Abstract
Background
Current studies of asthma history on coronavirus disease 2019 (COVID-19) outcomes are limited and lack consideration of disease status.
Objective
To conduct a population-based study to assess asthma disease status and chronic obstructive pulmonary disease (COPD) in relation to COVID-19 severity.
Methods
Patients diagnosed with COVID-19 (n = 61,338) in a large, diverse integrated health care system were identified. Asthma/COPD history, medication use, and covariates were extracted from electronic medical records. Asthma patients were categorized into those with and without clinical visits for asthma 12 or fewer months prior to COVID-19 diagnosis and labeled as active and inactive asthma, respectively. Primary outcomes included COVID-19–related hospitalizations, intensive respiratory support (IRS), and intensive care unit admissions within 30 days, and mortality within 60 days after COVID-19 diagnosis. Logistic and Cox regression were used to relate COVID-19 outcomes to asthma/COPD history.
Results
The cohort was 53.9% female and 66% Hispanic and had a mean age of 43.9 years. Patients with active asthma had increased odds of hospitalization, IRS, and intensive care unit admission (odds ratio 1.47-1.66; P < .05) compared with patients without asthma or COPD. No increased risks were observed for patients with inactive asthma. Chronic obstructive pulmonary disease was associated with increased risks of hospitalization, IRS, and mortality (odds ratio and hazard ratio 1.27-1.67; P < .05). Among active asthma patients, those using asthma medications had greater than 25% lower odds for COVID-19 outcomes than those without medication.
Conclusions
Patients with asthma who required clinical care 12 or fewer months prior to COVID-19 or individuals with COPD history are at increased risk for severe COVID-19 outcomes. Proper medication treatment for asthma may lower this risk.
Key words: Asthma, COPD, COVID-19, Hospitalization, Severity, Mortality
Abbreviations used: COPD, Chronic obstructive pulmonary disease; COVID-19, Coronavirus disease 2019; HR, Hazard ratio; ICU, Intensive care unit; IRS, Intensive respiratory support; KPSC, Kaiser Permanente Southern California; PCR, Polymerase chain reaction
What is already known about this topic? Past studies evaluating asthma and coronavirus disease 2019 (COVID-19) severity have had inconsistent findings and lack consideration of asthma disease status.
What does this article add to our knowledge? Increased risks for severe COVID-19 outcomes were observed for asthma patients who required recent clinical care, but not for those who did not require clinical care. Medication treatment for asthma appeared to lower this risk.
How does this study impact current management guidelines? These findings suggest that asthma patients, especially those who require clinical care, should continue taking control medications during the COVID-19 pandemic.
Introduction
The novel coronavirus disease 2019 (COVID-19) has caused over 3.9 million deaths across the world as of June 2021. Asthma and chronic obstructive pulmonary disease (COPD) are listed by the U.S. Centers for Disease Control and Prevention1 as potential comorbid conditions associated with severe COVID-19, but results from the current literature have been inconsistent. Whereas some studies have suggested that asthma or COPD histories increase the risk of severe illness from COVID-19,2 , 3 other studies have reported protective associations or no increased risk for patients with asthma or COPD for COVID-19 infection and severity.4, 5, 6, 7 These conflicting findings across studies may be attributed to relatively small sample sizes (n < 1,000), differences in study populations, and lack of full consideration of disease status, medication treatment history, and social characteristics.8
Given the discrepancies in the existing literature and high prevalence of asthma and COPD in the general population, there is a strong need for a more thorough assessment of the relationship between asthma and COPD and COVID-19 severity and mortality in a large and ethnically diverse population. In particular, a closer examination that accounts for the timing of asthma-related episodes, medication treatment history, and differences across certain patient characteristics (eg, race/ethnicity, age) may improve our understanding of these disease relationships and clarify the varying results from past studies.
To fill these gaps, we used a large cohort of adult COVID-19 patients from a large integrated health care system to assess the effects of preexisting asthma and COPD on a spectrum of COVID-19 severity outcomes. Potentially different effects of asthma disease status were investigated based on whether asthma history was accompanied with clinical visits for asthma and/or asthma medication use in the 12 months prior to the COVID-19 diagnosis. In addition, we sought to explore whether the relationship between asthma/COPD and COVID-19 outcomes varied across age groups, gender, and race/ethnicity.
Methods
Study population
This is a retrospective study with data prospectively collected in the electronic medical records from Kaiser Permanente Southern California (KPSC). KPSC is a large integrated health care system serving over 4 million members across Southern California. The patient population is representative of the racially diverse population of the geographic region. For this study, all adult KPSC patients with a confirmed COVID-19 diagnosis from March 1 to August 31, 2020 were included. Patients were defined as COVID-19 cases if they had a positive severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) polymerase chain reaction (PCR) laboratory test or a diagnosis code (International Classification of Diseases, Tenth Revision [ICD-10] and internal KPSC codes) for COVID-19 (Table E1; available in this article's Online Repository at www.jaci-inpractice.org) during this period. To eliminate potential false-positive cases, patients who had asymptomatic COVID-19 diagnosis codes and negative laboratory test results within 2 weeks after the diagnosis were excluded (n = 491). The index date was defined as the date of the earliest laboratory order for those with laboratory data and the date of the earliest COVID-19 diagnosis code for those with only diagnosis code data.
Patients were excluded if they were nonmembers or members for less than 1 year and thus had incomplete medical data (n = 16,702) or had other/unknown gender (n = 8). The KPSC institutional review board approved this study, with waiver of informed consent.
Exposures of interest
ICD-10 codes were used to identify patients with a history of asthma (J45) or COPD (J43-J44), as done in previous literature.9 Patients were defined as having asthma or COPD if they had at least 1 inpatient/emergency department code or at least 2 outpatient codes for the given condition prior to the COVID-19 diagnosis date. COPD history was only assessed for individuals aged 35 years and older. Individuals who had both asthma and COPD codes were included in the COPD group. Asthma disease status was further categorized based on whether the patient had any clinical visit (scheduled or unscheduled) with an asthma diagnosis code (primary or nonprimary diagnosis) in the 12 months prior to the COVID-19 diagnosis. Individuals with a clinical visit were labeled as active asthma, whereas those without a visit were labeled as inactive asthma. These labels were used to represent asthma status as defined by health care utilization, rather than by asthma severity.
For all patients with asthma, medication dispensation information for bronchodilators (short-acting or long-acting beta-adrenergic agonists and anticholinergics), leukotriene receptor antagonists, or corticosteroids (oral or inhaled) in the 12 months prior to COVID-19 diagnosis were extracted from the KPSC electronic pharmacy records. Risks by medication use for both active and inactive asthma groups were further assessed.
Covariates
Demographics (age, gender, race/ethnicity), insurance type, body mass index, smoking history, and history of other comorbidities at the time of COVID-19 diagnosis were obtained from the KPSC electronic medical records. Using all diagnosis codes within the past 12 months prior to COVID-19 diagnosis, we calculated a modified Charlson comorbidity index10 that excluded the chronic pulmonary disease category because the comorbidities in this category overlapped with our main exposure variables. Neighborhood-level education and income information were estimated from the geocoding database, which includes annual demographic data from Nielsen.
Outcomes
Severe COVID-19 disease was evaluated using 4 end points: COVID-19–related hospitalization, intensive respiratory support (IRS), and intensive care unit (ICU) admission within 30 days, and mortality within 60 days of the COVID-19 diagnosis. COVID-19–related hospitalization, IRS, and ICU were counted only if there was an associated COVID-19 diagnosis code during the given hospital encounter. IRS was defined as the need for invasive mechanical ventilation, noninvasive ventilation, high-flow mask, or high-flow nasal cannula. Hospitalization, IRS, and ICU information was obtained from inpatient records and out-of-network claims. Mortality information was assessed from inpatient records and death records.
Statistical analyses
Logistic regression was used to estimate the odds ratio (OR) for the associations between the exposures and hospitalization, IRS, and ICU within 30 days, whereas Cox regression was used to estimate the hazard ratios (HRs) for the associations with time to death within 60 days.
For the relationship between history of asthma and COVID-19 outcomes, we first assessed risks in the active asthma and inactive asthma groups compared with the neither asthma nor COPD group. We next assessed the risks by asthma medication use by running separate regression models in which both active and inactive asthma groups were further divided into those with or without asthma medication use within the 12 months prior to COVID-19 diagnosis. All medications were combined owing to similar point estimates and small sample sizes when assessing the individual medication types separately. To evaluate the relationship between COPD and the 4 COVID-19 outcomes, only patients aged 35 years and older were included in the analysis.
All models were adjusted for the following covariates: age group (18-34, 35-64, ≥65 y), gender, race/ethnicity (White, Black, Asian, Hispanic, other), household median income (<$40,000, $40,000-$79,999, ≥$80,000), college education, Medicaid insurance status, body mass index category (<25, 25-30, 30-40, ≥40 kg/m2), smoking history (current, former, never), and the modified Charlson comorbidity score (0, 1, ≥2). Owing to changes in COVID-19 testing availability and medical management of COVID-19 patients over the study period, we also included the month of the COVID-19 diagnosis as an additional covariate in all models.
For asthma, we also assessed effect modification by age category (18-34, 35-64, ≥65 y), gender, race/ethnicity, and obesity (≥30 kg/m2) and tested for interactions between each potential effect modifier and asthma history. We estimated the exposure-outcome relationships within each of these subgroups. For the age-stratified analyses for death, the analyses were limited to those aged 35 years and older because there were few deaths in the under 35 years' age group. We did not perform subgroup and effect modification analyses for COPD history owing to the lower proportion of patients with COPD in this cohort. All analyses were performed using SAS 9.4 (Cary, NC).
Results
The study cohort consisted of 61,338 adults who had COVID-19 diagnoses codes and/or positive PCR laboratory tests, with 50,540 (82.4%) having a positive PCR laboratory test. The mean age was 43.9 years, 53.9% were females, and 66.0% had Hispanic race/ethnicity.
A total of 2,751 patients (4.5%) were in the inactive asthma group (labeled for individuals without recent clinical visits for asthma care) and 2,775 (4.5%) were in in the active asthma group (labeled for individuals with recent clinical visits for asthma care). Among the active asthma group, 87% had an outpatient visit, 36% had an inpatient/emergency department visit, and 24% had both types of clinical visits for asthma in the past year. There were 820 individuals (1.3%) with a history of COPD, among whom 307 also had asthma. Patients with asthma were more likely to be female, White, and obese/severely obese (P < .0001; Table I ). They also had higher education, income, and more comorbidities (P < .0001; Table I). Asthma medication use in the 12 months prior to COVID-19 was observed in 1,320 patients (48.0%) in the inactive asthma group and 2,286 patients (82.4%) in the active asthma group.
Table I.
Cohort characteristics of 61,338 patients with a confirmed COVID-19 diagnosis from March 1 to August 31, 2020, stratified by history of asthma and/or COPD∗,†
| Characteristic | No asthma or COPD (n = 54,992) | Inactive asthma‡ (n = 2,751) | Active asthma‡ (n = 2,775) | COPD§ (n = 820) |
|---|---|---|---|---|
| Demographics | ||||
| Age (y), mean (SD) | 43.6 (15.81) | 41.5 (17.13) | 45.6 (17.46) | 71.7 (12.14) |
| Age category (y), n (%) | ||||
| 18-34 | 18,597 (33.8) | 1,145 (41.6) | 892 (32.1) | — |
| 35-64 | 30,886 (56.2) | 1,321 (48) | 1,456 (52.5) | 217 (26.5) |
| ≥65 | 5,509 (10) | 285 (10.4) | 427 (15.4) | 603 (73.5) |
| Gender, n (%) | ||||
| Female | 29,079 (52.9) | 1,714 (62.3) | 1,880 (67.7) | 398 (48.5) |
| Male | 25,913 (47.1) | 1,037 (37.7) | 895 (32.3) | 422 (51.5) |
| Race/ethnicity, n (%) | ||||
| Asian/Pacific Island | 3,760 (6.8) | 176 (6.4) | 182 (6.6) | 49 (6) |
| Black | 3,084 (5.6) | 257 (9.3) | 295 (10.6) | 123 (15) |
| Hispanic | 36,875 (67.1) | 1,676 (60.9) | 1,673 (60.3) | 244 (29.8) |
| White | 8,399 (15.3) | 567 (20.6) | 546 (19.7) | 386 (47.1) |
| Other | 2,874 (5.2) | 75 (2.7) | 79 (2.8) | 18 (2.2) |
| Smoking status, n (%) | ||||
| Current | 2,794 (5.1) | 135 (4.9) | 115 (4.1) | 88 (10.7) |
| Former | 9,323 (17) | 499 (18.1) | 555 (20) | 529 (64.5) |
| Never | 41,535 (75.5) | 2,114 (76.8) | 2,098 (75.6) | 202 (24.6) |
| Missing | 1,340 (2.4) | 3 (0.1) | 7 (0.3) | 1 (0.1) |
| BMI category, n (%) | ||||
| Underweight/normal | 9,878 (18) | 509 (18.5) | 401 (14.5) | 237 (28.9) |
| Overweight | 17,519 (31.9) | 732 (26.6) | 695 (25) | 242 (29.5) |
| Obese | 21,292 (38.7) | 1,133 (41.2) | 1,189 (42.8) | 262 (32) |
| Severely obese | 5,033 (9.2) | 376 (13.7) | 487 (17.5) | 78 (9.5) |
| Missing | 1,270 (2.3) | 1 (0) | 3 (0.1) | 1 (0.1) |
| Household median income, n (%) | ||||
| <$40,000 | 6,230 (11.3) | 256 (9.3) | 284 (10.2) | 113 (13.8) |
| $40,000-$79,999 | 32,048 (58.3) | 1,552 (56.4) | 1,645 (59.3) | 445 (54.3) |
| ≥$80,000 | 16,710 (30.4) | 943 (34.3) | 845 (30.5) | 262 (32) |
| Missing | 4 (0) | 0 (0) | 1 (0) | 0 (0) |
| College education, n (%) | ||||
| No | 33,891 (61.6) | 1,567 (57) | 1,630 (58.7) | 474 (57.8) |
| Yes | 21,097 (38.4) | 1,184 (43) | 1,144 (41.2) | 346 (42.2) |
| Missing | 4 (0) | 0 (0) | 1 (0) | 0 (0%) |
| Medicaid, n (%) | ||||
| No | 54,824 (99.7) | 2,737 (99.5) | 2,763 (99.6) | 815 (99.4) |
| Yes | 168 (0.3) | 14 (0.5) | 12 (0.4) | 5 (0.6) |
| Charlson score‖, n (%) | ||||
| 0 | 42,624 (77.5) | 2,097 (76.2) | 1,837 (66.2) | 108 (13.2) |
| 1 | 8,411 (15.3) | 422 (15.3) | 515 (18.6) | 167 (20.4) |
| ≥2 | 3,957 (7.2) | 232 (8.4) | 423 (15.2) | 545 (66.5) |
| Asthma medication use in past 12 mo¶, n (%) | – | 1,320 (48.0) | 2,286 (82.4) | – |
| Outcomes#, n (%) | ||||
| COVID-related hospitalization | 3,404 (6.2) | 154 (5.6) | 330 (11.9) | 194 (23.7) |
| COVID-related IRS∗∗ | 1,242 (2.3) | 48 (1.7) | 118 (4.3) | 92 (11.2) |
| COVID-related ICU admission | 796 (1.4) | 29 (1.1) | 78 (2.8) | 49 (6) |
| Death | 757 (1.4) | 31 (1.1) | 65 (2.3) | 144 (17.6) |
BMI, Body mass index; ICD-10, International Classification of Diseases, Tenth Revision.
All patient characteristics (except for Medicaid) and outcome event rates were significantly different across the 4 exposure groups (P < .0001).
Patients were identified as having a history of asthma or COPD if they had at least 1 inpatient/emergency department or 2 outpatient ICD-10 codes for the given condition (asthma J45; COPD J43-J44).
Asthma patients were categorized as active asthma if they had a clinical visit for asthma in the 12 mo prior to COVID-19 diagnosis, and as inactive asthma if otherwise.
COPD history was only assessed for patients aged 35 y and older. This group also includes 307 individuals with both COPD and asthma.
Modified Charlson score is based on all diagnosis codes excluding those in the chronic pulmonary disease category.
Bronchodilators (short-acting or long-acting beta-adrenergic agonists and anticholinergics), leukotriene receptor antagonists, or corticosteroids (inhaled or oral); only assessed for asthma patients.
Within 30 d for hospitalization, IRS, and ICU; within 60 d for death.
Need for invasive mechanical ventilation, noninvasive ventilation, high-flow mask, or high-flow nasal cannula.
Following COVID-19 diagnosis, 4,082 patients (6.7%) were hospitalized, 1,500 (2.4%) received IRS, and 952 (1.6%) were admitted to the ICU owing to a COVID-related reason within 30 days, whereas 997 (1.6%) died within 60 days. Rates of hospitalization, IRS, and ICU were higher for patients in the active asthma group or with a history of COPD, but lower for those in the inactive asthma group (P < .0001; Table I).
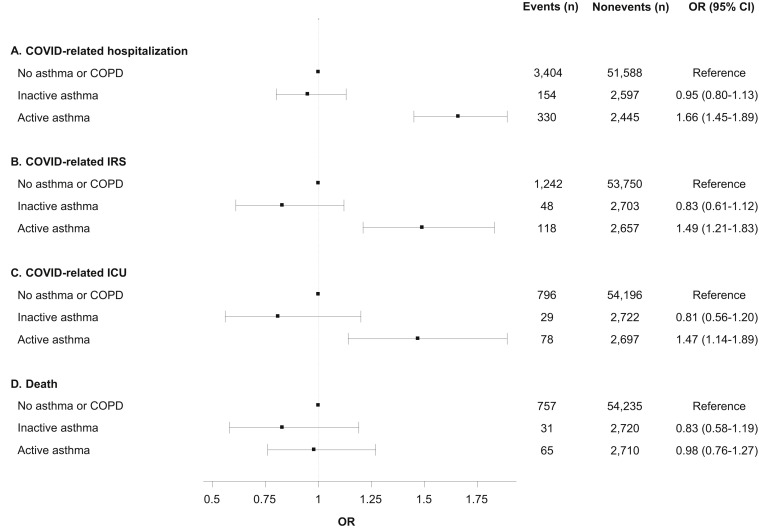
Compared with those with no history of asthma or COPD, patients in the active asthma group had a significantly increased odds of hospitalization (OR 1.66; 95% confidence interval [95% CI] 1.45-1.89), IRS (OR 1.49; 95% CI 1.21-1.83), and ICU (OR 1.47; 95% CI 1.14-1.89) within 30 days, but there was no association with mortality within 60 days (HR 0.98; 95% CI 0.76-1.27). There were no increased risks for any of the 4 outcomes for patients in the inactive asthma group (Figure 1 ).
Figure 1.
Associations between history of asthma and outcomes following COVID-19 diagnosis. All models are adjusted for age group, gender, race/ethnicity, income, college education, Medicaid insurance status, body mass index (BMI) category, smoking, and modified Charlson comorbidity score. Asthma patients were categorized as active asthma if they had a clinical visit for asthma in the 12 mo prior to COVID-19 diagnosis, and as inactive asthma if otherwise. For death, the measure of association is the hazard ratio (HR) from the Cox model.
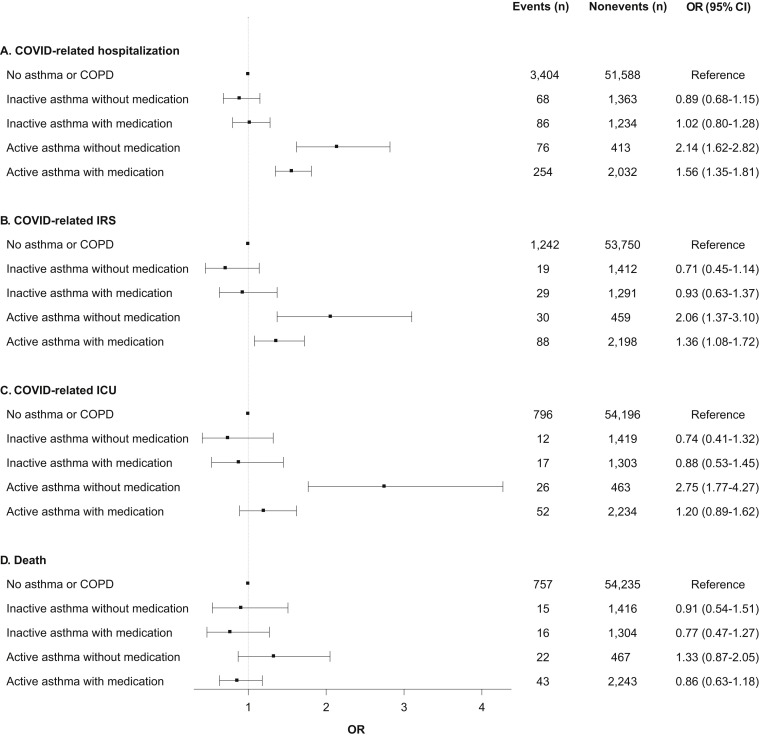
When splitting asthma patients into those with and without medication use in the past 12 months, we observed that the increased odds for hospitalization, IRS, and ICU were still present for patients in the active asthma group, but were attenuated for those who had used medications (Figure 2 ). Compared with those without a history of asthma or COPD, patients in the active asthma group who did not use medication had a 2- to 3-fold increased odds, whereas patients in this group who did use medications only had a 36% to 56% increased odds (Figure 2). Within the active asthma group, individuals with any asthma medication treatment had a 27% lower odds of hospitalization (OR 0.73; 95% CI 0.54-0.99), a 56% lower odds of ICU admission (OR 0.44; 95% CI 0.26-0.73), and nonsignificant reduced risks of ICU (OR 0.66; 95% CI 0.42-1.05) and death (HR 0.65; 95% CI 0.39-1.09) compared with those without medication. There were no significant risks for any of the COVID-19 outcomes for patients in the inactive asthma group, regardless of medication use (Figure 2).
Figure 2.
Associations between history of asthma with and without medication use and outcomes following COVID-19 diagnosis. All models are adjusted for age group, gender, race/ethnicity, income, college education, Medicaid insurance status, body mass index (BMI) category, smoking, and modified Charlson comorbidity score. Asthma patients were categorized as active asthma if they had a clinical visit for asthma in the 12 mo prior to COVID-19 diagnosis, and as inactive asthma if otherwise. For death, the measure of association is the hazard ratio (HR) from the Cox model.
Among patients aged 35 years and older, those with COPD had a 27% increased odds of hospitalization (OR 1.27; 95% CI 1.05-1.53), a 49% increased odds of IRS (OR 1.49; 95% CI 1.16-1.92), and a nonsignificant 21% increased odds of ICU admission (OR 1.21; 95% CI 0.87-1.68) within 30 days compared with those without asthma or COPD. In addition, COPD patients had a 67% increased hazard of death (HR 1.67; 95% CI 1.37-2.03) within 60 days (Table II ).
Table II.
| Outcome‡ | Exposure | Events (n) | Nonevents (n) | OR (95% CI)§ |
|---|---|---|---|---|
| COVID-related hospitalization | No asthma or COPD | 3,130 | 33,265 | Reference |
| COPD | 194 | 626 | 1.27 (1.05-1.53) | |
| COVID-related IRS | No asthma or COPD | 1,183 | 35,212 | Reference |
| COPD | 92 | 728 | 1.49 (1.16-1.92) | |
| COVID-related ICU admission | No asthma or COPD | 756 | 35,639 | Reference |
| COPD | 49 | 771 | 1.21 (0.87-1.68) | |
| Death | No asthma or COPD | 751 | 35,644 | Reference |
| COPD | 144 | 676 | 1.67 (1.37-2.03) |
This group also includes 307 individuals with both COPD and asthma.
Models were only run among individuals aged 35 y and older.
Within 30 d for hospitalization, IRS, and ICU; within 60 d for death.
For death, measure of association is the hazard ratio from the Cox model.
When examining effect modification for the association of asthma history and the 4 end points by age group, we observed stronger associations for the active asthma group among younger individuals than among older individuals. In the stratified analyses, the odds of hospitalization for active asthma were more pronounced for the younger (18-34 y: OR 2.21; 95% CI 1.53-3.20) and middle aged (35-64 y: OR 1.76; 95% CI 1.47-2.09) patients compared with the older individuals (≥65 y: OR 1.36; 95% CI 1.08-1.72; P interaction =.01). This pattern by age-group was also observed for IRS and ICU, but the interaction test was not statistically significant (Table E2; available in this article's Online Repository at www.jaci-inpractice.org). In addition, the hazard of death for the active asthma group was stronger among individuals aged 35 to 64 years (HR 1.62; 95% CI 1.05-2.48) than among those aged 65 years and older (HR 0.73; 95% CI 0.52-1.02; P interaction = .01). We did not detect significant heterogeneity for analyses stratified by gender, race/ethnicity, or obesity (data not shown).
Discussion
In this study of 61,338 COVID-19 patients identified from a diverse patient population in Southern California from March to August 2020, we found that patients with a history of asthma and clinical visits for asthma in the 12 months prior to COVID-19 diagnosis, as well as those with a history of COPD, were at higher risk of COVID-19–related hospitalization, IRS, and ICU admission. Chronic obstructive pulmonary disease, but not asthma, was associated with a higher risk of COVID-19 mortality. Patients with a history of asthma, but without clinical visits for asthma care in the 12 months prior to COVID-19 diagnosis, did not have an increased risk of COVID-19 severity or mortality. Among asthma patients who had clinical visits for asthma in the past 12 months, those who had received medication treatment had a lower risk of severe COVID-19 than individuals who were not treated by asthma medication. Furthermore, the associations with COVID-19 severity and mortality for those with clinical visits for asthma in the 12 months prior to COVID-19 diagnosis appeared to be stronger among individuals younger than 65 years.
Chronic respiratory diseases (primarily asthma and COPD), along with other comorbidities, have been suggested as risk factors of severe COVID-19.1 The preexisting perturbations of lung function, airway hypersensitivity, and immune alterations in asthma and COPD patients may cause patients to develop more severe outcomes with virus infections, including other coronaviruses such as Middle East respiratory syndrome–related coronavirus.11 Previous studies examining chronic respiratory diseases and COVID-19 severity have been inconsistent. Based on data from China and the United States during the early pandemic period from February to March 2020, the prevalence of asthma and COPD among hospitalized COVID-19 patients was much lower than the prevalence of asthma and COPD in the general population.12 However, more recent data from the United Kingdom and the United States have suggested that asthma and COPD are overrepresented in severe COVID-19 cases.13 , 14 The findings from this study support the prior evidence of an association between COPD history and COVID-19 severity and mortality. However, our study showed that the risk associated with asthma history is not uniform.
Although previous meta-analyses have suggested that there is no significant association between asthma and COVID-19 severity, there has been considerable variation in the associations observed across individual studies.15, 16, 17 This could be attributable to the fact that the association between asthma and COVID-19 severity may be influenced by disease status and medication treatment—factors that have not been thoroughly evaluated in earlier studies. Asthma is a chronic condition with large heterogeneity in symptom control and severity across individuals. Patients with uncontrolled asthma could be more vulnerable to virus infection and hyperinflammation than those with inactive asthma. Because we did not have data to specifically investigate asthma severity as defined by the Global Initiative for Asthma18 or the European Respiratory Society/American Thoracic Society Task Force,19 we focused our analysis on whether asthma patients received recent clinical care or used medications (eg, bronchodilators, leukotriene receptor antagonists, or corticosteroids). Our study showed that only asthma patients with clinical visits for asthma care during the 12 months before the COVID-19 diagnosis had a significantly increased risk of severe COVID-19. Moreover, patients in this active asthma group who did not use asthma medications had a higher risk of poor outcomes than those who did use medications. This suggests that recent clinical care and medication for asthma play important roles in the relationship between asthma and severe outcomes from COVID-19. Future studies with detailed histories of asthma symptoms and medication types are needed to further clarify the impact of asthma severity on COVID-19 outcomes.
We observed that medication use attenuated the risk of severe COVID-19 for asthma patients with recent clinical visits by a factor ranging from 27% for hospitalization to 56% for ICU admission. Conversely, asthma patients without recent clinical visits did not have an increased risk of COVID-19 severity and mortality, regardless of medication treatment status. Although many studies have been conducted to investigate the role of inhaled corticosteroids and other asthma medications in COVID-19 severity and mortality, the findings thus far have been conflicting. Some studies found no association or inverse associations,6 , 20, 21, 22 whereas another study of over 818,000 asthma patients suggested that using inhaled corticosteroids within 4 months before COVID-19 diagnosis was associated with an elevated risk of COVID-19 mortality.3 The results from our study indicate that asthma patients, particularly those who require clinical care and do not have proper medication treatment, are at an even greater risk of poor outcomes from COVID-19. These findings suggest that controlling asthma symptoms through medication treatment could be beneficial in reducing the risk of severe COVID-19 illness.
In this study, we also found that associations of asthma history and COVID-19–related hospitalization, IRS, and ICU admission were more pronounced among people younger than 65 years. The smaller effect size for COVID-19 severity observed in individuals 65 years old or older may be due to stronger associations from other comorbidities, such as hypertension and diabetes.
Our study was conducted among a large and unique COVID-19 patient cohort in Southern California, which comprises a diverse population of various racial/ethnic and socioeconomic groups. The detailed medical history of clinical visits, comorbidities, and medication use from the electronic health records allowed us to comprehensively investigate the associations of asthma disease status, COPD, and relevant medication treatment history with COVID-19 severity and mortality, while adjusting for important confounders. However, we acknowledge several limitations of our study. The study population was restricted to confirmed COVID-19 patients, which differs from past studies using random samples from the general population. Thus, our findings focus on the associations of asthma and COPD with disease severity and mortality among COVID-19 patients rather than the susceptibility to SARS-CoV-2 infection. Furthermore, the dose of medication treatment was not evaluated, which might have a small influence on the relationship between medication treatment history and severe COVID-19 among asthma patients. We also could not assess medication adherence with our pharmacy dispensation records. However, the finding of an attenuated increased risk among asthma patients with recent clinical visits who had used medications suggests that patients who require clinical care but are not treated by control medications may have a unique risk for severe COVID-19. Lastly, the health care utilization patterns among asthma patients may have been affected because our study period overlapped with the early phase of the pandemic. Thus, we defined recent clinical visits using a window of an entire year prior to the COVID-19 diagnosis in order to capture several months of prepandemic health care utilization. We also included a time variable as a covariate to minimize any potential bias. Our study design using the unanimous KPSC membership as the source population is likely the best approach to minimize bias in large population-based studies because all KPSC members have equal access to health care, testing, and treatment options with uniform guidelines across all medical centers.
In conclusion, the findings from this study indicate that, among asthma patients, only those with recent clinical visits for asthma care are at higher risk of severe COVID-19. Individuals with COPD are also susceptible to COVID-19 severity and mortality. Medication treatment for asthma can perhaps help to reduce the elevated risk for asthma patients.
Footnotes
This study was supported by the National Institute of Environmental Health Sciences (3R01ES029963-01 to A. H. Xiang and Z. Chen) and the National Cancer Institute (T32CA229110 and K99CA256525 to B. Z. Huang) at the National Institutes of Health, and the Keck School of Medicine Department of Preventive Medicine COVID-19 Pandemic Research Center (CPRC) at USC. The funding agencies had no role in the design or conduct of the study; in the analysis or interpretation of the data; or in the preparation, review, or approval of the manuscript.
Conflicts of interest: The authors declare that they have no relevant conflicts of interest.
Online Repository
Table E1.
ICD-10 codes used to identify COVID-19 cases∗
| ICD-10 code | Code description |
|---|---|
| U07.1 | COVID-19 disease |
| U07.1 | Asymptomatic COVID-19 disease |
| J80, U07.1 | COVID-19 acute respiratory distress syndrome |
| J12.89, U07.1 | COVID-19 pneumonia |
| J20.8, U07.1 | COVID-19 acute bronchitis |
| J22, U07.1 | COVID-19 lower respiratory infection |
ICD-10, International Classification of Diseases, Tenth Revision.
Other internal KPSC codes were also used to identify cases.
Table E2.
Associations between history of asthma∗ and outcomes following COVID-19 diagnosis, stratified by age group
| Outcome† | Subgroup | Exposure | Events (n) | Nonevents (n) | OR (95% CI)‡ |
|---|---|---|---|---|---|
| COVID-related hospitalization | 18-34 y | No asthma or COPD | 274 | 18,323 | Reference |
| Inactive asthma | 14 | 1,131 | 0.87 (0.50-1.51) | ||
| Active asthma | 40 | 852 | 2.21 (1.53-3.20) | ||
| 35-64 y | No asthma or COPD | 2,012 | 28,874 | Reference | |
| Inactive asthma | 93 | 1,228 | 1.11 (0.88-1.39) | ||
| Active asthma | 175 | 1,281 | 1.76 (1.47-2.09) | ||
| ≥65 y | No asthma or COPD | 1,118 | 4,391 | Reference | |
| Inactive asthma | 47 | 238 | 0.77 (0.55-1.07) | ||
| Active asthma | 115 | 312 | 1.36 (1.08-1.72) | ||
| P interaction§ | .01 | ||||
| COVID-related IRS | 18-34 y | No asthma or COPD | 59 | 18,538 | Reference |
| Inactive asthma | 3 | 1,142 | 0.92 (0.28-3.01) | ||
| Active asthma | 9 | 883 | 1.74 (0.80-3.77) | ||
| 35-64 y | No Asthma or COPD | 721 | 30165 | Reference | |
| Inactive asthma | 29 | 1292 | 0.97 (0.66-1.43) | ||
| Active asthma | 62 | 1394 | 1.60 (1.21-2.13) | ||
| ≥65 y | No Asthma or COPD | 462 | 5047 | Reference | |
| Inactive asthma | 16 | 269 | 0.66 (0.39-1.11) | ||
| Active asthma | 47 | 380 | 1.27 (0.91-1.76) | ||
| P interaction§ | .41 | ||||
| COVID-related ICU admission | 18-34 y | No asthma or COPD | 40 | 18,557 | Reference |
| Inactive asthma | 2 | 1,143 | 0.99 (0.23-4.17) | ||
| Active asthma | 7 | 885 | 1.86 (0.76-4.55) | ||
| 35-64 y | No asthma or COPD | 465 | 30,421 | Reference | |
| Inactive asthma | 15 | 1,306 | 0.78 (0.46-1.33) | ||
| Active asthma | 41 | 1,415 | 1.52 (1.07-2.15) | ||
| ≥65 y | No asthma or COPD | 291 | 5,218 | Reference | |
| Inactive asthma | 12 | 273 | 0.87 (0.47-1.59) | ||
| Active asthma | 30 | 397 | 1.29 (0.86-1.94) | ||
| P interaction§ | .62 | ||||
| Death‖ | 35-64 y | No asthma or COPD | 235 | 30,651 | Reference |
| Inactive asthma | 8 | 1,313 | 0.81 (0.40-1.64) | ||
| Active asthma | 25 | 1,431 | 1.62 (1.05-2.48) | ||
| ≥65 y | No asthma or COPD | 516 | 4,993 | Reference | |
| Inactive asthma | 23 | 262 | 0.81 (0.53-1.24) | ||
| Active asthma | 36 | 391 | 0.73 (0.52-1.02) | ||
| P interaction§ | .01 |
Asthma patients were categorized as active asthma if they had a clinical visit for asthma in the 12 mo prior to COVID-19 diagnosis, and as inactive asthma if otherwise.
Within 30 d for hospitalization, IRS, and ICU; within 60 d for death.
For death, measure of association is the hazard ratio from the Cox model.
Heterogeneity assessed using models with an interaction term for age group and asthma history.
Death models only run among those aged 35 y and older owing to the small number of deaths in the 18-34 y age group.
References
- 1.Centers for Disease Control and Prevention (CDC). People with Certain Medical Conditions. Available from: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html. Accessed November 16, 2020.
- 2.Wang L., Foer D., Bates D.W., Boyce J.A., Zhou L. Risk factors for hospitalization, intensive care, and mortality among patients with asthma and COVID-19. J Allergy Clin Immunol. 2020;146:808–812. doi: 10.1016/j.jaci.2020.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schultze A., Walker A.J., MacKenna B., Morton C.E., Bhaskaran K., Brown J.P. Risk of COVID-19-related death among patients with chronic obstructive pulmonary disease or asthma prescribed inhaled corticosteroids: an observational cohort study using the OpenSAFELY platform. Lancet Respir Med. 2020;8:1106–1120. doi: 10.1016/S2213-2600(20)30415-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lovinsky-Desir S., Deshpande D.R., De A., Murray L., Stingone J.A., Chan A. Asthma among hospitalized patients with COVID-19 and related outcomes. J Allergy Clin Immunol. 2020;146 doi: 10.1016/j.jaci.2020.07.026. 1027-34.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Doriane C., Sophie G., Nathalie M., Frix A.N., Thys M., Bonhomme O. Asthma and COPD are not risk factors for ICU stay and death in case of SARS-CoV-2 infection. J Allergy Clin Immunol Pract. 2020;9:160–169. doi: 10.1016/j.jaip.2020.09.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Choi Y.J., Park J.Y., Lee H.S., Suh J., Song J.Y., Byun M.K. Effect of asthma and asthma medication on the prognosis of patients with COVID-19. Eur Respir J. 2021;57:2002226. doi: 10.1183/13993003.02226-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Soeroto AY, Purwiga A, Pranggono EH, Roesli RMA. Asthma does not increase COVID-19 mortality and poor outcomes: a systematic review and meta-analysis. Asian Pac J Allergy Immunol. Published online April 18, 2021. 10.12932/ap-110920-0955 [DOI] [PubMed]
- 8.Kaye L., Theye B., Smeenk I., Gondalia R., Barrett M.A., Stempel D.A. Changes in medication adherence among patients with asthma and COPD during the COVID-19 pandemic. J Allergy Clin Immunol Pract. 2020;8:2384–2385. doi: 10.1016/j.jaip.2020.04.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhu Z., Hasegawa K., Ma B., Fujiogi M., Camargo C.A., Jr., Liang L. Association of asthma and its genetic predisposition with the risk of severe COVID-19. J Allergy Clin Immunol. 2020;146 doi: 10.1016/j.jaci.2020.06.001. 327-9.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Quan H., Sundararajan V., Halfon P., Fong A., Burnand B., Luthi J.C. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43:1130–1139. doi: 10.1097/01.mlr.0000182534.19832.83. [DOI] [PubMed] [Google Scholar]
- 11.Alqahtani F.Y., Aleanizy F.S., Mohamed R.A.E.H., Alanazi M.S., Mohamed N., Alrasheed M.M. Prevalence of comorbidities in cases of Middle East respiratory syndrome coronavirus: a retrospective study. Epidemiol Infect. 2018;147:1–5. doi: 10.1017/S0950268818002923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.CDC Covid–Response Team Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019—United States, February 12–March 28, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:382–386. doi: 10.15585/mmwr.mm6913e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Docherty A.B., Harrison E.M., Green C.A., Hardwick H.E., Pius R., Norman L. Features of 20,133 UK patients in hospital with Covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369:m1985. doi: 10.1136/bmj.m1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Garg S., Kim L., Whitaker M., O'Halloran A., Cummings C., Holstein R. Hospitalization rates and characteristics of patients hospitalized with laboratory-confirmed coronavirus disease 2019—COVID-NET, 14 States, March 1-30, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:458–464. doi: 10.15585/mmwr.mm6915e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu S., Cao Y., Du T., Zhi Y. Prevalence of comorbid asthma and related outcomes in COVID-19: a systematic review and meta-analysis. J Allergy Clin Immunol Pract. 2021;9:693–701. doi: 10.1016/j.jaip.2020.11.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Terry P.D., Heidel R.E., Dhand R. Asthma in adult patients with COVID-19. Prevalence and risk of severe disease. Am J Respir Crit Care Med. 2021;203:893–905. doi: 10.1164/rccm.202008-3266OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang Y., Ao G., Qi X., Xie B. The association between COVID-19 and asthma: a systematic review and meta-analysis. Clin Exp Allergy. 2020;50:1274–1277. doi: 10.1111/cea.13733. [DOI] [PubMed] [Google Scholar]
- 18.Global Initiative for Asthma . 2020. Global Strategy for Asthma Management and Prevention.www.ginasthma.org Available from: [Google Scholar]
- 19.Chung K.F., Wenzel S.E., Brozek J.L., Bush A., Castro M., Sterk P.J. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43:343–373. doi: 10.1183/09031936.00202013. [DOI] [PubMed] [Google Scholar]
- 20.Coutinho A.E., Chapman K.E. The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol Cell Endocrinol. 2011;335:2–13. doi: 10.1016/j.mce.2010.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Peters M.C., Sajuthi S., Deford P., Christenson S., Rios C.L., Montgomery M.T. COVID-19-related genes in sputum cells in asthma. Relationship to demographic features and corticosteroids. Am J Respir Crit Care Med. 2020;202:83–90. doi: 10.1164/rccm.202003-0821OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bloom C.I., Drake T.M., Docherty A.B., Lipworth B.J., Johnston S.L., Nguyen-Van-Tam J.S. Risk of adverse outcomes in patients with underlying respiratory conditions admitted to hospital with COVID-19: a national, multicentre prospective cohort study using the ISARIC WHO Clinical Characterisation Protocol UK. Lancet Respir Med. 2021;9:699–711. doi: 10.1016/S2213-2600(21)00013-8. [DOI] [PMC free article] [PubMed] [Google Scholar]