Abstract
Today's management of the ventilated patient still relies on the measurement of old parameters such as airway pressures and flow. Graphical presentations reveal the intricacies of patient-ventilator interactions in times of supporting the patient on the ventilator instead of fully ventilating the heavily sedated patient. This opens a new pathway for several bedside technologies based on basic physiologic knowledge; however, it may increase the complexity of measurements. The spread of the COVID-19 infection has confronted the anesthesiologist and intensivist with one of the most severe pulmonary pathologies of the last decades. Optimizing the patient at the bedside is an old and newly required skill for all physicians in the intensive care unit, supported by mobile technologies such as lung ultrasound and electrical impedance tomography. This review summarizes old knowledge and presents a brief insight into extended monitoring options.
Keywords: Respiratory mechanics, Mechanical ventilation, Monitoring
Key points
-
•
Measurement of respiratory mechanics is still the mainstay of bedside monitoring of mechanical ventilation.
-
•
Ideally, the energy applied to the lung is optimized based on the equation of mechanical power.
-
•
Setting the ventilator considers multiple aspects of energy transfer and protection against ventilator-associated injury.
-
•
Newer technologies appear promising; particularly, electrical impedance tomography and lung ultrasound in combination with conventional monitoring are currently the direction for the future.
Introduction
Mechanical ventilation can harm lung tissue through barotrauma, volutrauma, atelectrauma, oxytrauma, and biotrauma. These 5 mechanisms have their own specific pathophysiological features but share similarities in their pathways of injuring the lungs (ventilator-induced lung injury [VILI]).1 All these mechanisms together may result in inflammation through local production and release of inflammatory mediators, which is known as biotrauma. The so-called lung-protective ventilation strategies aim to minimize the occurrence of the pathophysiological features of VILI. To prevent VILI, the amount of energy transferred from the mechanical ventilator to the patient should be limited to a bare minimum. To do so, tidal volume (VT) and inspiratory pressures should be kept low to minimize the risk of barotrauma and volutrauma. This review provides a brief overview on the measurement of respiratory (also referred to as pulmonary) mechanics and current developments in the field to achieve this goal.
What are respiratory mechanics?
Respiratory mechanics refer to the expression of lung function through measures of pressure and flow. From these, a variety of derived indices can be determined, such as flow, pressure, volume, compliance, resistance, and work of breathing (WOB). These factors directly affect lung volumes and therefore functional residual capacity (FRC) and gas exchange. Waveforms are derived when one of the parameters of respiratory mechanics is plotted as a function of time or as a function of one of the other parameters. This produces scalar tracings of pressure-time, flow-time, and volume-time graphics, as well as flow-volume (-V) and pressure-volume (P-V) loops. All current-generation positive-pressure ventilators—including those in the operating room—provide some monitoring of pulmonary mechanics at the bedside. Additionally, advanced respiratory mechanics monitoring modalities, such as esophageal pressure and electrical activity of the diaphragm, are available to provide sophisticated analysis of breathing efforts and diaphragmatic function. They will only be touched upon briefly because they are beyond the scope of this article. Lung ultrasound has improved the diagnostic accuracy of these modalities and nowadays is the second mainstay of ventilator management at the bedside.
Why measure respiratory mechanics?
Artificial ventilation is a temporary measure to replace or augment the function of the inspiratory muscles, providing the necessary energy to ensure a flow of gas into the alveoli during inspiration. When this support is removed, gas is exhaled passively as the lung and chest wall recoil to their original volume. An understanding of respiratory mechanics is vital to patient assessment during mechanical ventilation in order to match the available technology to the patient's needs. The goals are optimizing the patient's pulmonary physiology, providing effective gas exchange, maintaining alveolar recruitment, reducing injury potential, and ensuring hemodynamic stability. Analyzing and incorporating measurements of respiratory mechanics during your assessment will provide the information required for optimal intraoperative mechanical ventilation. Optimizing settings requires that the physician understands the intricacies of patient-ventilator interactions, particularly in terms of the measured variables as they are displayed by ventilator graphics. They represent the interaction between the ventilator and the patient's respiratory mechanics described by the equation of motion and therefore the power applied to the lung.
Physiology of chest mechanics
The respiratory system can be simplified using a linear one-compartment model, which comprises a tube representing the airways and a balloon representing the alveoli and the chest wall.
The impedance to ventilation has numerous origins, the most important of which are the following:
-
•
Elastic resistance of lung tissue and the chest wall
-
•
Resistance from surface forces at the alveolar gas-liquid interface
-
•
Frictional resistance to gas flow through the airways
-
•
Frictional resistance from deformation of thoracic tissues (viscoelastic tissue resistance)
-
•
Inertia associated with movement of gas and tissue (negligible at normal respiratory rates)
The first two forms of impedance may be grouped together as elastic resistance. These are measured when gas is not flowing within the lung and represent the total compliance of the lung and chest wall:
-
•
Compliance Crs = Δ volume/Δ pressure
-
•
Elastance ELrs = Δ pressure/Δ volume = 1/C
The last three forms may be grouped together as nonelastic resistance or respiratory system resistance. They occur while gas is flowing within the airways, and work performed in overcoming this frictional resistance is dissipated as heat and lost. Impedance to flow represents resistance of the airways:
-
•
Resistance R = Δ pressure/flow
Note that the linear one-compartment model does not take into account the fact that resistance and compliance are not constant in the case of lung and chest wall disease; instead, they exhibit a flow and volume dependency. Work performed in overcoming elastic resistance is stored as potential energy, and elastic deformation during inspiration is the usual source of energy for expiration during both spontaneous and artificial breathing.
Concept of mechanical power
In the past, adjusting the ventilator often only considered the variables positive end-expiratory pressure (PEEP), tidal volume (VT), PPlat, and ΔP.2 Other components such as flow and respiratory rate were neglected. The problem is that any alterations of one component changes another and makes it even more difficult to understand the relationship of the variables, blurring the overall picture. With this in mind, Gattinoni and colleagues3 proposed the concept of mechanical power (MP) in 2016. The equation for MP is the product of ventilating frequency and the inflation energy of the tidal cycle. The latter consists of three components: (1) the power required to overcome tissue and airways resistance during gas movement (flow-resistive work), (2) the power required to inflate the lung and chest wall from their shared initial position (VT-associated work), and (3) the (nonrecurring) power required to overcome PEEP-related recoil of the lung and respiratory system. Ultimately, in the expression for MP, every component is subsumed:
where Pappl is the applied power to the lung and Pvent and PMus are the pressures applied by the ventilator and the muscles, respectively.
Elastance (E) relates P to V, and resistance relates P to (), so the equation of motion can be modified to explain how the pressure at the airway opening (Paw) can be partitioned into a resistive and an elastic pressure component.
Here, P0 is the starting pressure, either zero end expiratory pressure or PEEP.
Written differently, these components can be divided into the following more familiar variables, which can be measured easily at the bedside:
where ΔV marks VT, ELrs is the elastance of the respiratory system (the reciprocal of compliance), RR is the respiratory rate, and I:E is the time ratio of inspiration and expiration. To begin inflation, the lung requires an energy input greater than the potential energy stored in the system by PEEP at end-exhalation. The term Δ V⋅ PEEP is the energy required to equilibrate the potential energy stored in the system at the PEEP level (ie, PEEP-related MP, when related to time).
Recruitment diminishes and distention increases as airway pressure rises. Therefore, although its mechanical effects on atelectrauma may be, on balance, lung protective over its lower range, rising PEEP is unquestionably a component of MP and, as such, favors VILI by increasing lung stress and strain. MP can be calculated using the P-V curve. The power is defined as the area between the inspiratory limb of the Δ-transpulmonary pressure (x)-volume curve, and the volume axis (y) and is measured in joules.4
Following this basic idea, Collino and colleagues5 carried out a set of animal experiments in which the MP was modified by changing the PEEP. The total MP remained unchanged, with a PEEP between 0 and 7 cmH2O. The components of energy changed, however. While the PEEP-associated energy increases, the energy decreases owing to the components of the driving pressure (ΔP) and the flow resistance, with a PEEP level up to 7 cmH2O. If the PEEP is raised further (up to 11–18 cmH2O), all components of MP as well as the total energy are steadily increasing. VT, driving pressure, and inspiratory flow exponentially increased MP by a factor of 2. A 1.4 exponential increase in MP was registered with frequency, whereas a linear increase was observed with PEEP. The same MP may produce different effects in healthy or injured lungs. A power of 12 J/min may be a meaningful upper threshold of VILI and may be a predictor of survival. MP normalized to predicted body weight was a good ventilator variable in predicting mortality in patients with adult respiratory distress syndrome (ARDS).6
Stress
Stress is a force applied to an area, such as pressure applied to the lung parenchyma. Force applied at an angle generates shear stress. In clinical terms, lung stress refers to the distending pressure within the lung, and the counterforce (external load) is the chest wall. The best indicator of the amount of stress applied to the lungs is the transpulmonary pressure, which is difficult to measure in routine practice, but can be estimated with transpulmonary pressure measurements (eg, with an esophageal balloon, see the following information). At the bedside, plateau pressure is often used as a surrogate, although this comes with limitations. Plateau pressure does not represent the actual force on the lung fibers but the pressure needed to expand the lungs and the chest wall consisting of the rib cage and the diaphragm. Patients with a stiff chest wall, for example, during pneumoperitoneum, will have a high plateau pressure that cannot automatically be translated into lung overdistension.7 Maintaining a plateau pressure less than 25 cmH2O in most patients (<30 cmH2O in patients with ARDS) would limit lung strain to less than 2 cmH2O (considered detrimental) and lung stress to 22 to 24 cmH2O (considered the upper limit of stress).8
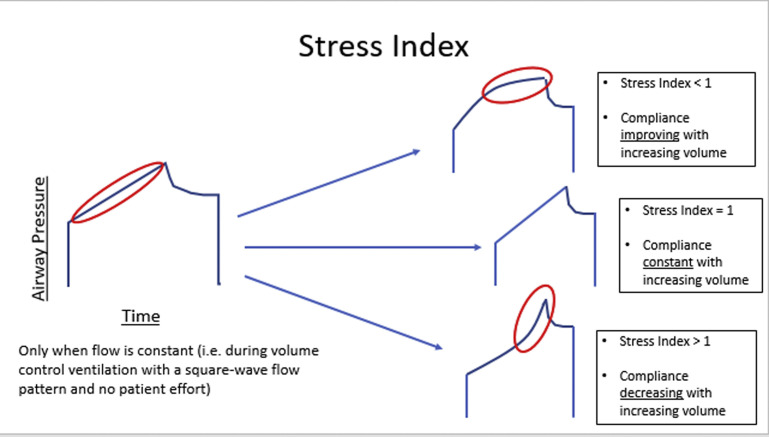
Stress index
The index is used to assess the shape of the pressure-time curve during constant -V control ventilation.9 A linear increase in pressure (constant compliance, index 1) suggests adequate alveolar recruitment without overdistention. If compliance worsens as the lungs are inflated (progressive decrease in compliance, upward concavity, index >1), this suggests overdistention, and the recommendation is to decrease the PEEP, VT, or both. If compliance improves as the lungs are inflated (progressive increase in compliance, downward concavity, index <1), this suggests tidal recruitment and potential for additional recruitment, and the recommendation is to increase PEEP (Fig. 1 ).
Fig. 1.
Stress Index.
(Courtesy of Willard Applefeld MD, Clinical Fellow in Critical Care Medicine, The National Institutes of Health, Bethesda, Maryland.)
Strain
Strain refers to the deformation, or change, in the shape of a structure compared with its resting condition. Lung strain is directly related to lung stress: stress = k × strain, where k is the specific lung elastance (13.5 cmH2O in humans).8 For the calculation of lung strain, values of FRC are obtained at zero end-expiratory pressure: lung strain = ΔV/FRC, where ΔV refers to the change in volume during inspiration. It is possible to measure strain and then calculate stress if the end-expiratory lung volume (EELV) is measured at FRC without PEEP. However, the concept of lung strain becomes complicated and less intuitive with the application of PEEP.8 , 10 , 11
Time constant
When a step change in pressure is applied to the respiratory system, the change in volume (and flow and alveolar pressure) follows an exponential curve. The speed of the volume change is described by the time constant (τ) (seconds). The time constant determines the rate of change in the volume of a lung unit that is passively inflated or deflated. It is expressed by the following relationship:
where Vt is the volume of a lung unit at time t, Vi is the initial volume of the lung unit, e is the base of the natural logarithm, and τ is the time constant.
The time constant can be calculated during inspiration or expiration. Mathematically, one time constant is equal to the product of resistance and compliance and describes the time needed to increase or decrease the volume by 63% of the total volume change: . Lung units with a higher resistance and/or compliance will have a longer time constant and require more time to fill and to empty.
Inspiratory Time Constant
Because the time constant represents the response to a step change (ie, a square pressure waveform), the inspiratory time constant (RCINSP) will be inaccurate to the extent that rise time is never zero. The inspiratory time constant is important because it determines the amount of inspiratory time required for complete VT delivery during pressure controlled modes.
Expiratory Time Constant
The expiratory time constant (RCEXP) is almost completely dependent on the patient (assuming passive expiration, PMUS = 0 and no leak) and independent of the ventilator settings to the extent that the pressure drops instantaneously to PEEP (which is never quite true because of resistance in the ventilator's expiratory circuit). RCEXP is therefore the preferred metric of the patient's dynamic respiratory mechanics. Some ventilators provide a measurement of the RCEXP in all ventilation modes, including non-invasive ventilation. Volume-time graphs can be used for calculating τ (Fig. 2 ). For the intubated patient with normal lungs, RCEXP is usually between 0.5 and 0.7 seconds. Values for different pathologies are shown in Table 1 . The expiratory time constant determines the time required for complete exhalation during any mode. Thus, if expiratory time is set less than five time constants, gas trapping with intrinsic PEEP will occur (auto-PEEP >0).
Fig. 2.
Ventilator screen image representing changes in airway pressure (PAW), flow, volume, and esophageal pressure (PES) versus time. PPEAK, inspiratory peak pressure; PPLAT, plateau airway pressure; PPLAT(ES), plateau esophageal pressure; P0, total PEEP at the airway pressure waveform; P0∗, total PEEP at the esophageal pressure waveform; VT, tidal volume.
(In: Cordioli, R and Brochard, L, Respiratory system compliance and resistance in the critically ill, Oxford Textbook of Critical Care, ISBN: 9780198855439.)
Table 1.
Overview of the static lung compliance (Cstat), inspiratory resistance (Rins) and expiratory time constant (Rexp) for different lung pathologies
| Normal Lung | ARDS | COPD | |
|---|---|---|---|
| Cstat (ml/cmH2O) | 50–70 | <40 | > 50 |
| Rins (cm HsO s/L) | <10 | 10–15 | 15–40 |
| RCEXP (s) | 0.5–0.7 | < 0.5 | > 0.7 |
Perioperative measurements
In general, modern ventilators are all able to measure airway pressure and flow. Volume is derived from the flow measurement. Apart from airway pressure displayed on the mechanical ventilator, additional sources of pressure measurements (eg, tracheal pressure, gastric pressure, and esophageal pressure) are useful for separating the effects of airway resistance and chest wall elastance on the lung mechanics.
Pressure Measurements
Airway pressure is measured universally during mechanical ventilation using a pressure transducer that converts pressure into an electrical signal (see Fig. 2). Pressure is ideally measured at the proximal airway, for example, at the tip of the endotracheal tube. The ventilator can approximate proximal airway pressures by measuring airway pressures proximal to the inspiratory and expiratory valve during zero-flow condition in the alternating limb of the ventilator circuit. Inspiratory pressure is measured proximal to the expiratory valve during inspiration and vice versa. Airway pressure is typically displayed on the ventilator screen as a function of time and can be predicted mathematically by the equation of motion.
Peak airway pressure
Peak airway pressure (Paw) is the maximum pressure recorded during inspiration. Inspiratory Paw is divided into two components, (1) resistive pressure to generate airflow through the airways and (2) alveolar pressure to distend the alveoli and chest wall. Peak Paw includes both resistive and alveolar components, but because there is no flow at end-inspiration (by brief inspiratory occlusion), plateau pressure can purely reflect alveolar pressure to distend the alveoli and chest wall.12 As per the equation of motion, PPEAK depends on PEEPTOT, flow, inspiratory resistance, VT, and respiratory system compliance (assuming muscle activity [PMUS = 0]). Therefore, any worsening of respiratory mechanics is associated with an increase in PPEAK. To distinguish between increased resistance and decreased compliance, the first step is to perform an end-inspiratory occlusion to measure plateau pressure (PPLAT). If PPLAT has not changed, the increase in PPEAK was due to an increase in resistance. If PPLAT is higher, the change in PPLAT resulted either from an increase in total PEEP or a decrease in compliance. Subsequently, an end-expiratory occlusion should be performed to measure total PEEP and detect occult or intrinsic PEEP.
End-inspiratory plateau pressure
Plateau pressure (PPLAT) is measured during mechanical ventilation by applying an end-inspiratory breath-hold for a short period of time, usually 0.5 to 2 seconds, or intermittently by manually performing an end-inspiratory occlusion. During the hold, pressure equilibrates throughout the system to approximate the proximal airway pressure Palv. As per the equation of motion, PPLAT depends on PEEPTOT, VT, and respiratory system compliance. Pplat is determined by VT and CRS during full ventilatory support: Pplat = VT/CRS. Measurement of Pplat is valid only during passive inflation of the lungs, but not during active breathing. During pressure control ventilation, the flow might decrease to zero at the end of the inspiratory phase; in this case, peak inspiratory pressure (PIP) and Pplat are equal.
The pressure drop between PPEAK and PPLAT is called transairway pressure and represents the resistive pressure. Owing to the airway resistance (Raw), proximal airway pressure will always be greater than alveolar pressure (Palv) during inspiration if flow is present. If the inspiratory flow is still positive at the end of inspiratory time, plateau pressure will be lower than the preset inspiratory pressure. In such a case, an end-inspiratory occlusion is required to measure plateau pressure. Pplat should ideally be kept at less than 30 cmH2O, or even lower in patients with ARDS. This assumes that chest wall compliance (CCW) is normal, whereas higher Pplat may be safe if CCW is decreased.
End-inspiratory occlusion pressure
In patients with normal lungs, an end-inspiratory occlusion of at least 0.5 seconds allows for an accurate measurement of PPLAT. However, in patients with associated lung inhomogeneity, a longer end-inspiratory occlusion of up to 5 seconds is required to reach a plateau. This long end-inspiratory occlusion must be performed manually. An end-inspiratory occlusion produces an immediate drop in peak airway pressure (PPEAK) down to a lower initial pressure (P1). Then, pressure continues to decline gradually—even after the ventilator valves are closed—to reach a plateau after 3 to 5 seconds (PPLAT) depending on lung mechanics. Maximum resistance, (PPEAK−PPLAT)/flow, is then partitioned into minimum resistance, (PPEAK−P1)/flow, and additional resistance, (P1−PPLAT)/flow. Minimum resistance represents the flow resistance of the airways and the endotracheal tube. Additional resistance represents the viscoelastic behavior or stress relaxation of the pulmonary tissues and decay of flow (pendelluft) among lung units with different time constants. Newer concepts use the expiratory time constant (τE) to provide real-time determinations of Pplat without the need for an end-inspiratory pause maneuver. This is helpful if expiration is not fully passive as in the awake patient.13
Mean airway pressure
The mean airway pressure is the average pressure over the whole ventilatory cycle. Graphically, it is represented by the area below the pressure time curve divided by the ventilatory period (inspiratory time plus expiratory time). During pressure control ventilation, the inspiratory pressure waveform is rectangular, and mean PAW can be estimated as follows:
| Mean PAW = (PIP - PEEP) × (Ti/Ttot) + PEEP |
Using a volume mode, the waveform is triangular, therefore cutting the aforementioned value in half:
| Mean PAW = 0.5 (PIP - PEEP) × (Ti/Ttot) + PEEP |
Many current microprocessor ventilators display mean PAW from integration of the PAW waveform as the average of samples taken over the ventilatory period (eg, every 20 milliseconds). Mean airway pressure is clinically important because Pa O 2 is proportional to mean airway pressure. On the other hand, cardiac output may be inversely proportional to mean airway pressure. Typical mean PAW values for fully ventilated patients are 5 to 10 cmH2O for patients with normal lungs, 10 to 20 cmH2O for patients with airflow obstruction, and 15 to 30 cmH2O for patients with ARDS. Anything that increases airway pressure or the I:E ratio by increasing inspiratory time or decreasing expiratory time increases mean airway pressure.
End-expiratory pressure and auto-positive end-expiratory pressure
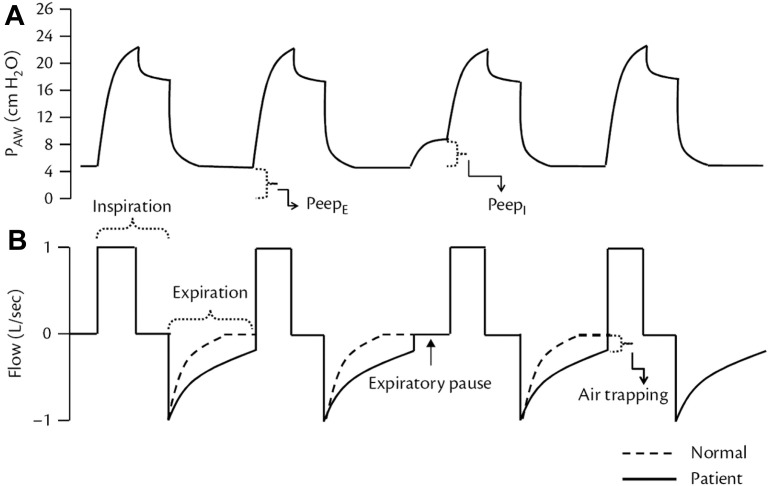
It is generally preferable that expiratory flow should reach zero before the end of expiration (Fig. 3 ). Incomplete emptying of the lungs occurs if the expiratory phase is terminated prematurely. The pressure produced by this trapped gas is called auto-PEEP, intrinsic PEEP, or occult PEEP. Auto-PEEP increases EELV and causes dynamic hyperinflation.14 , 15 Auto-PEEP is measured by applying an end-expiratory pause for 0.5 to 2 seconds or longer. The pressure measured at the end of this maneuver in excess of the PEEP set on the ventilator is defined as auto-PEEP. For a valid measurement, the patient must be relaxed and breathing in synchrony with the ventilator because active breathing during exhalation invalidates the measurement. The end-expiratory pause method can underestimate auto-PEEP when some airways close during exhalation, as may occur during ventilation of the lungs of patients with severe asthma (airway closure). Auto-PEEP reduces VT during PC ventilation and may contribute to ineffective trigger efforts and dyssynchrony. In spontaneously breathing patients, measurement of esophageal pressure (Pes) can be used to determine auto-PEEP (see the following information). Auto-PEEP can be decreased by decreasing minute ventilation (rate or VT), increasing expiratory time TE (decreasing rate or TI), or decreasing Raw (eg, bronchodilator administration). The pressure curve during expiration also reflects the status of the exhalation valve. If the exhalation valve demonstrates significant airflow resistance, the pressure drop at the beginning of expiration will be smooth. If the exhalation valve is leaking, the expiratory pressure will be lower than the set PEEP.
Fig. 3.
The occurrence of intrinsic PEEP can be observed on the screen of the ventilator from a patient with obstructive lung disease. (A) Airway pressure waveform with an expiratory pause showing the existence of PEEPI (or auto-PEEP). (B) Flow curve demonstrating the failure to exhale all gas during the expiratory time and, consequently, formation of gas trapping.
(In: Cordioli, R and Brochard, L, Respiratory system compliance and resistance in the critically ill, Oxford Textbook of Critical Care, ISBN: 9780198855439.)
Driving pressure
Driving pressure (ΔP) (more accurately, tidal pressure) is the pressure required to overcome elastic forces during tidal inflation of the respiratory system. Driving pressure is calculated as follows:
Driving pressure is one metric of the strain applied to the respiratory system and represents the risk of volutrauma. In retrospective studies on patients with ARDS, there is a clear association between driving pressure (ΔP) and mortality. This clinical observation confirms the assumption that ΔP is a measure of detrimental energy to the lung. The higher the ΔP, the more the MP is applied to the lung. ΔP represents an attractive shortcut to the equation of motion because it combines the variables PPlat and PEEP. PPlat represents total inspiratory forces, and PEEP represents the expiratory one. Increased PPlat is associated with overdistention and an insufficient amount of PEEP with atelectasis and atelectrauma. This is further complicated by the fact that changes in PEEP necessarily increase Pplat. It was shown that survival does not depend on the position of ΔP on the P-V graph, but rather on the absolute value of ΔP.16 , 17
Flow and volume
All current-generation critical care ventilators monitor flow. Note that, by convention, expiratory flow is negative and inspiratory flow is positive. The most frequently used airflow measurement is the Fleisch pneumotachograph, which is the gold standard for flow evaluation in respiratory mechanics research.18 Thermal cooling (or hot wire) pneumotachographs estimate V from the amount of heat loss as gas flows across the device, applying the principle of thermal convection. As is the case with PAW measurements, most ventilators measure I at the inspiratory valve and E at the expiratory valve rather than at the airway.
Inspiratory Flow
Peak inspiratory flow I depends on the following factors: the pressure gradient, driving flow, and the inspiratory resistance.
During volume control ventilation, I is set on the ventilator. During passive pressure control ventilation, flow is the pressure applied to the airway, Raw, and , where P is the pressure applied to the airway above PEEP, t is the elapsed time after initiation of the inspiratory phase, and e is the base of the natural logarithm.
Expiratory Flow
Expiratory flow (VE) is normally passive and is determined by Palv, Raw, the elapsed time since initiation of exhalation, and .
End-expiratory flow is present if Raw is high and expiratory time TE is not sufficient, indicating the presence of air trapping (auto-PEEP). It may be of value to determine whether auto-PEEP is due to flow limitation. If pushing on the abdomen results in no additional expiratory flow, flow limitation is present. The presence of missed triggers and flow limitation suggests that PEEP might effectively counterbalance auto-PEEP. Notching in the expiratory flow waveform suggests the presence of missed trigger efforts.19
Tidal Volume
Most ventilators do not measure volume directly but derive this from the integration of flow. Because flow is usually not measured directly at the proximal airway, volume output from the ventilator is less than the volume delivered to the patient. Modern ventilators correct volume for circuit compression, which can be as much as 0.5 to 1.5 mL/cmH2O.
Derived measurements
For an easy assessment, the ventilator is set in the volume-controlled mode with a constant inspiratory flow pattern. Care must be paid to minimize the patient's own respiratory efforts, which would invalidate or complicate these measurements. The relationship between respiratory mechanics and lung volume/volume-derived indices critically depends on the presence of aeration loss from predicted FRC: CRS reflects end-expiratory aerated volume, DP roughly measures dynamic strain, and PEEP-induced alveolar recruitment can be detected at the bedside by changes in these two parameters only if aerated lung volume is lower than the individual predicted FRC.20
Measurement of Compliance
Compliance measurements are key to ventilator management in patients with and without lung disease. Respiratory system static compliance (Crs) is the pressure burden exerted on the lung for any volume change. It is calculated as change in volume over the change in pressure as follows:
| Crs = ΔV/ΔP =VT/Plateau Pressure (Pplat) − PEEP. |
The normal respiratory compliance is in the range of 50 to 70 mL/cmH2O.
Static compliance is relatively simple to measure, but it has some limitations:
Most importantly, it does not distinguish between the lung and chest wall.
Next, it is measured at just one VT and assumes the respiratory system to be a single compartment, which may underestimate the complexity of regional compliances in clinical situations with great inhomogeneity, for example, ARDS. The distinction between the two requires measuring the intrathoracic (or pleural) pressure. Even in the absence of a direct measurement, a significant contribution of the chest wall to a low CRS can be suspected in the presence of morbid obesity, abdominal distention, tight chest bandages, and large pleural effusions.21
Dynamic compliance (DyCrs) is similar; however, it incorporates airway resistance (Raw) within the calculated value. For the single-compartment model of the respiratory system, CSTAT = CDYN and is independent of the respiratory rate. For a multiple-compartment model of the lungs, as the distribution of resistance and compliance becomes less homogeneous, CSTAT becomes greater than CDYN because flow persists among lung units with different mechanical properties (pendelluft), and this flow increases ΔP for the same ΔV. In this case, C decreases as the respiratory rate increases. Optimal compliance demonstrates ideal distending pressures, alveolar recruitment, V/Q matching, homogeneity, and therefore prevention of VILI. Static compliance values are relative to each patient and circumstance, often between 40 and 80 mL/cmH2O. The crucial factor is ensuring each patient's best and optimal compliance. Recruited open lungs are compliant, whereas atelectatic or overinflated lungs are not. Plateau pressure may also be trended as a surrogate of compliance when calculations are not expedient. It is determined by the compliance of the lungs and chest wall.
Chest Wall Compliance
To calculate CCW, changes in Pes (Ppl) are used during passive inflation: CCW =V/P= VT/Pes.
The normal CCW is 200 mL/cmH2O and is decreased in morbid obesity, abdominal compartment syndrome, chest wall edema, chest wall burns, and thoracic deformities (eg, kyphoscoliosis). CCW is also decreased with an increase in muscle tone (eg, a patient who is asynchronous with the ventilator). CCW is increased with flail chest and paralysis.
Lung Compliance
To calculate lung compliance (CL), the change in PL when the lungs are inflated is used:
| CL = V/P= VT/PL. |
The normal CL is 150 to 200 mL/cmH2O.22 The variability of CL may be related to the surface tension of alveoli and the viscosity of lung tissue. CL is decreased with ARDS, cardiogenic pulmonary edema, pneumothorax, consolidation, atelectasis, pulmonary fibrosis, pneumonectomy, bronchial intubation, and overdistention. CL is increased with emphysema.
Airway resistance
Raw reflects the resistance to ventilation imposed by the airways as well as by the endotracheal tube. However, the resistance of endotracheal tubes is known, and one can use this knowledge23 and clinical judgment (presence of secretions within the tube as seen in the flow curve) in estimating the airway-versus-equipment contribution onto Raw. The resistance imposed by the ventilator circuit (generally minimal) is not part of this measurement. Raw is affected by flow, lung volume, and the phase of respiration. Resistance forces disappear before reaching the deep lung and are not present in alveoli, therefore not contributing to the risk of VILI.
During volume control ventilation, RI can be estimated from the PIP, Pplat, and end-inspiratory flow:
| RI = (PIP - Pplat)/ V̇I |
RE can be estimated from the VEXH and the difference between Pplat and PEEP:24
| RE = (Pplat - PEEP)/ V̇EXH |
Common causes of increased Raw are bronchospasm, secretions, and a small inner diameter endotracheal tube. For intubated and mechanically ventilated patients, RI should be less than 10 cmH2O/L/s. Inspiratory Raw is typically lower than expiratory Raw owing to the increased diameter of the airways during inspiration, particularly in patients with chronic obstructive respiratory disease (COPD) and dynamic hyperinflation.25
The PIP-Pplat differential can offer a surrogate indicator of inspiratory Raw during rapid assessment. Raw can also be estimated from the time constant (τ = R × C) of the respiratory system. This method permits to calculate both inspiratory and expiratory Raw. Raw cannot be determined when using variable flow modes such as pressure control ventilation. Flow/time, volume/time, and pressure/time waveforms may demonstrate a failure to reach baseline, indicating high expiratory Raw, air trapping, dynamic hyperinflation, and/or auto-PEEP. The lowest Raw will often coincide with optimal compliance and PEEP. In the heterogeneous obstructive lung, optimal PEEP will splint airways, improve the distribution of ventilation, diminish air trapping, prevent auto-PEEP, and yield the lowest expiratory Raw.15 , 26 Removing externally set PEEP to decrease air trapping in obstructive lung disease is an erroneous technique not in-line with current empiric data. Raw may be minimized through optimal PEEP, treating airway abnormalities such as secretions or bronchospasm, and ensuring a patent airway.
Measurement of dead space
Approximately 33% of each breath does not participate in gas exchange, termed dead space (VDS) and averaging 2 mL/kg of ideal body weight. Dead space is divided into 2 categories: anatomic and alveolar. Together, the two encompass the physiologic (total) dead space and are measured in ratio to the VT (VDS/VT). Applying the Enghoff modification of the Bohr equation, VDS/VT =Pa co 2 – PeCO2/Paco 2, values for dead space fraction may be estimated. Although not technically equivalent, the end-tidal carbon dioxide (ETCO2) may act as a surrogate for PeCO2. CRS and alveolar dead space have been previously proposed to optimize the PEEP setting during surgery.27 Trending VDS/VT allows ventilation titration for the best possible V/Q matching and gas exchange. Intrapulmonary shunt measurements can offer additional data on oxygenation and V/Q mismatch, but shunt calculation is complex. Alveolar-to-arterial gradients from a blood gas analysis can act as a substitute (A/a gradient = Pao 2 – Pao 2). Confirming optimal Crs, DyCrs, Raw, VDS/VT, and gas exchange is the key facet in providing optimal mechanical ventilation, which in turn minimizes the risk of postoperative pulmonary complications (PPCs) and VILI.
Extended monitoring
Airway flow and pressure curves display the complex interaction between the ventilator settings and the patient's respiratory mechanics. Pressure, volume, and flow curves displayed by the ventilator are nothing more than graphical representations of the equation of motion.
P-V Loops
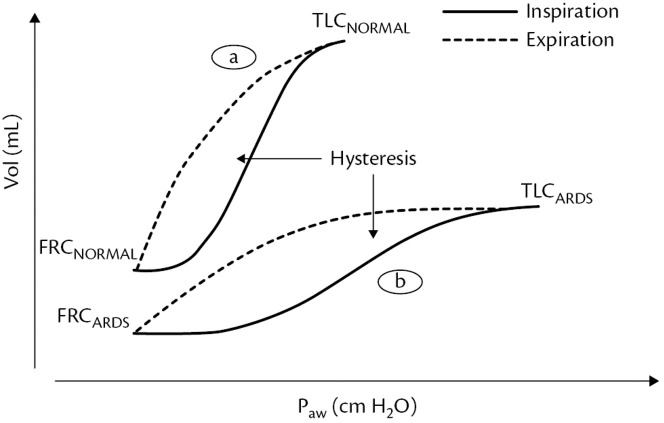
The P-V relation as displayed by the P-V loop is linear in a normal lung that is fully aerated at the beginning of the maneuver. This means that the compliance remains constant throughout inflation. The slope of the P-V curve is CRS. Deflation is also linear. There is a small degree of physiologic hysteresis (area between the inflation and deflation limbs of the P-V loop) that occurs owing to the viscoelastic property of tissues. In a patient with early-onset ARDS, the shape of the P-V loop may differ compared with that of a patient with normal lungs (see Fig. 4). The inflation and deflation limbs demonstrate a change in slope, which means that the respiratory system compliance varies at different levels of pressure. In addition, hysteresis is greater than in patients with normal lungs owing to recruitment occurring during inflation and derecruitment occurring during deflation. Recruitment occurs at a higher pressure than derecruitment. Therefore, a quasi-static P-V loop can be used to assess the potential for recruitment and predict the effect of a recruitment maneuver. The larger the volume difference between inflation and deflation, the higher the potential for recruitment. Hysteresis is quite awkward to calculate but can easily be estimated using the volume difference between inflation and deflation measured at 20 cmH2O of pressure. If the volume difference is higher than 500 mL, it means there is high potential for recruitment. A number of issues preclude routine use of P-V curves to set the ventilator in patients with ARDS.28 Correct interpretation of the P-V curve during nonconstant flow ventilation (eg, pressure control ventilation) and with higher VI is problematic. Measurement of the P-V curve requires deep sedation, and often paralysis, to correctly make the measurement. Chest wall mechanics potentially affect the shape of the P-V curve, necessitating Pes measurement to separate lung from chest wall effects. As with most measures of respiratory mechanics, the P-V curve treats the lungs as a single compartment, disregarding the inhomogeneity of the lungs of patients with ARDS. Performing subsequent P-V curves and measuring lung volume corresponding to different PEEP levels can be used to assess PEEP-induced lung recruitment.29 , 30 Lung recruitment at a given airway pressure is observed as the difference in lung volume between P-V curves starting at different lung volumes corresponding to different levels of PEEP (Fig. 4 ).
Fig. 4.
Pressure-volume curve (P/V) and its hysteresis. (a) P/V curve from a normal patient. (b) P/V curve from a patient with ARDS.
(In: Cordioli, R and Brochard, L, Respiratory system compliance and resistance in the critically ill, Oxford Textbook of Critical Care, ISBN: 9780198855439.)
-V Loops
-V loops are displayed with flow as a function of volume. Analysis of the -V loop may be helpful for identifying flow limitation during expiration and bronchodilator response.31 The inability to reach zero flow indicates that exhalation ends at a lung volume higher than FRC, which exerts an auto-PEEP (PEEPi). The -V curve can provide an indication of excessive secretions more reliably than clinical examination, with the presence of excessive secretions in the airways producing a sawtooth pattern on both the inspiratory and expiratory -V curves32 -V loops are also useful in the detection of air leaks that cause a loss of volume with each breath, as well as a difference between the delivered and the exhaled VT. Air leaks can occur all the way from within the ventilator system, between the ventilator and the patient and within the patient, for example, when a bronchopleural fistula is present. Regardless of their location, air leaks cause a characteristic failure of the V̇-V loop to close at the end of expiration because a portion of the inspired VT does not return to the site of measurement on the expiration side (Fig. 5 ).
Fig. 5.
Flow-volume curves in different situations. (A) Normal patient; (B) patient with COPD and dynamic hyperinflation and auto-PEEP and after bronchodilator treatment; (C) sudden interruption of exhalation flow representing an important gas leak from the patient (bronchopleural fistulae) or from the ventilator circuit; (D) a sawtooth pattern is observed in both inspiratory and expiratory limbs and indicates the presence of secretions in the airways.
(In: Cordioli, R and Brochard, L, Respiratory system compliance and resistance in the critically ill, Oxford Textbook of Critical Care, ISBN: 9780198855439.)
Best positive end-expiratory pressure ventilation
An optimal PEEP is a pressure at end-expiration, which should be set to an end-expiratory pressure that prevents injurious energy expenditure to the lung. Providing PEEP levels sufficient to maintain alveolar recruitment is instrumental in protective ventilation. All other parameters and settings rely on appropriate levels of PEEP to ensure adequate ventilation, FRC, and recruitment. If PEEP exceeds the level required to stabilize the lungs, overdistension may result, despite the use of low VT. Thus, moderate levels of PEEP in noninjured lungs may represent a compromise between cyclic overdistension and closing/reopening of lung units. The optimal PEEP to apply remains debated. Ventilation with higher PEEP (10–12 cmH2O) may be without clinical benefit because studies so far have shown no protection against development of PPCs, suggesting that the optimal setting has a wide intersubject variability. This might be attributable to its two-edged nature: PEEP generates overinflation with lung injury in already open alveoli (ie, static strain) but lowers dynamic strain when it is effective in recruiting new compartments.20 There are arguments against use of higher PEEP owing to its effect on circulation and the need for intraoperative administration of vasoactive drugs.7
What is the optimal positive end-expiratory pressure and how is it determined?
PEEP must be individually determined for each patient. The lowest sum of collapse and overdistension will be the optimal PEEP, which will lead to the least lung injury. Basic PEEP trial methods involve monitoring compliance and/or its surrogates (PIP, Pplat) with progressive changes in PEEP. During PEEP trials, DyCrs has proved to be a particularly valuable indicator, accounting for changes in Raw in addition to Crs. Alveolar-to-arterial gradient, oxygenation, and VDS/VT will generally improve in conjunction with compliance and resistance, indicating optimal settings. Suter and colleagues33 discovered that the best Crs coincided with maximum oxygen transport and the lowest VDS/VT. An increase in PIP/Pplat greater than the change in PEEP would indicate a drop in compliance, overdistention, and increased risk of VILI. For patients being ventilated with pressure control ventilation, the anesthesia provider may use a similar extrapolation in DyCrs or Crs, noting changes in the delivered VT for a set PIP and/or ΔP. Because pressure is fixed, VT changes are indicative of changes in compliance, recruitment, and FRC in contrast to PIP/Pplat changes during volume ventilation. Optimal PEEP promotes these goals without compromising hemodynamics; otherwise, oxygen delivery would be curtailed.
The most relevant clinical consequence of these considerations is that CRS and DP are the best available bedside tools to monitor aeration loss, dynamic strain, and PEEP-induced recruitment.
Intrathoracic or pleural pressure
Pleural pressure (PPL) is estimated by measuring the pressure in the lower third of the esophagus using an esophageal balloon catheter.11 , 34, 35, 36 Esophageal pressure (PESO) is used as a surrogate of pleural pressure to estimate CL and CCW, to quantify auto-PEEP and WOB during assisted modes of ventilation, and to evaluate the degree of diaphragmatic dysfunction. The measurement of PPL is of great value in the assessment of respiratory mechanics and can be used by the experienced clinician at the bedside.11
Transpulmonary pressure and esophageal pressure
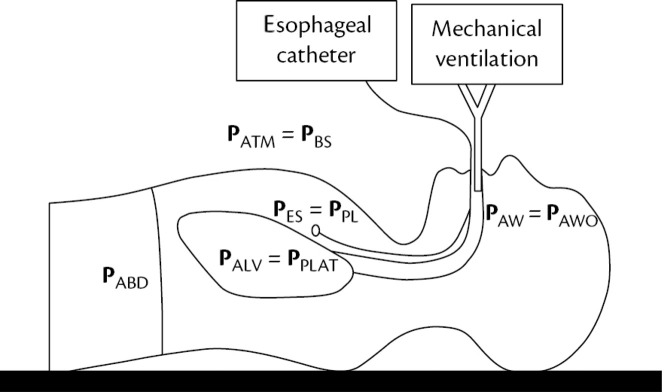
Transpulmonary Pressure (PTP) is the difference between the pressure inside the alveoli (PALV) and the pressure surrounding the lung (pleural pressure, PPL) (Fig. 6 ):
| PTP = PALV - PPL |
Fig. 6.
The main different pressures involved in pulmonary mechanics.
(In: Cordioli, R and Brochard, L, Respiratory system compliance and resistance in the critically ill, Oxford Textbook of Critical Care, ISBN: 9780198855439.)
As in vivo measurements of PALV and PPL are not feasible, PALV is usually assumed to be approximately equal to the static pressure at the airway opening (PPLAT, PEEP) and PPL is assumed to be equal to the esophageal pressure (PESO). PTP is usually described as the distending pressure of the lungs because it best describes the sum of the interactions of PPL and PALV across the lungs. Because PESO can be elevated in the setting of ARDS, obesity, or increased intraabdominal pressure, the use of PTP allows the titration of positive pressure based on the actual pressure applied to the lung. To allow optimal recruitment, PEEP could be increased until PTP becomes positive at end-expiration, to keep airways and alveoli open during the tidal cycle.36 End-inspiratory PTP is evaluated by calculating the PPLAT – PESO difference during an inspiratory hold. It is useful in the assessment of safety pressure thresholds: an end-inspiratory PTP lower than 20 cmH2O is generally considered safe.36
For the lung, only transpulmonary pressure (PL = Ppl – Palv, with Ppl being the intrapleural pressure) determines alveolar distention. Intrapleural pressure is not easily accessible, besides being unequally distributed throughout the lung. As a surrogate, esophageal pressure (Pes) can be used as an estimate of mean Ppl.37 This minimally invasive approach can be used to estimate PL, which is particularly advantageous in cases of a decreased extrathoracic compliance such as obesity or an elevated intraobdominal pressure. In the supine position, the weight of the mediastinum and abdominal pressure both increase Pes. Some studies have pointed out that Pes is higher than Ppl by about 5 cmH2O owing to the effects of body position and mediastinum.38 Pes can represent the average level of Ppl, and Pes and Ppl have a good correlation. For making sure Pes reflects Ppl accurately, the technical aspects of Pes measurement are important. It usually includes the characteristics of the esophageal manometers, placement position, balloon inflating volume, and data interpretation. In order to ensure the accuracy of Pes, its positioning needs to be tested by an occlusion test. In some cases, such as obesity, thoracic, or abdominal disease, Paw cannot reflect actual pressure gradient over the lung because the higher percentage of Paw is used to overcome the elastance of the chest wall. The monitoring of pleural pressure (Ppl) or esophageal pressure (Pes) may help to distinguish the pressure gradients acting on the lung and chest wall. On the other hand, when the patient makes spontaneous breathing efforts, the inspiratory muscles and the ventilator both participate in breathing activity during assisting ventilation mode. The pressure inflating the lung comes from the pressure generated by the ventilator and the patient's inspiratory muscles. However, excessive breathing effort also can induce spontaneous patient self-inflicted lung injury.39 Therefore, it is very important to monitor spontaneous breathing efforts and to balance the relationship between mechanical ventilation and spontaneous breathing effort during assisted ventilation. The Monitoring of Pes, intragastric pressure (Pga), and a series of parameters derived from Pes and Pga can be used to quantitatively assess spontaneous breathing efforts.
Choice of positive end-expiratory pressure using esophageal pressure
For the choice of PEEP levels, airway pressure Paw is primarily chosen. This, however, rather represents pressure at the artificial airway or the ventilator, respectively. Paw is easily displayed and measures the mechanical properties of the whole respiratory system including the artificial airway. Therefore, only during zero-flow conditions (end-inspiratory pause, end of expiration, or during occlusion) is Paw equal to tracheal pressure (Ptrach) and alveolar pressure (Palv). Initially used in ARDS to choose the right PEEP value, several studies have shown promising results in terms of improved oxygenation and compliance using Pes. Surprisingly, looking at the results, mortality and other end points such as measurements of PEEP, PTP, Paw, driving pressure (ΔP = PPlat-PEEP), and Pa o 2/FiO2 ratio were not significantly different.36 , 40 This may be due to the negative hemodynamic effects of too much PEEP on oxygen delivery. There are potential sources of error in the use of Pes to estimate Ppl. It is important to appreciate that Pes estimates Ppl mid-thorax. Ppl is more negative in the nondependent thorax and more positive in the dependent thorax. The weight of the heart can bias the Pes by as much as 5 cmH2O.41 Transpulmonary pressure (PTP) is the difference between pressure measured at the mouth and esophageal (pleural) pressure. During no flow (inspiratory or expiratory pause maneuvers), PTP becomes the alveolar distending pressure. In this article, the assumption is that PTP is measured under static conditions and thus represents alveolar distending pressure. The ventilator should be set to avoid a negative PTP during exhalation (contributing to cyclical opening and closing injury) and to avoid excessive PTP at the end of inspiration (overdistention).
Compliance of lungs and chest wall
Pes is required to separate CL and CCW. Measurement of end-inspiratory PESO (PESO,i) and end-expiratory PESO (PESO,e) allows to distinguish the two components of CRS: CCW and CL.
or
The partitioning of the elastic properties of the respiratory system is useful to understand whether a low CRS as a whole might be due to low CL, low CCW, or both. Impairment of the elastic properties of the chest wall, as in morbid obesity, pneumoperitoneum, or increased intra-abdominal pressure, gives us an indication that additional pressure needs to be applied to the lung and transmitted to the pleural space in order to achieve adequate thoracic inflation.29 As the chest wall becomes stiffer, the proportion of PAW that is spent for lung distention becomes smaller. The degree of stiffness of the chest wall (low CCW) must be taken into account when titrating PAW to recruit the lung of ARDS patients because higher PAW levels might be needed to achieve the desired recruitment, without the same risk of injuring the lung. Using PTP rather than PPLAT as a target of PAW titration has been shown to be safe and effective.30
Functional residual capacity and end-expiratory lung volume
FRC represents the unstressed volume at the end of lung expiration. In critically ill patients receiving mechanical ventilation and different levels of PEEP, it is better to speak of the EELV. Calculation of EELV is based on a step change in FiO2 and the assumption that N2 is the balance gas. Baseline determination is made of end-tidal N2 (FETN2). It is assumed that oxygen consumption and carbon dioxide (CO2) production remain constant throughout the measurement. A 20% step change in FiO2 then occurs, and the EELV is calculated as follows: VN2/FETN2, where FETN2 is the change following the step change in FiO2. The breath-to-breath changes are calculated over 20 breaths. The EELV measurement is implemented in some modern ventilators. For this application, nitrogen concentration in inhaled and exhaled gas is not directly measured but estimated from the end-tidal concentrations of oxygen and CO2.42, 43, 44
Why measure lung volumes during invasive ventilation?
The use of EELV during PEEP titration would seem attractive. An increase in PEEP will always increase EELV, and respiratory system compliance can predict the amount of volume it would increase. A PEEP-induced increase in EELV might be the result of recruitment, or it might be the result of overdistention of already open alveoli. Therefore, both PEEP and EELV might contribute to lung strain, and EELV by itself might not be useful to assess PEEP response.
Electrical impedance tomography
Electrical impedance tomography (EIT) is a noninvasive and radiation-free imaging technique that uses a minimum of 16 to 32 leads positioned around the thorax. EIT provides breath-by-breath dynamic imaging of regional ventilation distribution through measuring impedance changes across lung regions; such information cannot be obtained by global monitoring (eg, airway pressure, flow monitoring). EIT can detect the changes in lung impedance associated with a recruitment maneuver and an incremental or decremental PEEP trial, enabling the identification of the PEEP level at which tidal ventilation is most homogeneous.45 , 46 EIT combined with lung mechanics can avoid hyperinflation because of excessive PEEP, which may be reached if PEEP is titrated on the basis of gas exchange alone.47 Indeed, high impedance associated with reduced compliance is the hallmark of changes suggestive of hyperinflation, whereas low impedance associated with reduced compliance is suggestive of collapse.48, 49, 50 In patients with severe ARDS, EIT-guided PEEP titration has been associated with improved oxygenation, compliance, driving pressure, and weaning success rate.51
Mechanical ventilation in special subsets of patients
Spontaneously Breathing Patient
In spontaneously breathing patients, the pressure generated by the patient's muscle (PMUS) is added to the pressure applied by the ventilator:
There are two implications of this equation:
-
•
First is that for PC modes, increasing PMUS does not affect PAW (because this is preset), but it increases volume and flow (ie, it deforms the volume and flow curves). For volume controlled modes, increasing PMUS decreases PAW (ie, it deforms the pressure curve), but it does not affect volume or flow (because they are preset).
-
•
Second, it follows that PMUS must exceed PEEPTOT in order for PAW to drop (or flow to increase) enough to trigger inspiration. Otherwise a patient-ventilator asynchrony occurs, which is known as an ineffective trigger effort.
Increased Abdominal Pressure
Intra-abdominal pressure is the steady-state pressure in the abdominal cavity.52 The normal intra-abdominal pressure is 5 mm Hg; it increases during inhalation with diaphragmatic contraction. Direct measurement of intraperitoneal pressure is not practical, so the bladder method is thus most commonly used for intermittent intra-abdominal pressure measurement.53 Intra-abdominal pressure should be measured at the end of exhalation in the supine position, ensuring that abdominal muscle contractions are absent. In mechanically ventilated patients, an increase in intraabdominal pressure results in decreased CRS with flattening and a rightward shift of the P-V curve of the respiratory system. These changes are due to decreased CCW, whereas CL remains unchanged. In deeply sedated patients with ARDS, the diaphragm behaves as a passive structure, and thus moves upward in the rib cage, transmitting increased intra-abdominal pressure to the lower lobes of the lung, where it causes compression atelectasis. Surgical abdominal decompression recruits lung volume and increases Pao 2/Fio2. Upright positioning increases intra-abdominal pressure and decreases CRS, suggesting that this position might result in a deterioration of respiratory function in patients with intra-abdominal hypertension.54
Pneumoperitoneum
Insufflation of CO2 into the abdomen in the context of minimally invasive surgery leads to significant changes to the mechanical characteristics of the respiratory system. On the one hand, thoracic compliance changes with cranial shift of the diaphragm; on the other hand, compressive atelectasis leads to a reduction in EELV. Conventional monitoring, for example, of parameters relating to respiratory mechanics as provided by the mechanical ventilator (plateau and peak pressures, compliance), is not suitable for quantifying these two (lung/thorax) mechanical components. As such, faced with an increase in plateau pressure during otherwise unchanged volume-controlled ventilation, the anesthesiologist will be unable to discern whether this is solely the result of CO2 insufflation into the abdomen with a subsequent change in thoracic compliance or whether it instead represents an actual mismatch between VT and EELV, causing increased strain and potentially lung damage. Strict limitation of those pressures cannot as a concept simply be applied to the setting of minimally invasive surgery. One option to estimate pressure acting directly on the lungs is to measure transpulmonary pressure (see the aforementioned information). To date, however, there is no evidence that adjusting ventilation parameters, and especially PEEP, in accordance with transpulmonary pressure measurements can avoid PPCs. The increase in arterial CO2 caused by transperitoneal resorption of insufflated gas can require a significantly increased respiratory minute volume. Increasing alveolar ventilation primarily by increasing VTs may cause VILI. Increasing the respiratory rate is a useful measure but may, in the context of increased airway resistance in the setting of pneumoperitoneum, limit expiratory flow. Higher peak airway pressures do not necessarily reflect the pressure at the level of the alveoli and may instead be primarily associated with higher airway resistance in the face of increased flow. The aggressivity with which these respiratory measures are pursued can be tempered, however, if moderate hypercapnia based on pH is tolerated.
Chronic Obstructive Respiratory Disease/Asthma
Patients with obstructive lung disease cannot generate a normal expiratory flow. Normally, lung volume returns to the relaxed volume at the end of passive expiration. This relaxed lung volume is defined as FRC during spontaneous breathing. Dynamic hyperinflation occurs whenever the respiratory system cannot generate the necessary expiratory flow near FRC. In patients with increased expiratory resistance due to airflow obstruction, the EELV may increase higher than the predicted FRC. Along with the increase in EELV, end-expiratory alveolar pressure increases, which is called intrinsic PEEP.55 Mechanical causes of dynamic hyperinflation include an increased expiratory resistance resulting in a longer time constant and a reduced lung elastance which results in a decreased expiratory driving pressure. Expiratory flow limitation is defined as the inability of augmentation of expiratory flow regardless of an increased expiratory driving pressure.56 The consequences of hyperinflation and intrinsic PEEP are an increased inspiratory threshold during assisted mechanical ventilation. Patients have to generate an additional pleural pressure to counterbalance the intrinsic PEEP to be able to trigger the ventilator. This can be considered wasted energy cost of breathing because inspiratory muscle contraction to counterbalance intrinsic PEEP does not generate inspiratory flow. The increased inspiratory muscle load results in muscle fatigue and patient-ventilator asynchrony57, 58, 59, 60 and leads to an increasing pulmonary vascular resistance and right ventricular failure.
By conducting end-expiratory occlusion, static intrinsic PEEP can be measured. In combination with end-inspiratory occlusion, the static compliance of the respiratory system can be obtained. In patients with dynamic hyperinflation and intrinsic PEEP, the calculation of static compliance should be calibrated by intrinsic PEEP; otherwise, the true compliance will be underestimated.61 Dynamic hyperinflation-induced elevation of EELV can be measured at the bedside using release and prolonged expiration maneuver.62 No matter what ventilation model is selected, monitoring of expiration should be performed periodically to allow lung emptying. For patients with severe asthma, a relatively small VT and a higher inspiratory flow have been recommended to preserve expiratory time and minimize dynamic hyperinflation. To counterbalance intrinsic PEEP during ventilator triggering, PEEP may be applied in patients with COPD. The PEEP level is usually set as 80% of baseline intrinsic PEEP, which has been explained by the waterfall analogy.55
Adult Respiratory Distress Syndrome
In a heterogeneous lung in patients with ARDS, the local strain (lung tissue deformation or volume change) and the transpulmonary pressure (airway pressure-pleural pressure) are different in different locations.8 , 63 In patients with ARDS, high inspiratory and expiratory resistance as well as low compliances of the baby lung are the leading pathologies. Assuming that all alveoli are open, plateau pressure (Pplat) reflects end-inspiratory alveolar pressure.64 A short end-inspiratory occlusion (0.3 seconds) is sufficient to estimate injurious pressure applied to the alveoli, especially in passive breathing. Nowadays, maintaining Pplat at or lower than 25 cmH2O as used in a large trial seems to be a reasonable and safe threshold. At end-inspiration, PTP as derived by the insertion of an esophageal balloon is a more reliable measurement of the distending pressure of the lung because Pplat also depends on pleural pressure (Ppl). In some patients, Ccw is responsible for almost 50% of the CRS, whereas in other cases, it is only about 15% to 20%.65 Separation of these two factors improves ventilator settings and outcomes. PTP at end-expiration (PTP,ee) is the pressure distending the lungs at end-expiration. Negative PTP,ee values are common in ARDS, potentially favoring cyclic reopening and closing of alveoli during ventilation and atelectrauma.37 Setting PEEP to a positive PTP,ee was associated with improved physiologic parameters.36 A ΔP value higher than 14 to 15 cmH2O was independently associated with higher mortality.16 An optimal PEEP is essential in ARDS and has been discussed previously. Excluding lung imaging methods, the most common methods for the PEEP setting is the use of respiratory mechanics. Best compliance methods or the ARDS PEEP table are commonly used. Airway closure at end-expiration is another feature of ARDS, and close attention needs to be paid to the end-expiratory flow curve as well as esophageal pressures for the detection of airway closure. With the development of newer technologies, EIT has been used to select the optimal PEEP and may actually be the simplest and most promising way at the bedside. This method can be used to calculate the percentage of overdistention and collapse.
Summary
Contemporary management of the ventilated patient still relies on the measurement of old parameters such as airway pressures and flows. Graphical presentations—in particular of the pressure and flow curves at different locations—reveal the intricacies of patient-ventilator interactions. This increases the complexity of measurements and opens a new pathway for several bedside technologies based on basic physiologic knowledge. The spread of COVID-19 has confronted the anesthesiologist and intensivist with one of the most severe pulmonary pathologies of the last decades. Optimizing the patient's respiratory mechanics at the bedside is a valuable skill for the physician in the ICU, which is further refined and supported by mobile technologies such as lung ultrasound and electrical impedance tomography.
Clinics care points
-
•
Always evaluate the pressure-time and flow-time curves of the patient in combination with your clinical assessment.
-
•
Use additional imaging modalities such as radiological tests, ultrasound, and EIT.
-
•
Pressure-volume curves provide insight into compliance and recruitability.
-
•
Keep safe airway pressures in mind: ideally Pplat less than 30 cmH2O, or even lower in patients with ARDS, and driving pressure less than 15 cmH2O.
-
•
ΔP is a measure of detrimental energy to the lung and represents an attractive shortcut to the equation of motion because it combines the variables PPlat (inspiratory forces) and PEEP (expiratory forces). Survival does not depend on the position of ΔP on the pressure-volume graph, but rather on the absolute value of ΔP.
-
•
Mean airway pressure is clinically important because Pa o 2 is proportional to mean airway pressure and inversely proportional to cardiac output. Typical mean PAW values for fully ventilated patients are 5 to 10 cmH2O for patients with normal lungs, 10 to 20 cmH2O for patients with airflow obstruction, and 15 to 30 cmH2O for patients with ARDS.
-
•
Optimal compliance demonstrates ideal distending pressures, alveolar recruitment, V/Q matching, and homogeneity and therefore prevents VILI.
Acknowledgments
Disclosure
The author has nothing to disclose.
References
- 1.Slutsky A.S., Ranieri V.M. Ventilator-induced lung injury. N Engl J Med. 2013;369(22):2126–2136. doi: 10.1056/NEJMra1208707. [DOI] [PubMed] [Google Scholar]
- 2.Tonetti T., Vasques F., Rapetti F., et al. Driving pressure and mechanical power: new targets for VILI prevention. Ann Transl Med. 2017;5(14):286. doi: 10.21037/atm.2017.07.08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gattinoni L., Tonetti T., Cressoni M., et al. Ventilator-related causes of lung injury: the mechanical power. Intensive Care Med. 2016;42(10):1567–1575. doi: 10.1007/s00134-016-4505-2. [DOI] [PubMed] [Google Scholar]
- 4.Cressoni M., Gotti M., Chiurazzi C., et al. Mechanical power and development of ventilator-induced lung injury. Anesthesiology. 2016;124(5):1100–1108. doi: 10.1097/ALN.0000000000001056. [DOI] [PubMed] [Google Scholar]
- 5.Collino F., Rapetti F., Vasques F., et al. Positive end-expiratory pressure and mechanical power. Anesthesiology. 2019;130(1):119–130. doi: 10.1097/ALN.0000000000002458. [DOI] [PubMed] [Google Scholar]
- 6.Zhang Z., Zheng B., Liu N., et al. Mechanical power normalized to predicted body weight as a predictor of mortality in patients with acute respiratory distress syndrome. Intensive Care Med. 2019;45(6):856–864. doi: 10.1007/s00134-019-05627-9. [DOI] [PubMed] [Google Scholar]
- 7.Hol L., Nijbroek S., Schultz M.J. Perioperative lung protection: clinical implications. Anesth Analg. 2020;131(6):1721–1729. doi: 10.1213/ANE.0000000000005187. [DOI] [PubMed] [Google Scholar]
- 8.Chiumello D., Carlesso E., Cadringher P., et al. Lung stress and strain during mechanical ventilation for acute respiratory distress syndrome. Am J Respir Crit Care Med. 2008;178(4):346–355. doi: 10.1164/rccm.200710-1589OC. [DOI] [PubMed] [Google Scholar]
- 9.Grasso S., Stripoli T., De Michele M., et al. ARDSnet ventilatory protocol and alveolar hyperinflation: role of positive end-expiratory pressure. Am J Respir Crit Care Med. 2007;176(8):761–767. doi: 10.1164/rccm.200702-193OC. [DOI] [PubMed] [Google Scholar]
- 10.Terragni P.P., Rosboch G., Tealdi A., et al. Tidal hyperinflation during low tidal volume ventilation in acute respiratory distress syndrome. Am J Respir Crit Care Med. 2007;175(2):160–166. doi: 10.1164/rccm.200607-915OC. [DOI] [PubMed] [Google Scholar]
- 11.Benditt J.O. Esophageal and gastric pressure measurements. Respir Care Jan. 2005;50(1):68–75. [discussion 75-7] [PubMed] [Google Scholar]
- 12.Carvalho A.R., Spieth P.M., Pelosi P., et al. Ability of dynamic airway pressure curve profile and elastance for positive end-expiratory pressure titration. Intensive Care Med. 2008;34(12):2291–2299. doi: 10.1007/s00134-008-1301-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Al-Rawas N., Banner M.J., Euliano N.R., et al. Expiratory time constant for determinations of plateau pressure, respiratory system compliance, and total resistance. Crit Care. 2013;17(1):R23. doi: 10.1186/cc12500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Marini J.J. Dynamic hyperinflation and auto-positive end-expiratory pressure: lessons learned over 30 years. Am J Respir Crit Care Med. 2011;184(7):756–762. doi: 10.1164/rccm.201102-0226PP. [DOI] [PubMed] [Google Scholar]
- 15.Blanch L., Bernabe F., Lucangelo U. Measurement of air trapping, intrinsic positive end-expiratory pressure, and dynamic hyperinflation in mechanically ventilated patients. Respir Care. 2005;50(1):110–123. [discussion 123-4] [PubMed] [Google Scholar]
- 16.Amato M.B., Meade M.O., Slutsky A.S., et al. Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med. 2015;372(8):747–755. doi: 10.1056/NEJMsa1410639. [DOI] [PubMed] [Google Scholar]
- 17.Bellani G., Laffey J.G., Pham T., et al. The LUNG SAFE study: a presentation of the prevalence of ARDS according to the Berlin Definition! Crit Care. 2016;20:268. doi: 10.1186/s13054-016-1443-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Giannella-Neto A., Bellido C., Barbosa R.B., et al. Design and calibration of unicapillary pneumotachographs. J Appl Physiol (1985) 1998;84(1):335–343. doi: 10.1152/jappl.1998.84.1.335. [DOI] [PubMed] [Google Scholar]
- 19.Ninane V., Leduc D., Kafi S.A., et al. Detection of expiratory flow limitation by manual compression of the abdominal wall. Am J Respir Crit Care Med. 2001;163(6):1326–1330. doi: 10.1164/ajrccm.163.6.2004150. [DOI] [PubMed] [Google Scholar]
- 20.Fernandez-Bustamante A., Vidal Melo M.F. Bedside assessment of lung aeration and stretch. Br J Anaesth. 2018;121(5):1001–1004. doi: 10.1016/j.bja.2018.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ranieri V.M., Giuliani R., Mascia L., et al. Chest wall and lung contribution to the elastic properties of the respiratory system in patients with chronic obstructive pulmonary disease. Eur Respir J. 1996;9(6):1232–1239. doi: 10.1183/09031936.96.09061232. [DOI] [PubMed] [Google Scholar]
- 22.Faffe D.S., Zin W.A. Lung parenchymal mechanics in health and disease. Physiol Rev. 2009;89(3):759–775. doi: 10.1152/physrev.00019.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rossi A., Gottfried S.B., Higgs B.D., et al. Respiratory mechanics in mechanically ventilated patients with respiratory failure. J Appl Physiol (1985) 1985;58(6):1849–1858. doi: 10.1152/jappl.1985.58.6.1849. [DOI] [PubMed] [Google Scholar]
- 24.Hess D., Tabor T. Comparison of six methods to calculate airway resistance during mechanical ventilation in adults. J Clin Monit. 1993;9(4):275–282. doi: 10.1007/BF02886698. [DOI] [PubMed] [Google Scholar]
- 25.Smith T.C., Marini J.J. Impact of PEEP on lung mechanics and work of breathing in severe airflow obstruction. J Appl Physiol (1985) 1988;65(4):1488–1499. doi: 10.1152/jappl.1988.65.4.1488. [DOI] [PubMed] [Google Scholar]
- 26.Ranieri V.M., Grasso S., Fiore T., et al. Auto-positive end-expiratory pressure and dynamic hyperinflation. Clin Chest Med. 1996;17(3):379–394. doi: 10.1016/s0272-5231(05)70322-1. [DOI] [PubMed] [Google Scholar]
- 27.Maisch S., Reissmann H., Fuellekrug B., et al. Compliance and dead space fraction indicate an optimal level of positive end-expiratory pressure after recruitment in anesthetized patients. Anesth Analg. 2008;106(1):175–181. doi: 10.1213/01.ane.0000287684.74505.49. table of contents. [DOI] [PubMed] [Google Scholar]
- 28.Gattinoni L., Vagginelli F., Chiumello D., et al. Physiologic rationale for ventilator setting in acute lung injury/acute respiratory distress syndrome patients. Crit Care Med. 2003;31(4 Suppl):S300–S304. doi: 10.1097/01.CCM.0000057907.46502.7B. [DOI] [PubMed] [Google Scholar]
- 29.Gattinoni L., Chiumello D., Carlesso E., et al. Bench-to-bedside review: chest wall elastance in acute lung injury/acute respiratory distress syndrome patients. Crit Care. 2004;8(5):350–355. doi: 10.1186/cc2854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sarge T., Talmor D. Targeting transpulmonary pressure to prevent ventilator induced lung injury. Minerva Anestesiol. 2009;75(5):293–299. [PubMed] [Google Scholar]
- 31.Dhand R. Ventilator graphics and respiratory mechanics in the patient with obstructive lung disease. Respir Care Feb. 2005;50(2):246–261. [discussion 259-61] [PubMed] [Google Scholar]
- 32.Jubran A., Tobin M.J. Use of flow-volume curves in detecting secretions in ventilator-dependent patients. Am J Respir Crit Care Med. 1994;150(3):766–769. doi: 10.1164/ajrccm.150.3.8087350. [DOI] [PubMed] [Google Scholar]
- 33.Suter P.M., Fairley B., Isenberg M.D. Optimum end-expiratory airway pressure in patients with acute pulmonary failure. N Engl J Med. 1975;292(6):284–289. doi: 10.1056/NEJM197502062920604. [DOI] [PubMed] [Google Scholar]
- 34.Petit J.M., Milic-Emili G. Measurement of endoesophageal pressure. J Appl Physiol. 1958;13(3):481–485. doi: 10.1152/jappl.1958.13.3.481. [DOI] [PubMed] [Google Scholar]
- 35.Walterspacher S., Isaak L., Guttmann J., et al. Assessing respiratory function depends on mechanical characteristics of balloon catheters. Respir Care. 2014;59(9):1345–1352. doi: 10.4187/respcare.02974. [DOI] [PubMed] [Google Scholar]
- 36.Talmor D., Sarge T., Malhotra A., et al. Mechanical ventilation guided by esophageal pressure in acute lung injury. N Engl J Med. 2008;359(20):2095–2104. doi: 10.1056/NEJMoa0708638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Talmor D., Sarge T., O'Donnell C.R., et al. Esophageal and transpulmonary pressures in acute respiratory failure. Crit Care Med. 2006;34(5):1389–1394. doi: 10.1097/01.CCM.0000215515.49001.A2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Terragni P., Mascia L., Fanelli V., et al. Accuracy of esophageal pressure to assess transpulmonary pressure during mechanical ventilation. Intensive Care Med. 2017;43(1):142–143. doi: 10.1007/s00134-016-4589-8. [DOI] [PubMed] [Google Scholar]
- 39.Brochard L., Slutsky A., Pesenti A. Mechanical ventilation to minimize progression of lung injury in acute respiratory failure. Am J Respir Crit Care Med. 2017;195(4):438–442. doi: 10.1164/rccm.201605-1081CP. [DOI] [PubMed] [Google Scholar]
- 40.Beitler J.R., Sarge T., Banner-Goodspeed V.M., et al. Effect of Titrating Positive End-Expiratory Pressure (PEEP) with an esophageal pressure-guided strategy vs an empirical high PEEP-Fio2 strategy on death and days free from mechanical ventilation among patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2019;321(9):846–857. doi: 10.1001/jama.2019.0555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Loring S.H., O'Donnell C.R., Behazin N., et al. Esophageal pressures in acute lung injury: do they represent artifact or useful information about transpulmonary pressure, chest wall mechanics, and lung stress? J Appl Physiol (1985) 2010;108(3):515–522. doi: 10.1152/japplphysiol.00835.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Olegard C., Sondergaard S., Houltz E., et al. Estimation of functional residual capacity at the bedside using standard monitoring equipment: a modified nitrogen washout/washin technique requiring a small change of the inspired oxygen fraction. Anesth Analg Jul. 2005;101(1):206–212. doi: 10.1213/01.ANE.0000165823.90368.55. table of contents. [DOI] [PubMed] [Google Scholar]
- 43.Chiumello D., Cressoni M., Chierichetti M., et al. Nitrogen washout/washin, helium dilution and computed tomography in the assessment of end expiratory lung volume. Crit Care. 2008;12(6):R150. doi: 10.1186/cc7139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dellamonica J., Lerolle N., Sargentini C., et al. PEEP-induced changes in lung volume in acute respiratory distress syndrome. Two methods to estimate alveolar recruitment. Intensive Care Med. 2011;37(10):1595–1604. doi: 10.1007/s00134-011-2333-y. [DOI] [PubMed] [Google Scholar]
- 45.Blankman P., Hasan D., van Mourik M.S., et al. Ventilation distribution measured with EIT at varying levels of pressure support and Neurally Adjusted Ventilatory Assist in patients with ALI. Intensive Care Med. 2013;39(6):1057–1062. doi: 10.1007/s00134-013-2898-8. [DOI] [PubMed] [Google Scholar]
- 46.Dargaville P.A., Rimensberger P.C., Frerichs I. Regional tidal ventilation and compliance during a stepwise vital capacity manoeuvre. Intensive Care Med. 2010;36(11):1953–1961. doi: 10.1007/s00134-010-1995-1. [DOI] [PubMed] [Google Scholar]
- 47.Yoshida T., Piraino T., Lima C.A.S., et al. Regional ventilation displayed by electrical impedance tomography as an incentive to decrease positive end-expiratory pressure. Am J Respir Crit Care Med. 2019;200(7):933–937. doi: 10.1164/rccm.201904-0797LE. [DOI] [PubMed] [Google Scholar]
- 48.Hinz J., Hahn G., Neumann P., et al. End-expiratory lung impedance change enables bedside monitoring of end-expiratory lung volume change. Intensive Care Med. 2003;29(1):37–43. doi: 10.1007/s00134-002-1555-4. [DOI] [PubMed] [Google Scholar]
- 49.Meier T., Luepschen H., Karsten J., et al. Assessment of regional lung recruitment and derecruitment during a PEEP trial based on electrical impedance tomography. Intensive Care Med. 2008;34(3):543–550. doi: 10.1007/s00134-007-0786-9. [DOI] [PubMed] [Google Scholar]
- 50.Spadaro S., Mauri T., Bohm S.H., et al. Variation of poorly ventilated lung units (silent spaces) measured by electrical impedance tomography to dynamically assess recruitment. Crit Care. 2018;22(1):26. doi: 10.1186/s13054-017-1931-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhao Z., Chang M.Y., Chang M.Y., et al. Positive end-expiratory pressure titration with electrical impedance tomography and pressure-volume curve in severe acute respiratory distress syndrome. Ann Intensive Care. 2019;9(1):7. doi: 10.1186/s13613-019-0484-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Malbrain M.L., Deeren D., De Potter T.J. Intra-abdominal hypertension in the critically ill: it is time to pay attention. Curr Opin Crit Care. 2005;11(2):156–171. doi: 10.1097/01.ccx.0000155355.86241.1b. [DOI] [PubMed] [Google Scholar]
- 53.Malbrain M.L. Different techniques to measure intra-abdominal pressure (IAP): time for a critical re-appraisal. Intensive Care Med. 2004;30(3):357–371. doi: 10.1007/s00134-003-2107-2. [DOI] [PubMed] [Google Scholar]
- 54.Cheatham M.L., De Waele J.J., De Laet I., et al. The impact of body position on intra-abdominal pressure measurement: a multicenter analysis. Crit Care Med. 2009;37(7):2187–2190. doi: 10.1097/CCM.0b013e3181a021fa. [DOI] [PubMed] [Google Scholar]
- 55.Tobin M.J., Lodato R.F. PEEP, auto-PEEP, and waterfalls. Chest. 1989;96(3):449–451. doi: 10.1378/chest.96.3.449. [DOI] [PubMed] [Google Scholar]
- 56.Koutsoukou A., Pecchiari M. Expiratory flow-limitation in mechanically ventilated patients: a risk for ventilator-induced lung injury? World J Crit Care Med. 2019;8(1):1–8. doi: 10.5492/wjccm.v8.i1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Fernandez R., Benito S., Blanch L., et al. Intrinsic PEEP: a cause of inspiratory muscle ineffectivity. Intensive Care Med. 1988;15(1):51–52. doi: 10.1007/BF00255638. [DOI] [PubMed] [Google Scholar]
- 58.Nava S., Bruschi C., Fracchia C., et al. Patient-ventilator interaction and inspiratory effort during pressure support ventilation in patients with different pathologies. Eur Respir J. 1997;10(1):177–183. doi: 10.1183/09031936.97.10010177. [DOI] [PubMed] [Google Scholar]
- 59.Young I.H., Bye P.T. Gas exchange in disease: asthma, chronic obstructive pulmonary disease, cystic fibrosis, and interstitial lung disease. Compr Physiol. 2011;1(2):663–697. doi: 10.1002/cphy.c090012. [DOI] [PubMed] [Google Scholar]
- 60.MacDonald M.I., Shafuddin E., King P.T., et al. Cardiac dysfunction during exacerbations of chronic obstructive pulmonary disease. Lancet Respir Med. 2016;4(2):138–148. doi: 10.1016/S2213-2600(15)00509-3. [DOI] [PubMed] [Google Scholar]
- 61.Rossi A., Polese G., Brandi G., et al. Intrinsic positive end-expiratory pressure (PEEPi) Intensive Care Med. 1995;21(6):522–536. doi: 10.1007/BF01706208. [DOI] [PubMed] [Google Scholar]
- 62.Patroniti N., Bellani G., Cortinovis B., et al. Role of absolute lung volume to assess alveolar recruitment in acute respiratory distress syndrome patients. Crit Care Med. 2010;38(5):1300–1307. doi: 10.1097/CCM.0b013e3181d8cb51. [DOI] [PubMed] [Google Scholar]
- 63.Perchiazzi G., Rylander C., Derosa S., et al. Regional distribution of lung compliance by image analysis of computed tomograms. Respir Physiol Neurobiol. 2014;201:60–70. doi: 10.1016/j.resp.2014.07.001. [DOI] [PubMed] [Google Scholar]
- 64.Loring S.H., Topulos G.P., Hubmayr R.D. Transpulmonary pressure: the importance of precise definitions and limiting assumptions. Am J Respir Crit Care Med. 2016;194(12):1452–1457. doi: 10.1164/rccm.201512-2448CP. [DOI] [PubMed] [Google Scholar]
- 65.Gattinoni L., Pelosi P., Suter P.M., et al. Acute respiratory distress syndrome caused by pulmonary and extrapulmonary disease. Different syndromes? Am J Respir Crit Care Med. 1998;158(1):3–11. doi: 10.1164/ajrccm.158.1.9708031. [DOI] [PubMed] [Google Scholar]