Abstract
Calcified amorphous tumor (CAT) is a non-neoplastic tumor composed of calcified nodules consisting of amorphous fibrous material, and it may eventually cause cerebral infarction (CI). We experienced a 67-year-old woman with CAT who had recurrent CI. After excision of the CAT, the CI did not show recurrence. A review of previous papers on CI due to CAT in Pubmed revealed that 7 of 13 studies originated in Japan and that CI can occur even with small CAT. Surgical treatment is recommended to prevent CI recurrence, especially when CAT is accompanied by mitral annular calcification or has marked mobility.
Keywords: calcified amorphous tumor, cerebral infarction, mitral annular calcification, embolization
Introduction
Calcified amorphous tumor (CAT) is a non-neoplastic tumor composed of calcified nodules composed of amorphous fibrous material, and it may cause cerebral embolism. It was originally described in 1997 (1), and subsequently, several cases of CAT have been reported, but the clinical characteristics of cerebral infarction (CI) due to CAT remain unclear.
We herein report a patient with CAT who had recurrent CI in the posterior circulation territory and review the clinical features of previous cases in which CI was caused by CAT.
Case Report
A 67-year-old woman with a medical history of hypertension and hyperlipidemia and a 40-pack-year smoking history developed visual field impairment and was admitted to our hospital.
No neurological deficits were observed, with the exception of right hemianopsia and color blindness. Her fundoscopic findings were normal. Laboratory findings showed a white blood cell count of 8,700/mm3 with 5.7% eosinophils (calculated eosinophil count of 496/mm3), D-dimer of 1.85 μg/mL, serum calcium of 8.7 mg/dL, serum phosphorus of 4.2 mg/dL, and creatinine of 1.45 mg/dL.
An initial electrocardiogram showed a normal sinus rhythm. Magnetic resonance imaging (MRI) showed CI in the bilateral occipital lobes and cerebellum, and magnetic resonance angiography (MRA) showed a decreased blood flow in the left posterior cerebral artery (PCA). Contrast-enhanced computed tomography (CT) showed a high-density nodule in the mitral valve (MV) and left vertebral artery stenosis at the 5-6 cervical spine level. A transthoracic echocardiography examination (TTE) showed no intra-cardiac shunt and a normal left ventricular ejection function, but severe aortic valve (AV) stenosis and mitral annular calcification (MAC) were suggested. The mobility of the MAC appeared to be poor (Fig. 1A-E).
Figure 1.
Radiological and ultrasonography findings. (A) Diffusion weighted image showed multiple cerebral infarctions in bilateral occipital lobes and cerebellum. (B) Magnetic resonance angiography showed a decreased blood flow in the left posterior cerebral artery (arrow), and the vertebral artery was right-dominant. (C) Contrast-enhanced computed tomography showed a high-density nodule in the mitral valve. (D) Three-dimensional computed tomography angiography showed left vertebral artery stenosis at the C5/6 level (arrow). (E) A transthoracic echocardiography examination revealed mitral annular calcification (arrow). (F) Transesophageal echocardiography showed swinging calcified amorphous tumor (arrow).
Her symptoms were partially improved after medical treatment with anticoagulant agent and the free-radical scavenging agent Edaravone. On the 34th hospital day, she was transferred to another hospital for rehabilitation. Approximately five months later, she developed visual impairment again and visited our hospital. MRI showed a new CI in the left occipital lobe. A TTE showed no change in calcification at the base of the MV but did reveal the possibility of pedunculated CAT attached to the posterior leaflet of the MV, which usually indicates increased mobility. Transesophageal echocardiography (TEE) showed a tumor 14×9.2 mm in size and no other new lesions (Fig. 1F).
The patient was transferred to cardiovascular surgery and underwent MV foreign body removal and AV replacement. The combination of warfarin and aspirin was then started. The pathological findings revealed a calcified nodule with eosinophilic amorphous fibrinous material, which was diagnosed as CAT (Fig. 2). There was no recurrence of CI for over a year after the operation, indicating that the recurrence of the CIs had been due to MAC-related CAT.
Figure 2.

Pathological findings. (A) The gross anatomic pathology of the excised tumor is yellow-white in color and partially calcified. (B) A histological examination revealed multiple nodular calcifications surrounded by fibrous connective tissue and fibrin. There was no identifiable myxoma or inflammatory cell infiltration (Hematoxylin and Eosin staining; bar=500 μm).
Discussion
First described by Reynolds et al. in 1997, cardiac CAT is a non-neoplastic primary tumor of the heart (1). Primary cardiac tumors are rare entities, occurring with a lifetime incidence of 0.0017-0.02%, and CAT is estimated to account for 2.48% of primary cardiac tumors (2,3). It is characterized histologically by calcified nodules and amorphous fibrinous material and is usually discovered by cardiac imaging studies, although it may also be found following an instance of CI, as in this case. The majority of CAT cases have been identified in association with valvular heart disease (31%), end-stage renal disease (ESRD) (21%), MAC (14%), and diabetes mellitus (DM) (14%) (4). Various etiologies may induce CAT, including organized calcific mural thrombus, abnormality of coagulation system, abnormalities of calcium and phosphate metabolism, and chronic inflammation.
Since two episodes of CI in the present case involved the posterior circulation, the possibility of CI due to left vertebral artery stenosis was initially suspected. However, the emergence of pedunculated and mobile CAT over the course of time suggested that CAT was a more likely embolic source, and indeed, no more CI recurrence was observed postoperatively. In cases of CI due to CAT, recurrence of CI can be radically prevented by excision, and other issues, such as syncope and heart failure, may also be prevented by surgical treatment.
We reviewed all previous cases of CAT-induced CI and found 17 reported cases (1,5-19) (Table). First, we performed a PubMed search of all available literature from January 1, 1997, to July 31, 2020, using the terms “calcified amorphous tumor.” A total of 68 articles were found. We then selected all cases that mentioned “cerebral infarction (CI),” “cerebral embolism,” “stroke” or “cerebrovascular accident (CVA).” The cases cited in those articles were also included. Cases with only retinal artery occlusion or cerebral hemorrhaging without CI were excluded. Finally, 13 cases of CI due to CAT were found (Case No. 1-13). Seven of these 13 cases (No. 3, 7-13) were reported from Japan, so we conducted an additional search in J-STAGE, an electronic journal in Japan, and found 4 more case reports (No. 14-17). Racial differences in incidence may explain the large number of reports from Japan. Other reasons for the seemingly high prevalence in Japan may be the high dialysis induction rate in patients with ESRD and the accuracy of CI imaging diagnoses due to the prevalence of MRI examinations.
Table.
Clinical Characteristics of Published Cases of CAT Induced CI.
| Case No. | Age, Sex | CAT site | Size(mm) | MAC | Symptoms | CI site | Comorbidities/underlying disease | Treatment | Reference No. |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 48,F | MV | 30×2 | n.d. | n.d. | n.d. | MR,TR,cleft MV leaflet | Surgical | 1 |
| 2 | 60,F | LV | 15×15×15 | n.d. | retinal emboli | n.d. | MR,AR | Surgical | 1 |
| 3 | 66,F | MV | 14.7×13.7 | n.d. | right hemiplegia | n.d. | HD(renal tuberculosis) | Surgical | 5 |
| 4 | 69,F | LV | 20 | p | dyspnea (heart failure) | left hemisphere | HT,DM,OMI,COPD,BA | Surgical | 6 |
| 5 | 70,F | LA | n.d | n | dysarthria, right hemiparesis |
pons,bilateral hemisphere | DM,HT | Surgical | 7 |
| 6 | 67,M | MV | 9×7 | p | transient diplopia, facial dysesthaesia |
multiple disseminated punctate lesions | HT,HL | Surgical | 8 |
| 7 | 70,M | MV | 14×11 | p (mobile MAC-related CAT) | exotropia | right pons | HD(nephrosclerosis), HT | Aspirin | 9 |
| 8 | 68,F | MV | 12×12 | p | chest pain(AMI), blurred vision | left occipital lobe | 40-pack-year smoking | Surgical | 10 |
| 9 | 89,F | MV | 17.5×9.7 | p | decreased activity | multiple small infarctions (left SCA,PCA,MCA) | Af,HT,CHF,dementia | Warfarin | 11 |
| 10 | 38,F | MV | n.d | p | right hemiparesis, motor aphasia |
left upper branch of the MCA | HD(Lupus nephritis) | Surgical | 12 |
| 11 | 75,M | AV | 22×11 | n.d. | dysarthria | small multiple bilateral cerebral infarctions | HD(diabetic nephropathy) | Surgical | 13 |
| 12 | 59,M | MV | 10×16.9 | n.d. | speech disturbance, right arm weakness | left frontal and parietal lobes | HD | Surgical | 14 |
| 13 | 67,M | LV | 29×18 | n.d. | fatigue, stomatitis, diarrhea |
several small subacute infarctions | HD,IE,CHF,HT, colon and gastric cancer | Surgical | 15 |
| 14 | 47,M | MV | 20×7,2-3 | p | left hemiparesis | right MCA territory | HD(CKD), HT,HL,DM,SAS | Anti-coagrant | 16 |
| 15 | 64,M | MV | 6×15 | n | visual loss (retinal artery occlusion) | bilateral hemisphere and right thalamus | nothing | Surgical | 17 |
| 16 | 47,M | MV | 11×5 | n.d. | diplopia, balance disorder |
pons,bilateral hemisphere | HD,HT,DM | Surgical | 18 |
| 17 | 62,M | AV | 8×5 | n | left hemiparesis, dysarthria |
right parietal lobe | HD(diabetic nephropathy), DM,HT | Surgical | 19 |
| 18 | 67,F | MV | 14×9.2 | p | visual field defect, achromatopsia |
bilateral PCA and SCA territory | HT,HL,severe AS, left VA stenosis | Surgical | our case |
CAT: calcified amorphous tumor, CI: cerebral infarction, MAC: mitral annular calcification, MV: mitral valve, LV: left ventricular, LA: left atrium, AV: aortic valve, n.d.: no data, p: positive, n: negative, AMI: acute myocardial infarction, SCA: superior cerebellar artery, PCA: posterior cerebral artery, MCA: middle cerebral artery, MR: mitral regurgitation, TR: tricuspid regurgitation, AR: aortic regurgitation, HD: hemodialysis, HT: hypertension, DM: diabetic mellitus, OMI: old myocardial infarction, COPD: chronic obstructive pulmonary disease, BA: bronchial asthma, HL: hyperlipidemia, CHF: chronic heart failure, CKD: chronic kidney disease, AS: aortic stenosis, SAS: sleep apnea syndrome, VA: vertebral artery
Although there were more women than men in all previous reports on CAT (1,4,11,19), there was no marked difference between sexes in the CAT-induced CI cases (nine men and nine women). This discrepancy in findings may be due to gender differences in incidence and the small number of cases with CI morbidity, but the truth of the matter is unclear.
When the 18 total cases were divided according to the CAT site, MV was the most common in 12 cases (66.7%), left ventricle (LV) in 3 cases (16.7%), AV in 2 cases, and left atrium (LA) in 1 case. According to the paper by Singu et al., the MV was the site in 14 cases (33%), LA in 4 cases (9%), LV in 10 cases (23%), tricuspid valve in 2 cases (5%), right atrium in 6 cases (14%), and right ventricle in 7 cases (16%) (11), indicating that the MV is the most common site, regardless of the presence or absence of CI.
Benjamin et al. found that MAC was an independent risk factor for CI in the Framingham study because cases with MAC (n=160) had a higher rate of cerebrovascular accidents at subsequent follow-up than those without MAC (n=999) (20). Nakamaru et al. noted that MAC-related CAT was likely to occur due to ESRD, had high mobility, and might have a high embolic risk (21). Although our case lacked renal failure or dialysis, the recurrence of CI in association with highly mobile MAC-related CAT was compatible with this hypothesis.
Of the 18 cases described in Table, there were at least 8 (44.4%) with MAC, and dialysis was seen in 9 cases (50.0%). These rates were more frequent than those reported by de Hemptinne et al. (14% with MAC, 21% with ESRD) (4).
Of the 42 cases of CAT summarized by de Hemptinne et al., the maximum length of the CAT was 29.7 mm on average in 35 cases without CI (4), whereas the maximum length of CAT that caused CI was 16.8 mm on average, which was significantly smaller than the former value (p=0.004) (Fig. 3). The reason for the small size of embolic CAT is unclear but may be due to the detection of post-embolic remnant of CAT, or alternatively, a reduced detection rate of asymptomatic CAT. Alternatively, the risk of embolism may be independent of size and determined by qualitative factors, such as the degree of fibrin cap, as described below. However, physicians should not hesitate to perform surgical treatment even if the lesion is larger than average, and various aspects of clinical data should be considered to determine the course of treatment.
Figure 3.
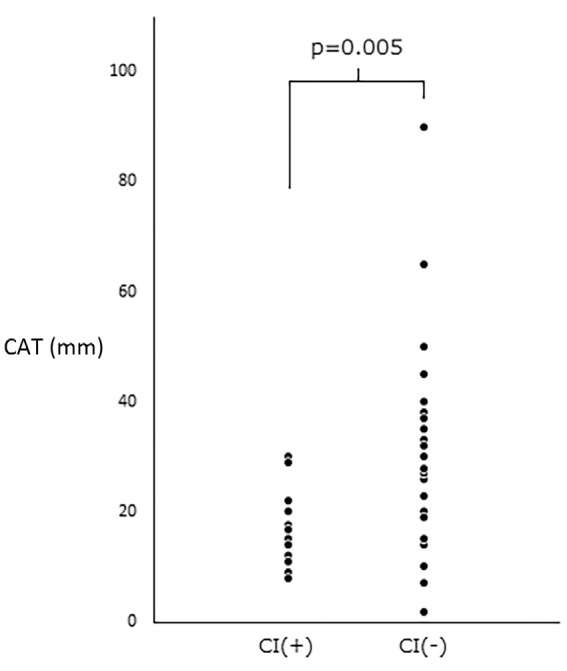
The maximum length of the CAT (mm). The CAT that caused CI was significantly shorter than the CAT without CI. CAT: calcified amorphous tumor, CI: cerebral infarction
Regarding the mechanism underlying the embolism, CAT itself may be an embolization source, or alternatively, the fibrin cap attached to CAT may be an embolization source. However, their differentiation is not easy (6). In the report by Nagao et al., intravascular thrombus with calcification was found on brain CT (14) and suggested embolization of CAT per se. In addition, in the report of Johno et al., the embolization of CAT has also been suggested by the stumped tip of the excised CAT specimen. In our case, there was no intracranial thrombus with calcification, and the tip of the excised specimen was not stumped, suggesting embolization of the fibrin cap (18).
Interestingly, there have been only a few cases of systemic circulation embolization. There were only three cases of myocardial infarction, one case of common iliac artery occlusion, and three cases of retinal artery occlusion, aside from the CI cases shown in Table.
One hypothesis is that the fibrin cap embolization is soft and small, so symptoms are unlikely to occur in areas other than those that are prone to focal signs, such as the cerebrovascular and retinal arteries. It is likely that there were many asymptomatic emboli in the systemic circulation. In addition, three cases of CI have shown relatively mild symptoms, as represented by a low NIHSS score (No. 7; 2 points, No. 16; 2 points, our case; 4 points), three cases have shown no symptoms (No. 9, 13, 15), and four cases have shown the improvement of neurological symptoms by natural remission or conservative treatment (No. 4, 5, 6, 17).
Conclusion
CAT is a non-neoplastic tumor composed of calcified nodules on a background of amorphous fibrous material, and it can cause CI. We have summarized cases of CAT-induced CI. Similar to previous reports, CAT was most commonly found in the MV and tended to be accompanied by MAC, but it was often smaller than in cases without CI. A fibrin cap was assumed to be the cause of embolism in the present case, but since the pathological condition is often unknown, the accumulation of further cases is desired.
The authors state that they have no Conflict of Interest (COI).
References
- 1.Reynolds C, Tazelaar HD, Edwards WD. Calcified amorphous tumor of the heart (cardiac CAT). Hum Pathol 28: 601-606, 1997. [DOI] [PubMed] [Google Scholar]
- 2.Elbardissi AW, Dearani JA, Daly RC, et al. Survival after resection of primary cardiac tumors: a 48-year experience. Circulation 118(Suppl): S7-S15, 2008. [DOI] [PubMed] [Google Scholar]
- 3.Watanabe Y, Naganuma T, Nakao T, Nakamura S. A calcified amorphous tumor originating in the sinus of valsalva. Echocardiography 33: 796-798, 2016. [DOI] [PubMed] [Google Scholar]
- 4.de Hemptinne Q, de Cannière D, Vandenbossche JL, Unger P. Cardiac calcified amorphous tumor: a systematic review of the literature. Int J Cardiol Heart Vasc 7: 1-5, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tsuchihashi K, Nozawa A, Marusaki S, et al. Mobile intracardiac calcinosis: a new risk of thromboembolism in patients with haemodialysed end stage renal disease. Heart 82: 638-640, 1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Greaney L, Chaubey S, Pomplun S, St Joseph E, Monaghan M, Wendler O. Calcified amorphous tumour of the heart: presentation of a rare case operated using minimal access cardiac surgery. BMJ Case Rep 2011: bcr0220113882, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Suh JH, Kwon JB, Park K, Park CB. Calcified amorphous tumor in left atrium presenting with cerebral infarction. J Thorac Dis 6: 1311-1314, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.de Hemptinne Q, Bar JP, deCannière D, Unger P. Swinging cardiac calcified amorphous tumour arising from a calcified mitral annulus in a patient with normal renal function. BMJ Case Rep 2015: bcr2014207401, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Saito K, Doi M, Karikusa M, Sakata K, Sasaguri H, Toru S. Cerebral infarction in right pons during the course of mobile mitral annular calcification-related calcified amorphous tumor during a long time hemodialysis. Rinsho Shinkeigaku (Clin Neurol) 56: 580-583, 2016. (in Japanese, Abstract in English). [DOI] [PubMed] [Google Scholar]
- 10.Kyaw K, Latt H, Aung SSM, Roongsritong C. A case of cardiac calcified amorphous tumor presenting with concomitant ST-elevation myocardial infarction and occipital stroke and a brief review of the literature. Case Rep Cardiol 2017: 8578031, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Singu T, Inatomi Y, Yonehara T, Ando Y. Calcified amorphous tumor causing shower embolism to the brain: a case report with serial echocardiographic and neuroradiologic images and a review of the literature. J Stroke Cerebrovasc Dis 26: e85-e89, 2017. [DOI] [PubMed] [Google Scholar]
- 12.Aizawa Y, Nakai T, Saito Y, et al. Calcified amorphous tumor-induced acute cerebral infarction. Int Heart J 59: 240-242, 2018. [DOI] [PubMed] [Google Scholar]
- 13.Kasai M, Osako M, Tanino T, Maeshima A. A calcified amorphous tumor originating in the aortic valve cusp. Ann Thorac Surg 105: e235-e237, 2018. [DOI] [PubMed] [Google Scholar]
- 14.Nagao Y, Nakajima M, Hirahara T, et al. Calcified cerebral embolism due to a calcified amorphous tumor. J Stroke Cerebrovasc Dis 27: e115-e116, 2018. [DOI] [PubMed] [Google Scholar]
- 15.Okazaki A, Oyama Y, Hosokawa N, Ban H, Miyaji Y, Moody S. The first report of calcified amorphous tumor associated with infective endocarditis: a case report and review of literature. Am J Case Rep 21: e922960, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kimura N, Haruta H, Tamaki T, et al. A case of calcified amorphous tumor found with cerebral infarction. Sinzo (Heart) 49: 502-507, 2017. (in Japanese). [Google Scholar]
- 17.Araga T, Hagiwara Y, Yanagisawa T, et al. A case of multiple cerebral embolism caused by calcified amorphous tumor incidentally found by central retinal artery occlusion: a case report. Neurosonology 32: 19-22, 2019. (in Japanese, Abstract in English). [Google Scholar]
- 18.Johno T, Kawano H, Hirano T. A case of calcified amorphous tumor with multiple cerebral embolism. Neurosonology 32: 43-45, 2019. (in Japanese). [Google Scholar]
- 19.Takahashi Y, Niino T. Cardiac calcified amorphous tumor presenting as cerebral infarction. Jpn J Cardiovasc Surg 49: 16-20, 2020. (in Japanese, Abstract in English). [Google Scholar]
- 20.Benjamin EJ, Plehn JF, D'Agostino RB, et al. Mitral annular calcification and the risk of stroke in an elderly cohort. N Engl J Med 327: 374-379, 1992. [DOI] [PubMed] [Google Scholar]
- 21.Nakamaru R, Oe H, Iwakura K, Masai T, Fujii K. Calcified amorphous tumor of the heart with mitral annular calcification: a case report. J Med Case Rep 11: 195, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]



