Abstract
We are currently experiencing the realities of the most severe pandemic within living memory, with major impacts on the health and economic well-being of our planet. The scientific community has demonstrated an unprecedented mobilization capability, with the rapid development of vaccines and drugs targeting the protection of human life and palliative measures for infected individuals. However, are we adequately prepared for ongoing defense against COVID-19 and its variants in the post-pandemic world? Moreover, are we equipped to provide a satisfactory quality of life for individuals who are recovering from COVID-19 disease? What are the possibilities for the acceleration of the recovery process? Here, we give special consideration to the potential and already-demonstrated role of probiotics and traditional medical approaches to the management of current and potential future encounters with our major virus adversaries.
Keywords: Coronavirus disease-19 (COVID-19), Lactic acid bacteria, Probiotics, Bacteriocins, Antimicrobial peptides, Traditional medicine
Introduction—Re-evaluation of Already Existing Antimicrobials
The antimicrobial specificity and efficacy of antagonistic proteins produced naturally by microorganisms and various other life forms continue and indeed now grows as a major research focus within the disciplines of microbiology and virology. The empirical application of proteinaceous materials as an infection treatment or preventative has been a cornerstone support for the growth of human populations, and a wide variety of either raw or fermented plant and natural product extracts have comprised a critical component of the arsenal of traditional medicine. An old Bulgarian proverb postulates that for every pain there is a medicinal plant remedy [1] and these days we can add that the remedy may also be a product derived from naturally occurring microbial processes. Interestingly, it seems that there has always been a strong belief that naturally occurring remedies are already available to help counter every pain, every disease, that it only requires our human ingenuity to discover and apply the appropriate active agents either in the form of simple extracts or in more purified formats.
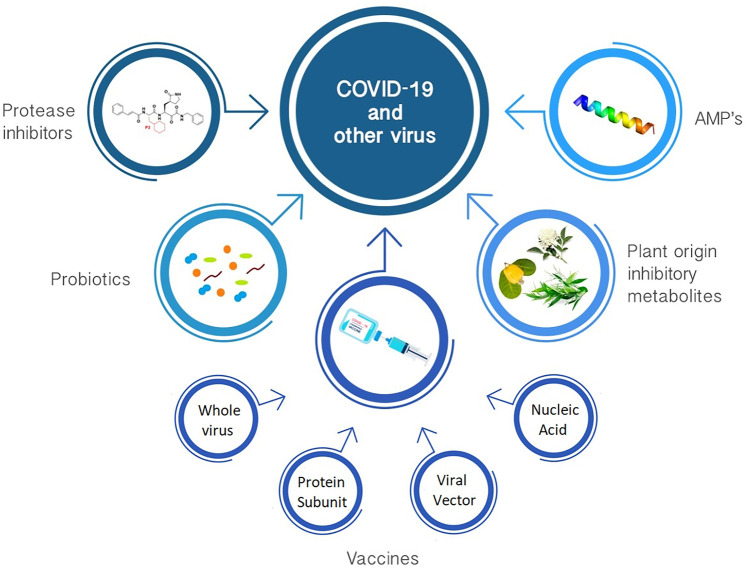
The aim of this opinion article is to focus attention upon some of the currently available information concerning the interaction of proteinaceous microbial products against viruses and in particular to consider the potential implications of such interactions in this post-COVID-19 world that we now live in (Fig. 1). We still do not know what might comprise the most effective pathways to achieve the elimination or at least the suppression of COVID-19 infections. In this article, we have tried to focus upon the currently known facts and credible speculations relating to the interaction between naturally occurring antibacterial and/or antiviral compounds and different types of viruses, and in particular, we have focused on the potential application of these agents to the management of COVID-19. Our underlying theme is that traditional medicine can provide a valuable natural addendum to modern Western medicine. Hippocrates proposed the medical professional codex, one of the foundation pillars of which has generally been taken to be “first do no harm”. With this in mind, it seems both justifiable and appropriate that traditional medicinal products should be given consideration for incorporation within the arsenal of approaches to apply synergistically with contemporary medical practices.
Fig. 1.
Some current available approaches for combat COVID-19 and other viruses
Now, in mid-2021, we are still confronted by a devastating global pandemic. On the one hand, COVID-19 has clearly illustrated the inherent fragility of human civilization as well as challenging the strength of human moral values. On the other hand, the urgent mobilization of a global scientific community response has resulted in the sometimes perhaps never seen before planned and development of potential vaccines targeting the control of COVID-19. Another, less frequently considered question relates to the management of adverse post-COVID-19 health consequences in individuals who have already experienced the infection. We support the thesis that some traditional medicine approaches including the sustained use of preparations containing naturally produced proteinase inhibitors or antimicrobial agents may have a role to play in patient recovery. Moreover, what beneficial role(s) might probiotics have in the recuperation of patients? The fact that vaccines have been developed and vaccination programs initiated does not of course in itself signal the termination of the current COVID-19 threat. Neither should this infer that new approaches are not needed. Indeed, it is now strikingly evident that a robust scientific capability to activate the rapid development of effective and optimized preparations having efficacy against both existing and novel viral infections and their complications will need to be retained as a priority focus for humanity for the foreseeable future.
Proteinase Inhibitors, a Possible Antiviral Drugs
In the past few decades, researchers have made considerable progress in their endeavors to define and to understand the physiological processes involved in virus replication and this knowledge has subsequently been used as the basis for the development of various antiviral pharmaceutical products. Although in principle the scientific basis for this approach is very simple, in reality, success has been difficult to achieve. Viruses hijack the biosynthetic machinery of their host cells in order to replicate. These processes typically include specific proteases, enzymes having a critical role in their replication cycle. Notably, however, there are already naturally occurring mechanisms in place for the specific regulation of protease activities, and so, the application of these protease inhibitor molecules represents a promising approach for the control of virus replication. The critical issue in this classical “magic bullet” approach to pathogen control is first to determine which protease(s) are to be targeted and then which inhibitor/s are most capable of interfering with virus replication, while at the same time exhibiting little or no toxicity for the host. These general principles have already been explored in the development of pharmaceutical preparations for the control of a wide variety of viruses including herpes simplex virus [2–7], rhinovirus [8, 9], Zika virus [10], picornavirus [5], poliovirus [11], human immunodeficiency virus [5, 12–14], hepatitis virus [5, 7], Ebola virus [15, 16], norovirus [7], dengue virus [17], and SARS and MERS coronaviruses [15, 16, 18–20] with variable levels of success.
Current anti-influenza drugs typically either target specific virus components such as the M2 proton channel blockers or function as inhibitors of either the neuraminidase or polymerase activities of the virus. Additionally, however, polyphenolic extracts from Geranium sanguineum have been shown to interfere with influenza virus protein expression, prompting suggestions that plant extracts such as this may provide a novel source of antiviral agents [21]. Are antiviral drugs able to control virus replication in virus-infected cells? This previously stated question explores the possibility of developing drugs that can overcome viral resistance and thereby would be suitable for different influenza viruses independently of their specific serotype [21]. It has already been postulated that the virulence of most influenza viruses is related to the ability of their HA precursor HA0 to be cleaved post-translationally into subunits HA1 and HA2 by trypsin-like proteases of the host [21]. The cleavage of HA is possibly the most critical step of the entire infectivity process since it can allow for the fusion of the viral and host cell membranes, prior to the release of the viral nucleocapsid into the host cytoplasm. Moreover, considering the importance of proteolytic processing for the successful reproduction of the virus, another potential target for effective control of influenza virus infection is blockage of the proteolytic cleavage of the virus proteins [21]. Success with this would lead to interference with subsequent rounds of virus replication and limitation of virus spread into the respiratory tract. This hypothesis has already been evaluated in chick embryos and in mice in experiments where exogenous inhibitors of serine proteases, including ε-aminocaproic acid (ACA) [22], aprotonin [23], and ambroxol [24], have achieved a reduction of HA cleavage and virus activation [21] for influenza viruses with monobasic HA. The Genus Streptomyces has been found to be a rich source of protease inhibitors having antiviral activity. Included among the documented Streptomyces antiviral protease inhibitors are MI 0114 [22] and SS 225b [25]. The replication of HIV-1 [26] and cytomegalovirus [27] has also been shown to be inhibited by Streptomyces proteinase inhibitors.
In other studies, the virus-inhibitory effect of metabolites from Streptomyces chromofuscus 34–1 was reported to be highly specific, strain related, and dose dependent [21, 28, 29]. The effectiveness of virus inhibition was demonstrated both in cell cultures and in experimental influenza virus infections in mice. The authors suggested that an inhibitory agent produced by Streptomyces chromofuscus 34–1 influenced virus protein proteolysis and hence indirectly affected the activation of the virus particles by increasing protease inhibitor activity [21, 28, 29].
Bacteriocins, Potential Antimicrobials with Antiviral Effect
Certain antimicrobial peptides produced by LAB have also been shown to be potential candidates for the control of some viruses. The Lactococcus lactis bacteriocin nisin is the most extensively studied and commercially utilized of all the bacteriocins. Its application to biopreservation has been approved both by EFSA and the FDA [30]. On the other hand, the medical application of bacteriocins is still largely either speculative or in the very early stages of implementation. Various strategies have been proposed for enhancing the bioactivity and in situ targeting efficacy of bacteriocins [31, 32]. One approach has been to generate modifications in the amino acid sequences of the bacteriocins by either introducing specific mutations within the bacteriocin structural gene or by post-translationally modifying the bacteriocin peptide sequences [33, 34]. As an example, the N-terminal modification of bacteriocins with specific polar polymers has been shown to increase their resistance to proteolytic enzymes in the gastrointestinal tract environment [32]. Traditionally, the study of bacteriocins has focused on their inhibitory activity against closely related competitor bacteria. More recently, however, research interest is increasingly being focused upon more unorthodox inhibitory activities of some bacteriocins as the search widens to identify potential agents to control relatively “exotic” pathogens of humans and other animals, including viruses. The mechanisms of the reported antiviral activities of bacteriocins are still being clarified [35–38]. Bacteriocins from certain strains of Lactobacillus spp., Lactococcus spp., Enterococcus spp., Bacillus spp., Staphylococcus spp., Erwinia spp., and Actinomadura spp. have already been shown to exhibit activity against various viruses including poliovirus, herpesvirus (HSV-1 and HSV-2), measles virus, Newcastle disease virus, coliphage HAS, and HIV-1 [35–39]. Experience gained from the attempted bacteriocin-mediated control of poliovirus may also have an application to the development of strategies for the management of SARS-CoV-2 [40]. Wachsman et al. [35] have proposed mechanisms underlying the interaction of an enterocin produced by E. mundtii CRL35 and herpesvirus in terms of the blocking of replication of the viral gamma protein (glycoprotein D) during the process of virus invasion.
In other studies, Serkedjieva et al. [41] reported that a Lactobacillus delbrueckii bacteriocin had virus-inhibitory activity. Previously, Dundarov and Andonov [42] had demonstrated a strong anti-herpesvirus effect of the ionophore antibiotic pandavir (nigericin). Pandavir inhibited virus reproduction, even at 0.01–0.02 ng/mL, by specific inhibition of virus DNA synthesis. The ionophore antibiotics monensin and A-23187 have also been shown to inhibit some RNA viruses by blocking the viral glycoproteins on the surface of infected cells [43].
Certain bacteriocins produced by LAB isolated from traditional fermented food products have demonstrated moderate antiviral activity against various viruses including herpesvirus [35–38]. A variety of beneficial properties have been attributed to these traditional fermented products, including enhancement of the responsiveness of the immune system [36]. Not all of the putative beneficial properties of fermented food products are of course likely to be attributable only to the presence of bacteriocins. In addition to their content of bacteriocin-producing microorganisms, these products will also contain a variety of bioactive metabolites, including vitamins, polypeptides, polysaccharides, short-chain fatty acids, inhibitors, and/or activators, called by some authors “postbiotics.” A combination of all factors will contribute to the beneficial efficacy of each product, and undoubtedly, the specific combinations of these factors could display some substantial variability from batch to batch of the product.
Pieces of evidence from different reports show that bacteriocins can be considered an effective alternative in viral control [35–41]. Similar to the proteinase inhibitors, bacteriocins are not killing the virus themselves, but interfere with viral replications via inhibiting some enzymes, critical for the virus life cycle [35]. Most probably, some different mechanisms can be involved in this interaction between virus and bacteriocin, and this can be relevant topics for intensive research. On the other side, vaccines are considered the gold standard in the prevention of diseases, including COVID-19, and currently, vaccination programs in several countries show their benefits. However, already new variants of the COVID-19 were challenging the health systems, and maybe combinations of different approaches in the prevention and treatment will be the correct alternative in the combat against these new variants of COVID-19.
Plant Extracts with Antiviral Application
Preliminary studies of various plant extracts have also yielded promising preliminary evidence of antiviral activities [44, 45]. Traditional medicine can undoubtedly be an important source both of beneficial information and of bioactive antiviral preparations, however, and to date, only a relatively small number of the traditional medicine leads have been systematically explored [45]. Undoubtedly, the main focus of antiviral research in the last few decades has been on synthetic chemistry as a way of identifying and evaluating putative novel antiviral compounds. Increasingly, however, public health concerns prompted by the relative absence of potent new antiviral agents have led to the re-evaluation of traditional medical practices and existing phyto-pharmaceutical knowledge as a possible pathway for identifying novel antiviral molecules or mechanisms of action [44, 45]. Thabti et al. [45] suggested that plants like mulberry (Morus spp.), jackfruit (Artocarpus heterophyllus), coffee, and others may be sources of new antiviral biotherapeutics. Extracts of the Morus species (mulberry: Morus alba var. alba, Morus alba var. rosa, and Morus rubra) have been proposed as candidates for the control of emerging SARS-CoV viruses (pandemic coronaviruses) and the Picornaviridae viruses (human poliovirus, human parechovirus, and human echovirus) [45]. In other studies, prenylated flavonoids (leachinone G and mulberroside C) from Morus alba var. alba were found to be inhibitory to HSV-1 [46] and ethyl acetate fractions showed strong activity against the hepatitis C virus [47]. Antiviral phenolics (especially cafeic and chlorogenic acid) and flavonol glycosides have been detected in mulberry leaves [48]. Cafeic acid was described as potentially inhibitory to the proliferation of HSV-2 and adenovirus type 3 (ADV-3) [49] and chlorogenic acid exhibited the strongest anti-ADV-11 activity. Chlorogenic acid and cafeic acid have been proposed as strong potential inhibitors of hepatitis B virus multiplication, reducing the number of viral particles in serum by blocking DNA synthesis [50]. More recently, Thabti et al. [45] have proposed that Morus extracts (especially the kuwanon G component) could possibly function as an inhibitor of SARS-CoV-2.
Some flavonoids of plant origin, such as rutin, quercetin (an aglycone of rutin), myricetin (3,3,4,5,5,7-hexahydroxyflavone), quercetagetin (3,3,4,5,6,7-hexahydroxyflavone), baicalein (5,6,7-trihydroxyflavone), apigenin (4,5,7-trihydroxyflavone), 7-galloyl catechin, kaempferol 3-O-β-(6″-O-galloyl)-glucopyranoside, quercetin 3-O-β-(6″-O-galloyl)-glucopyranoside, curcumin, kaempferolor-tri-terpenoidso-leanolic acid, gallic acid, and ursolic acid have been widely explored as potential antiviral compounds (reviewed by Ben-Shabat et al. [44] and El-Toumy et al. [51]).
A polyphenolic complex from the Bulgarian medicinal plant Geranium sanguineum L has been evaluated for its activity against different strains of human, avian, and equine influenza viruses [52]. The results were indicative of a strain-specific antiviral inhibitory effect, related to the expression of influenza virus proteins on infected cell surfaces. When administered intra-nasally as an aerosol, a plant extract of Geranium sanguineum reduced mortality in a mice model, with an index of protection of 64.47% [52]. The authors suggested that the virus-inhibitory effect was attributable to the presence of large amounts of polyphenolic substances in the extracts including gallotannins, catechins, kaempferol, myricetin, monne, quercetin, ramnasin, retusin, and the polyphenolic acids (caffeic, ellagic, and chinic) [52, 53].
Are Probiotics an Alternative in Antiviral Actions?
It is now evident that some probiotics can contribute to the prevention and even to the treatment of virus infections. The proposed mechanisms of these antiviral activities are often indirect and in many instances appear to hinge upon stimulation of the host’s immune system. Indeed, the application of a variety of immunity-stimulating natural products, sometimes including probiotics, is no longer a novelty within the therapeutic arsenal for the prevention and treatment of respiratory viral infections [36, 54].
The majority of upper respiratory tract infections is of viral etiology and the most common etiological agents include the rhinoviruses, coronaviruses, adenoviruses, respiratory syncytial virus, influenza, and parainfluenza viruses [54]. The host’s immune system plays a principal role not only in preventing the onset of these upper respiratory tract infections, but also in shortening the recovery period [54, 55]. Priming of the efficacy of the natural immune system is typically achieved via the use of an appropriate diet, supplemented by food additives and probiotics. Supplementation with vitamins C and/or D has been specifically recommended to enhance protection against the pandemic coronaviruses [56, 57], and historically, the directed intake of various vitamins, minerals, and antioxidants, as well as the liberal consumption of fiber, fruits, and vegetables, has been a foundation practice of traditional medicine to help stimulate the immune system and prevent viral infections [58]. The scientifically guided use of probiotics and prebiotics is another contemporary strategy, combining elements of traditional medicinal knowledge built up over many centuries with powerful novel food additives, identified and evaluated by sophisticated modern microbiology and biotechnology protocols. Probiotics can bring a wide variety of benefits to the consumer, including contributing to the prevention and treatment of upper respiratory tract infections [54]. On the other hand, probiotics and traditional medicine approaches do not provide a panacea and it cannot be claimed with an assurance that the use of probiotic microorganisms will automatically result in the effective treatment or prevention of infections. Appropriate strain selection and meta-analyses of their strain-specific attributes (both beneficial and potentially detrimental) need to carry out, in addition to carefully controlled evaluation of the probiotic interactions with the host, with a particular focus on the host’s immune system [54].
Various LAB have attracted attention and have been evaluated as potential probiotics over the past two decades, and on the basis of systematic clinical studies, some have been recommended as prophylactic and/or therapeutic agents for the prevention and/or control of viral infections [59]. Moreover, modern developments of analytical research tools have accelerated research and even facilitated the selection and development of recombinant new generation probiotic strains expressing specific antigens capable of inducing protective immune responses and increased resistance to infections [60]. The application of LAB probiotics as antiviral agents has already found application as a strategy in aquaculture [61] and in the poultry industry [62]. Increasingly, it is also being explored for use in human and veterinary medicine [36].
Different mechanisms of action have been proposed to better understand the interactions between probiotic LAB and viruses, but in most cases, the specific or relatively specific adsorption or trapping of the virus particles is the prerequisite first step. Botić et al. [63] described direct interactions (trapping) between probiotics (different Lactobacillus spp.) and the vesicular stomatitis virus and Wang et al. [64] reported the ability of strains of Enterococcus faecium to inhibit influenza viruses via direct physical interaction. Similarly, Al Kassaa [36] suggested that Lactobacillus gasseri CMUL57 can inhibit herpes simplex type 2 virus (HSV-2) through a trapping mechanism. Stimulation of the immune system of the host by LAB has been proposed for the influenza virus H1N1 [65, 66] and for protective effects against some respiratory virus infections in mouse models and in humans which have also been reported [66, 67]. In other studies, probiotics have enhanced the effects of the influenza virus vaccine [68, 69]. Lactobacillus rhamnosus GG reduced the incidence of respiratory virus infections [70] and Lactobacillus acidophilus NCFM reduced the occurrence of influenza-like symptoms [71]. Daily consumption of a probiotic dairy drink improved antibody responses to influenza virus after vaccination in elderly individuals [68] and oral administration of Lactobacillus casei Shirota to neonatal and infant mice showed that a boost of the immature immune system can play a positive role in the protection against influenza virus infection [72].
In addition to their influence on virus infections, probiotics can also be applied to the prevention and even treatment of other diseases, including allergic diseases and inflammatory episodes in the respiratory tract, including asthma. Asthma is characterized by airway inflammation and hyper-responsiveness of the host [73]. The efficacy of some probiotics in suppressing both allergic and autoimmune responses in the airways of the host has been illustrated in murine models of acute airway inflammation [74–78].
In the post-COVID-19 pandemic world, we will still be confronted with serious problems concerning patient recovery. Although by definition these post-COVID-19 infection patients have been freed of the presence of the virus, there will however often be residual damage to their tissues, especially in the respiratory tract. Taking into consideration the potential similarity of many of the post-COVID-19 infection problems to those currently faced by asthmatic patients, it seems that as an interim measure that some of the probiotics which have been developed in the last decade specifically to provide relief for asthmatic patients or for the treatment of upper respiratory tract infections may also have value for use by post-COVID-19 patients. Certainly, it seems there will now be progressively increasing scientific challenge to discover and commission probiotics and/or traditional medicine products that are capable of helping to restore normal lung function in individuals who have experienced lung tissue damage that is either directly virus mediated or that occurs as a pathological side effect of the host immune response to virus infection.
Even if the general effort of the scientific community was focus on developing the vaccine for the protection against COVID-19 based on mRNA technology or based on traditional approaches, some laboratories have explored different approaches—selecting from existing or generating modified bacterial species that can be effective in combat against COVID-19 and be consumed per oral. Probiotics can be good candidates to serve as oral vaccine delivery systems, including the fact that can facilitate elicitation of mucosal immunity without latent risks of pathogenicity. Knowledge build in the last decades on the deeper understanding of the interaction between probiotics and the mucosal immune system can only facilitate the development of effective oral vaccine vectors applying probiotic organisms [79]. Different strategies for developing effective COVID-19 vaccines, including comprising existing already in health practice and these still in the research processes, and different approaches for administration and effect on the host immunes system, including oral vaccines, were recently reviewed by Ashraf et al. [80].
One of the principal pathological effects of SARS-CoV-2 is leading to a several proinflammatory cytokine storm and chronic lung inflammation. The role of probiotics in the modulation of the immune system is supported by scientific pieces of evidence, showing clear antiviral and general immune-strengthening health effects [81]. From one side, it was previously suggested that some probiotic strains can trap the viruses and thus can reduced (or even eliminate) the virus binding to the host cell receptors [63]. Moreover, it was suggested that some strains such as Lactobacillus rhamnosus CRL1505, Lactobacillus gasseri SBT2055, Lactobacillus casei DK128, and some strains of Bifidobacterium bifidum can be considered as promising candidates for potential management of SARS-CoV-2 infections related to induction of strong anti-inflammatory response, results from tests in the appropriate animal model [81]. As well, pieces of evidence from clinical trials evaluating the role of Lactobacillus rhamnosus GG, Lactobacillus casei (including Shirota strain), Lactobacillus plantarum, Bifidobacterium lactis Bb-12, and Bifidobacterium longum different strains, reported on significant reduction of the prevalence of upper respiratory infections and flu-related symptoms [81]. All these probiotic trials can be critically evaluated and applied in clinical and post-clinical treatments of COVID-19 patients. Applications of probiotic strains can facilitate mitigation of the extreme levels of cytokines by establishing a balance between cellular and humoral immune responses. Moreover, it was suggested that some probiotics can be actively involved in the neutralization of free radicals, as several of them have antioxidant properties [81].
The role of probiotics in managing COVID-19 and other viral infections is currently generating more questions than answers. Even if we have adapted the concept of probiotics as live (or even dead) microbial cells that can have their benefits for the host, the scientific community and health professionals are still asking for more and more pieces of evidence for the positive role of probiotics on human (and other animals) health. Maybe a simple reason is the fact that several speculative products are on the market with not really proven health benefits, promising “miracles” to the consumers. There were always and will be always persons looking for their “15 min of fame.” However, solid scientific pieces of evidence are showing that right probiotics, delivered to the right person in the right doses, can have positive effects and even can be considered as potential candidates for the parallel active therapy of COVID-19 patients and post-COVID-19 recovery [81]. As well, we need to point that the GIT microbiota of that patient will be highly disordered after extensive treatments including highly active antibiotics and a specific diet during the hospital period. This is one more argument that appropriate re-establish GIT microbial balance will be some of the points in the recovery period of post-COVID-19 victims.
Acknowledgements
To Cecilia Alejandra Vazquez Bucheli for designing the figure.
Author Contribution
Original concept: S.D.T. and I.V.I.; writing of the manuscript: S.D.T. and I.V.I.; critical corrections and editing: S.D.T. and J.R.T.
Data Availability
All data presented, generated, or analyzed during this study are included in this published article and complied with research standards.
Declarations
Ethical Approval
This article does not contain any studies with human or animal subjects performed by authors. Any data on such experiments is obtained from appropriate, mentioned literature sources.
Conflict of Interest
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Bakhchevanov S (2010) Patient: for every pain - an herb. Millennium, Sofia, Bulgaria. pp. 208. ISBN: 9789545150623 (in bulgarian)
- 2.Waxman L, Darke PL. Theherpesivirus proteases as target for antiviral chemotherapy. A review Antiviral Chem Chemother. 2000;11(1):1–22. doi: 10.1177/095632020001100101. [DOI] [PubMed] [Google Scholar]
- 3.Lin K, Perni RB, Kwong AD, Lin C. VX-950, a novel hepatitis C virus (HCV) NS3-4A protease inhibitor, exhibits potent antiviral activities in HCV replicon cells. Antimicrob Agents Chemother. 2006;50(5):1813–1822. doi: 10.1128/AAC.50.5.1813-1822.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Malcolm BA, Liu R, Lahser F, Agrawal S, Belanger B, Butkiewicz N, Chase R, Gheyas F, Hart A, Heck D, Ingravallo P, Jiang C, Kong R, Lu J, Pichardo J, Prongay A, Skelton A, Tong X, Venkatraman S, Xia E, Girijavallabhan V, Njoroge FG. SCH 503034, a mechanism-based inhibitor of hepatitis C vírus NS3 protease, suppresses polyprotein maturation and enhances the antiviral activity of alpha interferon in replicon cells. Antimicrob Agents Chemother. 2006;50(3):1013–1020. doi: 10.1128/AAC.50.3.1013-1020.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Patrick AK, Potts KE. Protease inhibitor as antiviral agents. Clin Microbiol Rev. 1998;11(4):614–627. doi: 10.1128/CMR.11.4.614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Supuran CT, Casini A, Scozzafa A. Protease inhibitors of the sulfonamide type: anticancer, intiinflamatory, and antiviral agents. Med Res Rev. 2003;23(5):535–558. doi: 10.1002/med/10047. [DOI] [PubMed] [Google Scholar]
- 7.Chang K-O, Kim Y, Lovell S, Rathnayake AD, Groutas WC. Antiviral drug discovery: norovirus protease and development of inhibitors. Viruses. 2019;11:197. doi: 10.3390/v11020197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Patick AK, Binford SL, Brothers MA, Jackson RL, Ford CE, Diem MD, Maldonado F, Dragovich PS, Zhou R, Prins TJ, Fuhrman SA, Meador JW, Zalman LS, Matthews DA, Worland ST. In vitro antiviral activity of AG7088, a potent inhibitor of human rhinovirus 3C protease. Antimicrob Agents Chemother. 1999;43(10):2444–2450. doi: 10.1128/AAC.43.10.2444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mattews DA, Dragovich PS, Webber SE, Fuhrman SA, Patick AK, Zalman LS, Hendrickson TF, Love RA, Prins TJ, Marakovits JT, Zhou R, Tikhe J, Ford CE, Meador JW, Ferre RA, Brown EL, Binford SL, Brothers MA, Delisle DM, Worland ST. Structure-assisted design of mechanism-based irreversible inhibitors of human rhinovirus 3C protease with potent antiviral activity against multiple rhinovirus serotypes. Proc Nat Acad Sci USA. 1999;96:11000–11007. doi: 10.1073/pnas.96.20.11000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kang C, Keller TH, Luo D. Zika virus protease: an antiviral drug target. Trends Microbiol. 2017;25(10):797–808. doi: 10.1016/j.tim.2017.07.001. [DOI] [PubMed] [Google Scholar]
- 11.Rhoden E, Liu HM, Wang-Chern SW, Oberste MS. Anti-poliovirus activity of protease inhibitor AG-7404, and assessment of in vitro activity in combination with antiviral capsid inhibitor compounds. Antiviral Res. 2013;98(2):186–191. doi: 10.1016/j.antiviral.2013.03.003.Erratum.In:AntiviralRes.2013Sep,99(3):436. [DOI] [PubMed] [Google Scholar]
- 12.Molla A, Mo H, Vasavanonda S, Han L, Lin CT, Hsu A, Kempf DJ. In vitro antiviral interaction of lopinavir with other protease inhibitors. Antimicrob Agents Chemother. 2002;46(7):2279–2253. doi: 10.1128/AAC.46.7.2249-2253.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chauvin B, Drouot S, Barrail-Tran A, Taburet A-M. Drug-drug interactions between HMG-CoA reductase inhibitor (statins) and antiviral protease inhibitors. Clin Pharmacokinet. 2013;52:815–831. doi: 10.1007/s40262-013-0075-4. [DOI] [PubMed] [Google Scholar]
- 14.Glenn MP, Pattenden LK, Reid RC, Tyssen DP, Tyndall JDA, Birch CJ, Fairlie DP. β-Strand mimicking macrocyclic amino acids: templates for protease inhibitors with antiviral activity. J Med Chem. 2002;45:371–381. doi: 10.1021/jm010414i. [DOI] [PubMed] [Google Scholar]
- 15.Zhou N, Pan T, Zhang J, Li Q, Zhang X, Bai C, Hiang F, Peng T, Zhang J, Liu C, Tao L, Zhang H. Clycopeptide antibiotics potently inhibit cathepsin L in the late endosome/Lysosome and block the entry of Ebola virus, Middle East respiratory syndrom coronavirus (SARS-CoV) J Bio Chem. 2016;291(17):9218–9232. doi: 10.1074/jbc.M116.716100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gordon CJ, Tchesnokov EP, Feng JY, Porter DP, Gotte M. The antiviral compound remdesivir potently inhibits RNA-dependent RNA polymerase from Middle East respiratory syndrome corona virus. J Biol Chem. 2020;295:4773–4779. doi: 10.1074/jbc.AC120.013056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nitsche C, Schreier VN, Behnam MAM, Kumar A, Bartenschlager R, Klein CD. Thiazolidinone- peptide hybrids as dengue virus protease inhibitors with antiviral activity in cell culture. J Med Chem. 2013;56:8389–8403. doi: 10.1021/jm400828u. [DOI] [PubMed] [Google Scholar]
- 18.Tan ELC, Ooi EE, Lin C-Y, Tan HC, Ling AE, Lim B, Stanton LW. Inhibition of SARS corona virus in vitro with clinically approved antiviral drugs. Emerging Inf Dis. 2004;10(4):581–586. doi: 10.3201/eid1004.030458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kumar S, Zhi K, Mukherji A, Gerth K (2020) Repurposing antiviral protease inhibitors using extracellular vesicles for potential therapy of Covid-19. Viruses 12:486. 10.3390/v12050486 [DOI] [PMC free article] [PubMed]
- 20.Dai W, Zhang B, Jiang XM, Su H, Li J, Zhao Y, Xie X, Jin Z, Peng J, Liu F, Li C, Li Y, Bai F, Wang H, Cheng X, Cen X, Hu S, Yang X, Wang J, Liu X, Xiao G, Jiang H, Rao Z, Zhang LK, Xu Y, Yang H, Liu H. Structure-based design of antiviral drug candidates targeting the SARS-CoV-2 main protease. Science. 2020;368(6497):1331–1335. doi: 10.1126/science.abb4489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Serkedjieva J, Dalgalarrondo M, Angelova-Duleva L, Ivanova I. Antiviral potential of a proteolytic inhibitor from Streptomyces chromofuscus 34–1. Biotechnol Biotechnol Eq. 2012;26(1):2786–2793. doi: 10.5504/BBEQ.2011.0097. [DOI] [Google Scholar]
- 22.Lozitsky VP, Fedchuk AS, Puzis LE, Girlya BVP, YI, Bubnov VV, Participation of the proteolysis system in realization of influenza virus virulence and development of the infectious process: antiviral effect of protease inhibitors. Vopr Virusol. 1987;32:413–419. [PubMed] [Google Scholar]
- 23.Ovcharenko AV, Zhirnov OP. Aprotinin aerosol treatment of influenza and paramyxovirus bronchopneumonia of mice. Antiviral Res. 1994;23:107–118. doi: 10.1016/0166-3542(94)90038-8. [DOI] [PubMed] [Google Scholar]
- 24.Yang B, Yao DF, Ohuchi M, Ide M, Yano M, Okumura Y, Kido H. Ambroxol suppresses influenza-virus proliferation in the mouse airway by increasing antiviral factor levels. Eur Respir J. 2002;19(5):952–958. doi: 10.1183/09031936.02.00253302. [DOI] [PubMed] [Google Scholar]
- 25.Serkedjieva J, Ivanova I. In vitro and in vivo influenza virus inhibitory effects of a bacterial protease inhibitor. J Pure Appl Microbiol. 2009;3:441–452. [Google Scholar]
- 26.De Clercq E (2001) New developments in anti-HIV chemotherapy.Curr Med Chem 8(13):1543–1572. 10.2174/0929867013371842 [DOI] [PubMed]
- 27.Shu YZ, Ye Q, Kolb JM, Huang S, Veitch JA, Lowe SE, Manly SP (1997) Bripiodionen, a new inhibitor of human cytomegalovirus protease from Streptomyces sp. WC76599. J Nat Prod 60(5):529–532. 10.1021/np970054v [DOI] [PubMed]
- 28.Angelova L, Dalgalarrondo M, Minkov I, Danova S, Kirilov N, Serkedjieva J, Chobert J-M, Haertlé T, Ivanova I. Purification and characterisation of a protease inhibitor from Streptomyces chromofuscus 34–1 with an antiviral activity. Bioch Biophys Acta. 2006;1760:1210–1216. doi: 10.1016/j.bbagen.2006.03.002. [DOI] [PubMed] [Google Scholar]
- 29.Angelova L, Danova S, Iliev I, Ivanova I, Serkedjieva J. Characterization of production of an extracellular proteinase inhibitor from Streptomyces chromofuscus34–1 with alkaline phosphatase activity and antiviral effect. Biotechnol Biotechnol Eq. 2005;19(2):126–131. doi: 10.1080/13102818.2005.10817203. [DOI] [Google Scholar]
- 30.Favaro L, Penna ALB, Todorov SD (2015) Bacteriocinogenic LAB from cheeses - application in biopreservation? Trends Food Sci Techno l41(1):37–48. 10.1016/j.tifs.2014.09.001
- 31.Shin JM, Gwak JW, Kamarajan P, Fenno JC, Rickard AH, Kapila YL. Biomedical applications of nisin. J Appl Microbiol. 2016;120(6):1449–1465. doi: 10.1111/jam.13033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Umu ÖC, Bäuerl C, Oostindjer M, Pope PB, Hernández PE, Pérez-Martínez G, Diep DB. The potential of class II bacteriocins to modify gut microbiota to improve host health. PLoS One. 2016;11(10):e0164036. doi: 10.1371/journal.pone.0164036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Field D, Begley M, O’Connor PM, Daly KM, Hugenholtz F, Cotter PD, Hill C, Ross RP. Bioengineered nisin A derivatives with enhanced activity against both Gram positive and Gram negative pathogens. PLoS One. 2012;7(10):e46884. doi: 10.1371/journal.pone.0046884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Carroll J, Field D, O’Connor PM, Cotter PD, Coffey A, Hill C, O`Mahony J. Thegene encoded antimicrobial peptides, a template for the design of novel anti-mycobacterial drugs. Bioeng Bugs. 2010;1(6):408–412. doi: 10.4161/bbug.1.6.13642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wachsman MB, Castilla V, de Ruiz Holgado AP, de Torres RA, Sesma F, Coto CE. Enterocin CRL35 inhibits late stages of HSV-1 and HSV-2 replication in vitro. Antiviral Res. 2003;58(1):17–24. doi: 10.1016/s0166-3542(02)00099-2. [DOI] [PubMed] [Google Scholar]
- 36.Al Kassaa I, Hober D, Hamze M, Chihib NE, Drider D. Antiviral potential of lactic acid bacteria and their bacteriocins. Probiotics Antimicrob Proteins. 2014;6(3–4):177–185. doi: 10.1007/s12602-014-9162-6. [DOI] [PubMed] [Google Scholar]
- 37.Cavicchioli VQ, de Carvalho OV, de Paiva JC, Todorov SD, Júnior AS, Nero LA (2018) Inhibition of herpes simplex virus 1 (HSV-1) and poliovirus (PV-1) by bacteriocins from Lactococcus lactis subsp. lactis and Enterococcus durans strains isolated from goat milk. Int. J. Antimicrob. Agents 51:(1):33–37. 10.1016/j.ijantimicag.2017.04.020 [DOI] [PubMed]
- 38.Torres NI, Noll KS, Xu S, Li J, Huang Q, Sinko PJ, Wachsman MB, Chikindas ML. Safety, formulation, and in vitro antiviral activity of the antimicrobial peptide subtilosin against herpes simplex virus type 1. Probiotics Antimicrob Proteins. 2013;5(1):26–35. doi: 10.1007/s12602-012-9123-x.Erratum.In:ProbioticsAntimicrobProteins.2013,5(1):36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Todorov SD, Wachsman M, Tomé E, Dousset X, Destro MT, Dicks LMT, Franco BDGM, Vaz-Velho M, Drider D. Characterisation of an antiviral pediocin-like bacteriocin produced by Enterococcus faecium. Food Microbiol. 2010;27(7):869–879. doi: 10.1016/j.fm.2010.05.001. [DOI] [PubMed] [Google Scholar]
- 40.O’Reilly KM, Auzenbergs M, Jafari Y, Liu Y, Flasche S, Lowe R. Effective transmission across the globe: the role of climate in COVID-19 mitigation strategies. The Lancet Planetary health. 2020;4(5):e172. doi: 10.1016/S2542-5196(20)30106-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Serkedjieva J, Danova S, Ivanova I. Anti-influenza virus activity of a bacteriocin produced by Lactobacillus delbrueckii. Appl Biochem Biotechnol. 2000;88:285–298. doi: 10.1385/ABAB:88:1-3:285. [DOI] [Google Scholar]
- 42.Dundarov S, Andonov P. Medico-Biological Info. 1989;4:3–27. [Google Scholar]
- 43.Johnson D, Schlesinger M. Vesicular stomatitis and Sinbis virus glycoprotein transport to the cell surface is inhibited by ionophores. Virology. 1980;103:407–424. doi: 10.1016/0042-6822(80)90200-7. [DOI] [PubMed] [Google Scholar]
- 44.Ben-Shabat S, Yarmolinsky L, Porat D, Dahan A. Antiviral effect of phytochemicals from medicinal plants: applications and drug delivery strategies. Drug Deliv Transl Res. 2020;10(2):354–367. doi: 10.1007/s13346-019-00691-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Thabti I, Albert Q, Philippot S, Dupire F, Westerhuis B, Fontanay S, Risler A, Kassab T, Elfalleh W, Aferchichi A, Varbanov M (2020) Advances on antiviral activity of Morus spp. plant extracts: human coronavirus and virus-related respiratory tract infections in the spotlight. Molecules 25(8):1876. 10.3390/molecules25081876 [DOI] [PMC free article] [PubMed]
- 46.Du J, He ZD, Jiang RW, YeWC XuHX, Pui-Hay BP. Antiviral flavonoids from the root bark of Morus alba L. Phytochem. 2003;62:1235–1238. doi: 10.1016/s0031-9422(02)00753-7. [DOI] [PubMed] [Google Scholar]
- 47.Lee HY, Yum JH, Rho YK, Oh SJ, Choi HS, Chang HB, Choi DH, Leem MJ, Choi EJ, Ryu JM, Hwang SB. Inhibition of HCV replicon cell growth by 2-arylbenzofuran derivatives isolated from Mori Cortex Radicis. Planta Med. 2007;73(14):1481–1485. doi: 10.1055/s-2007-990249. [DOI] [PubMed] [Google Scholar]
- 48.Thabti I, Elfalleh W, Hannachi H, Ferchichi A, Da Graça CM. Identification and quantification of phenolic acids and flavonol glycosides in Tunisian Morus species by HPLC–DAD and HPLC–MS. J Funct Foods. 2012;4:367–374. doi: 10.1016/j.jff.2012.01.006. [DOI] [Google Scholar]
- 49.Chiang L, Chiang W, Chang M, Ng L, Lin C. Antiviral activity of Plantago major extracts and related compounds in vitro. Antivir Res. 2002;55:53–62. doi: 10.1016/S0166-3542(02)00007-4. [DOI] [PubMed] [Google Scholar]
- 50.Wang GF, Shi LP, Ren YD, Liu QF, Liu HF, Zhang RJ, Li Z, Zhu FH, He PL, Tang W, Tao PZ, Li C, Zhao WM, Zuo JP. Anti-hepatitis B virus activity of chlorogenic acid, quinic acid and cafeic acid in vivo and in vitro. Antivir Res. 2009;83:186–190. doi: 10.1016/j.antiviral.2009.05.002. [DOI] [PubMed] [Google Scholar]
- 51.El-Toumy SA, Salib JY, El-Kashak WA, Marty C, Bedoux G, Bourgougnon N. Antiviral effect of polyphenol rich plant extracts on herpes simplex virus type 1. Food Sci Human Wellness. 2018;7:91–101. doi: 10.1016/j.fshw.2018.01.001. [DOI] [Google Scholar]
- 52.Serkedjieva J. A polyphenolic extract from Geranium sanguineum L. inhibits influenza virus protein expression. Phytother Res. 1996;10:441–443. doi: 10.1002/(SICI)1099-1573(199608)10:5<441::AID-PTR867>3.0.CO;2-9. [DOI] [Google Scholar]
- 53.Serkedjieva J, Ivancheva S, Manolova N, Gegova G (1989) Studies on the antiviral activity of a polyphenolic complex isolated from the medicinal plant Geranium sanguineum L.X. Isolation of a biologically active butanolicfraction (1989) In:, Proceedings Vth International Conference on Chemistry and Biotechnology of Biologically Active Natural Products, 18–23 Sept., 1989, Varna, Bulgaria
- 54.Altadill T, Mazo JE, Liong M-L. Effects of a lactobacilli probiotic on reducing duration of URTI and fever, and use of URTI-associated medicine: a re-analysis of a randomized, placebo-controlled study. Microorganisms. 2021;9(3):528. doi: 10.3390/microorganisms9030528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rondanelli M, Miccono A, Lamburghini S, Avanzato I, Riva A, Allegrini P, Faliva MA, Peroni G, Nichetti M, Perna S. Self-care for common colds: the pivotal role of vitamin D, vitamin C, Zinc, and Echinacea in three main immune interactive clusters (physical barriers, innate and adaptive immunity) involved during an episode of common colds-practical advice on dosages and on the time to take these nutrients/botanicals in order to prevent or treat common colds. Evid Based Complement Alternat Med. 2018;2018:5813095. doi: 10.1155/2018/5813095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hemilä H, Chalker E (2013) Vitamin C for preventing and treating the common cold. Cochrane Database Syst Rev 2013(1):CD000980. 10.1002/14651858.CD000980.pub4 [DOI] [PMC free article] [PubMed]
- 57.Martineau AR, Jolliffe DA, Hooper RL, Greenberg L, Aloia JF, Bergman P, Dubnov-Raz G, Esposito S, Ganmaa D, Ginde AA, Goodall EC, Grant CC, Griffiths CJ, Janssens W, Laaksi I, Manaseki-Holland S, Mauger D, Murdoch DR, Neale R, Rees JR, Simpson S, Jr, Stelmach I, Kumar GT, Urashima M. Camargo Jr CA (2017) Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data. BMJ. 2017;356:i6583. doi: 10.1136/bmj.i6583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.van der Gaag E, Brandsema R, Nobbenhuis R, van der Palen J, Hummel T. Influence of dietary advice including green vegetables, beef, and whole dairy products on recurrent upper respiratory tract infections in children: a randomized controlled trial. Nutrients. 2020;12(1):272. doi: 10.3390/nu12010272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Mercenier A, Pavan S, Pot B. Probiotics as biotherapeutic agents: present knowledge and future prospects. Curr Pharm Des. 2003;9(2):175–191. doi: 10.2174/1381612033392224. [DOI] [PubMed] [Google Scholar]
- 60.Villena J, Oliveira ML, Ferreira P, Salva S, Alvarez S. Lactic acid bacteria in the prevention of pneumococcal respiratory infection: future opportunities and challenges. Int Immunopharmacol. 2011;11:1633–1645. doi: 10.1016/j.intimp.2011.06.004. [DOI] [PubMed] [Google Scholar]
- 61.Lakshmi B, Viswanath B, SaiGopal DV. Probiotics as antiviral agents in shrimp aquaculture. J Pathog. 2013;2013:424123. doi: 10.1155/2013/424123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Seo BJ, Rather IA, Kumar VJ, Choi UH, Moon MR, Lim JH, Park YH. Evaluation of Leuconostoc mesenteroides YML003 as a probiotic against low-pathogenic avian influenza (H9N2) virus in chickens. J Appl Microbiol. 2012;113:163–171. doi: 10.1111/j.1365-2672.2012.05326.x. [DOI] [PubMed] [Google Scholar]
- 63.Botić T, Klingberg TD, Weingartl H, Cencic A. A novel eukaryotic cell culture model to study antiviral activity of potential probiotic bacteria. Int J Food Microbiol. 2007;115:227–234. doi: 10.1016/j.ijfoodmicro.2006.10.044. [DOI] [PubMed] [Google Scholar]
- 64.Wang Z, Chai W, Burwinkel M, Twardziok S, Wrede P, Palissa C, Esch B, Schmid MFG. Inhibitory influence of Enterococcus faecium on the propagation of swine influenza a virus in vitro. PLoS One. 2013;8:e53043. doi: 10.1371/journal.pone.0053043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kawashima T, Hayashi K, Kosaka A, Kawashima M, Igarashi T, Tsutsui H, Tsuji NM, Nishimura I, Hayashi T, Obata A. Lactobacillus plantarum strain YU from fermented foods activates Th1 and protective immune responses. IntImmunopharmacol. 2011;11(12):2017–2024. doi: 10.1016/j.intimp.2011.08.013. [DOI] [PubMed] [Google Scholar]
- 66.Maeda N, Nakamura R, Hirose Y, Murosaki S, Yamamoto Y, Kase T, Yoshikai Y. Oral administration of heat-killed Lactobacillus plantarum L-137 enhances protection against influenza virus infection by stimulation of type I interferon production in mice. Int Immunopharmacol. 2009;9(9):1122–1125. doi: 10.1016/j.intimp.2009.04.015. [DOI] [PubMed] [Google Scholar]
- 67.Guillemard E, Tondu F, Lacoin F, Schrezenmeir J. Consumption of a fermented dairy product containing the probiotic Lactobacillus casei DN-114001 reduces the duration of respiratory infections in the elderly in a randomised controlled trial. Br J Nutr. 2010;103:58–68. doi: 10.1017/S0007114509991395. [DOI] [PubMed] [Google Scholar]
- 68.Boge T, Remigy M, Vaudaine S, Tanguy J, Bourdet-Sicard R, van der Werf S. A probiotic fermented dairy drink improves antibody response to influenza vaccination in the elderly in two randomised controlled trials. Vaccine. 2009;27:5677–5684. doi: 10.1016/j.vaccine.2009.06.094. [DOI] [PubMed] [Google Scholar]
- 69.Olivares M, Diaz-Ropero MP, Sierra S, Lara-Villoslada F, Fonolla J, Navas M, Rodriguez JM, Xaus J. Oral intake of Lactobacillus fermentum CECT5716 enhances the effects of influenza vaccination. Nutrition. 2007;23:254–260. doi: 10.1016/j.nut.2007.01.004. [DOI] [PubMed] [Google Scholar]
- 70.Rautava S, Salminen S, Isolauri E. Specific probiotics in reducing the risk of acute infections in infancy a randomised, double-blind, placebo-controlled study. Br J Nutr. 2009;101:1722–1726. doi: 10.1017/S0007114508116282. [DOI] [PubMed] [Google Scholar]
- 71.Leyer GJ, Li S, Mubasher ME, Reifer C, Ouwehand AC. Probiotic effects on cold and influenza-like symptom incidence and duration in children. Pediatrics. 2009;124(2):e172–179. doi: 10.1542/peds.2008-2666. [DOI] [PubMed] [Google Scholar]
- 72.Yasui H, Kiyoshima J, Hori T. Reduction of influenza virus titer and protection against influenza virus infection in infant mice fed Lactobacillus casei Shirota. Clin Diagn Lab Immunol. 2004;11:675–679. doi: 10.1128/CDLI.11.4.675-679.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mc Milan SJ, Lloyd CM. Prolonged allergen challenge in mice leads to persistent airway remodelling. Clin Exp Allergy. 2004;34:497–507. doi: 10.1111/j.1365-2222.2004.01895.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Eder W, Klimecki W, Yu L, von Mutius E, Riedler J, Braun-Fahrländer C, Nowak D, Holst O, Martinez FD, ALEX-Team Association between exposure to farming, allergies and genetic variation in CARD4/NOD1. Allergy. 2006;61(9):1117–1124. doi: 10.1111/j.1398-9995.2006.01128.x. [DOI] [PubMed] [Google Scholar]
- 75.Ezendam J, de Klerk A, Gremmer ER, van Loveren H. Effects of Bifidobacterium animals administered during lactation on allergic and autoimmune responses in rodents. Clin ExpImmunol. 2008;154:424–431. doi: 10.1111/j.1365-2249.2008.03788.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Forsythe P, Inman MD, Bienenstock J. Oral treatment with live Lactobacillus reuteri inhibits the allergic airway response in mice. Am J Respir Crit Care Med. 2007;175(6):561–569. doi: 10.1164/rccm.200606-821OC. [DOI] [PubMed] [Google Scholar]
- 77.Hougee S, Vriesema AJ, Wijering SC, Knippels LM, Folkerts G, Nijkamp FP, Knol J, Garssen J. Oral treatment with probiotics reduces allergic symptoms in ovalbumin-sensitized mice: a bacterial strain comparative study. Int Arch Allergy Immunol. 2010;151(2):107–117. doi: 10.1159/000236000. [DOI] [PubMed] [Google Scholar]
- 78.Nonaka Y, Izumo T, Izumi F, Maekawa T, Shibata H, Nakano A, Kishi A, Akatani K, Kiso Y. Antiallergic effects of Lactobacillus pentosus strain S-PT84 mediated by modulation of Th1/Th2 immunobalance and induction of IL-10 production. Int Arch Allergy Immunol. 2008;145(3):249–257. doi: 10.1159/000109294. [DOI] [PubMed] [Google Scholar]
- 79.Jiang B, Li Z, Ou B, Duan Q, Zhu G. Targeting ideal oral vaccine vectors based on probiotics: a systematical view. Appl Microbiol Biotechnol. 2019;103:3941–3953. doi: 10.1007/s00253-019-09770-7. [DOI] [PubMed] [Google Scholar]
- 80.Ashraf MU, Kim Y, Kumar S, Seo D, Ashraf M, Bae Y-S. COVID-19 vaccines (revisited) and oral-mucosa vector system as a potential vaccine platform. Vaccines. 2021;9:171. doi: 10.3390/vaccines9020171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Singh K, Rao A. Probiotics: A potential immunomodulator in COVID-19 infecton management. Nutr Res. 2021;87:1–12. doi: 10.1016/j.nutres.2020.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data presented, generated, or analyzed during this study are included in this published article and complied with research standards.