Abstract
Background:
Sclerotic-type chronic graft-vs-host disease (cGVHD) of the skin is an uncommon but potentially debilitating sequela of allogeneic hematopoietic stem cell transplantation. There is no standardized assessment measure for this form of cGVHD. Because a full-thickness incisional biopsy specimen to the level of the fascia may be needed to make a definitive histologic diagnosis of cGVHD-related fasciitis, a noninvasive technique for the assessment and monitoring of sclerotic-type cGVHD, particularly cGVHD-related fasciitis, would be of potential value.
Observations:
Sixty-two consecutive patients with cGVHD following allogeneic hematopoietic stem cell transplantation were evaluated for sclerotic skin disease. Forty-four patients (71%) had cutaneous cGVHD, and 28 patients (45%) had evidence of sclerotic involvement based on physical examination findings. Fifteen patients agreed to undergo research magnetic resonance imaging to evaluate quantifiable changes in the dermis, subcutaneous tissue, and muscle. Among 15 patients, magnetic resonance imaging identified abnormalities in the skin in 7 (47%), subcutaneous fibrous septa in 13 (87%), deep fascia in 12 (80%), epimysium in 9 (60%), and muscle in 3 (20%).
Conclusions:
Magnetic resonance imaging should be considered in the evaluation of patients with cGVHD suspected of having subcutaneous or fascial involvement. Additional studies are needed to validate this noninvasive modality for serial monitoring of disease activity and response to therapy.
Trial Registration:
clinicaltrials.gov Identifier: NCT00331968
SCLEROTIC-TYPE CHRONIC graft-vs-host disease (cGVHD) is associated with fibrosis of the dermis, subcutaneous tissue, or fascia. Superficial dermal fibrosis results in small atrophic plaques resembling lichen sclerosus, localized thickening of the dermis leads to firm circumscribed plaques resembling morphea, widespread hide-bound thickening resembles scleroderma, and subcutaneous involvement of the fat septae and deeper fascial tissues resembles eosinophilic fasciitis.1-4
Patients with sclerodermalike fibrosis and those with fibrosis of the subcutaneous and fascial tissues develop range-of-motion limitations and joint contractures that may significantly affect daily activities. In patients with fasciitis, functional limitation may occur in the absence of overt superficial skin thickening or pigmentary changes characteristic of the morphealike and sclerodermalike forms of cGVHD. Visible signs in these patients may consist of prominent “grooves” in the skin caused by thickening of fascia ensheathing muscle groups or large superficial veins. Thickening of the fibrous fat septae imparts a subtle rippled or cellulitelike appearance of the skin, as well as a “nodular” texture appreciated by deep palpation.2
There are no standardized assessment measures for sclerotic-type cGVHD involving the skin and subcutaneous tissues. Because a full-thickness incisional biopsy specimen to the level of the fascia may be needed to make a definitive histologic diagnosis of cGVHD-related fasciitis, a noninvasive technique for the assessment and monitoring of this form of cGVHD would be of value. Magnetic resonance (MR) imaging is of diagnostic and prognostic usefulness in the evaluation of several diseases associated with soft-tissue abnormalities. Short tau inversion recovery (STIR) MR imaging has been used in dermatomyositis to detect skin, subcutaneous, and fascial edema, as well as muscle disease. The soft-tissue MR images in dermatomyositis correlated with clinical severity and in some cases preceded development of calcinosis.5 Magnetic resonance imaging abnormalities in patients with eosinophilic fasciitis correlated with histologic findings in fascial biopsy specimens, and improvement on MR images correlated with clinical treatment response after therapy.6 Magnetic resonance imaging has also been used to determine the depth and extent of soft-tissue involvement in acute evaluation of limb swelling, a common presentation of new-onset GVHD-related fasciitis.7 However, there is limited experience using MR imaging in sclerotic-type cGVHD.7,8 In this study, we examined a cross-sectional cohort of patients with sclerotic-type cGVHD using MR imaging to determine the value of this imaging modality for the identification of cutaneous and soft-tissue abnormalities.
METHODS
Sixty-two consecutive adult and pediatric patients with a history of cGVHD involving 1 or more organ systems following allogeneic hematopoietic stem cell transplantation were evaluated by an interdisciplinary team (S.Z.P., S.M., M.L.T., and E.W.C.) at the National Institutes of Health (NIH) Clinical Center, Bethesda, Maryland, as a part of an observational cross-sectional protocol studying the natural history of cGVHD (clinicaltrials.gov identifier: NCT00331968). Clinical evidence of sclerotic-type cGVHD was determined by the presence of overt skin thickening, rippling or nodularity of subcutaneous tissues with deep palpation, groove sign on physical examination, or range-of-motion limitations. Every adult patient determined as having sclerotic involvement was invited to undergo an MR imaging procedure.
After intravenous administration of a gadolinium chelate (Magnevist; Bayer HealthCare Pharmaceuticals Inc, Wayne, New Jersey), magnetic resonance imaging was performed at 1.5 T and 3 T and included T1-weighted spin echo, STIR, 3-dimensional gradient recalled echo at out-of-phase echo time, and fat-suppressed 3-dimensional gradient recalled echo at inphase echo time. Structures analyzed on MR imaging included skin, subcutaneous septae and superficial fascia, deep fascia, epimysium, and muscle. The presence of edema was based on hyperintensity depicted by STIR imaging. Thickening of fascia and septae was assessed on T1-weighted spin echo and 3-dimensional gradient recalled echo out-of-phase imaging.
A 3-point scoring system (0 for negative, 1 for equivocal, or 2 for abnormal) was used to grade the degree of MR imaging abnormalities at the following 5 anatomical levels: skin, subcutaneous septae, deep fascia, epimysium, and muscle. Skin, subcutaneous septae, and deep fascia were each assessed for thickening, edema (STIR), and enhancement (gadolinium); epimysium was assessed for edema and enhancement; and muscle was assessed for edema. The highest score for thickening, edema, or enhancement at each anatomical level was used in the composite score for that site and in calculation of the overall MR imaging score. A maximum score of 10 indicated definite abnormalities at all 5 tissue levels.
The Institutional Review Board of the National Cancer Institute approved the research protocol. All subjects gave informed consent in accord with the Declaration of Helsinki.
RESULT
Sixty-two consecutive patients with cGVHD were evaluated. Forty-four patients (71%) had clinical evidence of cutaneous cGVHD, and 28 patients (45%) had diagnostic evidence of sclerotic involvement based on physical examination findings according to National Institutes of Health consensus criteria.1 Fifteen patients agreed to undergo research MR imaging (Table 1 and Table 2). The mean time from allogeneic hematopoietic stem cell transplantation to first sclerotic symptoms was 19.4 months (median, 17 months). Magnetic resonance imaging was performed a mean of 20.1 months (median, 17 months) after the development of sclerosis. Abnormality of the fat septae was the most frequently identified finding (13 of 15 patients [87%]). Twelve of fifteen patients (80%) had similar abnormalities of the deep fascia. Epimysial edema or enhancement was detected in 9 of 15 patients (60%), and muscle inflammation was observed in 3 of 15 patients (20%). Skin sclerosis was identifiable by MR imaging in 7 of 15 patients (47%). Figure 1 and Figure 2 show clinical and magnetic resonance images of abnormalities in 2 patients with cGVHD-related skin sclerosis and fasciitis. A mean total MR imaging score of 5.27 was calculated, suggesting that in most patients with sclerotic disease soft-tissue abnormalities are present in multiple tissue planes. In general, STIR imaging was more sensitive than contrast medium–enhanced MR imaging for detection of soft-tissue abnormalities. Three patients underwent second MR assessments several months later. In each patient, the MR images remained stable over time in concordance with physical examination findings.
Table 1.
Characteristics of 15 Patients With Sclerotic-Type Chronic Graft-vs-Host Disease (cGVHD)
| Patient No./Sex/Age, y | Primary Diagnosis | Time From Stem Cell Transplantation, moa | cGVHD |
|
|---|---|---|---|---|
| Onset | Other Organ Involvement at the Time of Evaluation | |||
| 1/F/40 | Non-Hodgkin lymphoma | 33 | Progressive | Ophthalmologic, GI, hepatic, J/F, oral, and vulvovaginal |
| 2/F/39 | AML | 16 | Quiescent | Ophthalmologic, GI, hepatic, J/F, oral, pulmonary, and vulvovaginal |
| 3/M/40 | AML | 53 | Progressive | Ophthalmologic, J/F, oral, and pulmonary |
| 4/F/42 | CML | 39 | De novo | Ophthalmologic, J/F, oral, and pulmonary |
| 5/M/56 | MM | 37 | De novo | J/F and oral |
| 6/M/62 | MM | 105 | Progressive | J/F and oral |
| 7/F/38 | AML | 29 | Progressive | Ophthalmologic, hepatic, J/F, oral, pulmonary, and vulvovaginal |
| 8/M/57 | Myelodysplastic syndrome | 45 | Progressive | Ophthalmologic, J/F, and oral |
| 9/M/51 | CML | 21 | De novo | Hepatic, J/F, and oral |
| 10/M/45 | T-cell lymphoblastic leukemia | 26 | De novo | Ophthalmologic, hepatic, J/F, and oral |
| 11/M/40 | AML | 67 | De novo | Ophthalmologic, J/F, and oral |
| 12/M/32 | T-cell lymphoma | 22 | De novo | GI, hepatic, J/F, and oral |
| 13/M/57 | Chronic lymphocytic leukemia | 52 | Progressive | J/F |
| 14/F/20 | Acute lymphoblastic leukemia | 26 | De novo | Ophthalmologic, GI, hepatic, J/F, oral, and pulmonary |
| 15/M/60 | Myeloid metaplasia with myelofibrosis | 21 | Progressive | Ophthalmologic, hepatic, and oral |
Abbreviations: AML, acute myelogenous lymphoma; CML, chronic myelogenous leukemia; GI, gastrointestinal; J/F, joint and fascia (physiatry assessment); MM, multiple myeloma.
Interval between stem cell transplantation and evaluation at the National Institutes of Health.
Table 2.
Magnetic Resonance (MR) Imaging Findings
| Total Body Surface Area With cGVHD, % |
MR Imaging Scorea |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Patient No. | Erythema Type | Sclerosis Type | Duration of Sclerosis, mo/Subtype | MR Imaging Location | Skin | Septa | Deep Fascia | Epimysium | Muscle | Total |
| 1 | 0 | 40.0 | 26/Subcutaneous fibrosis/fasciitis | Bilateral thighb | 0 | 2 | 2 | 2 | 2 | 8 |
| 2 | 9.9 | 62.1 | 4/Subcutaneous fibrosis/fasciitis, dermal | Bilateral thigh | 0 | 2 | 2 | 2 | 2 | 8 |
| 3 | 7.7 | 48.6 | 21/Subcutaneous fibrosis/fasciitis, dermal | Bilateral thigh | 2 | 2 | 2 | 2 | 0 | 8 |
| 4 | 0 | 1.4 | 17/Dermal, lichen sclerosus–like | Right arm | 0 | 0 | 0 | 0 | 0 | 0 |
| 5 | 3.4 | 25.6 | 20/Subcutaneous fibrosis/fasciitis, dermal, lichen sclerosus–like | Bilateral thigh | 2 | 2 | 2 | 2 | 0 | 8 |
| 6 | 1.8 | 39.6 | 90/Subcutaneous fibrosis/fasciitis, dermal | Bilateral thigh | 2 | 2 | 1 | 0 | 0 | 5 |
| 7 | 0 | 25.6 | 18/Subcutaneous fibrosis/fasciitis | Right elbow | 0 | 1 | 2 | 2 | 2 | 7 |
| 8 | 0 | 50.2 | 8/Subcutaneous fibrosis/fasciitis, dermal | Bilateral thigh | 0 | 1 | 2 | 1 | 0 | 4 |
| 9 | 0 | 62.3 | 6/Subcutaneous fibrosis/fasciitis, dermal | Bilateral thigh | 1 | 2 | 2 | 1 | 0 | 6 |
| 10 | 1.7 | 64.0 | 7/Subcutaneous fibrosis/fasciitis, lichen sclerosus–like | Bilateral thigh | 1 | 2 | 2 | 2 | 0 | 7 |
| 11 | 0 | 7.4 | 25/Subcutaneous fibrosis/fasciitis, dermal | Right ankle | 0 | 1 | 2 | 0 | 0 | 3 |
| 12 | 0 | 40.0 | 3/Subcutaneous fibrosis/fasciitis | Right forearm | 0 | 0 | 2 | 2 | 0 | 4 |
| 13 | 0 | 78.0 | 39/Subcutaneous fibrosis/fasciitis, dermal | Bilateral thighb | 2 | 2 | 2 | 0 | 0 | 6 |
| 14 | 0 | 15.8 | 16/Dermal, subcutaneous fibrosis/fasciitis | Abdomen | 0 | 1 | 0 | 0 | 0 | 1 |
| 15 | 0 | 20.0 | 1/Dermal, subcutaneous fibrosis/fasciitis, lichen sclerosus–like | Abdomenc | 2 | 2 | 0 | 0 | 0 | 4 |
Abbreviation: cGVHD, chronic graft-vs-host disease.
Score of 0 indicates no change; 1, equivocal; and 2, abnormal.
Using 3 T with gadolinium contrast medium.
Using 3 T without gadolinium contrast medium.
Figure 1.
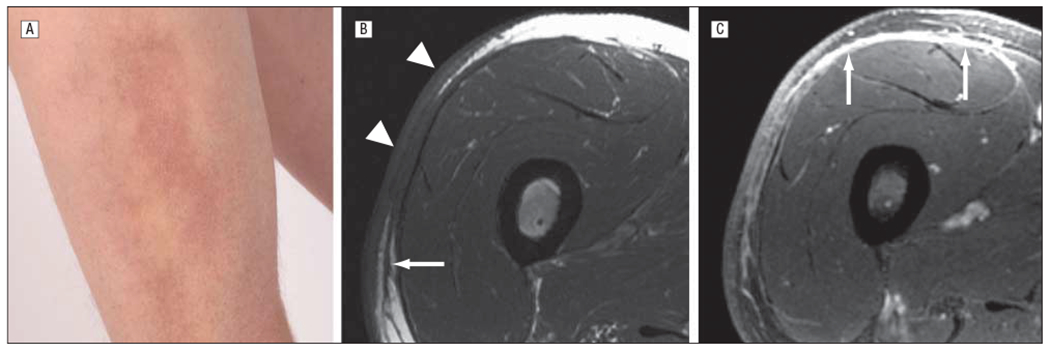
Fasciitis with overlying skin involvement in a 56-year-old man 37 months after allogeneic hematopoietic stem cell transplantation for multiple myeloma (patient 5 in Table 1 and Table 2). A, Erythema and induration on the anterolateral aspect of the right thigh. B, Axial T1-weighted spin-echo image demonstrates thickening of the skin, with intervening fat dropout (arrowheads) overlying a region of narrowing and sclerosis of the subcutis. There is also thickening of the adjacent superficial fascia (arrow). C, Axial 3-dimensional gradient recalled echo image after administration of contrast medium demonstrates mild enhancement of the skin and subcutis in the region of sclerosis, with more prominent and extensive enhancement along the deep fascia (arrows). Magnetic resonance imaging total score of 8 (score of 2 each for skin, septa, deep fascia, and epimysium).
Figure 2.
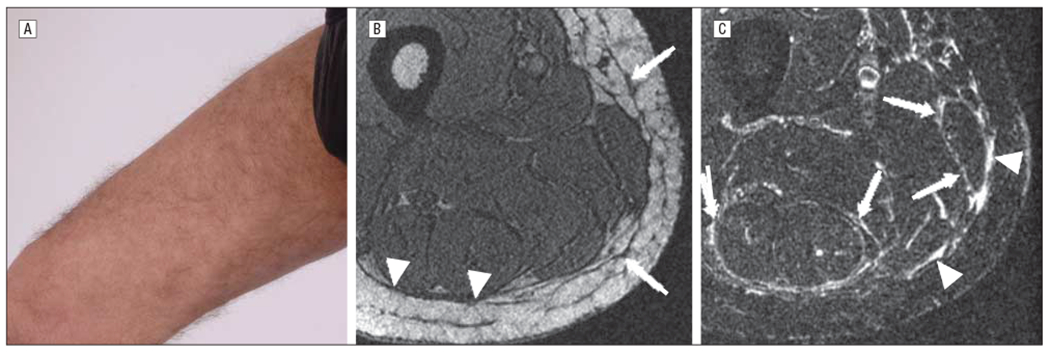
Fasciitis without overlying superficial skin sclerosis in a 45-year-old man 26 months after allogeneic hematopoietic stem cell transplantation for T-cell lymphoblastic leukemia (patient 10 in Table 1 and Table 2). A, A subtle rippled appearance is present on the right medial thigh. A firm nodular texture was detected on deep palpation. B, Axial 3-dimensional gradient recalled echo (out-of-phase echo time) image demonstrates mild thickening of the subcutaneous septa (arrows) and minimal thickening of the deep fascia (arrowheads). C, Axial short tau inversion recovery image demonstrates widespread linear increased signal along the deep fascia (arrowheads) and along the epimysium of various muscles (arrows). Magnetic resonance imaging total score of 7 (score of 1 for skin and 2 each for septa, deep fascia, and epimysium).
COMMENT
Imaging in 14 of 15 patients was consistent with the clinical impression, suggesting that careful clinical examination by a trained clinician may detect subcutaneous involvement and fasciitis. One patient (patient 4 in Table 1 and Table 2) with negative findings on MR images described a history of gradual improvement of tightness in the upper extremities. The patient had minimal superficial sclerosis of the upper extremities; therefore, the negative findings were not unexpected. In several other patients, MR imaging detected deeper and more extensive involvement than could be appreciated by physical examination alone. In 3 patients, muscle involvement was detected in the absence of an elevation in the serum creatine kinase level, suggesting that MR imaging may be particularly helpful for the detection of fascial and muscle inflammation.
Although most patients in this study agreed to provide punch skin biopsy specimens, fascial biopsy specimens were not obtained, and MR imaging abnormalities require clinical correlation. In addition, because MR imaging was performed only in patients with clinical evidence of sclerotic disease, the sensitivity of MR imaging for subclinical subcutaneous or fascial disease in patients with cGVHD is unknown. The high prevalence of sclerotic-type cGVHD among our cohort (28 of 45 patients [45%]) is not representative of cutaneous cGVHD in general, as the referral population for this study consisted primarily of severely affected patients with multiorgan involvement.
We believe that MR imaging is a useful adjunct to careful clinical assessment to evaluate soft-tissue fibrosis and may be considered in the clinical setting to confirm the presence of suspected muscle and fascial abnormalities before the development of extensive range-of-motion limitations. However, consideration must also be given to the cost of MR imaging and to the potential risk of nephrogenic systemic fibrosis associated with exposure to gadolinium contrast medium. Although we did not observe the development of nephrogenic systemic fibrosis in our cohort, patients with cGVHD may be at higher risk for nephrogenic systemic fibrosis owing to renal dysfunction from previous chemotherapy exposure or ongoing immunosuppressive therapy, particularly treatment with calcineurin inhibitors.
Better assessment tools are needed to differentiate disease activity vs damage and to evaluate treatment efficacy in patients with sclerotic-type cGVHD.9 Skin scoring systems based on body surface area estimates and degree of ability to pinchability or ability to move the skin are inadequate for precise monitoring of subcutaneous and fascial disease.10,11 Surrogate markers of skin fibrosis such as joint range of motion are helpful if sclerosis overlies a joint, but evaluation requires a trained professional to perform the assessment in a standardized and reproducible manner,9 and findings may be affected by other factors, including pain, deconditioning, and unrelated joint disease. Therefore, MR imaging may be useful in the research setting for serial long-term monitoring of patients with sclerotic soft-tissue disease. Optimization of MR imaging as a response assessment tool will require correlation of imaging, histologic findings, and meaningful clinical change over time.
Funding/Support:
This study was supported in part by the Intramural Research Program of the National Institutes of Health, Center for Cancer Research, National Cancer Institute. Dr Clark was jointly supported by the National Institutes of Health/Pfizer Clinical Research Training Program.
Footnotes
Financial Disclosure: None reported.
REFERENCES
- 1.Filipovich AH, Weisdorf D, Pavletic S, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease, I: Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant. 2005;11(12):945–956. [DOI] [PubMed] [Google Scholar]
- 2.Hymes SR, Turner ML, Champlin RE, Couriel DR. Cutaneous manifestations of chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2006;12(11):1101–1113. [DOI] [PubMed] [Google Scholar]
- 3.Schaffer JV, McNiff JM, Seropian S, Cooper DL, Bolognia JL. Lichen sclerosus and eosinophilic fasciitis as manifestations of chronic graft-versus-host disease: expanding the sclerodermoid spectrum. J Am Acad Dermatol. 2005;53(4):591–601. [DOI] [PubMed] [Google Scholar]
- 4.Häusermann P, Walter RB, Halter J, et al. Cutaneous graft-versus-host disease: a guide for the dermatologist. Dermatology. 2008;216(4):287–304. [DOI] [PubMed] [Google Scholar]
- 5.Kimball AB, Summers RM, Turner M, et al. Magnetic resonance imaging detection of occult skin and subcutaneous abnormalities in juvenile dermatomyositis: implications for diagnosis and therapy. Arthritis Rheum. 2000;43(8):1866–1873. [DOI] [PubMed] [Google Scholar]
- 6.Baumann F, Brühlmann P, Andreisek G, Michel BA, Marincek B, Weishaupt D. MRI for diagnosis and monitoring of patients with eosinophilic fasciitis. AJR Am J Roentgenol. 2005;184(1):169–174. [DOI] [PubMed] [Google Scholar]
- 7.Révelon G, Rahmouni A, Jazaerli N, et al. Acute swelling of the limbs: magnetic resonance pictorial review of fascial and muscle signal changes. Eur J Radiol. 1999;30(1):11–21. [DOI] [PubMed] [Google Scholar]
- 8.Dumford K, Anderson JC. CT and MRI findings in sclerodermatous chronic graft vs. host disease. Clin Imaging. 2001;25(2):138–140. [DOI] [PubMed] [Google Scholar]
- 9.Pavletic SZ, Martin P, Lee SJ, et al. ; Response Criteria Working Group. Measuring therapeutic response in chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease, IV: Response Criteria Working Group report. Biol Blood Marrow Transplant. 2006;12(3):252–266. [DOI] [PubMed] [Google Scholar]
- 10.Jacobsohn DA, Chen AR, Zahurak M, et al. Phase II study of pentostatin in patients with corticosteroid-refractory chronic graft-versus-host disease. J Clin Oncol. 2007;25(27):4255–4261. [DOI] [PubMed] [Google Scholar]
- 11.Greinix HT, Pohlreich D, Maalouf J, et al. A single-center pilot validation study of a new chronic GVHD skin scoring system. Biol Blood Marrow Transplant. 2007;13(6):715–723. [DOI] [PubMed] [Google Scholar]


