Abstract
Background
N6-methyladenosine (m6A) is the most common internal modification present in mRNAs and long noncoding RNAs (lncRNAs), associated with tumorigenesis and cancer progression. However, little is known about the roles of m6A and its regulatory genes in nonsmall cell lung cancer (NSCLC). Here, we systematically explored the roles and prognostic significance of m6A-associated regulatory genes in NSCLC.
Methods
The copy number variation (CNV), mutation, mRNA expression data, and corresponding clinical pathology information of 1057 NSCLC patients were downloaded from the cancer genome atlas (TCGA) database. The gain and loss levels of CNVs were determined by utilizing segmentation analysis and GISTIC algorithm. The GSEA was conducted to explore the functions related to different levels of m6A regulatory genes. Logrank test was utilized to assess the prognostic significance of m6A-related gene's CNV.
Results
The genetic alterations of ten m6A-associated regulators were identified in 102 independent NSCLC samples and significantly related to advanced tumor stage. Deletions or shallow deletions corresponded to lower mRNA expression while copy number gains or amplifications were related to increased mRNA expression of m6A regulatory genes. Survival analysis showed the patients with copy number loss of FTO with worse disease-free survival (DFS) or overall survival (OS). Besides, copy number loss of YTHDC2 was also with poor OS for NSCLC patients. Moreover, high FTO expression was significantly associated with oxidative phosphorylation, translation, and metabolism of mRNA.
Conclusion
Our findings provide novel insight for better understanding of the roles of m6A regulators and RNA epigenetic modification in the pathogenesis of NSCLC.
1. Introduction
Lung cancer is one of the most prevalent malignant tumors and also the most lethal cancer with an approximate 5-year survival rate of 16% all over the world [1]. The latest cancer statistic data indicate that there will be 19,300,000 new tumor patients and more than 10,000,000 deaths in 2020 [1, 2]. Nonsmall cell lung cancer (NSCLC) is the most prevalent type of lung tumor which accounts for 80% of all cases. In the past decades, a series of therapeutics including chemotherapy, surgery, radiotherapy, and immunotherapy were applied to lung tumor patients; however, the prognosis of patients is still unfavorable and is especially poor in advanced NSCLC [3]. NSCLC has become a serious health problem worldwide. Hence, to further explore the molecular pathogenesis underlying NSCLC to develop effective diagnostics and therapies is urgently needed.
The genetic and epigenetic alterations of nucleotides were involved in various regular bioprocesses such as regulation of gene expression, variable splicing, and protein translation, which play essential roles in the occurrence and progression of various diseases [4–6]. RNAs nucleotides modification is a common epigenetic alteration and more than 100 chemically modified nucleotides in different RNAs have been identified [7, 8]. Among these, methylation of N6-methyladenosine (m6A) is the most prevalent internal form of messenger RNAs (mRNAs) and long noncoding RNAs (lncRNAs) modification in eukaryotes [9]. Previous studies have shown that m6A modification is present in more than 7,600 mRNAs and in over 300 noncoding RNAs [10]. It is known as m6A modification closely related to RNA splicing, localization, stability, export, RNA-protein interactions, alternative polyadenylation, and translation [11–13]. The cellular m6A methylation is mediated by a group of regulatory enzymes including “writers” Wilms' tumor 1-associated protein (WTAP), methyltransferase-like 3 (METTL3) and METTL14, “erasers” fat mass and obesity-associated protein (FTO) and alkB homolog 5 (ALKBH5), and “readers” YTH domain containing 1 (YTHDC1/2) and YTH N6-methyladenosine RNA binding protein 1/2/3 (YTHDF1/2/3) [14–16]. Generally, m6A methylation is increased by writers, erased by FTO or ALKBH5, and deciphered by YTHDF1/2/3 or YTHDC1/2.
In recent years, growing evidences indicated that m6A dysregulation plays critical roles in tumorigenesis and cancer progression through diverse molecular mechanisms [14–18]. Meanwhile, the expression level of m6A-related regulatory proteins has been shown to be critically involved in tumorigenesis [19–21]. Knockout of m6A methyltransferase can regulate cancer occurrence by affecting the activity of p53 signaling pathway [22]. More recently, downregulation of FTO reduced lung cancer cell proliferation and invasion and promoted cell apoptosis [23, 24]. Another study also revealed that METTL14 by regulating its mRNA targets promotes leukemogenesis through mRNA m6A modification [25]. All these results show that m6A modification factors play essential roles in the occurrence of a variety of cancers. However, the connection between m6A-related regulatory factors and NSCLC remained not very clear. In the present work, we systematically explored the expression pattern of m6A regulators in NSCLC based on the data from TCGA database. We also analyzed the association between m6A-related genetic alterations and clinical features including age, sex, pathological stage, disease-free survival (DFS), and overall survival (OS).
2. Materials and Methods
2.1. Acquisition of NSCLC Data
The CNV, mutation, mRNA expression information, and corresponding clinicopathological information of 1057 NSCLC patients were obtained from the TCGA database (GDC data portal) (https://cancergenome.nih.gov/).
2.2. Data Preprocessing and Copy Number Variations Identifying
The gain and loss levels of copy number variations (CNVs) were determined by utilizing segmentation analysis and GISTIC algorithm.
The NSCLC samples were grouped into two classes: without CNVs and mutation of ten m6A regulators and with CNVs and/or mutation. The expression of mRNA in each CNV groups was calculated using R package “DESeq2.”
2.3. Gene Set Enrichment Analysis (GSEA)
The GSEA was implemented to explore the functions related to different levels of m6A regulatory genes.
The JAVA program with MSigDB v6.1 was used to execute GSEA. All samples were sorted into low- and high-FTO level groups. Then, significant enrichment of gene sets was calculated with a false discovery rate (FDR) value less than 0.25 and a normalized P value less than 0.05.
2.4. Survival Analysis
All NSCLC samples were grouped by with or without deletion/gain of each m6A regulator gene and then a survival analysis was conducted by utilizing R packages “survival” (https://cran.r-project.org/web/packages/survival/index.html) and R package “survminer” (https://cran.r-project.org/web/packages/survminer/index.html). The prognosis value of the CNV of m6A regulatory gene was assessed by logrank test. Moreover, the Kaplan–Meier plotter (https://kmplot.com/analysis/) was used to analyze the prognosis value of each m6A regulator.
2.5. Statistical Analysis
All data were processed by utilizing R (4.0). The relationship between the CNV of m6A regulators and clinical-pathological features was investigated with chi-square test or Kruskal–Wallis rank sum test. All P values less than 0.05 were treated to be statistically significant.
3. Results
3.1. Mutations and CNV Events of m6A Regulators in NSCLC Samples
In total, 1057 NSCLC specimens with sequencing data were included in the present research. Among these, the genetic alterations of m6A moderators were identified in 102 independent samples (Figure 1). In detail, the m6A “reader” genes YTHDF3 (8.8%, 93/1057), YTHDF1 (7.85%, 83/1057), and YTHDC2 (5.58%, 59/1057) are the top three genes in the CNVs frequency (Table 1, Figure 2(a)). Moreover, the CNVs of NSCLC-driven genes EGFR, KRAS, and TP53 were assessed and the results were 13.43%, 10.69%, and 6.53% (Table 1, Figure 2(a)), respectively. Subsequently, we counted all CNV patterns in NSCLC samples and found that the copy number loss events are the most of all CNVs (264/495) (Table 1, Figure 2(b)). The shallow deletions of YTHDC2 are the most frequent copy number loss of these ten m6A regulators, while the gain of YTHDF3 DNA copy number is the most common change in the CNVs of ten m6A regulatory genes (Table 1, Figure 2), suggesting a key significance of m6A reader genes in RNA m6A methylation in NSCLC patients.
Figure 1.
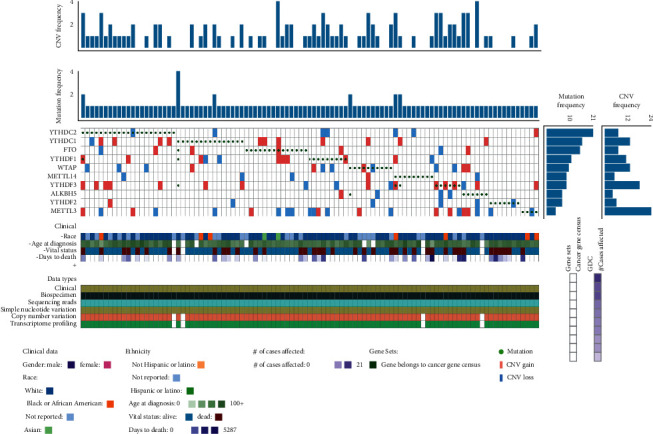
CNVs and mutations of top ten m6A regulators in NSCLC patients.
Table 1.
CNV patterns in NSCLC patients (N = 1057).
| Gene | Diploid | Deep deletion | Shallow deletion | Copy number gain | Amplification | CNV sum | Percentage | |
|---|---|---|---|---|---|---|---|---|
| Eraser | ALKBH5 | 999 | 0 | 45 | 11 | 2 | 58 | 5.49 |
| FTO | 1004 | 2 | 30 | 20 | 1 | 53 | 5.01 | |
|
| ||||||||
| Writer | METTL14 | 1034 | 1 | 21 | 1 | 0 | 23 | 2.18 |
| METTL3 | 1032 | 1 | 20 | 1 | 3 | 25 | 2.37 | |
| WTAP | 1004 | 1 | 39 | 12 | 1 | 53 | 5.01 | |
|
| ||||||||
| Reader | YTHDF1 | 974 | 0 | 8 | 67 | 8 | 83 | 7.85 |
| YTHDF2 | 1027 | 0 | 24 | 6 | 0 | 30 | 2.84 | |
| YTHDC1 | 1039 | 0 | 12 | 1 | 5 | 18 | 1.70 | |
| YTHDC2 | 998 | 1 | 53 | 4 | 1 | 59 | 5.58 | |
| YTHDF3 | 964 | 0 | 6 | 81 | 6 | 93 | 8.80 | |
|
| ||||||||
| Others | EGFR | 915 | 4 | 12 | 82 | 44 | 142 | 13.43 |
| KRAS | 944 | 4 | 9 | 72 | 28 | 113 | 10.69 | |
| TP53 | 988 | 4 | 55 | 8 | 2 | 69 | 6.53 | |
Figure 2.
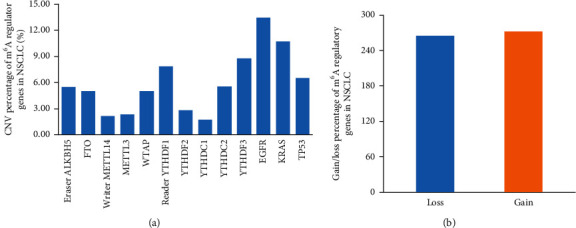
CNVs of ten m6A regulators in NSCLC. (a) CNV ratio of m6A regulators in NSCLC samples. (b) Number of gain or loss of DNA copy number of m6A-related regulators in NSCLC patients.
3.2. Association between the Alterations of m6A Regulators and Clinical-Pathological Characteristics
To investigate the connection between genetic variations of m6A regulators and the clinicopathological characteristics of NSCLC patients, we implemented a correlation analysis. The results showed that genetic alterations of m6A-related regulators were obviously correlated to advanced tumor stage (P < 0.05) (Table 2). Given that EGFR, TP53, and KRAS play crucial roles in tumorigenesis and progression of lung cancer, we examined the connection between m6A-related regulators and the variations of the above three tumor-related genes. We found that the variations of m6A regulatory genes were obviously linked to EGFR and TP53 alteration (P < 0.05) (Table 3). In detail, only 54 samples were missing from alterations of m6A regulators among the 316 patients with EGFR alteration and 57 samples were absent in 321 patients with TP53 alteration (Table 3). However, variations of m6A regulators did not correlate significantly with KRAS mutation (P > 0.05) (Table 3).
Table 2.
Clinical features of patients with NSCLC who are with or without genetic alterations of m6A regulators.
| Parameters | With mutation and/or CNVs | Without mutation and/or CNVs | P value | |
|---|---|---|---|---|
| Age | ≤60 | 90 | 176 | 0.217 |
| >60 | 223 | 524 | ||
|
| ||||
| Gender | Female | 123 | 283 | 0.787 |
| Male | 190 | 417 | ||
|
| ||||
| Primary diagnosis | Acinar cell carcinoma | 6 | 16 | 0.07 |
| Adenocarcinoma, NOS | 102 | 210 | ||
| Adenocarcinoma with mixed subtypes | 37 | 71 | ||
| Basaloid squamous cell carcinoma | 5 | 9 | ||
| Bronchiolo-alveolar carcinoma, mucinous | 2 | 3 | ||
| Bronchiolo-alveolar adenocarcinoma, NOS | 1 | 2 | ||
| Bronchiolo-alveolar carcinoma, nonmucinous | 6 | 12 | ||
| Clear cell adenocarcinoma, NOS | 1 | 1 | ||
| Micropapillary carcinoma, NOS | 0 | 3 | ||
| Mucinous adenocarcinoma | 1 | 12 | ||
| Papillary adenocarcinoma, NOS | 6 | 16 | ||
| Papillary squamous cell carcinoma | 1 | 3 | ||
| Signet ring cell carcinoma | 0 | 1 | ||
| Solid carcinoma, NOS | 2 | 4 | ||
| Squamous cell carcinoma, keratinizing, NOS | 3 | 9 | ||
| Squamous cell carcinoma, small cell, nonkeratinizing | 1 | 3 | ||
| Squamous cell carcinoma, NOS | 139 | 325 | ||
|
| ||||
| Tumor stage | Not reported | 3 | 8 | 0.001 ∗ |
| Stage I-II | 242 | 559 | ||
| Stage III-IV | 68 | 133 | ||
|
| ||||
| Tissue or organ of origin | Lower lobe, lung | 99 | 251 | 0.137 |
| Lung, NOS | 14 | 37 | ||
| Main bronchus | 4 | 5 | ||
| Middle lobe, lung | 14 | 23 | ||
| Overlapping lesion of lung | 6 | 5 | ||
| Upper lobe, lung | 176 | 379 | ||
NOS, not otherwise specified.
Table 3.
Relationship between EGFR/KRAS/TP53 and m6A genes.
| Gene | With alteration in 10 m6A genes | Without alteration in 10 m6A genes | χ 2 | P value | |
|---|---|---|---|---|---|
| EGFR | WT | 262 | 629 | 6.598 | 0.010 |
| n = 1023 | Alteration | 54 | 78 | ||
|
| |||||
| TP53 | WT | 264 | 694 | 96.505 | <0.001 |
| n = 1024 | Alteration | 57 | 9 | ||
|
| |||||
| KRAS | WT | 283 | 647 | 2.039 | 0.153 |
| n = 1036 | Alteration | 40 | 66 | ||
Subsequently, we further explored whether alterations in m6A-related genes affect mRNA expression. The results suggested that the expression of mRNA was significantly related to different CNV types in NSCLC patients. Deletions or shallow deletions were corresponding to lower mRNA expression while copy number gains or amplifications were linked to increased mRNA expression of the ten m6A regulators (Figure 3).
Figure 3.
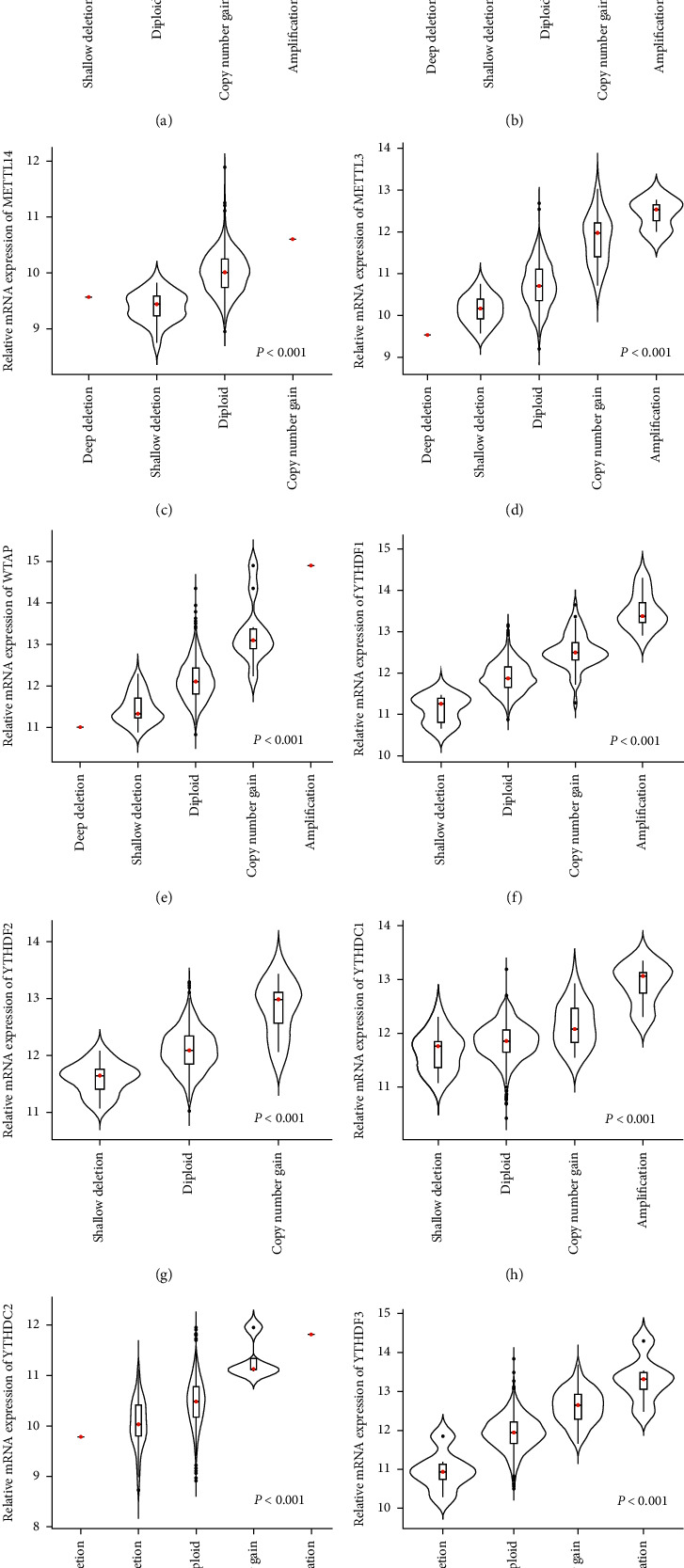
The relation between CNV types and m6A regulator expression.
3.3. Prognostic Significance of CNVs in m6A Regulatory Genes for NSCLC Patients
The prognostic significance of CNVs in the m6A regulators for DFS and OS among NSCLC patients was investigated, there was no significant correlation between patients with/without CNVs of m6A-related regulators and OS or DFS (Figures 4(a)-4(b)). However, further analysis showed copy number loss of FTO with worse DFS and OS (Figures 4(c)-4(d)). Besides, copy number loss of YTHDC2 is also with poor OS for NSCLC patients (Figure 4(e)). Furthermore, we further found low FTO expression was closely related to poor OS of lung cancer patients by using Kaplan–Meier plotter (Figure 4(f)).
Figure 4.
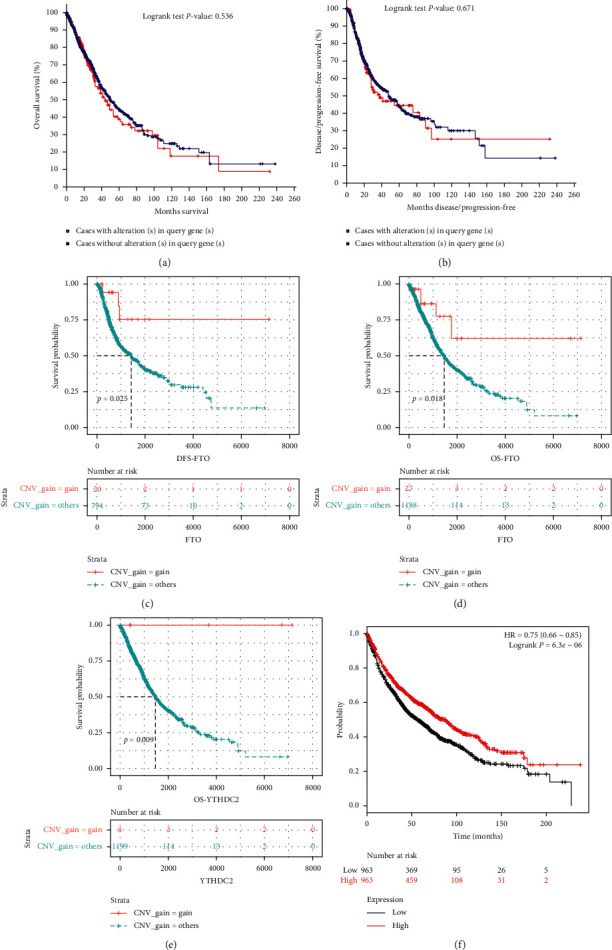
Survival analysis of NSCLC patients with CNVs of m6A-related regulators. ((a)-(b)) OS and DFS for NSCLC patients who have any alteration of m6A-related regulators, ((c)-(d)) DFS and OS for patients with NSCLC who have different CNV types of FTO, (e) OS for patients with NSCLC who have different CNV types of YTHDC2, and (f) OS for patients with FTO mRNA expression levels by Kaplan–Meier plotter.
3.4. Enrichment Analysis of FTO Gains of Function
Given that there is close correlation between CNVs and m6A regulator FTO and NSCLC prognosis, a GSEA was executed to investigate the gene enrichment in patients with different FTO mRNA expression. The results showed that high FTO expression was significantly associated with multiple key biological processes, including oxidative phosphorylation, ribosome, translation, 3′-UTR-mediated translational regulation, metabolism of mRNA, and influenza life cycle (Figure 5), which provides new clues for understanding the pathogenesis of NSCLC.
Figure 5.
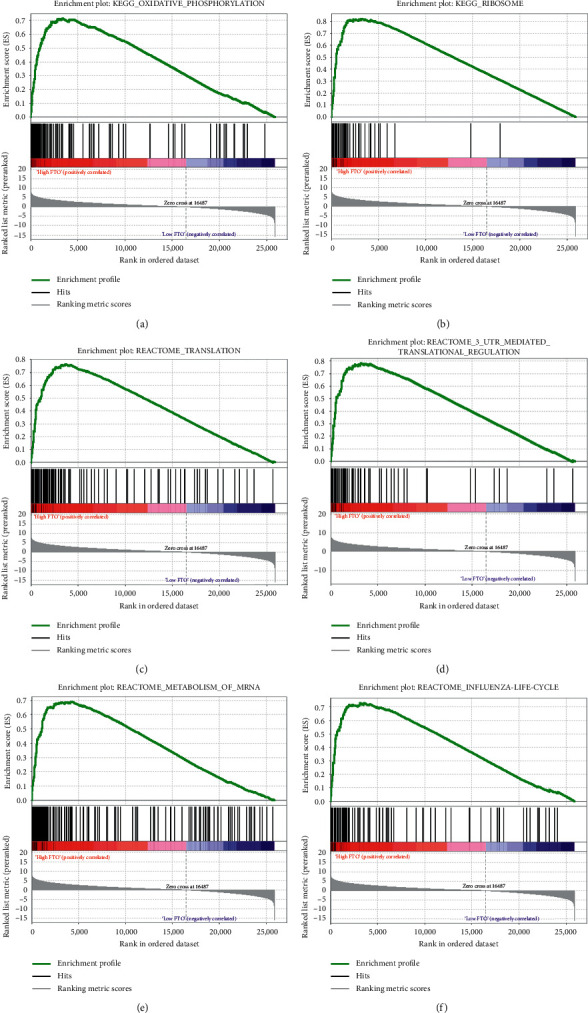
GSEA outcomes of different expression level of FTO. (a) Oxidative phosphorylation, (b) ribosome, (c) translation, (d) 3′-UTR-mediated translational regulation, (e) metabolism of mRNA, and (f) influenza life cycle.
4. Discussion
RNA modification is an emerging branch of epigenetics that is increasingly attracting the interest of related researchers. Currently, RNA modification is thought to be widespread in almost all forms of RNA, including mRNAs, lncRNAs, rRNAs, tRNAs, micro-RNAs, and small nucleolar RNAs [26–28]. Previous studies have shown that RNA modifications include multiple forms, such as pseudouridine, N7-methyladenosine, 2′-O-methylation, N1-methyladenosine, N6,2-O-dimethyladenosine (m6A), and 5-methylcytosine [29, 30]. Among them, m6A methylation is the most common mRNA modification form and it participates in the regulation of numerous biological processes in eukaryotes [29–31]. The cellular m6A status is dynamically regulated by methyltransferases, binding proteins, and demethylases. The alterations of these regulatory factors lead to dysregulation of m6A methylation and hence play an essential role in the progression of various diseases [31–33].
Bioinformatics analysis plays a fundamental role in disease diagnosis and pathogenesis research [34–36]. In this study, we used bioinformatics approaches to systematically identify the roles and prognostic values of m6A-related regulatory factors in NSCLC. A total of 1057 NSCLC samples with CNV information and clinical data from TCGA were included. Of these, 102 independent samples were identified as having the genetic variations of m6A-related regulators. Compared with clear cell renal cell carcinoma (ccRCC) and acute myeloid leukemia, the frequency of alterations in ten m6A-related genes in lung cancer is less [37, 38]. Among all CNV patterns in NSCLC, the copy number loss was the most important part of all CNV events, which was similar as the CNV patterns in ccRCC [37] and acute myeloid leukemia [38]. For all m6A regulators, deletions are the most important part of CNVs in “eraser” and “writer” genes, but the number of gain of CNV events in “eraser” genes is higher than those of “writer” genes, which eventually decreased the m6A level in NSCLC cells. Previous studies have shown that m6A levels were downregulated in various tumors, such as glioblastoma and breast cancer [39–41]. This may also be explained by the opposite effect on m6A status for“eraser” and “writer” genes.
Furthermore, we found that copy number gains or amplifications were positively correlated with mRNA levels of the 10 m6A-related regulators while deletions or shallow deletions were negatively related to mRNA levels, implying that alterations in CNVs affect m6A-related genes expression in NSCLC. Further analysis revealed that alterations of m6A-related regulators were positively linked to cancer stage of NSCLC. These results suggested that CNVs of m6A regulatory genes are involved in the regulation of tumor progression by affecting methylation modification of RNAs. A recent study found that overexpression of m6A methyltransferase METTL3 facilitates tumor development through AFF4/NF-κB/MYC signal pathway in bladder cancer [42]. Another study also indicated changes in the expression of m6A regulatory genes by regulating corresponding genes to promote breast cancer progression [41]. We also found that the changes of m6A modulators were significantly associated with EGFR and TP53 alteration. EGFR is an important oncogene, and its mediated pathways play crucial roles in tumor occurrence and development [43–45], while TP53 is a classic tumor suppressor gene [46]. Based on these findings, we speculate that dysregulated m6A status induced EGFR activation and TP53 inactivation to facilitate the tumorigenesis and progress of NSCLC.
Moreover, we explored the prognostic value of m6A regulator alterations in NSCLC. For all ten m6A-related genes, only copy number loss of eraser gene FTO and reader gene YTHDC2 was significantly associated with poor survival status for NSCLC patients. Besides, the results of Kaplan–Meier plotter analysis also indicate that low FTO expression is related to worse OS of lung cancer patients. Taking together, the present results showed that the FTO levels are inversely related to the survival time of patients with NSCLC. However, studies have revealed that FTO as an m6A demethylase participates in promoting the growth of lung cancer cells in vitro [23, 24]. These findings revealed that m6A regulatory genes are a “double-edged sword” in tumorigenesis, which could lead to not only tumor suppression but also tumor progression. Thus, restoring the balanced state of RNA methylation in tumor cells is a new anticancer strategy.
The present study displayed that m6A-related regulatory genes were also associated with multiple signaling pathways and biological processes of NSCLC occurrence and development. The results of the GSEA suggested that the expression of FTO was significantly related to oxidative phosphorylation, ribosome, translation, 3′-UTR-mediated translational regulation, and metabolism of mRNA. Similar to our results, a previous study reported that FTO expression was positively related to dextrose oxidation rates and levels of genes related to oxidative phosphorylation in skeletal muscle [47]. It has been shown that oxidative phosphorylation plays significant roles in lung cancer proliferation, invasion, metastasis, and drug resistance [48–50]. Therefore, it is likely that genetic alterations of FTO regulate the progression of NSCLC by affecting cellular oxidative phosphorylation levels. The specific molecular mechanism deserves to be explored through further work. In addition, several biological processes regulated by the m6A regulators have been identified, including RNA metabolism, translational regulation, and protein translation [23, 51], which are consistent with our GSEA results.
In conclusion, our work systematically displayed the genetic alterations, expression patterns, potential roles, and prognostic significance of m6A-related regulators in NSCLC and found that the alterations of m6A regulators are highly related to the malignant clinicopathological characteristics including survival. These results help us to find out the functions of m6A RNA methylation in the pathogenesis of NSCLC. However, these findings need to be validated with further clinical and molecular biology experiments.
Acknowledgments
This project was supported by the Natural Science Foundation of Hunan Province (2019JJ50425 and 2020JJ5909) and General Project of Hunan Provincial Education Department (18C1129).
Contributor Information
Tieming Zhou, Email: zhoutieming2008@126.com.
Wei Li, Email: xylw2015@csu.edu.cn.
Data Availability
Publicly available datasets were analyzed in this study. These data can be found at https://www.cancer.gov/.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Sung H., Ferlay J., Siegel R. L. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians. 2021;71:209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Siegel R. L., Miller K. D., Fuchs H. E., Jemal A. Cancer statistics. CA: A Cancer Journal for Clinicians. 2021;71:7–33. doi: 10.3322/caac.21654. [DOI] [PubMed] [Google Scholar]
- 3.Bray F., Ferlay J., Soerjomataram I., Siegel R. L., Torre L. A., Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 4.Grady W. M. Epigenetic alterations in the gastrointestinal tract: current and emerging use for biomarkers of cancer. Advances in Cancer Research. 2021;151:425–468. doi: 10.1016/bs.acr.2021.02.006. [DOI] [PubMed] [Google Scholar]
- 5.Koldobskiy M. A., Jenkinson G., Abante J., DiBlasi V. A. R., Zhou W., Pujadas E. Converging genetic and epigenetic drivers of paediatric acute lymphoblastic leukaemia identified by an information-theoretic analysis. Nature Biomedical Engineering. 2021;5:360–376. doi: 10.1038/s41551-021-00703-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rathore A. S., Birla H., Singh S. S., Zahra W., Dilnashin H., Singh R. Epigenetic modulation in Parkinson’s disease and potential treatment therapies. Neurochemical Research. 2021;46:1618–1626. doi: 10.1007/s11064-021-03334-w. [DOI] [PubMed] [Google Scholar]
- 7.Cantara W. A., Crain P. F., Rozenski J., et al. The RNA modification database, RNAMDB: 2011 update. Nucleic Acids Research. 2011;39:D195–D201. doi: 10.1093/nar/gkq1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Manfredonia I., Incarnato D. RNA post-transcriptional modification mapping data analysis using RNA framework. Methods in Molecular Biology. 2021;2298:3–13. doi: 10.1007/978-1-0716-1374-0_1. [DOI] [PubMed] [Google Scholar]
- 9.Luo J., Xu T., Sun K. N6-Methyladenosine RNA modification in inflammation: roles, mechanisms, and applications. Frontiers in Cell and Developmental Biology. 2021;9 doi: 10.3389/fcell.2021.670711.670711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Meyer K. D., Saletore Y., Zumbo P., Elemento O., Mason C. E., Jaffrey S. R. Comprehensive analysis of mRNA methylation reveals enrichment in 3′ UTRs and near stop codons. Cell. 2012;149:1635–1646. doi: 10.1016/j.cell.2012.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Han X., Guo J., Fan Z. Interactions between m6A modification and miRNAs in malignant tumors. Cell Death & Disease. 2021;12 doi: 10.1038/s41419-021-03868-5.598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Vasic R., Gao Y., Liu C., Halene S. The role of RNA epigenetic modification in normal and malignant hematopoiesis. Current Stem Cell Reports. 2020;6:144–155. doi: 10.1007/s40778-020-00178-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fang R., Ye L., Shi H. Understanding the roles of N(6)-methyladenosine writers, readers and erasers in breast cancer. Neoplasia. 2021;23:551–560. doi: 10.1016/j.neo.2021.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lan Q., Liu P. Y., Haase J., Bell J. L., Huttelmaier S., Liu T. The critical role of RNA m(6)A methylation in cancer. Cancer Research. 2019;79:1285–1292. doi: 10.1158/0008-5472.CAN-18-2965. [DOI] [PubMed] [Google Scholar]
- 15.Hagen F. V., Gundert L., Strick A., Klumper N., Schmidt D., Kristiansen G. N(6) -Methyladenosine (m(6) A) readers are dysregulated in renal cell carcinoma. Molecular Carcinogenesis. 2021;60:354–362. doi: 10.1002/mc.23297. [DOI] [PubMed] [Google Scholar]
- 16.Zou Z., Zhou S., Liang G., Tang Z., Li K., Tan S. The pan-cancer analysis of the two types of uterine cancer uncovered clinical and prognostic associations with m6A RNA methylation regulators. Molecular Omics. 2021;17:438–453. doi: 10.1039/d0mo00113a. [DOI] [PubMed] [Google Scholar]
- 17.Chen B., Li Y., Song R., Xue C., Xu F. Functions of RNA N6-methyladenosine modification in cancer progression. Molecular Biology Reports. 2019;46:1383–1391. doi: 10.1007/s11033-018-4471-6. [DOI] [PubMed] [Google Scholar]
- 18.Lin X., Chai G., Wu Y., Li J., Chen F., Liu J. RNA m(6)A methylation regulates the epithelial mesenchymal transition of cancer cells and translation of Snail. Nature Communications. 2019;10 doi: 10.1038/s41467-019-09865-9.2065 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 19.Yu J., Mao W., Sun S., Hu Q., Wang C., Xu Z. Identification of an m6A-related lncRNA signature for predicting the prognosis in patients with kidney renal clear cell carcinoma. Frontiers in Oncology. 2021;11 doi: 10.3389/fonc.2021.663263.663263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gu C., Shi X., Qiu W., Huang Z., Yu Y., Shen F. Comprehensive analysis of the prognostic role and mutational characteristics of m6A-related genes in lung squamous cell carcinoma. Frontiers in Cell and Developmental Biology. 2021;9 doi: 10.3389/fcell.2021.661792.661792 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ma S., Yan J., Barr T., Zhang J., Chen Z., Wang L. S. The RNA m6A reader YTHDF2 controls NK cell antitumor and antiviral immunity. Journal of Experimental Medicine. 2021;218 doi: 10.1084/jem.20210279.e20210279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dominissini D., Moshkovitz S. M., Schwartz S., Salmon-Divon M., Ungar L., Osenberg S. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq. Nature. 2012;485:201–206. doi: 10.1038/nature11112. [DOI] [PubMed] [Google Scholar]
- 23.Li J., Han Y., Zhang H., Qian Z., Jia W., Gao Y. The m6A demethylase FTO promotes the growth of lung cancer cells by regulating the m6A level of USP7 mRNA. Biochemical and Biophysical Research Communications. 2019;512:479–485. doi: 10.1016/j.bbrc.2019.03.093. [DOI] [PubMed] [Google Scholar]
- 24.Liu J., Ren D., Du Z., Wang H., Zhang H., Jin Y. m(6)A demethylase FTO facilitates tumor progression in lung squamous cell carcinoma by regulating MZF1 expression. Biochemical and Biophysical Research Communications. 2018;502:456–464. doi: 10.1016/j.bbrc.2018.05.175. [DOI] [PubMed] [Google Scholar]
- 25.Weng H., Huang H., Wu H., Qin X., Zhao B. S., Dong L. METTL14 inhibits hematopoietic stem/progenitor differentiation and promotes leukemogenesis via mRNA m(6)A modification. Cell Stem Cell. 2018;22:191–205. doi: 10.1016/j.stem.2017.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Roundtree I. A., Evans M. E., Pan T., He C. Dynamic RNA modifications in gene expression regulation. Cell. 2017;169:1187–1200. doi: 10.1016/j.cell.2017.05.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yang Y., Hsu P. J., Chen Y. S., Yang Y. G. Dynamic transcriptomic m(6)A decoration: writers, erasers, readers and functions in RNA metabolism. Cell Research. 2018;28:616–624. doi: 10.1038/s41422-018-0040-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Janostiak R., Wajapeyee N. RNA modification regulatory genes in DNA damage. Methods in Molecular Biology. 2019;1870:249–262. doi: 10.1007/978-1-4939-8808-2_19. [DOI] [PubMed] [Google Scholar]
- 29.Liu J., Harada B. T., He C. Regulation of gene expression by N(6)-methyladenosine in cancer. Trends in Cell Biology. 2019;29:487–499. doi: 10.1016/j.tcb.2019.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang Y., Su X., Zhao M., Xu M., Chen Y., Li Z. Importance of N(6)-methyladenosine RNA modification in lung cancer (Review) Molecular and Clinical Oncology. 2021;14 doi: 10.3892/mco.2021.2290.128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gu Y., Wu X., Zhang J., Fang Y., Pan Y., Shu Y. The evolving landscape of N(6)-methyladenosine modification in the tumor microenvironment. Molecular Therapy. 2021;29:1703–1715. doi: 10.1016/j.ymthe.2021.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen X., Yu C., Guo M., Zheng X., Ali S., Huang H. Down-regulation of m6A mRNA methylation is involved in dopaminergic neuronal death. ACS Chemical Neuroscience. 2019;10:2355–2363. doi: 10.1021/acschemneuro.8b00657. [DOI] [PubMed] [Google Scholar]
- 33.Du K., Zhang L., Lee T., Sun T. m(6)A RNA methylation controls neural development and is involved in human diseases. Molecular Neurobiology. 2019;56:1596–1606. doi: 10.1007/s12035-018-1138-1. [DOI] [PubMed] [Google Scholar]
- 34.Gao S., Wang Z. Comprehensive analysis of regulatory network for LINC00472 in clear cell renal cell carcinoma. Journal of Healthcare Engineering. 2021;2021:20. doi: 10.1155/2021/3533608.3533608 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 35.Senan E. M., Adhaileh M. H. A., Alsaade F. W., Aldhyani T. H. H., Alqarni A. A., Alsharif N. Diagnosis of chronic kidney disease using effective classification algorithms and recursive feature elimination techniques. Journal of Healthcare Engineering. 2021;2021:10. doi: 10.1155/2021/1004767.1004767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lang S., Xu Y., Li L., Wang B., Yang Y., Xue Y. Joint detection of tap and CEA based on deep learning medical image segmentation: risk prediction of thyroid cancer. Journal of Healthcare Engineering. 2021;2021:9. doi: 10.1155/2021/5920035.5920035 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 37.Zhou J., Wang J., Hong B., Ma K., Xie H., Li L. Gene signatures and prognostic values of m6A regulators in clear cell renal cell carcinoma-a retrospective study using TCGA database. Aging. 2019;11:1633–1647. doi: 10.18632/aging.101856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kwok C. T., Marshall A. D., Rasko J. E., Wong J. J. Genetic alterations of m(6)A regulators predict poorer survival in acute myeloid leukemia. Journal of Hematology & Oncology. 2017;10 doi: 10.1186/s13045-017-0410-6.39 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cui Q., Shi H., Ye P., Li L., Qu Q., Sun G. m(6)A RNA methylation regulates the self-renewal and tumorigenesis of glioblastoma stem cells. Cell Reports. 2017;18:2622–2634. doi: 10.1016/j.celrep.2017.02.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fry N. J., Law B. A., Ilkayeva O. R., Carraway K. R., Holley C. L., Mansfield K. D. N(6)-methyladenosine contributes to cellular phenotype in a genetically-defined model of breast cancer progression. Oncotarget. 2018;9 doi: 10.18632/oncotarget.25782.31231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wu L., Wu D., Ning J., Liu W., Zhang D. Changes of N6-methyladenosine modulators promote breast cancer progression. BMC Cancer. 2019;19 doi: 10.1186/s12885-019-5538-z.326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cheng M., Sheng L., Gao Q., Xiong Q., Zhang H., Wu M. The m(6)A methyltransferase METTL3 promotes bladder cancer progression via AFF4/NF-kappaB/MYC signaling network. Oncogene. 2019;38:3667–3680. doi: 10.1038/s41388-019-0683-z. [DOI] [PubMed] [Google Scholar]
- 43.Wang H., Yao F., Luo S., Ma K., Liu M., Bai L. A mutual activation loop between the Ca(2+)-activated chloride channel TMEM16A and EGFR/STAT3 signaling promotes breast cancer tumorigenesis. Cancer Letters. 2019;455:48–59. doi: 10.1016/j.canlet.2019.04.027. [DOI] [PubMed] [Google Scholar]
- 44.Ding Z., Zhu J., Zeng Y., Du W., Zhang Y., Tang H. The regulation of Neuropilin 1 expression by miR-338-3p promotes non-small cell lung cancer via changes in EGFR signaling. Molecular Carcinogenesis. 2019;58:1019–1032. doi: 10.1002/mc.22990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Phi L. T. H., Sari I. N., Wijaya Y. T., Kim K. S., Park K., Cho A. E. Ginsenoside rd inhibits the metastasis of colorectal cancer via epidermal growth factor receptor signaling Axis. IUBMB Life. 2019;71:601–610. doi: 10.1002/iub.1984. [DOI] [PubMed] [Google Scholar]
- 46.Ko H., Kim M. M. TP53 tumor-suppressor gene plays a key role in IGF1 signaling pathway related to the aging of human melanocytes. Anticancer Research. 2019;39:2447–2451. doi: 10.21873/anticanres.13363. [DOI] [PubMed] [Google Scholar]
- 47.Grunnet L. G., Nilsson E., Ling C., Hansen T., Pedersen O., Groop L. Regulation and function of FTO mRNA expression in human skeletal muscle and subcutaneous adipose tissue. Diabetes. 2009;58:2402–2408. doi: 10.2337/db09-0205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kikuchi R., Iwai Y., Tsuji T., Watanabe Y., Koyama N., Yamaguchi K. Hypercapnic tumor microenvironment confers chemoresistance to lung cancer cells by reprogramming mitochondrial metabolism in vitro. Free Radical Biology and Medicine. 2019;134:200–214. doi: 10.1016/j.freeradbiomed.2019.01.014. [DOI] [PubMed] [Google Scholar]
- 49.Dyrstad S. E., Lotsberg M. L., Tan T. Z., Pettersen I. K. N., Hjellbrekke S., Tusubira D. Blocking aerobic glycolysis by targeting pyruvate dehydrogenase kinase in combination with EGFR TKI and ionizing radiation increases therapeutic effect in non-small cell lung cancer cells. Cancers. 2021;13 doi: 10.3390/cancers13050941.2065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cheng C. C., Wooten J., Gibbs Z. A., McGlynn K., Mishra P., Whitehurst A. W. Sperm-specific COX6B2 enhances oxidative phosphorylation, proliferation, and survival in human lung adenocarcinoma. Elife. 2020;9 doi: 10.7554/eLife.58108.e58108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wang Y., Cheng Z., Xu J., Lai M., Liu L., Zuo M. Fat mass and obesity-associated protein (FTO) mediates signal transducer and activator of transcription 3 (STAT3)-drived resistance of breast cancer to doxorubicin. Bioengineered. 2021;12:1874–1889. doi: 10.1080/21655979.2021.1924544. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Publicly available datasets were analyzed in this study. These data can be found at https://www.cancer.gov/.


