Abstract
Background
This study aimed to clarify the protective role of dexmedetomidine in thoracoscopic-assisted thoracic surgery (TATS), including control of the intraoperative heart rate, blood pressure, and myocardial injury markers.
Material/Methods
The patients who underwent TATS were divided into 2 equal groups: the dexmedetomidine group (dexmedetomidine pumped at 0.5 μg/kg for >10 min before the administration of anesthesia and at 0.5 μg/kg in the maintenance period) and the control group (pumped normal saline for >10 min before the administration of anesthesia). The data recorded for each patient were heart rate (preoperative, maximum intraoperative, and minimum intraoperative), systolic and diastolic blood pressure, intraoperative hemodynamic data, and intraoperative cardiovascular drugs administered. An enzyme-linked immunosorbent assay was performed to assess the postoperative levels of cardiac troponin I (cTnI), creatine kinase isoenzyme, myoglobin, and N-terminal pro-B-type natriuretic peptide (NT-proBNP).
Results
There were no significant differences in the age, sex, body height, body weight, American Society of Anesthesiologists classification grade, resection mode, operation time, ejection fraction, basal heart rate, and systolic and diastolic blood pressure of the 2 groups. In the dexmedetomidine group, the patients’ maximum intraoperative heart rate and diastolic pressure decreased, and the postoperative hospital stay period was shorter. The postoperative peripheral blood test for the dexmedetomidine group showed higher NT-proBNP levels and lower cTnI levels.
Conclusions
Preoperative administration of dexmedetomidine can benefit hemodynamic stability, protect the cardiovascular system in the intraoperative and postoperative periods, and shorten postoperative hospitalization.
Keywords: Dexmedetomidine, Hemodynamics, Myocardial Bridging, Thoracoscopy
Background
Dexmedetomidine is a selective α-2 adrenoceptor agonist, which can exert sedative, analgesic, and anxiolytic effects. In surgical application, dexmedetomidine is administered to maintain the balance of hemodynamic factors and provide effective analgesia; this helps reduce the use of anesthetics [1,2]. For patients with endotracheal intubation under general anesthesia, it can maintain hemodynamic stability, facilitate intraoperative management, and reduce the occurrence of serious cardiac accidents, including myocardial ischemia in surgical patients with cardiovascular disease [3–5].
In video-assisted thoracoscopic surgery (VATS) or thoracoscopic-assisted thoracic surgery (TATS), myocardial hypoxia frequently occurs due to 1-lung ventilation, imbalance of the ventilation/blood-flow ratio, and other complications [6–9]. Factors including pressure on the heart and change of pulmonary circulation cause hemodynamic fluctuation, resulting in myocardial injury due to insufficient myocardial perfusion. Given the role of dexmedetomidine in surgery, we assumed it can provide myocardial protection in the perioperative period for patients undergoing TATS. Although dexmedetomidine was used in several VATS studies, the focus was its effect on the quality of recovery, postoperative pulmonary function, and postoperative intravenous analgesia [10–12]. The aim of this study is to illuminate its myocardial-protective role in TATS, including control of the intraoperative heart rate and blood pressure, and the levels of myocardial injury markers.
Material and Methods
Study Population
The design and workflow of this clinical trial are presented in Figure 1. The trial was conducted from June 2018 to December 2018. The inclusion criteria were patients aged 15 years to 80 years, American Society of Anesthesiologists (ASA) classification grade II or grade III and ready for VATS, body weight 40 kg to 80 kg, body mass index 19 kg/m2 to 24 kg/m2, no signs of difficult intubation, no history of mental disorders, and a normal ST segment in the preoperative electrocardiogram.
Figure 1.
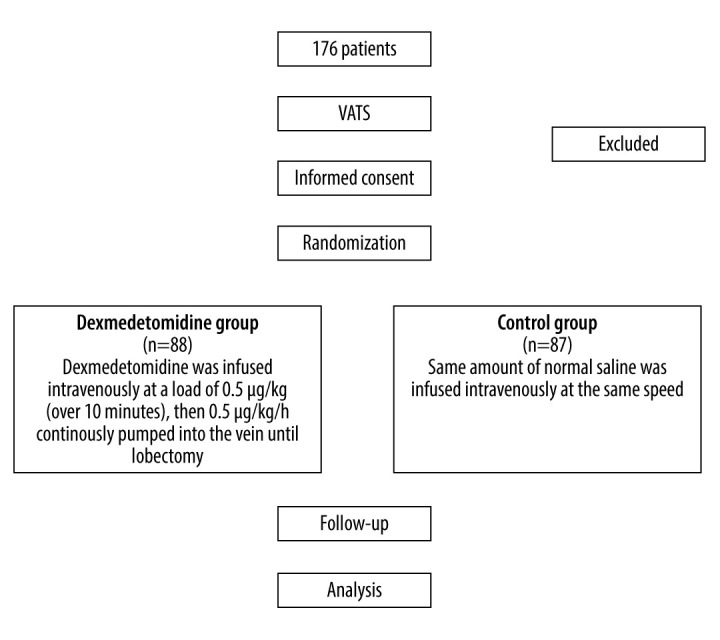
Flow diagram for the study.
The exclusion criteria were patients with arrhythmias, atrioventricular blocks, liver or kidney dysfunction, pulmonary diseases, endocrine diseases, preoperative bradycardia (heart rate [HR] <40 beats/min) or ≥2nd degree heart block, history of myocardial infarction, heart failure, myocarditis, and hypertrophic cardiomyopathy.
The 175 subjects were divided into 2 equal groups: the dexmedetomidine group and the control group (1: 1). Patients were assigned to the groups by randomization using the statistical software R (Bell Laboratories, New Jersey, USA). During the operation and drug administration, we referred to the methods by Du et al (2018) [13]. The patients were monitored throughout the operation by 5-lead electrocardiographs (ECG), 24-h ambulatory blood pressure, and end-tidal carbon dioxide pressure. In the dexmedetomidine group, the patients received a dose of dexmedetomidine (0.5 μg/kg) before anesthesia. Subsequently, dexmedetomidine was infused intravenously at 0.5 μg/kg/h. The control group patients continuously received saline for 10 min before the administration of anesthesia.
In the operating room, each patient’s ECG and blood oxygen saturation were continuously monitored. An electroencephalogram bispectral (BIS) index monitor (Aspect Medical, Inc., Newton, USA) was connected, oxygen was administered through a mask, and an upper-limb venous infusion channel was established. Then, the patients from both groups were given the standard induction regimen for anesthetic management (sufentanil 0.5 μg/kg, propofol 1.0–2.5 mg/kg, and cisatracurium 0.15 mg/kg). After performing a double-lumen intubation for single-lung ventilation and a fiberoptic bronchoscope positioning, TATS was performed on each patient. In the dexmedetomidine group, a 0.5 μg/kg dexmedetomidine intravenous injection was administered every hour for anesthesia maintenance and 1 ng/ml to 4 ng/ml remifentanil plus sevoflurane were administered every hour. The BIS index was maintained between 40 and 60. Cisatracurium was used to maintain muscle relaxation (if necessary) and the real-time ventilator parameters were adjusted to maintain normal end-tidal carbon dioxide levels.
The key intraoperative interventions and data monitoring were that all hemodynamic changes that lasted longer than 1 minute during the operation were intervened. The intraoperative mean arterial pressure was monitored; if this index was 30% lower than the baseline, it was considered to be intraoperative hypotension, and an intravenous infusion of 0.2 mg dopamine was rapidly administered. If the intravenous infusion was not effective, then 2 mg intravenous dopamine was injected. If the intraoperative mean arterial pressure index was 30% higher than the baseline, it was considered intraoperative hypertension, and 0.2 mg intravenous nicardipine was administered. During the operation, esmolol was used to treat tachycardia (heart rate >110 beats/min). Each patient was administered parecoxib (40 mg), sufentanil (0.1 μg/kg), and 1 ml ropivacaine (0.5%) by intramuscular injection around the incision to infiltrate the surgical wound.
Outcome Observation
The data recorded for each patient were the preoperative heart rate (HRpre), preoperative systolic pressure (SYSpre), preoperative diastolic pressure (DIApre), maximum intraoperative heart rate (HRmax), minimum intraoperative heart rate (HRmin), maximum intraoperative systolic pressure (SYSmax), minimum intraoperative systolic pressure (SYSmin), maximum intraoperative diastolic pressure (DIAmax), and minimum intraoperative diastolic pressure (DIAmin). Twenty-four hours after the operation, venous blood was drawn, and an enzyme-linked immunosorbent assay was performed to assess the concentrations of cardiac troponin I (cTnI), creatine kinase isoenzyme (CK-MB), myoglobin (MB), and N-terminal pro-B-type natriuretic peptide (NT-proBNP). The patients’ hospital-stay period (from the admission date to the date of discharge) was recorded. The criteria for discharge were good condition of the patient, and the absence of myocardial infarctions, severe arrhythmias, myocardial ischemia (other cardiovascular complications), cerebral infarction, cerebral hemorrhage, pulmonary embolism, pulmonary infection, atelectasis, pulmonary hemorrhage, obvious postoperative pain, and obvious postoperative cognitive impairment, as well as a daily thoracic drainage volume <200 ml.
Statistical Analysis
Continuous variables were presented as means with standard deviations (SD) and compared using the t test or medians with interquartile ranges and the Mann-Whitney U test. Categorical variables were presented as frequencies with percentages and compared using Fisher’s exact test or the chi-square test. All statistical analyses were performed using SAS version 9.2 (SAS Institute, Inc., Cary, NC, USA). Two-sided P values of <0.05 were considered statistically significant.
This study is registered on the Chinese Clinical Trial Registry (ID: ChiCTR-IPR-1800017118) and approved by the Ethics Committee of Shanghai Pulmonary Hospital. Each patient gave informed consent to be part of this study.
Results
Clinical Characteristics of Enrolled Patients
A total of 175 patients were enrolled in the study, with 88 patients assigned to the dexmedetomidine group and 87 patients assigned to the control group. There were no significant differences in age, sex, body height, body weight, ASA classification grade, resection mode, operation time, and ejection fraction between the 2 groups (P>0.05) (Table 1). The basal heart rate, systolic pressure, and diastolic pressure were comparable (P>0.05) between the 2 groups.
Table 1.
Clinical characteristics of the study population.
| Variables | Dexmedetomidine group (n=88) | Control group (n=87) | P value | |
|---|---|---|---|---|
| Age | (years) | 66.7±6.6 | 67.6±7.0 | 0.066 |
| Sex | Males | 66 (75.0%) | 49 (55.7%) | 0.672 |
| Females | 22 (25.0%) | 39 (44.3%) | ||
| Height | (cm) | 162.8±4.3 | 167.2±2.2 | 0.542 |
| Weight | (kg) | 53.9±2.4 | 54.1±1.9 | 0.924 |
| ASA classification grade | II | 79 (89.7%) | 82 (93.1%) | 0.669 |
| III | 9 (10.3%) | 6 (6.9%) | ||
| Resection mode | Lobectomy | 61 (69.3%) | 58 (65.9%) | 0614 |
| Wedge | 13 (14.8%) | 20 (22.7%) | 0.502 | |
| Segment | 14 (15.9%) | 10 (11.3%) | 0.707 | |
| Ejection fraction | 57.4±9.2% | 56.5±7.3% | 0.673 | |
| Operation time | (hours) | 2.3±0.4 | 2.6±03 | 0.704 |
| Preoperative heart rate | (beats/min) | 70.11±12.28 | 72.24±13.35 | 0.275 |
| Preoperative systolic pressure | (mmHg) | 145.82±108.98 | 134.70±19.49 | 0.348 |
| Preoperative diastolic pressure | (mmHg) | 76.60±16.44 | 75.84±10.40 | 0.716 |
ASA – American Society of Anesthesiologists. P<0.05 was considered statistically significant.
Dexmedetomidine Decreased the Maximum Intraoperative Heart Rate
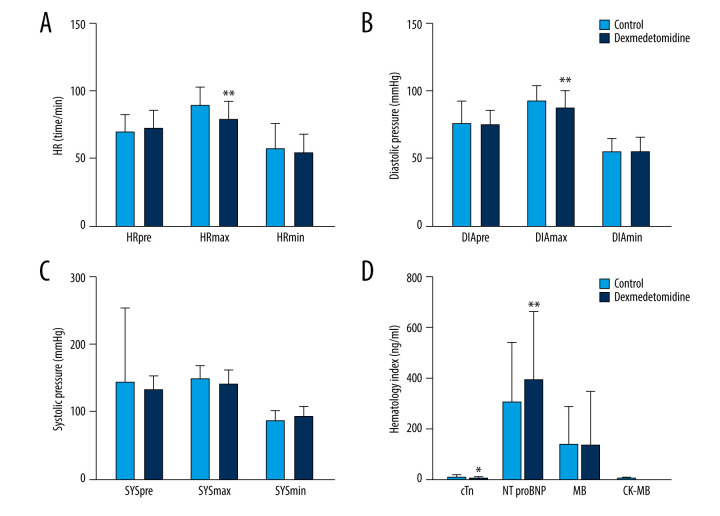
Three types of heart rate (HRpre, HRmax, and HRmin) were recorded in this trial. Only HRmax was significantly decreased in the dexmedetomidine group (t=5.142, P<0.01) (Figure 2A). The number of patients with heart rates >110 beats/min decreased in the dexmedetomidine group (1/88 vs 8/87, P<0.016) (Table 2).
Figure 2.
The intraoperative effect of dexmedetomidine on heart rate and blood pressure, and postoperative N-terminal pro-B-type natriuretic peptide. (A) Maximum intraoperative heart rate (HRmax) decreased in the dexmedetomidine group. (B) Maximum intraoperative diastolic pressure (DIAmax) significantly decreased in the dexmedetomidine group. (C) Dexmedetomidine did not impact the systolic pressure before or during the operation. (D) Dexmedetomidine increased the postoperative N-terminal pro-B-type natriuretic peptide levels and decreased the postoperative cTnI levels. * P<0.05; ** P<0.01.
Table 2.
Intraoperative hemodynamic results and the duration of hospital stay.
| Variables | Dexmedetomidine group (n=88) | Control group (n=87) | P value | |
|---|---|---|---|---|
| Intraoperative hypotension | No | 63 | 43 | 0.011 |
| Yes | 25 | 41 | ||
| Intraoperative hypertension | No | 77 | 60 | 0.003 |
| Yes | 11 | 27 | ||
| Heart rate >110 beats/min | No | 87 | 79 | 0.016 |
| Yes | 1 | 8 | ||
| Dopamine | (mg) | 0.34±0.97 | 1.70±2.10 | <0.001 |
| Nicardipine | (mg) | 0.24±0.83 | 0.70±1.50 | 0.013 |
| Atropine | (mg) | 0.25±0.33 | 0.06±0.18 | <0.001 |
| Esmolol | (mg) | 0.23±2.13 | 2.99±9.90 | 0.013 |
| Hospital stay | (days) | 5.00±1.27 | 5.57±1.44 | 0.006 |
P<0.05 was considered statistically significant.
Dexmedetomidine Decreased the Maximum Intraoperative Diastolic Pressure
The DIAmax of the dexmedetomidine group was significantly decreased compared to the control group (t=3.004, P<0.01) (Figure 2B), while no differences were observed in DIApre or DIAmin. Dexmedetomidine did not significantly affect the systolic pressure before or during the operation (P>0.05) (Figure 2C). The frequencies of intraoperative hypotension and hypertension were significantly lower in the dexmedetomidine group, and consequently the amounts of nicardipine (0.24±0.83 vs 0.70±1.50, P=0.013) and dopamine (0.34±0.97 vs 1.70±2.10, P<0.001) administered to these patients intraoperatively were significantly lower (Table 2).
Dexmedetomidine Shortened Postoperative Hospitalization
A comparison of the postoperative hospital stay periods between the 2 groups showed the dexmedetomidine group had a shorter postoperative hospital stay (5.00±1.27 days vs 5.57±1.44 days, P=0.006), which shows an additional economic advantage of dexmedetomidine administration.
Dexmedetomidine Increased Postoperative NT-ProBNP Levels
Out of the 4 postoperative indices (NT-proBNP, cTnI, CK-MB, and MB) measured in peripheral blood, the NT-proBNP levels had significantly increased in the dexmedetomidine group (t=3.757, P<0.01), and there was a significant decrease in cTnI (t=2.169, P<0.05). No changes were observed for CK-MB and MB between the 2 groups (Figure 2D). This result suggests that dexmedetomidine has a protective effect on the myocardium; however, this conclusion needs further confirmation.
Discussion
Perioperative myocardial infarction occurs in 3% of major surgery patients [14], and is the primary cause of death. Myocardial injury after a noncardiac surgery (MINS) has a 1-month mortality rate of 0.5% to 2% [15–17]. Perioperative myocardial infarction is frequently accompanied by an imbalance of the ventilation/blood flow ratio, and abnormal intraoperative blood pressure. Alpha-2 adrenoreceptor agonists exhibit strong analgesic and sedative properties; they can cause dilation of blood vessels and reduce intraoperative blood pressure.
Dexmedetomidine is widely used in current clinical anesthesia, Intensive Care Unit management, and pain treatment. It has been reported that dexmedetomidine administration improves cardiac outcomes during noncardiac surgery [18,19]. Dexmedetomidine preconditioning reduces the incidence of reperfusion-induced ventricular arrhythmias and the infarct area in animal myocardial ischemia/reperfusion models [20–22]. In patients undergoing VATS, the frequency of severe emergence agitations was lower in the dexmedetomidine group, and the length of hospital stay was significantly shorter [12]. Moreover, dexmedetomidine can exert an anti-inflammatory effect (in conditions including lung ischemia-reperfusion injury) [23], and the possible mechanisms include the reduction of serum levels of inflammatory cytokines. Animal studies have reported that dexmedetomidine can reduce myocardial ischemia-reperfusion injury in the isolated hearts of animals and plays a protective role in the heart in cardiac surgery or treatment [24–26].
This is one of the first studies to examine the intraoperative HRmax and DIAmax control for a medium-sized cohort of thoracic surgery patients under preoperative dexmedetomidine administration. We clearly demonstrated that dexmedetomidine can influence the extremum of intraoperative HR and blood pressure. Administration of dexmedetomidine at a dose of 0.5 μg/kg/h did not influence the minimum values, although it controlled the maximum values of HR and diastolic pressure. A similar effect of maximal heart rate control can be exerted by the preoperative use of a beta blocker [27], and is regarded as a potential benefit in reducing the physiologic stress and risk of perioperative cardiac morbidity.
An independent study compared the use of dexmedetomidine and propofol on hemodynamic effects and revealed that diastolic blood pressure was higher in the propofol group [28]. During thoracic surgery, when the patient is lying on their side, 1-lung ventilation can easily cause a ventilation/blood flow disorder, increase chest pressure on the ventilation side, and consequently cause a decrease in the return blood flow. At the same time, the change in pulmonary circulation pressure can lead to the insufficiency of coronary perfusion. The operation of pulling and compressing the heart can cause hemodynamic fluctuation, which can further aggravate the myocardial injury. We found HRmax was suppressed by dexmedetomidine, which helps reduce myocardial oxygen consumption and cardiac work.
The mean postoperative NT-proBNP level increased in the dexmedetomidine group. NT-proBNP is a well-established diagnostic and prognostic biomarker for heart failure and acute coronary syndromes [29]. The perioperative NT-proBNP level is a potential marker of congenital heart disease in children [30] and perioperative cardiac events in the elderly [31]. In patients receiving aortic valve intervention, the NT-proBNP level displayed a transient early postoperative increase with a peak on day 3 [29]. The correlation between the blood NT-proBNP level and dexmedetomidine has rarely been observed. Only 1 associated study showed the opposite result, where cTnI, NT-proBNP, and high-sensitivity C-reactive protein significantly decreased in the dexmedetomidine group [32]. This may be due to their study population being elderly patients diagnosed with coronary heart disease, who underwent gastric cancer surgery. The cohorts were distinct between the present study and the above study [32]. Nevertheless, they demonstrated a conclusion consistent with the present study; the incidence of bradycardia increased, although the incidence of silent myocardial ischemia/arrhythmia decreased in the dexmedetomidine group. Another possible explanation is that dexmedetomidine exerts a vasodilatory effect, and more fluid can be used to stabilize the hemodynamics. Perhaps the infusion rate or infused volume could affect the NT-proBNP for transient cardiac congestion or decreased renal function, which degrades the NT-proBNP. However, this hypothesis needs to be verified by further trials. To date, this is the first report that observed an increase of postoperative NT-proBNP in the dexmedetomidine-treated patients. However, more evidence is needed to confirm this conclusion and the potential mechanism.
There are some limitations in this study. First, the mechanism of the effect of dexmedetomidine on perioperative hemodynamics and myocardial protection is still unknown. In limited cases, there are large standard errors in some indices (eg, SYSpre in Figure 2C), which means the result is not completely conclusive. Finally, whether the postoperative NT-proBNP levels will increase with the dexmedetomidine treatment is still to be clarified; and if this is an adverse reaction to dexmedetomidine, avoiding this reaction is a challenge to overcome.
Conclusions
Preoperative and operative administration of dexmedetomidine can control the hemodynamic stability and have a protective effect on the cardiovascular system in the intraoperative and postoperative periods, as well as shorten the postoperative hospital stay.
Acknowledgements
We appreciate the participation of all the patients in this study.
Footnotes
Name of Department and Institution Where Work Was Done
Shanghai Pulmonary Hospital Affiliated to Tongji University, Shanghai, China.
Conflicts of Interest
None.
Source of support: This study was supported by the Academic Elite Program of Shanghai Pulmonary Hospital (FK1809) and the Special Program of Shanghai Aging and Women and Children’s Health Research (2020YJZX0136)
References
- 1.Ramsay MA, Luterman DL. Dexmedetomidine as a total intravenous anesthetic agent. Anesthesiology. 2004;101(3):787–90. doi: 10.1097/00000542-200409000-00028. [DOI] [PubMed] [Google Scholar]
- 2.Hassan ME, Mahran E. Evaluation of the role of dexmedetomidine in improvement of the analgesic profile of thoracic paravertebral block in thoracic surgeries: A randomised prospective clinical trial. Indian J Anaesth. 2017;61(10):826–31. doi: 10.4103/ija.IJA_221_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kamali A, Taghizadeh M, Esfandiar M, et al. A Comparison of the effects of dexmedetomidine and propofol in controlling the hemodynamic responses after intubation: A double-blind, randomized, clinical trial study. Open Access Maced J Med Sci. 2018;6(11):2045–50. doi: 10.3889/oamjms.2018.385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dere K, Sucullu I, Budak ET, et al. A comparison of dexmedetomidine versus midazolam for sedation, pain and hemodynamic control, during colonoscopy under conscious sedation. Eur J Anaesthesiol. 2010;27(7):648–52. doi: 10.1097/EJA.0b013e3283347bfe. [DOI] [PubMed] [Google Scholar]
- 5.Khetarpal M, Yadav M, Kulkarni D, et al. Role of dexmedetomidine and sevoflurane in the intraoperative management of patient undergoing resection of phaeochromocytoma. Indian J Anaesth. 2014;58(4):496–97. doi: 10.4103/0019-5049.139028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bashir MA, Lyle BC, Nasr AS, et al. Video-assisted thoracoscopic surgery approach for transmyocardial laser revascularization. Interact Cardiovasc Thorac Surg. 2017;25(5):848–49. doi: 10.1093/icvts/ivx146. [DOI] [PubMed] [Google Scholar]
- 7.Kaur M, Ramakumar N, Yadav S, et al. Recurrent syncope in a child and video assisted thoracoscopic surgery-The long and short of it. Indian J Anaesth. 2019;63(10):847–50. doi: 10.4103/ija.IJA_180_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gross DJ, Chang EH, Rosen PL, et al. Outcomes in video-assisted thoracoscopic surgery lobectomies: challenging preconceived notions. J Surg Res. 2018;231:161–66. doi: 10.1016/j.jss.2018.05.040. [DOI] [PubMed] [Google Scholar]
- 9.Anile M, Diso D, Mantovani S, et al. Uniportal video assisted thoracoscopic lobectomy: Going directly from open surgery to a single port approach. J Thorac Dis. 2014;6(Suppl 6):S641–43. doi: 10.3978/j.issn.2072-1439.2014.08.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Iwata Y, Hamai Y, Koyama T. Anesthetic management of nonintubated video-assisted thoracoscopic surgery using epidural anesthesia and dexmedetomidine in three patients with severe respiratory dysfunction. J Anesth. 2016;30(2):324–27. doi: 10.1007/s00540-015-2122-9. [DOI] [PubMed] [Google Scholar]
- 11.Tan XL, Chen YY, Hu B, et al. [Dose-response relationship of dexmedetomidine combined with sufentanil for postoperative intravenous analgesia in video-assisted thoracoscopic surgery]. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2019;41(3):373–78. doi: 10.3881/j.issn.1000-503X.10881. [in Chinese] [DOI] [PubMed] [Google Scholar]
- 12.Lee SH, Lee CY, Lee JG, et al. Intraoperative dexmedetomidine improves the quality of recovery and postoperative pulmonary function in patients undergoing video-assisted thoracoscopic surgery: A CONSORT – prospective, randomized, controlled trial. Medicine (Baltimore) 2016;95(7):e2854. doi: 10.1097/MD.0000000000002854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Du X, Yu J, Mi W. The effect of dexmedetomidine on the perioperative hemodynamics and postoperative cognitive function of elderly patients with hypertension: Study protocol for a randomized controlled trial. Medicine (Baltimore) 2018;97(43):e12851. doi: 10.1097/MD.0000000000012851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sellers D, Srinivas C, Djaiani G. Cardiovascular complications after non-cardiac surgery. Anaesthesia. 2018;73(Suppl 1):34–42. doi: 10.1111/anae.14138. [DOI] [PubMed] [Google Scholar]
- 15.Kristensen SD, Knuuti J, Saraste A, et al. 2014 ESC/ESA Guidelines on non-cardiac surgery: Cardiovascular assessment and management: The Joint Task Force on non-cardiac surgery: Cardiovascular assessment and management of the European Society of Cardiology (ESC) and the European Society of Anaesthesiology (ESA) Eur Heart J. 2014;35(35):2383–431. doi: 10.1093/eurheartj/ehu282. [DOI] [PubMed] [Google Scholar]
- 16.Costa MC, Furtado MV, Borges FK, et al. Perioperative troponin screening identifies patients at higher risk for major cardiovascular events in noncardiac surgery. Curr Probl Cardiol. 2021;46(3):100429. doi: 10.1016/j.cpcardiol.2019.05.002. [DOI] [PubMed] [Google Scholar]
- 17.Sessler DI, Devereaux PJ. Perioperative troponin screening. Anesth Analg. 2016;123(2):359–60. doi: 10.1213/ANE.0000000000001450. [DOI] [PubMed] [Google Scholar]
- 18.Biccard BM, Goga S, de Beurs J. Dexmedetomidine and cardiac protection for non-cardiac surgery: A meta-analysis of randomised controlled trials. Anaesthesia. 2008;63(1):4–14. doi: 10.1111/j.1365-2044.2007.05306.x. [DOI] [PubMed] [Google Scholar]
- 19.Duncan D, Sankar A, Beattie WS, et al. Alpha-2 adrenergic agonists for the prevention of cardiac complications among adults undergoing surgery. Cochrane Database Syst Rev. 2018;3(3):CD004126. doi: 10.1002/14651858.CD004126.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yoshitomi O, Cho S, Hara T, et al. Direct protective effects of dexmedetomidine against myocardial ischemia-reperfusion injury in anesthetized pigs. Shock. 2012;38(1):92–97. doi: 10.1097/SHK.0b013e318254d3fb. [DOI] [PubMed] [Google Scholar]
- 21.Willigers HM, Prinzen FW, Roekaerts PM, et al. Dexmedetomidine decreases perioperative myocardial lactate release in dogs. Anesth Analg. 2003;96(3):657–64. doi: 10.1213/01.ANE.0000048708.75957.FF. table of contents. [DOI] [PubMed] [Google Scholar]
- 22.Okada H, Kurita T, Mochizuki T, et al. The cardioprotective effect of dexmedetomidine on global ischaemia in isolated rat hearts. Resuscitation. 2007;74(3):538–45. doi: 10.1016/j.resuscitation.2007.01.032. [DOI] [PubMed] [Google Scholar]
- 23.Kong L, Lu XH. [Effect of dexmedetomidine on perioperative inflammatory response and cellular immune in patients undergoing radical operation of thoracoscopic lung cancer]. Zhonghua Yi Xue Za Zhi. 2018;98(36):2929–32. doi: 10.3760/cma.j.issn.0376-2491.2018.36.011. [in Chinese] [DOI] [PubMed] [Google Scholar]
- 24.Tan F, Chen Y, Yuan D, et al. Dexmedetomidine protects against acute kidney injury through downregulating inflammatory reactions in endotoxemia rats. Biomed Rep. 2015;3(3):365–70. doi: 10.3892/br.2015.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Taniguchi T, Kidani Y, Kanakura H, et al. Effects of dexmedetomidine on mortality rate and inflammatory responses to endotoxin-induced shock in rats. Crit Care Med. 2004;32(6):1322–26. doi: 10.1097/01.ccm.0000128579.84228.2a. [DOI] [PubMed] [Google Scholar]
- 26.Zhang JJ, Peng K, Zhang J, et al. Dexmedetomidine preconditioning may attenuate myocardial ischemia/reperfusion injury by down-regulating the HMGB1-TLR4-MyD88-NF-small ka, CyrillicB signaling pathway. PLoS One. 2017;12(2):e0172006. doi: 10.1371/journal.pone.0172006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mudumbai SC, Wagner T, Mahajan S, et al. Vascular surgery patients prescribed preoperative beta-blockers experienced a decrease in the maximal heart rate observed during induction of general anesthesia. J Cardiothorac Vasc Anesth. 2012;26(3):414–19. doi: 10.1053/j.jvca.2011.09.027. [DOI] [PubMed] [Google Scholar]
- 28.Białka S, Copik M, Karpe J, et al. Effect of dexmedetomidine or propofol sedation on haemodynamic stability of patients after thoracic surgery. Anaesthesiol Intensive Ther. 2018;50(5):359–66. doi: 10.5603/AIT.a2018.0046. [DOI] [PubMed] [Google Scholar]
- 29.Hultkvist H, Holm J, Svedjeholm R, et al. Rise and fall of NT-proBNP in aortic valve intervention. Open Heart. 2018;5(1):e000739. doi: 10.1136/openhrt-2017-000739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Qu J, Liang H, Zhou N, et al. Perioperative NT-proBNP level: Potential prognostic markers in children undergoing congenital heart disease surgery. J Thorac Cardiovasc Surg. 2017;154(2):631–40. doi: 10.1016/j.jtcvs.2016.12.056. [DOI] [PubMed] [Google Scholar]
- 31.Kaneko T. [Preoperative NT-proBNP predicts perioperative cardiac events in hip joint fracture surgery of the elderly]. Masui. 2012;61(6):574–78. [in Japanese] [PubMed] [Google Scholar]
- 32.Shen J, Sun Y, Han D, et al. [Effects of dexmedetomidine on perioperative cardiac adverse events in elderly patients with coronary heart disease]. Zhong NanDa Xue Xue Bao Yi Xue Ban. 2017;42(5):553–57. doi: 10.11817/j.issn.1672-7347.2017.05.012. [in Chinese] [DOI] [PubMed] [Google Scholar]