matched age and gender were included in this study. The body mass index (BMI) was not notably different between the three groups. According to the demographic data shown in Table 1 , a significant difference was observed in fasting blood sugar (FBS) level between mild (118.3 ± 31.25 mg/dl versus 104.9 ± 23.11 mg/dl, p = 0.0496) and severe (138.9 ± 49.95 versus 104.9 ± 23.11 mg/dl, p = 0.0001) COVID-19 patients, as compared to healthy controls. Moreover, the comorbidity of COVID-19 and diabetes was significant, not only in the mild group, but also in severe patients compared with healthy ones. Additionally, serum triglyceride (TG) levels were meaningfully higher in severe patients, as compared to healthy controls (186.5 ± 55.31 mg/dl versus 142.0 ± 55.99 mg/dl, p = 0.0004). Low density lipoprotein-cholesterol (LDL-C) level was significantly higher in severe COVID-19 patients compared to normal cases and patients with mild condition (144.9 ± 27.97 mg/dl versus 120.6 ± 31.52 mg/dl and 131.9 ± 24.34, p = 0.0003 and p = 0.0434, respectively). No statistically meaningful difference was observed regarding the total cholesterol and high-density lipoprotein cholesterol (HDL-C) levels between the three groups.
Table 1.
Demographics and laboratory findings of COVID-19 patients and healthy controls.
| Parameter | G1 mean ± SD (N = 50) | G2 mean ± SD (N = 50) | G3 mean ± SD (N = 50) | Significance Level |
||
|---|---|---|---|---|---|---|
| G1 vs G2 | G1 vs G3 | G2 vs G3 | ||||
| Age (Male-Female) |
52.8 ± 15.32 (24−26) |
51.1 ± 15.2 (23−27) |
52.2 ± 12.46 (28−22) |
NS | NS | NS |
| BMI (kg/m2) | 26.29 ± 3.273 | 27.1 ± 5.261 | 27.98 ± 4.564 | NS | NS | NS |
| SBP (mmHg) | 112.8 ± 11.73 | 114.5 ± 12 | 122.6 ± 19.05 | NS | 0.0078 | 0.0390 |
| DBP (mmHg) | 72.38 ± 8.588 | 72.52 ± 8.596 | 73.42 ± 12.05 | NS | NS | NS |
| FBS (mg/dl) | 104.9 ± 23.11 | 118.3 ± 31.25 | 138.9 ± 49.95 | 0.0496 | 0.0001 | 0.0455 |
| TG (mg/dl) | 142.0 ± 55.99 | 161.8 ± 62.33 | 186.5 ± 55.31 | NS | 0.0004 | NS |
| Cholesterol (mg/dl) | 180.8 ± 42.39 | 188 ± 40.3 | 196.6 ± 34.96 | NS | NS | NS |
| HDL (mg/dl) | 44.04 ± 8.877 | 44 ± 8.52 | 42.49 ± 7.153 | NS | NS | NS |
| LDL (mg/dl) | 120.6 ± 31.52 | 131. 9 ± 24.34 | 144.9 ± 27.97 | NS | 0.0003 | 0.0434 |
| Albumin (g/dl) | 3.461 ± 0.2154 | 3.367 ± 0.3766 | 3.251 ± 0.2881 | NS | 0.0003 | NS |
| Creatinine (mg/dl) | 1.554 ± 1.043 | 1.522±.845 | 1.450 ± 0.7022 | NS | NS | NS |
| GFR (cc/min) | 77.77 ± 24.11 | 70.84 ± 25.14 | 67.47 ± 22.71 | NS | NS | NS |
| Diabetic Patients | 7 | 9 | 14 | NS | 0.00854 | 0.0192 |
| Clinically COVID-19 Positive Subjects | – | 50 | 50 | |||
Data are presented as mean ± standard devision. P < 0.05 was considered as statistically significant. G1: Healthy Controls; G2: Patients with mild symptoms; G3: Patients with severe symptoms; BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; FBS: fasting blood sugar; TG: triglyceride; HDL: high density lipoprotein; LDL: low density lipoprotein; GFR: glomerular filtration rate.
3.2. The correlation between the exhaustion and reduction of T cells and the severity of COVID-19
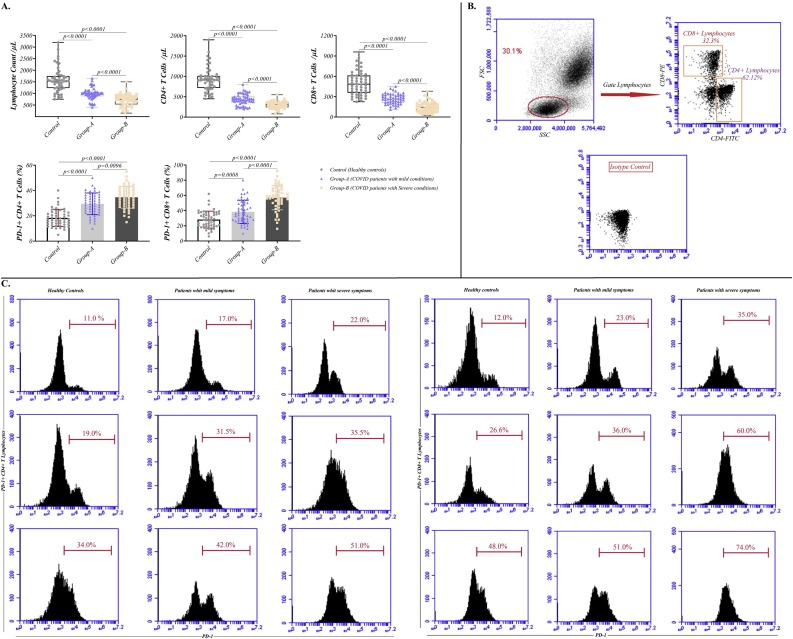
We examined the number and functional features of T cells in whole blood of the individuals. As illustrated in Fig. 1 , the absolute number of lymphocytes was markedly decreased in patients with mild and severe SARS-CoV-2, in comparison to controls (981.1 ± 257.1 and 724.4 ± 258.1 cell/μl versus 1579 ± 530 cell/μl, p < 0.0001 and p < 0.0001, respectively). Additionally, the volume of CD4+T and CD8+T cells was significantly decreased during the infection, such that it was considerably related to the severity of the disease. As shown in Fig. 1, the numbers of CD4 + T lymphocytes were 431.3 ± 134.4 and 301.2 ± 87.21 cell/μl in patients with mild and severe conditions, respectively, compared to 928.4 ± 299 cell/μl in healthy controls (p < 0.0001 for both of them). The same pattern was observed for CD8 + T lymphocytes (268.2 ± 75.97 and 149 ± 76.84 cell/μl versus 500.3 ± 175.3 cell/μl, p < 0.0001 for both of them) (Fig. 1A and Table 2 ).
Fig. 1.
Reduced and exhausted T lymphocytes during COVID-19 infection. (A) The incidence of total lymphocyte and T cell subsets in patients with mild and severe COVID-19. (B) Dot blot of lymphocytes gated to identify CD4+ and CD8 + T cells under isotype control. (C) Dot blot of CD4+ and CD8 + T lymphocytes expressed PD-1 in mild and severe COVID-19 infected patients versus healthy controls.
Table 2.
T cells number and PD-1 expression of patients with COVID-19 and healthy subjects.
| Target | G1 mean ± SD (N = 50) | G2 mean ± SD (N = 50) | G3 mean ± SD (N = 50) | P value |
||
|---|---|---|---|---|---|---|
| 1 vs 2 | 1 vs 3 | 2 vs 3 | ||||
| Flow Cytometery | ||||||
| Total Lymphocytes (cell/μl) | 1579 ± 530 | 981.1 ± 257.1 | 724.4 ± 258.1 | <0.0001 | <0.0001 | <0.0001 |
| CD4 + T Lymphocytes (cell/μl) | 928.4 ± 299 | 431.3 ± 134.4 | 301.2 ± 87.21 | <0.0001 | <0.0001 | <0.0001 |
| CD8 + T Lymphocytes (cell/μl) | 500.3 ± 175.3 | 268.2 ± 75.97 | 149 ± 76.84 | <0.0001 | <0.0001 | <0.0001 |
| PD-1+ CD4 + T Lymphocytes (%) | 18.35 ± 6.840 | 29.47 ± 8.595 | 34.66 ± 8.584 | <0.0001 | <0.0001 | 0.0096 |
| PD-1+ CD8 + T Lymphocytes (%) | 28.19 ± 11.11 | 38.34 ± 15.38 | 57.02 ± 15.96 | 0.0008 | <0.0001 | <0.0001 |
| ELISA | ||||||
| IL-2R (U/mL) | 500.5 ± 158.9 | 622.0 ± 161.8 | 879.6 ± 213.6 | 0.0008 | <0.0001 | <0.0001 |
| TNF-α (pg/mL) | 37.59 ± 16.34 | 52.60 ± 25.14 | 70.80 ± 30.43 | 0.0020 | <0.0001 | 0.0046 |
| IL-1β (pg/mL) | 80.84 ± 45.29 | 107.2 ± 37.46 | 139.8 ± 54.04 | 0.0061 | <0.0001 | 0.0022 |
Data are presented as mean ± standard devision. P < 0.05 was considered as statistically significant. G1: Healthy Controls; G2: Patients with mild symptoms; G3: Patients with severe symptoms; IL: interleukin; TNF-α: tumor necrosis factor-α, PD-1: programed cell death protein 1.
The expression of PD-1 was also increased in both CD4+T cells and CD8+T cells, indicating the exhaustion process in these cells. The percentage of PD-1+CD4+T cells in healthy subjects, mild infected cases, and severe patients was 18.35 ± 6.840 %, 29.47 ± 8.595 % and 34.66 ± 8.584 %, respectively (the p value for COVID-19 infected patients with both mild and severe symptoms compared to healthy controls was considered <0.0001). Similarly, the percentage of PD-1+CD8+T cells was 28.19 ± 11.11 %, 38.34 ± 15.38 %, and 57.02 ± 15.96 %, respectively. The p values in patients with mild and severe condition, compared to healthy controls, were considered 0.0008 and <0.0001, respectively).
3.3. Detecting high levels of inflammatory cytokines in patients’ plasma
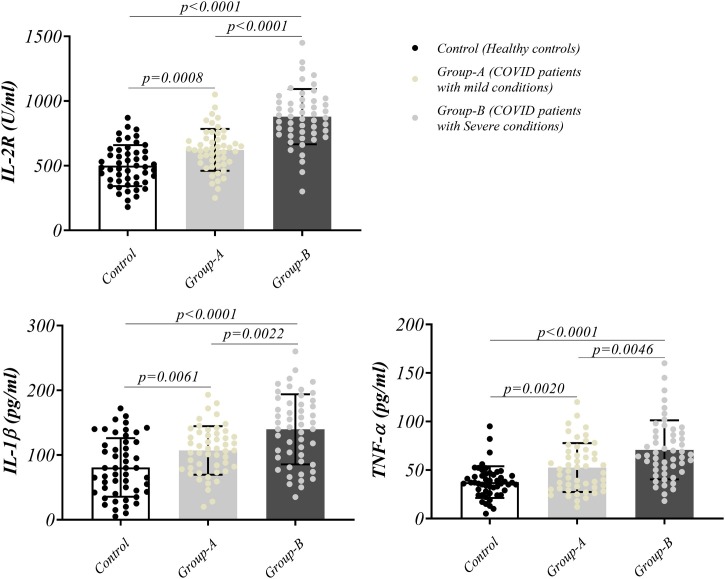
The plasma level of substantial secretory mediators related to the cytokine storm in patients and controls was also measured, and is illustrated in Fig. 2 . We found that the levels of IL-2R, IL-1β, and TNF-α were remarkably higher in severe and mild patients, as compared with the controls (p values for IL-2R: 0.0008 and <0.0001, IL-1β: 0.0061 and <0.0001, and TNF-α: 0.0020 and <0.0001 in patients with mild and severe symptoms compared to healthy controls, respectively). Moreover, these mediator levels were significantly higher in severe cases versus mild patients (Fig. 2 and Table 2).
Fig. 2.
Inflammatory cytokine secretion and soluble IL-2 receptor. The secretion of IL-2 receptor and proinflammatory cytokines, IL-1β and TNF-α in mild (group-A) and severe (group-B) COVID-19 patients.
4. Discussion
T cells are known as the main antiviral weapons which are equipped with two arms, CD8+ cytotoxic T cells (CTLs) that secrete antiviral mediators (Mescher et al., 2006) and CD4+ helper T (Th) cells that assist CTLs and B cells in improving their ability for pathogen clearance (Mullen et al., 2002). However, prolonged infection may lead to T cell exhaustion (Fenwick et al., 2019; Ng et al., 2013), which is a dysfunctional state of T cells characterized by the high expression level of immune checkpoint receptors such as PD-1, decreased proliferation and production of cytotoxic cytokines, and altered transcriptional and metabolic profiles (Wherry, 2011). In spite of the previous studies on COVID-19 concerning the reduction of number and functionality of lymphocytes, the correlation of these features with the severity of the disease, and whether serum metabolic changes affect T cell responses, have remained ambiguous. In this study, we described the overall serum changes on T cell compartments, and explained the correlation of serum metabolic status with T lymphocyte functionality and the severity of COVID-19 infection. To this end, the whole blood of patients with mild and severe COVID-19 was implicated to analyze the frequency and functionality of T lymphocytes in comparison to healthy controls.
Our findings showed that total lymphocytes decreased intensively in 100 COVID-19 patients, and especially in severe cases, and that the rate of T cell depletion was significant both for CD4+ and CD8 + T cell subsets. An earlier report conducted by Bo Diao et al. showed that 70.56 % of non-severe patients had reduced CD4+ and CD8 + T cells. However, 95 % of patients with severe signs displayed CD4 + T cell reduction, and all of them showed CD8 + T cell depletion (Diao et al., 2020). In parallel with this study, we observed that the decline in CD4+ and CD8 + T cell subsets in severe individuals was much higher than that of mild cases. However, how lymphopenia is developed remains unknown. Low levels of angiotensin-converting enzyme 2 (ACE2), the cell entry receptor for SARS-CoV-2 (Xu et al., 2020a; Zhou et al., 2020), are expressed by lymphocytes. Also, genome of the virus could hardly be detected in the peripheral blood of SARS-CoV-2 infected patients (Wölfel et al., 2020). Therefore, speculating that reduced peripheral lymphocyte is not directly related to SARS-CoV-2 infection is reasonable. However, it is assumed that such phenomenon results from the activation of induced cell death or T cell trafficking to the lung, where robust replication of the virus takes place (Song et al., 2020). Future studies are therefore expected to elucidate the underlying mechanisms of lymphopenia.
As opposed to their decreased absolute count, T cells show an increase in the expression of T cell inhibitory molecules. Moreover, we identified a broad range of PD-1 over expression in CD4+ and CD8 + T cell subsets in infected patients, and especially in severe cases, emphasizing limited T cell functions as a result of exhaustion during SARS-CoV-2 infection (Zheng et al., 2020). Bellesi et al. also outlined higher expression of CD95 and PD-1 in old patients compared to inpatients, with a trend for PD-1 expression in CD8+cells (Bellesi et al., 2020). In line with this finding, our observations showed that in severe COVID-19 patients, the percentage of cycling peripheral blood PD-1+ CD8 + T cells (57.02 %±15.96) was higher than PD-1+ CD4 + T cells (34.66 %±8.584), reflecting the vulnerability of CD8 + T cell exhaustion in the viral infection. Nonetheless, it is not clear whether and how the infection implicates such pathway that suppresses efficient antiviral immune responses. Given this, CD8 + T cells play crucial roles in defeating the virus through exerting extreme cytotoxic mediators, including perforin and granzymes (Mescher et al., 2006).
Since the process of T cell exhaustion is managed to elicit functional unresponsiveness and prevent aggressive immunoactivation, and is in turn regarded as relative autoimmune tissue damage (Cao, 2020), imbalanced immunoactivation is observed in COVID-19 patients as a result of the secretion of hyper pro-inflammatory cytokines and mediators which can be detrimental and eventuate in tissue injuries, especially in severe cases. Despite the reduction of T cell functionality during COVID-19 infection, it seems that the source of these cytokines may be an interesting issue. Some studies recognized macrophages and monocytes, but not T cells, as probable producers of these mediators (Diao et al., 2020; Kany et al., 2019; Minciullo et al., 2016). In our attempt, excessive concentrations of pro-inflammatory cytokines, such as IL-1β and TNF-α, and mediators, including IL-2R, were detected in the serum of severe COVID-19 patients, which were totally comparable with mild subjects. Many SARS-CoV-2 infected patients have an overproduction of several inflammatory cytokines and chemokines (Yang et al., 2020). Severely ill patients with SARS-CoV-2 pneumonia develop a hyper-immune state with a dramatic increase in several pro-inflammatory cytokines IL-1, IL-6 and TNF-α (Mehta et al., 2020). Meanwhile, a meta-analysis report by Leisman et al., indicated that cytokine storm is not directly the cause of COVID-19- induced organ dysfunction as cytokine elevations in COVID-19 could not address an adequate inflammatory response to combat SARS-CoV-2 infection (Leisman et al., 2020). However, Diao et al. reported that high serum concentrations of TNF-α, IL-6, and IL-10 in COVID-19 patients were correlated with decreased number of T cells as negative regulators of T cell survival or proliferation (Diao et al., 2020). It has also been indicated that circulating sIL-2Rs regulate the activation of T lymphocytes in multiple immunological disorders, and enhanced plasma sIL-2R concentrations predict reduced cellular responses against IL-2 (Gooding et al., 1995). In spite of the significant functions of IL-2 signaling in T cells, sIL-2R possibly possesses central functions in the onset of lymphopenia in COVID-19 patients. This could be evidenced by the study of Zhang et al., in which the expression level of IFN-γ in T cells was impaired in the presence of high concentrations of recombinant CD25, where the proliferation and function of T cells were hampered. Besides, soluble IL-2R levels were negatively correlated with the percentage of T cells as a regulatory factor, especially for CD8 + T cells (Song et al., 2020). Our study was in consistence with these findings, such that in severe SARS-CoV-2 cases, low T cell frequency coincided with high level of sIL-2R. However, it is not obvious how this correlation takes place in COVID-19 infected patients.
Other scenarios in our study were the remarkably high levels of TG and LDL-C, as well as their association with the severity of the disease in COVID-19 patients, as compared to the controls. Cholesterol is a crucial component of cell membrane, which not only affects membrane fluidity, but also influences cellular functions including gene expression and metabolism (Ikonen, 2008). Meanwhile the role of cholesterol in T cell activation is a sort of mystery yet, and it has been proven that cholesterol could promote CD8 + T cell exhaustion in the tumor microenvironment through inducing endoplasmic reticulum (ER) stress (Ma et al., 2019). Ma X et al. demonstrated that the expression of immune checkpoints and the CD8 + T cell exhaustion state were progressively and positively correlated with cholesterol accumulation in mouse tumor models (Ma et al., 2019). Additionally, gamma radiation increases TG, cholesterol, LDL-C, TNF-α, and IL-6 contents, as well as elevating T cell exhaustion through the up-regulation of PD-1, while decreasing HDL-C levels in rats (Guida et al., 2016). In our study, patients suffering from severe COVID-19 also had higher LDL-C and TG levels, in addition to extensive PD-1+ T cells and pro-inflammatory cytokines. However, total cholesterol level was not significantly different between the three groups. Supportively, it has been demonstrated that oxidized LDL-C and cholesterol crystals can trigger IL-1β and IL-18 production (Duewell et al., 2010; Fessler, 2017), which in the case of hyper-cholesterolemia could be expected to cultivate to an exaggerated inflammatory response against SARS-CoV-2 infection.
Moreover, diabetes is recognized as a common co-morbidity of COVID-19, which induces a severe manifestation and worse clinical outcomes (Fang et al., 2020). Regarding the chronic and low grade inflammation of diabetes mellitus, there exist several evidence showing that immune cells participate in the progression of the pathogenesis of diabetes. Shi et al. reported an increased PD-1 expression in CD4+CD28−cells in diabetic patients (Shi et al., 2013). Nishimura et al. also revealed that CD8+T cells are responsible for macrophage activation during diabetes progression, while the expression of PD-1 in CD8+T cells could mediate cell exhaustion (Nishimura et al., 2009). In consistence with these studies, we found that COVID-19 infection was closely related to diabetes background and could contribute to extended pro-inflammatory cytokines and PD1 + T lymphocytes, as described in affected patients and particularly in severe subjects. On the contrary, in an interesting report, it was declared that PD-1 was downregulated in CD4+ and CD8 + T cells in diabetic patients, such that the macrophages were activated by T cells to secret various inflammatory cytokines and deteriorate the diabetic state (Sun et al., 2019). Therefore, the serum profile of infected individuals could be considered as a promising prognostic factor for recognizing potential severe cases and making better therapeutic decisions.
By putting the analytical differences between COVID-19 mild and severe patients aside, the interesting point of our results was the independency to gender disparities as no meaningful changes were detected in circulating lymphocytes and serum analysis of patients in the context of gender. This was on the contrary to previous findings that declared gender and sex affect the consequence of COVID-19 infection (ABULUDE and Abulude, 2020). For instance, Gebhard et al. displayed that the fatality ratio of male to female cases was notably elevated among all age groups (Gebhard et al., 2020). Ongoing researches could be valuable to clarify the impacts of ethnicity and geographic issues, and how some unknown mechanisms influence the gender tendency in COVID-19.
5. Conclusion
SARS-CoV-2 infection affects T lymphocytes that are substantial players in the fight against viruses, through reducing number and impairing functionality, which may become worse as the disease progresses. Meanwhile, pro-inflammatory cytokines, such as TNF-α and IL-1β, as well as IL-2R, are elevated in severe cases in comparison to mildly infected individuals. Besides, serum metabolic changes, such as hyper-lipidemia and diabetic background, may be valuable as feasible prognostic factors in COVID-19 progression. Possibly, further researches could clarify the precise mechanism through which T cells are affected by COVID-19, and focus on serum changes to predict and manage the consequence of a patient’s condition.
Author contributions
Ata Mahmoodpoor: Conceptualization; Investigation; Methodology; Roles/Writing - original draft.
Maryam Hosseini: Roles/Writing - original draft; Writing - review & editing.
Mohammad Sadegh Soltani-Zangbar: Formal analysis; Investigation; Methodology; Software; Writing - review & editing.
Sarvin Sanaie: Investigation; Methodology.
Leili Aghebati-Maleki: Investigation; Methodology.
Seyed Hadi Saghaleini: Investigation; Methodology.
Zohreh Ostadi: Investigation; Methodology.
Mahsa Hajivalili: Investigation; Methodology.
Zhinous Bayatmakoo: Data curation; Writing - review & editing.
Mostafa Haji-Fatahaliha: Investigation; Methodology.
Zohreh Babaloo: Project administration.
Sima Shahmohammadi Farid: Investigation; Methodology; Software.
Javad Ahmadian Heris: Data curation; Investigation; Validation.
Leila Roshangar: Writing - review & editing.
Reza Rikhtegar Ghiasi: Data curation; Investigation.
Hossein Samadi Kafil: Investigation; Project administration; Visualization.
Mehdi Yousefi: Conceptualization; Data curation; Project administration; Supervision.
Declaration of Competing Interest
The authors report no declarations of interest.
Acknowledgments
This study was supported by Stem Cell Research Center, Tabriz University of Medical Sciences, Tabriz, Iran [Grant NO. 65300].
Footnotes
Supplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.molimm.2021.06.001.
Appendix A. Supplementary data
The following is Supplementary data to this article:
References
- Abulude F., Abulude I.A. SARS-CoV-2 pandemic in Nigeria: the impact on age and sex distributions. Aurum J. Health Sci. 2020;2:173–189. [Google Scholar]
- Aggarwal S., Gollapudi S., Gupta S. Increased TNF-α-induced apoptosis in lymphocytes from aged humans: changes in TNF-α receptor expression and activation of caspases. J. Immunol. 1999;162:2154–2161. [PubMed] [Google Scholar]
- Bellesi S., Metafuni E., Hohaus S., Maiolo E., Marchionni F., D’Innocenzo S., La Sorda M., Ferraironi M., Ramundo F., Fantoni M. Increased CD95 (Fas) and PD‐1 expression in peripheral blood T lymphocytes in COVID‐19 patients. Br. J. Haematol. 2020;191:207–211. doi: 10.1111/bjh.17034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhimraj A., Morgan R.L., Shumaker A.H., Lavergne V., Baden L., Cheng V.C.-C., Edwards K.M., Gandhi R., Muller W.J., O’Horo J.C. Infectious Diseases Society of America guidelines on the treatment and management of patients with COVID-19. Clin. Infect. Dis. 2020:1–13. doi: 10.1093/cid/ciaa478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bordoni V., Sacchi A., Cimini E., Notari S., Grassi G., Tartaglia E., Casetti R., Giancola L., Bevilacqua N., Maeurer M. An inflammatory profile correlates with decreased frequency of cytotoxic cells in COVID-19. Clin. Infect. Dis. 2020;71(16):2272–2275. doi: 10.1093/cid/ciaa577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun J., Loyal L., Frentsch M., Wendisch D., Georg P., Kurth F., Hippenstiel S., Dingeldey M., Kruse B., Fauchere F. SARS-CoV-2-reactive T cells in healthy donors and patients with COVID-19. Nature. 2020;587:270–274. doi: 10.1038/s41586-020-2598-9. [DOI] [PubMed] [Google Scholar]
- Cao X. COVID-19: immunopathology and its implications for therapy. Nat. Rev. Immunol. 2020;20:269–270. doi: 10.1038/s41577-020-0308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X., Ling J., Mo P., Zhang Y., Jiang Q., Ma Z., Cao Q., Hu W., Zou S., Chen L. Restoration of leukomonocyte counts is associated with viral clearance in COVID-19 hospitalized patients. MedRxiv. 2020 10(2020.03) pp.03-20030437. [Google Scholar]
- Diao B., Wang C., Tan Y., Chen X., Liu Y., Ning L., Chen L., Li M., Liu Y., Wang G. Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID-19) Front. Immunol. 2020;11:827. doi: 10.3389/fimmu.2020.00827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duewell P., Kono H., Rayner K.J., Sirois C.M., Vladimer G., Bauernfeind F.G., Abela G.S., Franchi L., Nuñez G., Schnurr M. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature. 2010;464:1357–1361. doi: 10.1038/nature08938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Etemadi J., Bordbar S., Soltani-Zangbar M.S., Hajivalili M., Aghebati-Maleki L., Motavalli R., Mahmoodpoor A., Shahmohammadi-Farid S., Abedi Azar S., Niknafs B. Prevalence of SARS-CoV-2 specific antibodies in asymptomatic hemodialysis patients. Immunol. Invest. 2021:1–12. doi: 10.1080/08820139.2021.1899202. [DOI] [PubMed] [Google Scholar]
- Fang L., Karakiulakis G., Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir. Med. 2020;8:e21. doi: 10.1016/S2213-2600(20)30116-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenwick C., Joo V., Jacquier P., Noto A., Banga R., Perreau M., Pantaleo G. T‐cell exhaustion in HIV infection. Immunol. Rev. 2019;292:149–163. doi: 10.1111/imr.12823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fessler M.B. A new frontier in immunometabolism. Cholesterol in lung health and disease. Ann. Am. Thorac. Soc. 2017;14:S399–S405. doi: 10.1513/AnnalsATS.201702-136AW. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gebhard C., Regitz-Zagrosek V., Neuhauser H.K., Morgan R., Klein S.L. Impact of sex and gender on COVID-19 outcomes in Europe. Biol. Sex Differ. 2020;11:1–13. doi: 10.1186/s13293-020-00304-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gooding R., Riches P., Dadian G., Moore J., Gore M. Increased soluble interleukin-2 receptor concentration in plasma predicts a decreased cellular response to IL-2. Br. J. Cancer. 1995;72:452. doi: 10.1038/bjc.1995.354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guida M.S., Abd El-Aal A., Kafafy Y., Salama S.F., Badr B.M., Badr G. Thymoquinone rescues T lymphocytes from gamma irradiation-induced apoptosis and exhaustion by modulating pro-inflammatory cytokine levels and PD-1, Bax, and Bcl-2 signaling. Cell. Physiol. Biochem. 2016;38:786–800. doi: 10.1159/000443034. [DOI] [PubMed] [Google Scholar]
- Gupta S., Bi R., Kim C., Chiplunkar S., Yel L., Gollapudi S. Role of NF-κ B signaling pathway in increased tumor necrosis factor-α-induced apoptosis of lymphocytes in aged humans. Cell Death Differ. 2005;12:177–183. doi: 10.1038/sj.cdd.4401557. [DOI] [PubMed] [Google Scholar]
- Ikonen E. Cellular cholesterol trafficking and compartmentalization. Nat. Rev. Mol. Cell Biol. 2008;9:125–138. doi: 10.1038/nrm2336. [DOI] [PubMed] [Google Scholar]
- Kany S., Vollrath J.T., Relja B. Cytokines in inflammatory disease. Int. J. Mol. Sci. 2019;20:6008. doi: 10.3390/ijms20236008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leisman D.E., Ronner L., Pinotti R., Taylor M.D., Sinha P., Calfee C.S., Hirayama A.V., Mastroiani F., Turtle C.J., Harhay M.O. Cytokine elevation in severe and critical COVID-19: a rapid systematic review, meta-analysis, and comparison with other inflammatory syndromes. Lancet Respir. Med. 2020;8(12):1233–1244. doi: 10.1016/S2213-2600(20)30404-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao M., Liu Y., Yuan J., Wen Y., Xu G., Zhao J., Cheng L., Li J., Wang X., Wang F., Liu L. Single-cell landscape of bronchoalveolar immune cells in patients with COVID-19. Nat med. 2020;26(6):842–844. doi: 10.1038/s41591-020-0901-9. [DOI] [PubMed] [Google Scholar]
- Ma X., Bi E., Lu Y., Su P., Huang C., Liu L., Wang Q., Yang M., Kalady M.F., Qian J. Cholesterol induces CD8+ T cell exhaustion in the tumor microenvironment. Cell Metab. 2019;30:143–156. doi: 10.1016/j.cmet.2019.04.002. e145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minciullo P.L., Catalano A., Mandraffino G., Casciaro M., Crucitti A., Maltese G., Morabito N., Lasco A., Gangemi S., Basile G. Inflammaging and anti-inflammaging: the role of cytokines in extreme longevity. Archivum immunologiae et therapiae experimentalis . 2016;64(2):111–126. doi: 10.1007/s00005-015-0377-3. [DOI] [PubMed] [Google Scholar]
- Mehra M.R., Ruschitzka F. American College of Cardiology Foundation; Washington DC: 2020. COVID-19 Illness and Heart Failure: a Missing Link? [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mescher M.F., Curtsinger J.M., Agarwal P., Casey K.A., Gerner M., Hammerbeck C.D., Popescu F., Xiao Z. Signals required for programming effector and memory development by CD8+ T cells. Immunol. Rev. 2006;211:81–92. doi: 10.1111/j.0105-2896.2006.00382.x. [DOI] [PubMed] [Google Scholar]
- Mullen A.C., Hutchins A.S., High F.A., Lee H.W., Sykes K.J., Chodosh L.A., Reiner S.L. Hlx is induced by and genetically interacts with T-bet to promote heritable TH 1 gene induction. Nat. Immunol. 2002;3:652–658. doi: 10.1038/ni807. [DOI] [PubMed] [Google Scholar]
- Ng C., Snell L., Brooks D., Oldstone M.A. Networking at the level of host immunity: immune cell interactions during persistent viral infections. Cell Host Microbe. 2013;13:652–664. doi: 10.1016/j.chom.2013.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishimura S., Manabe I., Nagasaki M., Eto K., Yamashita H., Ohsugi M., Otsu M., Hara K., Ueki K., Sugiura S. CD8+ effector T cells contribute to macrophage recruitment and adipose tissue inflammation in obesity. Nat. Med. 2009;15:914–920. doi: 10.1038/nm.1964. [DOI] [PubMed] [Google Scholar]
- Ross S.H., Cantrell D.A. Signaling and function of interleukin-2 in T lymphocytes. Annu. Rev. Immunol. 2018;36:411–433. doi: 10.1146/annurev-immunol-042617-053352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen K.-L., Yang Y.-H., Jiang R.-M., Wang T.-Y., Zhao D.-C., Jiang Y., Lu X.-X., Jin R.-M., Zheng Y.-J., Xu B.-P. Updated diagnosis, treatment and prevention of COVID-19 in children: experts’ consensus statement (condensed version of the second edition) World J. Pediatr. 2020;16:232–239. doi: 10.1007/s12519-020-00362-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi B., Du X., Wang Q., Chen Y., Zhang X. Increased PD-1 on CD4+ CD28− T cell and soluble PD-1 ligand-1 in patients with T2DM: association with atherosclerotic macrovascular diseases. Metabolism. 2013;62:778–785. doi: 10.1016/j.metabol.2012.12.005. [DOI] [PubMed] [Google Scholar]
- Soltani-Zangbar M.S., Aghebati-Maleki L., Hajivalili M., Haji-Fatahaliha M., Motavalli R., Mahmoodpoor A., Kafil H.S., Farhang S., Pourakbari R., Jadidi-Niaragh F. Application of newly developed SARS-CoV2 serology test along with real-time PCR for early detection in health care workers and on-time plasma donation. Gene Rep. 2021 doi: 10.1016/j.genrep.2021.101140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song J.-W., Zhang C., Fan X., Meng F.-P., Xu Z., Xia P., Cao W.-J., Yang T., Dai X.-P., Wang S.-Y. Immunological and inflammatory profiles in mild and severe cases of COVID-19. Nat. Commun. 2020;11:1–10. doi: 10.1038/s41467-020-17240-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun P., Jin Q., Nie S., Jia S., Li Y., Li X., Guo F. Unlike PD-L1, PD-1 is downregulated on partial immune cells in type 2 diabetes. J. Diabetes Res. 2019;2019 doi: 10.1155/2019/5035261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wherry E.J. T cell exhaustion. Nat. Immunol. 2011;12:492–499. doi: 10.1038/ni.2035. [DOI] [PubMed] [Google Scholar]
- Wölfel R., Corman V., Guggemos W., Seilmaier M., Zange S., Müller M., Niemeyer D., Jones T., Vollmar P., Rothe C., Hoelscher M., Bleicker T., Brünink S., Schneider J., Ehmann R., Zwirglmaier K., Drosten C., Wendtner C. Virological assessment of hospitalized patients with COVID-2019. Nature. 2020;581:465–469. doi: 10.1038/s41586-020-2196-x. [DOI] [PubMed] [Google Scholar]
- Xu X., Chen P., Wang J., Feng J., Zhou H., Li X., Zhong W., Hao P. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci. China Life Sci. 2020;63:457–460. doi: 10.1007/s11427-020-1637-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C., Liu S., Zhao P., Liu H., Zhu L. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020;8:420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L., Liu S., Liu J., Zhang Z., Wan X., Huang B., Chen Y., Zhang Y. COVID-19: immunopathogenesis and Immunotherapeutics. Signal Transduct. Target. Ther. 2020;5:1–8. doi: 10.1038/s41392-020-00243-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y., Wang X., Li X., Xi D., Mao R., Wu X., Cheng S., Sun X., Yi C., Ling Z. Potential contribution of increased soluble IL-2R to lymphopenia in COVID-19 patients. Cell. Mol. Immunol. 2020;17:878–880. doi: 10.1038/s41423-020-0484-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng H.-Y., Zhang M., Yang C.-X., Zhang N., Wang X.-C., Yang X.-P., Dong X.-Q., Zheng Y.-T. Elevated exhaustion levels and reduced functional diversity of T cells in peripheral blood may predict severe progression in COVID-19 patients. Cell. Mol. Immunol. 2020;17:541–543. doi: 10.1038/s41423-020-0401-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou P., Yang X.-L., Wang X.-G., Hu B., Zhang L., Zhang W., Si H.-R., Zhu Y., Li B., Huang C.-L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.