Abstract
Coronavirus disease 2019 (COVID-19) is an ongoing pandemic that to date has spread to >100 countries. Acute kidney injury is not uncommon with this disease. The most common kidney biopsy finding is acute tubular injury. Glomerular diseases such as collapsing glomerulopathy and vasculitis, and thrombotic microangiopathy have been reported. Viral inclusion particles with distinctive spikes in the tubular epithelium and podocytes, and endothelial cells of the glomerular capillary loops, have been visualized by electron microscopy by some but disputed by others as non-viral structures. Interstitial infiltrates have not commonly been described in the published kidney biopsy series from patients with COVID-19. Medications used to treat COVID-19 can lead to interstitial nephritis, but very few have been reported. In summary, interstitial kidney disease is a rare finding in COVID-19.
Keywords: acute tubulointerstitial nephritis, coronavirus, SARS-CoV-2
CASE VIGNETTE
A 59-year-old male with a past medical history of hypertension, coronary artery disease presents with cough and shortness of breath. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection is confirmed. In 24 h, he gets intubated for worsening pulmonary function. Within 8 h of intubation, acute kidney injury (AKI) ensues. He is on steroids and tocilizumab. He is started on remdisivir, Vitamin C and pantoprozole as well. Within the next 24 h, he requires kidney replacement therapy.
Coronaviruses (CoVs) are enveloped, single positive-stranded RNA viruses, which belong to the subfamily Coronavirinae. CoVs have long been recognized as causative agents in self-limited upper respiratory tract infections affecting humans [1] and can be divided into low pathogenic and highly pathogenic CoVs [2]. The low pathogenic CoVs account for 10–30% of upper respiratory tract infections and typically cause mild respiratory diseases [3]. In contrast, the highly pathogenic CoVs, known as novel coronavirus (nCoV), including SARS, Middle East respiratory syndrome (MERS) CoV and the more recent 2019-nCoV or SARS-CoV-2 predominantly infect lower airways and cause fatal pneumonia [4]. These three highly pathogenic β-CoVs have posed a substantial threat to public health.
SARS-CoV was first described as a new human infection in Guangdong province, China in 2002 (World Health Organization 2020) and ended in July 2003. It infected 8096 people and caused 774 deaths with an overall mortality rate of about 9.6% [5, 6]. According to the National Health Service, no new cases of SARS-CoV infection have been reported in humans since 2004 [7].
MERS CoV was first identified in Jordan in 2012 (World Health Organization 2019) and has caused a total of 2494 laboratory-confirmed cases and 858 deaths from 27 countries (mortality rate, 34.4%) since September 2012 [8]. In addition, MERS is still considered to be an ongoing epidemic but appears to be limited to its area of origin [8, 9].
SARS-CoV-2/coronavirus disease 2019 (COVID-19) is an ongoing pandemic that to date has spread to >100 countries (World Health Organization 2020). The estimated case fatality rates (CFRs) for COVID-19 are to be around 2.3% [10]. Nonetheless, despite its lower CFR, COVID-19 has now caused orders of magnitude more deaths than SARS and MERS combined [11].
Viral nephropathy may be caused by viruses themselves or virus-induced immune mechanisms. This article summarizes current evidence on tubulointerstitial kidney disease during CoVs infection.
AKI IN CoV INFECTION
Epidemiology
Kidney involvement of human CoV was noticed when the SARS-CoV epidemic occurred in the early 2000s. AKI was reported in 6.7% of 536 patients with SARS-CoV [12] and was an independent risk factor predicting mortality [13] SARS-CoV patients developed AKI at a median duration of 20 days from the onset of viral infection [14]. The detection of polymerase chain reaction fragments of coronavirus in urine from 21% to 50% of SARS patients between the second and the third week of the viral infection implied a possibility of kidney tropism of the CoV [15].
AKI appears to be frequent in the context of MERS. In a Saudi cohort, 30 out of 70 MERS patients (42.9%) developed AKI during the course of their illness [16]. Among critically ill MERS patients, AKI was reported to develop in 75% of them [13, 17]. Progressive impairment of renal function and AKI start 9–12 days after symptom onset among MERS patients, compared with a median of 20 days for SARS patients [12, 17, 18]. However, despite the presence of viral particles in renal proximal tubular epithelial cell, no evidence of concurrent tubulointerstitial nephritis was identified [19].
AKI is a frequent complication of SARS-CoV-2 (Table 1) and is associated with mortality especially when dialysis is required [20–48].
Table 1.
| Ref. | City/country | Patients, n | AKI (%) | RRT (%) | Mortality |
|---|---|---|---|---|---|
| [23] | Wuhan | 116 | 0 | 4 | NR |
| [27] | Wuhan | 99 | 3 | 9; 39 in ICU | NR |
| [28] | Wuhan | 138 | 4; 8 in ICU | 2; 6 in ICU | NR |
| [29] | Wuhan | 333 | 11; 43 in ICU | 3 in ICU | 57% |
| [30] | Wuhan | 701 | 5 | NR | 34% |
| [20] | Wuhan | 41 | 7; 23 in ICU | 7; 23 in ICU | NR |
| [31] | Wuhan | 274 | 11 | 1 | NR |
| [9] | Wuhan | 191 | 15 | 5 | NR |
| [32] | Wuhan | 52 | 29 | 17 | NR |
| [33] | Wuhan | 102 | 20 | 6 | NR |
| [35] | China | 1099 | 0.5; 6 in ICU | 0.8; 12 in ICU | NR |
| [36] | Jiangsu | 80 | 3 | 1 | NR |
| [21] | New York | 5700 | 24 | 4 | NR |
| [37] | New York | 1000 | 34; 78 in ICU | 14; 35 in ICU | NR |
| [38] | New York | 257 | NR | 31 | NR |
| [39] | New York | 5449 | 37; 76 in ICU | 23 in ICU |
|
| [43] | New York | 3235 | 43; 68 in ICU | 45% with AKI died (7% in non-AKI) | |
| [40] | New York | 3099 | 20.6 | 63.3% | |
| [41] | New York | 9657 | 39.9 | 6.6 |
|
| [42] | Washington | 21 | 19 | NR | NR |
| [44] | Louisiana | 575 | 28; 61 in ICU | 15; 73 in ICU | 50%; 72% in patients on RRT |
| [45] | USA | 2215 | 43 | 20 | NR |
| [46] | UK | 10 547 | NR | 27 | 57% |
| [47] | France | 71 | 80 | 18 | 7% |
| [48] | France | 100 | 81 | 16 | 35% with AKI (5% in non-AKI) |
NR, not reported.
Assessment of data from major published cohorts on COVID-19, combining results from intensive care unit (ICU) admissions with non-ICU admission, reveals an overall AKI incidence of around 4.2% [32, 42, 49–52]. Among the non-survivors, the incidence of AKI is ∼30% and renal replacement therapy (RRT) is required in 19.5%. Comparatively, in the SARS outbreak in 2003, the incidence of AKI was around 6.7% and multivariate analysis showed AKI as a significant independent risk factor for predicting mortality [relative risk: 4.057; 99% confidence interval (CI): 1.461–11.27; P < 0.001] [12].
In China, the reported incidence of AKI in hospitalized patients with COVID-19 ranged from 0.5% to 29% [31, 32, 35, 53] and occurred within a median of 7–14 days after admission [31, 53].
Studies from the USA have reported much higher rates of AKI. In a study of nearly 5500 patients admitted with COVID-19 in a New York City hospital system, AKI occurred in 37%, with 14% of the patients requiring dialysis [39]. About one-third were diagnosed with AKI within 24 h of admission in this study. Of note, these rates are much higher than those reported during the SARS-CoV epidemic [12]. AKI occurred at much higher rates in critically ill patients admitted to New York City hospitals, ranging from 78% to 90% [19, 37–39, 54]. Of 257 patients admitted to ICUs in a study from New York City, 31% received RRT [38].
In a multicentre cohort study of 3099 critically ill adults with COVID-19 admitted to ICUs at 67 hospitals across the USA, AKI-RRT is common among critically ill patients with COVID-19 (20.6%) and is associated with a hospital mortality rate of >60%. Among those who survive to discharge, 33.8% still depend on RRT at discharge and 18.1% remain RRT dependent 60 days after ICU admission. Patient-level risk factors for AKI-RRT included chronic kidney disease (CKD), men, non-White race, hypertension, diabetes mellitus, higher body mass index, higher d-dimer and greater severity of hypoxaemia on ICU admission. Predictors of 28-day mortality in patients with AKI-RRT were older age, severe oliguria and admission to a hospital with fewer ICU beds or one with greater regional density of COVID-19 [40].
In an observational retrospective cohort study including 9657 patients admitted with COVID-19 limited to the New York metropolitan area during the peak of the COVID-19 pandemic (between 1 March 2020 and 27 April 2020), AKI in hospitalized patients with COVID-19 was associated with significant risk for death. Among patients with AKI Stages 1–3 who survived, 74.1% achieved kidney recovery by the time of discharge. Among those with AKI receiving dialysis who survived, 30.6% remained on dialysis at discharge, and pre-hospitalization CKD was the only independent risk factor associated with needing dialysis at discharge (adjusted odds ratio: 9.3; 95% CI: 2.3–37.8) [41].
Spectrum of renal pathologic abnormalities during SARS-CoV-2 infection
Case reports and autopsy series have revealed that most patients with COVID-19-associated AKI have evidence of prominent acute tubular necrosis and diffuse erythrocyte aggregation, specific dysfunction of the proximal tubules (loss of the brush border, intratubular debris and reduced expression of the endocytosis receptor megalin in the brush border), endothelial damage and capillary occlusions, tubules and glomerular complement complex deposit [55–59]. More recent findings support SARS-CoV-2 kidney tropism. A higher SARS-CoV-2 viral load in urine sediments from COVID-19 patients correlated with increased incidence of AKI and mortality [60]. A mild associated interstitial infiltrate may be present [60]. Other biopsy findings have included collapsing glomerulopathy associated with African ancestry and a high-risk APOL1 genotype [59, 61, 62], thrombotic microangiopathy and diverse underlying kidney diseases [56, 59, 63–65]. Kidney infarction has also been reported [66] (Table 2).
Table 2.
| Acute tubulointerstitial injury |
| Acute tubule epithelial damage of varying degrees |
| Acute damage of the proximal tubules |
| Systemic capillary leak syndrome |
| Interstitial edema and/or nephritis |
| Glomerular disease |
| − Podocytopathy: |
| Minimal change disease |
| Collapsing focal segmental hyalinosis in African ancestry patient with high-risk APOL1 genotype |
| − Endotheliopathy: TMA |
| − Others: |
| Membranous nephropathy, crescentic GN (ANCA vasculitis and anti-GBM), IgA nephropathy and lupus nephritis |
| Small fibrin thrombi in the glomerular capillaries consistent with disseminated intravascular coagulation |
| Miscellaneous |
| Myoglobin cast nephropathy |
| Oxalate nephropathy |
| Renal cortical necrosis |
| Renal infarction |
| Renal artery stenosis |
ANCA, anti polynuclear neutrophil cytoplasmic antibodies; GBM, glomerular basement membrane; GN, glomerulonephritis; TMA, thrombotic microangiopathy.
Viral inclusion particles with distinctive spikes in the tubular epithelium and podocytes, and endothelial cells of the glomerular capillary loops, have been visualized by electron microscopy [57, 58, 67] and may support this possibility, although the clinical significance of this remains unknown [68, 69].
The demonstration of lymphocytic endothelialitis in the kidney, in addition to viral inclusion particles in glomerular capillary endothelial cells, suggests that microvascular dysfunction is secondary to endothelial damage [67]. Other potential etiologies of AKI common to critical illness presentations, including acute respiratory distress syndrome, rhabdomyolysis, volume depletion and interstitial nephritis, all remain relevant in patients with COVID-19 [70].
Acute tubulointerstitial nephritis during SARS-CoV-2 infection
Acute interstitial nephritis (AIN) is not a rare cause of AKI but it remains a Cinderella among other causes, being underrecognized and underdiagnosed despite the fact that it is a serious reversible disease that can cause CKD and end-stage renal disease . The same probably applies to COVID-19.
AIN is a disease of tubular dysfunction with insidious onset that usually starts as non-oliguric acute renal failure with signs of tubulointerstitial damage that are present but rarely dominate, therefore high clinical suspicion is necessary for timely diagnosis at its reversible stage and early treatment that highly impacts its outcome.
AIN, as one of the differential diagnoses of kidney injury in COVID-19 patients, should therefore be important part of the equation when deciding on medications including steroids.
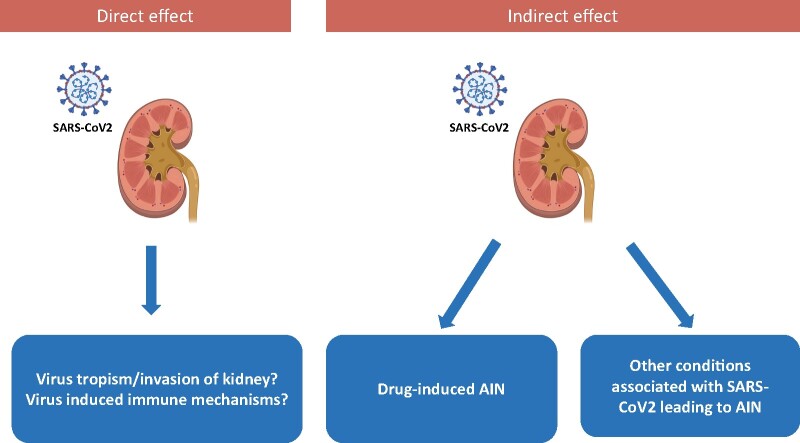
Although AIN remains uncommon during COVID-19 infection (Table 3) [64, 71–74], several mechanisms may explain such findings (Figure 1).
Table 3.
AIN and COVID-19
| Ref. | Study (age, gender) | Presentation | Pathological finding | Outcome related to AIN |
|---|---|---|---|---|
| [73] |
|
AKI |
|
|
| [74] |
|
|
|
|
| [75] |
|
AKI | Mononuclear cell infiltration (80% of cases); CD3+T and CD68+ macrophages | NA |
| [76] |
|
|
Diffuse interstitial infiltration in 8/13 cases but no isolated case of AIN | NA |
| [66] |
|
|
FSGS, ATI and AIN in 3/17 patients | NA |
M, male; pts, patients; ATI, acute tubular injury; FSGS, focal segmental glomerulosclerosis; MMF, mycophenolate mofetil; SCr, serum creatinine; CVVHDF, continuous veno-venous haemodiafiltration; Hu haematuria; NA, not available.
FIGURE 1:
AIN during SARS CoV2 infection: mechanisms
Specific interstitial infiltration
Interstitial infiltrates have not commonly been described in the published kidney biopsy series from patients with COVID-19 [56, 58, 63].
SARS-CoV-2 has been detected in a kidney allograft [71] associated with a mononuclear cell infiltrate, indicating that the virus is able to enter renal parenchyma and may cause interstitial nephritis. Recent post mortem histopathological analyses showed positive immunostaining with SARS-CoV nucleoprotein antibody in tubules, which supports these findings [57].
It is important to pay attention to low molecular weight proteinuria (Pu) and ionic disorders, as they precede AKI and tubulointerstitial involvement [75].
Werion et al. [76] established that SARS-CoV-2 causes specific manifestations of proximal tubule dysfunction as attested by low molecular weight Pu (70–80%), neutral aminoaciduria (46%) and defective handling of uric acid (46%) or phosphate (19%), but not of glucose, corresponding to a partial renal Fanconi syndrome. Proximal tubule dysfunction was independent of pre-existing comorbidities, nephrotoxic medications or viral load. At the structural level, kidneys from patients with COVID-19 showed prominent tubular injury, including in the initial part of the proximal tubule, with brush border loss, acute tubular necrosis, intraluminal debris and a marked decrease in the expression of megalin in the brush border [76]. Among features of proximal tubule dysfunction, hypouricaemia with inappropriate uricosuria was independently associated with disease severity and with a significant increase in the risk of respiratory failure requiring invasive mechanical ventilation. The molecular mechanisms accounting for such specific defects remain unknown [76].
Even though studies argue against SARS-CoV-2 nephropathy [77], we know that there are many cases of AIN caused by infections remote to the kidney (Legionella, leptospirosis and streptococcal organisms), and that might also be a case with SARS-CoV-2. Finding virus or its particles within kidney does not necessarily mean direct clinical–pathological connection with AKI, and vice versa.
Drug-induced AIN
Medication-induced nephrotoxicity is a relatively common cause affecting the tubulointerstitial compartment [78, 79]. However, despite patients with severe COVID-19 in the ICU likely receive multiple medications, drug-induced AIN remains uncommon in this setting.
Only case reports of drug reaction with eosinophilia and systemic symptoms syndrome associated with hydroxychloroquine or azithromycin [80] and a drug-induced granulomatous AIN [72] have been reported in patients with COVID-19, with a good response to corticosteroid therapy.
Furthermore, specific therapeutic treatments for COVID-19 itself such as the use of antiviral agents can potentially induce tubulointerstitial diseases [81–84].
Thus, treatment-related complications need to be considered when determining the etiology of AKI.
Other disease-associated AIN
Several diseases reported to be associated with COVID-19, such as secondary haemophagocytic lymphohistiocytosis [85] and autoimmune haemolytic anaemia [86–88], can induce AIN [88, 89].
CONCLUSION
In summary, even though ATN probably remains the most frequent cause of AKI in clinical settings like COVID-19, and proof of viral replication in human kidney cells remains to be confirmed, one should not forget that it is often hard to differentiate it from AIN that must be one of our differential diagnoses. Besides numerous medications used for COVID-19, AIN in that setting might also be caused not only through direct pathogen invasion of the kidney, but also through different immunologic mechanisms.
CONFLICT OF INTEREST STATEMENT
KDJ serves as a consultant for Astex Pharmaceuticals and Natera and is a paid contributor to Uptodate.com
No disclosure for the other authors.
REFERENCES
- 1.Falsey AR, McCann RM, Hall WJ. et al. The “common cold” in frail older persons: impact of rhinovirus and coronavirus in a senior daycare center. J Am Geriatr Soc 1997; 45: 706–711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cui J, Li F, Shi ZL.. Origin and evolution of pathogenic coronaviruses. Nat Rev Microbiol 2019; 17: 181–192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Su S, Wong G, Shi W. et al. Epidemiology, genetic recombination, and pathogenesis of coronaviruses. Trends Microbiol 2016; 24: 490–502 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Channappanavar R, Perlman S.. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin Immunopathol 2017; 39: 529–539 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Drosten C, Günther S, Preiser W. et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med 2003; 348: 1967–1976 [DOI] [PubMed] [Google Scholar]
- 6.Ksiazek TG, Erdman D, Goldsmith CS. et al. ; SARS Working Group. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med 2003; 348: 1953–1966 [DOI] [PubMed] [Google Scholar]
- 7.National Health Service. SARS (Severe Acute Respiratory Syndrome). National Health Service, 2019. https://www.nhs.uk/conditions/sars [Google Scholar]
- 8.WHO. Middle East Respiratory Syndrome Coronavirus (MERS-CoV). 2020.http://www.who.int/emergencies/mers-cov/en/ (3 March 2021, date last accessed)
- 9.Al-Tawfiq JA, Kattan RF, Memish ZA.. Middle East respiratory syndrome coronavirus disease is rare in children: an update from Saudi Arabia. World J Clin Pediatr 2016; 5: 391–396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Porcheddu R, Serra C, Kelvin D. et al. Similarity in case fatality rates (CFR) of COVID-19/SARS-COV-2 in Italy and China. J Infect Dev Ctries 2020; 14: 125–128 [DOI] [PubMed] [Google Scholar]
- 11.Mahase E.Coronavirus: covid-19 has killed more people than SARS and MERS combined, despite lower case fatality rate. BMJ 2020; 368: m641. [DOI] [PubMed] [Google Scholar]
- 12.Chu KH, Tsang WK, Tang CS. et al. Acute renal impairment in coronavirus-associated severe acute respiratory syndrome. Kidney Int 2005; 67: 698–705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cha RH, Joh JS, Jeong I. et al. ; Critical Care Team of National Medical Center. Renal complications and their prognosis in Korean patients with Middle East respiratory syndrome-coronavirus from the central MERS-CoV designated hospital. J Korean Med Sci 2015; 30: 1807–1814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lai KN, Tsang KW, Seto WH. et al. Clinical, laboratory, and radiologic manifestation of SARS. Curr Infect Dis Rep 2004; 6: 213–219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Peiris JS, Chu CM, Cheng VC. et al. HKU/UCH SARS Study Group. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet 2003; 361: 1767–1772 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Saad M, Omrani AS, Baig K. et al. Clinical aspects and outcomes of 70 patients with Middle East respiratory syndrome coronavirus infection: a single-center experience in Saudi Arabia. J Glob Infect Dis 2014; 29: 301–306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Eckerle I, Muller MA, Kallies S. et al. In-vitro renal epithelial cell infection reveals a viral kidney tropism as a potential mechanism for acute renal failure during Middle East Respiratory Syndrome (MERS) Coronavirus infection. Virol J 2013; 10: 359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zaki AM, van Boheemen S, Bestebroer TM. et al. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med 2012; 367: 1814–1820 [DOI] [PubMed] [Google Scholar]
- 19.Alsaad KO, Hajeer AH, Al Balwi M. et al. Histopathology of Middle East respiratory syndrome coronovirus (MERS-CoV) infection—clinicopathological and ultrastructural study. Histopathology 2018; 72: 516–524 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Huang C, Wang Y, Li X. et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan. Lancet 2020; 395: 497–506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Richardson S, Hirsch JS, Narasimhan M. et al. ; The Northwell COVID-19 Research Consortium. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA 2020; 323: 2052–2059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wendel Garcia PD, Fumeaux T, Guerci P. et al. RISC-19-ICU investigators. Prognostic factors associated with mortality risk and disease progression in 639 critically ill patients with COVID-19 in Europe: initial report of the international RISC-19-ICU prospective observational cohort. EClinicalMedicine 2020; 25: 100449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pelayo J, Lo KB, Bhargav R. et al. Clinical characteristics and outcomes of community- and hospital-acquired acute kidney injury with COVID-19 in a US inner city hospital system. Cardiorenal Med 2020; 10: 223–231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Becher T, Frerichs I.. Mortality in COVID-19 is not merely a question of resource availability. Lancet Respir Med 2020; 8: 832–833 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Karagiannidis C, Mostert C, Hentschker C. et al. Case characteristics, resource use, and outcomes of 10 021 patients with COVID-19 admitted to 920 German hospitals: an observational study. Lancet Respir Med 2020; 8: 853–862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang L, Li X, Chen H. et al. Coronavirus disease 19 infection does not result in acute kidney injury: an analysis of 116 hospitalized patients from Wuhan, China. Am J Nephrol 2020; 51: 343–348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen N, Zhou M, Dong X. et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 2020; 395: 507–513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang D, Hu B, Hu C. et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus infected pneumonia in Wuhan, China. JAMA 2020; 323: 1061–1069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pei G, Zhang Z, Peng J. et al. Renal involvement and early prognosis in patients with COVID-19 pneumonia. J Am Soc Nephrol 2020; 31: 1157–1165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cheng Y, Luo R, Wang K. et al. Kidney disease is associated with in hospital death of patients with COVID-19. Kidney Int 2020; 97: 829–838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chen T, Wu D, Chen H. et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ 2020; 368: m1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhou F, Yu T, Du R. et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020; 395: 1054–1062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yang X, Yu Y, Xu J. et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med 2020; 8: 475–481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cao J, Tu WJ, Cheng W. et al. Clinical features and short-term outcomes of 102 patients with corona virus disease 2019 in Wuhan, China. Clin Infect Dis 2020; 71: 748–755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Guan WJ, Ni ZY, Hu Y. et al. ; China Medical Treatment Expert Group for Covid-19. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382: 1708–1720 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wu J, Liu J, Zhao X. et al. Clinical characteristics of imported cases of COVID-19 in Jiangsu province: a multicenter descriptive study. Clin Infect Dis 2020; 71: 706–712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Argenziano MG, Bruce SL, Slater CL. et al. Characterization and clinical course of 1000 patients with COVID-19 in New York: retrospective case series. BMJ 2020; 369: m1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cummings MJ, Baldwin MR, Abrams D. et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York city: a prospective cohort study. Lancet 2020; 395: 1763–1770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hirsch JS, Ng JH, Ross DW. et al. ; Northwell COVID-19 Research Consortium; Northwell Nephrology COVID-19 Research Consortium. Acute kidney injury in patients hospitalized with COVID-19. Kidney Int 2020; 98: 209–218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gupta S, Coca SG, Chan L. et al. ; The STOP-COVID Investigators. AKI treated with renal replacement therapy in critically Ill patients with COVID-19. J Am Soc Nephrol 2021; 32: 161–176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ng JH, Hirsch JS, Hazzan A. et al. ; Northwell Nephrology COVID-19 Research Consortium. Outcomes among patients hospitalized with COVID-19 and acute kidney injury. Am J Kidney Dis 2021; 77: 204–215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Arentz M, Yim E, Klaff L. et al. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State. JAMA 2020; 323: 1612–1614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chan L, Chaudhary K, Saha A. et al. Acute kidney injury in hospitalized patients with COVID-19. J Am Soc Nephrol 2021; 32: 151–160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Mohamed MM, Lukitsch I, Torres-Ortiz AE. et al. Acute kidney injury associated with coronavirus disease 2019 in urban New Orleans. Kidney360 2020; 1: 614–622 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gupta S, Hayek SS, Wang W. et al. ; STOP-COVID Investigators. Factors associated with death in critically ill patients with coronavirus disease 2019 in the US. JAMA Intern Med 2020; 180: 1436–1412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Intensive Care National Audit and Research Centre. ICNARC Report on COVID-19 in Critical Care Case Mix Programme Database, 2020. www.icnarc.org (15 April 2021, date last accessed) [DOI] [PMC free article] [PubMed]
- 47.Rubin S, Orieux A, Prevel R. et al. Characterization of acute kidney injury in critically ill patients with severe coronavirus disease 2019. Clin Kidney J 2020; 13: 354–361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Joseph A, Zafrani L, Mabrouki A. et al. Acute kidney injury in patients with SARS-CoV-2 infection. Ann Intensive Care 2020; 10: 117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Grasselli G, Pesenti A, Cecconi M.. Critical care utilization for the COVID-19 outbreak in Lombardy, Italy: early experience and forecast during an emergency response. JAMA 2020; 323: 1545–1546 [DOI] [PubMed] [Google Scholar]
- 50.Luigi Palmieri XA, Bella A, Bellino S. et al. Characteristics of COVID-19 Patients Dying in Italy Report Based on Available Data on March 20th, 2020 Epidemiology for Public Health. Higher Institute of Health, 2020. https://www.epicentro.iss.it/coronavirus/bollettino/Report-COVID-2019_20_marzo_eng.pdf (3 March 2021, date last accessed)
- 51.Ronco C, Reis T, Husain-Syed F.. Management of acute kidney injury in patients with COVID-19. Lancet Respir Med 2020; 8: 738–742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gupta A, Madhavan MV, Sehgal K. et al. Extrapulmonary manifestations of COVID-19. Nat Med 2020; 26: 1017–1032 [DOI] [PubMed] [Google Scholar]
- 53.Wu C, Chen X, Cai Y. et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med 2020; 180: 934–943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Petrilli CM, Jones SA, Yang J. et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ 2020; 369: m1966 (doi: 10.1136/bmj.m1966) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bradley BT, Maioli H, Johnston R. et al. Histopathology and ultrastructural findings of fatal COVID-19 infections in Washington State: a case series. Lancet 2020; 396: 320–332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sharma P, Uppal NN, Wanchoo R. et al. ; Northwell Nephrology COVID-19 Research Consortium. COVID-19-associated kidney injury: a case series of kidney biopsy findings. J Am Soc Nephrol 2020; 31: 1948–1958 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Su H, Yang M, Wan C, et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int 2020; 98: 219–227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Puelles VG, Lütgehetmann M, Lindenmeyer MT. et al. Multiorgan and renal tropism of SARS-CoV-2. N Engl J Med 2020; 383: 590–592 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sharma P, Ng JH, Bijol V. et al. Pathology of COVID-19-associated acute kidney injury. Clin Kidney J 2021; 14: i30–i39 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Caceres P, Savickas G, Murray S. et al. High SARS-CoV-2 viral load in urine sediment correlates with acute kidney injury and poor COVID-19 outcome. J Am Soc Nephrol 2021; doi: 10.1681/ASN.2021010059. Online ahead of print [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Peleg Y, Kudose S, D’Agati V. et al. Acute kidney injury due to collapsing glomerulopathy following COVID-19 infection. Kidney Int Rep 2020; 5: 940–945 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gaillard F, Ismael S, Sannier A. et al. Tubuloreticular inclusions in COVID-19-related collapsing glomerulopathy. Kidney Int 2020; 98: 241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kudose S, Batal I, Santoriello D. et al. Kidney biopsy findings in patients with COVID-19. J Am Soc Nephrol 2020; 31: 1959–1968 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Akilesh S, Nast CC, Yamashita M. et al. Multicenter clinicopathologic correlation of kidney biopsies performed in COVID-19 patients presenting with acute kidney injury or proteinuria. Am J Kidney Dis 2021; 77: 82–93e1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Case R, Ramaniuk A, Martin P. et al. Systemic capillary leak syndrome secondary to coronavirus disease 2019. Chest 2020; 158: e267–e268 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Post A, den Deurwaarder ESG, Bakker SJL. et al. Kidney infarction in patients with COVID-19. Am J Kidney Dis 2020; 76: 431–435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Varga Z, Flammer AJ, Steiger P. et al. Endothelial cell infection and endothelilitis in COVID-19. Lancet 2020; 395: 1417–1418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ng JH, Bijol V, Sparks MA. et al. Pathophysiology and pathology of acute kidney injury in patients with COVID-19. Adv Chronic Kidney Dis 2020; 27: 365–376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Farkash EA, Wilson AM, Jentzen JM.. Ultrastructural evidence for direct renal infection with SARS-CoV-2. J Am Soc Nephrol 2020; 31: 1683–1687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Peerapornratana S, Manrique-Caballero CL, Gómez H. et al. Acute kidney injury from sepsis: current concepts, epidemiology, pathophysiology, prevention and treatment. Kidney Int 2019; 96: 1083–1099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Westhoff TH, Seibert FS, Bauer F. et al. Allograft infiltration and meningoencephalitis by SARS-CoV-2 in a pancreas-kidney transplant recipient. Am J Transplant 2020; 20: 3216–3220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Szajek K, Kajdi ME, Luyckx VA. et al. Granulomatous interstitial nephritis in a patient with SARS-CoV-2 infection. BMC Nephrol 2021; 22: 19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ferlicot S, Jamme M, Gaillard F. et al. ; AP-HP/Universities/Inserm COVID-19 Research Collaboration. The spectrum of kidney biopsies in hospitalized patients with COVID-19, acute kidney injury, and/or proteinuria. Nephrol Dial Transplant 2021; doi: 10.1093/ndt/gfab042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Nasr SH, Alexander MP, Cornell LD. et al. Kidney biopsy findings in patients with COVID-19, kidney injury, and proteinuria. Am J Kidney Dis 2021; 77: 465–468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Karras A, Livrozet M, Lazareth H. et al. Proteinuria and clinical outcomes in hospitalized COVID-19 patients: a retrospective single-center study. Clin J Am Soc Nephrol 2021; 16: 514–521 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Werion A, Belkhir L, Perrot M. et al. ; Cliniques universitaires Saint-Luc (CUSL) COVID-19 Research Group. SARS-CoV-2 causes a specific dysfunction of the kidney proximal tubule. Kidney Int 2020; 98: 1296–1307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Rossi GM, Delsante M, Pilato FP. et al. Kidney biopsy findings in a critically ill COVID-19 patient with dialysis-dependent acute kidney injury: a case against “SARS-CoV-2 nephropathy”. Kidney Int Rep 2020; 5: 1100–1105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Sales GTM, Foresto RD.. Drug-induced nephrotoxicity. Rev Assoc Med Bras (1992) 2020; 66 (Suppl 1): s82–s90 [DOI] [PubMed] [Google Scholar]
- 79.Naughton CA.Drug-induced nephrotoxicity. Am Fam Physician 2008; 78: 743–750 [PubMed] [Google Scholar]
- 80.Herman A, Matthews M, Mairlot M. et al. Drug reaction with eosinophilia and systemic symptoms syndrome in a patient with COVID-19. J Eur Acad Dermatol Venereol 2020; 34: e768–e700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Dordal Culla MT, Herrera-Lasso Regás V, Martí-Garrido J. et al. Treating COVID-19: review of drug hypersensitivity reactions. J Investig Allergol Clin Immunol 2020; 30: 385–399 [DOI] [PubMed] [Google Scholar]
- 82.Manjaly Thomas ZR, Leuppi-Taegtmeyer A, Jamiolkowski D. et al. Emerging treatments in COVID-19: adverse drug reactions including drug hypersensitivities. J Allergy Clin Immunol 2020; 146: 786–789 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Izzedine H, Jhaveri KD, Perazella MA.. COVID-19 therapeutic options for patients with kidney disease. Kidney Int 2020; 97: 1297–1298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Mehta P, McAuley DF, Brown M. et al. ; HLH Across Speciality Collaboration, UK. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet 2020; 395: 1033–1034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Hindilerden F, Yonal-Hindilerden I, Akar E. et al. Severe autoimmune hemolytic anemia in COVID-19 infection, safely treated with steroids. Mediterr J Hematol Infect Dis 2020; 12: e2020053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Lazarian G, Quinquenel A, Bellal M. et al. Autoimmune haemolytic anaemia associated with COVID-19 infection. Br J Haematol 2020; 190: 29–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lopez C, Kim J, Pandey A. et al. Simultaneous onset of COVID-19 and autoimmune haemolytic anaemia. Br J Haematol 2020; 190: 31–32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Cho E, Cha I, Yoon K. et al. Hemophagocytic syndrome in a patient with acute tubulointerstitial nephritis secondary to hepatitis A virus infection. J Korean Med Sci 2010; 25: 1529–1531 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Demirkaya E, Atay AA, Musabak U. et al. Ceftriaxone-related hemolysis and acute renal failure. Pediatr Nephrol 2006; 21: 733–736 [DOI] [PubMed] [Google Scholar]