T cells have long been known as the core of adaptive immunity and play pivotal roles in host defense (Zhu et al., 2010). There are two main types of T cells in our body, cytotoxic T cells (CD8+) and helper T (Th) cells (CD4+), the latter comprised of Th1, Th2, Th17, and Treg subsets. Increasing evidence supports that metabolic reprogramming of T cells leads to dramatic changes in tissue microenvironments, which may alter whole-body energy homeostasis and metabolism, beyond their roles in adaptive immunity (Varanasi et al., 2020).
Obesity is accompanied by low-grade chronic inflammation in adipose tissue, where pro-inflammatory Th1 and CD8+ T cells gradually overweigh the anti-inflammatory Th2 and Treg cells. Evidence accumulated over the past decade strongly suggests that pro-inflammatory T cells play a major contributing role in accelerating adipose inflammation and insulin resistance. More recently, increasing studies have uncovered a potential role of T cells in regulating energy homeostasis. Enhancing sympathetic tone greatly increases Treg cells in brown adipose tissue (BAT) and inguinal adipose tissue (iWAT), whereas Treg cell ablation significantly impairs BAT thermogenic capacity (Kalin et al., 2017). Treg cell-induced BAT thermogenesis is also found to be promoted by IL-17-producing γδ T cells (Kohlgruber et al., 2018). However, beige fat development, which is critical for thermogenesis in iWAT, is inhibited by CD8+ T cell-mediated IFN-γ secretion (Moysidou et al., 2018). Despite these studies, the roles and mechanisms of fat-resident T cells in the regulation of adipose tissue function and energy expenditure remain largely unclear.
Zhou et al. (2021) recently showed that mimicking high-fat diet (HFD) feeding by β3 adrenergic receptor stimulation decreased the expression of the mitochondrial-localized chaperone protein disulfide bond A oxidoreductase-like protein (DsbA-L) in T cells. T cell-specific knockout of DsbA-L significantly impaired T cell mitochondrial function but promoted diet-induced thermogenesis in BAT, which was accompanied with reduced HFD-induced obesity, alleviated hepatosteatosis, and improved insulin resistance in mice. They further showed that DsbA-L deficiency in T cells reduced IFN-γ production, concurrently with enhanced BAT thermogenesis by increasing cyclic adenosine monophosphate (cAMP)‒protein kinase A (PKA) signaling. These results identify T cells as a key regulator of BAT function and provide new insights into the mechanism by which adipose-resident T cells communicate with neighboring brown adipocytes to regulate BAT thermogenesis and whole-body energy homeostasis.
How DsbA-L deficiency inhibits IFN-γ production in BAT? T cell differentiation and function are orchestrated by optimal mitochondrial metabolism. In this study, Zhou et al. (2021) demonstrated that DsbA-L deficiency in T cells reduces oxygen consumption rates, an indicator of mitochondrial oxidative phosphorylation, in both CD4+ and CD8+ T cells. They also found that T cell mitochondrial DNA content, adenosine triphosphate production, mitochondrial fusion, and mitochondrial calcium levels are all decreased upon T cell receptor stimulation. These results further demonstrate that DsbA-L deficiency decreases T cell mitochondrial respiration. Interestingly, changes in mitochondrial respiration have been suggested as a key cellular mechanism regulating T cytokine production (Tsai et al., 2018). Indeed, components of the electron transport chain complex have been found to play distinct roles in regulating T cell polarization and cytokine production (Bailis et al., 2019). Therefore, DsbA-L deficiency may reprogram T cells via a mitochondria-dependent mechanism, which could be tested by restoring mitochondrial function via genetic or pharmacological approaches in T cell-specific DsbA-L knockout mice.
The mechanisms by which T cells adopt different metabolic programs within different non-lymphoid tissues remain largely unknown. It is suggested that T cells in different tissues may be shaped by a tissue-specific microenvironment (Varanasi et al., 2020). Consistent with this, overnutrition had distinct effects on T cell mitochondrial function in mouse BAT and iWAT, with the mitochondrial function of BAT-resident T cells more significantly impaired than that of iWAT-resident T cells (Zhou et al., 2021). In addition, DsbA-L ablation in T cells led to a greater decrease of IFN-γ-producing Th1 and CD8+ T cells in BAT compared with that in iWAT. To be noted, no obvious changes in IFN-γ levels were observed in T cells from the spleen, reflecting a crucial role of metabolic milieu in T cell function (Zhou et al., 2021). A possible explanation for the tissue-specific effect on IFN-γ production is that BAT displays more sympathetic innervation compared to iWAT, thus taking the advantage of transmitting diet-induced signals to regulate BAT thermogenic capacity (Vaughan et al., 2014). Moreover, we found that uncoupling protein 1 (UCP1) expression is elevated in BAT but decreased in iWAT after HFD feeding, further suggesting a difference in metabolic milieu between BAT and iWAT upon HFD feeding. The findings by Zhou et al. (2021) expand the view on how different metabolic climates imprint T cells in different adipose tissues and how the reprogramming coordinates tissue function.
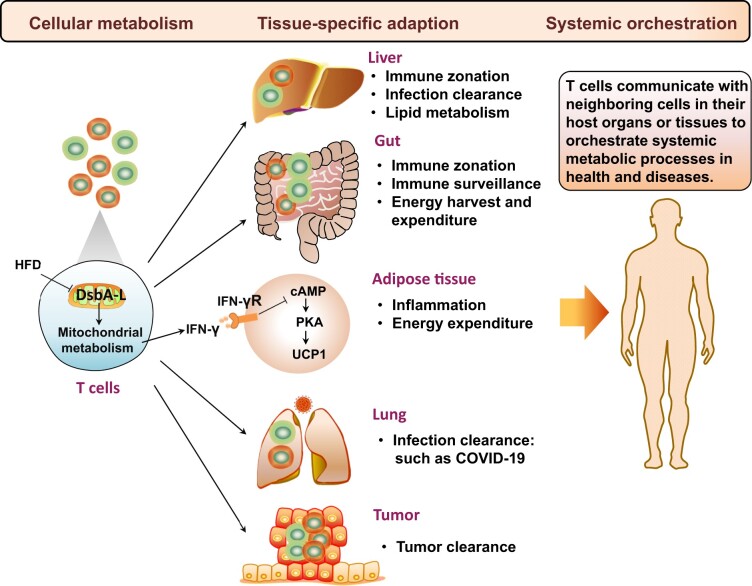
In addition to adipose tissue, T cells are widely distributed in other non-lymphoid tissues including liver, gut, and lung (Figure 1). However, the mechanisms regulating the tissue-specific metabolic adaption of T cells under specific metabolic climate in health and diseases remain largely unknown. Importantly, the gut-derived microbiota establishes immune zonation in the liver, forming an effective, spatially organized protective barrier against incoming blood-borne threats (Gola et al., 2021). Such zonation can also be found in the gut, where different segments are occupied by different ecologies of gut microbiota, dietary stimuli, and metabolites (Mowat and Agace, 2014). Since IFN-γ is a major effector cytokine in pathogen clearance, it is of interest to determine whether DsbA-L deficiency in T cells would affect liver and gut immune zonation and function. In addition, T cells play a critical role in antiviral immunity, but they appear functionally exhausted and their numbers are reduced significantly in COVID-19 patients (Diao et al., 2020). As mitochondrial dysfunction is a hallmark of effector T cell exhaustion, it would be worth determining whether DsbA-L deficiency contributes to T cell exhaustion in the lung. T cell exhaustion also occurs in tumor tissues, where Treg cells overwhelm effector T cells due to abundant lactate exposure. Since mitochondrial respiration is critical for lactate utilization, exploring whether mitochondrial reprogramming induced by DsbA-L deficiency contributes to effector T cell exhaustion or Treg cell persistency in tumors would be an interesting subject for further investigations. Lastly, with an appreciation to assess the spatiotemporal profiles of genes involved in T cell metabolism, one could anticipate identifying more intrinsic and extrinsic factors to modulate T cell function in health and diseases.
Figure 1.
T cell metabolism and function in a cell-intrinsic, tissue-specific, and system-orchestrated manner. DsbA-L, a mitochondrial localized protein, is essential for T cell mitochondrial respiration. HFD feeding decreases DsbA-L expression in adipose-resident T cells. T cell-specific DsbA-L knockout protects mice from HFD-induced obesity, alleviates hepatosteatosis, and improves insulin resistance due to enhance diet-induced thermogenesis. Mechanically, DsbA-L deficiency reduces IFN-γ-producing Th1 and CD8+ T cells in BAT, leading to elevated cAMP/PKA signaling in brown adipocytes and consequently increased UCP1 expression. Beyond adipose tissues, it is predictable that different microenvironment factors would also instruct T cell reprogramming and the specific function in distinct tissues. Overall, T cell homeostasis is firstly regulated by cellular metabolic pathways and then undergo tissue-specific metabolic reprogramming and adaption, ultimately orchestrated to regulate global state in health and diseases. IFN-γR, IFN-γ receptor. T cells in red: pro-inflammatory T cells; T cells in green: anti-inflammatory T cells.
In closing, mitochondrial metabolism plays a critical role in coordinating T cell adaptation within different tissues. Targeting T cell adaptation and function in a tissue-specific manner would not only shed new light on the development of novel therapeutic treatment for obesity, but also contribute to better understanding of the mechanisms modulating liver disease, gut inflammation, virus clearance, as well as tumor therapy.
[The work described was supported by grants from the National Natural Science Foundation of China (81730022 and 81600671), the National Key R&D Program of China (2018YFC2000100 and 2019YFA0801903), and Natural Science Foundation of Hunan Province, China (2019JJ50867).]
References
- Bailis W., Shyer J.A., Zhao J., et al. (2019). Distinct modes of mitochondrial metabolism uncouple T cell differentiation and function. Nature 571, 403–407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diao B., Wang C., Tan Y., et al. (2020). Reduction and functional exhaustion of T cells in patients with Coronavirus disease 2019 (COVID-19). Front. Immunol. 11, 827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gola A., Dorrington M.G., Speranza E., et al. (2021). Commensal-driven immune zonation of the liver promotes host defence. Nature 589, 131–136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalin S., Becker M., Ott V.B., et al. (2017). A Stat6/Pten axis links regulatory T cells with adipose tissue function. Cell Metab. 26, 475–492.e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohlgruber A.C., Gal-Oz S.T., LaMarche N.M., et al. (2018). γδ T cells producing interleukin-17A regulate adipose regulatory T cell homeostasis and thermogenesis. Nat. Immunol. 19, 464–474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mowat A.M., Agace W.W. (2014). Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 14, 667–685. [DOI] [PubMed] [Google Scholar]
- Moysidou M., Karaliota S., Kodela E., et al. (2018). CD8+ T cells in beige adipogenesis and energy homeostasis. JCI Insight 3, e95456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai S., Clemente-Casares X., Zhou A.C., et al. (2018). Insulin receptor-mediated stimulation boosts T cell immunity during inflammation and infection. Cell Metab. 28, 922–934.e4. [DOI] [PubMed] [Google Scholar]
- Varanasi S.K., Kumar S.V., Rouse B.T. (2020). Determinants of tissue-specific metabolic adaptation of T cells. Cell Metab. 32, 908–919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaughan C.H., Zarebidaki E., Ehlen J.C., et al. (2014). Analysis and measurement of the sympathetic and sensory innervation of white and brown adipose tissue. Methods Enzymol. 537, 199–225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou H., Peng X., Hu J., et al. (2021). DsbA-L deficiency in T cells promotes diet-induced thermogenesis through suppressing IFN-γ production. Nat. Commun. 12, 326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu J., Yamane H., Paul W.E. (2010). Differentiation of effector CD4 T cell populations*. Annu. Rev. Immunol. 28, 445–489. [DOI] [PMC free article] [PubMed] [Google Scholar]