Abstract
The prevailing coronavirus disease-19 (COVID-19) caused by a novel severe acute respiratory syndrome coronavirus (SARS-CoV-2) has presented some neurological manifestations including hyposmia, hypogeusia, headache, stroke, encephalitis, Guillain–Barre syndrome, and some neuropsychiatric disorders. Although several cell types in the brain express angiotensin-converting enzyme-2 (ACE2), the main SARS-CoV-2 receptor, and other related proteins, it remains unclear whether the observed neurological manifestations are attributed to virus invasion into the brain or just comorbidities caused by dysregulation of systemic factors. Here, we briefly review the neurological manifestations of SARS-CoV-2, summarize recent evidence for the potential neurotropism of SARS-CoV-2, and discuss the potential mechanisms of COVID-19-associated neurological diseases.
Keywords: SARS-CoV-2, ACE2, brain invasion, neurological manifestations
Introduction
In less than two decades, three coronavirus outbreaks prevailed to form major epidemics that mainly attack the respiratory system of patients, including severe acute respiratory syndrome (SARS) caused by SARS-coronavirus (SARS-CoV) in 2002–2003, Middle East respiratory syndrome (MERS) by MERS-CoV in 2012, and the ongoing coronavirus disease-19 (COVID-19). SARS affected a total of 8096 people and caused ∼9.56% of deaths (De Wit et al., 2016), and MERS was responsible for 34.4% of deaths in 2494 total patients (Berger, 2020). As of May 10, 2021, COVID-19 has caused nearly 160 million cases and over 3.3 million deaths globally and it is becoming the most severe pandemic over the last century.
The most common symptoms of COVID-19 are fever, cough, fatigue, muscle pain, and shortness of breath (Garg et al., 2020; Grasselli et al., 2020; Guan et al., 2020; Huang et al., 2020a). Some reports showed that >36.4% of confirmed cases presented neurological manifestations, including dizziness, headache, hypogeusia, and hyposmia (Helms et al., 2020; Mao et al., 2020). Cerebrovascular diseases such as stroke have also been identified in some patients (Giustozzi et al., 2020; Oxley et al., 2020; Viguier et al., 2020). The neurological involvement is not specific for COVID-19, because similar neurological abnormalities are also observed following infections from other coronaviruses, such as SARS-CoV and MERS-CoV (Glass et al., 2004; Li et al., 2016; Ellul et al., 2020).
Like SARS-CoV, SARS-CoV-2 also uses protease-processed spike (S) protein to invade host cells through angiotensin-converting enzyme-2 (ACE2) (Li et al., 2003; Hoffmann et al., 2020; Walls et al., 2020). The cellular serine proteases transmembrane protease serine 2 (TMPRSS2) and furin are responsible for S protein priming and pre-activated cleavage, respectively, to facilitate the access to host cells (Hoffmann et al., 2020; Shang et al., 2020). In addition, other cellular receptors, such as Neuropilin-1 (NRP-1) and CD147, may mediate the virus entry into the host cell through ACE2-independent manner (Cantuti-Castelvetri et al., 2020; Daly et al., 2020; Zhou et al., 2020). Using pseudovirus system, it has been shown that SARS-CoV-2 entry and replication can be facilitated by the proprotein convertase furin, the cell surface protease TMPRSS2, and the lysosomal proteases cathepsins (Shang et al., 2020). The presence of a polybasic furin-type cleavage site in the SARS-CoV-2 S protein might explain why SARS-CoV-2 spreads much more rapidly than SARS-CoV (Coutard et al., 2020).
ACE2 protein was found to be expressed in neurons of the brain (Song et al., 2021), and NRP-1 expression was observed in the olfactory system (Kawakami et al., 1996), including neurons of the olfactory epithelium (Cantuti-Castelvetri et al., 2020). The expression of putative SARS-CoV-2 receptors in several neuronal cell types raised possibility of direct invasion of SARS-CoV-2 into the brain (Yang et al., 2020).
COVID-19-associated neurological manifestations
Accumulating case reports have shown that COVID-19 patients exhibit a wide range of neurological manifestations of either the central nervous system (CNS) or the peripheral nervous system (PNS), in isolation or together with other features (Ellul et al., 2020; Iadecola et al., 2020; Table 1). For example, a retrospective study from Wuhan, China, reported that 53 of 214 COVID-19 patients (∼25%) had CNS symptoms, including 28 with headache, 36 with dizziness, and 16 with consciousness impairment (Mao et al., 2020). Notably, only ∼50% of those with CNS symptoms (27/53) showed severe respiratory problem. In another study carried out in France for 58 COVID-19 patients, 49 (84%) had neurological complications, including 40 with encephalopathy (Helms et al., 2020), which was defined based on alterations in mental status, such as confusion, agitation, disorientation, or delirium. The alterations in mental status were most likely caused by systemic disorder, rather than encephalitis resulted from direct SARS-CoV-2 infection, because only a few cases showed modest amount of SARS-CoV-2 RNA in the cerebrospinal fluid (CSF) (Huang et al., 2020b; Moriguchi et al., 2020).
Table 1.
Neurological and neuropsychiatric features of COVID-19 patients.
| Neurological symptoms | Etiology/pathophysiology | References |
|---|---|---|
|
Neuroimaging, viral presence in CSF, inflammatory factors | Poyiadji et al. (2020); Mao et al. (2020); Pleasure et al. (2020); Jin et al. (2020); Ellul et al. (2020) |
|
Neuroimaging, viral presence in CSF, CNS infection, spine imaging | Helms et al. (2020); Poyiadji et al. (2020); Mao et al. (2020); Duong et al. (2020); Asadi-Pooya and Simani (2020) |
|
PNS infection, nerve demyelination, neuromuscular junction, immune dysregulation | Sedaghat and Karimi (2020); (,Zhao et al., 2020a) |
|
Olfactory viral invasion, CNS autoimmunity | Poyiadji et al. (2020); Najjar et al. (2020); Mao et al. (2020); Sohal and Mansur (2020) |
| Seizure | Neuroimaging, viral presence in CSF | Mao et al. (2020); Poyiadji et al. (2020); Duong et al. (2020); Sohal and Mansur (2020) |
|
Not reported | Mao et al. (2020); Rogers et al. (2020); Varatharaj et al. (2020) |
In addition to encephalopathy, cerebrovascular manifestations are also seen in some of COVID-19 patients. Ischemic stroke has been reported in 1%–6% COVID-19 patients (Klok et al., 2020; Mao et al., 2020; Merkler et al., 2020; Yaghi et al., 2020), and notably, the rates are several folds higher than that in patients with common influenza (Merkler et al., 2020). The COVID-19-associated endothelial dysfunction might be related with increased susceptibility to cerebrovascular disease (Klok et al., 2020).
Compared to influenza patients, much higher ratio of COVID-19 patients reported olfactory dysfunction or gustatory disorders (Ellul et al., 2020). In a multicenter European study of 417 COVID-19 patients, 85.6% reported decreased sense of smell, and 79.6% reported a complete loss of taste (Lechien et al., 2020). Although anosmia and ageusia are emerging as common symptoms of COVID-19, it remains unclear whether these abnormalities were caused by direct invasion of olfactory nerve or just because of coryza, which can occur in many other respiratory viral infections.
Systemic inflammation and dysregulated immune responses elicited after acute phase of viral infection are known to cause CNS or PNS neurological complications. Up to now, only rare cases of post-infectious CNS inflammations, typically encephalomyelitis and myelitis, were reported (Zanin et al., 2020; Zhao et al., 2020b; Zhang et al., 2021). More cases of Guillain–Barre syndrome resulted from immune attack on PNS were reported in COVID-19 patients, with evidence of axonal demyelination and consequent symptoms of muscle weakness, ataxia, or areflexia (Dinkin et al., 2020; Gutiérrez-Ortiz et al., 2020; Marta-Enguita et al., 2020; Toscano et al., 2020). Interestingly, a recent study showed that all the brain regions with SARS-CoV-2 signals had minimal lymphocyte or leukocyte infiltration, suggesting that SARS-CoV-2 may not elicit strong post-infectious immune response (Song et al., 2021). This feature is distinct from other neurotropic viruses like ZIKV, rabies virus, and herpes virus. In line with this notion, SARS-CoV-2-infected brain organoids exhibited little change in the pathways related with defense response to virus, cellular response to type I interferon, and innate immune response, which were observed in ZIKV-infected organoids (Song et al., 2021).
Neurotropic characteristics of SARS-CoV-2
Of the seven types of coronaviruses that infect humans, at least two strains have been shown to be able to enter and persist in the brain, including SARS-CoV (Arbour et al., 2000; Gu et al., 2005). The high similarity of SARS-CoV-2 in its nucleotide sequence to SARS-CoV, in particular the receptor-binding domain, indicates that it may also invade the host cells through ACE2 (Lu et al., 2020; Wang et al., 2020a). In support of this conclusion, intranasal infection of mice expressing human ACE2 with SARS-CoV-2 caused high viral loads in the lung, trachea, and brain (Sun et al., 2020).
In line with frequently observed loss of smell in COVID-19 patients, ACE2 and TMPRSS2 that mediates proteolytic processing of the S protein were observed in the olfactory epithelium, but not olfactory neurons, in both murine and human samples (Bilinska et al., 2020; Brann et al., 2020). Varied expression of ACE2 was observed in human neurons in a subtype- and region-specific manner, and high expression of ACE2 and TMPRSS2 was observed in pericytes, endothelial cells, or choroid plexus cells (He et al., 2020; Jacob et al., 2020; Ramani et al., 2020; Chen et al., 2021; Song et al., 2021).
Other evidence supporting the neurotropism of SARS-CoV-2 came from in vitro cellular or organoid models. In a cultured human brain organoid model, SARS-CoV-2 exhibited neuroinvasive ability in an ACE2-dependent manner, leading to neuronal death (Song et al., 2021). Several independent studies using brain organoids have also shown neurotropism of SARS-CoV-2 in their brain organoid systems (Ramani et al., 2020; Yang et al., 2020; Zhang et al., 2020). However, the results for targeted cell types were inconsistent among different studies. Some studies showed that SARS-CoV-2 preferably targets mature neurons (Ramani et al., 2020; Song et al., 2021), whereas other studies state that SARS-CoV-2 cannot target neuronal cell types (Pellegrini et al., 2020) or only has minimal neuronal infection ability (Jacob et al., 2020). Interestingly, in the brain organoids of hippocampal-like regions, SARS-CoV-2 signals were found to be enriched in choroid plexus epithelia-like cells and SARS-CoV-2 entry can lead to damages for the integrity of choroid plexus epithelium (Jacob et al., 2020; Pellegrini et al., 2020), suggesting that barrier disruption might facilitate SARS-CoV-2 invasion into the brain.
It is worth noting that SARS-CoV-2-positive neurons in human brain organoids exhibit mis-localization of axonal protein Tau to the cell soma and aberrant Tau phosphorylation, a phenomenon often seen in tauopathy-related neurodegeneration diseases (Ramani et al., 2020). In accordance with positive association between the ApoE4, a strong genetic risk factor for Alzheimer disease, and severe COVID-19 (Kuo et al., 2020), the ApoE4/4 neurons, astrocytes, or brain organoids exhibit increased rate of SARS-CoV-2 infection (Wang et al., 2021). These results indicate that particular attention should be paid to the potential long-term effects of SARS-CoV-2 infection on different patient populations, in particular the neurological and neuropsychological morbidities.
In histological analyses of the deceased individuals with COVID-19, SARS-CoV-2 RNA and protein were found in epithelial tissues of the olfactory mucosa and defined CNS regions receiving olfactory neuronal projections (Meinhardt et al., 2021). Notably, the SARS-CoV-2 S protein was observed in cortical neurons of ischemic regions in brains of some cases of postmortem COVID-19 patients, who were diagnosed with severe encephalopathy and multiple ischemic infarcts (Song et al., 2021). These studies have provided direct evidence supporting neurotropism of SARS-CoV-2 in COVID-19 patients and substantiated the findings obtained in human brain organoids.
Possible mechanisms of neurological manifestations
Viral entry to the brain via the olfactory bulb is a possible route given frequent anosmia in COVID-19 patients (Figure 1). In support of this possibility, the brain has been shown to be one of the major organs in hACE2 transgenic mice after intranasal infection with either SARS-CoV or SARS-CoV-2 (Tseng et al., 2007; Netland et al, 2008; Sun et al., 2020). Notably, magnetic resonance imaging analysis in a COVID-19 patient with anosmia showed a transient hyperintensity in the olfactory cortex, as well as a subtle hyperintensity in the olfactory bulb suggestive of viral infection (Politi et al., 2020). The presence of SARS-CoV-2 RNA and protein in the olfactory mucosa, its neuronal projections, and several CNS regions has provided direct evidence supporting the olfactory route of CNS entry by SARS-CoV-2 (Meinhardt et al., 2021). The presence of SARS-CoV-2 in defined neuroanatomical areas receiving olfactory tract projections, including the primary respiratory and cardiovascular control center, raises the possibility of axonal transport and/or trans-synaptic spread of virus (Meinhardt et al., 2021; Zubair et al., 2020). The trans-synaptic spread of virus has been demonstrated for several other types of coronaviruses, such as HCoV-OC43 (Dubéet al., 2018).
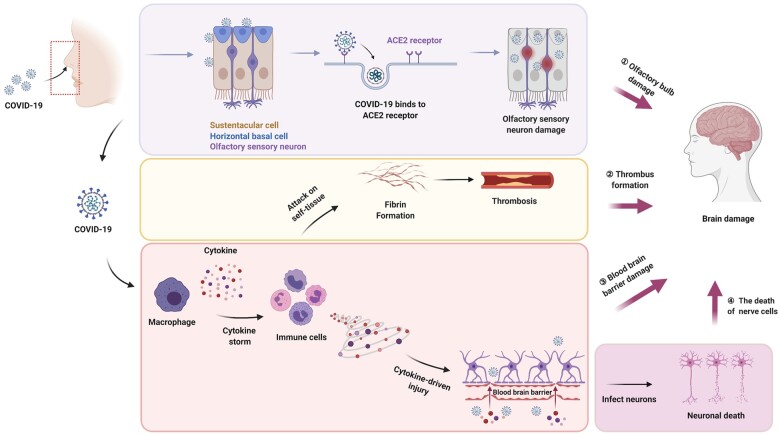
Figure 1.
Possible mechanism for the effects of SARS-CoV-2 on the nervous system. SARS-CoV-2 may cause damage to the nervous system through direct or indirect pathways. The direct infection of SARS-CoV-2 into sustentacular cells and horizontal basal cells impairs the function of olfactory neurons, which leads to the loss of smell sense and may further cause brain damage. SARS-CoV-2 induces an immune response after it enters the human body. The cytokine storm formed in the process of immune response can cause thrombosis and destruction of the BBB, and thus indirectly cause brain damage. When the BBB is broken, SARS-CoV-2 may cross the barrier and directly infect nerve cells by binding to specific receptors on nerve cells.
Pathogens like viruses can enter the blood and circulate to cerebrovascular after crossing the blood–brain barrier (BBB), following viremia or through infected immune cells (Bohmwald et al., 2018). Of note, remarkable percentage of the blood specimen, varying from 1% to 22%, derived from COVID-19 patients, displayed SARS-CoV-2 RNAemia suggestive of viremia (Huang et al., 2020a; Peng et al., 2020; Wang et al., 2020,b). The lack of detailed clinical information of patients limits the correlation of viremia with prognosis or disease course. The BBB formed by tight junctions consisted of endothelial cells together with surrounding pericytes and astrocytes plays a crucial role in maintaining the homeostasis of the brain and prevents pathogen invasion into the brain. Virions in the blood can directly attach to endothelial and pericytes through viral entry receptors for SARS-CoV-2, leading to alterations of the tight junction. High expression of ACE2 and TMPRSS2 in pericytes and endothelial cells supports this possibility (He et al., 2020; Chen et al., 2021). The transcytosis transport across the endothelia and pericytes by endocytic vesicles is an alternative pathway, as that done by other neuroinvasive respiratory viruses (Suen et al., 2014; Bohmwald et al., 2018). The presence of SARS-CoV-2 S protein in cerebral and leptomeningeal endothelial cells also supports the possible route of viral entry along CNS endothelia (Meinhardt et al., 2021).
Dysregulated immune response like cytokine storm is a common phenomenon in many severe COVID-19 patients (Mehta et al., 2020). High levels of inflammatory cytokines saturate in the blood can cause breakdown of the BBB (Poyiadji et al., 2020; Quirch et al., 2020), which may facilitate virion entry into the brain (Figure 1). Furthermore, cytokines may cause damage to the CNS by facilitating the formation of thrombosis, leading to cerebrovascular diseases (Hoffmann et al., 2020; Figure 1). It has been suggested that the infected immune cells can cross the BBB via the paracellular route (Suen et al., 2014; Bohmwald et al., 2018). Indeed, a report demonstrated that circulating monocytes, B lymphocytes, and CD4+ T lymphocytes are the main cell types frequently detected with positive signals for SARS-CoV-2 (Pontelli et al., 2020). Interestingly, immune cells with SARS-CoV-2 settled down in the lung of COVID-19 patients (Pontelli et al., 2020), implying the ‘Trojan Horse’ invasion mechanism—infected immune cell may mediate viral invasion into organs, including the brain. This entry strategy has been suggested to be used by immunodeficiency virus (HIV-1) (Albright et al., 2003; Spindler and Hsu, 2012) and another coronavirus HCoV-229E (Desforges et al., 2007). Further studies are needed to confirm whether SARS-CoV-2 entry into the brain is also via this route.
Conclusion and perspective of future study
In addition to the potential direct cytolytic effect, SARS-CoV-2 can also lead to brain dysfunction and neuronal damage through indirect systemic effects such as secondary inflammatory response (Van den Pol, 2006). In the absence of direct viral invasion, the neurotoxicity of the cytokine storm itself may cause damage to the CNS, such as acute necrotizing encephalopathy (Ouattara et al., 2011). Besides, cytokines may also cause brain damage by activating the thrombotic pathway (Hoffmann et al., 2020), which may lead to ischemic stroke.
Although many reports demonstrated that SARS-CoV-2 could cause nervous system diseases, the underlying mechanism is not clear. The effects of SARS-CoV-2 on the CNS most likely involve the direct entry of viruses into neurons, which causes neuronal cell death, and indirect pathway through the cytokine storm and thrombosis (Figure 1). In addition, SARS-CoV-2 may invade the CNS through axonal transport and trans-synaptic transfer via the olfactory nerve and through infection of pericytes or endothelial cells that make up BBB or leukocytes that pass through BBB (Zubair et al., 2020; Alquisiras-Burgos et al., 2021; Figure 1). These possibilities can be thoroughly verified using animal models such as hACE2 transgenic mice, which can be used to resemble the systemic effects of the disease.
Besides ACE2, other receptors and accessary proteins have been shown to facilitate SARS-CoV-2 entry into the cell. What factors mediate the interaction of SARS-CoV-2 with human brain remains to be studied systematically. Their expression and subcellular localization in human cell types, including various neurons, glia, and vascular cells, would be essential information. Furthermore, CNS control of systematic functions, such as autonomic pathways that control breathing, would help to disentangle neurological and systematic complications. Given the ongoing trends of COVID-19 pandemic, the long-term neurological and psychiatric sequelae of SARS-CoV-2 infection need serious consideration and follow-up tracking studies.
Acknowledgements
Due to the rapid progress in the field, the authors apologize for the ignorance of other relevant works.
Funding
This study was partially supported by grants from the Frontier Key Project of the Chinese Academy of Sciences (QYZDJ-SSW-SMC025), Shanghai Municipal Science and Technology Projects (2018SHZDZX05 and 201409001700), and the National Key R&D Program of China (2017YFA0700500).
Conflict of interest: none declared.
References
- Albright A.V., Soldan S.S., González-Scarano F. (2003). Pathogenesis of human immunodeficiency virus-induced neurological disease. J. Neurovirol. 9, 222–227. [DOI] [PubMed] [Google Scholar]
- Alquisiras-Burgos I., Peralta-Arrieta I., Alonso-Palomares, et al. (2021). Neurological complications associated with the blood–brain barrier damage induced by the inflammatory response during SARS-CoV-2 infection. Mol. Neurobiol. 58, 520–535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arbour N., Day R., Newcombe J., et al. (2000). Neuroinvasion by human respiratory coronaviruses. J. Virol. 74, 8913–8921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asadi-Pooya A.A., Simani L. (2020). Central nervous system manifestations of COVID-19: a systematic review. J. Neurol. Sci. 413, 116832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger J.R. (2020). COVID-19 and the nervous system. J. Neurovirol. 26, 143–148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bilinska K., Jakubowska P., Von Bartheld C.S., et al. (2020). Expression of the SARS-CoV-2 entry proteins, ACE2 and TMPRSS2, in cells of the olfactory epithelium: identification of cell types and trends with age. ACS Chem. Neurosci. 11, 1555–1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohmwald K., Galvez N.M.S., Rios M., et al. (2018). Neurologic alterations due to respiratory virus infections. Front. Cell. Neurosci. 12, 386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brann D.H., Tsukahara T., Weinreb C., et al. (2020). Non-neuronal expression of SARS-CoV-2 entry genes in the olfactory system suggests mechanisms underlying COVID-19-associated anosmia. Sci. Adv. 6, eabc5801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantuti-Castelvetri L., Ojha R., Pedro L.D., et al. (2020). Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity. Science 370, 856–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen R., Wang K., Yu J., et al. (2021). The spatial and cell-type distribution of SARS-CoV-2 receptor ACE2 in the human and mouse brains. Front. Neurol. 11, 573095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coutard B., Valle C., de Lamballerie X., et al. (2020). The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same clade. Antiviral Res. 176, 104742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daly J.L., Simonetti B., Klein K., et al. (2020). Neuropilin-1 is a host factor for SARS-CoV-2 infection. Science 370, 861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desforges M., Miletti T.C., Gagnon M., et al. (2007). Activation of human monocytes after infection by human coronavirus 229E. Virus Res. 130, 228–240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Wit E., van Doremalen N., Falzarano D., et al. (2016). SARS and MERS: recent insights into emerging coronaviruses. Nat. Rev. Microbiol. 14, 523–534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinkin M., Gao V., Kahan J., et al. (2020). COVID-19 presenting with ophthalmoparesis from cranial nerve palsy. Neurology 95, 221. [DOI] [PubMed] [Google Scholar]
- Dubé M., Le Coupanec A., Wong A.H., et al. (2018). Axonal transport enables neuron-to-neuron propagation of human coronavirus OC43. J. Virol. 92, e00404-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duong L., Xu P., Liu A. (2020). Meningoencephalitis without respiratory failure in a young female patient with COVID-19 infection in Downtown Los Angeles, early April 2020. Brain Behav. Immun. 87, 33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellul M.A., Benjamin L., Singh B., et al. (2020). Neurological associations of COVID-19. Lancet Neurol. 19, 767–783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glass W.G., Subbarao K., Murphy B., et al. (2004). Mechanisms of host defense following severe acute respiratory syndrome-coronavirus (SARS-CoV) pulmonary infection of mice. J. Immunol. 173, 4030. [DOI] [PubMed] [Google Scholar]
- Garg S., Kim L., Whitaker M., et al. (2020). Hospitalization rates and characteristics of patients hospitalized with laboratory-confirmed coronavirus disease 2019—COVID-NET, 14 states, March 1–30, 2020. Morb. Mortal. Wkly. Rep. 69, 458–464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giustozzi M., Acciarresi M., Agnelli G., et al. (2020). Safety of anticoagulation in patients treated with urgent reperfusion for ischemic stroke related to atrial fibrillation. Stroke 51, 2347–2354. [DOI] [PubMed] [Google Scholar]
- Grasselli G., Zangrillo A., Zanella A., et al. (2020). Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy Region, Italy. JAMA 323, 1574–1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guan W.J., Ni Z.Y., Hu Y., et al. (2020). Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 382, 1708–1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu J., Gong E., Zhang B., et al. (2005). Multiple organ infection and the pathogenesis of SARS. J. Exp. Med. 202, 415–424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutiérrez-Ortiz C., Méndez-Guerrero A., Rodrigo-Rey S., et al. (2020). Miller Fisher syndrome and polyneuritis cranialis in COVID-19. Neurology 95, e601. [DOI] [PubMed] [Google Scholar]
- He L., Mäe M.A., Muhl L., et al. (2020). Pericyte-specific vascular expression of SARS-CoV-2 receptor ACE2—implications for microvascular inflammation and hypercoagulopathy in COVID-19. bioRxiv, 10.1101/2020.05.11.088500 [DOI] [Google Scholar]
- Helms J., Kremer S., Merdji H., et al. (2020). Neurologic features in severe SARS-CoV-2 infection. N. Engl. J. Med. 382, 2268–2270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., et al. (2020). SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 181, 271–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C., Wang Y., Li X., et al. (2020a). Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395, 497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang Y.H., Jiang D., Huang J.T. (2020b). SARS-CoV-2 detected in cerebrospinal fluid by PCR in a case of COVID-19 encephalitis. Brain Behav. Immun. 87, 149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iadecola C., Anrather J., Kamel H. (2020). Effects of COVID-19 on the nervous system. Cell 183, 16–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacob F., Pather S.R., Huang W.-K., et al. (2020). Human pluripotent stem cell-derived neural cells and brain organoids reveal SARS-CoV-2 neurotropism predominates in choroid plexus epithelium. Cell Stem Cell 27, 937–950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin Y., Ji W., Yang H., et al. (2020). Endothelial activation and dysfunction in COVID-19: from basic mechanisms to potential therapeutic approaches. Signal Transduct. Target. Ther. 5, 293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawakami A., Kitsukawa T., Takagi S., et al. (1996). Developmentally regulated expression of a cell surface protein, neuropilin, in the mouse nervous system. J. Neurobiol. 29, 1–17. [DOI] [PubMed] [Google Scholar]
- Klok F.A., Kruip M.J.H.A., van der Meer N.J.M., et al. (2020). Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 191, 145–147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo C.-L., Pilling L.C., Atkins J.L., et al. (2020). APOE e4 genotype predicts severe COVID-19 in the UK biobank community cohort. J. Gerontol. A Biol. Sci. Med. Sci. 75, 2231–2232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lechien J.R., Chiesa-Estomba C.M., De Siati D.R., et al. (2020). Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study. Eur. Arch. Otorhinolaryngol. 277, 2251–2261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li K., Wohlford-Lenane C., Perlman S., et al. (2016). Middle East Respiratory Syndrome coronavirus causes multiple organ damage and lethal disease in mice transgenic for human dipeptidyl peptidase 4. J. Infect. Dis. 213, 712–722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W., Moore M.J., Vasilieva N., et al. (2003). Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 426, 450–454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu R., Zhao X., Li J., et al. (2020). Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet 395, 565–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao L., Jin H., Wang M., et al. (2020). Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 77, 683–690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marta-Enguita J., Rubio-Baines I., Gastón-Zubimendi I. (2020). Fatal Guillain–Barre syndrome after infection with SARS-CoV-2. Neurologia 35, 265–267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta P., McAuley D. F., Brown M., et al. (2020). COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 395, 1033–1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meinhardt J., Radke J., Dittmayer C., et al. (2021). Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nat. Neurosci. 24, 168–175. [DOI] [PubMed] [Google Scholar]
- Merkler A.E., Parikh N.S., Mir S., et al. (2020). Risk of ischemic stroke in patients with coronavirus disease 2019 (COVID-19) vs patients with influenza. JAMA Neurol. 77, 1366–1372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moriguchi T., Harii N., Goto J., et al. (2020). A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 94, 55–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Najjar S., Najjar A., Chong D.J., et al. (2020). Central nervous system complications associated with SARS-CoV-2 infection: integrative concepts of pathophysiology and case reports. J. Neuroinflammation 17, 231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Netland J., Meyerholz D.K., Moore S., et al. (2008). Severe acute respiratory syndrome coronavirus infection causes neuronal death in the absence of encephalitis in mice transgenic for human ACE2. J. Virol. 82, 7264–7275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ouattara L.A., Barin F., Barthez M.A., et al. (2011). Novel human reovirus isolated from children with acute necrotizing encephalopathy. Emerg. Infect. Dis. 17, 1436–1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oxley T.J., Mocco J., Majidi S., et al. (2020). Large-vessel stroke as a presenting feature of Covid-19 in the young. N. Engl. J. Med. 382, e60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pellegrini L., Albecka A., Mallery D.L., et al. (2020). SARS-CoV-2 infects the brain choroid plexus and disrupts the blood–CSF barrier in human brain organoids. Cell Stem Cell 27, 951–961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng L., Liu J., Xu W., et al. (2020). SARS-CoV-2 can be detected in urine, blood, anal swabs, and oropharyngeal swabs specimens. J. Med. Virol. 92, 1676–1680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pleasure S.J., Green A.J., Josephson S.A. (2020). The spectrum of neurologic disease in the severe acute respiratory syndrome Coronavirus 2 pandemic infection: neurologists move to the frontlines. JAMA Neurol. 77, 679–680. [DOI] [PubMed] [Google Scholar]
- Politi L.S., Salsano E., Grimaldi M. (2020). Magnetic resonance imaging alteration of the brain in a patient with coronavirus disease 2019 (COVID-19) and anosmia. JAMA Neurol. 77, 1028–1029. [DOI] [PubMed] [Google Scholar]
- Pontelli M., Castro I., Martins R., et al. (2020). Infection of human lymphomononuclear cells by SARS-CoV-2. bioRxiv, 10.1101/2020.07.28.225912 [DOI] [Google Scholar]
- Poyiadji N., Shahin G., Noujaim D., et al. (2020). COVID-19-associated acute hemorrhagic necrotizing encephalopathy: imaging features. Radiology 296, e119–e120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quirch M., Lee J., Rehman S. (2020). Hazards of the cytokine storm and cytokine-targeted therapy in patients with COVID-19: review. J. Med. Internet. Res . 22, e20193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramani A., Müller L., Ostermann P.N., et al. (2020). SARS-CoV-2 targets neurons of 3D human brain organoids. EMBO J. 39, e106230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers J.P., Chesney E., Oliver D., et al. (2020). Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry 7, 611–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sedaghat Z., Karimi N. (2020). Guillain Barre syndrome associated with COVID-19 infection: a case report. J. Clin. Neurosci. 76, 233–235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shang J., Wan Y., Luo C., et al. (2020). Cell entry mechanisms of SARS-CoV-2. Proc. Natl Acad. Sci. USA 117, 11727–11734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sohal S., Mansur M. (2020). COVID-19 presenting with seizures. IDCases 20, e00782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song E., Zhang C., Israelow B., et al. (2021). Neuroinvasion of SARS-CoV-2 in human and mouse brain. J. Exp. Med. 218, e20202135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spindler K.R., Hsu T.-H. (2012). Viral disruption of the blood–brain barrier. Trends Microbiol. 20, 282–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suen W.W., Prow N.A., Hall R.A., et al. (2014). Mechanism of West Nile virus neuroinvasion: a critical appraisal. Viruses 6, 2796–2825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun S.-H., Chen Q., Gu H.-J., et al. (2020). A mouse model of SARS-CoV-2 infection and pathogenesis. Cell Host Microbe 28, 124–133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toscano G., Palmerini F., Ravaglia S., et al. (2020). Guillain–Barré syndrome associated with SARS-CoV-2. N. Engl. J. Med. 382, 2574–2576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tseng C.-T. K., Huang C., Newman P., et al. (2007). Severe acute respiratory syndrome coronavirus infection of mice transgenic for the human Angiotensin-converting enzyme 2 virus receptor. J. Virol. 81, 1162–1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van den Pol A.N. (2006). Viral infections in the developing and mature brain. Trends Neurosci. 29, 398–406. [DOI] [PubMed] [Google Scholar]
- Varatharaj A., Thomas N., Ellul M.A., et al. (2020). Neurological and neuropsychiatric complications of COVID-19 in 153 patients: a UK-wide surveillance study. Lancet Psychiatry 7, 875–882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viguier A., Delamarre L., Duplantier J., et al. (2020). Acute ischemic stroke complicating common carotid artery thrombosis during a severe COVID-19 infection. J. Neuroradiol. 47, 393–394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walls A.C., Park Y.J., Tortorici M.A., et al. (2020). Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell 181, 281–292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C., Zhang M., Garcia G., et al. (2021). ApoE-isoform-dependent SARS-CoV-2 neurotropism and cellular response. Cell Stem Cell 28, 331–342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q., Zhang Y., Wu L., et al. (2020a). Structural and functional basis of SARS-CoV-2 entry by using human ACE2. Cell 181, 894–904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang W., Xu Y., Gao R., et al. (2020b). Detection of SARS-CoV-2 in different types of clinical specimens. JAMA 323, 1843–1844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yaghi S., Ishida K., Torres J., et al. (2020). SARS-CoV-2 and stroke in a New York healthcare system. Stroke 51, 2002–2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L., Han Y., Nilsson-Payant B.E., et al. (2020). A human pluripotent stem cell-based platform to study SARS-CoV-2 tropism and model virus infection in human cells and organoids. Cell Stem Cell 27, 125–136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zanin L., Saraceno G., Panciani P.P., et al. (2020). SARS-CoV-2 can induce brain and spine demyelinating lesions. Acta Neurochir. 162, 1491–1494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang B.-Z., Chu H., Han S., et al. (2020). SARS-CoV-2 infects human neural progenitor cells and brain organoids. Cell Res. 30, 928–931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang T., Hirsh E., Zandieh S., et al. (2021). COVID-19-associated acute multi-infarct encephalopathy in an asymptomatic CADASIL patient. Neurocrit. Care 34, 1099–1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao, H., , Shen,D., , Zhou,H., , et al, . (2020a). Guillain–Barré syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol. 19, 383–384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao K., Huang J., Dai D., et al. (2020b). Acute myelitis after SARS-CoV-2 infection: a case report. medRxiv, 10.1101/2020.03.16.20035105 [DOI] [Google Scholar]
- Zhou P., Yang X.L., Wang X.G., et al. (2020). A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579, 270–273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zubair A.S., McAlpine L.S., Gardin T., et al. (2020). Neuropathogenesis and neurologic manifestations of the coronaviruses in the age of coronavirus disease 2019: a review. JAMA Neurol. 77, 1018–1027. [DOI] [PMC free article] [PubMed] [Google Scholar]