Abstract
Purpose
To study and analyse the radiological and surgical findings of 25 cochlear implantees with SMS type—I cochleovestibular malformation and to compare their outcomes in terms of hearing and speech gains pre- and post-operatively.
Methods
Retrospective analysis of cochlear implanted candidates over a period of 8 year from 3 institutions was undertaken and 25 patients suffering from pre-lingual profound congenital sensori-neural hearing loss along with presence of SMS type I cochleovestibular malformation were studied. Pre-operative radiology, surgical difficulties and complication, and post-operative hearing and speech outcomes upto a period of 2 years, using IT-MAIS scores were noted. Statistical comparison pre- and post-implantation was done using Wilcoxon signed rank test and a p-value of < 0.05 was considered significant.
Results
In 25 cases, we encountered 2 posterior SCC dysplasia, 15 enlarged vestibular aqueducts (LVA), 2 dilated vestibules, 4 superior SCC dysplasia, 2 cases with both posterior and superior SCC dysplasia and of those 1 had a dilated vestibule also. Intra-operatively, CSF leak was observed in 10 patients. Complete electrode insertion and good electrical response (NRT) was detected in all. No facial nerve anomaly was encountered. IT-MAIS scores increased from a pre-operative mean of 4.32 to 34.56 two years post-operatively and this difference was found to be statistically significant.
Conclusion
Our experience with SMS Type I malformations show promising and motivating results with less chances of complications, similar to implant candidates with normal anatomy. The outcomes in this group of patients are definitely not affected by any malformations in semicircular canals, vestibule or vestibular aqueduct.
Introduction
Since the advent of Cochlear implants, they have become the standard of care for management of children suffering from congenital severe to profound sensori-neural hearing loss, as well as adults who lost their hearing ability afterwards in life, post-lingually. However, as much as 20% of these congenital deafened children have one or other kind of structural abnormality of the inner ear thus making the implantation and decision making process in these children very challenging [1]. Their outcomes also differ from their counterparts with normal anatomy and also vary amongst themselves depending on the type and extent of malformation of cochleo-vestibular structures. Accurate definition of the degree and kind of malformations from computed tomography (CT) and magnetic resonance imaging (MRI) pre-implantation is imperative for proper counselling of the parents regarding expected realistic outcomes and for going through an uneventful surgical procedure. While Cochlear and/or vestibular anomaly may not contraindicate implant surgery, the relationship between degree of abnormality and outcomes (surgical and communication) requires clarification [2].
Jackler et al. were the first to provide us with an accepted classification for various types of cochleo-vestibular malformations in 1987 [3]. Sennaroglu et al. further described and detailed this classification in 2002, 2010 and then again recently in 2017, giving us currently the most accepted classification of inner ear malformations [4–6]. However, all these classifications are based on embryology of the developing labyrinth and fail to take into consideration many surgical and prognosticating factors. Also, in order to include a wide range of abnormalities, these lose simplicity and get laborious to remember and apply. Various confusions also arise from the fact that few overlapping and intermediate forms exist in the Sennaroglu classification leading to a lack of uniformity during communication amongst surgeons, radiologists and speech therapists. Classifying these anomalous cochleo-vestibular anatomy cases according to the newly proposed SMS (Sawai Man Singh) classification provides us with ease and simplicity [7]. It is based on the morphology of the cochlea, modiolus and lamina cribrosa rather than on embryogenesis and thus overcomes the aforementioned shortcomings of previous classifications. It is easier to understand and apply radiologically and gives correct estimate of prognosis in a descending fashion. (Table 1).
Table 1.
SMS classification of cochleovestibular malformations [7]
| Cochleovestibular malformation type | Cochlear morphology | Modiolus | Lamina cribrosa | Other anomalies |
|---|---|---|---|---|
| I | Normal | Normal | Normal | Present |
| IIa | Abnormal | Complete but smaller | Normal | Present or absent |
| IIb | Abnormal | Partially defective | Normal | Present or absent |
| IIIa | Abnormal | Absent | Normal | Present or absent |
| IIIb | Abnormal | Absent | Deficient | Present or absent |
| IV | Absent | Absent | Absent | Present or absent |
As per the SMS classification, type-I anomaly has a normal cochlea, modiolus and lamina cribrosa with malformations in other parts of the inner ear like dilated vestibular aqueduct, vestibular dysplasia, and semi-circular canal dysplasia or agenesis (Fig. 1). Type-II cochleovestibular anomaly has an abnormal cochlea, with a normal modiolus and lamina cribrosa. Type-III includes an abnormal cochlear morphology with a completely absent modiolus. Type-III is further sub classified as type-IIIa, having a normal lamina cribrosa and type-IIIb with a deficient lamina. Lastly, type-IV refers to an abnormal cochlea along with absence of both modiolus and lamina cribrosa, thus giving it a poor prognosis and making it a contra-indication for cochlear implantation. The prognosis of outcomes in this classification goes on decreasing from type-I through type-IV and the chances of encountering a complication intra-operatively increases.
Fig1.
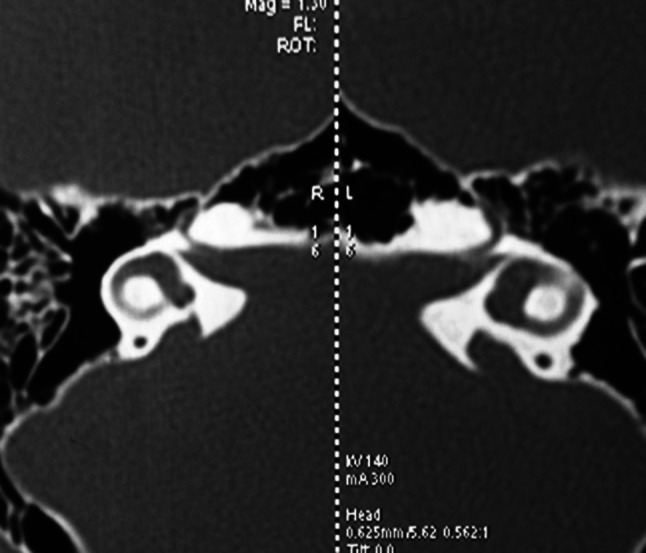
HRCT temporal bone scan of a patient with SMS type I malformation (bilateral enlarged vestibular aqueducts)
Cochleovestibular malformation type I (Fig. 2) includes all those malformations of the inner ear that have no direct bearing on cochlear implantation. The cochlea itself is normal. Malformation is limited to other inner ear structures, and includes semicircular canal (SCC) dysplasia or agenesis, a dysplastic vestibule, or an enlarged vestibular aqueduct. With the exception of a vestibular duct anomaly, the surgery will not be any different to that conducted in a patient with normal cochlear anatomy. Even in patient with an enlarged vestibular aqueduct, the only difference will be a minor pulsatile cerebrospinal fluid (CSF) leak during cochleostomy, with no significant egress of fluid. This will not have any effect on electrode insertion and therefore any electrode can be used. Thus here we studied and analysed 25 cochlear implanted cases across three tertiary care institutions over a span of 8 years who were classified using the SMS classification instead of the Sennaroglu classification and all were designated as type-I SMS malformation.
Fig2.
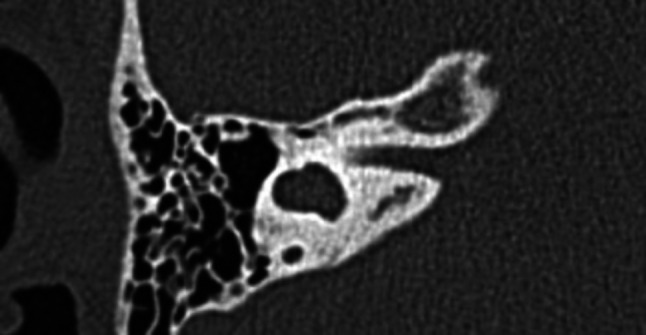
HRCT temporal bone scan of a patient with SMS type I malformation (Dilated vestibule with dysplastic SCC)
Material and Methods
The study was a retrospective review carried out from records of over a period of 8 years from January 2012 to December 2019 across three tertiary care institutions of India. Children with congenital bilateral profound sensori-neural hearing loss who were radiologically diagnosed with cochleovestibular malformation type-I according to the SMS classification and underwent cochlear implantation were taken into consideration as the study population. Children with any other type of malformation or children who did not undergo cochlear implantation were excluded from the study. All patients were operated on using the Transmastoid posterior tympanotomy approach through the facial recess and all findings, including intra-operative and post-operative complications that we faced along with their management strategies were noted.
Post-operatively each implanted candidate underwent switch-on of the implant 4 weeks after surgery as per the institutional protocols. Speech therapy and rehabilitation was then carried out by a trained audiologist and speech therapist who was also involved in pre-operative counselling of the parents regarding post-operative therapy sessions. The children were followed up by their respective audiologists in each institution and their hearing and speech progress was recorded using the Infant toddler—Meaningful auditory integration scale (IT-MAIS)[8].
The Infant–Toddler Meaningful Auditory Integration Scale (IT-MAIS) is a modification of the Meaningful Auditory Integration Scale (MAIS) given by Robbins et al. in 1991 [9]. It is a structured interview schedule designed to assess the child’s spontaneous responses to sound in her/his everyday environment. The assessment is based upon information provided by the child’s parent(s) in response to 10 probes each with specific scoring criteria. These 10 probes assess three main areas including vocalization behaviour, alerting to sounds and deriving meaning from sound. It is a more practical scale since it does not depend on assessment in hospital environment alone. IT-MAIS scoring system was done pre-operatively and compared with 2 year post-operative scores.
The age-adjusted mean scores in accordance with MAIS were compared between the pre-implantation and post-implantation group. Statistical significance was ascertained using the Wilcoxon signed rank test. p—value when < 0.05was considered to be significant. SPSS® version 24.0 was used to conduct analysis of the results.
Results
On searching through complete records, we found 25 patients who had a type-I SMS malformation and underwent cochlear implantation along with meeting all other inclusion criteria. The mean age of these patients was 4 years ranging between 1.6 to 7.8 years. Two of these had posterior SCC dysplasia, 15 had an enlarged vestibular aqueduct (taken as diameter larger than 1.5 mm at midpoint [10]), 2 had a dilated vestibule, 4 had dysplasia of superior SCC, 1 suffered from both posterior and superior SCC dysplasia and 1 had dysplasia of both superior and posterior SCC along with a dilated vestibule.
All patients were operated using the transmastoid posterior tympanotomy-facial recess approach. Intra-operatively, CSF leak was observed in 10 patients. All of these were cases of large vestibular aqueduct anomaly, although not all cases with LVA had a CSF leak. These were comfortably managed with allowing the CSF to egress a little and then using soft tissue plugs around the electrodes to seal the cochleostomy site. Rest no major complication was encountered. Six of our patients were implanted with ‘HiFocus’ electrodes from Advance Bionics® which are mid-scalar, half-banded partially curved electrodes and the rest 18 cases were implanted with ‘Rest’ electrodes from Cochlear® which are full banded straight electrodes. The choice of electrodes was taken bearing the availability, choice and financial constraints of the family of the candidate to be implanted.
All cases had complete electrode insertion and Neural Response Telemetry (NRT) detected a good response in all the patients. No patient had any kind of facial nerve anomaly and none suffered from facial paresis/palsy. There were no incidences of facial nerve twitching or meningitis in the post-operative period. No patient reported any imbalance, difficulty in walking or persisting nausea/vomiting.
All patients had a mastoid X-ray post implantation (modified stenver’s view) to check the correct positioning of electrodes inside the cochlea. All patients had a stable hospital course and were discharged post-operatively on day third on oral antibiotics.
Post discharge, switch on of device was done after 3 weeks and then further follow was kept by the audiologist and speech therapist. The IT-MAIS scores of all the candidates were recorded pre-operatively as well as up till 2 years of follow up. Pre-operatively these scores ranged from 2 to 8 with a mean score of 4.32 and after two years of auditory-verbal therapy post implantation these scores increased to a mean score of 34.56 ranging from 32 to 38. Using the Wilcoxon signed rank test, the difference between the pre and post-operative scores was found to be statistically significant (p-value < 0.05), thus indicating significant benefit (Table 2).
Table 2.
Patient profile along with surgical, radiological and audiological data of all the cases
| Patient number | Age at surgery (years) | CT scan and MRI findings | Facial nerve status | IT-MAIS scores | Intra-operative complications | Implant type | Surgical approach | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Cochlear morphology | Modiolus | Lamina cribrosa | Any other | Pre-operative | Post-operative | ||||||
| 1 | 3 | Normal | Normal | Normal | PSCC dysplasia | Normal | 5 | 37 | None | Rest | PT-FR |
| 2 | 4.5 | Normal | Normal | Normal | LVA | Normal | 3 | 34 | Mild CSF leak | Rest | PT-FR |
| 3 | 2.8 | Normal | Normal | Normal | Dilated vestibule | Normal | 4 | 36 | None | Rest | PT-FR |
| 4 | 5.9 | Normal | Normal | Normal | LVA | Normal | 6 | 33 | Mild CSF leak | Rest | PT-FR |
| 5 | 2.2 | Normal | Normal | Normal | SSCC dysplasia | Normal | 3 | 36 | None | Rest | PT-FR |
| 6 | 6.5 | Normal | Normal | Normal | LVA | Normal | 5 | 32 | Mild CSF leak | Rest | PT-FR |
| 7 | 2.3 | Normal | Normal | Normal | SSCC dysplasia | Normal | 3 | 37 | None | Rest | PT-FR |
| 8 | 2.7 | Normal | Normal | Normal | LVA | Normal | 3 | 35 | None | Rest | PT-FR |
| 9 | 5 | Normal | Normal | Normal | LVA | Normal | 5 | 32 | Mild CSF leak | Rest | PT-FR |
| 10 | 4 | Normal | Normal | Normal | LVA | Normal | 3 | 33 | Mild CSF leak | Rest | PT-FR |
| 11 | 6.8 | Normal | Normal | Normal | LVA | Normal | 7 | 33 | Mild CSF leak | Rest | PT-FR |
| 12 | 7.8 | Normal | Normal | Normal | LVA | Normal | 7 | 38 | Mild CSF leak | HiFocus mid-scala | PT-FR |
| 13 | 1.6 | Normal | Normal | Normal | PSCC and SSCC dysplasia | Normal | 2 | 36 | None | HiFocus mid-scala | PT-FR |
| 14 | 3.7 | Normal | Normal | Normal | SSCC dysplasia | Normal | 4 | 33 | None | HiFocus mid-scala | PT-FR |
| 15 | 3.9 | Normal | Normal | Normal | LVA | Normal | 4 | 35 | Mild CSF leak | HiFocus mid-scala | PT-FR |
| 16 | 3 | Normal | Normal | Normal | LVA | Normal | 4 | 36 | Mild CSF leak | HiFocus mid-scala | PT-FR |
| 17 | 2 | Normal | Normal | Normal | LVA | Normal | 3 | 34 | None | HiFocus mid-scala | PT-FR |
| 18 | 2.8 | Normal | Normal | Normal | PSCC dysplasia | Normal | 4 | 35 | None | Rest | PT-FR |
| 19 | 4.3 | Normal | Normal | Normal | LVA | Normal | 6 | 32 | None | Rest | PT-FR |
| 20 | 6.8 | Normal | Normal | Normal | LVA | Normal | 8 | 34 | None | Rest | PT-FR |
| 21 | 3 | Normal | Normal | Normal | LVA | Normal | 4 | 35 | None | Rest | PT-FR |
| 22 | 4.2 | Normal | Normal | Normal | Dilated vestibule | Normal | 4 | 33 | None | Rest | PT-FR |
| 23 | 3 | Normal | Normal | Normal | SSCC dysplasia | Normal | 3 | 35 | None | Rest | PT-FR |
| 24 | 6.2 | Normal | Normal | Normal | LVA | Normal | 5 | 35 | Mild CSF leak | Rest | PT-FR |
| 25 | 2.6 | Normal | Normal | Normal | PSCC and SSCC dysplasia; dilated vestibule | Normal | 3 | 35 | None | Rest | PT-FR |
‘Rest’ electrodes (Cochlear, Sydney, Australia) are full−banded, straight electrodes. HiFocus mid−scala electrodes (Advance Bionics, Valencia, California) are half−banded, partially curved electrodes. SSCC Superior Semi−circular Canal; PSCC Posterior Semi−circular Canal; LVA Large Vestibular Aqueduct; PT−FR Posterior Tympanotomy−Facial Recess
Discussion
Cochlear implants have had a great impact in management of severe to profoundly deaf individuals. The greatest benefit is seemingly derived in congenital sensori-neural hearing loss children where life altering outcomes are obtained, thus making their use in this population very widespread. Although many surgeons have shared their experience of dealing with such cases with good results, very few are actually dealing with those handfuls of patients who deviate from the normal and end up having an abnormal cochleovestibular anatomy.
Patients with cochleovestibular malformation make up about 20% of all the patients undergoing an HRCT temporal bone scan for congenital sensori-neural deafness [1]. This is a significantly huge percentage number, thus making it very clear of the need of a system to classify these malformations, for uniformity in exchange and discussion of ideas amongst various surgeons as well as for protocol formation for management of these patients. The most accepted classification till date has been that by Sennaroglu et al., but it falls short on many levels the most obvious of which is simplicity thus making its uniform application and understanding very difficult [6]. Also it has many overlapping areas, thus making it difficult at times to correctly categorise an anomaly based on radiology. Finally, it is based on embryology which in many cases leads to its deviation from correct surgical and prognostication chronology, thus leading to confusion. Thus here in our study, we decided to implement and use the SMS system of classification for our subjects. It is a simple classification covering all types of cochleovestibular malformations. It uses the radiological criteria only instead of relying on embryology, covering the malformations from a surgeon’s point of view allowing for better and chronological pre-operative grouping of anomalies leading to easier prognosis assessment and counselling of parents [11].
The SMS type I class of cochleovestibular malformations is the most favourable scenario, both for the surgeon as well as the patient. It has a normal cochlear anatomy and the malformation is limited to other vestibular structures like the SCC, the vestibule and the endolymphatic duct. In our study, we encountered 25 class I patients, all of which were implanted with ease. The only complication we encountered was a minor pulsatile CSF leak in 10 cases with large vestibular aqueduct. Various studies have reported complications similar to ours with some even giving incidences of CSF gushers with SMS type I malformation, especially with LVA anomaly. Lee et al. in their study of 23 children published in 2010 encountered a CSF gusher in 13% of cases operated for cochlear implantation with LVA anomaly [12]. Dettman et al. studied 48 cases with cochleovestibular malformation out of which they encountered a CSF gusher in 11 cases, 2 of which were of purely vestibular pathology [2]. Hongjian et al. described a 4.8% incidence of CSF gusher in LVA cases in their study of 442 patients [13]. Grover et al. in their study of 436 cochlear implanted cases reported 19 to be of SMS type I and of these 7 had a mild CSF leak with no incidence of gusher [7]. Harker et al. also reported no incidence of CSF gusher in implantation of patients with LVA [14]. In totality the literature is varied on the occurrence of CSF gusher in purely vestibular pathologies, similar to SMS type I. In our study we did not encounter any case of CSF gusher out of all 25 cases, 10 of these although had a mild leak which was managed by packing the cochleostomy site with soft tissue of temporalis muscle. Additional measures have been mentioned in literature including placing the patient in reverse Trendelenburg position to slow the flow of CSF, intra-operative lowering of pCO2 and administration of mannitol and acetazolamide, though none of these were used in this study [15–17].
According to Papsin, reports that include minor CSF leaks may lead to an overestimation of the number of cases with CSF gushers and may help explain the wide range in incidence of gushers and leaks in literature [18]. A ‘gusher’ is usually mentioned as an egress of profuse clear fluid upon making an opening into the inner ear [5, 18]. Whereas a gentle intermittent flow of clear fluid in small quantities upon opening the cochlea which stops after a few minutes is termed as ‘oozing’ [19, 20]. Children with vestibular anomalies should not have increased risk of CSF gushers, despite the size of endolymphatic sac or duct and vestibule, as there is usually no communication with the CSF [2]. A CSF gusher may occur in these cases with associated cochlear malformations, or it is more likely to occur in cases with abnormal patency of the cochlear aqueduct or deficiency of the modiolus and fundus of the internal auditory canal where there is abnormal communication between the subarachnoid and perilymphatic space, as has been stated in SMS classification originally. Thus a CSF gusher seems to be unlikely in SMS type I class including LVA patients, as was the finding in this study.
The facial nerve might also have an anomalous course in cases with abnormal cochleovestibular anatomy [21]. Anomalous facial nerve probably originates because of a dilated vestibule and malformed lateral SCC [5]. The facial nerve in such scenarios gets displaced antero-medially, increasing the chances of injury during the procedure thus necessitating an alternative route for implantation to avoid a posterior tympanotomy [11]. In our study we found no such displacement of facial nerve both pre-operatively on radiology and intra-operatively and posterior tympanotomy could be safely done in all the cases.
We did not have to use any specific type of electrodes for these patients and the choice was based completely on the parents of the patient, subject to availability. This is also clearly stated in SMS classification that any kind of electrodes can be used in type I and II malformations [7].
Discussion is also warranted on the speech and auditory outcomes of these SMS type I cases, as in this study we found a significant improvement in IT-MAIS scores of all the implanted children post-operatively. Decreased performance expectations in patients with inner ear malformations secondary to reduced spiral ganglion counts have led some to question whether cochlear implantation in patients with LVA or other vestibular malformations can safely improve auditory functions [22, 23]. Significant auditory gains have been reported in studies of Lee et al. [12] and Grover et al. [24], who in a study in 2017 concluded that children with LVA (which is part of spectrum of SMS type I) perform similar to patients with normal cochleo-vestibular anatomy. Isaiah et al. also conclude in their study with 381 cases that outcomes in patients with LVA and other vestibular anomalies were similar to those obtained in implantees with normal anatomy [25]. Our experience here, with SMS type I cases clearly shows that these cases are perfectly suitable candidates for implant surgery and can even have results similar to normal implanted. Also, the surgery can be performed in a similar fashion to normal candidates with few more considerations and very rare incidences of serious complications. Multiple other complications such as wound infections, post-operative pain and vomiting, local swelling, vertigo and meningitis have been described in literature [26]. Till the last follow up we had of our patients no such complication was reported.
Thus, children with inner ear malformations present a significant challenge to even the most experienced clinician due to uncertainty regarding surgical feasibility and performance outcomes [24]. The need of a simple and uniform classification is must so as to not add diagnostic and management difficulty in these cases. The SMS classification used here was found to be simple in implementation and useful for predicting the intra-operative complications as well in giving reasonable prognosis guide for the pre-operative counselling of patient’s parents. The results we obtained in type I malformed cases were encouraging and comparable to implanted candidates with normal anatomy without any association with significant complications.
Conclusion
Cochlear malformations, although a challenge for the surgeon, are not contraindications to cochlear implantation. A simple and uniformly applicable classification system for these is a must for appropriate prognostication and correct decision making. Our experience with SMS Type I malformations show promising and motivating results, similar to implant candidates with normal anatomy. The outcomes in this group of patients are definitely not affected by any malformations in semicircular canals, vestibule or vestibular aqueduct.
Compliance with Ethical Standards
Conflict of Interest
All Authors declare that they have no conflict of Interest. This research did not receive any specific grant from funding agencies in the public, commercial, or not for profit sectors.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent
Informed consent was obtained from all individual participants included in the study..
Author Contributions Statement
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Mohnish Grover and Shivam Sharma. The first draft of the manuscript was written by Shivam Sharma and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Mohnish Grover, Email: drmohnish.aiims@gmail.com.
Shivam Sharma, Email: shivam92@hotmail.com.
Sunil Samdani, Email: sunilsamdhani@gmail.com.
Gaurav Gupta, Email: drgauravgupta24@gmail.com.
Chappati Preetam, Email: drcpreetam@aiimsbhubaneswar.edu.in.
Kriti Gera, Email: kriti.gera1095@gmail.com.
Jairaj Kumar Vaishnav, Email: jai2016@gmail.com.
Mahendra Kumar Hada, Email: drmshada@rediffmail.com.
References
- 1.Jensen S. Malformation of the inner ear in deaf children. Acta Radio. 1969;286:1–97. [PubMed] [Google Scholar]
- 2.Dettman S, Sadeghi-Barzalighi A, Ambett R, Dowell R, Trotter M, Briggs R. Cochlear implants in forty-eight children with cochlear and/or vestibular abnormality. Audio Neurotol. 2011;16(4):222–232. doi: 10.1159/000320608. [DOI] [PubMed] [Google Scholar]
- 3.Jackler RK, Luxfor WM, House WF. Congenital malformations of the inner ear: a classification based on embryogenesis. The Laryngoscope. 1987;97(S40):2–14. doi: 10.1002/lary.5540971301. [DOI] [PubMed] [Google Scholar]
- 4.Sennaroglu L, Saatci I. A new classification for cochleovestibular malformations. The Laryngoscope. 2002;112(12):2230–2241. doi: 10.1097/00005537-200212000-00019. [DOI] [PubMed] [Google Scholar]
- 5.Sennaroglu L. Cochlear implantation in inner ear malformations–a review article. Cochlear implants Int. 2010;11:4–41. doi: 10.1002/cii.416. [DOI] [PubMed] [Google Scholar]
- 6.Sennaroğlu L, Bajin MD. Classification and current management of inner ear malformations. Balkan Med J. 2017;34(5):397. doi: 10.4274/balkanmedj.2017.0367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Grover M, Sharma S, Preetam C, Gupta G, Samdani S, Agarwal S, Singh SN, Sharma MP. New SMS classification of cochleovestibular malformation and its impact on decision-making. J Laryngol Otol. 2019;133(5):368–375. doi: 10.1017/S0022215119000884. [DOI] [PubMed] [Google Scholar]
- 8.Zimmerman-Phillips S, Robbins AM, Osberger MJ. Assessing cochlear implant benefit in very young children. Ann Otol Rhinol Laryngol Suppl. 2000;109(12):42–43. doi: 10.1177/0003489400109S1217. [DOI] [PubMed] [Google Scholar]
- 9.Robbins AM, Renshaw JJ, Berry SW. Evaluating meaningful auditory integration in profoundly hearing-impaired children. Am J otol. 1991;12:144–150. [PubMed] [Google Scholar]
- 10.Valvassori GE, Clemis JD. The large vestibular aqueduct syndrome. Laryngo. 1978;88(5):723–728. doi: 10.1002/lary.1978.88.5.723. [DOI] [PubMed] [Google Scholar]
- 11.Grover M, Kanodia A, Gupta G, Preetam C, Samdani S. Application of the new SMS system of cochleovestibular anomalies: our experience with nine cases of type III anomaly. Eur Arch Otorhinolaryngol. 2020;11:1–6. doi: 10.1007/s00405-020-06114-4. [DOI] [PubMed] [Google Scholar]
- 12.Lee KH, Lee J, Isaacson B, Kutz JW, Roland PS. Cochlear implantation in children with enlarged vestibular aqueduct. Laryngo. 2010;120(8):1675–1681. doi: 10.1002/lary.20987. [DOI] [PubMed] [Google Scholar]
- 13.Hongjian L, Guangke W, Song M, Xiaoli D, Daoxing Z. The prediction of CSF gusher in cochlear implants with inner ear abnormality. Acta Otolaryngol. 2012;132(12):1271–1274. doi: 10.3109/00016489.2012.701328. [DOI] [PubMed] [Google Scholar]
- 14.Harker LA, Vanderheiden S, Gentile N, Veazey D, McCleary E (1999) Multichannel cochlear implantation in children with large vestibular aqueduct syndrome. Annals Otol Rhinol Laryngol 108(4_suppl):39–43 [DOI] [PubMed]
- 15.Wootten CT, Backous DD, Haynes DS. Management of cerebrospinal fluid leakage from cochleostomy during cochlear implant surgery. The Laryngoscope. 2006;116(11):2055–2059. doi: 10.1097/01.mlg.0000240286.43289.87. [DOI] [PubMed] [Google Scholar]
- 16.Loundon N, Rouillon I, Munier N, Marlin S, Roger G, Garabedian EN. Cochlear implantation in children with internal ear malformations. Otol Neurotol. 2005;26(4):668–673. doi: 10.1097/01.mao.0000178126.58859.a9. [DOI] [PubMed] [Google Scholar]
- 17.Eisenman DJ, Ashbaugh C, Zwolan TA, Arts HA, Telian SA. Implantation of the malformed cochlea. Otol Neurotol. 2001;22(6):834–841. doi: 10.1097/00129492-200111000-00020. [DOI] [PubMed] [Google Scholar]
- 18.Papsin BC. Cochlear implantation in children with anomalous cochleovestibular anatomy. The Laryngoscope. 2005;115(S106):1–26. doi: 10.1097/00005537-200501001-00001. [DOI] [PubMed] [Google Scholar]
- 19.Phelps PD, King A, Michaels L. Cochlear dysplasia and meningitis. Am J otology. 1994;15(4):551–557. [PubMed] [Google Scholar]
- 20.Eftekharian A, Amizadeh M. Cerebrospinal fluid gusher in cochlear implantation. Cochlear Implants Int. 2014;15(3):179–184. doi: 10.1179/1754762814Y.0000000069. [DOI] [PubMed] [Google Scholar]
- 21.Sennaroglu L, Sarac S, Ergin T. Surgical results of cochlear implantation in malformed cochlea. Otol Neurotol. 2006;27(5):615–623. doi: 10.1097/01.mao.0000224090.94882.b4. [DOI] [PubMed] [Google Scholar]
- 22.Lin CY, Lin SL, Kao CC, Wu JL. The remediation of hearing deterioration in children with large vestibular aqueduct syndrome. Auris Nasus Larynx. 2005;32(2):99–105. doi: 10.1016/j.anl.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 23.Boston M, Halsted M, Meinzen-Derr J, Bean J, Vijayasekaran S, Arjmand E, Choo D, Benton C, Greinwald J (2007) The large vestibular aqueduct: a new definition based on audiologic and computed tomography correlation. Otolaryngol Head Neck Surg 136(6):972–7 [DOI] [PubMed]
- 24.Grover M, Sharma S, Bhargava S, Singh SN, Gupta G, Sharma MP. Cochlear implantation in children with anomalous cochleovestibular anatomy: our experience. Indian J Otolaryngol Head Neck Surg. 2017;69(4):504–508. doi: 10.1007/s12070-017-1209-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Isaiah A, Lee D, Lenes-Voit F, Sweeney M, Kutz W, Isaacson B, Roland P, Lee KH. Clinical outcomes following cochlear implantation in children with inner ear anomalies. Int J Pediatr Otorhinolaryngol. 2017;1(93):1–6. doi: 10.1016/j.ijporl.2016.12.001. [DOI] [PubMed] [Google Scholar]
- 26.Ciorba A, Bovo R, Trevisi P, Rosignoli M, Aimoni C, Castiglione A, Martini A. Postoperative complications in cochlear implants: a retrospective analysis of 438 consecutive cases. Eur Arch Otorhinolaryngol. 2012;269(6):1599–1603. doi: 10.1007/s00405-011-1818-1. [DOI] [PubMed] [Google Scholar]


