Abstract
Background
Macrophage migration inhibitory factor (MIF) is a pleotropic inflammatory cytokine that is overexpressed in a number of cancer types including most types of human cancer. Inhibition of MIF signaling can restore anticancer immune responses in tumor microenvironments. In this study, we aimed to develop a therapeutic vaccine capable of inhibiting tumor development by inducing anti-MIF immune responses.
Methods
We introduced a recombinant Mycobacterium smegmatis (rSmeg-hMIF-hIL-7) vaccine that could deliver a fusion protein of human macrophage migration inhibitory factor (MIF) and interleukin 7, which could act as a target antigen and as an adjuvant of cancer vaccine, respectively. We checked the anticancer potential of the vaccine in a tumor-bearing mouse model.
Results
We found that rSmeg-hMIF-hIL-7 showed enhanced oncolytic activity compared with PBS, BCG or Smeg in MC38-bearing mice, and there was an increase in the humoral and cell-mediated immune responses against MIF. rSmeg-hMIF-hIL-7 can also induce a neutralizing effect regarding MIF tautomerase activity in the serum of vaccinated mice. We also found downregulation of MIF, CD74, and CD44, which are related to the MIF signaling pathway and PI3K/Akt and MMP2/9 signaling, which are regulated by MIF in the tumor tissue of rSmeg-hMIF-hIL-7-vaccinated mice, suggesting a significant role of the anti-MIF immune response to rSmeg-hMIF-hIL-7 in its anticancer effect. In addition, rSmeg-hMIF-hIL-7 treatment led to enhanced activation of CD4+ and CD8+ T cells in the tumor regions of vaccinated mice, also contributing to the anticancer effect. This trend was also found in LLC-bearing and PanO2-bearing mouse models. In addition, rSmeg-hMIF-hIL-7 treatment exerted an enhanced anticancer effect with one of the immune checkpoint inhibitors, the anti-PD-L1 antibody, in a tumor-bearing mouse model.
Conclusions
In conclusion, our data showed that rSmeg-hMIF-hIL-7 exerts a strong antitumor immune response in mice, possibly by inhibiting the MIF-dependent promotion of tumorigenesis by the anti-MIF immune response and via enhanced cytotoxic T cell recruitment into tumor microenvironments. We also found that it also exerted an enhanced anticancer effect with immune checkpoint inhibitors. These results suggest that rSmeg-hMIF-hIL-7 is a potential adjuvant for cancer immunotherapy. This is the first report to prove anticancer potential of immunotherapeutic vaccine targeting immune response against MIF.
Keywords: tumor microenvironment, immunotherapy, immunogenicity, vaccine, biomarkers, tumor
Background
Cancer immunotherapy is promising as a replacement of other treatments or as adjunctive therapy to enhance the treatment effect of current cancer therapies for a range of different cancer types.1 2 Many immunotherapeutic regimens are being approved for the treatment of several types of cancer and are being investigated as immunotherapeutic interventions alone or in combination with conventional treatments.3–5
Mycobacterium smegmatis (Smeg) is a fast-growing saprophytic environmental mycobacterium6 that can be transformed effectively with many heterologous genes, rendering it an ideal vaccine vector. Furthermore, unlike BCG, Smeg is rapidly destroyed by phagolysosomal proteases in the phagosomes of infected cells,7 8 which could facilitate better cross-presentation of antigen into T cells. Smeg can also induce the maturation of dendritic cells better than BCG and can trigger CD8+ T cell mediated immune responses, suggesting that it has an advantage in inducing the cytotoxic T lymphocyte (CTL) response, which is necessary for cancer immunotherapy.9 10 These findings highlight the potential role of Smeg as a recombinant vaccine delivery vector for cancer immunotherapy.
Together with the selection of a proper delivery vector system, immunogenic target antigens for cancer immunotherapy are another crucial option for developing a successful cancer vaccine.6 10 Macrophage migration inhibitory factor (MIF) has been characterized as a cytokine implicated in numerous inflammatory diseases, including rheumatoid arthritis, asthma and atherosclerosis.11–13 In addition, MIF can also function as a link between inflammation and tumorigenesis.14 Recently, MIF expression has been reported to be elevated in various solid tumors with a positive correlation with poor prognosis in patients with cancer.15 16
Recently, MIF has been reported to exert its protumorigenic effects through modulation of the immunosuppressive tumor microenvironment, mainly via MIF–CD74 signaling to macrophages and dendritic cells (DCs).17 Therefore, inhibiting MIF–CD74 signaling on macrophages and DCs via anti-MIF antibodies or MIF inhibitors could restore the antitumor immune response in tumor microenvironments.18 19 In addition, loss of MIF expression in tumor microenvironments can lead to robust induction of a specialized form of cell death, immunogenic cell death (ICD), also contributing to anticancer effects.20 These findings highlight the significant role of anti-MIF immune responses or drug inhibitors capable of interfering with MIF signaling in tumor microenvironments in inhibiting tumor development.
Adjuvant interleukin 7 (IL-7) treatment can elicit a significant increase in the number of antigen-specific effector and memory CD8+ T cells in several types of vaccine modules, including lentivirus-based or DNA-based vaccines.21 22 In particular, IL-7 can boost vaccine-induced antitumor immunity and enhance mouse survival in a tumor-bearing mouse model by enhancing the survival of activated T cells and cytolytic activity in vivo.23
Therefore, in this study, to elicit an anti-MIF immune response as an anticancer vaccine, we developed a recombinant Smeg vaccine delivering a fusion protein of human MIF and IL-7 (rSmeg-hMIF-hIL-7), which can act as a target antigen and an adjuvant of cancer vaccine, respectively, via a novel Mycobacterium–Escherichia coli shuttle vector system, pMyong2, to guarantee stable and enhanced expression of delivered heterologous genes in recombinant Smeg or BCG.24 25 We also sought to explore the immunotherapeutic potential of rSmeg-hMIF-hIL-7 in a tumor-bearing mouse model system. We found that rSmeg-hMIF-hIL-7 treatment led to a significant cancer inhibitory effect mainly by restoring the CTL response in tumor microenvironments by inducing anti-MIF immune responses. In addition, its challenge can exert an enhanced anticancer effect with one of the immune checkpoint inhibitors, anti-PD-L1 antibody, in a tumor-bearing mouse model.
Materials and methods
Mice experiments
Female C57BL/6 mice were purchased from Orient Bio and maintained in an ABL-2 laboratory. Seven-week-old C57BL/6 mice were inoculated with MC38, LLC, and PanO2 cells (3×106 cells/mouse) by subcutaneous injection on day 0. For bacterial injection, the mice were treated with peritumoral injections of mycobacterium (2×106 bacteria/mouse) on days 3, 7, and 14. For anti-PD-L1 immunotherapy, the mice were treated by intraperitoneal injection of anti-PD-L1 on days 7 and 14. The tumor size was measured once every 2 or 3 days and calculated following the formula: tumor volume (mm3) = (longest diameter × shortest diameter2)/2. The mice were observed until the tumor diameter was over 3 mm. Tumor-infiltrating lymphocytes (TILs) were isolated from mice by Ficoll assay26 on day 23 and subcutaneously injected into other tumor-bearing mice (3×106 cells/mouse).
Statistical analysis
Data are shown as the mean±SD and were analyzed by using GraphPad Prism V.9 statistical software (GraphPad, California, USA). Significant differences among multiple groups were analyzed by one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison test. A p value of <0.05 was considered to denote statistical significance. *p<0.05, **p<0.01, and ***p<0.001.
Results
Construction of recombinant Mycobacterium smegmatis expressing the hMIF-hIL-7 fusion protein
In a previous study, the protein expression efficacy of the pMyong2 vector system, a novel Mycobacterium–E. coli shuttle vector, was found to be significantly higher than the protein expression efficacy of other vector systems.27 Using the pMyong2-TOPO vector system, we generated three types of recombinant Smeg strains expressing human macrophage migration inhibitory factor (rSmeg-pMyong2-hMIF), human IL-7 (rSmeg-pMyong2-hIL-7), and fusion protein (rSmeg-pMyong2-hMIF-hIL-7) to enhance the anticancer effect of Smeg10 (figure 1A). Protein expression was detected by western blotting assay against human MIF (12 kDa) and human IL-7 (17.5 kDa) after lysis of cultured bacteria. The hMIF-hIL-7 fusion protein expressed in rSmeg-hMIF-hIL-7 was size shifted compared with the single human MIF and human IL-7 proteins. The protein expression levels were also confirmed by ELISA. Human MIF and hMIF-hIL-7 fusion proteins in each bacterial lysate were detected by ELISA against human MIF. In the same way, human IL-7 and hMIF-hIL-7 proteins were detected against IL-7 (figure 1B). Thereafter, to compare the cytotoxic effect induced by mycobacteria, MC38 cancer cells were cocultured with CD8+ T cells that had been cocultured with bone marrow-derived DCs infected with various types of rSmeg. There was no difference in T cell activation among the mycobacteria-infected DCs (online supplemental figure S1). However, rSmeg-hMIF-hIL-7-infected dendritic cells significantly induced CD8+ T cells capable of killing MC38 cancer cells compared with rSmeg-hMIF or rSmeg-hIL-7, leading to decreased MIF secretion from the cancer cells (figure 1C and D). Interestingly, rSmeg-hMIF-hIL-7-induced CD8+ T cells significantly secreted inflammatory cytokines in the presence of MC38 cells (figure 1E). Collectively, these results suggested that CD8+ T cells cocultured with DCs infected with rSmeg-hMIF-hIL-7 could exert the strongest CTL response against cancer cells, resulting in reduced MIF secretion from cancer cells.
Figure 1.
Construction of recombinant Mycobacterium smegmatis expressing the hMIF-hIL-7 fusion protein. (A) Maps of a novel Mycobacterium–Escherichia coli shuttle vector using pMyong2 expressing the hMIF-hIL-7 fusion protein. (B) Bacteria were cultured until the OD600 value reached 1, and the pellets were sonicated. The bacterial lysates were subjected to SDS-PAGE and analyzed by immunoblot assay. Target protein amounts in the bacterial lysates of rSmeg-hMIF-hIL-7 were assessed by ELISA. (C) For in vitro T cell-mediated cytotoxicity assay, MC38 cells were cocultured with CD8+ T cells that had been cocultured with bone marrow-derived dendritic cells infected with mycobacteria and analyzed by apoptosis assay. (D) Cell culture supernatants were detected by ELISA. (E) Cytokine-releasing T cell responses in the coculture experiment were analyzed by flow cytometry. Significance differences (*p<0.05, **p<0.01, and ***p<0.001) among the different groups are shown in the related figures, and the data are presented as the means±SEM of four independent experiments.
jitc-2021-003180supp001.pdf (81.2KB, pdf)
rSmeg-hMIF-hIL-7 suppressed tumor progression by downregulating MIF and PI3K/Akt signaling
Previous studies have shown that recombinant Mycobacterium smegmatis has cancer immune therapeutic potential.6 10 C57BL/6 mice were subcutaneously inoculated with MC38 colon cancer cells and injected with mycobacteria on days 3, 7, and 14 after cancer injection. In the MC38 tumor-bearing mouse model, rSmeg-hMIF-hIL-7 significantly inhibited tumor progression compared with PBS, BCG, and Smeg. Tumor growth was significantly suppressed by therapeutic administration of rSmeg-hMIF-hIL-7 compared with the other groups (figure 2A). Tumor growth inhibition of rSmeg-hMIF-hIL-7 was also observed in the PanO2-bearing and LLC-bearing mouse model (online supplemental figures S2 and S4). Serum MIF levels, an important modulator in tumor angiogenesis, were decreased in both Smeg and rSmeg-hMIF-hIL-7 compared with PBS, but rSmeg-hMIF-hIL-7-cells showed the most pronounced reduction in MIF levels compared with Smeg. Moreover, administration of rSmeg-hMIF-hIL-7 induced increased production of anti-human MIF IgG1, IgG2c, and total IgG in serum compared with all other groups (figure 2B). The surface expression of MIF coreceptor CD74 and CD44, which promote downstream signaling pathways for cancer cell proliferation and migration,28 were decreased in rSmeg-hMIF-hIL-7 compared with all other groups (figure 2C). MIF downstream signaling proteins in primary tumor cells were detected by western blotting. rSmeg-hMIF-hIL-7 administration led to decreased MIF and downstream ERK and PI3K/Akt signaling pathways in primary tumor cells. Also, monocyte chemoattractant protein 1 (MCP-1), one inflammatory cytokine implicated in MDSC tumor infiltration and cancer development, was decreased in response to rSmeg-hMIF-hIL-7. Notably, the production of matrix metalloproteinases MMP-2 and MMP-9, which are crucial for cancer cell invasion and metastasis, was significantly suppressed in response to rSmeg-hMIF-hIL-7 treatment (figure 2D). The transcriptional levels of the corresponding genes were significantly decreased in rSmeg-hMIF-hIL-7 (figure 2E). These results indicate that rSmeg-hMIF-hIL-7 reduced the tumorigenic effect of MIF, enhancing its therapeutic efficacy against tumors.
Figure 2.
rSmeg-hMIF-hIL-7 suppressed tumor progression by downregulating MIF and PI3K/Akt signaling. C57BL/6 mice were subcutaneously injected with MC38 cells, and mycobacteria were administered on day 3, 7, and 14 after tumor implantation. (A) Growth curve shows tumor volume as the mean±SD at the indicated time points. Representative pictures were taken from tumors of each group, and tumor weight was measured at day 25 post injection (p.i.). (B) Human MIF-specific immunoglobulin subtypes (IgG1, IgG2c, and total IgG) and mouse MIF in serum were assessed by ELISA. (C) Immunohistochemistry staining for the MIF coreceptor CD74 and CD44 (scale bar=50 μm). (D) Protein expression of ERK and PI3K/Akt signaling and MCP-1, matrix metalloproteinases MMP-2 and MMP-9 in primary tumor cells. (E) The transcription level of mRNA in primary tumor cells was quantified by RT-qPCR. Significance differences (*p<0.05, **p<0.01, and ***p<0.001) among the different groups are shown in the related figures, and the data are presented as the mean±SEM of mice (n=4). IL-7, interleukin 7; MIF, macrophage migration inhibitory factor.
jitc-2021-003180supp002.pdf (247.5KB, pdf)
jitc-2021-003180supp003.pdf (383.2KB, pdf)
rSmeg-hMIF-hIL-7 induced antitumor immune responses through the recruitment of functional T cells in the tumor environment
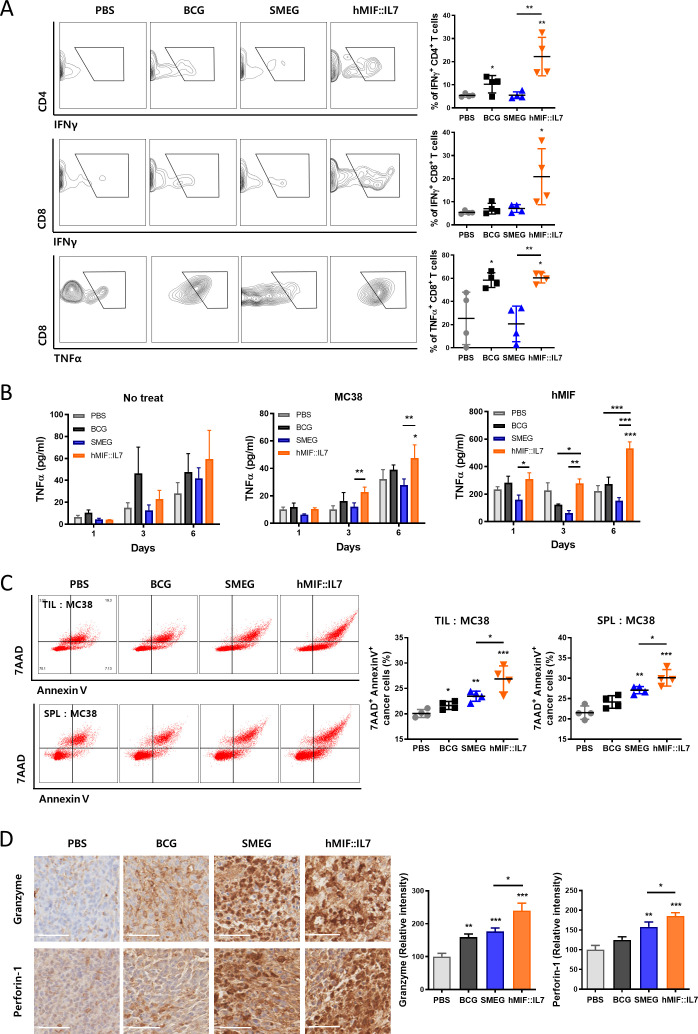
As tumor progression was successfully inhibited through administration of rSmeg-hMIF-hIL-7, a cell-mediated immune response in addition to elevated humoral anti-MIF IgG may work together to inhibit tumor growth. Eleven days after the last mycobacteria injection, rSmeg-hMIF-hIL-7 induced the recruitment of tumor-infiltrating functional T cells. Infiltration of IFNγ-releasing CD8+ cytotoxic T cells and CD4+ helper T cells in the tumor environment was noticeably induced after rSmeg-hMIF-hIL-7 administration compared with Smeg administration. Additionally, the population of TNFα-releasing CD8+ T cells was significantly increased in response to rSmeg-hMIF-hIL-7 compared with Smeg (figure 3A). These enhanced cell-mediated immune responses were also observed in PanO2-bearing and LLC-bearing mouse models (online supplemental figures S3 and S4). In addition, rSmeg-hMIF-hIL-7 significantly enhanced the inflammatory response against MIF in mouse splenocytes compared with Smeg (figure 3B). To assess the cytotoxicity of tumor-infiltrating lymphocytes and splenocytes, lymphocytes were isolated from tumor tissue and spleen and cocultured with MC38 for 24 hours. The 7AAD+ AnnexinV+ cancer cells were significantly increased by lymphocytes in rSmeg-hMIF-hIL-7 group, indicating that rSmeg-hMIF-hIL-7 administration induced increased lymphocytes capable of directly killing cancer cells in the tumor-bearing mouse (figure 3C). Since interferon-gamma (IFNγ) and tumor necrosis factor-alpha (TNFα) in the tumor environment play a vital role in antitumor activity via the stimulation of tumor-specific cytotoxic T cells,29 cytolytic responses by CTLs were also observed by immunohistochemistry (IHC) staining. Secretion of the pore-forming molecule Perforin-1 and proapoptotic protease Granzyme B, which can lead to granule exocytosis of cancer cells, was induced by the administration of rSmeg-hMIF-hIL-7 (figure 3D).
Figure 3.
rSmeg-hMIF-hIL-7 induced antitumor immune responses through the recruitment of functional T cells in the tumor environment. (A) The population of IFNγ-releasing CD8+ T cells and CD4+ T cells and TNFα-releasing CD4+ T cells in tumors were analyzed by flow cytometry. (B) Splenocytes were incubated with MC38 lysates or h-MIF for 6 days. Inflammatory cytokine secretion of splenocytes in response to the antigen stimulation was detected by ELISA. (C) Four days after the last mycobacteria injection, lymphocytes from the tumor tissue and spleen were isolated and cocultured with carboxyfluorescein succinimidyl ester (CFSE)-labeled MC38 (E:T=5:1) cancer cells, and cytotoxicity of the lymphocytes were detected by apoptosis assay. (D) IHC image for cytolytic responses by secretion of the pore-forming molecule perforin-1 and proapoptotic protease granzyme B in tumor tissue (scale bar=50 µm). Significance differences (*p<0.05, **p<0.01, and ***p<0.001) among the different groups are shown in the related figures, and the data are presented as the mean±SEM of mice (n=4).
jitc-2021-003180supp004.pdf (382.8KB, pdf)
rSmeg-hMIF-hIL-7 inhibited the infiltration of myeloid-derived suppressor cells (MDSCs) into the tumor environment
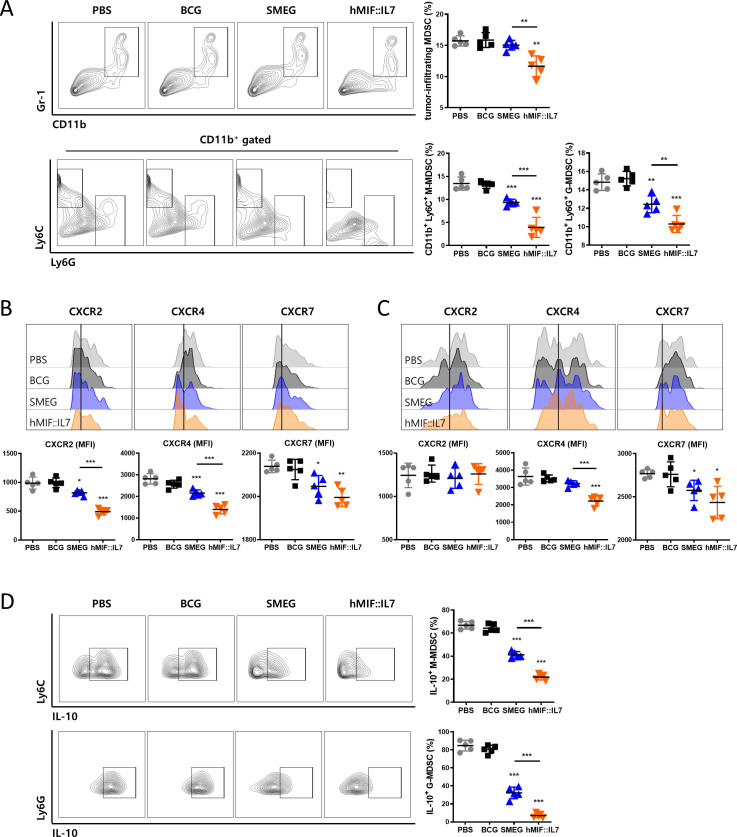
As rSmeg-hMIF-hIL-7 administration decreased MIF level in serum and tumor, immune cells population in tumor microenvironment were changed. myeloid-derived suppressor cells (MDSCs), an obstacle to cancer immunotherapies, accumulate in cancer and promotes tumor progression.30 Since MDSC accumulation in tumor is dependent on MIF,31 MDSC subsets in tumor after rSmeg-hMIF-hIL-7 administration were assessed by flow cytometry. The population of total MDSC, monocytic MDSC (M-MDSC), and granulocytic MDSC (G-MDSC) were significantly decreased in rSmeg-hMIF-hIL-7 group compared with all other group (figure 4A). In addition, among the surface MIF receptors, surface expression of CXCR2 and CXCR4 in M-MDSC, and CXCR4 and CXCR7 in G-MDSC were decreased in response to rSmeg-hMIF-hIL-7, indicating that rSmeg-hMIF-hIL-7 suppressed the infiltration of MDSC into tumor (figure 4B and C). Also, among tumor-infiltrating MDSCs, IL-10-releasing M-MDSC and G-MDSC were decreased in rSmeg-hMIF-hIL-7 compared with all other groups (figure 4D).
Figure 4.
rSmeg-hMIF-hIL-7 inhibited the infiltration of myeloid-derived suppressor cells (MDSCs) into the tumor environment. (A) The population of total MDSC, monocytic MDSC and granulocytic MDSC in mice tumor tissue 4 days after the last mycobacteria injection. Surface expression levels of MIF receptors CXCR2, CXCR4, and CXCR7 of (B) M-MDSC and (C) G-MDSC were detected by flow cytometry. The population of IL-10-releasing (D) M-MDSC and G-MDSC in tumors were analyzed by flow cytometry. Significance differences (*p<0.05, **p<0.01, and ***p<0.001) among the different groups are shown in the related figures, and the data are presented as the mean±SEM of mice (n=5).
rSmeg-hMIF-hIL-7 inhibited cancer cell proliferation by downregulating the biological activity of serum MIF
Since the anti-MIF IgG level in mouse serum was increased in response to rSmeg-hMIF-hIL-7, the serum of vaccinated mice may neutralize MIF and regulate the malignant character of cancer cells. Therefore, next to address this, we checked the biological activity of MIF in vaccinated mice via a tautomerase activity assay. Serum MIF in the rSmeg-hMIF-hIL-7 group showed significantly decreased enzymatic activity compared with the PBS and Smeg groups (figure 5A). Thereafter, MC38 cancer cells showed the decreased expression level of CD74 on the cell surface and downregulated MIF downstream signaling pathway during incubation with serum in rSmeg-hMIF-hIL-7 compared with Smeg, indicating that MIF, which plays a crucial role in cancer cell proliferation, was neutralized by anti-MIF IgG in the serum (figure 5B and C). Serum in the rSmeg-hMIF-hIL-7 and Smeg groups significantly increased the population of apoptotic cancer cells compared with the population of apoptotic cancer cells in the PBS group (figure 5D). Furthermore, the malignant characteristics of cancer cells were suppressed by neutralizing MIF activity. The migration and invasion activities of cancer cells were inhibited after incubation with serum in rSmeg-hMIF-hIL-7 compared with the PBS group (figure 5E and F). Altogether, rSmeg-hMIF-hIL-7 treatment increased neutralization of MIF in serum and suppressed the migration and invasion of cancer cells by neutralizing the biological activity of MIF.
Figure 5.
rSmeg-2-hMIF-hIL-7 inhibited cancer cell proliferation by downregulating the biological activity of serum MIF. (A) Tautomerase activity of serum MIF from MC38-bearing mice. (B) Surface expression of CD74 in MC38 cancer cells during incubation with mouse serum. (C) Protein expression of PI3K/Akt signaling and matrix metalloproteinases MMP-2 and MMP-9 in serum-treated MC38 cells. (D) Induction of apoptosis of MC38 cancer cells after 24 h and 48 h incubation in the media containing 50% mouse serum. Inhibition of (E) migration and (F) invasion ability of MC38 cancer cells after incubation with serum from in vivo studies. Significance differences (*p<0.05, **p<0.01, and ***p<0.001) among the different groups are shown in the related figures, and the data are presented as the mean±SEM of mice (n=4). MIF, macrophage migration inhibitory factor.
rSmeg-hMIF-hIL-7 induced tumor-infiltrating lymphocytes capable of downregulating the MIF downstream signaling pathway
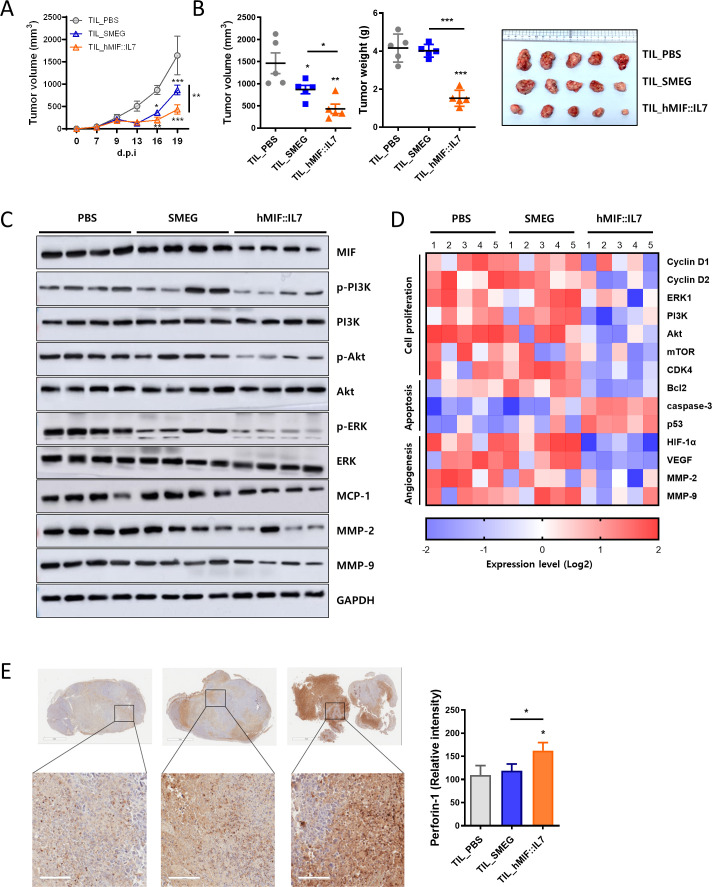
To further confirm the role of tumor-infiltrating lymphocytes in rSmeg-hMIF-hIL-7-mediated function, the anticancer effect of TILs isolated from rSmeg-hMIF-hIL-7-injected mice was observed by adoptively transferring TILs into tumor-bearing recipient mice. TILs from rSmeg-hMIF-hIL-7 treatment significantly suppressed tumor volume compared with those from Smeg treatment (figure 6A and B). Tumor weight at day post injection (d.p.i.) 23 was reduced only in the rSmeg-hMIF-hIL-7 group, not in the PBS and Smeg groups, suggesting that rSmeg-hMIF-hIL-7 induced TILs capable of inhibiting tumor growth in a way that is distinct from Smeg (figure 6B). Notably, protein expression in the ERK and PI3K/Akt downstream pathway of MIF was significantly inhibited by treatment with TILs from the rSmeg-hMIF-hIL-7 group compared with the Smeg group (figure 6C). Moreover, the transcription levels of the corresponding genes were downregulated by treatment with TILs from rSmeg-hMIF-hIL-7 (figure 6D). The cytolytic protein perforin-1 was significantly increased by TILs from rSmeg-hMIF-hIL-7 compared with the TILs from Smeg, suggesting that TILs in response to rSmeg-hMIF-hIL-7 more effectively induced tumor cell death (figure 6E). Interestingly, tumor infiltration of MDSC and surface MIF receptors in MDSCs were decreased in response to treatment with TILs from rSmeg-hMIF-hIL-7 (online supplemental figure S5). Collectively, rSmeg-hMIF-hIL-7 enhanced cell-mediated immune responses against MIF and regulated the MIF-mediated PI3K/Akt pathway in the tumor environment via functional TILs.
Figure 6.
rSmeg-hMIF-hIL-7 induced tumor-infiltrating lymphocytes capable of downregulating the MIF downstream signaling pathway. Tumor-infiltrating lymphocytes from tumor-bearing mice treated with PBS, Smeg, or rSmeg-hMIF-hIL-7 were adoptively transferred into other tumor-established mice. (A) Growth curve represents the tumor volume of tumor-bearing mice as the mean±SD at the indicated time points. (B) Tumor volume and tumor weight were measured at d.p.i. 23. Representative images were taken from tumors of each group at d.p.i. 23. (C) MIF-mediated ERK and PI3K/Akt signaling proteins were detected by western blotting. (D) The transcription level of mRNA in primary tumor cells was quantified by RT-qPCR. (E) Cytolytic protein perforin-1 in tumors was observed by IHC staining (scale bar=100 μm). Significance differences (*p< 0.05, **p<0.01, and ***p<0.001) among the different groups are shown in the related figures, and the data are presented as the mean±SEM of mice (n=5). MIF, macrophage migration inhibitory factor.
of mice (n=5). MIF, macrophage migration inhibitory factor.
jitc-2021-003180supp005.pdf (389.3KB, pdf)
rSmeg-hMIF-hIL-7 exerted an enhanced anticancer effect with anti-PD-L1 immunotherapy
Next, we checked the enhanced effect of the combination treatment of rSmeg-hMIF-hIL-7 and anti-PD-L1. Our data showed that combination treatment with the immunotherapeutic agents rSmeg-hMIF-hIL-7 and anti-PD-L1 exerted an enhanced anticancer effect. rSmeg-hMIF-hIL-7 alone suppressed tumor growth and body weight change compared with PBS treatment, but the combinational treatment of rSmeg-hMIF-hIL-7 and anti-PD-L1 showed significant reduction in tumor volume and body weight change compared with rSmeg-hMIF-hIL-7 treatment alone or anti-PD-L1 alone (figure 7A and B). Excised tumor weight and size at d.p.i. 23 were drastically decreased by the combination therapy compared with rSmeg-hMIF-hIL-7 alone or anti-PD-L1 alone (figure 7C and F and online supplemental figure S6A). Interestingly, the combinational therapy resulted in the highest spleen/body weight ratio and increased population of cytokine-releasing T cells in the spleen, suggesting that the combination therapy could promote the recruitment of functional immune cells in the spleen (online supplemental figure S6A, E). rSmeg-hMIF-hIL-7 and anti-PD-L1 worked together to induce the recruitment of cytokine-releasing T cells in tumor tissue and suppress tumor-infiltration of MDSCs (figure 7G, online supplemental figures S6D, S7, S8). Notably, the combinational treatment of rSmeg-hMIF-hIL-7 and anti-PD-L1 resulted in decreased serum MIF and biological activity of serum MIF compared with rSmeg-hMIF-hIL-7, indicating that anti-PD-L1 treatment maximizes the ability of rSmeg-hMIF-hIL-7 to neutralize MIF (figure 7D and E and online supplemental figure S6B). These results indicated that rSmeg-hMIF-hIL-7 exerted an enhanced anticancer effect with cancer immune checkpoint drugs, suggesting the potential use of rSmeg-hMIF-hIL-7 as an adjunctive immunotherapy capable of enhancing the effect of immunotherapy.
Figure 7.
rSmeg-hMIF-hIL-7 exerted an enhanced anticancer effect with anti-PD-L1 immunotherapy. C57BL/6 mice were subcutaneously injected with MC38 cells, anti-PD-L1 was administered by the intraperitoneal route on d.p.i. 7 and 14, and mycobacteria were injected on d.p.i. 3, 7, and 14. Growth curve shows (A) tumor volume and (B) body weight change of tumor-bearing mice as the mean±SD at the indicated time points. (C) The tumor weight was measured at d.p.i. 23. (F) Representative pictures were taken from tumors and spleens of each group at d.p.i. 23. (D and E) Serum MIF and tautomerase activity of serum MIF were detected by ELISA. (G) The population of functional tumor-infiltrating lymphocytes. Significance differences (*p<0.05, **p<0.01, and ***p<0.001) among the different groups are shown in the related figures, and the data are presented as the mean±SEM of mice (n=5). MIF, macrophage migration inhibitory factor.
jitc-2021-003180supp006.pdf (546.7KB, pdf)
jitc-2021-003180supp007.pdf (384KB, pdf)
jitc-2021-003180supp008.pdf (198.4KB, pdf)
Discussion
In the present study, we developed a recombinant Smeg strain, rSmeg-hMIF-hIL-7 as a new anticancer therapeutic vaccine, expressing the chimeric fusion protein of h-MIF as the target Ag of the cancer vaccine and h-IL-7 as an adjuvant of the cancer vaccine to induce anti-MIF immune responses (graphical abstract). We also proved that rSmeg-hMIF-hIL-7 can exert strong anticancer immune responses in various tumor-bearing mouse models (MC38, PanO2 and LLC), which was mediated mainly by eliciting anti-MIF immune responses.
MIF, a member of the tautomerase family of cytokines, is highly expressed in numerous cancers, including colon cancer, lung cancer, breast cancer, pancreatic cancer, melanoma, nasopharyngeal carcinoma, cervical adenocarcinoma, and prostate cancer.28 MIF is now recognized as a pleiotropic cytokine that plays pivotal roles in cancer promotion and the inflammatory cascade, and MIF is constitutively expressed in almost all cancer cell types.32 MIF can exert various biological activities, including cancer-promoting effects, mainly via its interaction with the receptor complex of the cognitive receptor CD74 and coreceptor CD44. Signaling by binding of MIF with the receptor complex can lead to a cancer-promoting effect mainly by inducing the PI3K/Akt axis,33 p53 inhibition by sustained ERK activation34 or enhanced angiogenesis following VEGF and MMP activation by upregulation of HIF-1. In addition to cancer cells, it has been proven that MIF could affect innate myeloid, neutrophil and adaptive Th1, Th2, and NK T cells, influencing tumor-associated immune responses.35–37 MIF promotes the tumor infiltration and immune suppressive activity of MDSCs in mouse models of breast, lung, and glioblastoma cancer.31 35 Also, MIF induces Th2 and Th17 expansion and suppresses CTL activation, which are primary target of immune checkpoint inhibitor immunotherapies.37 Moreover, since the expression of MIF has been reported to be positively correlated with the severity of the cancer phenotype,38 MIF is an attractive target for the development of cancer therapeutic inhibitors.
Currently, many efforts have focused on the development of small molecule inhibitors that target the unique tautomerase active site of MIF and anti-MIF antibodies capable of neutralizing MIF. For example, ISO-1 treatment capable of inhibiting MIF tautomerase activity has been reported to markedly inhibit the tumorigenic growth of various cancers, including prostate cancer, colon cancer and pancreatic cancers, by effectively blocking the interaction between MIF and receptors, particularly CD74,32 leading us to hypothesize that an immunotherapeutic vaccine approach eliciting an anti-MIF immune response can also play a pivotal role in cancer treatment. To date, an immunotherapeutic vaccine targeting MIF for cancer treatment has not been introduced. Theoretically, immunotherapeutic vaccines have merit over chemical inhibitors or anti-MIF antibodies since the former can also lead to a cell-mediated immune response against MIF-producing cancer cells in addition to a humoral immune response against MIF capable of neutralizing MIF activity.
Since MIF has various biological functions, including inflammation induction or cancer promotion,32 direct MIF protein challenge as a vaccine component could have a risk of generating unwanted biological phenotypes. Therefore, to use MIF as a vaccine, its accurate loading to antigen-presenting cells (APCs) such as macrophages or DCs is an essential step. For this reason, we chose M. smegmatis for vaccine delivery of MIF in this study, which generally infects APCs, and loaded antigens can easily be processed and presented into CD4+ or CD8+ T cells, resulting in strong immune responses against delivered Ags. Importantly, since it leads to an enhanced CTL response against cancer and grows more rapidly than BCG,10 it seems more appropriate for the delivery of cancer vaccines in terms of treatment efficacy or production cost.
IL-7 has various vaccine potentiating effects, including differentiating naïve T cells into memory T cells or activating γδ T cells. Therefore, we generated rSmeg strains expressing fusion proteins of both hMIF and hIL-7 to maximize anti-MIF immune responses in vaccinated mice. Indeed, we found that rSmeg-hMIF-hIL-7 elicited an enhanced CTL response against CD4+ and CD8+ T cells cocultured with infected dendritic cells compared with rSmeg expressing h-MIF alone or h-IL-7 alone (figure 1), suggesting an adjuvant effect of IL-7 in cancer immunotherapy.
Our data indicated that the anticancer effect of rSmeg-hMIF-hIL-7 in tumor-bearing mice could be mediated mainly by two major mechanisms: attenuation of MIF signaling by interaction with CD74, perhaps via anti-MIF antibody-mediated reduction of MIF levels or neutralization of MIF activity, and attenuation of the CD4+ or CD8+ T cell mediated immune response against MIF, particularly the CD8+ T cell mediated CTL response in tumor microenvironments. Our data indicated that rSmeg-hMIF-hIL-7 can lead to an enhanced humoral immune response against MIF (an increase in anti-MIF IgG capable of neutralizing tautomerase activity in serum) in vaccinated mice (figures 2 and 3). Given that MIF tautomerase activity is positively correlated with MIF cancer-promoting signaling,39 an increase in anti-MIF IgG induced by rSmeg-hMIF-hIL-7 vaccination is likely to elicit anticancer effects by blocking MIF signaling. It has been reported that MIF could also regulate expression of receptor complex, CD74 and CD44,40–42 resulting in affecting MIF signaling. Indeed, we found that MIF, CD74, and CD44, which are related to the MIF signaling pathway, and PI3K/Akt and MMP2/9 signaling-mediated regulation MIF were downregulated in the tumor tissue of rSmeg-hMIF-hIL-7-vaccinated mice (figure 2), suggesting a significant role of the anti-MIF humoral immune response by rSmeg-hMIF-hIL-7 in its anticancer effect. In addition to the anti-MIF humoral immune response, a reduction in the entire secreted MIF level in serum or tumor microenvironments could also contribute to the anticancer effect via robust induction of a specialized form of cell death, immunogenic cell death (ICD),20 and inhibition of MIF signaling. Our data showed that T cells cocultured with dendritic cells that had been infected with rSmeg-hMIF-hIL-7 could reduce secreted MIF levels via enhanced killing of MIF-producing cancer cells (figure 1), suggesting that the reduced MIF level found in mice vaccinated with rSmeg-hMIF-hIL-7 may be due to dual actions of anti-MIF antibodies and an enhanced CTL response.
T lymphocytes play a key role in electing antitumor immunity and are the major targets of immune checkpoint inhibitor (ICI) immunotherapies. Tumor-derived MIF can compromise T lymphocyte mediated anticancer effects, particularly the CTL response in tumor microenvironments.43 Therefore, efforts to reduce MIF levels in tumor microenvironments using anti-MIF antibodies or small inhibitors have been reported to be able to restore MHC I expression on tumors to allow for more effective CTL-mediated immunotherapy in tumor microenvironments.44 Indeed, our data indicated that the anticancer potential of CTLs from the tumor tissue of rSmeg-hMIF-hIL-7-vaccinated mice was enhanced compared with the anticancer potential of CTLs from control mice (figure 3), suggesting that the reduction in tumor-derived MIF levels by rSmeg-hMIF-hIL-7 vaccination can contribute to anticancer effects by recovering CTL function in the tumor microenvironment in vaccinated mice. Moreover, we found that adoptive transfer of isolated TILs from rSmeg-hMIF-hIL-7-injected mice led to a strong anticancer effect in a tumor-bearing mouse model via inhibition of MIF signaling (figure 5), indicating that cell-mediated immune responses against MIF could play a major role in the anticancer effect. Of note, our immunotherapeutic approach using rSmeg-hMIF-hIL-7 showed an enhanced anticancer effect with one of the ICI drugs, anti-PD-L1 treatment, in a tumor-bearing mouse model (figure 6), suggesting its further feasibility in cancer treatment.
Of the potential small molecule inhibitors of MIF signaling, ibudilast has gained a great attention due to its strong anti-inflammatory effect and suppressive effect of MDSCs by MCP-1 inhibition via blocking MIF signaling.45 Of note, it can lead to the most powerful reduction of MDSC generation compared with other MIF inhibitors 4-IPP and ISO-1.45 Ibudilast can also contribute to cancer treatment via increasing CD8+ T cell infiltration and suppressing MDSC functions in tumor microenvironments45 46 in a MIF-dependent manner. As shown in ibudilast treatment, our data also indicated that rSmeg-hMIF-hIL7 can decrease population of total MDSC, M-MDSC, and G-MDSC in tumor microenvironments of vaccinated mice (figure 4), possibly due to of its inhibition of MIF signaling, suggesting that it can contribute to recovery of T cell-mediated anticancer function in tumor microenvironments via MDSC suppression. In T cell-mediated anticancer effect, rSmeg-hMIF-hIL7 may have merit over ibudilast in that the former can lead to vaccine-mediated activation of T lymphocytes capable of killing MIF-producing cancer cells as well as recovery of T cell function mediated by MDSC inhibition.
The present study has several limitations. Our results include rSmeg-hMIF-hIL7 anticancer therapy only in female mice. However, there exists gender disparity in immune responses.47 So, rSmeg-hMIF-hIL7 anticancer effect should also be evaluated in male mice in the future. Furthermore, synergistic effect of rSmeg-hMIF-hIL7 anticancer therapy with other MIF inhibitors such as ISO-1 or Ibudilast remains to be evaluated in the future.
In conclusion, our results demonstrate that rSmeg-hMIF-hIL-7 therapy elicits a strong anticancer immune response in mice, mainly via reduction of tumor-derived MIF level, neutralization of MIF or the recruitment of activated lymphocytes capable of downregulating the MIF downstream signaling pathway, indicating the potential use of rSmeg-hMIF-hIL-7 as adjunctive immunotherapy capable of enhancing the efficacy of immunotherapy.
jitc-2021-003180supp009.pdf (2.7MB, pdf)
jitc-2021-003180supp010.pdf (37.7KB, pdf)
jitc-2021-003180supp011.pdf (311.5KB, pdf)
Footnotes
Contributors: Bum-Joon Kim and Hyein Jeong conceived of the presented idea and developed the theory. Hyein Jeong and So-Young Lee carried out animal experiments and assays. Hyejun Seo supported DNA work. Bum-Joon Kim supervised overall experiments and wrote the manuscript. Hyein Jeong wrote the manuscript and discussed the results. All authors contributed to the article and approved the submitted version.
Funding: This research was supported by CLIPSBnC Co. Ltd. (Grant No. 800-20190038) and by the Ministry of Education (Grant No. NRF-2019R1A2C1084511).
Competing interests: No, there are no competing interests.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
All data relevant to the study are included in the article or uploaded as supplementary information. The original contributions presented in the study are included in the article/supplementary material; further inquiries can be directed to the corresponding author.
Ethics statements
Patient consent for publication
Not required.
Ethics approval
The study was carried out according to the guidelines approved by the Institutional Animal Care and Use Committee of Seoul National University (Approval No. SNU-200706–5) and by the Institutional Biosafety Committee of SNU (Approval No. SNUIBC-R200707-1-1).
References
- 1.Kruger S, Ilmer M, Kobold S, et al. Advances in cancer immunotherapy 2019 - latest trends. J Exp Clin Cancer Res 2019;38:268. 10.1186/s13046-019-1266-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kudo M. Combination cancer immunotherapy in hepatocellular carcinoma. Liver Cancer 2018;7:20–7. 10.1159/000486487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Patyar S, Joshi R, Byrav DSP, et al. Bacteria in cancer therapy: a novel experimental strategy. J Biomed Sci 2010;17:21. 10.1186/1423-0127-17-21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shrimali RK, Ahmad S, Verma V, et al. Concurrent PD-1 blockade negates the effects of OX40 agonist antibody in combination immunotherapy through inducing T-cell apoptosis. Cancer Immunol Res 2017;5:755–66. 10.1158/2326-6066.CIR-17-0292 [DOI] [PubMed] [Google Scholar]
- 5.Beatty GL, Chiorean EG, Fishman MP, et al. CD40 agonists alter tumor stroma and show efficacy against pancreatic carcinoma in mice and humans. Science 2011;331:1612–6. 10.1126/science.1198443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Young SL, Murphy M, Zhu XW, et al. Cytokine-modified Mycobacterium smegmatis as a novel anticancer immunotherapy. Int J Cancer 2004;112:653–60. 10.1002/ijc.20442 [DOI] [PubMed] [Google Scholar]
- 7.Beltan E, Horgen L, Rastogi N. Secretion of cytokines by human macrophages upon infection by pathogenic and non-pathogenic mycobacteria. Microb Pathog 2000;28:313–8. 10.1006/mpat.1999.0345 [DOI] [PubMed] [Google Scholar]
- 8.Yadav M, Roach SK, Schorey JS. Increased mitogen-activated protein kinase activity and TNF-alpha production associated with Mycobacterium smegmatis- but not Mycobacterium avium-infected macrophages requires prolonged stimulation of the calmodulin/calmodulin kinase and cyclic AMP/protein kinase A pathways. J Immunol 2004;172:5588–97. 10.4049/jimmunol.172.9.5588 [DOI] [PubMed] [Google Scholar]
- 9.Cayabyab MJ, Hovav A-H, Hsu T, et al. Generation of CD8+ T-cell responses by a recombinant nonpathogenic Mycobacterium smegmatis vaccine vector expressing human immunodeficiency virus type 1 env. J Virol 2006;80:1645–52. 10.1128/JVI.80.4.1645-1652.2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rich FJ, Kuhn S, Hyde EJ, et al. Induction of T cell responses and recruitment of an inflammatory dendritic cell subset following tumor immunotherapy with Mycobacterium smegmatis. Cancer Immunol Immunother 2012;61:2333–42. 10.1007/s00262-012-1291-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Baugh JA, Chitnis S, Donnelly SC, et al. A functional promoter polymorphism in the macrophage migration inhibitory factor (MIF) gene associated with disease severity in rheumatoid arthritis. Genes Immun 2002;3:170–6. 10.1038/sj.gene.6363867 [DOI] [PubMed] [Google Scholar]
- 12.Mizue Y, Ghani S, Leng L, et al. Role for macrophage migration inhibitory factor in asthma. Proc Natl Acad Sci U S A 2005;102:14410–5. 10.1073/pnas.0507189102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Verschuren L, Kooistra T, Bernhagen J, et al. MIF deficiency reduces chronic inflammation in white adipose tissue and impairs the development of insulin resistance, glucose intolerance, and associated atherosclerotic disease. Circ Res 2009;105:99–107. 10.1161/CIRCRESAHA.109.199166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Meyer-Siegler KL, Vera PL, Iczkowski KA, et al. Macrophage migration inhibitory factor (MIF) gene polymorphisms are associated with increased prostate cancer incidence. Genes Immun 2007;8:646–52. 10.1038/sj.gene.6364427 [DOI] [PubMed] [Google Scholar]
- 15.Xu X, Wang B, Ye C, et al. Overexpression of macrophage migration inhibitory factor induces angiogenesis in human breast cancer. Cancer Lett 2008;261:147–57. 10.1016/j.canlet.2007.11.028 [DOI] [PubMed] [Google Scholar]
- 16.Wilson JM, Coletta PL, Cuthbert RJ, et al. Macrophage migration inhibitory factor promotes intestinal tumorigenesis. Gastroenterology 2005;129:1485–503. 10.1053/j.gastro.2005.07.061 [DOI] [PubMed] [Google Scholar]
- 17.Gordon-Weeks AN, Lim SY, Yuzhalin AE, et al. Macrophage migration inhibitory factor: a key cytokine and therapeutic target in colon cancer. Cytokine Growth Factor Rev 2015;26:451–61. 10.1016/j.cytogfr.2015.03.002 [DOI] [PubMed] [Google Scholar]
- 18.Figueiredo CR, Azevedo RA, Mousdell S, et al. Blockade of MIF-CD74 signalling on macrophages and dendritic cells restores the antitumour immune response against metastatic melanoma. Front Immunol 2018;9:1132. 10.3389/fimmu.2018.01132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zheng Y-X, Yang M, Rong T-T, et al. CD74 and macrophage migration inhibitory factor as therapeutic targets in gastric cancer. World J Gastroenterol 2012;18:2253–61. 10.3748/wjg.v18.i18.2253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Balogh KN, Templeton DJ, Cross JV. Macrophage migration inhibitory factor protects cancer cells from immunogenic cell death and impairs anti-tumor immune responses. PLoS One 2018;13:e0197702. 10.1371/journal.pone.0197702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee S-J, Rossi RJ, Lee S-K, et al. CD134 costimulation couples the CD137 pathway to induce production of supereffector CD8 T cells that become IL-7 dependent. J Immunol 2007;179:2203–14. 10.4049/jimmunol.179.4.2203 [DOI] [PubMed] [Google Scholar]
- 22.Markley JC, Sadelain M. IL-7 and IL-21 are superior to IL-2 and IL-15 in promoting human T cell-mediated rejection of systemic lymphoma in immunodeficient mice. Blood 2010;115:3508–19. 10.1182/blood-2009-09-241398 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pellegrini M, Calzascia T, Elford AR, et al. Adjuvant IL-7 antagonizes multiple cellular and molecular inhibitory networks to enhance immunotherapies. Nat Med 2009;15:528–36. 10.1038/nm.1953 [DOI] [PubMed] [Google Scholar]
- 24.Kim B-J, Kim B-R, Kook Y-H, et al. Development of a live recombinant BCG expressing human immunodeficiency virus type 1 (HIV-1) Gag using a pMyong2 vector system: potential use as a novel HIV-1 vaccine. Front Immunol 2018;9:643. 10.3389/fimmu.2018.00643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kim B-J, Gong J-R, Kim G-N, et al. Recombinant Mycobacterium smegmatis with a pMyong2 vector expressing human immunodeficiency virus type I Gag can induce enhanced virus-specific immune responses. Sci Rep 2017;7:44776. 10.1038/srep44776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tan YS, Lei YL. Isolation of tumor-infiltrating lymphocytes by Ficoll-Paque density gradient centrifugation. Methods Mol Biol 2019;1960:93–9. 10.1007/978-1-4939-9167-9_8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lee H, Kim B-J, Kim B-R, et al. The development of a novel Mycobacterium-Escherichia coli shuttle vector system using pMyong2, a linear plasmid from Mycobacterium yongonense DSM 45126T. PLoS One 2015;10:e0122897. 10.1371/journal.pone.0122897 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Oliveira CS, de Bock CE, Molloy TJ, et al. Macrophage migration inhibitory factor engages PI3K/Akt signalling and is a prognostic factor in metastatic melanoma. BMC Cancer 2014;14:630. 10.1186/1471-2407-14-630 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Martínez-Lostao L, Anel A, Pardo J. How do cytotoxic lymphocytes kill cancer cells? Clin Cancer Res 2015;21:5047–56. 10.1158/1078-0432.CCR-15-0685 [DOI] [PubMed] [Google Scholar]
- 30.Ostrand-Rosenberg S, Fenselau C. Myeloid-derived suppressor cells: immune-suppressive cells that impair antitumor immunity and are sculpted by their environment. J Immunol 2018;200:422–31. 10.4049/jimmunol.1701019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Otvos B, Silver DJ, Mulkearns-Hubert EE, et al. Cancer stem cell-secreted macrophage migration inhibitory factor stimulates myeloid derived suppressor cell function and facilitates glioblastoma immune evasion. Stem Cells 2016;34:2026–39. 10.1002/stem.2393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Guda MR, Rashid MA, Asuthkar S, et al. Pleiotropic role of macrophage migration inhibitory factor in cancer. Am J Cancer Res 2019;9:2760–73. [PMC free article] [PubMed] [Google Scholar]
- 33.Su H, Na N, Zhang X, et al. The biological function and significance of CD74 in immune diseases. Inflamm Res 2017;66:209–16. 10.1007/s00011-016-0995-1 [DOI] [PubMed] [Google Scholar]
- 34.Fingerle-Rowson G, Petrenko O, Metz CN, et al. The p53-dependent effects of macrophage migration inhibitory factor revealed by gene targeting. Proc Natl Acad Sci U S A 2003;100:9354–9. 10.1073/pnas.1533295100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Simpson KD, Templeton DJ, Cross JV. Macrophage migration inhibitory factor promotes tumor growth and metastasis by inducing myeloid-derived suppressor cells in the tumor microenvironment. J Immunol 2012;189:5533–40. 10.4049/jimmunol.1201161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dumitru CA, Gholaman H, Trellakis S, et al. Tumor-derived macrophage migration inhibitory factor modulates the biology of head and neck cancer cells via neutrophil activation. Int J Cancer 2011;129:859–69. 10.1002/ijc.25991 [DOI] [PubMed] [Google Scholar]
- 37.Bacher M, Metz CN, Calandra T, et al. An essential regulatory role for macrophage migration inhibitory factor in T-cell activation. Proc Natl Acad Sci U S A 1996;93:7849–54. 10.1073/pnas.93.15.7849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Soumoy L, Kindt N, Ghanem G, et al. Role of macrophage migration inhibitory factor (MIF) in melanoma. Cancers 2019;11:529. 10.3390/cancers11040529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mawhinney L, Armstrong ME, O’Reilly C, et al. Macrophage migration inhibitory factor (MIF) enzymatic activity and lung cancer. Mol Med 2014;20:729–35. 10.2119/molmed.2014.00136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Calandra T, Bernhagen J, Metz CN, et al. MIF as a glucocorticoid-induced modulator of cytokine production. Nature 1995;377:68–71. 10.1038/377068a0 [DOI] [PubMed] [Google Scholar]
- 41.Beswick EJ, Das S, Pinchuk IV, et al. Helicobacter pylori-induced IL-8 production by gastric epithelial cells up-regulates CD74 expression. J Immunol 2005;175:171–6. 10.4049/jimmunol.175.1.171 [DOI] [PubMed] [Google Scholar]
- 42.Yoo S-A, Leng L, Kim B-J, et al. MIF allele-dependent regulation of the MIF coreceptor CD44 and role in rheumatoid arthritis. Proc Natl Acad Sci U S A 2016;113:E7917–26. 10.1073/pnas.1612717113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yan X, Orentas RJ, Johnson BD. Tumor-derived macrophage migration inhibitory factor (MIF) inhibits T lymphocyte activation. Cytokine 2006;33:188–98. 10.1016/j.cyto.2006.01.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Garrido F, Aptsiauri N, Doorduijn EM, et al. The urgent need to recover MHC class I in cancers for effective immunotherapy. Curr Opin Immunol 2016;39:44–51. 10.1016/j.coi.2015.12.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Alban TJ, Bayik D, Otvos B, et al. Glioblastoma myeloid-derived suppressor cell subsets express differential macrophage migration inhibitory factor receptor profiles that can be targeted to reduce immune suppression. Front Immunol 2020;11:1191. 10.3389/fimmu.2020.01191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cho Y, Crichlow GV, Vermeire JJ, et al. Allosteric inhibition of macrophage migration inhibitory factor revealed by ibudilast. Proc Natl Acad Sci U S A 2010;107:11313–8. 10.1073/pnas.1002716107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Klein SL, Flanagan KL. Sex differences in immune responses. Nat Rev Immunol 2016;16:626–38. 10.1038/nri.2016.90 [DOI] [PubMed] [Google Scholar]
- 48.Grote A, Hiller K, Scheer M, et al. JCat: a novel tool to adapt codon usage of a target gene to its potential expression host. Nucleic Acids Res 2005;33:W526–31. 10.1093/nar/gki376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Garai J, Lóránd T, Molnár V. Ketone bodies affect the enzymatic activity of macrophage migration inhibitory factor. Life Sci 2005;77:1375–80. 10.1016/j.lfs.2005.03.010 [DOI] [PubMed] [Google Scholar]
- 50.Rao X, Huang X, Zhou Z, et al. An improvement of the 2ˆ(-delta delta CT) method for quantitative real-time polymerase chain reaction data analysis. Biostat Bioinforma Biomath 2013;3:71–85. [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jitc-2021-003180supp001.pdf (81.2KB, pdf)
jitc-2021-003180supp002.pdf (247.5KB, pdf)
jitc-2021-003180supp003.pdf (383.2KB, pdf)
jitc-2021-003180supp004.pdf (382.8KB, pdf)
jitc-2021-003180supp005.pdf (389.3KB, pdf)
jitc-2021-003180supp006.pdf (546.7KB, pdf)
jitc-2021-003180supp007.pdf (384KB, pdf)
jitc-2021-003180supp008.pdf (198.4KB, pdf)
jitc-2021-003180supp009.pdf (2.7MB, pdf)
jitc-2021-003180supp010.pdf (37.7KB, pdf)
jitc-2021-003180supp011.pdf (311.5KB, pdf)
Data Availability Statement
All data relevant to the study are included in the article or uploaded as supplementary information. The original contributions presented in the study are included in the article/supplementary material; further inquiries can be directed to the corresponding author.