Abstract
Whether low diastolic blood pressure (DBP) is a risk factor for recurrent cardiovascular disease (CVD) events in persons with isolated systolic hypertension (ISH) is controversial. We studied 791 individuals (mean age 75 years, 47% female, mean follow-up time: 8±6 years) with DBP<70 (N=225) versus 70–89 mmHg (N=566) following initial CVD events in the original and offspring cohorts of the Framingham Heart Study. Recurrent CVD events occurred in 153 (68%) participants with lower DBP and 271 (48%) with higher DBP (p<0.0001). Risk of recurrent CVD events in risk factor-adjusted Cox regression was higher in those with DBP<70 mmHg versus DBP 70–89 mmHg in both treated [HR, 5.1 (95% CI: 3.8–6.9) p<0.0001] and untreated individuals [HR, 11.7 (6.5–21.1) p<0.0001] (treatment interaction: p=0.10). Individually, coronary heart disease, heart failure, and stroke recurrent events were more likely with DBP<70 mmHg versus 70–89 mmHg (p<0.0001). To examine for an effect of wide pulse pressure on excess risk associated with low DBP, we defined 4 binary groupings of pulse pressure (≥ 68 verses <68 mmHg) and DBP (<70 versus 70–89 mmHg). CVD incidence rates were higher only in the group with PP≥68 and DBP<70 mmHg (76% versus 46–54%, p<0.001). Persons with ISH and prior CVD events have increased risk for recurrent CVD events in the presence of DBP<70 mmHg versus DBP 70–89 mmHg, whether treated or untreated, supporting wide pulse pressure as an important risk-modifier for the adverse effect of low DBP.
Keywords: isolated systolic hypertension, antihypertensive treatment, diastolic J-curves, epidemiology, population study, cardiovascular disease
For more than 30 years1, 2 there has been controversy regarding the clinical significance of low diastolic blood pressure (DBP) in patients treated for hypertension as it relates to increased cardiovascular disease (CVD) risk in general and to coronary heart disease (CHD) risk in particular. This has not been studied in those with pre-existing CVD, so in the present study, we limited our investigation to persons who survived an initial CVD event—CHD, heart failure (HF), or stroke. We divided these persons into 2 groups: (1) those with isolated systolic hypertension (systolic blood pressure [SBP] ≥140 and DBP<90 mm Hg) (ISH) with DBP 70–89 mm Hg, in the presence or absence of antihypertensive treatment; these persons are at higher risk due to widened pulse pressure and increased arterial stiffness3–5; and (2) persons with ISH with a low mean arterial pressure (MAP) that is associated with a very low DBP, i.e. <70 mm Hg in the absence or presence of antihypertensive treatment6, 7; we hypothesized that group 2 persons would be at even higher risk than group 1.
We addressed the following questions in the present investigation: First, is there increased risk for recurrent CVD events in individuals with a DBP of <70 mm Hg versus those with a DBP 70–89 mm Hg in those with ISH, regardless of the presence or absence of antihypertensive treatment? Second, is there increased risk for individual CVD components examined separately (CHD, HF or stroke events) in the presence versus absence of antihypertensive treatment in individuals with a DBP of <70 mm Hg versus a DBP 70–89 mm Hg? Third, can we judge the relative importance of wide pulse pressure versus low DBP in predicting CVD events?
Methods:
Overview:
The Framingham Heart Study began in 1948 enrolling 5209 men and women, 30 to 62 years of age who underwent repeated examinations biennially.8–10 In 1971, 5124 men and women who were children or the spouses of children of the original Framingham Heart Study were enrolled in the Framingham Offspring cohort.11The offspring cohort underwent repeated examinations every 4 years. A detailed description of this study design, the method for assessing BP, and the method of classifying CVD end points have been published elsewhere.12, 13 Initial and recurrent CVD events consisted of CHD (myocardial infarctions, coronary insufficiency, or sudden cardiac death), HF, or stroke. Inclusion criteria included post-primary CVD event survivors with the presence of ISH, with or without receiving antihypertensive treatment. Exclusion criteria were the absence of a study visit within 5-years post-initial CVD event.
Study Sample:
As shown in the flow diagram (Figure 1), inclusion criteria resulted in 791 participants who survived their initial CVD event, appeared for at least ≥ one post-initial CVD event visit within 5-years of their initial event, and had evidence of ISH. There were an additional 130 initial CVD event survivors that were excluded from the analysis: 121 had insufficient risk-factor covariates for Cox regression modeling and 19 had their first visits beyond the 5-years post-initial CVD exclusion criterion. Of the initial 791 event survivors with post-initial event visits, 367 were without recurrent CVD events and therefore were censored: 72 with DBP <70 mm Hg and 295 with DBP 70–89 mm Hg. Of the 424 subjects with recurrent CVD events, 153 had DBP <70 mm Hg and 271 had DBP 70–89 mm Hg. We began the study with the second biannual examination (1950) and extended it through 2005. Because of the long duration of the study, we tested for secular trend by adjusting for consecutive decades of occurrence of index primary events (<1960s, 1960–1969, 1970–1979, ≥1980).
Figure 1:
Flow diagram depicts the 791 initial hypertensive cardiovascular disease (CVD) event survivors with ≥1 visits who qualified for this study. *An additional 130 persons with recurrent CVD events and office visits were excluded from the analysis: 121 with missing covariates and 19 with first visits beyond the 5-year post-initial CVD event exclusion criterion.
Assessment of risk factors and cardiovascular events:
Current cigarette smoking was defined as regularly smoking cigarettes at any time during the prior year. Body mass index (BMI) was calculated as body weight (in kilograms) divided by the square of height (in meters). A fasting blood glucose level of ≥7.0 mmol/L (126 mg/dL) (in the offspring cohort), a nonfasting glucose of ≥11.0 mmol/L (≥200 mg/dL) (in the original cohort), or the use of hypoglycemic medications (in both cohorts) defined diabetes mellitus, and serum cholesterol (high-density-lipoprotein cholesterol was not included as it was not available for baseline visits prior to 1970). Hypertensive treatment refers to any medication(s) prescribed specifically for hypertension at the examination after the primary CVD event and/or prior to the recurrent CVD event; in the absence of this information, the participant was defined as not receiving antihypertensive therapy.
Data Analysis:
At the first available visit after the initial event, comparisons were made between participants with DBP <70 versus DBP 70–89 mm Hg (Table 1). In addition, the 826 individuals who had secondary events without visits between their initial and recurrent events did not qualify for Cox regression analysis; nevertheless, we compared characteristics of these 826 with 424 persons with visits and recurrent events. Comparisons between various groups utilized chi-square for categorical analysis and paired t-tests for continuous data.
Table 1:
Comparisons Between Subjects at the Post-Primary Clinic Visit
| Category | DBP 70–89 mmHg | DBP <70 mmHg | Total number |
|---|---|---|---|
|
| |||
| Number | 566 | 225 | 791* |
| Number Male (%) | 332 (59%) ‡ | 92 (41%) | 424 (53%) |
| Mean age at Primary event (y) | 66.2 ‡ | 74.6 | 68.6 |
| Mean age at Recurrent event (y) | 72.4 ‡ | 81.4 | 75.1 |
| Last SBP before Primary event (mmHg) | 154.4 ‡ | 148.7 | 152.8 |
| Last DBP before Primary event (mmHg) | 87.1 ‡ | 72.6 | 82.9 |
| * Last SBP before Recurrent event (mmHg) | 153.1 ‡ | 145.9 | 151.0 |
| * Last DBP before Recurrent event (mmHg) | 84.5 ‡ | 64.4 | 78.6 |
| * Last PP before Recurrent Event (mmHg) | 68.6 ‡ | 81.5 | 72.4 |
| BMI | 27.5 | 27.3 | 27.4 |
| Total Cholesterol (mg/dL) | 235.2 ‡ | 223.1 | 231.8 |
| Smokers (%) | 215 (38%) ‡ | 51 (23%) | 266 (34%) |
| DM (%) | 66 (12%) ‡ | 43 (19%) | 109 (14%) |
| Mean duration between Primary and Recurrent events (y) | 7.7 | 8.4 | 7.9 |
| Primary CHD (%) | 267 (47%) † | 84 (37%) | 351 (44%) |
| Primary HF (%) | 76 (13%) ‡ | 47 (21%) | 123 (16%) |
| Primary Stroke (%) | 223 (39%) | 94 (42%) | 317 (40%) |
| Recurrent CHD (%) | 135 (24%) | 49 (22%) | 184 (23%) |
| Recurrent HF (%) | 107 (19%) † | 58 (26%) | 165 (21%) |
| Recurrent Stroke (%) | 122 (22%) | 46 (20%) | 168 (21%) |
| Recurrent CVD (%) | 271 (48%) † | 153 (68%) | 424 (54%) |
| Mortality (%) | 65 (12%) | 16 (7%) | 81 (10%) |
Last BP category before recurrent CVD event or censoring
p<0.05,
p<0.01:
BP 70–89 mmHg versus DBP <70 mmHg
Cox regression models were performed calculating hazard ratios (HRs) and 95% confidence intervals (1) unadjusted, (2) adjusted for age and sex, and (3) adjusted additionally for BMI, total cholesterol, smoking, and diabetes to test the relation of DBP<70 mm Hg versus DBP 70–89 mm Hg for recurrent CVD event risk (CHD, HF, and stroke events) examined together and separately as individual events in models for (a) combined antihypertensive treated and untreated, (b) antihypertensive treated, and (c) untreated individuals. All models were stratified by quintiles of time from the date of the primary CVD event to the first post-examination visit (Cut-points for quintiles in days were <165, 166–317, 318–471, 472–650, and 651–1806). Lastly, interaction terms were added to test for homogeneity of the DBP effect by treatment status
To judge the potential modifying effect of wide pulse pressure on excess risk associated with low DBP, we defined 4 groups from binary groupings of median pulse pressure (≥ 68 verses <68 mmHg) and categorical DBP (<70 versus 70–89 mmHg), followed by Cox modeling with those in the lowest pulse pressure-DBP group defined as reference category.
Results:
Comparison between participants with DBP<70 mm Hg versus those with DBP 70–89 mm Hg:
The participants with DB <70 mm Hg were older by almost a decade at the time of their recurrent event (p<0.01) (Table 1). The prevalence of metabolic abnormalities was comparable in participants with DBP<70 mm Hg versus DBP 70–89 mm Hg. There were 408 persons with initial and 211 recurrent CHD events; 138 initial and 181 recurrent HF events; and 375 initial and 185 recurrent stroke events. The mean blood pressures at the last available clinic visit in participants treated with antihypertensive medication before the recurrent CVD events (not shown in Table 1) were 146/64 mm Hg in those with DBP <70 mm Hg and 153/85 mm Hg in those with DBP 70–89 mm Hg (p<0.01).
Comparison between individuals with visits versus without visits between events:
Participants without visits between initial and recurrent events were older by 5.2 years than in those with visits between events (p<0.01). The most striking difference in those without visits between events was the close temporal relation between initial and recurrent CVD events: 47% of treated and 42% of untreated recurrent CVD events occurred within one week of the initial CVD events (p<0.01); 64% of the treated and 56% of untreated subjects occurred within one month of the initial events (p<0.01). The mean follow-up time to recurrent events was 1.9 years in those without visits between events and 7.9 years in those with ≥1 visits between events (p<0.01). The proportion of the 3 types of recurrent CVD events (CHD, HF and stroke) did not differ between those with and without post-initial visits. However, CVD deaths were significantly more common in people without post-initial CVD event visits (40% untreated, 32% treated) in comparison to those with visits between events (12% untreated, 10% treated) (p<0.01).
Bivariate analysis:
Of 791 hypertensive participants (mean age 75 years, female 47%), who survived their initial CVD event, 225 (28%) had DBP<70 mm Hg and 566 (72%) had DBP 70–89 mm Hg, of which 153 of 225 (68%) and 271 of 566 (48%), respectively, experienced CVD events (p<0.0001). The mean follow-up time between the initial and recurrent events was 8.0±6 years.
Cox proportional hazard regression modeling:
There was an increased risk of recurrent CVD events in risk factor-adjusted Cox regression models with DBP<70 mmHg versus DBP 70–89 mmHg in combined treated and untreated persons [HR, 5.9 (95% CI: 4.6–7.5) p<0.0001] after adjusting for age, sex, BMI, total cholesterol, smoking, and diabetic status (Table 2 and Figure 2). Adjustment for SBP as a continuous variable in the regression model enhanced the hazard ratio slightly [HR, 6.2 (95% CI: 4,8–8.1) p<0.0001. In addition, the increased CVD risk associated with low DBP was present in both treated [HR, 5.1 (95% CI: 3.8–6.9) p<0.0001] and untreated persons [HR, 11.7 (6.5–21.1) p<0.0001], (treatment interaction: p=0.9). Furthermore, DBP<70 mm Hg was associated with increased risk for single CVD entities of CHD, HF, and stroke in treated and untreated groups (Figure 2). All treatment interactions were not significant. The test for secular trend in Cox modeling did not weaken HRs for increased risk in persons with DBP<70 mm Hg versus DBP 70–89 mm Hg; in fact they were slightly strengthened (HR, 6.8 (95% CI: 5.2–8.8) p<0.0001).
Table 2.
Cox Regressions for Combined CVD Events
| Treated and Untreated |
| Total CVD | Chi-Square | p-value | HR | 95% CI |
|---|---|---|---|---|
|
| ||||
| Unadjusted | ||||
| DBP<70 vs. 70–89 mm Hg | 238.4 | <.0001 | 5.9 | 4.7–7.4 |
| Age- and Sex-Adjusted | ||||
| DBP<70 vs. 70–89 mm Hg | 190.8 | <.0001 | 5.7 | 4.5–7.3 |
| Age (per SD) | 4.3 | 0.0373 | 1.0 | 1.0–1.0 |
| Sex (male vs. female) | 7.5 | 0.0063 | 0.7 | 0.6–0.9 |
| * Fully Adjusted | ||||
| DBP<70 vs. 70–89 mm Hg | 189.6 | <.0001 | 5.9 | 4.6–7.5 |
| Age (per SD) | 9.1 | 0.0026 | 1.2 | 1.1–1.4 |
| Sex (male vs. female) | 10.8 | 0.001 | 0.7 | 0.6 –0.9 |
| BMI (per SD) | 0.016 | 0.898 | 0.99 | 0.9 –1.1 |
| Total Cholesterol (per SD) | 10.0 | 0.0016 | 1.2 | 1.1 –1.3 |
| Smoking (current vs. other) | 0.8 | 0.3701 | 1.1 | 0.9–1.4 |
| Diabetes (yes vs. no) | 14.1 | 0.0002 | 1.7 | 1.3–2.2 |
| Treated Only |
| Total CVD | Chi-Square | p-value | HR | 95% CI |
|---|---|---|---|---|
|
| ||||
| Unadjusted | ||||
| DBP<70 vs. 70–89 mm Hg | 155.9 | <.0001 | 5.3 | 4.1–6.9 |
| Age- and Sex-Adjusted | ||||
| DBP<70 vs. 70–89 mm Hg | 121.4 | <.0001 | 4.9 | 3.7– 6.5 |
| Age (per SD} | 10.4 | 0.0012 | 1.0 | 1.0–1.0 |
| Sex (male vs. female) | 5.5 | 0.0188 | 0.7 | 0.6–1.0 |
| * Fully Adjusted | ||||
| DBP<70 vs. 70–89 mm Hg | 122.3 | <.0001 | 5.1 | 3.8–6.9 |
| Age (per SD) | 16.4 | <.0001 | 1.4 | 1.2–1.7 |
| Sex (male vs. female) | 9.5 | 0.0021 | 0.7 | 0.5–0.9 |
| BMI (per SD) | 0.4 | 0.5071 | 1.0 | 0.9–1.2 |
| Total Cholesterol (per SD) | 10.4 | 0.0013 | 1.3 | 1.1–1.4 |
| Smoking (current vs. other) | 0.02 | 0.8762 | 0.9 | 0.7–1.3 |
| Diabetes (yes vs. no) | 10.1 | 0.0015 | 1.7 | 1.2–2.3 |
| Treated Only |
| Total CVD | Chi-Square | p-value | HR | 95% CI |
|---|---|---|---|---|
|
| ||||
| Unadjusted | ||||
| DBP < 70 vs. 70–89 mm Hg | 73.8 | <.0001 | 9.2 | 5.5–15.2 |
| Age and sex-adjusted | ||||
| DBP < 70 vs. 70–89 mm Hg | 70.3 | <.0001 | 12.0 | 6.7–21.5 |
| Age (per *SD) | 3.2 | 0.0742 | 0.9 | 0.9–1.0 |
| Sex | 0.7 | 0.4033 | 0.8 | 0.6–1.2 |
| * Fully Adjusted | ||||
| DBP < 70 vs. 70–89 mm Hg | 67.0 | <.0001 | 11.7 | 6.5–21.0 |
| Age (per SD) | 1.6 | 0.2063 | 0.9 | 0.7–1.1 |
| Sex (male vs. female) | 1.4 | 0.2332 | 0.8 | 0.5–1.2 |
| BMI (per SD) | 3.0 | 0.0823 | 0.8 | 0.7–1.0 |
| Total Cholesterol (per SD) | 1.7 | 0.1981 | 1.1 | 0.9–1.3 |
| Smoking (yes vs. no) | 0.7 | 0.3967 | 1.2 | 0.8–1.8 |
| Diabetes (yes vs. no) | 3.9 | 0.0492 | 1.7 | 1.0–2.9 |
N = 791 for all Cox models; Strata (quintiles) were determined using time between primary event and first post-primary event visit; SD = standard deviation;
Adjusted for Age per SD, Sex (male vs. female), BMI (per SD), Total Cholesterol (per SD), Smoking (yes vs. no), Diabetes (yes vs. no)
Figure 2:
Fully adjusted hazard ratios (HRs) for total and individual cardiovascular disease (CVD) events [coronary heart disease (CHD), heart failure (HF), and stroke)], respectively, occurring in subjects with DBP<70 mmHg versus DBP 70–89 mmHg in (a) treated and untreated, (b) treated, and (c) untreated groups, respectively. (All HRs p<0.0001). The Y-axis refers to the comparison between DBP<70 mm Hg versus DBP 70–89 mm Hg.
Wide pulse pressure modifies the effect of low DBP on CVD risk.
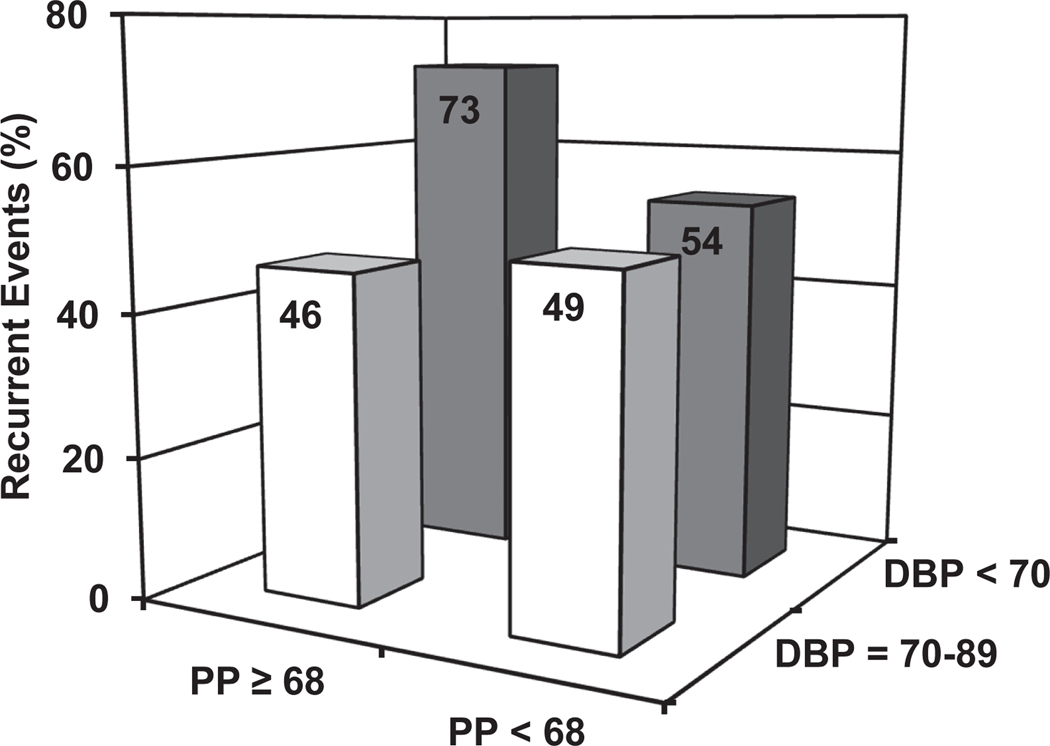
For the 791 post-initial CVD event survivors, the combination of pulse pressure ≥ 68 together with DBP <70 mm Hg had the highest incidence of subjects who developed CVD events; (Figure 3). In Cox regression, with the lowest PP-DBP grouping as the reference category, the adjusted HR for DBP<70 mm Hg and pulse pressure ≥68 mm Hg was 2.40 (95%CI: 1.6–3.7) p<0.0001, consistent with the hypothesis that increased CVD risk was associated with a pulse pressure ≥68 mm Hg and a DBP <70 mm Hg.
Figure 3:
Bar graph depicts the 4 possible binary combinations of median pulse pressure cut-points (≥68 verses <68 mm Hg) and DBP cut-points (<70 versus 70–89 mm Hg) that predicted recurrent cardiovascular disease (CVD) risk in the 791 initial CVD event survivors with ≥1 office visits. The highest event rate occurred in individuals with DBP<70 mm Hg and pulse pressures of ≥68 mm Hg that predicted CVD events significantly (p<0.0001 across the 4 DBP x pulse pressure groupings. Chi Square = 32.6). No other binary paring of pulse pressure and DBP showed significant prediction of CVD events.
Discussion
In a previous Framingham report14, we showed that middle-aged and older individuals with ISH in the absence of prior CVD events (CHD, HF, or stroke) and without antihypertensive treatment were at greater CVD risk in the presence of DBP<70 versus DBP 70–89 mm Hg; these conclusions were confirmed by the MORGAM Project study.15 Our present investigation is the first community-based study to show that both treated and untreated individuals with ISH and DBP <70 mm Hg were at greater risk for recurrent CVD events than those with DBP 70–89 mm Hg. Second, similar findings were present when examining single CVD endpoints of CHD, HF, or stroke. Third, CVD event rates were highest in the group with widened pulse pressure and DBP< 70 mm Hg as compared to those with both low pulse pressure and low DBP, whether treated or untreated, supporting wide pulse pressure as an important risk-modifier of the adverse effect from low DBP.
Frequency and CVD risk of DBP <70 mm Hg in persons with ISH.
We have previously shown in United States adults16 that DBP <70 mm Hg is present in 30% of untreated persons with ISH (versus 35% in treated). There was a 3-fold greater prevalence of CVD events from the highest to the lowest DBP strata in untreated ISH. Furthremore, advanced age, female sex, and diabetes mellitus, but not treatment status, were associated with low DBP. Similarly, Unger et al.17 showed that low ambulatory DBP was associated with greater all-cause mortality in older patients with ISH after adjusting for antihypertensive treatment and other covariates. Indeed, the Framingham Heart Study14 showed that a DBP of <70 mm Hg versus DBP 70–89 mm Hg in the absence of antihypertensive therapy could add a risk equivalent of about 20 mm Hg of increase in SBP—in other words, a potential risk-equivalent shift from prehypertension to stage 1 systolic hypertension or from stage 1 to stage 2 systolic hypertension. Moreover, as shown by Wang et al18, antihypertensive therapy will maximize the decrease in SBP and minimize the reduction in DBP in direct proportion to age-related widening of pulse pressure. Therefore, not only is DBP<70 mm Hg common and associated with increased CVD risk in untreated elderly persons with ISH, but also antihypertensive therapy has only a minimal effect in further lowering of DBP as compared with the lowering of SBP.
Antihypertensive Treatment as a cause of Recurrent CHD Events.
Because post-hoc analyses of intervention trials are fraught with bias and confounding, they cannot answer whether antihypertensive treatment induces CHD events in the presence of a low DBP. 19–21 Indeed, DBP declines with age, female sex, diabetes, chronic kidney disease, heart failure, incident cancer, and wider pulse pressure. If such patients experience CHD events in intervention trials, it is not necessarily that antihypertensive treatment caused the event, but rather that a low baseline DBP (and increased pulse pressure) predicted their future event. In a previous Framingham investigation, Kannel, et al.22 noted that CHD events in persons with low DBP were mainly in those with increased SBP, suggesting that ISH with increased pulse pressure may have been responsible, rather than the presence of antihypertensive treatment. Importantly, excessive treatment was not likely the cause of the low DBP because a lower frequency of treatment was related to a lower DBP.22 Furthermore, antihypertensive therapy in an elderly person with ISH preferentially decreases SBP over DBP18, 20, lowers pulse pressure, decreases arterial stiffness, and therefore theoretically, may improve the coronary oxygen supply/demand ratio of the left ventricle—thereby providing protection from ischemia. Indeed, the Framingham Heart Study results22 have been confirmed by the CALIBER programme (Linked Bespoke studies and the Electronic health Records of 1.25 million patients), which showed an absence of DBP J-curve except when accompanied by SBP≥140 mm Hg.23
Taken together, the findings in the present study suggest that antihypertensive treatment was not related to recurrent CHD events. First, the increase in CVD risk with DBP <70 mm Hg versus DBP 70–89 mm Hg was equally distributed in treated and untreated persons. Second, the increased CVD risk in association with DBP <70 mm Hg versus DBP 70–89 mm Hg was present separately for CHD, HF and stroke outcomes. Third, the absence of an interaction by treatment status post-DBP<70 mm Hg versus DBP 70–89 mm Hg was further evidence that the increased CHD event rate was not treatment-induced. In summary, the present study does not support antihypertensive treatment as a significant cause of increased CVD event risk in association with the onset of low DBP.
Significance of Subjects with Recurrent CVD Events Without Return Visits.
Of the 826 individuals who did not have visits between their initial and recurrent CVD events, there was a striking difference in clinical course as compared to the 577 who did attend one or more visits. First, the majority of participants without visits had a significantly shorter duration between events versus those with visits. Second, there was a very high mortality occurring in individuals that had their recurrent event soon after their initial event, both off and on antihypertensive treatments. Third, there were comparable mean BPs prior to pre-initial CVD events in both persons with visits (155/84 mm Hg) and without visits (155/80 mm Hg), suggesting that there were few persons with DBP<70 mm Hg prior to the onset of primary events.
Previous Framingham publications have shown that a significant post-infarction reduction in SBP is an indication of impaired cardiac function.24, Therefore, we can postulate that many subjects with early secondary events and high mortality after their initial events had impaired cardiac function and reversed causality.24, 25 Therefore, those who did attend office visits after their initial event to quality for our study likely had a significant survivor bias in comparison to those with post-initial CVD events without subsequent visits.
Causes of Increased Recurrent CVD Events in Association with Low DBP.
Aging plays an important role as an effect modifier in influencing the presence or absence of DBP in predicting CVD risk.26 From age 60 onward there is a shift in favor of pulse pressure over SBP as a predictor of CVD risk; thus, DBP now displays a J-curve of CVD risk in older people with ISH.26 Traditionally, increased pulse pressure as a marker of large artery stiffness has been shown to be an independent CVD risk factor.3–5 After 60 years of age, the fall in DBP and rapid widening of pulse pressure become surrogate markers of arterial stiffness. Furthermore, we postulate that the increase in CVD risk is in part related to a pernicious combination of faulty microvascular function resulting from increased elastic artery stiffness in combination with low diastolic perfusion pressure. Interestingly, our Cox regression analyses showed that low DBP is a much greater contributor to risk than age, sex, BMI, total cholesterol, smoking, or diabetes—attesting to the singular importance of the combination of wide pulse pressure and low DBP to recurrent CVD risk in persons with initial CVD events.
The J-curve controversy has been recently summarized in two articles entitled “Aggressive Blood Pressure Lowering is Dangerous: The J-Curve”: Con Side27 of the argument versus the Pro Side28 of the argument. However, neither of the authors considered a low DBP in association with ISH as predisposing to J-curve CVD events.
Strengths and Limitations.
The strengths of our investigation include a wide time interval of data collection in initial CVD event survivors and the standardized measurements from the well-characterized Framingham Heart Study. However, our data on antihypertensive therapy only indicates the presence of treatment for hypertension being prescribed after the index examination, with no information on adherence, type, or change in the number of agents or dosage from one visit to another, which could impact on our BP measures or outcomes. Furthermore, even though our medication variable specified an antihypertensive indication, these may have been prescribed for both antihypertensive and other cardiovascular indications. Lastly, we do not have information on physical functioning nor the possible presence of significant orthostatic hypotension. Nevertheless, the absence of higher HRs in the treatment versus untreated subjects supports a minimal role of antihypertensive treatment in predicting the increased risk of CVD events in those persons with DBP<70 mm Hg. Finally, of the 826 subjects without return visits after their initial CVD events that did not qualify for our main analysis, we can only speculate that many of these individuals had a likelihood of reverse causation with a high mortality rate within a short time after their primary CVD event.
Many factors interact to lower DBP and influence CHD risk, so only an intervention trial including subjects with low DBP, randomized to various active treatment levels of SBP-lowering, can establish whether high-baseline CVD risk can be modified by treatment. Furthermore, the optimal therapeutic reduction in SBP and DBP in the older patient with ISH that maximizes benefit is a separate question from the safety of lowering high SBP in the presence of low DBP.
Perspective:
Not only are elevated SBP and wide pulse pressure markers of increased arterial stiffness and contributors to increased CVD risk in persons with ISH, but also low DBP, particularly in association with elevated pulse pressure, contributes to increased CVD risk for CHD, HF, and stroke events regardless of the presence or absence of antihypertensive treatment. Whether specific risk factor modifications might be considered to address this possible excess risk in such individuals may be the subject of future investigations.
Novelty and Significance:
What is New?
Increased recurrent cardiovascular disease (CVD) risk in persons with isolated systolic hypertension, whether treated or untreated, was related to wide pulse pressure as a risk-modifier for the adverse effect of a low DBP<70 mm Hg.
What is Relevant?
We postulate that increased CVD risk is related to the pernicious effects of faulty microvascular function resulting from increased elastic artery stiffness in combination with low diastolic perfusion pressure.
Summary:
Persons with an initial CVD event and persistent isolated systolic hypertension who subsequently have DBP <70 mm Hg versus DBP 70–89 mmHg, have increased risk for a recurrent CVD event, supporting wide pulse pressure in combination with low DBP as important risk factors independent of treatment status.
Footnotes
Disclosures
None
References:
- 1.Stewart IMG. Relation of reduction in pressure to first myocardial infarction in patients receiving treatment for severe hypertension. Lance.t 1979;1:861–865. [DOI] [PubMed] [Google Scholar]
- 2.Cruickshank JM, Thorp JM Zacharias FJ. Benefits and potential harm of lowering high blood pressure. Lancet. 1987;1:581–584. [DOI] [PubMed] [Google Scholar]
- 3.Franklin SS, Gustin W, Wong ND, Larson MG, Weber MA, Kannel WB, Levy D. Hemodynamic patterns of age-related changes in blood pressure: the Framingham Heart study. Circulation. 1997;96:308–315. [DOI] [PubMed] [Google Scholar]
- 4.Benetos A, Safar M, Rudnichi A, Smulyan H, Richard JL, Ducimetiere P, Guize L. Pulse pressure: a predictor of long-term cardiovascular mortality in a French male population. Hypertension. 1997;30:1410–1415. [DOI] [PubMed] [Google Scholar]
- 5.Mitchell GF, Wang N, Palmisano JN, Larson MG, Hamburg NM, Vita JA, Levy D, Benjamin EJ, Vasan RS. Hemodynamic correlates of blood pressure across the adult age spectrum: noninvasive evaluation in the Framingham Heart Study. Circulation. 2010;122:1379–1386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Messerli FH, Mancia G, Conti CR, Hewkin AC, Kupfer S, Champion A, Kolloch R, Benetos A, Pepine CJ. Dogma disputed: Can aggressively lowering blood pressure in hypertensive patients with coronary artery disease be dangerous? Ann Intern Med. 2006;144:884–893. [DOI] [PubMed] [Google Scholar]
- 7.Bangalore S, Messerli FH, Wun CC, Zuckerman AL, DeMicco D, Kostis JB, LaRosa JC, for the Treating to New Targets Steering Committee and Investigators. J-curve revisited: an analysis of blood pressure and cardiovascular events in the Treating to New Targets (TNT) Trial. European Heart Journal. 2010;31:2897–2908. [DOI] [PubMed] [Google Scholar]
- 8.Dawber TR, Meadors GF, Moore FE Jr. Epidemiological approaches to heart disease. The Framingham Study. Am J public Health. 1951;41:279–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dawber TR, Kannel WB, Lyell LP. An approach to longitudinal studies in a community: the Framingham Study. Ann NY Acad Sci. 1963;107:539–556. [DOI] [PubMed] [Google Scholar]
- 10.Feinleib M, Kannel WB, Garrison RJ, McNamara PM, Castelli WP. The Framingham Offspring Study: design and preliminary data. Prev. Med. 1975:4:518–525. [DOI] [PubMed] [Google Scholar]
- 11.. Kannel WB, Wolf P, Garrison R. The Framingham Study: Section 35: Survival Following Initial Cardiovascular Events. Bethesda, Md. National Institutes of Health. 1998-5. [Google Scholar]
- 12.Cupple LA, D’Agostino RB, Some risk factors related to the annual incidence of cardiovascular disease and death using pooled repeated biennial measurments: In Kannel WB, Wolff PA Garrison RJ. Eds. The Framingham Heart Study; an Epidemiologic Investigation of Cardiovascular Disease. Washington DC: National Institutes of Health:1987:87:87–203. [Google Scholar]
- 13.Kannel WB, Wolff Pa, Garrison RJ. Eds. The Framingham Heart Study; an Epidemiologic Investigation of cardiovascular Disease. Washington DC: National Institutes of Health:1987:87–203. [Google Scholar]
- 14.Franklin SS, Lopez VA, Wong ND, Mitchell GF, Larson MG, Vasan RS, Levy D. Single versus combined blood pressure components and risk for cardiovascular disease. Circulation. 2009;119:243–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vishram JKK, Borglykke A, andreasen AH, et al. Impact of age on the importance of systolic and diastolic blood pressures for stroke risk: The Monica, Risk, Genetics, Archiving, and Monograph (MORGAM) Project. Hypertension. 12;60:1117–123. [DOI] [PubMed] [Google Scholar]
- 16.Franklin SS, Chow VH, Mori AD, Wong ND. The significance of low DBP in US adults with isolated systolic hypertension. J Hypertens. 2011;29:1101–1108. [DOI] [PubMed] [Google Scholar]
- 17.Unger A, Pepe G, Lambertucci L, Fedeli A, Monami M. Mannucci E, Gabbani L, Masotti G, Marchionni N, Di Bari M. Low diastolic ambulatory blood pressure is associated with greater all-cause mortality in older patients with hypertension. J Am Geriatr Soc. 2009;57:291–296. [DOI] [PubMed] [Google Scholar]
- 18.Wang JG, Staessen JA, Franklin SS, Fagard R, Gueyffier F. Systolic and diastolic blood pressure lowering as determinants of cardiovascular outcome. Hypertension. 2005;45:907–913. [DOI] [PubMed] [Google Scholar]
- 19.Williams B. Hypertension and the “J-Curve” J Am Coll Cardiol. 2009;54:1835–1836. [DOI] [PubMed] [Google Scholar]
- 20.Franklin SS. Isolated systolic hypertension and the J-curve of cardiovascular risk. Artery Research. 2010;4:1–6. [Google Scholar]
- 21.Kannel WB, Vasan RS. Aggressive lowering of blood pressure. Lancet. 2006;368:627–628. [DOI] [PubMed] [Google Scholar]
- 22.Kannel WB, Wilson PWF, Nam BH, D’Agostino RB. A likely explanation for the J-curve of blood pressure cardiovascular risk. Am J Cardiol. 2004;94:380–384. [DOI] [PubMed] [Google Scholar]
- 23.. Rapsomaniki E, Timmis A, George J, Pujades-Rodiguez M, Shah AD, Denaxas S, White IR, Caulfield MJ, Deanfield JE, Smeeth L, Williams B, Hingorani A, Hemingway H. Blood pressure and incidence of twelve cardiovascular diseases: lifetime risks, healthy life-years lost, and age-specific associations in 1.25 million people. Lancet. 2014;383:1899–1911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kannel WB, Sorlie P, Castelli WP, Mcgee D. Blood pressure and survival after myocardial infarction. Am J Cardiol. 1980;45:326–330. [DOI] [PubMed] [Google Scholar]
- 25.Marschner IC, Simes RJ, Keech A. Biases in the identification of risk factors thresholds and J-curves. Am J Epidemiol. 2007;166:824–831. [DOI] [PubMed] [Google Scholar]
- 26.Franklin SS, Larson MG, Khan SA, Wong ND, Leip EP, Kannel WB, Levy D. Does the relation of blood pressure to coronary heart disease risk change with aging? The Framingham Heart Study. Circulation. 2001;103:1245–1249. [DOI] [PubMed] [Google Scholar]
- 27.Verdecchia P, Angeli F, Mazzotta G, Garofoli M, Reboldi G. Aggressive blood pressure lowering is dangerous: the J-curve: con side of the argument. Hypertension. 2013;63:37–40. [DOI] [PubMed] [Google Scholar]
- 28.Mancia G, Grassi G. Aggressive blood pressure lowering is dangerous: the J-curve: pro side of the argument. Hypertension. 2013;63:29–36. [DOI] [PubMed] [Google Scholar]