Abstract
Background
Since the coronavirus disease 2019 (COVID-19) has risen, several risk factors have been identified, predicting a worse outcome. It has been speculated that patients with Multiple sclerosis (MS) have an increased risk for a severe course of COVID-19 due to a suspected higher vulnerability. Therefore, we aimed to analyze the impact of comorbid MS on the outcome of patients with COVID-19 in Germany.
Methods
We conducted a retrospective cross-sectional study using the administrative database of all hospitalized patients diagnosed with PCR-confirmed COVID-19 (n = 157,524) in Germany during 2020. The cohort was stratified according to the presence (n = 551) or absence (n = 156,973) of comorbid MS, including discrimination of MS subtypes. Primary outcome measures were admission to the intensive care unit (ICU), use of invasive or non-invasive ventilation, and in-hospital mortality. Differences were investigated using rates and odds ratios as estimates. Pooled overall estimates, sex-stratified estimates, age-group stratified estimates, and MS subtype stratified estimates were calculated for all outcomes under the random-effects model.
Results
Among 157,524 patients hospitalized with COVID-19, 551 had a concurrent MS diagnosis (0.3%). Overall, univariate analysis showed lower rates of ICU admission (17.1% versus 22.7%, p < 0.001), lower use of ventilation (9.8% versus 14.5%, p < 0.001) and lower in-hospital mortality (11.1% versus 19.3%, p < 0.001) among COVID-19 patients with comorbid MS. This finding was stable across the subgroup analysis of sex and MS subtype but was attenuated by age-stratification, confirming equal odds of in-hospital mortality between COVID-19 patients with and without MS (log OR: 0.09 [95% CI: − 0.40, 0.59]).
Conclusions
Although there might be differences in risk within the MS patients’ population, this large-scale nationwide analysis found no evidence for a worse outcome of COVID-19 in patients with comorbid MS compared to non-MS individuals.
Supplementary Information
The online version contains supplementary material available at 10.1186/s42466-021-00143-y.
Keywords: Multiple sclerosis, COVID-19, SARS-CoV-2, Germany
Background
The coronavirus disease 2019 (COVID-19) pandemic has spread rapidly, threatening global health [1]. The severe acute respiratory coronavirus 2 (SARS-CoV-2) high virulence can cause severe pneumonia with extensive lung inflammation that has caused innumerable victims worldwide. Since the beginning of the pandemic, there have been global efforts to gather information on the risk of infection and the specific consequences for patients with Multiple sclerosis (MS) [2, 3]. MS is the most common demyelinating disease of the central nervous system (CNS). The immunopathogenesis of MS involves CNS inflammation, including autoreactive lymphocytes, resulting in immunomodulating and immunosuppressive treatment approaches [4–6]. It has been speculated that patients with MS are at higher risk for a severe course of COVID-19, although robust evidence is lacking. Nevertheless, such assumptions have led to uncertainties in the MS community [7].
Different risk factors for worse outcomes of COVID-19 have been identified in the general population, including older age, male sex, and other comorbid conditions, such as hypertension, diabetes, and chronic obstructive pulmonary disease [8–11]. These comorbidities are also common in patients with Parkinson’s disease [12] or stroke, where comorbid COVID-19 has been associated with high in-hospital mortality [13]. Various reports and studies in patients with MS have evolved age and disability as important risk factors for a more severe course of COVID-19 within the MS patients’ population [3, 14–16]. However, there is lacking knowledge about the general role of comorbid MS as a factor influencing the outcome in COVID-19 patients compared to non-MS individuals.
Therefore, this study aimed to investigate the impact of comorbid MS on the outcome of patients with COVID-19 using comprehensive administrative diagnosis-related group (DRG) data from Germany.
Methods
Data source and study sample
The data that support the findings of this study are available from the corresponding author upon reasonable request. This is a German retrospective cross-sectional study using the administrative diagnosis-related group (DRG) database (Data transmission according to §21 KHEntgG and §24 para. 2 KHG; official data on file, source: Institut für das Entgeltsystem im Krankenhaus, InEK, www.g-drg.de). In Germany, all inpatient cases are encoded according to ICD-10-GM and operating and procedure keys (OPS) issued by the Federal Institute for Drugs and Medical Devices (BfArM). We included all patients hospitalized in Germany between January 1, 2020, to December 31, 2020, with the ICD-10 diagnosis of U07.1 (COVID-19, PCR-confirmed, n = 157,524). This cohort was divided into patients with (n = 551) and without (n = 156,973) comorbid MS defined by concurrent ICD-10 diagnosis of G35.1- (relapsing-remitting MS, n = 106), G35.2- (primary progressive MS, n = 76), G35.3- (secondary progressive MS, n = 99), and G35.9 (unspecified MS, n = 270). Patients being transferred once or multiple times from one hospital to another were censored appropriately to avoid multiple counting cases (excluding “discharge key 06”). The patient and outcome identification process of this study is given in the online supplement.
Outcomes
Primary outcomes were admission to the intensive care unit (ICU), use of ventilation, and in-hospital mortality among COVID-19 patients with and without a concurrent main or secondary diagnosis of MS. The use of ventilation was defined by invasive or non-invasive ventilation for at least 1 h. In-hospital mortality was obtained using discharge key 07 (death during hospital stay).
Statistical analysis
For descriptive analysis, results are reported as mean and standard deviation (SD) for continuous variables and absolute numbers and percentages for categorical variables. Univariate analysis was performed with the chi-squared test (χ2) and t-test, respectively. Subgroup analysis of the primary outcomes was investigated using odds ratio (OR) as estimate to compare the likelihoods between COVID-19 patients with and without a concurrent MS diagnosis. Pooled overall estimates, sex-stratified estimates, age-group stratified estimates, and MS subtype stratified estimates were calculated for all outcomes under the random-effects model (DerSimonian-Laird). Differences of subgroup-stratified estimates within and in-between the primary outcome analysis were investigated with the Cochran test for heterogeneity and I2 statistics. P < 0.05 was set as the level of statistical significance. Calculations were performed with the Stata Statistical Software Release 17 for Mac (StataCorp LP, College Station, TX).
Results
Demographics
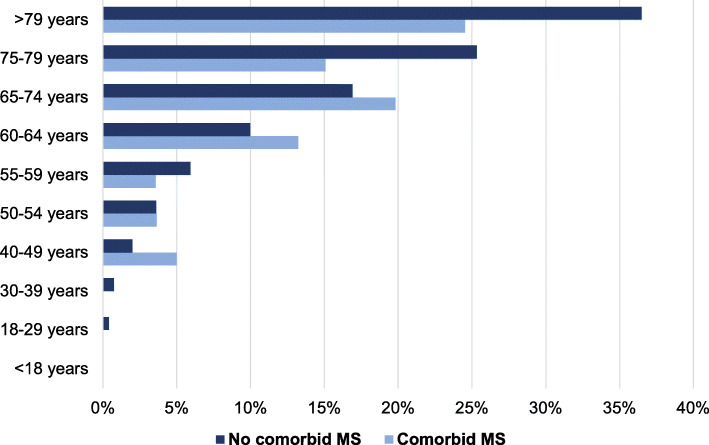
Among 157,524 patients hospitalized with COVID-19, 551 had a concurrent diagnosis of MS (0.3%). There was a higher percentage of women in the cohort of patients with comorbid MS (62.8% versus 48.2%, p < 0.001). Age was lower in patients with comorbid MS (60.6 ± 4.5 versus 66.9 ± 7.3, p < 0.001), in line with a lower rate of patients over 60 years (52.5% versus 68.2%, p < 0.001). COVID-19 patients with comorbid RRMS patients were younger compared to those with comorbid PPMS (51.2 ± 3.4 versus 61.6 ± 4.7, p < 0.001), SPMS (63.5 ± 6.2, p < 0.001), or unspecified MS (62.2 ± 4.7, p < 0.001).
Univariate analysis
Univariate analysis of the primary outcome measures revealed lower rates for ICU admission (17.1% versus 22.7%, p < 0.001), lower use of ventilation (9.8% versus 14.5%, p < 0.001), and lower in-hospital mortality (11.1% versus 19.3%, p < 0.001) among COVID-19 patients with comorbid MS (Table 1).
Table 1.
General characteristics of COVID-19 patients stratified by the presence of comorbid MS
| Comorbid MS (n = 551) | No comorbid MS (n = 156,973) | P-Value | |
|---|---|---|---|
| Female (n, %) | 346, 62.8 | 75,593, 48.2 | < 0.001 |
| Male (n, %) | 205, 37.2 | 81,380, 51.8 | < 0.001 |
| Age (mean ± SD) | 60.3 ± 4.5 | 66.9 ± 7.3 | < 0.001 |
| Age ≥ 60 years (n, %) | 289, 52.5 | 106,986, 68.2 | < 0.001 |
| Admission to ICU (n, %) | 94, 17.1 | 35,564, 22.7 | < 0.001 |
| Ventilation (n, %) | 54, 9.8 | 22,692, 14.5 | < 0.001 |
| In-hospital mortality (n, %) | 61, 11.1 | 30,246, 19.3 | < 0.001 |
Subgroup analysis
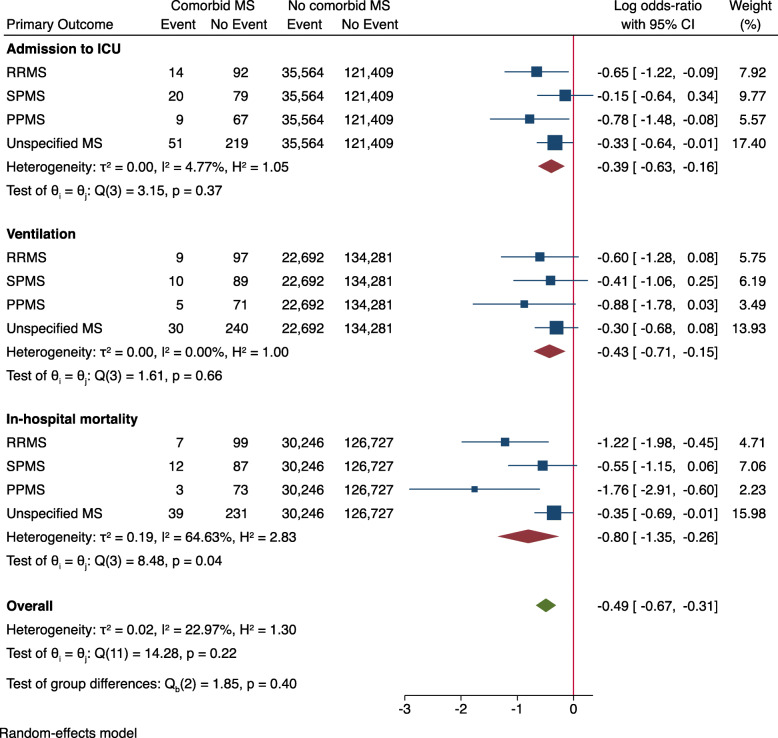
Sex- and MS subtype-stratified analysis demonstrated lower OR in patients with comorbid MS for all primary outcome measures (Figs. 1 and 2). When stratified by age groups, there was no significant difference in the OR for in-hospital mortality between patients with and without comorbid MS (log OR: 0.09 [95% CI: − 0.40, 0.59], Fig. 3). Age-stratified analysis was in line with the sex and MS subtype stratified findings, showing lower OR of admission to ICU (log OR: -0.43 [95% CI: − 0.66, − 0.21]) and use of invasive or non-invasive ventilation (log OR: -0.51 [95% CI: − 0.79, − 0.23]) in patients with comorbid MS.
Fig. 1.
Primary outcome parameters stratified by sex. Log odds-ratio < 0 indicate lower risk for patients with comorbid MS. Abbreviations: ICU = intensive care unit
Fig. 2.
Primary outcome parameters stratified by MS-subtype. Log odds-ratio < 0 indicates a lower risk for patients with comorbid MS. Abbreviations: ICU = intensive care unit
Fig. 3.
Primary outcome parameters stratified by age-group. Log odds-ratio < 0 indicates a lower risk for patients with comorbid MS. Abbreviations: ICU = intensive care unit
Discussion
This is the first large-scale nationwide analysis from Germany investigating the outcome of COVID-19 in patients with comorbid MS. Overall, 0.3% of all hospitalized COVID-19 patients had a concurrent MS diagnosis. We found no evidence for a worse outcome of COVID-19 in patients with MS compared to non-MS patients.
Indeed, all primary outcome rates were lower in MS patients compared to the non-MS cohort. The overall in-hospital mortality rate of COVID-19 patients with comorbid MS was 10.9%. Although the lower odds for MS patients were stable over the subgroup analysis of MS subtypes and sex, the age-group analysis revealed no difference between patients with and without comorbid MS in terms of in-hospital mortality. This is the most reasonable explanation for the observed discrepancies of the pooled univariate analysis, as the MS patients in our study were younger than the non-MS individuals. In general, higher age is known to be an essential risk factor for a severe course of COVID-19 that was strongly associated with higher mortality. Recent data from England, Geneva in Switzerland, and Spain confirmed age as an independent predictor of dying from a SARS-CoV-2 infection [17–20]. The colleagues found an increase in the infection fatality ratio with higher age [17]. Although there is limited in-between comparability of these data due to different age-group definitions, the data from Geneva indicates an infection fatality ratio of 5.6% in patients older than 64 years, England report 11.6% in patients older than 74 years and Spain reports 7.2% in patients older than 79 years. All countries observed an infection fatality ratio far below 1% in SARS-CoV-2 antibody-positive individuals under 50 years. The infection fatality ratios of these different studies were calculated based on the serological status (presence of SARS-CoV-2 antibodies). Therefore, they are not directly transferable to our findings because our data refer to all hospitalized patients with laboratory-confirmed COVID-19 in Germany. However, we observed an equal dynamic of the in-hospital mortality that mirrors the observations of the colleagues. We also found that the in-hospital mortality of COVID-19 patients increased with age, which we document for both groups, patients with and without comorbid MS (Fig. 4).
Fig. 4.
In-hospital mortality rates of COVID-19 patients stratified by age and the presence of comorbid MS. P-Value > 0.05 for all age-group comparisons among patients with and without comorbid MS
Besides age, the male sex was also associated with higher mortality among individuals with SARS-CoV-2 infection [17]. This also applies to patients with comorbid MS, as we also found a higher likelihood to die in male compared to female COVID-19 patients with comorbid MS (Online supplement). Although there was a higher frequency of women in our cohort of COVID-19 patients with comorbid MS, the sex-stratified analysis confirmed lower odds for all outcome measures, indicating age rather than sex as the major confounding factor for the observed differences between COVID-19 patients with and without comorbid MS.
The OR for admission to ICU and use of invasive or non-invasive ventilation were lower in patients with comorbid MS across all subgroup stratifications. Although the effect was attenuated in the age-group-stratified analysis, it remained statistically significant. The reason for this is speculative as there is a lack of information regarding other clinical and comorbid conditions, which is the major limitation of this study. Considering the high heterogenicity in the in-hospital mortality analysis across MS subtypes, such information is particularly important. We had no information about the use of immunosuppressive medication and the degree of disability of the study cohort. Unfortunately, this data is not consistently coded in the DRG database, making it impossible to account for it. Furthermore, admission to ICU might differ between hospitals that could also have biased the findings.
Besides age, Black race, cardiovascular comorbidities, and recent treatment with corticosteroids, an increased disability has recently been linked with clinical severity of COVID-19 in patients with comorbid MS [Salter 2021]. The German nursing institutions were almost sealed off and had to comply with strict quarantine rules during 2020. Additionally, highly disabled MS patients may have been more prudent and therefore better protected against a SARS-CoV-2 infection in- and outside of these institutions, resulting in the overall lower OR of the predefined outcomes. This hypothesis is also supported by the substantial decline in hospitalizations of MS patients with a progressive disease course in Germany, that was most remarkable during the first wave the pandemic in March 2020 [21].
Another punt for speculation is that PPMS patients had a lower OR for in-hospital mortality compared to SPMS and RRMS patients, although the age of these subgroups was similar or younger. As the information is not registered, this dataset cannot answer whether this is due to a lower use of immunosuppressive drugs in patients with PPMS. Despite the lack of confounding factors, this large-scale nationwide analysis used comprehensive administrative data from Germany based on the documented diagnoses and procedures in the G-DRG system. The administrative data have high quality and accuracy because registration of all inpatient cases and procedures is a prerequisite to getting financial compensation. Although generally not all coding of, e.g., secondary diagnoses, lead to higher reimbursement, all codes within the G35.- category are relevant for determining the case severity. In this respect, systematic under-determination cannot be ruled out, but it is unlikely.
Nevertheless, reimbursement by coding within the G35.- category is irrespective of the MS subtype coding. This might be reflected by the fact that nearly half of the MS patients were coded with an unspecified disease course. Therefore, another explanation for the low mortality in the PPMS group might be a wrong attribution. However, the coding is closely controlled by medical services of the medical health insurances to ensure the proper financial compensation for all German hospitals. The almost complete coverage of all hospitalized patients in Germany with a shallow risk of missing patients or double coding procedures is an essential strength of this study.
Conclusion
This study shows that simply having MS does not increase the likelihood of a severe course of COVID-19 compared to patients without comorbid MS. Nevertheless, there might be differences in risk within the MS patients’ population. Therefore, the knowledge of MS-associated risk factors is vital to help clinicians identify patients at higher risk for a severe course of COVID-19. Future studies should also question the incidence and impact of Long-COVID syndromes in patients with comorbid MS.
Supplementary Information
Additional file 1: Figure S1. Patient and outcome identification process.
Additional file 2: Figure S2. In-hospital mortality of COVID-19 patients with comorbid MS stratified by age and sex. Log odds-ratio > 0 indicates a higher risk for males.
Acknowledgments
None.
Abbreviations
- MS
Multiple sclerosis
- COVID-19
Coronavirus disease 2019
- SARS-CoV-2
Severe acute respiratory coronavirus 2
- G-DRG
German Diagnosis-Related Groups
- ICU
Intensive care unit
Authors’ contributions
DR and CK participated in the study design. DR drafted the first version of the manuscript. DR performed the statistical analysis. DB conducted the data abstraction. DR, SF, DB, LT, KH, IA, CK, and RG analyzed and discussed data. SF, LT, KH, IA, CK, and RG revised the manuscript. The author(s) read and approved the final manuscript.
Funding
None.
Availability of data and materials
Data according to §21 KHEntgG and §24 para. 2 KHG; official data on file, source: Destatis, www.destatis.de.
Declarations
Ethics approval and consent to participate
No informed consent or ethical approval was required for this study because completely anonymized data were used and provided by the German Federal Statistical Office to comply with the German data protection regulations. The latest Good Practice in Secondary Data Analysis recommendations (version 2; 2008) of the German Working Group for the Survey and Utilization of Secondary Data (with representatives from the German Society for Social Medicine and Prevention and the German Society for Epidemiology) and the Working Group for Epidemiological Methods (with representatives from the German Society for Epidemiology, the German Society for Medical Informatics, Biometry and Epidemiology and the German Society for Social Medicine and Prevention) state in their respective Guideline 1 that “ … recommendation to consult with an ethics committee need not apply to secondary data analyses if all the data protection provisions on pseudo-anonymization of all personal data are fulfilled … and no link to primary data is intended.”
Consent for publication
Not applicable.
Competing interests
Daniel Richter receives support from the Medical Faculty of Ruhr-University Bochum (FoRUM grant K136–20). Simon Faissner received speaker’s and/or board honoraria from Biogen, BMS, Celgene, Novartis and Roche and grant support from Ruhr-University Bochum, DMSG, Stiftung für therapeutische Forschung and Novartis, none related to this work. Dirk Bartig received orders for analysis of the German Diagnosis-Related Groups system from Boehringer Ingelheim and Sanofi Aventis. Lars Tönges LT has received travel funding and/or speaker honoraria from Abbvie, Bayer, Bial, Desitin, GE, UCB, Zambon and consulted for Abbvie, Bayer, Bial, Desitin, Stadapharm, UCB, Zambon in the last 3 years.
Kerstin Hellwig: received speaker’s and/or research support from Biogen, Bayer, Teva, Sanofi Genzyme, Novartis, and Roche and grant support from the Innovation Fond, DMSG and DFG, all not related to this work. Ilya Ayzenberg has received travel grants and speaker honoraria from Biogen Idec and Guthy-Jackson Charitable Foundation, Alexion, Santhera, Merck, served on scientific advisory boards for Roche and Alexion, and received research support from Diamed, none related to this study. Christos Krogias received speaker honoraria or travel grants for scientific meetings from Bayer Vital and Daichii Sankyo. Ralf Gold serves on scientific advisory boards for Teva Pharmaceutical Industries Ltd., Biogen Idec, Bayer Schering Pharma, and Novartis; has received speaker honoraria from Biogen Idec, Teva Pharmaceutical Industries Ltd., Bayer Schering Pharma, and Novartis; serves as editor for Therapeutic Advances in Neurological Diseases and on the editorial boards of Experimental Neurology and the Journal of Neuroimmunology; and receives research support from Teva Pharmaceutical Industries Ltd., Biogen Idec, Bayer.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.World Health Organization . Novel coronavirus – China 2020. 2020. [Google Scholar]
- 2.Peeters LM, Parciak T, Walton C, Geys L, Moreau Y, De Brouwer E, Raimondi D, Pirmani A, Kalincik T, Edan G, Simpson-Yap S, De Raedt L, Dauxais Y, Gautrais C, Rodrigues PR, McKenna L, Lazovski N, Hillert J, Forsberg L, Spelman T, et al. COVID-19 in people with multiple sclerosis: A global data sharing initiative. Multiple Sclerosis (Houndmills, Basingstoke, England) 2020;26(10):1157–1162. doi: 10.1177/1352458520941485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sormani MP, Italian Study Group on COVID-19 infection in multiple sclerosis An Italian programme for COVID-19 infection in multiple sclerosis. The Lancet Neurology. 2020;19(6):481–482. doi: 10.1016/S1474-4422(20)30147-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Faissner S, Plemel JR, Gold R, Yong VW. Progressive multiple sclerosis: from pathophysiology to therapeutic strategies. Nature Reviews Drug Discovery. 2019;18(12):905–922. doi: 10.1038/s41573-019-0035-2. [DOI] [PubMed] [Google Scholar]
- 5.Hauser SL, Oksenberg JR. The neurobiology of multiple sclerosis: genes, inflammation, and neurodegeneration. Neuron. 2006;52(1):61–76. doi: 10.1016/j.neuron.2006.09.011. [DOI] [PubMed] [Google Scholar]
- 6.Kleinewietfeld M, Hafler DA. Regulatory T cells in autoimmune neuroinflammation. Immunological Reviews. 2014;259(1):231–244. doi: 10.1111/imr.12169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Deutsche Gesellschaft für Neurologie . Welchen Verlauf nimmt COVID-19 bei Patienten mit Multipler Sklerose? 2020. [Google Scholar]
- 8.CDC COVID-19 Response Team Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019 - United States, February 12-march 28, 2020. MMWR Morbidity and Mortality Weekly Report. 2020;69(13):382–386. doi: 10.15585/mmwr.mm6913e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Guan WJ, Liang WH, Zhao Y, Liang HR, Chen ZS, Li YM, Liu XQ, Chen RC, Tang CL, Wang T, Ou CQ, Li L, Chen PY, Sang L, Wang W, Li JF, Li CC, Ou LM, Cheng B, Xiong S, Ni ZY, Xiang J, Hu Y, Liu L, Shan H, Lei CL, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Cheng LL, Ye F, Li SY, Zheng JP, Zhang NF, Zhong NS, He JX, China Medical Treatment Expert Group for COVID-19 Comorbidity and its impact on 1590 patients with COVID-19 in China: a nationwide analysis. The European Respiratory Journal. 2020;55(5):2000547. doi: 10.1183/13993003.00547-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui D, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, China Medical Treatment Expert Group for Covid-19 Clinical characteristics of coronavirus disease 2019 in China. The New England Journal of Medicine. 2020;382(18):1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Instituto Superiore di Sanità . Characteristics of COVID-19 patients dying in Italy. 2020. [Google Scholar]
- 12.Richter D, Bartig D, Krogias C, Tönges L. Letter to the editor: Risk comorbidities of COVID-19 in Parkinson's disease patients in Germany. Neurological Research and Practice. 2020;2(1):22. doi: 10.1186/s42466-020-00069-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Richter D, Krogias C, Eyding J, Bartig D, Grau A, Weber R. Comparison of stroke care parameters in acute ischemic stroke patients with and without concurrent Covid-19. A Nationwide analysis. Neurological Research and Practice. 2020;2:48. doi: 10.1186/s42466-020-00095-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Parrotta E, Kister I, Charvet L, Sammarco C, Saha V, Charlson RE, Howard J, Gutman JM, Gottesman M, Abou-Fayssal N, Wolintz R, Keilson M, Fernandez-Carbonell C, Krupp LB, Zhovtis Ryerson L. COVID-19 outcomes in MS: observational study of early experience from NYU multiple sclerosis comprehensive care center. Neurology(R) Neuroimmunology & Neuroinflammation. 2020;7(5):e835. doi: 10.1212/NXI.0000000000000835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Louapre C, Collongues N, Stankoff B, Giannesini C, Papeix C, Bensa C, Deschamps R, Créange A, Wahab A, Pelletier J, Heinzlef O, Labauge P, Guilloton L, Ahle G, Goudot M, Bigaut K, Laplaud DA, Vukusic S, Lubetzki C, De Sèze J, et al. Clinical characteristics and outcomes in patients with coronavirus disease 2019 and multiple sclerosis. JAMA Neurology. 2020;77(9):1079–1088. doi: 10.1001/jamaneurol.2020.2581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Salter, A., Fox, R. J., Newsome, S. D., Halper, J., Li, D., Kanellis, P., … Cross, A. H. (2021). Outcomes and risk factors associated with SARS-CoV-2 infection in a north American registry of patients with multiple sclerosis. JAMA Neurology, 78(6), e210688. [DOI] [PMC free article] [PubMed]
- 17.Mallapaty S. The coronavirus is most deadly if you are older and male - new data reveal the risks. Nature. 2020;585(7823):16–17. doi: 10.1038/d41586-020-02483-2. [DOI] [PubMed] [Google Scholar]
- 18.Pastor-Barriuso R, Pérez-Gómez B, Hernán MA, Pérez-Olmeda M, Yotti R, Oteo-Iglesias J, Sanmartín JL, León-Gómez I, Fernández-García A, Fernández-Navarro P, Cruz I, Martín M, Delgado-Sanz C, Fernández de Larrea N, León Paniagua J, Muñoz-Montalvo JF, Blanco F, Larrauri A, Pollán M, ENE-COVID Study Group Infection fatality risk for SARS-CoV-2 in community dwelling population of Spain: Nationwide seroepidemiological study. BMJ (Clinical research ed.) 2020;371:m4509. doi: 10.1136/bmj.m4509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.O'Driscoll M, Ribeiro Dos Santos G, Wang L, Cummings D, Azman AS, Paireau J, Fontanet A, Cauchemez S, Salje H. Age-specific mortality and immunity patterns of SARS-CoV-2. Nature. 2021;590(7844):140–145. doi: 10.1038/s41586-020-2918-0. [DOI] [PubMed] [Google Scholar]
- 20.Ward H, Atchison C, Whitaker M, Ainslie K, Elliott J, Okell L, Redd R, Ashby D, Donnelly CA, Barclay W, Darzi A, Cooke G, Riley S, Elliott P. SARS-CoV-2 antibody prevalence in England following the first peak of the pandemic. Nature Communications. 2021;12(1):905. doi: 10.1038/s41467-021-21237-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Richter D, Faissner S, Bartig D, Tönges L, Hellwig K, Ayzenberg I, Krogias C, Gold R. The impact of the COVID-19 pandemic on hospitalizations and plasmapheresis therapy in multiple sclerosis and neuromyelitis optica spectrum disorder: a nationwide analysis from Germany. Therapeutic Advances in Neurological Disorders. 2021;14:175628642110306. doi: 10.1177/17562864211030656. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Figure S1. Patient and outcome identification process.
Additional file 2: Figure S2. In-hospital mortality of COVID-19 patients with comorbid MS stratified by age and sex. Log odds-ratio > 0 indicates a higher risk for males.
Data Availability Statement
Data according to §21 KHEntgG and §24 para. 2 KHG; official data on file, source: Destatis, www.destatis.de.