Abstract
The coronavirus disease 2019 (COVID-19) pandemic is wreaking havoc across the globe. This pandemic has given rise to a mindset where physicians tend to neglect other causes of pneumonia, especially if the patient presents with signs and symptoms commonly associated with COVID-19. Herein, we report a case of a young man presenting to the emergency department with common clinical, radiological, and laboratory features of COVID-19 pneumonia such as shortness of breath, hypoxia, pulmonary embolism, elevated D-dimer, and bilateral ground glass opacities on computed tomography of the chest but was later diagnosed with Pneumocystis pneumonia that was treated with appropriate antibiotics and corticosteroids. This case highlights the importance of performing a thorough clinical history and differentiating the clinical and radiological features of COVID-19 pneumonia from pneumonia of other etiologies.
1. Introduction
Since the first case of COVID-19 was diagnosed in December 2019, it has rapidly spread across the globe, resulting in the COVID-19 pandemic. Presently, with more than 100 million cases of COVID-19 globally, it is not uncommon for physicians to think of COVID-19 as an initial diagnosis for patients presenting with pneumonia [1]. In this report, we discuss a case of a young adult with unremarkable past medical history presenting to the emergency department with shortness of breath and diarrhea. Computed tomography (CT) of the chest revealed diffuse pulmonary bilateral ground glass opacities and pulmonary embolism. Recognizing the differences in radiological and clinical features of COVID-19 and Pneumocystis pneumonia helped to reach the correct diagnosis and, thus, initiate the appropriate treatment for the patient.
2. Case Presentation
A 23-year-old male presented to the emergency department (ED) with a chief complaint of diarrhea for the last one week. On examination, his vital signs were as follows: temperature, 101.8 F; blood pressure, 125/76 mmHg; pulse rate, 126/min; and respiratory rate, 24/min. His blood oxygen saturation was 85% at room air and 95% with 5 L oxygen via nasal cannula. His body mass index (BMI) was 25.1 kg/m2. Chest examination revealed decreased breath sounds with bilateral crackles in the lower part of lung fields and normal first and second heart sounds.
Initial laboratory investigations revealed these results: white blood cell count (WBC), 7700/ul with lymphopenia; prothrombin time (PT), 16.6 seconds; international normalized ratio (INR), 1.4; activated partial thromboplastin time (APTT), 38.5 seconds; D-dimer, 3.85 mcg/mL, serum lactate dehydrogenase (LDH), 402 U/L; and C-reactive protein (CRP), 3.63 mg/dl. Arterial blood gas (ABG) analysis showed a pH of 7.53, paCO2 of 33 mmHg, paO2 of 62 mmHg, serum HCO3− level of 27.6 mmol/L, and alveolar-arterial (A-a) oxygen gradient of 46.5 mmHg.
A chest X-ray (Figure 1) showed patchy bilateral pulmonary opacities. The patient's clinical presentation of shortness of breath and hypoxia together with elevated D-dimer levels prompted a chest CT to rule out pulmonary embolism. The chest CT demonstrated notable diffuse bilateral ground glass opacities (Figure 2) and arterial embolism in the pulmonary arteries supplying the right middle and lower lobes.
Figure 1.
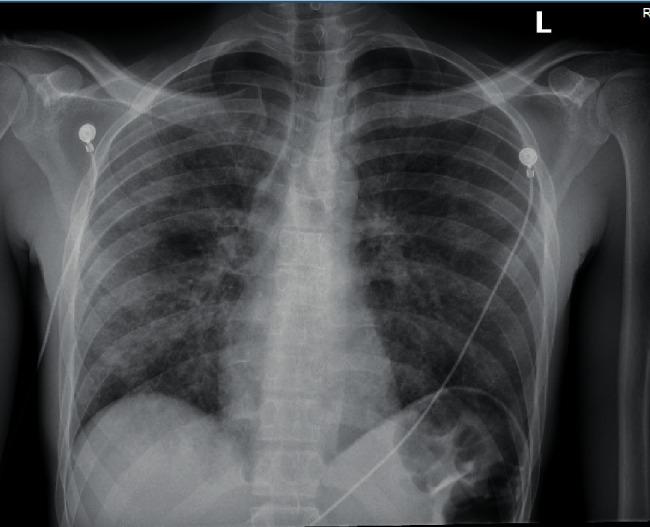
CXR showing bilateral pulmonary infiltrates.
Figure 2.
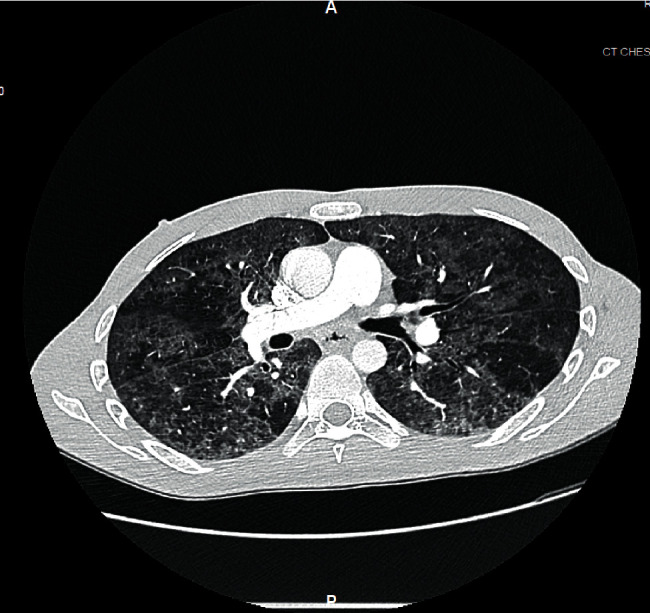
Chest CT showing diffuse bilateral ground glass opacities.
Reverse-transcription polymerase chain reaction (RT-PCR) for COVID-19 ribonucleic acid (RNA) conducted on a nasopharyngeal swab sample of the patient revealed a negative result. However, the patient was still considered to have clinical COVID-19, given he had typical features of COVID-19 such as hypoxia, shortness of breath, elevated D-dimer, pulmonary embolism, and ground glass opacities on chest CT. Thereby, dexamethasone (6 mg) for COVID-19 and apixaban for pulmonary embolism were initiated. Decision was made to hold remdesivir since COVID-19 was not confirmed in this patient.
During further questioning, the patient reported productive yellowish cough during the last month coupled with anorexia and significant weight loss of 15 pounds. The insidious onset of the long-lasting symptoms and the radiological features characterized by ground glass opacities seen in Pneumocystis pneumonia as opposed to patchy and peripheral ground glass opacities of typical COVID-19 pneumonia (Figure 3) prompted further investigation for potential occult immunodeficiency. The fourth-generation Ag/Ab combination human immunodeficiency virus (HIV)-1/2 immunoassay showed a positive result for HIV-1 antibody and negative for HIV-2 antibody. Repeat PCR tests for COVID-19 on day 3 and day 5 of hospitalization were negative as well. Thereafter, IV trimethoprim-sulfamethoxazole 400 mg every 8 hours was started for possible Pneumocystis pneumonia (PCP) given the new diagnosis of HIV. Dexamethasone was switched to prednisone 40 mg oral twice a day.
Figure 3.
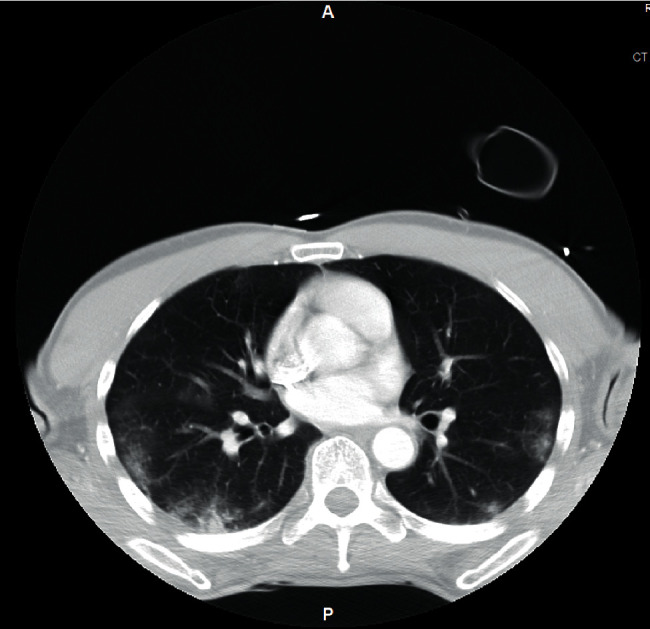
Chest CT of the COVID-19 patient showing patchy and peripheral ground glass opacities mid to lower lobe predominant (taken from another COVID-19-positive patient for comparison).
Bronchoscopy was performed on day 4 of hospitalization, and bronchoalveolar culture, Gram stain, acid-fast bacilli (AFB) stain, silver stain, and Pneumocystis jirovecii PCR were performed on the specimen obtained. Cells obtained via bronchial washing were negative for malignancy, and no fungal elements were visualized by methenamine silver stain. No acid-fast bacilli were detected on AFB staining, and neither any Mycobacterium species was isolated in the mycobacterial culture. HIV viral load was 1.4 million copies/ml, and total cluster of differentiation 4 (CD4) count was <20 cells/mcl. Treatment with oral emtricitabine/tenofovir and oral dolutegravir was, thus, initiated.
PCR for Pneumocystis jirovecii on material obtained via BAL revealed a positive result. The patient's presentation of chronic symptoms as opposed to acute presentation of COVID-19 patients together with a positive HIV test, high viral load, low CD4 count, and positive PCR for Pneumocystis jirovecii contributed to establishing the diagnosis of Pneumocystis pneumonia. After one more week of continued treatment with IV antibiotics and prednisone, the patient's condition improved and he was discharged with instructions for home oxygen therapy and prescription for apixaban, trimethoprim-sulfamethoxazole (800 mg/160 mg), prednisone, dolutegravir, and emtricitabine/tenofovir.
3. Discussion
In the current situation of the COVID-19 pandemic, there have been an increasing number of patients presenting with severe symptoms of the disease, such as cough, fever, shortness of breath, hypoxia, and diarrhea. Pneumocystis pneumonia is also characterized by an insidious onset of similar symptoms when found in association with other opportunistic infections. Given these similarities, the diagnosis of Pneumocystis pneumonia may take a backseat in the setting of increasing COVID-19 incidence in the present scenario [2]. In the present case, the patient's presentation of hypoxia, shortness of breath, and diarrhea together with elevated D-dimer and LDH levels along with pulmonary ground glass opacities and pulmonary embolism seen on chest CT made it difficult to differentiate between COVID-19 and Pneumocystis pneumonia.
Elevated levels of serum LDH, inflammatory markers (CRP and ferritin), and D-dimer are common laboratory findings associated with COVID-19 [3–5]. Diagnostic tests for COVID-19 include nucleic acid amplification tests (NAAT) such as RT-PCR and antigen testing. RT-PCR assay of COVID-19 RNA from the upper respiratory tract is the preferred initial diagnostic test for COVID-19 with a reported sensitivity of 77% [6, 7].
On the other hand, the common laboratory findings of Pneumocystis pneumonia are low CD-4 counts (<200 cells/microliter), widened alveolar-arterial oxygen gradient, and elevated LDH level [8–10]. Diagnostic tests for Pneumocystis pneumonia include microscopy with staining and a PCR test. Gomori-methenamine silver is the commonly used stain to detect Pneumocystis pneumonia. The sensitivity and specificity of Grocott-Gomori-methenamine silver stain (GMS) are 79.4 and 99.2%, respectively [11]. PCR has a sensitivity of as high as 96% and specificity of more than 90% in detecting Pneumocystis jirovecii in respiratory samples [12–14]. PCR is the most sensitive method for the detection of Pneumocystis and should be considered the diagnostic test of choice for patients with high suspicion of Pneumocystis pneumonia with negative results on GMS stain, as was encountered in the present case [15].
Both COVID-19 and Pneumocystis pneumonia can present with ground glass opacities in the lungs on chest CT, and there are no pathognomonic radiographic findings associated with either of the two pathologies that would lead to a definite diagnosis. In general, Pneumocystis pneumonia is characterized by diffuse infiltrates mainly involving the upper lobe while sparing the subpleural regions, whereas COVID-19 is characterized by patchy infiltrates, mainly in the lower lobes with peripheral and subpleural distribution [16–18]. COVID-19 is commonly associated with hypercoagulability which can lead to pulmonary embolism [19–21], whereas pulmonary embolism is not a common feature of Pneumocystis pneumonia [22]. Elevated LDH is a common laboratory abnormality associated with both COVID-19 and Pneumocystis pneumonia, while widened alveolar-arterial oxygen gradient is more commonly found in Pneumocystis pneumonia.
Since the treatment protocols of COVID-19 and Pneumocystis pneumonia are entirely different, it is very important to reach the correct diagnosis. COVID-19 treatment includes remdesevir, baricitinib, or tocilizumab, while the standard regimen for Pneumocystis pneumonia is trimethoprim-sulfamethoxazole for 21 days [23–26]. Corticosteroid of choice used in COVID-19 treatment is dexamethasone with a suggested dosing of 6 mg once daily for 10 days [27–29], while the corticosteroid indicated for moderate-to-severe Pneumocystis pneumonia (partial pressure of oxygen <70 mmHg at standard room conditions and/or an A-a oxygen gradient of ≥35 mmHg) is prednisone 40 mg twice a day with subsequent taper [30, 31]. Baricitinib and tocilizumab are immunomodulatory drugs that are indicated for COVID-19 treatment. In the present case, it was of utmost importance to reach the correct diagnosis in order to avoid administration of immunomodulatory agents to an HIV patient with opportunistic infection of Pneumocystis jirovecii, which could have led to deterioration in the patient's condition.
4. Conclusions
We report a case of Pneumocystis pneumonia presenting with features mimicking COVID-19 pneumonia. Increased COVID-19 cases in the scenario of the pandemic have led physicians into neglecting other causes of pneumonia and respiratory failure. This case highlights the importance of medical history taking and detailed clinical examination. It also highlights the importance of understanding the differences between clinical and radiological features of COVID-19 and Pneumocystis pneumonia.
Abbreviation
- F:
Fahrenheit
- mmHg:
Millimeter of mercury
- min:
Minute
- g/dl:
Gram per deciliter
- mg/dl:
Milligram per deciliter
- k/ul:
Kilo per microliter
- mcg/mL FEU:
Microgram/mL fibrinogen equivalent units
- U/L:
Unit per liter
- mmol/L:
Millimole per liter
- ml:
Milliliter
- mcl:
Microliter.
Data Availability
No data were used to support this study.
Consent
No written consent has been obtained from the patients as there are no patient-identifiable data included in this case report.
Conflicts of Interest
None of the authors have any conflicts of interest.
References
- 1.Coleman J. J., Manavi K., Marson E. J., Botkai A. H., Sapey E. COVID-19: to be or not to be; that is the diagnostic question. Postgraduate Medical Journal. 2020;96(1137):392–398. doi: 10.1136/postgradmedj-2020-137979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mang S., Kaddu-Mulindwa D., Metz C., et al. Pneumocystis jirovecii pneumonia and severe acute respiratory syndrome coronavirus 2 coinfection in a patient with newly diagnosed HIV-1 infection. Clinical Infectious Diseases. 2021;72(8):1487–1489. doi: 10.1093/cid/ciaa906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhou F., Yu T., Du R., et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wu C., Chen X., Cai Y., et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Internal Medicine. 2020;180(7):934–943. doi: 10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shi S., Qin M., Shen B., et al. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China. JAMA Cardiology. 2020;5(7):802–810. doi: 10.1001/jamacardio.2020.0950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Patel A., Jernigan D. B. Initial public health response and interim clinical guidance for the 2019 novel coronavirus outbreak-United States, december 31, 2019-February 4, 2020. Morbidity and Mortality Weekly Report. 2020;69(5):140–146. doi: 10.15585/mmwr.mm6905e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Clerici B., Muscatello A., Bai F., et al. Sensitivity of SARS-COVID-2 detection with nasopharyngeal swabs. Front Public Health. 2021;8 doi: 10.3389/fpubh.2020.593491.593491 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Stansell J. D., Osmond D. H., Charlebois E., et al. Predictors of pneumocystis carinii pneumonia in HIV-infected persons. Pulmonary complications of HIV infection study group. American Journal of Respiratory and Critical Care Medicine. 1997;155(1):60–66. doi: 10.1164/ajrccm.155.1.9001290. [DOI] [PubMed] [Google Scholar]
- 9.Miller R. F., Huang L., Walzer P. D. Pneumocystis pneumonia associated with human immunodeficiency virus. Clinics in Chest Medicine. 2013;34(2):229–241. doi: 10.1016/j.ccm.2013.02.001. [DOI] [PubMed] [Google Scholar]
- 10.Zaman M. K., White D. A. Serum lactate dehydrogenase levels and Pneumocystis carinii pneumonia. Diagnostic and prognostic significance. The American Review of Respiratory Disease. 1988;137(4):796–800. doi: 10.1164/ajrccm/137.4.796. [DOI] [PubMed] [Google Scholar]
- 11.Procop G. W., Haddad S., Quinn J., et al. Detection of Pneumocystis jiroveci in respiratory specimens by four staining methods. Journal of Clinical Microbiology. 2004;42(7):3333–3335. doi: 10.1128/JCM.42.7.3333-3335.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alvarez-Martinez M. J., Miro J. M., Valls M. E., et al. Sensitivity and specificity of nested and real-time PCR for the detection of Pneumocystis jiroveci in clinical specimens. Diagnostic Microbiology and Infectious Disease. 2006;56:153–160. doi: 10.1016/j.diagmicrobio.2006.03.010. [DOI] [PubMed] [Google Scholar]
- 13.Flori P., Bellete B., Durand F., et al. Comparison between real-time PCR, conventional PCR and different staining techniques for diagnosing Pneumocystis jiroveci pneumonia from bronchoalveolar lavage specimens. Journal of Medical Microbiology. 2004;53(Pt 7):603–607. doi: 10.1099/jmm.0.45528-0. [DOI] [PubMed] [Google Scholar]
- 14.Caliendo A. M., Hewitt P. L., Allega J. M., Keen A., Ruoff K. L., Ferraro M. J. Performance of a PCR assay for detection of Pneumocystis carinii from respiratory specimens. Journal of Clinical Microbiology. 1998;36(4):979–982. doi: 10.1128/JCM.36.4.979-982.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Doyle L., Vogel S., Procop G. W. Pneumocystis PCR: it is time to make PCR the test of choice. Open Forum Infectious Diseases. 2017;4(4) doi: 10.1093/ofid/ofx193.ofx193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Salehi S., Abedi A., Balakrishnan S., Gholamrezanezhad A. Coronavirus disease 2019 (COVID-19): a systematic review of imaging findings in 919 patients. American Journal of Roentgenology. 2020;215(1):87–93. doi: 10.2214/AJR.20.23034. [DOI] [PubMed] [Google Scholar]
- 17.Fujii T., Nakamura T., Iwamoto A. Pneumocystis pneumonia in patients with HIV infection: clinical manifestations, laboratory findings, and radiological features. Journal of Infection and Chemotherapy. 2007;13(1):1–7. doi: 10.1007/s10156-006-0484-5. [DOI] [PubMed] [Google Scholar]
- 18.Hani C., Trieu N. H., Saab I., et al. COVID-19 pneumonia: a review of typical CT findings and differential diagnosis. Diagn Interv Imaging. 2020;101(5):263–268. doi: 10.1016/j.diii.2020.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ackermann M., Verleden S. E., Kuehnel M., et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in COVID-19. The New England Journal of Medicine. 2020;383(2):120–128. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Menter T., Haslbauer J. D., Nienhold R., et al. Postmortem examination of COVID-19 patients reveals diffuse alveolar damage with severe capillary congestion and variegated findings in lungs and other organs suggesting vascular dysfunction. Histopathology. 2020;77(2):198–209. doi: 10.1111/his.14134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Connors J. M., Levy J. H. Thromboinflammation and the hypercoagulability of COVID-19. Journal of Thrombosis and Haemostasis. 2020;18(7):1559–1561. doi: 10.1111/jth.14849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. https://ijisrt.com/assets/upload/files/IJISRT20JUL095.pdf%20.
- 23.Eli Lilly and Company. Fact Sheet for Healthcare Providers: Emergency Use Authorization (EUA) of Baricitinib. Indianapolis, IN, USA: Eli Lilly and Company; 2020. https://www.fda.gov/media/143823/download. [Google Scholar]
- 24.National Institutes of Health. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. Bethesda, MD, USA: National Institutes of Health; 2021. https://covid19treatmentguidelines.nih.gov/ [PubMed] [Google Scholar]
- 25.IDSA. Infectious Diseases Society of America Guidelines on the Treatment and Management of Patients with COVID-19. Arlington, VA, USA: IDSA; 2020. https://www.idsociety.org/practice-guideline/covid-19-guideline-treatment-and-management. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.UpToDate. Treatment-and-Prevention-of-Pneumocystis-Infection-In-Patients-with-HIV. Waltham, MA, USA: UpToDate; 2021. https://www.uptodate.com/contents/treatment-and-prevention-of-pneumocystis-infection-in-patients-with-hiv. [Google Scholar]
- 27.Sterne J. A. C., Murthy S., Diaz J. V., et al. Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19: a meta-analysis. JAMA. 2020;324(13):1330–1341. doi: 10.1001/jama.2020.17023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Siemieniuk R. A., Bartoszko J. J., Ge L., et al. Drug treatments for COVID-19: living systematic review and network meta-analysis. BMJ. 2020;370 doi: 10.1136/bmj.m2980.m2980 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Horby P., Lim W. S., Linsell L., et al. Dexamethasone in hospitalized patients with COVID-19. The New England Journal of Medicine. 2021;384(8):693–704. doi: 10.1056/NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.National Institutes of Health-University of California. Consensus statement on the use of corticosteroids as adjunctive therapy for pneumocystis pneumonia in the acquired immunodeficiency syndrome. The New England Journal of Medicine. 1990;323(21):1500–1504. doi: 10.1056/NEJM199011223232131. [DOI] [PubMed] [Google Scholar]
- 31. Available at: https://clinicalinfo.hiv.gov/en/guidelines/adult-and-adolescent-opportunistic-infection/whats-new-guidelines.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data were used to support this study.


