Abstract
Background
We describe the epidemiology of COVID-19 in South Africa following importation and during implementation of stringent lockdown measures.
Methods
Using national surveillance data including demographics, laboratory test data, clinical presentation, risk exposures (travel history, contacts and occupation) and outcomes of persons undergoing COVID-19 testing or hospitalised with COVID-19 at sentinel surveillance sites, we generated and interpreted descriptive statistics, epidemic curves, and initial reproductive numbers (Rt).
Findings
From 4 March to 30 April 2020, 271,670 SARS-CoV-2 PCR tests were performed (462 tests/100,000 persons). Of these, 7,892 (2.9%) persons tested positive (median age 37 years (interquartile range 28–49 years), 4,568 (58%) male, cumulative incidence of 13.4 cases/100,000 persons). Hospitalization records were found for 1,271 patients (692 females (54%)) of whom 186 (14.6%) died. Amongst 2,819 cases with data, 489/2819 (17.3%) travelled internationally within 14 days prior to diagnosis, mostly during March 2020 (466 (95%)). Cases diagnosed in April compared with March were younger (median age, 37 vs. 40 years), less likely female (38% vs. 53%) and resident in a more populous province (98% vs. 91%). The national initial Rt was 2.08 (95% confidence interval (CI): 1.71–2.51).
Interpretation
The first eight weeks following COVID-19 importation were characterised by early predominance of imported cases and relatively low mortality and transmission rates. Despite stringent lockdown measures, the second month following importation was characterised by community transmission and increasing disease burden in more populous provinces.
Keywords: COVID-19, SARS-CoV-2, South Africa, Public Health, Lockdown, Epidemiology, Outbreak
Research in context.
Evidence before this study
The African continent appears to have been relatively less affected by the COVID-19 pandemic. Cases of COVID-19 were documented later from the African continent, and reported case burdens and mortality rates have consistently been lower relative to other continents. The need for and the effectiveness of stringent public health measures (including lockdowns) in African countries have been questioned, particularly in the context of the negative social and economic impact of these measures. Few African countries have reported on the epidemiology of COVID-19 following importation. Many epidemiological descriptions reflect limited availability of laboratory testing and/or incomplete surveillance.
Added value of this study
South Africa has an extensive public and private laboratory network and was able to initiate testing for SARS-CoV-2 prior to importation and to achieve testing rates substantially higher than other sub-Saharan African countries. The ‘lockdown’ implemented by South African authorities was one of the most stringent on the continent, with all persons except essential service workers confined to their place of residence for 5 weeks from March 27th to April 30th, 2020. This paper documents the importation of SARS-CoV-2 and subsequent community transmission during the first eight weeks of the COVID-19 pandemic in South Africa whilst lockdown measures were in place.
Implications of all the available evidence
This paper comprehensively describes the epidemiology of COVID-19 and progression of the pandemic in an African upper-middle income country whilst robust disease surveillance and stringent lockdown measures were in place .
Alt-text: Unlabelled box
Introduction
The Department of Health in the Republic South Africa confirmed the country's first case of SARS-CoV-1 on March 5, 2020 in a symptomatic traveller who had returned from Italy on March 3, 2020 [1]. This case was identified eight weeks after the World Health Organization (WHO) reported a cluster of pneumonia cases of unknown cause in Wuhan City, Hubei Province (on 5 January 2020) [2] and three weeks after the first case was reported elsewhere in Africa (in Egypt on 14 February 2020 [3]). By 15 March 2020, 51 coronavirus disease (COVID-19) cases had been confirmed in South Africa.
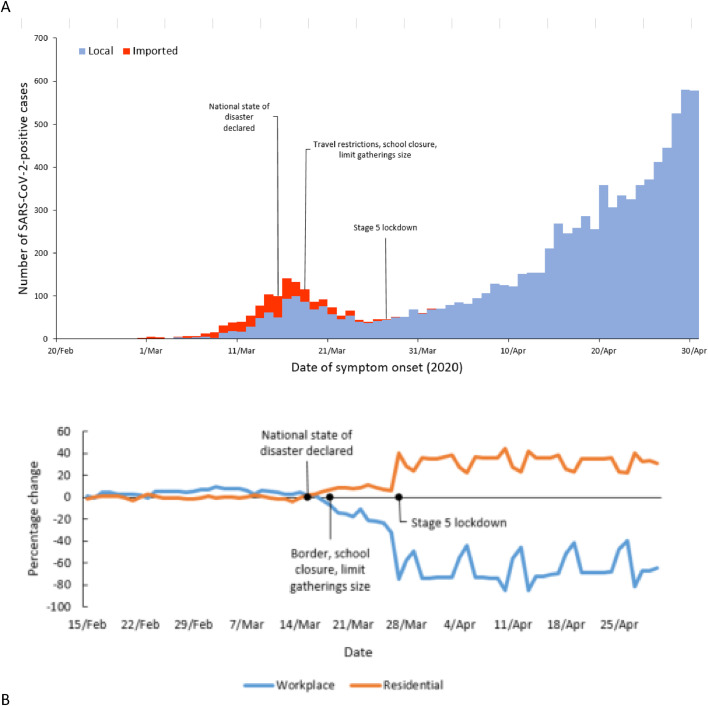
In an effort to prevent community transmission, the South African government responded swiftly, declaring a national state of disaster. Restrictions on international travel, school closures and limits on the size of public gatherings were implemented [1]. Interventions to strengthen laboratory testing and in-patient facilities, train and recruit staff, procure personal protective equipment and implement infection prevention and control protocols were intensified to meet the anticipated health requirements of the pandemic [1]. On 27 March 2020, after community-transmitted cases had been detected, the President declared a nationwide ‘level 5′ lockdown, halting non-essential business and confining all persons to their residences until easing of restrictions on 30 April 2020. These interventions were implemented early during the evolving COVID-19 pandemic when limited information was available on the impact of age and highly prevalent conditions such as tuberculosis and HIV on the natural history and outcome of infection with SARS-CoV-2 [4,5].
We describe the epidemiology of COVID-19 from 5 March to 30 April 2020 following importation of SARS-CoV-2 while the ‘level 5′ lockdown was in place. We describe characteristics of cases identified during this eight-week period and explore changes in disease epidemiology over time. Our findings provide data on COVID-19 epidemiology in an African country where testing rates were substantially higher than other sub-Saharan countries [6].
Methods
Setting
South Africa is an upper middle-income country (UMIC) and a regional economic hub with an approximate population of 58 million, of whom one third are under 18 years of age [7]. The country carries an enormous burden of tuberculosis (TB, approximately 520 cases/100,000 persons [8]) and HIV (7.8 million persons with 5.2 million on antiretroviral treatment) [9] Data from South Africa's long-standing influenza-like-illness sentinel surveillance programme indicated that approximately 20% of the population develops influenza each season, with approximately 11,500 annual deaths [10]. Approximately 15% of South Africans have access to private medical services through medical insurance.
Public health responses to the South African COVID-19 pandemic
Disaster Management Act regulations issued on 15 March 2020: 1) prohibited international air travel into and from South Africa for non-citizens; 2) limited the size of gatherings to fewer than 100 persons, or 50 persons at places where liquor is sold3) closed schools until 15 April 2020, and suspended visits to correctional service facilities, institutional care homes and hospitals; and 4) limited the sale, dispensing and transportation of liquor. These limitations were broadened to a complete ‘lockdown’ on 27 March 2020 that closed all land and sea ports, prohibited all gatherings except funerals (limited to 50 persons), instructed all businesses and commuter transport services to cease operations save those providing essential goods or services, banned the sale of tobacco and alcohol, and required every person to remain at home except for emergencies and procurement of essential services. These regulations were eased on 30 April 2020 when South Africa moved to ‘Level 4′ restrictions. We ascertained the impact of lockdown on mobility trends using data provided by Google [11].
Laboratory testing for SARS-CoV-2
The National Institute for Communicable Diseases (NICD) commenced in-house reverse-transcription polymerase chain reaction (RT-PCR) testing for SARS-CoV-2 [12] on 28 January 2020. Later testing was expanded to private (6 March 2020) and National Health Laboratory Service (NHLS) (9 March 2020) laboratories using commercial assays. Positive and indeterminate RT-PCR results were defined according to manufacturers’ instructions. All testing was conducted in a formal laboratory setting. Respiratory specimens including oropharyngeal (OP) or nasopharyngeal (NP) swabs, and lower respiratory tract specimens (sputum, tracheal aspirate and broncho-alveolar lavage fluid) from persons meeting the case definition or who participated in community screening and testing (CST) activities, were transported to laboratories in viral transport media (VTM). During April 2020, if flocked swabs (preferred) and VTM were unavailable, NP or OP specimens were obtained using cotton-tipped swabs moistened with sterile normal saline.
Case definitions
A confirmed case of COVID-19 was defined as a first positive SARS-CoV-2 RT-PCR test from a resident or traveller entering South Africa. A suspected case was initially defined as a person with an acute respiratory illness with at least one of: cough, sore throat, shortness of breath or fever ≥38 °C or history of fever and: 1) had contact with a confirmed or probable case of COVID-19; or 2) had a travel history to an area with SARS-CoV-2 community transmission; or 3) worked or visited a health care facility where COVID-19 patients were treated. In early April 2020, the suspected case definition was expanded to include persons who were admitted with severe pneumonia of unknown aetiology. In late March 2020, public and private sector clinicians started requesting SARS-CoV-2 tests outside the specified testing criteria, including from asymptomatic contacts of persons with laboratory-confirmed SARS-CoV-2, and from asymptomatic persons identified during community screening.
Surveillance and data collection methodology
From early February 2020, screening for the presence of fever amongst returning travellers was implemented at all ports of entry. Case definitions were widely disseminated to emergency departments, general practitioners, intensive treatment units, district and tertiary hospitals and primary care facilities. Training on all aspects of COVID-19 was provided. The NICD 24-hour hotline was expanded to support clinical consultations and testing of suspected cases.
Community screening and testing was implemented across the country from 8 April 2020 onwards with varying methodologies including: 1) mass screening of persons in public venues followed by SARS-CoV-2 testing of symptomatic persons, and/or 2) SARS-CoV-2 testing of asymptomatic contacts of essential service workers with confirmed SARS-CoV-2 infection in retail, healthcare and manufacturing sectors, and/or 3) SARS-CoV-2 testing of asymptomatic persons who lived adjacent to confirmed cases.
COVID-19 is a ‘Category 1′ notifiable medical condition (NMC) and requires immediate notification to health authorities following laboratory confirmation. Results from all testing laboratories were shared electronically with the NICD/NHLS data warehouse, daily. From 28 January 2020, clinicians were requested to submit by email a ‘patient under investigation’ (PUI) form (adapted from the WHO case report form), which included travel history, risk factors, clinical symptoms, admission details and hospitalisation outcome. During March 2020, surveillance teams collected missing clinical data on confirmed cases directly from patients, next-of-kin, healthcare providers or provincial officials. From May 2020, the PUI form was no longer requested by the NICD as over April 2020, data quality became increasingly poor, many clinicians did not submit forms, and the surveillance team collecting additional data was overwhelmed.
An electronic hospital system (DATCOV) was set up in early April 2020 to collect demographic and clinical information on confirmed COVID-19 cases admitted to public and private sector hospitals. Participating hospitals provided data at multiple time points (admission, daily clinical updates, discharge/death) pertaining to risk factors, clinical presentation, treatment and outcome at the end of hospitalisation. Up to 30 April, 197 facilities (69 public-sector and 128 private-sector) from nine provinces and representing 19.5% of public and 52.5% of private hospitals respectively, submitted data to DATCOV. National death data were reported by the National Department of Health following collation of COVID-19 deaths notified to provincial health authorities. COVID-19 deaths were defined and reported according to WHO criteria.
Data management and analysis
Case line lists were generated from laboratory testing data and NMC reports. Line lists were routinely de-duplicated and updated based on feedback from district health teams. For this data analysis we abstracted from line lists and the DATCOV surveillance system including patient demographics and laboratory test data, symptom onset date, clinical symptoms, risk factors for acquisition and death, travel history, hospital admission data and outcome. All data is available in summary form on the NICD website in epidemiologic, laboratory testing or hospital admission reports (https://www.nicd.ac.za/diseases-a-z-index/covid-19/surveillance-reports/)
Table 1 describes epidemiological measures reported in this paper and their definitions. An epidemic curve was plotted using date of symptom onset. We estimated the initial reproductive number (Rt) using data from imported and locally acquired infections based on date of symptom onset for cases identified during the exponential growth phase of the epidemic before control measures were instituted. For imported cases, we used the date of arrival in the country if the date of symptom onset preceded the arrival date. We implemented the analysis nationally and for provinces where sufficient data for the estimation were available. We used the maximum likelihood estimator for known serial interval (SI) described by White and Pagano [13] (using a fixed serial interval, gamma distributed with mean of 5.3 and standard deviation of 1.8 days [14]). To assess the impact of imported infections on Rt estimation, we allowed the imported cases to transmit only (main analysis) or to acquire and transmit infection (for comparability with Rt estimates obtained without differentiation between imported and locally acquired infections). In addition, at national level only, we used likelihood-based estimation methodology for the simultaneous estimation of the initial Rt and SI [13] allowing imported cases to transmit only. As only 533/9230 (58%) of cases until 27 March 2020 had a date of symptom onset, missing dates were imputed using chained equation multiple (100) imputations [15]. A negative-binomial model was fitted to confirmed COVID-19 cases for which the date of symptom onset was available and used to impute symptom onset date for cases with missing information. The model predictors for imputation were the following: health sector (private versus public), age, day of the week of sample collection, month of sample collection and province. The initial Rt was estimated for each imputed dataset (100 time series generated through multiple imputations encompassing observed and imputed dates of symptom onset)[15,16] for all cases reported before 27 March 2020. Estimates are reported as the median across the 100 imputations. We combined the results from all 100 imputations to obtain a confidence interval that incorporated both imputation error, as well as random error [15]. The Rt analysis was implemented using the R0 package of R version 4.0.2. We compared demographic features of cases with specimen collection date in March versus April 2020 using frequencies and proportions (categorical variables) and median and interquartile ranges (continuous variables). We used univariate logistic regression to determine the strength of association between diagnosis during the month of March 2020 (predominantly imported cases) versus April 2020 (after international travel ceased) with selected variables having relatively complete datasets (age, sex, province and health sector).
Table 1.
Epidemiological measures and respective definitions used to describe the importation and establishment of SARS-CoV-2 in South Africa, 4 March- 30 April 2020.
| Epidemiological measure | Definition |
|---|---|
| Percentage test positive | The number of SARS-CoV-2 positive tests in a defined period/total number of tests performed in the same period |
| Testing rate | The number of laboratory tests for SARS-CoV-2 divided by the 2019 mid-year population estimates (obtained from Statistics SA) expressed as tests per 100,000 persons in a defined time period |
| Incidence risk | The number of laboratory-confirmed cases of COVID-19 divided by the 2019 mid-year population estimates (obtained from Statistics SA) expressed as cases per 100,000 persons in a defined time period. |
| In-hospital case-fatality ratio | The number of in-hospital deaths in a defined period/the number of laboratory-confirmed hospitalised cases of COVID-19 in the same period |
Ethics statement
Surveillance activities for NMC including SARS-COV-2 infection are conducted by the NICD according to National Health Act Regulations which allow for the use of case data for public health surveillance without individual consent in the interests of safeguarding public health Publication of surveillance data in peer-reviewed literature was approved by the University of the Witwatersrand Human Research Ethics Committee
Role of funding sources
Collection and analysis of these data were funded through the NICD, national and provincial health departments by South African taxpayers who played no role in the interpretation nor decision to publish this data.
Results
Laboratory testing
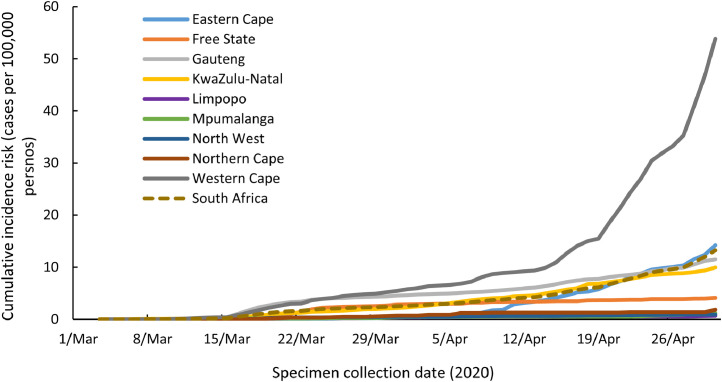
From 4 March to 30 April 2020, 271,670 RT-PCR tests for SARS-CoV-2 were performed equating to 462 tests/100,000 persons (Table 2 and Figure S1). Initially the majority of tests were conducted in private sector laboratories (Figure S2). Of the 271,670 performed tests, 271,345 had conclusive test results with 7892 (2.9%) positive and 415 (0.2%) with indeterminate results. The testing rate ranged from 85 tests/100,000 persons in North West Province (NW) to 835 tests/100,000 persons in the Western Cape (WC) Province (Table 2).
Table 2.
SARS-CoV-2testing rate (tests per 100,000 persons), and the number and incidence risk of laboratory-confirmed cases of COVID-19 by province, South Africa, 4 March–30 April 2020, and official reported number of COVID-19 deaths, 2 May 20,201.
| Province | Mid-2019 population* | Number of tests performed (%) | Tests per 100,000 persons | Number of confirmed cases (%) | Incidence risk (cases per 100 000 persons) | Number of admissions to hospital participating in the surveillance network2 (%) | COVID-19 deaths by 30 April 2020 |
||
|---|---|---|---|---|---|---|---|---|---|
| Officially reported1 | In-hospital deaths3 (DATCOV) | Case-fatality ratio amongst admitted cases | |||||||
| Eastern Cape | 6712,276 | 27,139 (10) | 404 | 957 (12.1) | 14.4 | 159 (15.2) | 13 | 39 | 39/223 (17.5) |
| Free State | 2887,465 | 12,239 (4.5) | 424 | 121 (1.5) | 4.4 | 33 (3.2) | 6 | 5 | 5/32 (15.6) |
| Gauteng4 | 15,176,115 | 100,361 (36.9) | 661 | 1787 (22.6) | 11.1 | 212 (20.3) | 12 | 18 | 18/252 (7.1) |
| KwaZulu-Natal5 | 11,289,086 | 54,440 (20) | 482 | 1145 (14.5) | 9 | 232 (22.2) | 34 | 39 | 39/330 (11.8) |
| Limpopo | 5982,584 | 5801 (2.1) | 97 | 40 (0.5) | 0.5 | 6 (0.6) | 2 | 1 | 1/6 (16.7) |
| Mpumalanga | 4592,187 | 6679 (2.5) | 145 | 55 (0.7) | 1.6 | 1 (0.1) | 0 | 0 | 0/1 (0) |
| North West | 4027,160 | 3432 (1.2) | 85 | 36 (0.5) | 0.7 | 12 (1.2) | 0 | 0 | 0/12 (0) |
| Northern Cape | 1263,875 | 1722 (0.6) | 136 | 23 (0.3) | 2.1 | 7 (0.7) | 0 | 0 | 0/6 (0) |
| Western Cape6 | 6844,272 | 57,170 (21) | 835 | 3728 (47.2) | 57.5 | 385 (36.8) | 56 | 84 | 84/377 (22.3) |
| South Africa | 58,750,220 | 271,670 | 462 | 7892 | 13.4 | 1047 | 123 | 186 | 186/1239 (15.0) |
These data were released on 2 May 2020 in the National Department of Health's official COVID Media Release and reflect those reported by provincial health authorities to NDoH from 3 March until that day. No death figures were reported in official statistics released on 1 May 2020.
2,3These data were not available in real time, as the DATCOV surveillance network was rolled out incrementally, and cases were added retrospectively.
The most populous province encompassing the cities of Johannesburg and Tshwane (Pretoria).
The city of eThekwini (Durban), a large port and economic hub is located here.
The city of Cape Town, a tourist destination and economic hub is located here.
Statistics South Africa.
Demographic features, clinical presentation and outcomes of cases
The median age of COVID-19 cases was 37 years (interquartile range [IQR], 28–49 years) with the largest proportion of cases in the 30–39-year age group (2151/7892; 27.3%) (Table 3). Fifty-eight per cent (4568/7892) of cases were male. The majority of cases were reported in WC (3728, 47.2%), followed by Gauteng (GP, 1787, 22.6%) and the Eastern Cape (EC) provinces (957, 12.1%). Amongst cases where these data were provided, 2710/3215 (84%) were symptomatic. Just under 60% of cases (4641/7892; 58.1%) were diagnosed in public-sector healthcare facilities or through the CST program. Amongst patients with available information, the commonest reported symptoms were fever/rigors/chills (1523/2286; 66.6%), cough (1868/2995; 62.4%) and sore throat (1197/2274; 53%). Co-morbidities were present in 1047/3043 (34%) of cases with available data and included diabetes mellitus (DM) (344/2927; 11.8%), hypertension (HT) (322/2824; 11.4%), asthma (227/2940; 7.7%), HIV (173/2330, 7.4%), obesity (132/2369, 5.6%), and heart disease (161/2931; 5.5%). By 2 May 2020, 123 COVID-19-related deaths had been officially reported (Table 2). Cases diagnosed in April 2020, compared with March 2020 were more likely to be younger, female and resident in one of the more populous provinces (EC, GP, KwaZulu-Natal (KZN) or WC, Table 3).
Table 3.
Clinical and demographic characteristics of COVID-19 cases diagnosed amongst persons resident in or travelling through South Africa by month of diagnosis, 4 March 2020–30 April 2020 (n = 7892).
| Characteristic | All cases (n = 7892) | Month of diagnosis amongst all cases (N = 7791)* |
Admitted to a sentinel surveillance hospital | ||
|---|---|---|---|---|---|
| March 2020 (n = 1480) | April 2020 (n = 6311) | Unadjusted odds ratio comparing diagnosis in April versus March 2020 (95% CI)1 | (n = 1271) | ||
| Age (years) | |||||
| Median (IQR) –years | 37 (28–49) | 40 (29–54) | 37 (28–48) | 47 (33–60) | |
| Distribution – no./ total no. (%) | |||||
| 0–9 years | 232 (2.9) | 28 (1.9) | 200 (3.2) | 1.34 (0.89–2.03) | 36 (2.8) |
| 10–19 years | 388 (4.9) | 49 (3.3) | 332 (5.3) | 1.28 (0.93–1.76) | 33 (2.6) |
| 20–29 years | 1630 (20.7) | 300 (20.3) | 1311 (20.8) | 0.82 (0.69–0.97) | 153 (12) |
| 30–39 years | 2151 (27.3) | 335 (22.6) | 1796 (28.5) | Reference | 227 (17.9) |
| 40–49 years | 1558 (19.7) | 266 (18.0) | 1277 (20.2) | 0.90 (0.75–1.07) | 224 (17.6) |
| 50–59 years | 1063 (13.5) | 261 (17.6) | 782 (12.4) | 0.57 (0.47–0.68) | 229 (18) |
| 60–69 years | 500 (6.3) | 137 (9.3) | 353 (5.6) | 0.49 (0.39–0.62) | 160 (12.6) |
| 70–79 years | 247(3.1) | 87 (5.9) | 157 (2.5) | 0.34 (0.25–0.45) | 105 (8.3) |
| ≥80 years | 83 (1.1) | 14 (1.0) | 67 (1.1) | 0.91 (0.51–1.63) | 50 (3.9) |
| Unknown | 39 (0.5) | 3 (0.2) | 36 (0.6) | 2.21 (0.68–7.23) | |
| Sex | |||||
| Male | 4568 (57.9) | 690 (46.6) | 3843 (60.9) | Reference | 578 (45.5) |
| Female | 3262 (41.3) | 789 (53.3) | 2411 (38.2) | 1.62 (1.42–1.86) | 692 (54.5) |
| Unknown | 62 (0.8) | 1 (0.1) | 57 (0.9) | – | 1 (0.1) |
| South African province | |||||
| Eastern Cape | 957 (12.1) | 24 (1.6) | 930 (14.7) | 22.34 (13.44 −37.14) | 234 (18.4) |
| Gauteng | 1787 (22.6) | 699 (47.2) | 1054 (16.7) | 2.16 (1.58–2.94) | 257 (20.2) |
| KwaZulu-Natal | 1145 (14.5) | 242 (16.4) | 883 (14.0) | 5.69 (4.09–7.91) | 336 (26.4) |
| Western Cape | 3728 (47.2) | 374 (25.3) | 3312 (52.5) | 7.83 (5.73–10.69) | 385 (30.3) |
| Free State | 121 (1.5) | 81 (5.5) | 38 (0.6) | Reference (these provinces combined) | 33 (2.6) |
| Limpopo | 40 (0.5) | 18 (1.2) | 22 (0.4) | 6 (0.5) | |
| Mpumalanga | 55 (0.7) | 15 (1.0) | 40 (0.6) | 1 (0.1) | |
| Northern Cape | 23 (0.3) | 9 (0.6) | 14 (0.2) | 7 (0.6) | |
| North West | 36 (0.5) | 18 (1.2) | 18 (0.3) | 12 (0.9) | |
| Sector where case was tested | |||||
| Private | 3244 (41.1) | 1294 (87.4) | 1905 (30.2) | Reference | 564 (44.4) |
| Public | 4641 (58.8) | 180 (12.2) | 4406 (69.8) | 12.19 (10.23–14.54) | 707 (55.6) |
| Unknown | 7 (0.1) | 6 (0.4) | 0 | – | |
| Exposure to source of transmission within past 14 days – no./ total no. (%) | |||||
| International travel to region with local transmission | 489/2819 (17.3)2 | 466/674 (69.1) | 5/2098 (0.2) | – | |
| Close contact with confirmed case | 642/2056 (31.2) | 138/579 (23.8) | 496/2079 (23.9) | – | |
| Healthcare worker | 344/3441 (9.9) | 64/871 (7.4) | 278/2512 (11.1) | – | 91/1271 (7.2) |
| No reported exposure | 4396 /7892 (55.7) | 538/1480 (35.7) | 3799/6311 (60.2) | – | |
| Self-reported symptoms – no./ total no. (%) | |||||
| Cough | 1868/2995 (62.4) | 513/795 (64.5) | 1238/2151 (61.7) | – | 126/406 (31.0) |
| Fever | 1380/2871 (48.1) | 435/790 (55.1) | 917/2030 (45.2) | – | 102/406 (25.1) |
| Fever/chill/rigors | 1523/2286 (66.6) | 489/803 (60.9) | 1005/2032 (49.5) | – | – |
| Sore throat | 1197/2274 (52.6) | 420/691 (60.8) | 753/1540 (48.9) | – | – |
| Myalgia | 881/2700 (32.6) | 298/708 (42.1) | 565/1946 (29.0) | – | 42/406 (10.3) |
| Rigors/chills | 552/2044 (27.0) | 210/593 (35.4) | 335/1411 (23.7) | – | – |
| Fatigue/malaise | 622/2659 (23.4) | 236/692 (34.1) | 371/1921 (19.3) | – | 19/406 (4.7) |
| Dyspnoea | 732/2707 (27.0) | 217/725 (29.9) | 499/1934 (25.8) | – | 104/406 (25.6) |
| Diarrhoea | 264/2648 (10.0) | 99/671 (14.8) | 156/1930 (8.1) | – | 12/406 (3.0) |
| Irritability or confusion | 94/2614 (3.6) | 33/654 (5.1) | 57/1915 (3.0) | – | 5/406 (1.2) |
| One or more | 2701/3215 (84.0) | 772/890 (86.7) | 1884/2271 (83.0) | – | 151/406 (37.2) |
| None | 465/3218 (14.5) | 106/894 (11.7) | 350/2270 (15.4) | – | – |
| Co-existing condition – no./ total no. (%) | |||||
| Asthma | 227/2940 (7.7) | 79/798 (9.9) | 146/2091 (7.0) | – | 74/1061 (7.0) |
| Heart disease | 161/2931 (5.5) | 39/786 (5.0) | 120/2095 (5.7) | – | 41/1061 (3.9) |
| Hypertension | 322/2824 (11.4) | 32/724 (4.4) | 284/2050 (13.9) | – | 317/1061 (29.9) |
| Diabetes mellitus | 344/2927 (11.8) | 45/780 (5.8) | 293/2096 (14.0) | – | 227/1061 (21.4) |
| Obesity | 132/2369 (5.6) | 22/710 (3.1) | 108/1615 (6.7) | – | |
| Chronic kidney disease | 72/2908 (2.5) | 13/777 (1.7) | 58/2081 (2.8) | – | 29/1061 (2.7) |
| Chronic lung disease | 80/2921 (2.7) | 12/786 (1.5) | 67/2085 (3.2) | – | |
| Malignancy | 24/1061 (2.3) | ||||
| HIV | 173/2330 (7.4) | 5/654 (0.8) | 166/1642 (10.8) | – | 108/1061 (10.2) |
| Immunodeficiency other than HIV | 23/2335 (1.0) | 5/707 (0.7) | 18/1584 (1.1) | – | |
| Neurological disease | 19/2375 (0.8) | 4/720 (0.6) | 15/1612 (0.9) | – | |
| Chronic liver disease | 6/1540 (0.4) | 5/711 (0.7) | 13/1684 (0.8) | – | |
| Pregnancy (females 15–49 years)⁎⁎ | 58/1305 (4.4) | 5/317 (1.6) | 53/974 (5.4) | – | 28/385 (7.3) |
| Any pre-existing co-morbidities | 1047/3043 (34.4) | 201/810 (24.8) | 831/2182 (38.1) | – | 520/1061 (49.0) |
| Highest level of care received | – | ||||
| General | NR | 84/174 (48.3) | 602/870 (69.2) | – | 909/1271 (71.5) |
| High-care | NR | 10/174 (10.3) | 87/870 (10.0) | – | 160/1271 (12.6) |
| Intensive care | NR | 34/174 (19.5) | 171/870 (19.7) | – | 202/1271 (15.9) |
| Total hospital admission duration | – | ||||
| Median (IQR) - days | NR | – | 9 (4–14) | ||
| Outcome at end of hospitalisation | – | ||||
| Discharged | – | 1053/1271 (82.9) | |||
| Transferred to another facility | 32/1271 (2.5) | ||||
| Died | 186/1271 (14.6) | ||||
101 missing date of collection but included in overall NICD database.
Amongst female cases.
Confidence interval.
Origins of 461 cases where this was reported include Europe (318, 69%), Americas (57, 12%), Middle East (26, 6%), South East Asia (20, 4%) or mixed (14, 4%).
Clinical characteristics and outcomes of hospitalised cases
Amongst 7892 cases identified between 4 March and 30 April 2020, hospitalisation records were found for 1271 (16.1%) cases of whom 54.4% (692/1271) were female. The median age of hospitalised persons was 47 years (IQR, 33–60 years), with 71 (5.6%) admissions recorded amongst patients aged <20 years. The majority of admissions were reported from three provinces, namely WC (385; 30.3%), KZN (336; 26.4%) and GP (257; 20.2%). Fifty-six per cent (707/1271) of admissions were reported from public-sector hospitals. Amongst 1061 (83.5%) hospitalised patients with data provided, 274 (25.8%) and 246 (23.2%) had one or two comorbid conditions, respectively. The most commonly reported comorbid conditions were HT (317/1061, 29.9%), DM (227/1061, 21.4%) and HIV infection (108/1061, 10.2%). A total of 100 (7.9%) patients required invasive mechanical ventilation and 300 (23.6%) required supplemental oxygen. Amongst 1271 admissions, 186 (14.6%) patients died in hospital, with the highest in-hospital mortality amongst the ≥80 years age category (21/50, 42%) (Table 4). Amongst patients with outcomes other than death, 843 (80.5%) were discharged and 31 (3%) were transferred to other facilities.
Table 4.
Age-related distribution of officially reported national COVID-19 deaths as of 2 May 2020, and in-hospital COVID-19 deaths (DATCOV sentinel surveillance network) as of 30 April 2020.
| Age group (years) | Officially reported deaths, n (%) | In-hospital deaths |
|
|---|---|---|---|
| Number (%) | Case fatality ratio (n/N,%) | ||
| 0–9 | 0 | 0 | 0/36 (0) |
| 10–19 | 0 | 1 (0.5) | 1/33 (3.0) |
| 20–29 | 1 (1) | 1 (0.5) | 1/153 (0.7) |
| 30–39 | 6 (5) | 10 (5.4) | 10/227 (4.4) |
| 40–49 | 19 (16) | 26 (14.0) | 26/224 (11.6) |
| 50–59 | 23 (19) | 43 (23.1) | 43/229 (18.8) |
| 60–69 | 30 (24) | 47 (25.3) | 47/160 (29.4) |
| 70–79 | 28 (23) | 37 (19.9) | 37/105 (35.2) |
| ≥80 | 16 (13) | 21 (11.3) | 21/50 (42.0) |
| Total | 123 | 186 | |
Epidemiological measures
Fig. 1A illustrates the epidemic curve of COVID-19 cases by actual or imputed date of symptom onset for the period 4 March to 30 April 2020. Fig. 1B quantifies changes in national geospatial mobility relative to 2–3 January 2020 amongst Google users. At eight weeks following importation, the national cumulative incidence was 13.4 cases/100,000 persons with the highest risk in the WC province (57.5 cases/100,000 persons). The cumulative incidence increased over time (Fig. 2) with the WC province exceeding that of the other provinces from the third week following importation (5 April 2020). Through to 30 April 2020, the cumulative incidence risk was highest amongst those in the 40–59 years age group, with the lowest risk amongst children aged 0–19 years (Figure S2A). The cumulative incidence risk was higher amongst females than males (29.5 cases/100,000 persons [95% CI 28.9–30.1] versus 22.5 cases/100,000 persons [95% CI 21.9–23.1] and varied by age group (Figure S2B).
Fig. 1.
(A) Epidemic curve of the number of SARS-CoV-2 RT-PCR/ PCR positive samples by date of symptom onset (with missing data imputed), South Africa, 4 March to 30 April 2020, (B) The percentage change in mobility relative to baseline (as measured 2–3 January 2–2020) from residential (orange) and workplace (blue) by Google users in South Africa, 15 February 2020–30 April 2020 (data publically available at https://www.google.com/covid19/mobility/).
Fig. 2.
Cumulative incidence risk of laboratory-confirmed COVID-19 cases by specimen collection date, for South Africa and provinces, 1 March 4–April 30, 2020.
Amongst 2819 cases where data were provided, 489 (17.3%) reported international travel during their incubation period, of which 466 (95%) were reported in March 2020 (Table 3). Other reported risk factors included close contact with a confirmed case (642/2056, 31.2%) and being a healthcare worker (344/3441, 10%). No risk exposure for COVID-19 was reported for 4396 /7892 (55.7%) persons.
The initial Rt at national level (calculated assuming imported cases were transmitters only and using a serial interval of 5.3 days) was 2.08 (95% CI: 1.71–2.51) (Table 5). When imported cases were modelled as being infected and capable of transmitting SARS-CoV-2, the initial Rt was calculated to be 4.15 (95% CI: 3.60–4.74) and the mean serial interval was 5.3 days (SD: ±2.7 days). Using the simultaneous estimation method for Rt and the serial interval, and assuming imported cases to be transmitters only, the national initial Rt was 1.82 (95% CI: 1.57–2.22) and the mean serial interval was 4.6 days (SD: ±2.2 days). For the four provinces where Rt estimation was possible, the initial Rt ranged from 1.76 to 2.18 when imported cases were assumed to transmit only and from 2.10 to 4.90 when imported cases were allowed to be infected and transmit.
Table 5.
Estimated SARS-CoV-2 initial reproductive number in South Africa before implementation of control measures on 18 March 2020.
| Location | Rt (95% CI*) using a SI* of 5.3 days |
|
|---|---|---|
| Imported cases as transmitters only | Imported cases treated as local cases | |
| National | 2.08 (1.71–2.51) | 4.15 (3.60–4.74) |
| Eastern Cape Province | 1.85 (1.13–2.82) | 2.10 (1.32–3.13) |
| Gauteng Province | 2.18 (1.83–2.57) | 3.51 (3.06–4.01) |
| KwaZulu-Natal Province | 1.82 (1.21–2.60) | 3.24 (2.40–4.24) |
| Western Cape Province | 1.76 (1.11–2.62) | 4.90 (3.75–6.26) |
CI=confidence interval; SI=serial interval.
Discussion
In this description and analysis of the epidemiology of COVID-19 in South Africa, a UMIC with high testing rates, we report that the first eight weeks following the introduction of SARS-CoV-2 were characterised by early predominance of imported cases, followed by a transition to community-level transmission despite rapid implementation of public health interventions and scale up of testing, case-finding, quarantine and isolation programmes. Cases during the first eight weeks had a typical COVID-19 clinical presentation with a relatively low COVID-19-associated case-fatality ratio amongst hospitalised patients [17], [18], [19]. In this discussion, we reflect on how case detection strategies, testing capacity, clinical presentation and outcomes, the initial reproductive rate and the timing and nature of public health interventions impacted the epidemiology of COVID-19 in South Africa during the first eight weeks following importation.
Despite high volumes of international travel, early implementation of screening measures and strengthening of passive surveillance, COVID-19 cases were detected later in South Africa compared with other countries. Unlike other African countries (such as Uganda [20], Zambia [21]) South Africa did not implement active monitoring and case-finding amongst in-bound travellers. It is likely that initial imported cases may have gone undetected and that early testing criteria were too restrictive as phylogenetic analyses of South African strains showed evidence of multiple introduction events and some evidence of prior local transmission [22,23]. In May 2020 the testing criteria were altered to ‘the presence of any respiratory symptoms with or without fever’, with no requirement for a travel history nor exposure risk.
Unlike most African countries [24], South Africa was able to scale up RT-PCR testing rapidly through well-equipped and extensive NHLS and private laboratory networks. The percentage test positivity rate during the first eight weeks was considerably lower than reported from other African countries [6], suggesting adequate test coverage and expansion of testing capacity. The geographical variation in testing rate across the provinces of South Africa therefore reflects the relative predominance of cases in more populous provinces following importation to large urban centres, combined with patient health seeking behaviour and clinician awareness of case definitions. Asymptomatic cases accounted for less than 15% of all cases. These were likely under-represented, as up to half of SARS-CoV-2 infections may be asymptomatic [25] and early testing criteria required the presence of symptoms.
Our case series was characterized by typical COVID-19 clinical presentations, comparable prevalence of comorbid illness and risk factors for poor outcome as described in preceding months in Europe and North America [17,19]. HIV infection was found to increase the risk of death approximately two-fold (adjusted hazard ratio [aHR] 2.14, (95% confidence interval [CI], 1.70–2.70) in a risk factor analysis amongst a provincial cohort of South African patients [26]. The disparities between officially reported deaths and deaths amongst hospitalised persons (Table 2) may reflect under-reporting to health departments on account of health system delays. Whilst not possible to calculate nor compare meaningful case fatality ratios in our country or other African countries because of limitations in testing capacity and death reporting mechanisms, the relatively low case-fatality ratio amongst hospitalized patients (14.6%) compared with reported case series from Europe [17], [18], [19] may reflect the early practice by clinicians of precautionary admission, a health system that was not yet overwhelmed, and the younger age-composition of hospitalised patients in our cohort [27].
Our finding of a reproductive number just over two immediately following importation and until limitation of international travel indicated potential for exponential increase in case numbers. Our reproductive number falls within the range of 1.00–2.79 reported for India, Syria, the USA, Yemen, China, France, Nigeria and Russia for the period until 30 July 2020 [28]. It was hoped that the restriction of international flights and implementation of lockdown measures would stop importation of new cases and limit local transmission. However, despite implementation of a more generalised and restrictive lockdown compared with other sub-Saharan African countries [6], our analysis reveals a rapid transition from predominantly-imported cases or cases amongst their contacts, to community transmission during lockdown. This is evidenced by a decrease in the proportion of cases reporting a history of international travel, a greater odds of younger people acquiring illness and a shift from a male to a female preponderance (due to lockdown outbreaks occurring amongst essential service workers who tend to be female [29]) in April compared to March 2020.. Whilst the lockdown slowed the trajectory of the outbreak, clustered transmission amongst essential workers during lockdown [30,31], socio-economic factors limiting the capacity for social distancing in poorer communities, delays in laboratory testing, health system challenges in contact tracing and transmission from persons with asymptomatic infection [25] most likely contributed to the inability to contain SARS-CoV-2.
Incomplete case detection and admission data before and during the initial weeks following importation and missing data elements including date of symptom onset, travel history, risk exposures, outcomes and hospitalisation events preclude a deeper understanding of the transition from imported to community transmission. We elected not to calculate the daily reproductive rate using testing data or deaths because of inconsistent application of case definitions, changes in testing practices and incomplete death notification.
Our description of the importation and establishment of community transmission of SARS-CoV-2 in an African country with a robust testing infrastructure and high testing rates allows for an understanding of COVID-19 epidemiology and impact of public health interventions in an African country. Despite the most stringent lockdown on the continent, transmission of COVID-19 continued, leading to community infections and a widespread epidemic which progressed over subsequent months. The longer-term epidemiological, social and economic impact of South Africa's lockdown should be fully investigated.
Declaration of Competing Interest
KMM, PNN, JE, GN, YP, LE, DM, AMS, KS, MM, KM, NW, TM, JK, TK, WJ, LHB, SM, ST, SW declare no competing interests. AvG reports receipt of funding and equipment to the NICD to support surveillance for SARS-CoV-2 from the South African Medical Research Committee (funding), Wellcome Trust (funding), European Union, Germany (equipment), the Jack Ma Foundation (Allibaba) (testing equipment and reagents), the Centres for Disease Control, Atlanta (funding), the EDCTP (funding), the Africa CDC (funding) and the Fleming Fund (funding). CC reports grants from US CDC, grants from Wellcome Trust, grants from South Africa MRC during the conduct of the study and grants from Sanofi Pasteur outside the submitted work; NPG reports grants from NIH, grants from US CDC, grants from CDC Foundation, grants from Bill and Melinda Gates Foundation, grants from NHLS Research Trust, grants from UK MRC (JGHT scheme) all outside the submitted work. The findings and conclusions in this manuscript are those of the authors and do not necessarily represent the official position of the authors’ affiliated institutions or funding agencies.
Acknowledgments
Acknowledgements
We gratefully acknowledge the contributions of staff at private laboratories, NHLS laboratory staff, and provincial health department officials in supporting the collection and cleaning of laboratory test result data. We thank the health care workers and administrators who submitted data to the DATCOV hospital system. We pay tribute to persons who were affected by COVID-19 and remember the families and friends of those who passed away during this time.
Contributors
NPG, CC, LE and LHB conceptualised the Article. ST, TK, JK, NW and WJ ran the analyses and have verified the underlying data; TK, JK, NW, WJ, JE, AvG, MM, PNM, TM, SM, SW, GN, AMS, DM, KC, KM, NPG supported initial data collection, cleaning and analysis; KMM, YP, DM, ST, TK, JK, NW, WJ, AvG, PM, TM, SM, SW, LHB, CC, NPG interpreted data; NPG, LE and KMM wrote the first draft and revised it based on feedback from co-authors. All authors reviewed and approved the Article.
Funding statement
Surveillance for SARS-CoV-2 is conducted by the National Institute for Communicable Diseases, and South African national and provincial health departments with funding, resources and material provided by the South African government and other sources as detailed in declaration of interest.
Data sharing statement
All data are publically available in summary form in NICD epidemiology, laboratory testing or hospital admission reports (https://www.nicd.ac.za/diseases-a-z-index/covid-19/surveillance-reports/).
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.eclinm.2021.101072.
Appendix. Supplementary materials
References
- 1.Abdool Karim S.S. The South African Response to the Pandemic. N Engl J Med. 2020;382(e95) doi: 10.1056/NEJMc2014960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.WHO. Timeline: wHO's COVID-19 response. 2020. https://www.who.int/news-room/detail/29-06-2020-covidtimeline (accessed 21 September 2020 2020).
- 3.Khattab N.M., Vermund S.H., Hu Y. How coronavirus disease 2019 entered Africa and the Middle East: a case study from Egypt. Trans R Soc Trop Med Hyg. 2020;114(10):715–717. doi: 10.1093/trstmh/traa065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Togun T., Kampmann B., Stoker N.G., Lipman M. Anticipating the impact of the COVID-19 pandemic on TB patients and TB control programmes. Ann Clin Microbiol Antimicrob. 2020;19(1):21. doi: 10.1186/s12941-020-00363-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Klugman K.P., Zewdu S., Mahon B.E. Younger ages at risk of Covid-19 mortality in communities of color. Gates Open Res. 2020;4:69. doi: 10.12688/gatesopenres.13151.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Haider N., Osman A.Y., Gadzekpo A. Lockdown measures in response to COVID-19 in nine sub-Saharan African countries. BMJ Glob Health. 2020;5(10) doi: 10.1136/bmjgh-2020-003319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.SA S. Statistics South Africa; Pretoria: 2020. Midyear Population Estimates 2020. [Google Scholar]
- 8.WHO . WHO; Geneva: 2019. Global Tuberculosis Report 2019. [Google Scholar]
- 9.UNAIDS. UNAIDS. 2020. https://www.unaids.org/en/regionscountries/countries/southafrica (accessed 22 September 2020 2020).
- 10.Tempia S., Walaza S., Moyes J. Quantifying how different clinical presentations, levels of severity, and healthcare attendance shape the burden of influenza-associated illness: a modeling study from South Africa. Clin Infect Dis. 2019;69(6):1036–1048. doi: 10.1093/cid/ciy1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Google. COVID-19 Community mobility reports. 2020. https://www.google.com/covid19/mobility/ (accessed 22 September 2020 2020).
- 12.Corman V.M., Landt O., Kaiser M. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 2020;25(3) doi: 10.2807/1560-7917.ES.2020.25.3.2000045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.White L.F., Pagano M. A likelihood-based method for real-time estimation of the serial interval and reproductive number of an epidemic. Stat Med. 2008;27(16):2999–3016. doi: 10.1002/sim.3136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pitzer V.E., Chitwood M., Havumaki J. The impact of changes in diagnostic testing practices on estimates of COVID-19 transmission in the United States. medRxiv. 2020 doi: 10.1093/aje/kwab089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Archer B.N., Tempia S., White L.F., Pagano M., Cohen C. Reproductive number and serial interval of the first wave of influenza A(H1N1)pdm09 virus in South Africa. PLoS ONE. 2012;7(11):e49482. doi: 10.1371/journal.pone.0049482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.White L.F., Wallinga J., Finelli L. Estimation of the reproductive number and the serial interval in early phase of the 2009 influenza A/H1N1 pandemic in the USA. Influenza Other Respir Viruses. 2009;3(6):267–276. doi: 10.1111/j.1750-2659.2009.00106.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Karagiannidis C., Mostert C., Hentschker C. Case characteristics, resource use, and outcomes of 10 021 patients with COVID-19 admitted to 920 German hospitals: an observational study. The Lancet Respiratory medicine. 2020;8(9):853–862. doi: 10.1016/S2213-2600(20)30316-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Guan W.J., Ni Z.Y., Hu Y. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382(18):1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Richardson S., Hirsch J.S., Narasimhan M. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA. 2020;323(20):2052–2059. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lumu I. COVID-19 Response in Sub-Saharan Africa: lessons From Uganda. Disaster Med Public Health Prep. 2020:1–3. doi: 10.1017/dmp.2020.248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chipimo P.J., Barradas D.T., Kayeyi N. First 100 Persons with COVID-19 - Zambia, March 18-April 28, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(42):1547–1548. doi: 10.15585/mmwr.mm6942a5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Giandhari J., Pillay S., Wilkinson E. Early transmission of SARS-CoV-2 in South Africa: an epidemiological and phylogenetic report. medRxiv. 2020 doi: 10.1016/j.ijid.2020.11.128. 2020.05.29.20116376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tegally H., Wilkinson E., Lessells R.J. Sixteen novel lineages of SARS-CoV-2 in South Africa. Nat Med. 2021;27(3):440–446. doi: 10.1038/s41591-021-01255-3. [DOI] [PubMed] [Google Scholar]
- 24.Ondoa P., Kebede Y., Loembe M.M. COVID-19 testing in Africa: lessons learnt. The Lancet Microbe. 2020;1(3):e103. doi: 10.1016/S2666-5247(20)30068-9. -e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Emery J.C., Russell T.W., Liu Y. The contribution of asymptomatic SARS-CoV-2 infections to transmission on the Diamond Princess cruise ship. Elife. 2020;9 doi: 10.7554/eLife.58699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Boulle A., Davies M.A., Hussey H. Risk factors for COVID-19 death in a population cohort study from the Western Cape Province, South Africa. Clin Infect Dis. 2020 doi: 10.1093/cid/ciaa1198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Diop B.Z., Ngom M., Pougué Biyong C., Pougué Biyong J.N. The relatively young and rural population may limit the spread and severity of COVID-19 in Africa: a modeling study. BMJ global health. 2020;5(5) doi: 10.1136/bmjgh-2020-002699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Al-Raeei M. The basic reproduction number of the new coronavirus pandemic with mortality for India, the Syrian Arab Republic, the United States, Yemen, China, France, Nigeria and Russia with different rate of cases. Clin Epidemiol Glob Health. 2020 doi: 10.1016/j.cegh.2020.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lessells R., Moosa Y., de Oliveira T. South Africa: KwaZulu-Natal Research Innovation and Sequencing Platform (KRISP); Durban: 2020. Report into a Nosocomial Outbreak of Coronavirus Disease 2019 (COVID-19) at netcare st. Augustine's hospital. [Google Scholar]
- 30.Peleker M., Tembo T., Davies M.A. Accepted manuscript PHA-11-20-0069.R2 Asymptomatic SARS-CoV-2 infection in workplaces in South Africa – implications for control of transmission. Public Health Action. 2021 doi: 10.5588/pha.20.0069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mahomed H., Gilson L., Boulle A. The evolution of the COVID-19 pandemic and health system responses in South Africa and the Western Cape Province – how decision-making was supported by data. In: Massyn N, Day D, Ndlovu NT, Padayachee T, editors. District health barometer 2019/2020. Health Systems Trust; Durban: 2020. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.