Abstract
Camel urine has traditionally been used to treat multiple human diseases and possesses the most beneficial effects amongst the urine of other animals. However, scientific review evaluating the anticancer, antiplatelet, gastroprotective and hepatoprotective effects of camel urine is still scarce. Thus, this scoping review aimed to provide scientific evidence on the therapeutic potentials of camel urine. Three databases were searched to identify relevant articles (Web of Science, PubMed and Scopus) up to September 2020. Original articles published in English that investigated the effects of camel urine in various diseases were included. The literature search identified six potential articles that met all the inclusion criteria. Three articles showed that camel urine possesses cytotoxic activities against different types of cancer cells. Two studies revealed camel urine’s protective effects against liver toxicity and gastric ulcers, whilst another study showed the role of camel urine as an antiplatelet agent. All studies demonstrated significant positive effects with different effective dosages. Thus, camel urine shows promising therapeutic potential in treating human diseases, especially cancer. However, the standardised dosage and potential side effects should be determined before camel urine could be offered as an alternative treatment.
Keywords: Animal urine, Camelus dromedarius, Anticancer, Urotherapy, Biological activities
1. Introduction
Urine therapy or urotherapy is an ancient method for treating multiple human diseases. Animal urine, including that of goat, sheep, buffalo, horse, camel and donkey, has traditionally been used to treat abdominal tumours, abdominal enlargement, tuberculosis, haemorrhoids, colic and anaemia (Al-Abdalall, 2010). Camel urine (CU) is considered the prototype of urotherapy and possesses more beneficial effects than other animal urine (Abdel Gader & Alhaider, 2016). It consists of various chemical constituents that contribute to its anticancer, antiplatelet, gastroprotective and hepatoprotective effects. A recent study by Anwar et al. (2021) showed that CU possesses cytotoxic but no clastogenic properties, with studies both in vitro and in vivo showing that CU failed to cause clastogenicity. This finding suggests that CU is safe and has no risk of developing genetic toxicity; thus, it could be offered as a potential therapeutic agent against various diseases.
In Asia, especially in the Arabian Peninsula, CU has been consumed as a traditional remedy. It has been reported that the Bedouins drink approximately 100 ml of CU daily, either alone or mixed with camel milk (CM) (Al-Yousef et al., 2012). Shoeib and Ba-hatheq (2007) reported that heated CU exhibited excellent antibacterial effects against various pathogenic bacteria, including Pseudomonas aeruginosa and Escherichia coli. There is also a belief that CU can be prophylactic and therapeutic for diseases, including cancer. A study conducted in 2018 showed that 15.7% of patients with cancer in Saudi Arabia consumed CU, either alone or in combination with CM, as alternative medicine (Abuelgasim et al., 2018). This practice is supported by the more than 80% apoptosis observed in cancer cells after treatment with 216 mg/ml of lyophilised CU. Cyclin-D1 and β-catenin, cancer-enhancing proteins, were down-regulated following that treatment (Al-Yousef et al., 2012). CU treatment also demonstrated significant anti-metastatic effects on breast cancer cells in mice (Romli et al., 2017). Moreover, The Saudi Center for Medical Research revealed the possibility of producing a medical capsule containing CU as cancer treatment after observing various promising therapeutic effects of CU in cancer cells (Ghamdi and Khorshid, 2012, Romli et al., 2017).
Chemical analysis using gas chromatography-mass spectrometry revealed the presence of 20 metabolites in CU compared with only 14 metabolites in bovine urine. Amongst the metabolites, canavanine, erythritol, benzenepropanoic acid and melibiose were exclusively present in CU (Ahamad et al., 2017). Reports have indicated that 2% of canavanine is excreted in CU, with previous studies suggesting that canavanine has potent antineoplastic activity (Rosenthal and Nkomo, 2000, Vynnytska et al., 2011, Vynnytska-Myronovska et al., 2012). Meanwhile, benzenepropanoic acid, a fatty acid derivative, may be responsible for CU’s antiplatelet properties. (Ahamad et al., 2017). Another prominent feature of CU is its pH of 7.8 compared with human urine with pH 5, which is acidic (Anwar et al., 2021). CU also contains about 10-folds more mineral salts than human urine (Read, 1925). The presence of chemical constituents contributes to the anticancer, antiplatelet, antibacterial, antifungal and antiviral properties of CU (Al-Yousef et al., 2012).
Various studies have been conducted to prove the effectiveness of CU as alternative medicine. However, to the best of our knowledge, the effects of CU consumption on health have not been reviewed previously. Thus, this scoping review aimed to provide scientific evidence on the anticancer, antiplatelet, gastroprotective and hepatoprotective effects of CU.
2. Methods
2.1. Literature review
A systematic search was undertaken to identify relevant studies focusing on the therapeutic potentials of CU on various health conditions. A comprehensive search in the Web of Science, PubMed and Scopus databases (published until September 2020) was conducted. The search strategy involved a combination of three (3) keywords: [1] ‘camel’ OR ‘camelus’ OR ‘dromedary’ AND [2] ‘urine’ OR ‘body fluids’ AND [3] ‘health’ OR ‘medicine’ OR ‘therapeutics’.
2.2. Selection of research articles
Non-English articles were excluded from this review. Review articles, proceedings, books, book chapters, bibliographies, letters to the editor, case reports and consensus/statements/guidelines/updates were also excluded from this review. Only studies that reported CU’s therapeutic potentials on various health conditions were included.
2.3. Inclusion and exclusion criteria
For this review’s intent, only studies involving the direct effect of CU on any health conditions or diseases were included. The studies were considered if only CU was used as an intervention on humans, animals or cells.
2.4. Data extraction and management
The extraction process proceeded by screening the abstract of the remaining articles, and articles were further excluded if they did not meet the inclusion criteria. A thorough reading by two independent reviewers was essential in finalising the selection of articles that met the inclusion criteria. A data extraction form was designed to independently obtain a standardised data collection comprising these data: (1) population and sample size; (2) type of camel; (3) camel urine dosage; (4) methodology of camel urine treatment; (5) result and (6) conclusion.
3. Results
3.1. Search results
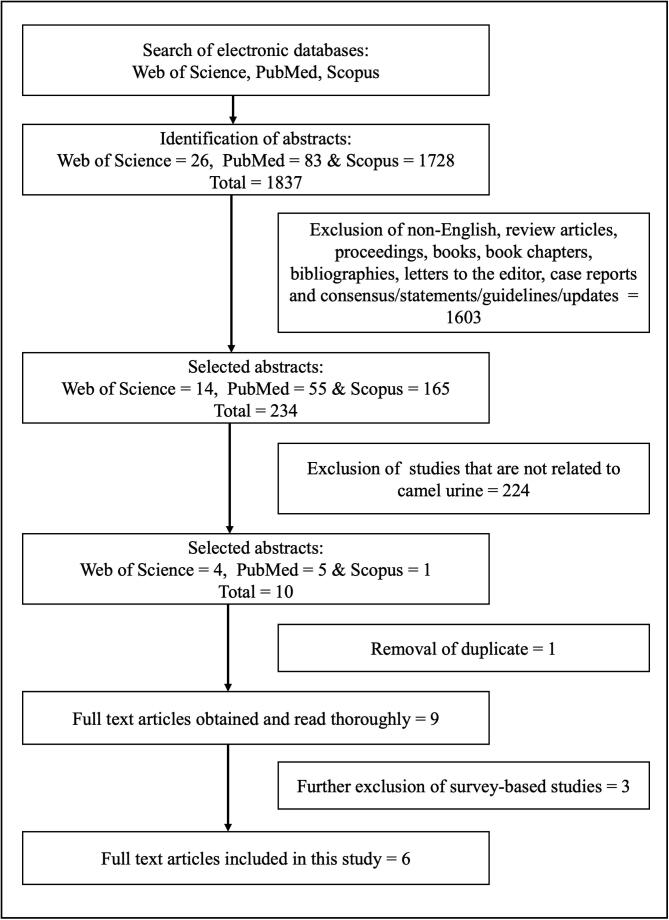
The literature search in Web of Science, PubMed and Scopus databases obtained 1837 potentially relevant articles. The two independent reviewers excluded 1603 non-related articles based on the title, keywords and abstract. A total of 224 articles were further excluded because they did not use CU as an intervention. Before data were extracted, the reviewers assessed the literature thoroughly and selected six studies from the nine selected articles. Any difference of opinion between reviewers was resolved through discussion. Fig. 1 shows the overall article selection and exclusion process.
Fig. 1.
Flowchart of the selection process.
3.2. Study characteristics
Table 1 shows the characteristics of the articles that met all the requirements. All studies were published until September 2020. Out of nine studies, six studies were meticulously reviewed. CU was collected from the same species, Camelus dromedarius. Four studies used female camel (Al-Yousef et al., 2012, Alhaidar et al., 2011, Alhaider et al., 2011, Mahmoud et al., 2019), one used a male camel (Al-Harbi et al., 1996), and another study did not mention the sex of the camel used (Hu et al., 2017). Two studies further divided CU types from virgin, pregnant and lactating camels (Alhaidar et al., 2011, Alhaider et al., 2011). This review included three animal studies, two in vitro studies and one human study targeting cancer (Al-Harbi et al., 1996, Al-Yousef et al., 2012, Alhaider et al., 2011), liver toxicity (Mahmoud et al., 2019), gastric ulcer (Hu et al., 2017) and platelet inhibitory activity (Alhaidar et al., 2011). Overall, the studies showed promising results with different effective dosages.
Table 1.
Data extraction table.
| Author | Subject (n) | Dosage | Type of camel |
Methodology |
Results | Conclusion | |
|---|---|---|---|---|---|---|---|
| Treatment Group | Measurement | ||||||
| (Al-Harbi et al., 1996) | Mice (60) | Camel urine (CU) with a 5%–100% concentration as daily drinking water for 7 days. Average of 5.4–5.82 ml camel urine per mouse. |
Male camel (Camelus dromedarius) from Riyadh, Saudi Arabia | All animals were divided into 12 groups (5 mice per group). Each group was treated with;
|
|
1) Micronucleus test:
|
CU shows cytotoxic activity and non-clastogenic nature in mice induced with CP. |
| (Mahmoud et al., 2019) | Adult male Sprague Dawley rats (30) | 2 ml/100 g by oral intubation for 60 days | Female lactating camel from Egypt | All animals were divided into three groups (10 rats per group). Each group was treated with:
|
|
1. In vitro assessment of CU antioxidant activity:
|
CU shows a protective and curative role against hepatic dysfunction via antioxidative, anti-free radical scavenging activities of its present volatile metabolites and essential inorganic elements. |
| (Al-Yousef et al., 2012) | Human cancer cell line: MCF 10A, MDA-MB-231, U2OS, DAOY, LoVo, HCT-116, MED-1, MED-8, MED-13 and HFSN1 | 16 mg/ml was added to every cell line | Young female camel (Camelus dromedaries) | All cell lines were divided into two groups (control and treatment). Control groups were not treated, and treatment groups received 16 mg/ml CU. For PBMC cytotoxicity test, cells were treated with different concentrations of CU for 3 days. |
|
1. Cytotoxicity test:
|
Camel urine has anticancer effects on the various human cancer cell lines. |
| (Alhaider et al., 2011) | Hepa 1c1c7 cell line | Camel urine (CU) 15 µl/ml added to Hepa 1c1c7 cell line | Female virgin, prenant and lactating camel (Camelus dromadaries) | Hepa 1c1c7 cells were preincubated with either CU or BU (15 µl/ml) before incubation with 1 nM TCDD |
|
|
Camel urine inhibits the TCDD-mediated effect, at least in part by inhibiting the expression of Cyp1a1, a cancer-activating gene, at both the transcriptional and the post-translational levels through an AhR-dependent mechanism. |
| (Hu et al., 2017) | Sprague–Dawley rats (95) | 5 ml/kg of camel milk (CM) and urine (CU) were given orally | Camel from Hargeisa, Somaliland | All animals (5 rats per group) were treated with saline (5 ml/kg), cimetidine (100 mg/kg), CM (5 ml/kg) or CU (5 ml/kg) and induced with any one of the ulcer agents (HCl/EtOH (0.2 ml/animal), indomethacin (50 mg/kg) or water restraint stress-induced ulcer (3 h)) |
|
|
Administration of CM and CU may have strengthened the mucosal barrier against endogenous and exogenous ulcerogenic agents in HCl/EtOH, non-steroidal anti-inflammatory drugs and WRS-induced gastric damage. CM and CU also showed a strong ulcer-healing effect in indomethacin-induced gastric damage. |
| (Alhaidar et al., 2011) | Healthy human volunteers | 0.05 ml CU was added to human platelet-rich plasma (PRP) | Female virgin, pregnant and lactating domesticated camels (Camelus dromedaries) | 0.05 ml of CU was added to human PRP before 0.05 ml of ADP or AA (aggregating agent) was added | Platelet inhibitory activity, aggregation responses to adenosine diphosphate (ADP) and arachidonic Acid (AA), PFA-100 closure time |
|
CU showed antiplatelet actions and provided an essential foundation of scientific evidence to explore CU as a therapeutic antiplatelet agent. |
4. Discussion
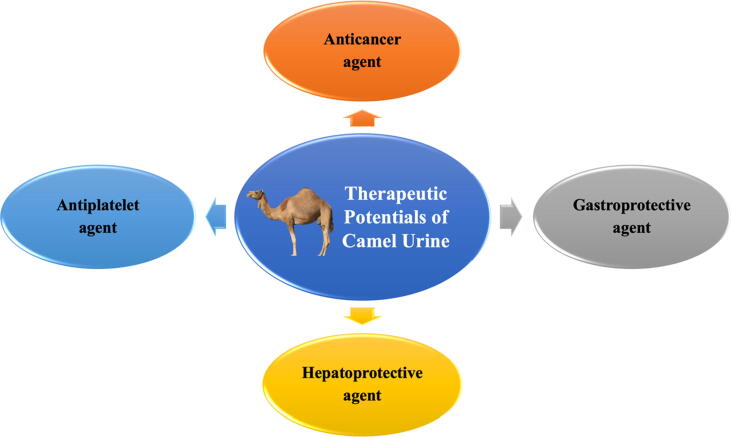
For over a century, camels continue to be an essential source of food and transportation amongst desert dwellers in Asia and Africa. Interestingly, not only CM is beneficial in maintaining good health, but CU has also been used to treat various ailments (Adams & Jewell, 2007). CU can effectively treat stomach infections, tuberculosis and cancer (Kabarity et al., 1988). A survey conducted in 2018 found that 15.2% of patients with cancer in Saudi Arabia consumed CU alone or in combination with CM as an alternative treatment (Abuelgasim et al., 2018). Fig. 2 shows the therapeutic potentials of CU.
Fig. 2.
Therapeutic potentials of camel urine.
4.1. Camel urine as an anticancer agent
In this review, three out of six articles investigated the effect of CU on cancer cell lines (in vitro) and mice (in vivo). Al-Harbi et al. (1996) found that CU with at least 50% concentration produced a significant cytotoxic effect in mouse bone marrow cells; a considerable reduction in the polychromatic erythrocyte (PCE) to normochromic erythrocyte (NCE) ratio indicates the CU cytotoxic potential. These findings were supported by the decreased nucleic acids and glutathione (GSH) levels and increased malondialdehyde (MDA) in the same study. This cytotoxic effect was comparable with that of cyclophosphamide (CP), a standard drug for chemotherapy in patients with cancer. However, CU-treated mice did not show any clastogenic activities, in contrast to CP, which has high clastogenic activity. A similar finding was noted where CU was found to be mitodepressive but not clastogenic (Kabarity et al., 1988). Additionally, CU showed no clastogenic effect on the bone marrow cells of mice, and 25 and 50 ml/kg of CU treatment significantly improved the cyclophosphamide-induced clastogenic effect (Anwar et al., 2021). Meanwhile, the presence of antioxidative and antimutagenic components, such as creatinine and uric acid, in CU may contribute to the non-clastogenic nature of CU (Al-Bekairi et al., 1991, Ames et al., 1981). Uric acid is a potent scavenger of peroxyl radicals, hydroxyl radicals and singlet oxygen (Ames et al., 1981) and can chelate metal ions by converting them to poorly reactive forms that cannot catalyse free-radical reactions (Davies et al., 1986, Einsele et al., 1987, Halliwell and Gutteridge, 2015, Miura et al., 1993).
Al-Yousef et al. (2012) have found that CU exhibits anticancer properties on various cancer cells. Ten types of cancer cells were treated with 16 mg/ml of CU. After treatment, the cells were divided into two groups: 1) CU-resistant cells, which include MCF 10A, HFSN-1, U2OS, MCF-7, MED-8, LoVo and HCT-116 and 2) CU-sensitive cells, which killed more than 50% cells, including MDA-MB-231, DAOY, MED-4 and MED-13. Surprisingly, CU killed more than 80% of MDA-MB-231 (breast cancer cell) but not MCF 10A cells (non-tumourigenic breast epithelial cells). This finding suggests that CU is cytotoxic but has a particular effect on breast cancer cells and avoids healthy breast cells.
CU also induced apoptosis (90%) in the CU-sensitive cells group, with a slight proportion of necrosis. High levels of caspase-3 and PARP, 18.6- and 3.4-folds higher than normal, respectively, were believed to trigger apoptosis via the mitochondrial pathway. CU also showed potent inhibitory effects on two major apoptosis inhibitor proteins, Bcl-2 and survivin, which are related to breast cancer pathology and therapeutic outcome (Altieri, 2008, Callagy et al., 2006, Tanaka et al., 2000). Furthermore, CU possesses a significant anti-proliferative effect on breast cancer cells, where MDA-MB-231 cells stop proliferating immediately following treatment with CU. However, no effects were observed on non-tumourigenic breast epithelial cells. This effect could be mediated by the induction of the cyclin-dependent kinase inhibitor p21, associated with the acquisition of senescence phenotypes in breast cancer cells (Caldon et al., 2010). This finding is supported by Lacroix et al. (2006), which found an up-regulation of p21 in the p53-defective MDA-MB-231 cells, indicating a p53-dependent effect. p53 transcriptionally activates genes, which induce cell cycle arrest or apoptosis. In turn, it eliminates and inhibits the proliferation of abnormal cells and prevents neoplastic development (Gasco et al., 2002).
CU also acts as an anticancer agent by enhancing the production of IFN-ɣ and inhibiting IL-4, IL-6 and IL-10. Both IL-4 and IL-10 levels were almost undetectable after treatment with CU. IL-4 is linked to tumour cell growth (Nagai & Toi, 2000), whilst IL-6 is a potent growth factor for breast cancer (Knüpfer & Preiß, 2007); a high level of IL-10 correlated well with poor survival of cancer patients (Mocellin et al., 2005). Thus, inhibition of these three cytokines holds a promising therapeutic strategy for breast cancer.
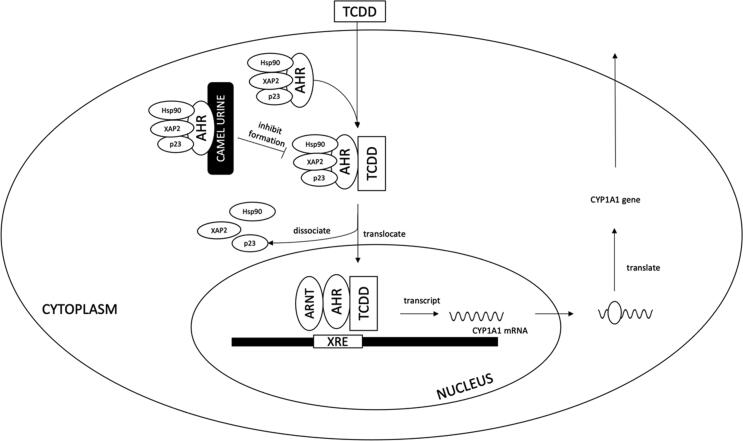
The effect of CU on cancer cells was also studied by Alhaider et al. (2011). They used Hepa 1c1c7 cells, a hepatoma cell line, to evaluate the ability of CU to inhibit cytochrome P450 1a1 (CYP1A1) gene expression. CYP1A1 is a known cancer-activating gene and is strongly correlated with an increased incidence of colon, rectal and lung cancers (Shah et al., 2009, Slattery et al., 2004). 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) is the most potent inducer of CYP1A1 (Wei et al., 2002). CYP1A1 stimulates the bioactivity of procarcinogens to regenerate reactive metabolites (Rendic & Guengerich, 2012). These reactive metabolites will form DNA adducts and contribute to mutagenesis, which eventually leads to the development of various types of cancer (Wogan et al., 2004). Correlations between DNA-adduct formation and exposure, hepatocyte initiation and hepatocellular carcinoma have been demonstrated (Dyroff et al., 1986). A concentration up to 25 µl/ml was not toxic to Hepa 1c1c7 cells for virgin, pregnant and lactating CU in the cytotoxic test.
Bovine urine (BU) starts to reduce cell viability at 25 µl/ml. Preincubation with virgin, lactating and pregnant CU reduced TCDD-induced CYP1A1 catalytic activity by 80%, 70% and 54%, respectively. Interestingly, these inhibitory effects are similar to that of a standard aryl hydrocarbon receptor (AhR) antagonist, resveratrol, which significantly reduced CYP1A1 catalytic activity by 50%. CU might modulate CYP1A1 catalytic activity by altering mRNA and protein expression (Fig. 3). CU, particularly virgin CU, demonstrated a significant ability in inhibiting CYP1A1 mRNA expression, whilst lactating and pregnant CU did not alter CYP1A1 mRNA expression (Alhaider et al., 2011). Virgin and lactating CU also significantly down-regulated CYP1A1 protein expression. This finding strongly suggests that inhibition of CYP1A1 by CU, particularly virgin CU, might be mediated at the transcription and translational levels. Virgin CU was further tested to determine its ability to inhibit AhR transformation and xenobiotic responsive element (XRE) binding. The results revealed that virgin CU could inhibit AhR from transforming into DNA-binding form, preventing TCDD binding to XRE. This ability of virgin CU eventually inhibits CYP1A1, an AhR-dependent gene expression.
Fig. 3.
Modulation of CYP1A1 catalytic activity by camel urine.
The abundance of lactoferrin in CU might contribute to its ability as an anticancer agent. Roseanu et al. (2010) found that lactoferrin is responsible for exerting in vitro and in vivo anti-tumour activity. It can also significantly inhibit CYP1A1 activation in 7,12-dimethylbenz[a]anthracene-induced buccal pouch carcinoma in hamster and thus inhibit cancer development (Alhaider et al., 2011). Additionally, approximately 2% of canavanine, a toxic anti-metabolite of L-arginine, is excreted in CU. This compound has been shown to have a significant effect as an anticancer agent (Vynnytska et al., 2011, Vynnytska-Myronovska et al., 2012). Therefore, CU is a potential therapeutic agent against various cancers due to its anticancer properties, cytotoxic activity and non-clastogenic effect.
4.2. Camel urine as a gastroprotective agent
CU exhibits a significant anti-ulcer effect in three models of the gastroprotective method: 1) HCl/ethanol (HCl/EtOH)-induced ulcer, 2) water-restraint stress (WRS)-induced ulcer and 3) non-steroidal anti-inflammatory drug (indomethacin)-induced ulcer. Ulcer formation was confirmed by the presence of lesions and inflammation in the stomach glandular portion (HCl/EtOH model), the appearance of haemorrhagic streaks across the whole gastric mucosa (WRS model) and extensive gastric mucosal damage lesion with the presence of blackish-red haemorrhagic spots (indomethacin model). Treatment with 5 ml/kg of CU showed 100% protection against a peptic ulcer in HCl/EtOH and WRS models. In the indomethacin model, CU showed 66.7% ulcer inhibition. Also, CU demonstrated a 100% healing rate with no ulcers observed in indomethacin-induced gastric damage (healing model) compared with cimetidine, a standard drug, which only resulted in a 60.5% healing rate. This result suggests that CU has better healing properties than cimetidine. This powerful action is attributed to the presence of antioxidants in CU. Trace elements, such as magnesium and zinc present in CU, were found to reduce oxidative stress and help prevent the tissue injury caused by a toxic agent (Shazia et al., 2012). Therefore, CU appears to strengthen the mucosal barrier against endogenous and exogenous ulcerogenic agents and showed a strong ulcer healing effect in indomethacin-induced gastric damage, which could be considered an ulcer treatment in the future.
4.3. Camel urine as a hepatoprotective agent
Mahmoud et al. (2019) investigated the hepatoprotective effects of CU against carbon tetrachloride (CCl4)-induced liver damage in rats after 60 days. They revealed that CU significantly alleviated hepatotoxicity induced by CCl4. This effect was established through the antioxidant property of the active metabolites, which prevent cellular oxidative free radicals induced by CCl4 from attacking the liver tissues. Twenty different components that are related to glycolysis, fatty acid synthesis, nucleoside, arachidonic acid androgens, mineralocorticoids, tyrosine inhibitor and the arachidonic acid pathway have been identified in CU. Furthermore, another 28 essential inorganic elements have been identified in CU with sodium, potassium, magnesium, manganese and chromium present in the highest quantities. The presence of these inorganic elements at a dose of 100 µg CU significantly increased SOR scavenging, H2O2 scavenging and FRAPS free radical activities.
Meanwhile, essential organic elements that act as antioxidants mediate the high activity of free radical scavenging (Abdel-Salam et al., 2010). A significant increment in aspartate transferase (AST/SGPT) and alanine transaminase (ALT/SGOT) levels were observed following CU treatment, plus the reduction of albumin level. Low AST and ALT levels suggested improving liver function and protection against CCl4 (Elhag et al., 2017). Hepatic tissue sections treated with CU also showed the absence of lytic necrosis or portal inflammation and less marked fibrous tissue expansion with some steatosis evidence with focal confluent necrosis. Together, CU provided a protective effect on liver damage induced by CCl4. Thus, CU can be considered as a potential hepatoprotective agent due to the antioxidative and anti-free radical scavenging activities of its present volatile metabolites and essential inorganic elements.
4.4. Camel urine as an antiplatelet
Alhaidar et al. (2011) studied the antiplatelet properties of CU; they found that CU showed significant platelet inhibitory activity against human blood and can block the aggregation responses of human platelets to adenosine diphosphate (ADP) and arachidonic acid (AA). ADP is a potent pro-aggregatory agent and contributes to tumour cell-induced platelet aggregation (TCIPA) induced by breast carcinoma MCF7. The release of ADP during MCF7-induced TCIPA activates the P2Y12 purinergic receptor and eventually causes platelet aggregation (Alonso-Escolano et al., 2004). Thus, CU’s inhibitory activity is suggested as an important feature of its anticancer properties because it may help prevent aggregation of platelets and reduce the metastatic potential of cancer cells (Gasic, 1984, Pearlstein et al., 1981, Radomski et al., 1991). Moreover, CU also prolonged the PFA-100 closure time. PFA-100 is a test to measure the duration through which platelets can block the whole blood flow, and it is a measurement of overall platelet-related hemostasis (Paniccia et al., 2015). PFA-100 closure time is inversely correlated with platelet count (Kundu et al., 1996). This reduced platelet count is in line with the inhibition of platelet aggregation. The presence of benzenepropanoic acid in CU is thought to be responsible for the antiplatelet activity, but further research is needed to confirm its roles as a potential therapeutic antiplatelet agent (Ahamad et al., 2017).
5. Conclusion
In this review, CU showed promising therapeutic effects in the treatment of certain human diseases, especially cancer. However, the treatment dosages on cancer cells in all three studies varied. The effective dosage must be standardised, and extensive research must be conducted to determine any potential side effects of CU before it can be recommended as an alternative treatment for cancer. Issues, such as the potential of Middle East respiratory syndrome coronavirus infections, should be addressed. Further studies should be conducted to determine the protective effects of CU in gastric ulcer and liver toxicity, as well as its mechanism as an antiplatelet agent. This review has few limitations, including 1) the reviewed articles were selected from three databases only, and there might be relevant articles in other databases that were not retrieved. 2) Non-English articles were excluded to avoid any misinterpretations as we have limited resources for translating the articles.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Peer review under responsibility of King Saud University.
Contributor Information
Norizam Salamt, Email: norizam_salamt@ukm.edu.my.
Mohd Helmy Mokhtar, Email: helmy@ukm.edu.my.
References
- Abdel Gader A., Alhaider A. The unique medicinal properties of camel products: a review of the scientific evidence. J. Taibah Univ. Med. Sci. 2016 doi: 10.1016/j.jtumed.2015.12.007. [DOI] [Google Scholar]
- Abdel-Salam A.M., El-Mergawi R.A., Al-Humaid A.I., Mousa H.M. Chemical composition and antioxidant activity of dates and dates-camel-milk mixtures as a protective meal against lipid peroxidation in rats. Am. J. Food Technol. 2010 Retrieved from http://docsdrive.com/pdfs/academicjournals/ajft/2010/22-30.pdf. [Google Scholar]
- Abuelgasim K.A., Alsharhan Y., Alenzi T., Alhazzani A., Ali Y.Z., Jazieh A.R. The use of complementary and alternative medicine by patients with cancer: a cross-sectional survey in Saudi Arabia. BMC Complement. Alternative Med. 2018;18(1):88. doi: 10.1186/s12906-018-2150-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adams M., Jewell A.P. The use of Complementary and Alternative Medicine by cancer patients. Int. Semin. Surg. Oncol. 2007;4 doi: 10.1186/1477-7800-4-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahamad S.R., Alhaider A.Q., Raish M., Shakeel F. Metabolomic and elemental analysis of camel and bovine urine by GC–MS and ICP–MS. Saudi J. Biol. Sci. 2017;24(1):23–29. doi: 10.1016/j.sjbs.2015.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Abdalall A. The inhibitory effect of camels urine on mycotoxins and fungal growth. Afr. J. Agric. Res. 2010;5:1331–1337. [Google Scholar]
- Al-Bekairi A.M., Qureshi S., Chaudhry M.A., Shah A.H. Uric acid as an inhibitor of cyclophosphamide-induced micronuclei in mice. Mutat. Res. 1991;262(2):115–118. doi: 10.1016/0165-7992(91)90116-l. [DOI] [PubMed] [Google Scholar]
- Al-Harbi M.M., Qureshi S., Ahmed M.M., Raza M., Baig M.Z., Shah A.H. Effect of camel urine on the cytological and biochemical changes induced by cyclophosphamide in mice. J. Ethnopharmacol. 1996;52(3):129–137. doi: 10.1016/0378-8741(96)01399-2. [DOI] [PubMed] [Google Scholar]
- Al-Yousef N., Gaafar A., Al-Otaibi B., Al-Jammaz I., Al-Hussein K., Aboussekhra A. Camel urine components display anti-cancer properties in vitro. J. Ethnopharmacol. 2012;143(3):819–825. doi: 10.1016/j.jep.2012.07.042. [DOI] [PubMed] [Google Scholar]
- Alhaidar A., Abdel Gader A.G., Mousa S.A. The antiplatelet activity of camel urine. J. Altern. Complement. Med. 2011;17(9):803–808. doi: 10.1089/acm.2010.0473. [DOI] [PubMed] [Google Scholar]
- Alhaider A.A., El Gendy M.A., Korashy H.M., El-Kadi A.O. Camel urine inhibits the cytochrome P450 1a1 gene expression through an AhR-dependent mechanism in Hepa 1c1c7 cell line. J. Ethnopharmacol. 2011;133(1):184–190. doi: 10.1016/j.jep.2010.09.012. [DOI] [PubMed] [Google Scholar]
- Alonso-Escolano D., Strongin A.Y., Chung A.W., Deryugina E.I., Radomski M.W. Membrane type-1 matrix metalloproteinase stimulates tumour cell-induced platelet aggregation: role of receptor glycoproteins. Br. J. Pharmacol. 2004;141(2):241–252. doi: 10.1038/sj.bjp.0705606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altieri D.C. Survivin, cancer networks and pathway-directed drug discovery. Nat. Rev. Cancer. 2008;8(1):61–70. doi: 10.1038/nrc2293. [DOI] [PubMed] [Google Scholar]
- Ames B.N., Cathcart R., Schwiers E., Hochstein P. Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: a hypothesis. Proc. Natl. Acad. Sci. USA. 1981;78(11):6858–6862. doi: 10.1073/pnas.78.11.6858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anwar S., Ansari S.A., Alamri A., Alamri A., Alqarni A., Alghamdi S., Rengasamy K.R.R. Clastogenic, anti-clastogenic profile and safety assessment of Camel urine towards the development of new drug target. Food Chem. Toxicol. 2021;151 doi: 10.1016/j.fct.2021.112131. [DOI] [PubMed] [Google Scholar]
- Caldon C., Sutherland R., Musgrove E. Cell cycle proteins in epithelial cell differentiation: Implications for breast cancer. Cell Cycle (Georgetown, Tex.) 2010;9:1918–1928. doi: 10.4161/cc.9.10.11474. [DOI] [PubMed] [Google Scholar]
- Callagy G.M., Pharoah P.D., Pinder S.E., Hsu F.D., Nielsen T.O., Ragaz J., Caldas C. Bcl-2 is a prognostic marker in breast cancer independently of the Nottingham Prognostic Index. Clin. Cancer Res.: Off. J. Ame. Assoc. Cancer Res. 2006;(8):2468–2475. doi: 10.1158/1078-0432.ccr-05-2719. [DOI] [PubMed] [Google Scholar]
- Davies K.J., Sevanian A., Muakkassah-Kelly S.F., Hochstein P. Uric acid-iron ion complexes. A new aspect of the antioxidant functions of uric acid. Biochem. J. 1986;235(3):747–754. doi: 10.1042/bj2350747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dyroff M.C., Richardson F.C., Popp J.A., Bedell M.A., Swenberg J.A. Correlation of O4-ethyldeoxythymidine accumulation, hepatic initiation and hepatocellular carcinoma induction in rats continuously administered diethylnitrosamine. Carcinogenesis. 1986;7(2):241–246. doi: 10.1093/carcin/7.2.241. [DOI] [PubMed] [Google Scholar]
- Einsele H., Clemens M.R., Wegner U., Waller H.D. Effect of free radical scavengers and metal ion chelators on hydrogen peroxide and phenylhydrazine induced red blood cell lipid peroxidation. Free Radical Res. Commun. 1987;3(1–5):257–263. doi: 10.3109/10715768709069791. [DOI] [PubMed] [Google Scholar]
- Elhag A., Faye B., El Badwi S. Protective activity of camel’s milk and urine mixture (camelus dromedaries) against ethanol-induced hepatotoxicity in rats. Adv. Biosci. Biotechnol. 2017;08:378–387. doi: 10.4236/abb.2017.810027. [DOI] [Google Scholar]
- Gasco M., Shami S., Crook T. The p53 pathway in breast cancer. Breast Cancer Res.: BCR. 2002;4(2):70–76. doi: 10.1186/bcr426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gasic G.J. Role of plasma, platelets, and endothelial cells in tumor metastasis. Cancer Metastasis Rev. 1984;3(2):99–114. doi: 10.1007/bf00047657. [DOI] [PubMed] [Google Scholar]
- Ghamdi Z., Khorshid F. Cytotoxicity of the urine of different camel breeds on the proliferation of lung cancer cells, A549. J. Nat. Sci. Res. 2012;2 [Google Scholar]
- Halliwell B., Gutteridge J.M. Oxford University Press; USA: 2015. Free Radicals in Biology and Medicine. [Google Scholar]
- Hu Z., Chang X., Pan Q., Gu K., Okechukwu P.N. Gastroprotective and ulcer healing effects of camel milk and urine in HCl/EtOH, non-steroidal anti-inflammatory drugs (indomethacin), and water-restraint stress-induced ulcer in rats. Pharmacogn. Mag. 2017;13(52):559–565. doi: 10.4103/pm.pm_135_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kabarity A., Mazrooei S., Elgindi A. Camel urine as a possible anticarcinogenic agent. Arab Gulf J. Sci. Res. 1988;6:55. [Google Scholar]
- Knüpfer H., Preiß R. Significance of interleukin-6 (IL-6) in breast cancer (review) Breast Cancer Res. Treat. 2007;102(2):129–135. doi: 10.1007/s10549-006-9328-3. [DOI] [PubMed] [Google Scholar]
- Kundu S.K., Heilmann E.J., Sio R., Garcia C., Ostgaard R.A. Characterization of an in vitro platelet function analyzer, PFA-100™. Clin. Appl. Thromb. Hemost. 1996;2(4):241–249. doi: 10.1177/107602969600200404. [DOI] [Google Scholar]
- Lacroix M., Toillon R.A., Leclercq G. p53 and breast cancer, an update. Endocr. Relat. Cancer. 2006;13(2):293–325. doi: 10.1677/erc.1.01172. [DOI] [PubMed] [Google Scholar]
- Mahmoud H., Elsaed W., Gabr S. Camel urotherapy and hepatoprotective effects against carbon tetrachloride-induced liver toxicity. Int. J. Pharmacol. 2019;15:696–705. doi: 10.3923/ijp.2019.696.705. [DOI] [Google Scholar]
- Miura T., Muraoka S., Ogiso T. Inhibitory effect of urate on oxidative damage induced by adriamycin-Fe3+ in the presence of H2O2. Res. Commun. Chem. Pathol. Pharmacol. 1993;79(1):75–85. [PubMed] [Google Scholar]
- Mocellin S., Marincola F.M., Young H.A. Interleukin-10 and the immune response against cancer: a counterpoint. J. Leukoc. Biol. 2005;78(5):1043–1051. doi: 10.1189/jlb.0705358. [DOI] [PubMed] [Google Scholar]
- Nagai S., Toi M. Interleukin-4 and breast cancer. Breast Cancer. 2000;7(3):181–186. doi: 10.1007/BF02967457. [DOI] [PubMed] [Google Scholar]
- Paniccia R., Priora R., Liotta A.A., Abbate R. Platelet function tests: a comparative review. Vascular Health Risk Manage. 2015;11:133–148. doi: 10.2147/vhrm.s44469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearlstein E., Ambrogio C., Gasic G., Karpatkin S. Inhibition of the platelet-aggregating activity of two human adenocarcinomas of the colon and an anaplastic murine tumor with a specific thrombin inhibitor, dansylarginine N-(3-ethyl-1,5-pentanediyl)amide. Cancer Res. 1981;41(11 Pt 1):4535–4539. [PubMed] [Google Scholar]
- Radomski M.W., Jenkins D.C., Holmes L., Moncada S. Human colorectal adenocarcinoma cells: differential nitric oxide synthesis determines their ability to aggregate platelets. Cancer Res. 1991;51(22):6073. http://cancerres.aacrjournals.org/content/51/22/6073.abstract Retrieved from. [PubMed] [Google Scholar]
- Read B.E. Chemical constituents of camel's urine. J. Biol. Chem. 1925;64(3):615–617. Retrieved from http://www.jbc.org/content/64/3/615.short. [Google Scholar]
- Rendic S., Guengerich F.P. Contributions of human enzymes in carcinogen metabolism. Chem. Res. Toxicol. 2012;25(7):1316–1383. doi: 10.1021/tx300132k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romli F., Abu N., Khorshid F.A., Syed Najmuddin S.U.F., Keong Y.S., Mohamad N.E., Nik Abd Rahman N.M.A. The growth inhibitory potential and antimetastatic effect of camel urine on breast cancer cells in vitro and in vivo. Integrative Cancer Therapies. 2017;16(4):540–555. doi: 10.1177/1534735416656051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roseanu A., Florian P.E., Moisei M., Sima L.E., Evans R.W., Trif M. Liposomalization of lactoferrin enhanced its anti-tumoral effects on melanoma cells. Biometals: Int. J. Role Metal Ions Biol. Biochem. Med. 2010;23(3):485–492. doi: 10.1007/s10534-010-9312-6. [DOI] [PubMed] [Google Scholar]
- Rosenthal G.A., Nkomo P. The natural abundance of L-canavanine, an active anticancer agent, in alfalfa, medicago sativa (L.) Pharm. Biol. 2000;38(1):1–6. doi: 10.1076/1388-0209(200001)3811-bft001. [DOI] [PubMed] [Google Scholar]
- Shah P.P., Saurabh K., Pant M.C., Mathur N., Parmar D. Evidence for increased cytochrome P450 1A1 expression in blood lymphocytes of lung cancer patients. Mutat. Res. 2009;670(1–2):74–78. doi: 10.1016/j.mrfmmm.2009.07.006. [DOI] [PubMed] [Google Scholar]
- Shazia Q., Mohammad Z.H., Rahman T., Shekhar H. Correlation of oxidative stress with serum trace element levels and antioxidant enzyme status in beta thalassemia major patients: a review of the literature. Anemia. 2012;2012 doi: 10.1155/2012/270923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shoeib A. Effect of camel’s urine on pathogenic P.aeruginosa and E.coli isolates, towards its maintains to their antibiotic(s) resistance and the presence of plasmid(s) Saudi J. Biol. Sci. 2007;14(2):177–184. [Google Scholar]
- Slattery M.L., Samowtiz W., Ma K., Murtaugh M., Sweeney C., Levin T.R., Neuhausen S. CYP1A1, cigarette smoking, and colon and rectal cancer. Am. J. Epidemiol. 2004;160(9):842–852. doi: 10.1093/aje/kwh298. [DOI] [PubMed] [Google Scholar]
- Tanaka K., Iwamoto S., Gon G., Nohara T., Iwamoto M., Tanigawa N. Expression of and its relationship to loss of apoptosis in breast carcinomas. Clin. Cancer Res. 2000;6(1):127. Retrieved from http://clincancerres.aacrjournals.org/content/6/1/127.abstract. [PubMed] [Google Scholar]
- Vynnytska B.O., Mayevska O.M., Kurlishchuk Y.V., Bobak Y.P., Stasyk O.V. Canavanine augments proapoptotic effects of arginine deprivation in cultured human cancer cells. Anticancer Drugs. 2011;22(2):148–157. doi: 10.1097/CAD.0b013e32833e0334. [DOI] [PubMed] [Google Scholar]
- Vynnytska-Myronovska B., Bobak Y., Garbe Y., Dittfeld C., Stasyk O., Kunz-Schughart L.A. Single amino acid arginine starvation efficiently sensitizes cancer cells to canavanine treatment and irradiation. Int. J. Cancer. 2012;130(9):2164–2175. doi: 10.1002/ijc.26221. [DOI] [PubMed] [Google Scholar]
- Wei C., Caccavale R., Weyand E., Chen S., Iba M. Induction of CYP1A1 and CYP1A2 expressions by prototypic and atypical inducers in human lung. Cancer Lett. 2002;178:25–36. doi: 10.1016/S0304-3835(01)00809-6. [DOI] [PubMed] [Google Scholar]
- Wogan G.N., Hecht S.S., Felton J.S., Conney A.H., Loeb L.A. Environmental and chemical carcinogenesis. Semin. Cancer Biol. 2004;14(6):473–486. doi: 10.1016/j.semcancer.2004.06.010. [DOI] [PubMed] [Google Scholar]