Abstract
The health of insulin-producing β cells is critical for normoglycemia. Wu et al.1 and Tang et al.2 provide evidence in vitro that β cells can be infected by SARS-CoV-2 virus, possibly contributing to worsening hyperglycemia seen during the COVID-19 pandemic.
The health of insulin-producing β cells is critical for normoglycemia. Wu et al.1 and Tang et al.2 provide evidence in vitro that β cells can be infected by SARS-CoV-2 virus, possibly contributing to worsening hyperglycemia seen during the COVID-19 pandemic.
Main text
The SARS-CoV-2 virus has created the COVID-19 global pandemic, leading to over 4 million deaths worldwide, according to the World Health Organization. Whereas the acute respiratory effects of SARS-CoV-2 infection are apparent, the long-term consequences of the virus remain unknown. Since the initial months of the pandemic, there has been much discussion of the relationship between SARS-CoV-2 and diabetes. Studies have highlighted the increased COVID-19 severity and mortality in patients with diabetes.3 Moreover, there appear to be higher rates of hyperglycemia and diabetic ketoacidosis in patients with diabetes who develop COVID-19.4 However, what remains unclear at present is whether infection with SARS-CoV-2 might itself precipitate new-onset diabetes, or possibly predispose patients to the development of diabetes in the longer term. Regardless, a key aspect of either exacerbation or initiation of diabetes is the health and viability of islet β cells, the relatively small number of which are responsible for the secretion of insulin. The loss or dysfunction of β cells is considered an underlying feature of all forms of diabetes, and therefore the potential that SARS-CoV-2 might infect these cells becomes salient with respect to both short- and long-term sequelae of COVID-19. Prior studies have demonstrated expression of the major SARS-CoV-2 entry factors ACE2 and TMPRSS2 in human β cells.5, 6, 7 However, in the absence of functional data, it has remained speculative if levels of these entry factors correlate with the ability of the virus to infect β cells.
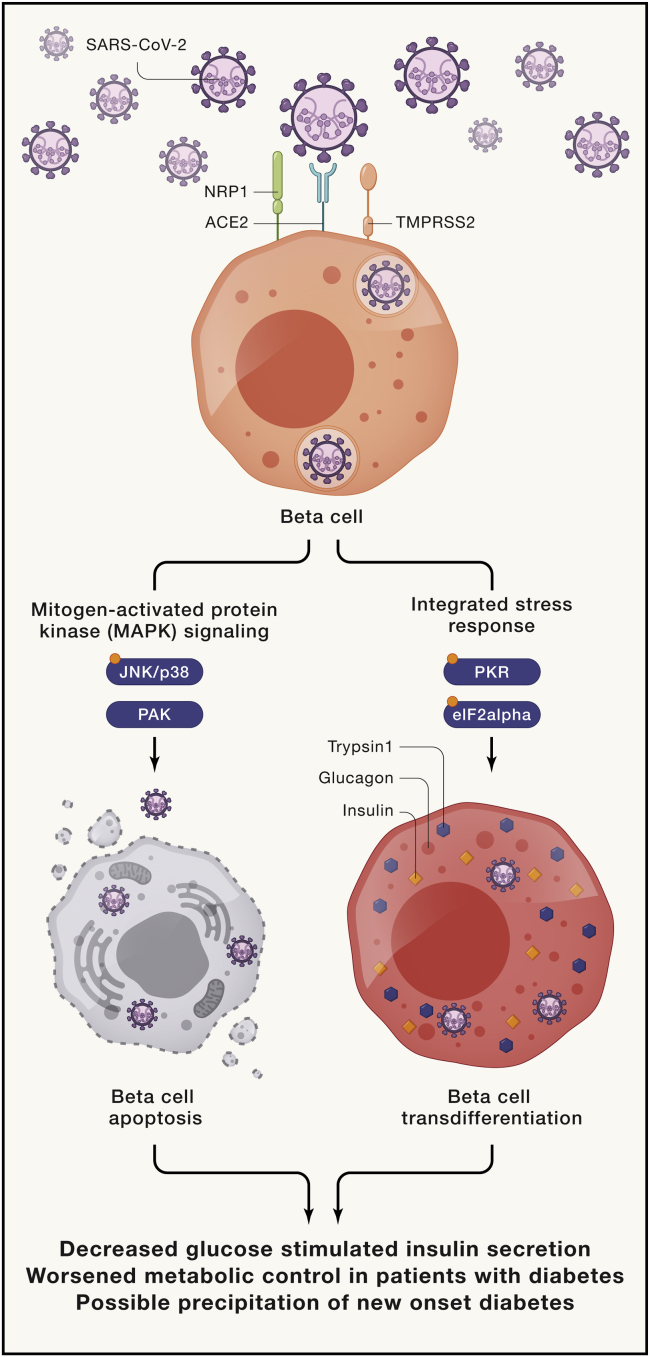
Two studies published recently by Wu et al.1 and Tang et al.2 address the potential for β cell infection by SARS-CoV-2. The authors of both studies asked several key questions: Do β cells contain factors conducive to viral entry? Can the virus enter β cells under conditions in vitro? What is the consequence of viral infection with respect to β cell function and survival? In the studies of Wu et al., the authors confirm the previously identified low levels of ACE2 and TMPRSS2 in β cells5,7 but suggest that additional entry factors—NRP1 and TRFC—may provide conduits for viral entry. The authors show that infection of isolated human islets with SARS-CoV-2 virus results in viral entry and islet dysfunction, as evidenced by reduced insulin secretion to glucose (Figure 1, left arm). Phosphoproteomics analysis revealed that the mitogen-activated protein kinase (MAPK)/c-Jun N-terminal kinase (JNK) pathways are subsequently activated and contribute to apoptosis of β cells. The importance of NRP1 as an entry factor was supported by experiments showing that its chemical inhibition partially reversed the dysfunction seen upon viral infection.
Figure 1.
SARS-CoV-2 virus infects human β cells, causing cell death and transdifferentiation leading to reduced insulin output
Upon infection with SARS-CoV-2 virus, human islets activate the mitogen-activated protein kinase (MAPK) signaling pathway (left) and the integrated stress response pathway (right), highlighted by key phosphorylation events (indicated by orange circles on JNK/p38, PKR, and eIF2alpha). Activation of these pathways lead to β cell apoptosis and transdifferentiation, respectively, resulting in decreased β cell insulin levels and lower glucose-stimulated insulin secretion. JNK, c-Jun N-terminal kinase; PAK, p21-activated kinase; PKR, protein kinase R; eIF2alpha, eukaryotic translation initiation factor 2 alpha.
The studies of Tang et al. also demonstrate the presence of entry factors—ACE2 and NRP1—in human β cells and show that β cells can be infected by SARS-CoV-2 in vitro. Additionally, these authors show that reduced insulin levels in infected cells are accompanied by increases in glucagon (a characteristic of α cells) and trypsin1 (a characteristic of exocrine cells) (Figure 1, right arm). This phenotypic alteration, or transdifferentiation, appears to be mediated by the activation of the kinase PKR and its inhibition of the translation factor eIF2α—events collectively referred to as the integrated stress response. An inhibitor of this response, trans-ISRIB, attenuated transdifferention and restored insulin production in β cells following infection.
Limitations should be noted in these studies. Both were conducted using human islets infected in vitro, and it is unknown if conditions conducive to viral entry into β cells exist in vivo in individuals with COVID-19. Although both studies demonstrated evidence of viral antigens in some autopsy samples from COVID-19 patients, the possibility that this represents uptake of viral debris (rather than actual virus) cannot be excluded. Also, given the heterogeneity of human islets, it remains possible that infectivity observed in vitro might be restricted to certain individuals. In this regard, not all autopsy samples interrogated in these studies showed clear evidence of viral particles in β cells. Therefore, a clearer understanding of the clinical characteristics of individuals who might be more susceptible to β cell infection should be investigated.
Collectively, the studies by Wu et al. and Tang et al. and another recent study by Müller et al.8 highlight several key concepts that have arisen about current perspectives on the pathogenesis of diabetes: First, it has been proposed that viral infection may be a trigger that initiates development of autoimmunity against β cells in type 1 diabetes. In a recent publication, Qadir et al.9 demonstrated pancreatic fibrosis and thrombosis accompanying new-onset diabetes in both human patients and non-human primates with COVID-19. Despite these findings, there does not appear to be a clear increase in type 1 diabetes incidence during the pandemic.10 Because type 1 diabetes develops months to years after the initiation of autoimmune β cell destruction, longer-term follow-up studies of COVID-19 patients are needed to determine the effect of infection upon development of type 1 diabetes. Second, in the case of type 2 diabetes, exacerbation of the disease is thought to be closely related to the death and/or phenotypic alteration of β cells. Both of these processes were observed in the studies noted here (see Figure 1), and both studies showed evidence of viral presence in β cells in autopsy specimens, lending credence to the notion that direct infection of β cells by SARS-CoV-2 might be exacerbating hyperglycemia. Finally, these studies emphasize potential therapeutic approaches in the setting of acute COVID-19, whereby therapies targeting viral entry and/or the integrated stress response might attenuate short-term hyperglycemia and possibly longer-term sequelae on β cells that are yet to be defined.
References
- 1.Wu C.-T., Lidsky P.V., Xiao Y., Lee I.T., Cheng R., Nakayama T., Jiang S., Demeter J., Bevacqua R.J., Chang C.A. SARS-CoV-2 infects human pancreatic β cells and elicits β cell impairment. Cell Metab. 2021 doi: 10.1016/j.cmet.2021.05.013. S1550-4131(21)00230-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tang X., Uhl S., Zhang T., Xue D., Li B., Vandana J.J., Acklin J.A., Bonnycastle L.L., Narisu N., Erdos M.R. SARS-CoV-2 infection induces beta cell transdifferentiation. Cell Metab. 2021 doi: 10.1016/j.cmet.2021.05.015. S1550-4131(21)00232-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Apicella M., Campopiano M.C., Mantuano M., Mazoni L., Coppelli A., Del Prato S. COVID-19 in people with diabetes: understanding the reasons for worse outcomes. Lancet Diabetes Endocrinol. 2020;8:782–792. doi: 10.1016/S2213-8587(20)30238-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li J., Wang X., Chen J., Zuo X., Zhang H., Deng A. COVID-19 infection may cause ketosis and ketoacidosis. Diabetes Obes. Metab. 2020;22:1935–1941. doi: 10.1111/dom.14057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Coate K.C., Cha J., Shrestha S., Wang W., Gonçalves L.M., Almaça J., Kapp M.E., Fasolino M., Morgan A., Dai C., HPAP Consortium SARS-CoV-2 Cell Entry Factors ACE2 and TMPRSS2 Are Expressed in the Microvasculature and Ducts of Human Pancreas but Are Not Enriched in β Cells. Cell Metab. 2020;32:1028–1040.e4. doi: 10.1016/j.cmet.2020.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fignani D., Licata G., Brusco N., Nigi L., Grieco G.E., Marselli L., Overbergh L., Gysemans C., Colli M.L., Marchetti P. SARS-CoV-2 Receptor Angiotensin I-Converting Enzyme Type 2 (ACE2) Is Expressed in Human Pancreatic β-Cells and in the Human Pancreas Microvasculature. Front. Endocrinol. (Lausanne) 2020;11:596898. doi: 10.3389/fendo.2020.596898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kusmartseva I., Wu W., Syed F., Van Der Heide V., Jorgensen M., Joseph P., Tang X., Candelario-Jalil E., Yang C., Nick H. Expression of SARS-CoV-2 Entry Factors in the Pancreas of Normal Organ Donors and Individuals with COVID-19. Cell Metab. 2020;32:1041–1051.e6. doi: 10.1016/j.cmet.2020.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Müller J.A., Groß R., Conzelmann C., Krüger J., Merle U., Steinhart J., Weil T., Koepke L., Bozzo C.P., Read C. SARS-CoV-2 infects and replicates in cells of the human endocrine and exocrine pancreas. Nat Metab. 2021;3:149–165. doi: 10.1038/s42255-021-00347-1. [DOI] [PubMed] [Google Scholar]
- 9.Qadir M.M.F., Bhondeley M., Beatty W., Gaupp D.D., Doyle-Meyers L.A., Fischer T., Bandyopadhyay I., Blair R.V., Bohm R., Rappaport J. SARS-CoV-2 infection of the pancreas promotes thrombo-fibrosis and is associated with new-onset diabetes. JCI Insight. 2021 doi: 10.1172/jci.insight.151551. Published online July 9, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tittel S.R., Rosenbauer J., Kamrath C., Ziegler J., Reschke F., Hammersen J., Mönkemöller K., Pappa A., Kapellen T., Holl R.W., DPV Initiative Did the COVID-19 Lockdown Affect the Incidence of Pediatric Type 1 Diabetes in Germany? Diabetes Care. 2020;43:e172–e173. doi: 10.2337/dc20-1633. [DOI] [PMC free article] [PubMed] [Google Scholar]