Abstract
Background
The study aimed to compare the predictive values of the thrombolysis in myocardial infarction (TIMI); History, Electrocardiography, Age, Risk factors and Troponin (HEART) and Global Registry in Acute Coronary Events (GRACE) scoring systems for major adverse cardiovascular events (MACEs) in acute chest pain (ACP) patients admitted to the emergency department (ED).
Methods
We systematically searched PubMed, Embase and the Cochrane Library from their inception to June 2020; we compared the following parameters: sensitivity, specificity, positive and negative likelihood ratios (PLR and NLR), diagnostic OR (DOR) and area under the receiver operating characteristic curves (AUC).
Results
The pooled sensitivity and specificity for TIMI, HEART and GRACE were 0.95 and 0.36, 0.96 and 0.50, and 0.78 and 0.56, respectively. The pooled PLR and NLR for TIMI, HEART and GRACE were 1.49 and 0.13, 1.94 and 0.08, and 1.77 and 0.40, respectively. The pooled DOR for TIMI, HEART and GRACE was 9.18, 17.92 and 4.00, respectively. The AUC for TIMI, HEART and GRACE was 0.80, 0.80 and 0.70, respectively. Finally, the results of indirect comparison suggested the superiority of values of TIMI and HEART to those of GRACE for predicting MACEs, while there were no significant differences between TIMI and HEART for predicting MACEs.
Conclusions
TIMI and HEART were superior to GRACE for predicting MACE risk in ACP patients admitted to the ED.
Keywords: adult cardiology, accident & emergency medicine, coronary heart disease
Strengths and limitations of this study.
The analysis is based on prospective studies and used consistent cut-off values.
The pooled results were stable owing to a large sample size.
The indirect comparisons among thrombolysis in myocardial infarction, History, Electrocardiography, Age, Risk factors and Troponin and Global Registry in Acute Coronary Events scoring systems were provided.
The analysis is based on crude data; the predictive values could be affected by covariates.
Substantial heterogeneity was not fully explained.
Introduction
Acute chest pain (ACP) is a common symptom accounting for a significant proportion of attendance and burden in the emergency department (ED).1 ACP patients require effective risk stratification to ensure timely initiation of proper treatment in high-risk cases to achieve better prognoses. The early identification of cardiovascular disease (CVD) in ACP patients is important although CVD accounts for only a small proportion of ACP patients with ECGs on presentation.2–4 Moreover, patients diagnosed with acute coronary syndrome (ACS) should remain hospitalised, whereas non-ACS patients are unnecessarily admitted to hospitals due to the heavy burden on resource constraints.5 Therefore, accurate risk stratification for ACP patients is essential to improve hospital efficacy by administering timely interventions to high-risk patients, avoiding unnecessary tests and minimising admissions for low-risk patients.
Currently, the thrombolysis in myocardial infarction (TIMI); History, ECG, Age, Risk factors and Troponin (HEART) and Global Registry in Acute Coronary Events (GRACE) scores are widely used for the risk stratification of ACP patients; however, the predictive values using these methods on major adverse cardiovascular events (MACEs) have not been elucidated. The TIMI scoring system was established in 2000 for evaluating patients with unstable angina or non-ST-segment elevation myocardial infarction.6–10 The HEART score, developed in 2008, aims to improve the accuracy of diagnosing ACS for patients with undifferentiated chest pain.11 The GRACE score was developed in 2001 for adults with symptoms of ACS; it comprises the following factors: age, vital signs, kidney function, ECG and troponin levels.12 13 However, the predictive values of risk stratification measured by the TIMI, HEART and GRACE scoring systems on MACEs have not been fully compared. Therefore, this study was conducted based on prospective cohort studies to provide comprehensive results regarding the risk stratification assessed by the TIMI, HEART and GRACE scoring systems on MACEs in ACP patients admitted to the ED. Furthermore, the predictive values of risk stratification assessed by the TIMI, HEART and GRACE scoring systems on MACEs were compared through an indirect analytic approach.
Methods
Data sources, search strategy and selection criteria
This study was conducted and reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Statement issued in 2009.14 Any prospective cohort studies investigating the predictive value of TIMI, HEART and GRACE on MACEs in ACP patients were eligible for inclusion in this study. No restrictions were placed on publishing language and status. The electronic databases of PubMed, Embase and the Cochrane library were systematically searched for studies from their inception up to June 2020, and the search strategy was performed using the following terms with Medical Subject Heading and free words: (“TIMI” or “HEART” or “GRACE”) and “emergency department” and “chest pain” and (“prospective” or “cohort”). The search strategy details are summarised in online supplemental file. The reference lists of retrieved studies were also searched manually to find new eligible studies.
bmjopen-2020-048356supp001.pdf (1.1MB, pdf)
Two authors independently performed the literature search and study selection; any conflicts were resolved through group discussion until a consensus was reached. A study was included if they met the following inclusion criteria: (1) study design: the study had a prospective design; (2) patients: ACP patients admitted to the ED; (3) risk stratifying tools: TIMI, HEART or GRACE; (4) outcomes: the study had to report the incidence of MACEs and provided clear definitions of MACEs; (5) data abstracted: true and false positives or negatives, or data could transform into the above information must be reported and (6) cut-off value: the cut-off value of TIMI and HEART was 0–3, and the cut-off value of GRACE was 55–110. Retrospective studies were excluded due to various confounding factors. Additionally, studies that used other cut-off values were excluded.
Data collection and quality assessment
Two authors independently abstracted data items and assessed the quality of the included studies, and any disagreement was settled by an additional author reviewing the original article. The collected information from retrieved studies including the first author’s name, publication year, country, sample size, age at baseline, percentage of males, risk stratifying tools, patients’ status, MACE definition, follow-up duration and true and false positives/negatives. Study quality was assessed using the Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2), which is based on patient selection, index test, reference standard, risk of bias and concerns about applicability.15
Statistical analysis
The pooled sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR) and the area under the receiver operating characteristic curves (AUC) for the risk stratification by TIMI, HEART and GRACE on MACEs were calculated using a bivariate generalised linear mixed model,16 17 while the pooled diagnostic OR (DOR) was calculated using a random-effects model.18 The I2 and Q statistic were used for assessing the heterogeneity across included studies, and p<0.10 was considered as significant heterogeneity.19 20 The robustness of pooled results was also assessed by sensitivity analyses, which were also performed for studies using all three scoring systems and the endpoint MACE.21 TIMI-based, HEART-based and GRACE-based risk stratification were assessed using an indirect comparison analysis, and the ratios among these scoring systems were calculated.22 Furthermore, subgroup analyses for the predictive values of TIMI, HEART and GRACE on subsequent MACE risk were also estimated based on country, mean age, percentage of males, follow-up duration and study quality. The funnel plots and Deeks’ asymmetry tests were used to assess publication bias.23 24 The inspection level for pooled diagnostic parameters was two sided, and p<0.05 was considered statistically significant. All statistical analyses were performed using Stata software (V.10.0; Stata).
Patient and public involvement
There was no patient or public involvement in the design or conduct of this study.
Results
Literature search
The details regarding the literature search and study selection of eligible studies are presented in figure 1. A total of 2794 articles were identified through the electronic search from PubMed, Embase and the Cochrane library, and 1981 were excluded because of term duplications. Subsequently, the remaining 813 studies were selected through title and abstract review; 732 were excluded based on irrelevance. A total of 81 full texts were retrieved for further evaluation, and 48 studies were excluded due to the following reasons: used other cut-off values (n=21), retrospective study design (n=14) and insufficient data (n=13). An additional 135 potential studies identified from the reference lists of retrieved studies were excluded because of duplication with the electronic search. Subsequently, 33 prospective cohort studies that recruited 40 262 ACP patients were selected for final quantitative meta-analysis.13 25–56
Figure 1.
Flow diagram of the literature search and study selection process.
Study characteristics
The baseline characteristics of included studies are summarised in table 1. The retrieved studies were published from 2005 to 2020, and 255–4333 ACP patients were included in each study. Nine studies were conducted in Eastern countries, and the remaining 24 studies were conducted in Western countries. The mean age of enrolled patients ranged from 48.0 to 69.0 years, and the percentage of males ranged from 40.0% to 68.8%. Risk stratification by the TIMI score was available in 25 studies published between 2005 and 2020, 16 studies used the HEART score and were published between 2013 and 2020 and and 16 studies employed GRACE and were published between 2007 and 2020.55 The definition of MACEs across included studies contained all-cause death, cardiac death, myocardial infarction, revascularisation, cardiac arrest, cardiogenic shock, unstable angina, ACS, percutaneous coronary intervention, coronary artery bypass grafting, coronary angiography revealing procedurally correctable stenosis managed conservatively, ventricular arrhythmia needing intervention, high-degree atrioventricular block needing intervention and life-threatening arrhythmias requiring emergency intervention. The study quality of the included studies was assessed by QUADAS-2 (figure 2).
Table 1.
Baseline characteristics of studies included in the systematic review and meta-analysis
| Study | Country | Sample size | Mean age (years) | Percentage male (%) | Risk stratifying tools | Patients’ status | MACE definition | Follow-up |
| Tong 2005 25 | USA | 957 | 60.0 | 52.0 | TIMI | Chest pain and a nondiagnostic ECG | Death and MI | 30 days |
| Sanchis 200526 | Spain | 646 | 64.0 | 65.8 | TIMI | Acute chest pain | Death, MI or urgent revascularisation | 14 days |
| Pollack 200627 | USA | 3929 | 51.6 | 40.0 | TIMI | Chest pain in the ED | Death, acute MI and revascularisation | 30 days |
| Pelliccia 200628 | Italy | 4333 | 58.4 | 68.8 | TIMI | Acute chest pain | MI | In-hospital |
| Lyon 200729 | UK | 954 | 60.0 | 62.0 | TIMI, GRACE | Undifferentiated chest pain | MI, cardiac arrest, revascularisation, unstable angina with myocardial damage and death | 30 days |
| Ramsay 200713 | UK | 347 | 65.2 | 62.3 | TIMI, GRACE | Suspected cardiac pain | Death, non-fatal MI and emergency revascularisation | 3.0 months |
| Body 200930 | UK | 796 | 58.9 | 60.4 | TIMI | Chest pain in the ED | Death, acute MI or urgent coronary revascularisation | 30 days |
| Campbell 200931 | USA | 3169 | 53.6 | 45.0 | TIMI | Chest pain in the ED | Death, MI or revascularisation | 30 days |
| Hess 201032 | Canada | 1017 | 59.3 | 60.6 | TIMI | ED patients with chest pain and possible ACS | Acute MI, revascularisation or death | 30 days |
| Stracke 201033 | Germany | 1014 | 66.0 | 55.0 | GRACE | Chest pain in the ED | Death | In-hospital |
| van der Zee 201134 | The Netherlands | 524 | 57.7 | 60.5 | GRACE | Chest pain in the ED | Death | 9.4 years |
| Graham 201335 | China | 315 | 69.0 | 54.9 | TIMI | Chest pain in the ED | Death, MI, troponin positive ACS and PCI |
30 days |
| Holly 201336 | USA | 552 | 54.1 | 46.0 | TIMI | Chest pain in the ED | MI, revascularisation or death | 30 days |
| Backus 201337 | The Netherlands | 2388 | 60.6 | 57.5 | HEART | Chest pain in the ED | Acute MI, PCI, CABG, coronary angiography revealing procedurally correctable stenosis managed conservatively and death | 6 weeks |
| Cullen 201338 | Australia | 948 | 54.0 | 59.9 | TIMI, and GRACE | Chest pain in the ED | Cardiac death, acute MI and unstable angina | 30 days |
| Graham 201439 | China | 925 | 68.0 | 51.7 | TIMI | Chest pain in the ED | Death, readmission with MI, ACS not diagnosed at initial ED presentation and coronary revascularisation | 30 days |
| Visser 201540 | The Netherlands | 255 | 64.0 | 56.0 | HEART | Chest pain in the ED | MI, or PCI, or CABG, or coronary angiography revealing significant stenosis or death | 6 weeks |
| Boubaker 201541 | Tunisia | 3125 | 57.7 | 58.3 | TIMI, GRACE | Chest pain in the ED | All-cause mortality, ACS and coronary non-ED planned revascularisation | 30 days |
| Wang 201642 | China | 986 | 54.0 | 55.0 | TIMI, HEART, GRACE | Chest pain in the ED | Death, MI and/or the need for revascularisation by CABG or PCI | 6 months |
| Chen 201643 | China | 833 | 65.1 | 55.3 | TIMI, HEART, GRACE | Chest pain in the ED | Coronary revascularisation, ventricular arrhythmia needing intervention and high-degree atrioventricular block needing intervention | 30 days |
| Sakamoto 201644 | Singapore | 604 | 60.8 | 69.2 | TIMI, HEART, GRACE | Chest pain in the ED | Death, acute MI, PCI, CABG | 30 days |
| Leung 201745 | China | 602 | 66.0 | 48.8 | TIMI, HEART | Chest pain in the ED | Death, cardiac arrest, MI and cardiogenic shock | 30 days |
| Poldervaart 201746 | The Netherlands | 1748 | 62.0 | 54.0 | TIMI, HEART, GRACE | Chest pain in the ED | UA, MI, PCI, CABG, stenosis managed conservatively and death | 3.0 months |
| McCord 201747 | Europe, Australia and the USA | 661 | 58.3 | 58.2 | HEART | Chest pain in the ED | Death or acute MI | 30 days |
| Reaney 201848 | UK | 1000 | 62.5 | 57.6 | TIMI, HEART, GRACE | Chest pain in the ED | Acute MI, PCI, CABG, cardiac death, cardiogenic shock and life-threatening arrhythmias requiring emergency intervention. | 30 days |
| Greenslade 201849 | Australia | 1760 | 60.4 | 59.3 | TIMI | Chest pain in the ED | Cardiac death, cardiac arrest, cardiogenic shock, acute MI, UA, emergency or urgent revascularisation, high-level atrioventricular block and ventricular arrhythmias | 30 days |
| Moumneh 201850 | France | 641 | 53.3 | 53.4 | HEART | Non-traumatic chest pain patients | MI, coronary angioplasty, coronary bypass and sudden unexplained death | 6 weeks |
| Ishak 201851 | The Netherlands | 1127 | 63.8 | 57.7 | HEART | Chest pain in the ED | Acute MI, PCI, CABG or death | 30 days |
| Wong 201852 | China | 1081 | 48.0 | 52.3 | TIMI, HEART, GRACE | Undifferentiated chest pain | Acute MI, PCI, CABG and death | 30 days |
| Al-Zaiti 201953 | USA | 750 | 59.0 | 58.0 | TIMI, HEART, GRACE | Chest pain in the ED | Resuscitated or unresuscitated sudden cardiac arrest, all-cause death, postdischarge reinfarction requiring cardiac revascularisation | 30 days |
| Huang 202054 | China | 509 | 59.8 | 53.4 | HEART, GRACE | Chest pain in the ED | Acute MI, PCI, CABG, cardiac death, cardiogenic shock or life-threatening arrhythmias requiring emergency intervention or resulting in mortality | 30 days |
| Torralba 202055 | Colombia | 519 | 64.3 | 56.1 | TIMI, HEART, GRACE | Chest pain in the ED | Death from any cause, MI and surgical or percutaneous myocardial revascularisation | 30 days |
| Shin 202056 | Korea | 1247 | 62.0 | 60.8 | TIMI, HEART, GRACE | Chest pain in the ED | Acute MI, PCI, CABG or death from cardiac causes | 30 days |
ACS, acute coronary syndrome; CABG, coronary artery bypass grafting; ED, emergency department; GRACE, Global Registry in Acute Coronary Events; HEART, History, Electrocardiography, Age, Risk factors, and Troponin; MI, myocardial infarction; PCI, percutaneous coronary intervention; TIMI, thrombolysis in myocardial infarction; UA, unstable angina.
Figure 2.
QUADAS-2 scoring of included studies. QUADAS-2, Quality Assessment of Diagnostic Accuracy Studies 2.
Thrombolysis in myocardial infarction
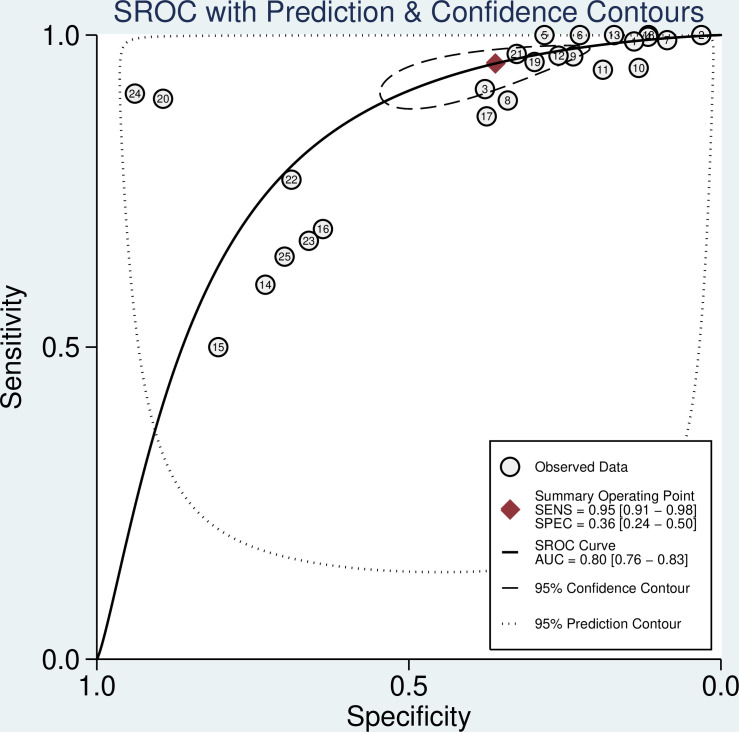
The predictive value of risk stratification by the TIMI score on MACEs in ACP patients was available in 25 studies. The pooled sensitivity and specificity of the TIMI score for predicting MACEs were 0.95 (95% CI: 0.91 to 0.98; I2=98.10%) and 0.36 (95% CI: 0.24 to 0.50; I2=99.64%), respectively (online supplemental file). Moreover, the pooled PLR and NLR of the TIMI score for predicting MACEs were 1.49 (95% CI: 1.25 to 1.79; I2=99.21%) and 0.13 (95% CI: 0.07 to 0.21; I2=95.38%), respectively (online supplemental file). The pooled DOR of the TIMI score for predicting MACEs was 9.18 (95% CI: 6.22 to 13.55; p<0.001) with significant heterogeneity across the included studies (I2=87.6%; p<0.001) (online supplemental file). The AUC of the TIMI score for predicting MACEs was 0.80 (95% CI: 0.76 to 0.83; figure 3). No significant publication bias for the TIMI score was detected (p=0.17; online supplemental file).
Figure 3.
The summary receiver operating characteristic curve (SROC) of risk stratification assessed by the TIMI score. AUC, area under the curve; TIMI, thrombolysis in myocardial infarction.
History, electrocardiography, age, risk factors and troponin
The predictive value of risk stratification by the HEART score on MACEs in ACP patients was available in 16 studies. The pooled sensitivity and specificity of the HEART score for predicting MACEs were 0.96 (95% CI: 0.91 to 0.98; I2=94.87%) and 0.50 (95% CI: 0.41 to 0.60; I2=98.84%), respectively (online supplemental file). The pooled PLR and NLR of the HEART score for predicting MACEs were 1.94 (95% CI: 1.61 to 2.35; I2=98.01%) and 0.08 (95% CI: 0.03 to 0.17; I2 =94.65%), respectively (online supplemental file). The pooled DOR for the HEART score was 17.92 (95% CI: 9.40 to 34.18; p<0.001) with significant heterogeneity across the included studies (I2=88.9%; p<0.001) (online supplemental file). The AUC of the HEART score for predicting MACEs was 0.80 (95% CI: 0.77 to 0.84; figure 4). There was no significant publication bias for the HEART score (p=0.98; online supplemental file).
Figure 4.
The summary receiver operating characteristic curve (SROC) of risk stratification assessed by the HEART score. AUC, area under the curve; HEART, History, Electrocardiography, Age, Risk factors and Troponin.
Global Registry in Acute Coronary Events
The predictive value of risk stratification by the GRACE score on MACEs in ACP patients was available in 16 studies. The pooled sensitivity and specificity of the GRACE score for predicting MACEs were 0.78 (95% CI: 0.64 to 0.87; I2=96.78%) and 0.56 (95% CI: 0.46 to 0.66; I2=99.39%), respectively (online supplemental file). The pooled PLR and NLR of the GRACE score for predicting MACEs were 1.77 (95% CI: 1.51 to 2.08; I2 =96.34%) and 0.12 (95% CI: 0.06 to 0.26; I2 =94.07%), respectively (online supplemental file). The DOR of the GRACE score for predicting MACEs was 4.00 (95% CI: 2.78 to 5.74; p<0.001) with significant heterogeneity across the included studies (I2=88.7%; p<0.001) (online supplemental file). The AUC of the GRACE score for predicting MACEs was 0.70 (95% CI: 0.66 to 0.74; figure 5). No significant publication bias for the GRACE score was observed (p=0.36; online supplemental file).
Figure 5.
The summary receiver operating characteristic curve (SROC) of risk stratification assessed by the grace score. AUC, area under the curve.
Indirect comparisons
Indirect comparisons of the diagnostic parameters (sensitivity, specificity, PLR, NLR, DOR and AUC) among the TIMI, HEART and GRACE scoring systems for predicting MACEs are summarised in table 2. First, the sensitivity of TIMI (ratio: 1.22; 95% CI: 1.04 to 1.43) and HEART (ratio: 1.23; 95% CI: 1.05 to 1.44) was significantly higher than that of GRACE for predicting MACEs. Second, the specificity of TIMI was significantly lower than that of GRACE for predicting MACEs (ratio: 0.64; 95% CI: 0.43 to 0.97). Third, the PLR of TIMI was lower than that of HEART for predicting MACEs (ratio: 0.77; 95% CI: 0.59 to 1.00). Fourth, TIMI (ratio: 0.32; 95% CI: 0.17 to 0.64) and HEART (ratio: 0.20; 95% CI: 0.08 to 0.52) were associated with a lower NLR than GRACE for predicting MACEs. Fifth, the DOR of TIMI (ratio: 2.29; 95% CI: 1.35 to 3.91) and HEART (ratio: 4.48; 95% CI: 2.14 to 9.39) was significantly higher than that of GRACE for predicting MACEs. Finally, the AUC of TIMI (ratio: 1.14; 95% CI: 1.06 to 1.23) and HEART (ratio: 1.14; 95% CI: 1.06 to 1.23) was significantly higher than that of GRACE for predicting MACEs.
Table 2.
Systematic comparisons of the sensitivity, specificity, PLR and NLR, DOR and the AUC of risk stratifying measured by TIMI, HEART and GRACE
| Parameters | TIMI | HEART | GRACE | TIMI versus HEART | TIMI versus GRACE | HEART versus GRACE |
| Sensitivity and 95% CI | 0.95 (0.91–0.98) | 0.96 (0.91–0.98) | 0.78 (0.64–0.87) | 0.99 (0.94–1.04) | 1.22 (1.04–1.43) | 1.23 (1.05–1.44) |
| Specificity and 95% CI | 0.36 (0.24–0.50) | 0.50 (0.41–0.60) | 0.56 (0.46–0.66) | 0.72 (0.48–1.09) | 0.64 (0.43–0.97) | 0.89 (0.69–1.16) |
| PLR and 95% CI | 1.49 (1.25–1.79) | 1.94 (1.61–2.35) | 1.77 (1.51–2.08) | 0.77 (0.59–1.00) | 0.84 (0.66–1.07) | 1.10 (0.86–1.40) |
| NLR and 95% CI | 0.13 (0.07–0.21) | 0.08 (0.03–0.17) | 0.40 (0.27–0.59) | 0.46 (0.15–1.39) | 0.32 (0.17–0.64) | 0.20 (0.08–0.52) |
| DOR and 95% CI | 9.18 (6.22–13.55) | 17.92 (9.40–34.18) | 4.00 (2.78–5.74) | 0.51 (0.24–1.09) | 2.29 (1.35–3.91) | 4.48 (2.14–9.39) |
| AUC and 95% CI | 0.80 (0.76–0.83) | 0.80 (0.77–0.84) | 0.70 (0.66–0.74) | 1.00 (0.94–1.06) | 1.14 (1.06–1.23) | 1.14 (1.06–1.23) |
AUC, the area under the receiver operating characteristic curve; DOR, diagnostic OR; GRACE, Global Registry in Acute Coronary Events; HEART, History, Electrocardiography, Age, Risk factors and Troponin; NLR, negative likelihood ratio; PLR, positive likelihood ratio; TIMI, thrombolysis in myocardial infarction.
Sensitivity and subgroup analysis
The results of sensitivity analyses found that the predictive values of TIMI, HEART and GRACE for predicting MACEs were stable and unaltered by sequential removal of one study from the overall analysis (data not shown). Sensitivity analyses were also performed after removing studies not using all three scoring systems (table 3). We noted that TIMI had lower sensitivity that that of HEART (ratio: 0.86; 95% CI: 0.75 to 0.99). Moreover, TIMI had lower NLR (ratio: 0.55; 95% CI: 0.31 to 0.99), higher DOR (ratio: 2.71; 95% CI: 1.17 to 6.24) and higher AUC (ratio: 1.19; 95% CI: 1.11 to 1.27), compared with GRACE. Furthermore, HEART had higher sensitivity (ratio: 1.32; 95% CI: 1.11 to 1.58), DOR (ratio: 4.36; 95% CI: 1.63 to 11.64) and AUC (ratio: 1.14; 95% CI: 1.06 to 1.23) and lower NLR (ratio: 0.22; 95% CI: 0.09 to 0.56) as compared with GRACE. Additionally, table 4 presents the subgroup analysis results for the predictive values of TIMI, HEART and GRACE. First, TIMI has lower specificity than HEART in the pooled studies from Western countries. Furthermore, TIMI versus HEART showed lower PLR in the pooled studies of Western countries (mean age <60.0 years). Moreover, the DOR of TIMI was lower than of HEART in the pooled studies of Eastern countries, while TIMI had a lower AUC than HEART for mean age <60.0 years. Second, TIMI with higher sensitivity than GRACE when pooled studies conducted in Eastern countries, percentage of males≥60.0%, follow-up duration ≤30.0 days and studies of moderate quality. Moreover, TIMI versus GRACE showed lower specificity if mean age <60.0 years, percentage of males ≥60.0% and follow-up duration ≤30.0 days. Furthermore, TIMI has lower PLR than GRACE if percentage of males ≥60.0%, while TIMI has lower NLR than GRACE in the pooled studies of Eastern countries, irrespective of the mean age or percentage male status, follow-up duration ≤30.0 days, and studies of moderate quality. In addition, TIMI versus GRACE showed higher DOR and AUC in most subgroups. Third, HEART versus GRACE showed higher sensitivity in the pooled studies of Eastern countries, mean age ≥60.0 years, follow-up duration ≤30.0 days and studies of moderate quality. Moreover, HEART has lower NLR than GRACE in most subgroups, except that of mean age <60.0 years. Furthermore, HEART versus GRACE showed higher DOR and AUC in most subgroups.
Table 3.
Sensitivity analysis for direct comparisons of the sensitivity, specificity, PLR and NLR, DOR, and the AUC of risk stratifying measured by TIMI, HEART and GRACE
| Parameters | TIMI | HEART | GRACE | TIMI versus HEART | TIMI versus GRACE | HEART versus GRACE |
| Sensitivity and 95% CI | 0.81 (0.70–0.89) | 0.94 (0.84–0.98) | 0.71 (0.58–0.81) | 0.86 (0.75–0.99) | 1.14 (0.94–1.39) | 1.32 (1.11–1.58) |
| Specificity and 95% CI | 0.70 (0.54–0.83) | 0.53 (0.39–0.66) | 0.59 (0.44–0.73) | 1.32 (0.94–1.86) | 1.19 (0.85–1.65) | 0.90 (0.62–1.29) |
| PLR and 95% CI | 2.72 (1.67–4.41) | 1.99 (1.51–2.63) | 1.74 (1.33–2.28) | 1.37 (0.78–2.39) | 1.56 (0.90–2.72) | 1.14 (0.78–1.68) |
| NLR and 95% CI | 0.27 (0.16–0.44) | 0.11 (0.05–0.28) | 0.49 (0.36–0.66) | 2.45 (0.90–6.66) | 0.55 (0.31–0.99) | 0.22 (0.09–0.56) |
| DOR and 95% CI | 9.45 (4.77–18.73) | 15.21 (6.45–35.88) | 3.49 (2.16–5.63) | 0.62 (0.21–1.86) | 2.71 (1.17–6.24) | 4.36 (1.63–11.64) |
| AUC and 95% CI | 0.83 (0.80–0.86) | 0.80 (0.76–0.83) | 0.70 (0.66–0.74) | 1.04 (0.98–1.10) | 1.19 (1.11–1.27) | 1.14 (1.06–1.23) |
AUC, the area under the receiver operating characteristic curve; DOR, diagnostic OR; GRACE, Global Registry in Acute Coronary Events; HEART, History, Electrocardiography, Age, Risk factors, and Troponin; NLR, negative likelihood ratio; PLR, positive likelihood ratio; TIMI, thrombolysis in myocardial infarction.
Table 4.
Subgroup analyses
| Parameters | Factors | Groups | TIMI | HEART | GRACE | TIMI versus HEART | TIMI versus GRACE | HEART versus GRACE |
| Sensitivity | Country | Western | 0.97 (0.93–0.99) | 0.97 (0.93–0.99) | 0.88 (0.72–0.95) | 1.00 (0.96–1.05) | 1.10 (0.96–1.27) | 1.10 (0.96–1.27) |
| Eastern | 0.86 (0.69–0.95) | 0.94 (0.79–0.98) | 0.60 (0.49–0.70) | 0.91 (0.75–1.11) | 1.43 (1.13–1.82) | 1.57 (1.27–1.93) | ||
| Mean age (years) | ≥60.0 | 0.97 (0.92–0.99) | 0.97 (0.94–0.99) | 0.83 (0.67–0.92) | 1.00 (0.96–1.05) | 1.17 (0.99–1.38) | 1.17 (1.00–1.37) | |
| <60.0 | 0.93 (0.81–0.97) | 0.91 (0.70–0.98) | 0.68 (0.44–0.85) | 1.02 (0.84–1.24) | 1.37 (0.97–1.92) | 1.34 (0.92–1.94) | ||
| Percentage male (%) | ≥60.0 | 0.98 (0.92–0.99) | – | 0.73 (0.50–0.88) | – | 1.34 (1.01–1.79) | – | |
| <60.0 | 0.92 (0.85–0.96) | 0.96 (0.91–0.99) | 0.80 (0.62–0.90) | 0.96 (0.89–1.03) | 1.15 (0.95–1.40) | 1.20 (0.99–1.45) | ||
| Follow-up duration | ≤30.0 days | 0.96 (0.91–0.98) | 0.97 (0.92–0.99) | 0.76 (0.61–0.87) | 0.99 (0.94–1.04) | 1.26 (1.05–1.51) | 1.28 (1.06–1.53) | |
| >30.0 days | – | 0.93 (0.78–0.98) | – | – | – | – | ||
| Study quality | High | 0.94 (0.83–0.98) | 0.97 (0.91–0.99) | 0.77 (0.50–0.92) | 0.97 (0.88–1.06) | 1.22 (0.89–1.67) | 1.26 (0.93–1.71) | |
| Moderate | 0.96 (0.90–0.98) | 0.95 (0.85–0.98) | 0.78 (0.61–0.89) | 1.01 (0.93–1.10) | 1.23 (1.01–1.49) | 1.22 (1.00–1.49) | ||
| Specificity | Country | Western | 0.33 (0.20–0.50) | 0.56 (0.47–0.64) | 0.50 (0.34–0.66) | 0.59 (0.36–0.96) | 0.66 (0.37–1.16) | 1.12 (0.78–1.61) |
| Eastern | 0.46 (0.27–0.67) | 0.44 (0.27–0.62) | 0.64 (0.55–0.73) | 1.05 (0.56–1.94) | 0.72 (0.45–1.16) | 0.69 (0.44–1.07) | ||
| Mean age (years) | ≥60.0 | 0.35 (0.19–0.55) | 0.45 (0.34–0.56) | 0.51 (0.37–0.64) | 0.78 (0.43–1.40) | 0.69 (0.38–1.25) | 0.88 (0.61–1.28) | |
| <60.0 | 0.38 (0.23–0.56) | 0.60 (0.44–0.74) | 0.63 (0.49–0.76) | 0.63 (0.38–1.06) | 0.60 (0.37–0.99) | 0.95 (0.68–1.34) | ||
| Percentage male (%) | ≥60.0 | 0.21 (0.10–0.37) | – | 0.56 (0.45–0.66) | – | 0.38 (0.19–0.74) | – | |
| <60.0 | 0.45 (0.30–0.61) | 0.52 (0.42–0.63) | 0.56 (0.42–0.70) | 0.87 (0.58–1.30) | 0.80 (0.52–1.24) | 0.93 (0.67–1.29) | ||
| Follow-up duration | ≤30.0 days | 0.35 (0.23–0.50) | 0.46 (0.35–0.57) | 0.56 (0.45–0.67) | 0.76 (0.48–1.20) | 0.63 (0.40–0.97) | 0.82 (0.60–1.13) | |
| >30.0 days | – | 0.60 (0.43–0.75) | – | – | – | – | ||
| Study quality | High | 0.41 (0.24–0.62) | 0.47 (0.35–0.60) | 0.61 (0.36–0.81) | 0.87 (0.51–1.51) | 0.67 (0.36–1.25) | 0.77 (0.47–1.25) | |
| Moderate | 0.33 (0.19–0.51) | 0.54 (0.39–0.68) | 0.54 (0.44–0.64) | 0.61 (0.35–1.08) | 0.61 (0.36–1.04) | 1.00 (0.72–1.40) | ||
| PLR | Country | Western | 1.45 (1.16–1.80) | 2.20 (1.79–2.70) | 1.75 (1.37–2.23) | 0.66 (0.49–0.89) | 0.83 (0.60–1.15) | 1.26 (0.91–1.73) |
| Eastern | 1.61 (1.24–2.08) | 1.66 (1.30–2.13) | 1.67 (1.37–2.03) | 0.97 (0.68–1.39) | 0.96 (0.70–1.33) | 0.99 (0.72–1.36) | ||
| Mean age (years) | ≥60.0 | 1.49 (1.13–1.96) | 1.76 (1.44–2.16) | 1.67 (1.34–2.09) | 0.85 (0.60–1.19) | 0.89 (0.63–1.27) | 1.05 (0.78–1.42) | |
| <60.0 | 1.50 (1.23–1.83) | 2.26 (1.61–3.18) | 1.86 (1.61–2.15) | 0.66 (0.45–0.98) | 0.81 (0.63–1.03) | 1.22 (0.84–1.02) | ||
| Percentage male (%) | ≥60.0 | 1.23 (1.06–1.43) | – | 1.67 (1.44–1.93) | – | 0.74 (0.60–0.91) | – | |
| <60.0 | 1.68 (1.29–2.19) | 2.02 (1.64–2.50) | 1.81 (1.45–2.28) | 0.83 (0.59–1.17) | 0.93 (0.66–1.31) | 1.12 (0.82–1.52) | ||
| Follow-up duration | ≤30.0 days | 1.47 (1.22–1.79) | 1.79 (1.47–2.19) | 1.74 (1.45–2.09) | 0.82 (0.62–1.08) | 0.84 (0.65–1.10) | 1.03 (0.78–1.35) | |
| >30.0 days | – | 2.34 (1.63–3.35) | – | – | – | – | ||
| Study quality | High | 1.61 (1.20–2.17) | 1.84 (1.45–2.34) | 1.97 (1.32–2.95) | 0.88 (0.60–1.28) | 0.82 (0.50–1.35) | 0.93 (0.58–1.49) | |
| Moderate | 1.44 (1.15–1.79) | 2.04 (1.53–2.72) | 1.70 (1.46–1.97) | 0.71 (0.49–1.01) | 0.85 (0.65–1.11) | 1.20 (0.87–1.66) | ||
| NLR | Country | Western | 0.10 (0.05–0.18) | 0.05 (0.02–0.14) | 0.25 (0.13–0.49) | 2.00 (0.62–6.41) | 0.40 (0.16–1.01) | 0.20 (0.06–0.65) |
| Eastern | 0.30 (0.18–0.48) | 0.14 (0.06–0.37) | 0.63 (0.51–0.77) | 2.14 (0.76–6.02) | 0.48 (0.28–0.81) | 0.22 (0.09–0.56) | ||
| Mean age (years) | ≥60.0 | 0.08 (0.03–0.20) | 0.06 (0.02–0.14) | 0.34 (0.19–0.60) | 1.33 (0.34–5.19) | 0.24 (0.08–0.71) | 0.18 (0.06–0.55) | |
| <60.0 | 0.19 (0.11–0.35) | 0.15 (0.04–0.51) | 0.50 (0.31–0.81) | 1.27 (0.31–5.13) | 0.38 (0.18–0.81) | 0.30 (0.08–1.17) | ||
| Percentage male (%) | ≥60.0 | 0.11 (0.05–0.24) | – | 0.48 (0.27–0.87) | – | 0.23 (0.09–0.61) | – | |
| <60.0 | 0.17 (0.10–0.28) | 0.07 (0.03–0.17) | 0.36 (0.22–0.60) | 2.43 (0.89–6.66) | 0.47 (0.23–0.97) | 0.19 (0.07–0.53) | ||
| Follow-up duration | ≤30.0 days | 0.12 (0.07–0.22) | 0.07 (0.02–0.18) | 0.42 (0.27–0.65) | 1.71 (0.50–5.92) | 0.29 (0.14–0.59) | 0.17 (0.05–0.54) | |
| >30.0 days | – | 0.12 (0.04–0.35) | – | – | – | – | ||
| Study quality | High | 0.13 (0.05–0.33) | 0.06 (0.02–0.20) | 0.38 (0.20–0.71) | 2.17 (0.49–9.60) | 0.34 (0.11–1.07) | 0.16 (0.04–0.59) | |
| Moderate | 0.12 (0.06–0.24) | 0.10 (0.04–0.26) | 0.40 (0.24–0.69) | 1.20 (0.37–3.85) | 0.30 (0.13–0.72) | 0.25 (0.09–0.73) | ||
| DOR | Country | Western | 12.68 (7.19–22.38) | 30.41 (10.41–88.82) | 6.32 (3.35–11.92) | 0.42 (0.12–1.40) | 2.01 (0.86–4.70) | 4.81 (1.38–16.72) |
| Eastern | 4.56 (3.67–5.65) | 10.05 (5.30–19.06) | 2.59 (1.83–3.67) | 0.45 (0.23–0.89) | 1.76 (1.17–2.65) | 3.88 (1.87–8.04) | ||
| Mean age (years) | ≥60.0 | 12.86 (6.11–27.07) | 23.01 (10.84–48.85) | 4.55 (2.50–8.28) | 0.56 (0.19–1.61) | 2.83 (1.09–7.35) | 5.06 (1.93–13.23) | |
| <60.0 | 6.24 (4.46–8.75) | 11.52 (3.44–38.55) | 3.41 (2.25–5.16) | 0.54 (0.15–1.90) | 1.83 (1.07–3.12) | 3.38 (0.94–12.12) | ||
| Percentage male (%) | ≥60.0 | 8.06 (4.08–15.91) | 12.32 (1.74–87.48) | 3.08 (1.76–5.37) | 0.65 (0.08–5.20) | 2.62 (1.09–6.31) | 4.00 (0.52–30.66) | |
| <60.0 | 9.16 (5.69–14.75) | 19.66 (9.31–41.51) | 4.51 (2.82–7.22) | 0.47 (0.19–1.13) | 2.03 (1.04–3.97) | 4.36 (1.80–10.54) | ||
| Follow-up duration | ≤30.0 days | 9.45 (6.19–14.41) | 19.21 (8.02–45.99) | 3.70 (2.46–5.55) | 0.49 (0.19–1.30) | 2.55 (1.42–4.59) | 5.19 (1.98–13.61) | |
| >30.0 days | 8.42 (5.14–13.80) | 15.90 (8.10–31.21) | 5.53 (2.34–13.03) | 0.53 (0.23–1.22) | 1.52 (0.57–4.10) | 2.88 (0.96–8.57) | ||
| Study quality | High | 7.96 (4.90–12.91) | 21.21 (7.10–63.34) | 5.17 (2.51–10.64) | 0.38 (0.11–1.24) | 1.54 (0.65–3.67) | 4.10 (1.11–15.22) | |
| Moderate | 9.44 (5.17–17.23) | 16.25 (6.88–38.41) | 3.33 (2.22–5.01) | 0.58 (0.20–1.66) | 2.83 (1.37–5.86) | 4.88 (1.88–12.63) | ||
| AUC | Country | Western | 0.85 (0.82–0.88) | 0.82 (0.78–0.85) | 0.74 (0.70–0.78) | 1.04 (0.98–1.10) | 1.15 (1.08–1.23) | 1.11 (1.03–1.19) |
| Eastern | 0.73 (0.69–0.77) | 0.74 (0.70–0.78) | 0.66 (0.62–0.70) | 0.99 (0.91–1.07) | 1.11 (1.02–1.20) | 1.12 (1.03–1.22) | ||
| Mean age (years) | ≥60.0 | 0.85 (0.82–0.88) | 0.83 (0.80–0.86) | 0.70 (0.66–0.74) | 1.02 (0.97–1.08) | 1.21 (1.14–1.30) | 1.19 (1.11–1.27) | |
| <60.0 | 0.73 (0.69–0.77) | 0.79 (0.75–0.82) | 0.70 (0.65–0.73) | 0.92 (0.86–0.99) | 1.04 (0.96–1.13) | 1.13 (1.05–1.21) | ||
| Percentage male (%) | ≥60.0 | 0.74 (0.70–0.77) | – | 0.65 (0.60–0.69) | – | 1.14 (1.05–1.24) | – | |
| <60.0 | 0.82 (0.79–0.85) | 0.81 (0.78–0.85) | 0.72 (0.68–0.76) | 1.01 (0.96–1.07) | 1.14 (1.07–1.22) | 1.13 (1.05–1.21) | ||
| Follow-up duration | ≤30.0 days | 0.81 (0.77–0.84) | 0.79 (0.75–0.82) | 0.69 (0.65–0.73) | 1.03 (0.96–1.09) | 1.17 (1.09–1.26) | 1.14 (1.06–1.23) | |
| >30.0 days | – | 0.83 (0.79–0.86) | – | – | – | – | ||
| Study quality | High | 0.79 (0.75–0.83) | 0.80 (0.76–0.83) | 0.74 (0.70–0.78) | 0.99 (0.92–1.06) | 1.07 (0.99–1.15) | 1.08 (1.01–1.16) | |
| Moderate | 0.80 (0.76–0.83) | 0.81 (0.77–0.84) | 0.67 (0.62–0.71) | 0.99 (0.93–1.05) | 1.19 (1.10–1.29) | 1.21 (1.05–1.14) |
AUC, area under the receiver operating characteristic curves; GRACE, Global Registry in Acute Coronary Events; HEART, History, Electrocardiography, Age, Risk factors and Troponin; NLR, negative likelihood ratio; PLR, positive likelihood ratio; TIMI, thrombolysis in myocardial infarction.
Discussion
This study was the first meta-analysis to conduct indirect comparisons of the predictive values of risk stratification assessed by the TIMI, HEART and GRACE scores on MACEs in ACP patients. The current study included a total of 40 262 ACP patients from 33 prospective cohort studies and across a wide range of patient characteristics. The findings of this study suggest that the predictive values of TIMI, HEART and GRACE scoring systems were better for MACEs in ACP patients admitted to the ED. Moreover, an indirect analysis indicated that the predictive value of TIMI and HEART was superior to that of GRACE for predicting MACEs, while there were no significant differences between TIMI and HEART for predicting MACEs. The results of sensitivity analyses for studies using all three scoring systems were consistent with those of the overall analysis. Meanwhile, we noted that the sensitivity of TIMI was lower than HEART for predicting MACEs.
Several systematic reviews and meta-analyses have illustrated the predictive values of the TIMI, HEART and GRACE scoring systems on MACEs in ACP patients.57–59 Hess et al included eight prospective studies and found that the TIMI score provided effective risk stratification for predicting MACEs in potential ACS patients, whereas it should not be used as the sole means for determining patient disposition.57 Van Den Berg et al identified 2 prospective and 10 retrospective cohort studies and suggested that the HEART score could be used to identify MACEs in patients with a suspected diagnosis of ACS.58 Roche et al included 11 studies and found that using 100 as the cut-off value of the GRACE score could predict the discharge of nearly 70% of presentations, while the predictive value for subsequent MACE risk was not obtained.59 However, the above studies only reported the diagnostic value of a single scoring system for predicting MACEs in ACS patients. Therefore, we performed the current meta-analysis of prospective studies to evaluate the predictive values of the TIMI, HEART and GRACE scoring systems on the risk of MACEs in ACP patients and systematically compared the predictive values among them.
The predictive value of the TIMI score for MACEs in ACP patients was statistically significant, whereas several studies reported inconsistent results. Sanchis et al found that the TIMI score was not associated with the risk of MACEs when 0 was used as the cut-off value.26 Graham et al found that a low TIMI score could not rule out cardiac causes of chest pain.35 Holly et al suggested that the TIMI score was not associated with the risk of MACE at 30 days when 0 was used as the cut-off value.36 Leung et al indicated that a modified TIMI score of 0 could not rule out 30-day MACEs in ACP patients admitted to the ED.45 The potential reasons for this could be that the TIMI score was designed for risk stratification in patients with non-ST-segment elevation ACS, which is mainly based on appropriate ECG changes or elevations of biomarkers of necrosis. Moreover, the presentation characteristics in ACP patients were not entered into the TIMI score. Finally, the prevalence of MACEs during the follow-up in these studies was lower than expected, resulting in broad 95% CIs, that is, no statistically significant difference.26 35 36 45
The predictive value of the HEART score for predicting MACEs in ACP patients was statistically significant. Nearly all included studies reported a similar conclusion, whereas the study conducted by McCord et al suggested that the HEART score after 4 hours of the presentation was associated with marginal predictive values for the risk of MACEs.47 The potential reason for this may be that this study used a modified HEART score, and the original HEART score only considered the initial cTn value, without taking serial sampling into account, which is associated with a lower prevalence of MACEs.60
The predictive value of the GRACE score for predicting MACE in ACP patients was statistically significant, and all included studies reported similar conclusions for the predictive value of the GRACE score on the risk of MACEs. Especially, the GRACE score was initially designed for post-ACS risk stratification, including unstable angina and non-ST-elevation ACS.11 The American Heart Association suggested the use of the GRACE score for admission and discharge of ACS patients. Moreover, the risk assessment for patients evaluated outside the hospital should be recommended to use GRACE.61 Therefore, the predictive value of the GRACE score in low-risk individuals was restricted, which should be addressed in clinical practice.
We noted that the predictive value of the GRACE score was inferior to that of TIMI and HEART scores. Sensitivity analyses were performed for studies reporting all three scoring systems, which included nine studies,42–44 46 48 52 53 55 56 and the results indicated TIMI and HEART having higher predictive values for MACEs than GRACE. The results of the sensitivity analyses were more reliable owing to the analysis of three scoring systems based on direct comparisons. Moreover, the GRACE score was initially developed for ACS patients but not for ACP patients, and the potential risk factors for the progression of MACEs were not considered in the GRACE score. Interestingly, we noted no significant difference between TIMI and HEART for predicting MACEs, while several studies reported that the predictive value of HEART for MACEs was superior to that of TIMI.44 46 48 52 53 55 56 Subgroup analysis found TIMI with lower AUC compared with HEART if the mean age of patients was <60.0 years. The potential explanation could be the use of HEART score in the absence of exact definitions for medical history across included studies,11 and the predictive value of HEART was more suitable in low-risk individuals.
Three strengths of this quantitative meta-analysis should be highlighted: (1) the study was based on prospective cohort studies and used relatively uniform cut-off values, which were associated with lower selective and informative biases; (2) the analysis of this study was based on a large sample size, and the findings in our study were more robust than any individual study and (3) the predictive values of the TIMI, HEART and GRACE scoring systems on the risk of MACEs in ACP patients were compared through an indirect analytical approach.
Despite the above-mentioned findings, the predictive values of the TIMI, HEART and GRACE could be affected by the definitions of MACEs and the ranges from a single endpoint (death or myocardial infarction) to a composite endpoint. Subgroup analysis based on MACE definition were not performed because of the definition of MACE across included studies are various. Therefore, MACE definition could affect the predictive value and follow-up durations owing to these factors that attribute to the weight of pooled conclusion. Moreover, this meta-analysis was based on crude data, and the adjusted results were not available. Furthermore, substantial heterogeneity was detected across the included studies, and the heterogeneity was not fully explained by sensitivity and subgroup analyses. In addition, the analysis was conducted on published articles, which causes inevitable publication biases. Finally, the current study was based on indirect comparisons between the predictive values of the TIMI, HEART and GRACE scoring systems as direct comparisons were not available.
The findings of this study indicated that risk stratification assessed by the TIMI, HEART and GRACE scores provides relatively appropriate predictive values for MACEs in ACP patients. The results of indirect comparison analysis indicated that TIMI and HEART had relatively relative better predictive values than GRACE on subsequent MACE risk. Further prospective cohort studies should be conducted to directly compare the predictive values of TIMI, HEART and GRACE on MACEs in ACP patients.
Supplementary Material
Footnotes
JK and Yc contributed equally.
Contributors: JK: study design and manuscript drafting. YC: study supervision, data analysis and result interpretation. XW: literature search and data collection. ZW: data curation. FC: manuscript revising and final approval.
Funding: The present study was supported by Pilot Project of Sci-Tech Innovation of Fujian Provincial Science & Technology Department (2018Y0009) and Provincial Natural Science Foundation of Fujian (2020J011058).
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available on reasonable request. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics statements
Patient consent for publication
Not required.
Ethics approval
Neither ethics approval nor participant consent was required as this study was based solely on the summary results of previously published articles. Individual patient data were not obtained or accessed.
References
- 1. Goodacre S, Cross E, Arnold J, et al. The health care burden of acute chest pain. Heart 2005;91:229–30. 10.1136/hrt.2003.027599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. World Health Organization . Cardiovascular diseases. Available: https://www.who.int/en/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds) [Accessed June 2019].
- 3. Launbjerg J, Fruergaard P, Hesse B, et al. Long-Term risk of death, cardiac events and recurrent chest pain in patients with acute chest pain of different origin. Cardiology 1996;87:60–6. 10.1159/000177061 [DOI] [PubMed] [Google Scholar]
- 4. Hollander JE. Risk stratification of emergency department patients with chest pain: the need for standardized reporting guidelines. Ann Emerg Med 2004;43:68–70. 10.1016/s0196-0644(03)00726-1 [DOI] [PubMed] [Google Scholar]
- 5. Lee TH, Goldman L. Evaluation of the patient with acute chest pain. N Engl J Med 2000;342:1187–95. 10.1056/NEJM200004203421607 [DOI] [PubMed] [Google Scholar]
- 6. Chase M, Robey JL, Zogby KE, et al. Prospective validation of the thrombolysis in myocardial infarction risk score in the emergency department chest pain population. Ann Emerg Med 2006;48:252–9. 10.1016/j.annemergmed.2006.01.032 [DOI] [PubMed] [Google Scholar]
- 7. Antman EM, Cohen M, Bernink PJ, et al. The TIMI risk score for unstable angina/non-ST elevation MI: a method for prognostication and therapeutic decision making. JAMA 2000;284:835–42. 10.1001/jama.284.7.835 [DOI] [PubMed] [Google Scholar]
- 8. Conway Morris A, Caesar D, Gray S, et al. Timi risk score accurately risk stratifies patients with undifferentiated chest pain presenting to an emergency department. Heart 2006;92:1333–4. 10.1136/hrt.2005.080226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Golabchi A, Sadeghi M, Sanei H, et al. Can TIMI risk score predict angiographic involvement in patients with ST-elevation myocardial infarction? ARYA Atheroscler 2010;6:69–73. [PMC free article] [PubMed] [Google Scholar]
- 10. Lee B, Chang AM, Matsuura AC, et al. Comparison of cardiac risk scores in ED patients with potential acute coronary syndrome. Crit Pathw Cardiol 2011;10:64–8. 10.1097/HPC.0b013e31821c79bd [DOI] [PubMed] [Google Scholar]
- 11. Six AJ, Backus BE, Kelder JC. Chest pain in the emergency room: value of the heart score. Neth Heart J 2008;16:191–6. 10.1007/BF03086144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. GRACE Investigators . Rationale and design of the grace (global registry of acute coronary events) project: a multinational registry of patients hospitalized with acute coronary syndromes. Am Heart J 2001;141:190–9. 10.1067/mhj.2001.112404 [DOI] [PubMed] [Google Scholar]
- 13. Ramsay G, Podogrodzka M, McClure C, et al. Risk prediction in patients presenting with suspected cardiac pain: the grace and TIMI risk scores versus clinical evaluation. QJM 2007;100:11–18. 10.1093/qjmed/hcl133 [DOI] [PubMed] [Google Scholar]
- 14. Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097. 10.1371/journal.pmed.1000097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Whiting PF, Rutjes AWS, Westwood ME, et al. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med 2011;155:529–36. 10.7326/0003-4819-155-8-201110180-00009 [DOI] [PubMed] [Google Scholar]
- 16. DerSimonian R, Laird N. Meta-Analysis in clinical trials. Control Clin Trials 1986;7:177–88. 10.1016/0197-2456(86)90046-2 [DOI] [PubMed] [Google Scholar]
- 17. Walter SD. Properties of the summary receiver operatingcharacteristic (SROC) curve for diagnostic test data. Stat Med 2002;9:1237–56. [DOI] [PubMed] [Google Scholar]
- 18. Ades AE, Lu G, Higgins JPT. The interpretation of random-effects meta-analysis in decision models. Med Decis Making 2005;25:646–54. 10.1177/0272989X05282643 [DOI] [PubMed] [Google Scholar]
- 19. Deeks JJ, Higgins JPT, Altman DG. Analyzing data and undertaking meta-analyses. In: Higgins J, Green S, eds. Cochrane Handbook for systematic reviews of interventions 5.0.1. Oxford, UK: The Cochrane Collaboration, 2008: chap 9. [Google Scholar]
- 20. Higgins JPT, Thompson SG, Deeks JJ, et al. Measuring inconsistency in meta-analyses. BMJ 2003;327:557–60. 10.1136/bmj.327.7414.557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Tobias A. Assessing the influence of a single study in meta-analysis. Stata Tech Bull 1999;47:15–17. [Google Scholar]
- 22. Woodward M. Epidemiology: study design and data analysis. 2nd edn. Boca Raton, FL, USA: Chapman and Hall/CRC, 2005. [Google Scholar]
- 23. Deeks JJ, Macaskill P, Irwig L. The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. J Clin Epidemiol 2005;58:882–93. 10.1016/j.jclinepi.2005.01.016 [DOI] [PubMed] [Google Scholar]
- 24. Trinquart L, Chatellier G, Ravaud P. Adjustment for reporting bias in network meta-analysis of antidepressant trials. BMC Med Res Methodol 2012;12:150. 10.1186/1471-2288-12-150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Tong KL, Kaul S, Wang X-Q, et al. Myocardial contrast echocardiography versus thrombolysis in myocardial infarction score in patients presenting to the emergency department with chest pain and a nondiagnostic electrocardiogram. J Am Coll Cardiol 2005;46:920–7. 10.1016/j.jacc.2005.03.076 [DOI] [PubMed] [Google Scholar]
- 26. Sanchis J, Bodí V, Núñez J, et al. New risk score for patients with acute chest pain, non-ST-segment deviation, and normal troponin concentrations: a comparison with the TIMI risk score. J Am Coll Cardiol 2005;46:443–9. 10.1016/j.jacc.2005.04.037 [DOI] [PubMed] [Google Scholar]
- 27. Pollack CV, Sites FD, Shofer FS, et al. Application of the TIMI risk score for unstable angina and non-ST elevation acute coronary syndrome to an unselected emergency department chest pain population. Acad Emerg Med 2006;13:13–18. 10.1197/j.aem.2005.06.031 [DOI] [PubMed] [Google Scholar]
- 28. Pelliccia F, Salvini P, Cartoni D, et al. Frequency and clinical correlates of changes in Thrombolysis In Myocardial Infarction risk score during observation period at emergency department in "low-risk" patients with acute chest pain. Am J Cardiol 2006;97:781–4. 10.1016/j.amjcard.2005.09.126 [DOI] [PubMed] [Google Scholar]
- 29. Lyon R, Morris AC, Caesar D, et al. Chest pain presenting to the Emergency Department--to stratify risk with GRACE or TIMI? Resuscitation 2007;74:90–3. 10.1016/j.resuscitation.2006.11.023 [DOI] [PubMed] [Google Scholar]
- 30. Body R, Carley S, McDowell G, et al. Can a modified thrombolysis in myocardial infarction risk score outperform the original for risk stratifying emergency department patients with chest pain? Emerg Med J 2009;26:95–9. 10.1136/emj.2008.058495 [DOI] [PubMed] [Google Scholar]
- 31. Campbell CF, Chang AM, Sease KL, et al. Combining thrombolysis in myocardial infarction risk score and clear-cut alternative diagnosis for chest pain risk stratification. Am J Emerg Med 2009;27:37–42. 10.1016/j.ajem.2008.01.028 [DOI] [PubMed] [Google Scholar]
- 32. Hess EP, Perry JJ, Calder LA, et al. Prospective validation of a modified thrombolysis in myocardial infarction risk score in emergency department patients with chest pain and possible acute coronary syndrome. Acad Emerg Med 2010;17:368–75. 10.1111/j.1553-2712.2010.00696.x [DOI] [PubMed] [Google Scholar]
- 33. Stracke S, Dörr O, Heidt MC, et al. Grace risk score as predictor of in-hospital mortality in patients with chest pain. Clin Res Cardiol 2010;99:627–31. 10.1007/s00392-010-0160-8 [DOI] [PubMed] [Google Scholar]
- 34. van der Zee PM, Verberne HJ, Cornel JH, et al. Grace and TIMI risk scores but not stress imaging predict long-term cardiovascular follow-up in patients with chest pain after a rule-out protocol. Neth Heart J 2011;19:324–30. 10.1007/s12471-011-0154-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Graham CA, Tsay SXH, Rotheray KR, et al. Validation of the TIMI risk score in Chinese patients presenting to the emergency department with chest pain. Int J Cardiol 2013;168:597–8. 10.1016/j.ijcard.2013.01.233 [DOI] [PubMed] [Google Scholar]
- 36. Holly J, Fuller M, Hamilton D, et al. Prospective evaluation of the use of the thrombolysis in myocardial infarction score as a risk stratification tool for chest pain patients admitted to an ED observation unit. Am J Emerg Med 2013;31:185–9. 10.1016/j.ajem.2012.07.006 [DOI] [PubMed] [Google Scholar]
- 37. Backus BE, Six AJ, Kelder JC, et al. A prospective validation of the heart score for chest pain patients at the emergency department. Int J Cardiol 2013;168:2153–8. 10.1016/j.ijcard.2013.01.255 [DOI] [PubMed] [Google Scholar]
- 38. Cullen L, Greenslade J, Hammett CJ, et al. Comparison of three risk stratification rules for predicting patients with acute coronary syndrome presenting to an Australian emergency department. Heart Lung Circ 2013;22:844–51. 10.1016/j.hlc.2013.03.074 [DOI] [PubMed] [Google Scholar]
- 39. Graham CA, Chan JWM, Chan CPY, et al. Prospective validation of thrombolysis in myocardial infarction and front door thrombolysis in myocardial infarction risk scores in Chinese patients presenting to the ED with chest pain. Am J Emerg Med 2014;32:1339–44. 10.1016/j.ajem.2014.08.032 [DOI] [PubMed] [Google Scholar]
- 40. Visser A, Wolthuis A, Breedveld R, et al. Heart score and clinical gestalt have similar diagnostic accuracy for diagnosing ACS in an unselected population of patients with chest pain presenting in the ED. Emerg Med J 2015;32:595–600. 10.1136/emermed-2014-203798 [DOI] [PubMed] [Google Scholar]
- 41. Boubaker H, Beltaief K, Grissa MH, et al. Inaccuracy of thrombolysis in myocardial infarction and global registry in acute coronary events scores in predicting outcome in ED patients with potential ischemic chest pain. Am J Emerg Med 2015;33:1209–12. 10.1016/j.ajem.2015.05.019 [DOI] [PubMed] [Google Scholar]
- 42. Wang H, Watson K, Robinson RD, et al. Chest pain risk scores can reduce emergent cardiac imaging test needs with low major adverse cardiac events occurrence in an emergency department observation unit. Crit Pathw Cardiol 2016;15:145–51. 10.1097/HPC.0000000000000090 [DOI] [PubMed] [Google Scholar]
- 43. Chen X-H, Jiang H-L, Li Y-M, et al. Prognostic values of 4 risk scores in Chinese patients with chest pain: prospective 2-centre cohort study. Medicine 2016;95:e4778. 10.1097/MD.0000000000004778 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Sakamoto JT, Liu N, Koh ZX, et al. Comparing heart, TIMI, and grace scores for prediction of 30-day major adverse cardiac events in high acuity chest pain patients in the emergency department. Int J Cardiol 2016;221:759–64. 10.1016/j.ijcard.2016.07.147 [DOI] [PubMed] [Google Scholar]
- 45. Leung Y-K, Cheng N-M, Chan CP-Y, et al. Early exclusion of major adverse cardiac events in emergency department chest pain patients: a prospective observational study. J Emerg Med 2017;53:287–94. 10.1016/j.jemermed.2017.05.006 [DOI] [PubMed] [Google Scholar]
- 46. Poldervaart JM, Langedijk M, Backus BE, et al. Comparison of the grace, heart and TIMI score to predict major adverse cardiac events in chest pain patients at the emergency department. Int J Cardiol 2017;227:656–61. 10.1016/j.ijcard.2016.10.080 [DOI] [PubMed] [Google Scholar]
- 47. McCord J, Cabrera R, Lindahl B, et al. Prognostic utility of a modified heart score in chest pain patients in the emergency department. Circ Cardiovasc Qual Outcomes 2017;10:e003101. 10.1161/CIRCOUTCOMES.116.003101 [DOI] [PubMed] [Google Scholar]
- 48. Reaney PDW, Elliott HI, Noman A, et al. Risk stratifying chest pain patients in the emergency department using heart, grace and TIMI scores, with a single contemporary troponin result, to predict major adverse cardiac events. Emerg Med J 2018;35:420–7. 10.1136/emermed-2017-207172 [DOI] [PubMed] [Google Scholar]
- 49. Greenslade JH, Chung K, Parsonage WA, et al. Modification of the thrombolysis in myocardial infarction risk score for patients presenting with chest pain to the emergency department. Emerg Med Australas 2018;30:47–54. 10.1111/1742-6723.12913 [DOI] [PubMed] [Google Scholar]
- 50. Moumneh T, Richard-Jourjon V, Friou E, et al. Reliability of the care rule and the heart score to rule out an acute coronary syndrome in non-traumatic chest pain patients. Intern Emerg Med 2018;13:1111–9. 10.1007/s11739-018-1803-4 [DOI] [PubMed] [Google Scholar]
- 51. Ishak M, Ali D, Fokkert MJ, et al. Fast assessment and management of chest pain patients without ST-elevation in the pre-hospital gateway (famous triage): ruling out a myocardial infarction at home with the modified heart score. Eur Heart J Acute Cardiovasc Care 2018;7:102–10. 10.1177/2048872616687116 [DOI] [PubMed] [Google Scholar]
- 52. Wong CP, Lui CT, Sung JG, et al. Prognosticating clinical prediction scores without clinical gestalt for patients with chest pain in the emergency department. J Emerg Med 2018;54:176–85. 10.1016/j.jemermed.2017.10.006 [DOI] [PubMed] [Google Scholar]
- 53. Al-Zaiti SS, Faramand Z, Alrawashdeh MO, et al. Comparison of clinical risk scores for triaging high-risk chest pain patients at the emergency department. Am J Emerg Med 2019;37:461–7. 10.1016/j.ajem.2018.06.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Huang Z, Wang K, Yang D, et al. The predictive value of the heart and grace scores for major adverse cardiac events in patients with acute chest pain. Intern Emerg Med 2021;16:193–200. 10.1007/s11739-020-02378-0 [DOI] [PubMed] [Google Scholar]
- 55. Torralba F, Navarro A, la Hoz JC. Heart, TIMI, and grace scores for prediction of 30-day major adverse cardiovascular events in the era of high-sensitivity troponin. OS Escores heart, TIMI E grace para Predição de Eventos Cardiovasculares Adversos Maiores no Período de 30 Dias Na era de Troponina I de Alta Sensibilidade. Arq Bras Cardiol 2020;114:795–802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Shin YS, Ahn S, Kim Y-J, et al. Risk stratification of patients with chest pain or anginal equivalents in the emergency department. Intern Emerg Med 2020;15:319–26. 10.1007/s11739-019-02230-0 [DOI] [PubMed] [Google Scholar]
- 57. Hess EP, Agarwal D, Chandra S, et al. Diagnostic accuracy of the TIMI risk score in patients with chest pain in the emergency department: a meta-analysis. CMAJ 2010;182:1039–44. 10.1503/cmaj.092119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Van Den Berg P, Body R. The heart score for early rule out of acute coronary syndromes in the emergency department: a systematic review and meta-analysis. Eur Heart J Acute Cardiovasc Care 2018;7:111–9. 10.1177/2048872617710788 [DOI] [PubMed] [Google Scholar]
- 59. Roche T, Jennings N, Clifford S, et al. Review article: diagnostic accuracy of risk stratification tools for patients with chest pain in the rural emergency department: a systematic review. Emerg Med Australas 2016;28:511–24. 10.1111/1742-6723.12622 [DOI] [PubMed] [Google Scholar]
- 60. Mahler SA, Hiestand BC, Goff DC, et al. Can the heart score safely reduce stress testing and cardiac imaging in patients at low risk for major adverse cardiac events? Crit Pathw Cardiol 2011;10:128–33. 10.1097/HPC.0b013e3182315a85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Ang DSC, Wei L, Kao MPC, et al. A comparison between B-type natriuretic peptide, global registry of acute coronary events (grace) score and their combination in ACS risk stratification. Heart 2009;95:1836–42. 10.1136/hrt.2008.160234 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2020-048356supp001.pdf (1.1MB, pdf)
Data Availability Statement
Data are available on reasonable request. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.