Abstract
Objective.
Pathologic extranodal extension (ENE) is an important adverse feature for human papillomavirus (HPV)–negative head and neck squamous cell carcinoma (HNSCC), but the prognostic significance of microscopic ENE (ENEmi) and role of adjuvant concurrent chemoradiation (CRT) for ENEmi remain unclear. This study evaluates (1) the prognostic significance of ENEmi in HPV-negative HNSCC and (2) whether adjuvant CRT is associated with improved overall survival (OS) for these patients.
Study Design.
Retrospective cohort study.
Setting.
Commission on Cancer (CoC)–accredited facilities.
Methods.
This retrospective cohort study included patients in the National Cancer Database from 2009 to 2015 with pathologic node-positive (pN+) HPV-negative HNSCC with either pathologic ENEmi or no ENE who had undergone margin-negative surgery. The association of ENEmi with OS was evaluated using Cox proportional hazard analyses. Analyses were repeated in patients with ENEmi receiving adjuvant therapy to evaluate the association of adjuvant CRT with OS.
Results.
We included 5483 patients with pN+ HPV-negative HNSCC, of whom 24% had ENEmi. On multivariable analysis, ENEmi was associated with decreased OS relative to no ENE (adjusted hazard ratio [aHR], 1.43; 95% CI, 1.28-1.59). Among patients with ENEmi who received ≥60 Gy of adjuvant radiation therapy (RT) (n = 617), adjuvant CRT was not associated with improved OS relative to RT (aHR, 0.91; 95% CI, 0.66-1.27).
Conclusion.
For patients with HPV-negative HNSCC, pN+ with ENEmi is associated with worse OS than pN+ without ENE. However, for patients with ENEmi, concurrent CRT is not associated with improved OS relative to RT. The optimal adjuvant paradigm for ENEmi requires additional investigation.
Keywords: extranodal extension, head and neck squamous cell carcinoma, adjuvant therapy
For patients with human papillomavirus (HPV)–negative head and neck squamous cell carcinoma (HNSCC), pathologic extranodal extension (ENE) is an established adverse feature that significantly increases the risk of locoregional recurrence and distant metastasis while decreasing overall survival (OS).1-4 As a result, ENE is now incorporated into the N-category in the eighth edition of the American Joint Committee on Cancer (AJCC) staging manual.5 In addition, ENE is a critically important factor in determining risk-adjusted adjuvant therapy. Based on the pooled analysis of 2 landmark clinical trials (EORTC 22931 and RTOG 9501), standard of care for patients with ENE is high-dose bolus cisplatin-based chemotherapy concurrent with adjuvant radiation therapy (RT).6-9
Since these landmark trials, emerging evidence has suggested an association between the extent of ENE and prognosis in the HPV-negative setting.10-12 Grading systems to characterize the amount of ENE (beyond present/absent) have been developed.13,14 In addition, AJCC8 and College of American Pathologist (CAP) guidelines recommend categorizing ENE as absent, microscopic (ENEmi) (≤2 mm), or macroscopic (>2 mm beyond the lymph node [LN] capsule).5,15
Although ENE is a negative prognosticator in the HPV-negative setting, the prognostic and therapeutic implications of ENEmi are less well understood. Findings from single-institution retrospective studies using varied definitions of ENEmi have produced conflicting results about its prognostic significance.11,12,16 In addition, although standard of care remains adjuvant chemoradiation therapy (CRT),8 results from recent single-institution retrospective studies for patients with HPV-negative HNSCC with ENEmi suggest that adjuvant CRT may not improve disease-free survival (DFS) or OS relative to RT.10,11 These studies have been limited by their single-institution nature, small sample size, and varied definitions of ENEmi. As a result, the prognostic significance of ENEmi and its implications for adjuvant therapy require further elucidation in a large, national data set using the AJCC 8 and CAP definition of ENEmi.
The aims of this study are therefore to (1) evaluate the prognostic significance of ENEmi in patients with HPV-negative HNSCC and (2) investigate whether the addition of chemotherapy concurrent with adjuvant RT improves OS for patients with ENEmi.
Methods
Study Design
We conducted a retrospective cohort study using data from the National Cancer Database (NCDB), a hospital-based cancer registry capturing cancer cases treated at Commission on Cancer (CoC)–accredited facilities across the United States.17,18 This study was exempt from review by the institutional review board of the Medical University of South Carolina.
Study Cohort
Included patients had a first primary HPV-negative squamous cell carcinoma (SCC)19 of the oral cavity, oropharynx, hypopharynx, or larynx (see Suppl. Table S1 in the online version of the article); were node positive (pN+) with either no ENE or ENEmi; and had undergone primary margin-negative resection and neck dissection between 2009 and 2015. Because the prognostic significance and therapeutic implications of ENEmi for HPV-positive oropharyngeal squamous cell carcinoma (OPSCC) are areas of significant clinical controversy and ongoing research,3,4 only patients with confirmed HPV-negative OPSCC were included (ie, patients with HPV-positive or HPV-unknown OPSCC were excluded). Patients with positive or unknown margin status were excluded to prevent confounding of the association between ENEmi, CRT, and OS. To avoid including LN biopsies without neck dissection, only patients with >2 examined LNs were included.20 ENE status was ascertained using the Collaborative Stage Site-Specific (CSSS) factor 9, which separates pathologic ENE into microscopic (≤2 mm) and macroscopic (>2 mm).15,21 We identified 13,508 patients with HPV-negative HNSCC with ENEmi or no ENE who had undergone primary margin-negative resection and neck dissection (without preoperative chemotherapy or RT), of whom 5483 were included in the cohort to evaluate the prognostic significance of ENEmi (Figure 1A). To assess the association of adjuvant CRT with OS for patients with ENEmi, we created a second cohort consisting of patients with ENEmi treated with adjuvant RT or CRT (Figure 1B). Only patients receiving ≥60 Gy were included in this cohort, to ensure that treatment resembled RT dosing from cooperative group clinical trials.6,7
Figure 1.
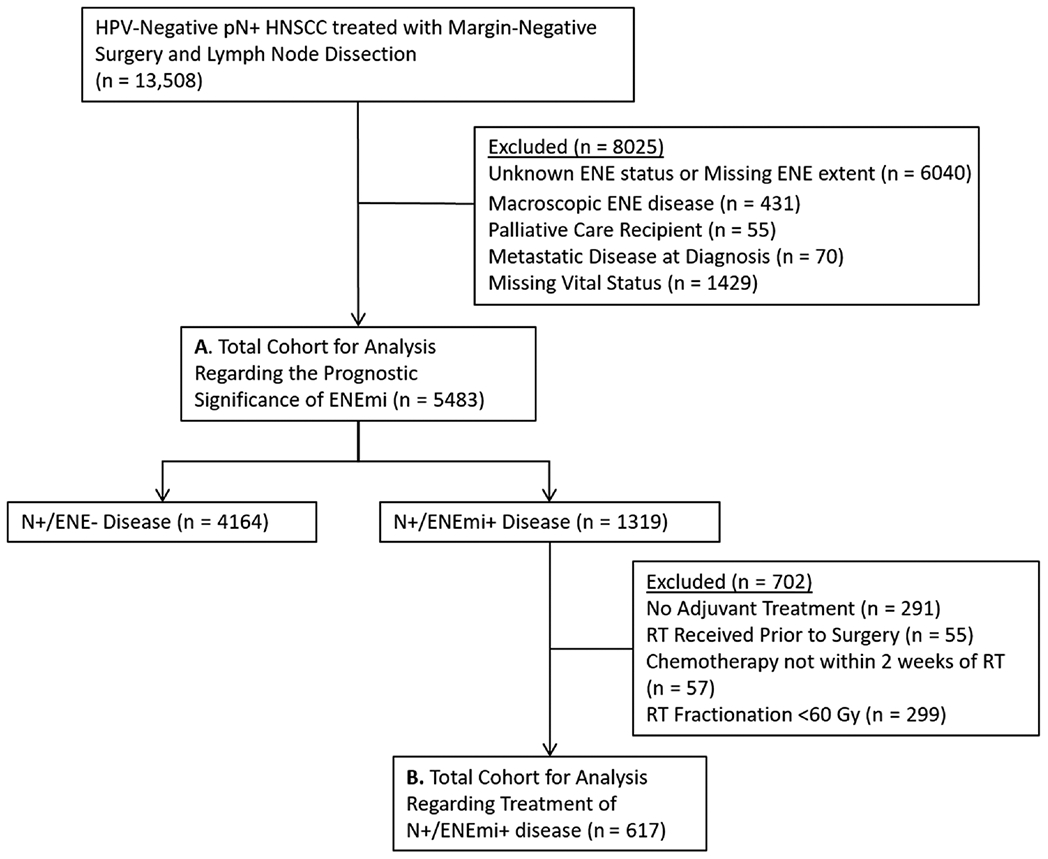
Prognostic significance of ENEmi (a) and adjuvant chemoradiation on overall survival (b). ENEmi, microscopic extranodal extension; HNSCC, head and neck squamous cell carcinoma; HPV, human papillomavirus; pN+, pathologic node positive; RT, radiation therapy.
Endpoints
The primary endpoint was OS, the time from date of first treatment to death or last follow-up. In the analysis of the prognostic significance of ENEmi, the primary explanatory variable of interest was extent of ENE (ENEmi vs No ENE). To analyze the oncologic impact of concurrent CRT in patients with ENEmi, the primary explanatory variable of interest was the type of adjuvant therapy (CRT vs RT).
Covariates
We analyzed the following covariates: demographics (sex, age, race/ethnicity, and severity of comorbidity as measured by the Charlson-Deyo combined comorbidity score [CDCC]), socioeconomic characteristics (insurance status, ZIP code income quartile, and ZIP code education level quartile), oncologic characteristics (tumor site, T-category, N-category, lymphovascular invasion, and number of metastatically involved LNs), facility-related variables (facility type, number of CoC-accredited facilities), and measures of treatment timeliness (time to postoperative radiotherapy [PORT] and treatment package time) using previously described thresholds.22-24
Statistical Analysis
Statistical analyses were conducted from December 1, 2019, to August 9, 2020. For descriptive analyses, categorical variables were compared between study groups using χ2 or 2-sided Fisher exact tests depending on the sample size. Kaplan-Meier estimates of OS were used to examine unadjusted survival time distributions; comparisons were performed using the log-rank test. Uni- and multivariable Cox proportional hazards regression models were created to test the hypotheses that (1) ENEmi is associated with worse OS relative to no ENE and (2) adding chemotherapy concurrent with RT is not associated with improved OS relative to RT in patients with ENEmi. Prespecified demographic covariates (age, sex, CDCC) and covariates associated with OS with a P value ≤.15 on univariable analysis were included for consideration in the multivariable Cox proportional hazard models.25,26 Ratios of rates of death between comparative arms were expressed as hazard ratios (HRs) and corresponding 95% CIs. To evaluate whether the association of CRT with OS was moderated by other adverse oncologic characteristics, interaction effects between adjuvant therapy and T-category, N-category, number of metastatically involved LNs, and tumor subsites were analyzed. To ensure that study findings were not biased by the use of less efficacious systemic agents (eg, cetuximab-based therapy, which is associated with worse outcomes than cisplatin in a variety of settings and was not used in EORTC 22931 or RTOG 9501),27-29 we performed a sensitivity analysis excluding those known to have received targeted therapy (eg, cetuximab) with adjuvant RT.30 A 2-sided P value <.05 was considered statistically significant. Statistical analyses were performed using SAS, version 9.4 (SAS Institute).
Results
Patient Characteristics
In total, 5483 patients with HPV-negative HNSCC treated with margin-negative surgical resection and neck dissection with ENEmi or no ENE were included (Table 1). The mean (SD) age was 60.4 (12.1) years; 3694 (67%) were men and 4671 (85%) were white. The median follow-up time was 26.2 months (range, 1.0-85.7 months). Seventy-six percent (n = 4164) of patients were ENE negative, and 24% (n = 1319) had ENEmi. Patients with ENEmi were more likely to have T3/T4 category HNSCC (57% vs 46%, P < .001), lymphovascular invasion (45% vs 32%, P < .001), and ≥3 metastatically involved LNs (57% vs 27%, P < .001) than patients without ENE.
Table 1.
Characteristics of the Study Cohort With HPV-Negative Head and Neck Squamous Cell Carcinoma With Microscopic ENE or pN1 Without ENE
| Variable | Total (N = 5483), No. (%) |
ENE negative (n =4164), No. (%) |
Microscopic ENE (n = 1319), No. (%) |
P value |
|---|---|---|---|---|
| Age, y | .292 | |||
| <50 | 885 (16.1) | 678 (16.3) | 207 (15.7) | |
| 50-59 | 1789 (32.6) | 1374 (33.0) | 415 (31.5) | |
| 60-69 | 1574 (28.7) | 1199 (28.8) | 375 (28.4) | |
| ≥70 | 1235 (22.5) | 913 (21.9) | 322 (24.4) | |
| Sex | .444 | |||
| Men | 3694 (67.4) | 2794 (67.1) | 900 (68.2) | |
| Women | 1789 (32.6) | 1370 (32.9) | 419 (31.8) | |
| Race | .967 | |||
| White | 4671 (85.2) | 3550 (85.3) | 1121 (85.0) | |
| Black | 511 (9.3) | 387 (9.3) | 124 (9.4) | |
| Other/unknown | 301 (5.5) | 227 (5.4) | 74 (5.6) | |
| Insurance type | .224 | |||
| Private | 2204 (40.2) | 1673 (40.2) | 531 (40.3) | |
| Medicaid | 778 (14.2) | 592 (14.2) | 186 (14.1) | |
| Medicare | 1984 (36.2) | 1486 (35.7) | 498 (37.8) | |
| Uninsured | 335 (6.1) | 269 (6.5) | 66 (5.0) | |
| Other government/unknown | 182 (3.3) | 144 (3.5) | 38 (2.9) | |
| Charlson-Deyo combined comorbidity score | .495 | |||
| 0 | 4037 (73.6) | 3082 (74.0) | 955 (72.4) | |
| 1 | 1131 (20.6) | 848 (20.4) | 283 (21.5) | |
| 2 or more | 315 (5.8) | 234 (5.6) | 81 (6.1) | |
| Educationa | .864 | |||
| Highest quartile | 1080 (19.8) | 814 (19.6) | 266 (20.2) | |
| Second highest quartile | 1795 (32.9) | 1360 (32.8) | 435 (33.1) | |
| Second lowest quartile | 1495 (27.4) | 1147 (27.6) | 348 (26.5) | |
| Lowest quartile | 1094 (20.0) | 829 (20.0) | 265 (20.2) | |
| Median household incomea | .338 | |||
| Highest quartile | 1469 (26.9) | 1097 (26.4) | 372 (28.3) | |
| Second highest quartile | 1423 (26.1) | 1093 (26.4) | 330 (25.1) | |
| Second lowest quartile | 1452 (26.6) | 1119 (27.0) | 333 (25.3) | |
| Lowest quartile | 1118 (20.5) | 839 (20.2) | 279 (21.2) | |
| Facility typea | .057 | |||
| Community cancer program | 207 (4.0) | 152 (3.8) | 55 (4.4) | |
| Comprehensive community | 1151 (21.9) | 909 (22.8) | 242 (19.2) | |
| Academic/research | 3369 (64.2) | 2535 (63.6) | 834 (66.2) | |
| Integrated network | 518 (9.9) | 390 (9.8) | 128 (10.2) | |
| No. of CoC facilities | .355 | |||
| 1 | 4278 (78.0) | 3261 (78.3) | 1017 (77.1) | |
| >1 | 1205 (22.0) | 903 (21.7) | 302 (22.9) | |
| Regiona | .017 | |||
| Northeast | 1026 (19.6) | 750 (18.8) | 276 (21.9) | |
| South | 1898 (36.2) | 1484 (37.2) | 414 (32.9) | |
| Midwest | 1473 (28.1) | 1115 (28.0) | 358 (28.4) | |
| West | 848 (16.2) | 637 (16.0) | 211 (16.8) | |
| Geographya | .866 | |||
| Metropolitan | 4337 (80.6) | 3293 (80.6) | 1044 (80.8) | |
| Nonmetropolitan | 1041 (19.4) | 793 (19.4) | 248 (19.2) | |
| Adjuvant therapya | <.001 | |||
| None | 1420 (26.8) | 1174 (29.2) | 246 (19.3) | |
| RT | 1864 (35.2) | l628 (40.4) | 236 (18.5) | |
| CRT | 2018 (38.1) | 1226 (30.4) | 792 (62.2) | |
| Cancer primary site | .130 | |||
| Oral cavity | 3804 (69.4) | 2905 (69.8) | 899 (68.2) | |
| Oropharynx (HPV negative) | 473 (8.6) | 368 (8.8) | 105 (8.0) | |
| Hypopharynx/larynx | 1206 (22.0) | 891 (21.4) | 315 (23.9) | |
| AJCC 6/7 pathologic stage | <.001 | |||
| III | 1547 (29.0) | 1362 (33.7) | 185 (14.3) | |
| IV | 3791 (71.0) | 2683 (66.3) | 1108 (85.7) | |
| AJCC 6/7 pathologic tumor categorya | <.001 | |||
| T1/T2 | 2814 (51.6) | 2249 (54.3) | 565 (42.9) | |
| T3/T4 | 2644 (48.4) | 1893 (45.7) | 751 (57.1) | |
| AJCC 6/7 pathologic nodal category | <.001 | |||
| N1 | 2149 (39.2) | 1896 (45.5) | 253 (19.2) | |
| N2 | 3278 (59.8) | 2243 (53.9) | 1035 (78.5) | |
| N3 | 56 (1.0) | 25 (0.6) | 31 (2.4) | |
| Lymphovascular invasiona | <.001 | |||
| Absent | 3106 (65.1) | 2473 (68.4) | 633 (54.9) | |
| Present | 1662 (34.9) | 1142 (31.6) | 520 (45.1) | |
| No. of positive nodesa | <.001 | |||
| 1 | 2345 (42.8) | 2043 (49.1) | 302 (22.9) | |
| 2 | 1251 (22.8) | 979 (23.5) | 272 (20.6) | |
| ≥3 | 1887 (34.4) | 1142 (27.4) | 745 (56.5) |
Abbreviations: AJCC, American Joint Committee on Cancer; CoC, Commission on Cancer; CRT, chemoradiation therapy; ENE, extranodal extension; HPV, human papillomavirus; RT, radiation therapy.
May not sum to total patient number due to missing data.
Prognostic Significance of ENEmi
Table 2 demonstrates the results of univariable and multivariable Cox regression analyses examining the prognostic significance of ENEmi relative to absent ENE in patients with HPV-negative HNSCC following margin-negative surgical resection and neck dissection. On univariable analysis, ENEmi was associated with worse OS (HR, 1.67; 95% CI, 1.53-1.83) (see Suppl. Figure S1 in the online version of the article). On multivariable analysis adjusting for demographics, tumor characteristics, and adjuvant treatment, ENEmi remained associated with worse OS (adjusted HR [aHR], 1.43; 95% CI, 1.28-1.59). In addition, age, insurance type, CDCC score, adjuvant therapy, cancer primary site, T-category N-category, and number of positive LNs were also associated with OS.
Table 2.
Cox Regression Analysis of the Survival Impact of Microscopic ENE in Margin-Negative HPV-Negative Head and Neck Squamous Cell Carcinoma (n = 5483).
| Variable | Univariable HR (95% CI) | P value | Multivariable HR (95% CI) | P value |
|---|---|---|---|---|
| Pathologic ENE status | ||||
| No ENE | 1 [Reference] | 1 [Reference] | ||
| Microscopic ENE | 1.67 (1.53-1.83) | <.001 | 1.43 (1.28-1.59) | <.001 |
| Age, y | ||||
| <50 | 1 [Reference] | 1 [Reference] | ||
| 50-59 | 1.15 (1.00-1.32) | .044 | 1.00 (0.85-1.19) | .964 |
| 60-69 | 1.41 (1.23-1.62) | <.001 | 1.14 (0.96-1.35) | .146 |
| ≥70 | 1.99 (1.73-2.29) | <.001 | 1.44 (1.19-1.75) | <.001 |
| Sex | ||||
| Men | 1 [Reference] | 1 [Reference] | ||
| Women | 0.91 (0.83-0.99) | .034 | 0.96 (0.86-1.06) | .391 |
| Race | ||||
| White | 1 [Reference] | 1 [Reference] | .909 | |
| Black | 1.17 (1.02-1.34) | .027 | 1.01 (0.86-1.19) | .513 |
| Other/unknown | 0.92 (0.76-1.11) | .384 | 0.93 (0.75-1.16) | .256 |
| Insurance type | ||||
| Private | 1 [Reference] | 1 [Reference] | ||
| Medicaid | 1.50 (1.32-1.71) | <.001 | 1.27 (1.09-1.48) | .002 |
| Medicare | 1.74 (1.58-1.91) | <.001 | 1.25 (1.09-1.43) | .001 |
| Uninsured | 1.40 (1.17-1.67) | <.001 | 1.24 (1.00-1.52) | .045 |
| Other government/unknown | 1.14 (0.88-1.47) | .333 | 0.85 (0.62-1.17) | .317 |
| Charlson-Deyo combined comorbidity score | ||||
| 0 | 1 [Reference] | 1 [Reference] | ||
| 1 | 1.23 (1.11-1.36) | <.001 | 1.07 (0.95-1.20) | .256 |
| 2 or more | 1.69 (1.45-1.98) | <.001 | 1.46 (1.21-1.75) | <.001 |
| Education | —a | |||
| Highest quartile | 1 [Reference] | |||
| Second highest quartile | 1.09 (0.96-1.23) | .181 | ||
| Second lowest quartile | 1.27 (1.12-1.44) | <.001 | ||
| Lowest quartile | 1.25 (1.09-1.43) | .001 | ||
| Median household income | —a | |||
| Highest quartile | 1 [Reference] | |||
| Second highest quartile | 1.05 (0.93-1.18) | .420 | ||
| Second lowest quartile | 1.08 (0.96-1.21) | .217 | ||
| Lowest quartile | 1.29 (1.14-1.45) | <.001 | ||
| Facility type | —a | |||
| Community cancer program | 1 [Reference] | |||
| Integrated network | 1.07 (0.85-1.36) | .561 | ||
| Comprehensive community | 1.06 (0.84-1.31) | .636 | ||
| Academic/research | 1.01 (0.78-1.31) | .927 | ||
| No. of CoC facilities | —a | |||
| 1 | 1 [Reference] | |||
| >1 | 1.11 (1.00-1.22) | .047 | ||
| Region | —a | |||
| Northeast | 1 [Reference] | |||
| South | 1.18 (1.05-1.33) | .006 | ||
| Midwest | 1.03 (0.91-1.17) | .650 | ||
| West | 1.09 (0.95-1.26) | .225 | ||
| Geography | —a | |||
| Metropolitan | 1 [Reference] | |||
| Nonmetropolitan | 1.08 (0.98-1.20) | .137 | ||
| Adjuvant therapy | ||||
| None | 1 [Reference] | 1 [Reference] | ||
| RT | 0.69 (0.62-0.76) | <.001 | 0.70 (0.63-0.79) | <.001 |
| CRT | 0.75 (0.67-0.83) | <.001 | 0.59 (0.52-0.66) | <.001 |
| Cancer primary site | ||||
| Oral cavity | 1 [Reference] | 1 [Reference] | ||
| Oropharynx (HPV negative) | 0.34 (0.27-0.42) | <.001 | 0.44 (0.34-0.57) | <.001 |
| Hypopharynx/larynx | 1.04 (0.94-1.14) | .473 | 0.69 (0.62-0.78) | <.001 |
| AJCC 6/7 pathologic tumor category | ||||
| T1/T2 | 1 [Reference] | 1 [Reference] | ||
| T3/T4 | 1.95 (1.79-2.13) | <.001 | 1.73 (1.56-1.93) | <.001 |
| AJCC 6/7 pathologic nodal category | ||||
| N1 | 1 [Reference] | 1 [Reference] | ||
| N2 | 1.55 (1.31-1.69) | <.001 | 0.89 (0.70-1.14) | .374 |
| N3 | 2.26 (1.58-3.24) | <.001 | 1.62 (1.02-2.55) | .040 |
| Lymphovascular invasion | ||||
| Absent | 1 [Reference] | 1 [Reference] | ||
| Present | 1.32 (1.21-1.45) | <.001 | 1.10 (1.00-1.22) | .051 |
| No. of metastatically involved nodes | ||||
| 1 | 1 [Reference] | 1 [Reference] | ||
| 2 | 1.27 (1.14-1.43) | <.001 | 1.36 (1.06-1.75) | .015 |
| ≥3 | 2.06 (1.88-2.27) | <.001 | 2.13 (1.67-2.71) | <.001 |
Abbreviations: AJCC, American Joint Committee on Cancer; CoC, Commission on Cancer; CRT, chemoradiation therapy; ENE, extranodal extension; HPV, human papillomavirus; HR, hazard ratio; RT, radiation therapy.
Dropped out of final multivariable model.
Adjuvant CRT vs RT for ENEmi
To evaluate whether the addition of chemotherapy concurrent with adjuvant RT improves OS in patients with HPV-negative HNSCC with ENEmi, we identified a subset of 617 patients with ENEmi who received ≥60 Gy of adjuvant RT (n = 133; 22%) or CRT (n = 484; 78%) (Table 3). The median follow-up time was 24.1 months (range, 4.0-90.9 months). Patients who received adjuvant CRT were more likely to be <70 years of age (86% vs 62%, P < .001), have AJCC stage IV HNSCC (89% vs 77%, P = .001), and have ≥3 positive LNs (60% vs 42%, P < .001) than patients receiving RT. No differences in treatment package time or time to PORT were seen (P = .691 and P = .875, respectively).
Table 3.
Characteristics of Patients With Microscopic ENE Treated With Adjuvant RT or CRT.
| Variable | Total (N = 617), No. (%) |
Adjuvant RT (n = 133), No. (%) |
Adjuvant CRT (n = 484), No. (%) |
P value |
|---|---|---|---|---|
| Age, y | <.001 | |||
| <50 | 106 (17.2) | 11 (8.3) | 95 (19.6) | |
| 50-59 | 200 (32.4) | 31 (23.3) | 169 (34.9) | |
| 60-69 | 194 (31.4) | 40 (30.1) | 154 (31.8) | |
| ≥70 | 117 (19.0) | 51 (38.3) | 66 (13.6) | |
| Sex | .997 | |||
| Men | 437 (70.7) | 94 (70.7) | 342 (70.7) | |
| Women | 181 (29.3) | 39 (29.3) | 142 (29.3) | |
| Racea | .710 | |||
| White | 524 (84.9) | 110 (82.7) | 414 (85.5) | |
| Black | 62 (10.1) | 15 (11.3) | 47 (9.7) | |
| Insurance typea | <.001 | |||
| Private | 266 (43.1) | 45 (33.8) | 221 (45.7) | |
| Medicaid | 91 (14.8) | 13 (9.8) | 78 (16.1) | |
| Medicare | 216 (35.0) | 67 (50.4) | 149 (30.8) | |
| Charlson-Deyo combined comorbidity scorea | .166 | |||
| 0 | 461 (74.7) | 91 (68.4) | 370 (76.5) | |
| 1 | 121 (19.6) | 33 (24.8) | 88 (18.2) | |
| Educationb | .629 | |||
| Highest quartile | 130 (21.1) | 28 (21.1) | 102 (21.2) | |
| Second highest quartile | 217 (35.3) | 42 (31.6) | 175 (36.3) | |
| Second lowest quartile | 159 (25.9) | 35 (26.3) | 124 (25.7) | |
| Lowest quartile | 109 (17.7) | 28 (21.1) | 81 (16.8) | |
| Median household incomeb | .449 | |||
| Highest quartile | 182 (29.6) | 37 (27.8) | 146 (30.1) | |
| Second highest quartile | 158 (25.7) | 39 (29.3) | 119 (24.7) | |
| Second lowest quartile | 149 (24.2) | 35 (26.3) | 114 (23.7) | |
| Lowest quartile | 126 (20.5) | 22 (16.5) | 104 (21.6) | |
| Facility typeb | .290 | |||
| Comprehensive community | 120 (20.3) | 32 (24.1) | 88 (19.3) | |
| Academic/research | 363 (61.5) | 80 (60.2) | 283 (61.9) | |
| Integrated networks | 84 (14.2) | 19 (14.3) | 65 (14.2) | |
| No. of CoC facilities | .691 | |||
| 1 | 484 (78.4) | 106 (79.7) | 378 (78.1) | |
| >1 | 133 (21.6) | 27 (20.3) | 106 (21.9) | |
| Regionb | .131 | |||
| Northeast | 113 (19.2) | 20 (15.0) | 93 (20.4) | |
| South | 189 (32.0) | 44 (33.1) | 145 (31.7) | |
| Midwest | 182 (30.9) | 37 (27.8) | 145 (31.7) | |
| West | 106 (18.0) | 32 (24.1) | 74 (16.2) | |
| Geographyb | .673 | |||
| Metropolitan | 491 (81.0) | 107 (82.3) | 384 (80.7) | |
| Nonmetropolitan | 115 (19.0) | 23 (17.7) | 92 (19.3) | |
| Cancer primary site | .631 | |||
| Oral cavity | 397 (64.3) | 90 (67.7) | 307 (63.4) | |
| Oropharynx (HPV negative) | 66 (10.7) | 12 (9.0) | 54 (11.2) | |
| Hypopharynx/larynx | 154 (25.0) | 31 (23.3) | 123 (25.4) | |
| AJCC 6/7 pathologic stageb | <.001 | |||
| III | 84 (13.9) | 30 (22.9) | 54 (11.4) | |
| IV | 520 (86.1) | 101 (77.1) | 419 (88.6) | |
| AJCC 6/7 pathologic tumor categoryb | .182 | |||
| TI/T2 | 283 (46.0) | 68 (51.1) | 216 (44.6) | |
| T3/T4 | 332 (54.0) | 65 (48.9) | 267 (55.4) | |
| AJCC 6/7 pathologic nodal categorya | .002 | |||
| N1 | 111 (18.0) | 38 (28.6) | 73 (15.1) | |
| N2 | 486 (78.8) | 92 (69.2) | 394 (81.4) | |
| Lymphovascular invasionb | .054 | |||
| Absent | 303 (55.7) | 72 (63.7) | 231 (53.6) | |
| Present | 241 (44.3) | 41 (36.3) | 200 (46.4) | |
| No. of positive nodesb | <.001 | |||
| 1 | 138 (22.4) | 46 (34.6) | 92 (19.0) | |
| 2 | 134 (21.7) | 31 (23.3) | 103 (21.3) | |
| ≥3 | 345 (55.9) | 56 (42.1) | 289 (59.7) | |
| Time to PORT | .875 | |||
| ≤42 days | 175 (28.4) | 37 (27.8) | 138 (28.5) | |
| >42 days | 442 (71.6) | 96 (72.2) | 346 (71.5) | |
| Total package time | .691 | |||
| <87 days | 136 (22.0) | 31 (23.3) | 105 (21.7) | |
| ≥87 days | 481 (78.0) | 102 (76.7) | 379 (78.3) |
Abbreviations: AJCC, American Joint Committee on Cancer; CoC, Commission on Cancer; CRT, chemoradiation therapy; ENE, extranodal extension; HPV, human papillomavirus; PORT, postoperative radiation therapy; RT, radiation therapy.
May not sum to total patient number to omit cells with values <10 per National Cancer Database policy.
May not sum to total patient number due to missing data.
The median OS of patients with ENEmi treated with adjuvant RT was 40.0 months, compared to 41.9 months of patients treated with adjuvant CRT (see Suppl. Figure S2 in the online version of the article). Adjuvant CRT was not associated with improved OS on univariable analysis (HR, 0.95; 95% CI, 0.71-1.26). On Cox proportional hazards multivariable analysis adjusted for age, sex, insurance status, severity of comorbidity, primary tumor site, pathologic AJCC overall stage, number of positive LNs, and treatment package time, adjuvant CRT was still not associated with improved OS relative to RT (aHR, 0.91; 95% CI, 0.66-1.27) (Table 4). Primary site, pathologic AJCC overall stage, and the number of positive LNs were associated with OS on multivariable analysis. There was no effect modification of the association of CRT with OS by T-category, N-category, number of metastatically involved LNs, or tumor subsite (P > .05 for all interactions).
Table 4.
Cox Regression Analysis of the Survival Impact of Adjuvant Therapy Among Patients With HPV-Negative HNSCC With Microscopic Extranodal Extension Undergoing Margin-Negative Surgery (n = 617).
| Variable | Univariable HR (95% CI) | P value | Multivariable HR (95% CI) | P value |
|---|---|---|---|---|
| Adjuvant treatment | ||||
| RT | 1 [Reference] | 1 [Reference] | ||
| CRT | 0.95 (0.71-1.26) | .709 | 0.91 (0.66-1.27) | .584 |
| Age, y | ||||
| <50 | 1 [Reference] | 1 [Reference] | ||
| 50-59 | 1.16 (081-1.68) | .419 | 1.22 (0.78-1.88) | .382 |
| 60-69 | 1.36 (0.95-1.97) | .097 | 1.36 (0.86-2.14) | .192 |
| ≥70 | 1.55 (1.04-2.30) | .031 | 1.65 (0.95-2.84) | .074 |
| Sex | ||||
| Men | 1 [Reference] | 1 [Reference] | ||
| Women | 0.81 (0.63-1.05) | .117 | 0.87 (0.65-1.16) | .331 |
| Race | ||||
| White | 1 [Reference] | —a | ||
| Black | 1.18 (0.82-1.69) | .384 | ||
| Other/unknown | 1.17 (0.69-2.01) | .558 | ||
| Insurance type | ||||
| Private | Reference | Reference | ||
| Medicaid | 1.32 (0.94-1.86) | .112 | 1.23 (0.82-1.83) | .320 |
| Medicare | 1.25 (0.96-1.63) | .092 | 0.96 (0.67-1.37) | .805 |
| Uninsured | 1.46 (0.87-2.47) | .153 | 1.76 (0.98-3.18) | .059 |
| Other government/unknown | 0.67 (0.28-1.65) | .387 | 0.73 (0.27-2.02) | .547 |
| Charlson-Deyo combined comorbidity score | ||||
| 0 | 1 [Reference] | 1 [Reference] | ||
| 1 | 1.22 (0.91-1.61) | .178 | 0.99 (0.71-1.39) | .965 |
| 2 or more | 1.42 (0.89-2.27) | .144 | 1.45 (0.86-2.45) | .161 |
| Education | —a | |||
| Highest quartile | 1 [Reference] | |||
| Second highest quartile | 1.19 (0.86-1.64) | .295 | ||
| Second lowest quartile | 1.12 (0.79-1.59) | .510 | ||
| Lowest quartile | 1.18 (0.81-1.72) | .400 | ||
| Median household income | —a | |||
| Highest quartile | 1 [Reference] | |||
| Second highest quartile | 1.08 (0.80-1.47) | .608 | ||
| Second lowest quartile | 0.86 (0.62-1.19) | .365 | ||
| Lowest quartile | 1.02 (0.73-1.41) | .916 | ||
| Facility type | —a | |||
| Community cancer program | 1 [Reference] | |||
| Integrated network | 1.44 (0.74-2.81) | .282 | ||
| Comprehensive community | 1.27 (0.67-2.40) | .464 | ||
| Academic/research | 1.46 (0.73-2.92) | .287 | ||
| No. of CoC facilities | —a | |||
| 1 | 1 [Reference] | |||
| >1 | 0.87 (0.66-1.14) | .315 | ||
| Region | —a | |||
| Northeast | 1 [Reference] | |||
| South | 1.36 (0.97-1.91) | .073 | ||
| Midwest | 1.00 (0.70-1.42) | .984 | ||
| West | 1.08 (0.73-1.60) | .704 | ||
| Geography | —a | |||
| Metropolitan | 1 [Reference] | |||
| Nonmetropolitan | 1.18 (0.89-1.56) | .253 | ||
| Cancer primary site | ||||
| Oral cavity | 1 [Reference] | 1 [Reference] | ||
| Oropharynx (HPV negative) | 0.40 (0.24-0.66) | <.001 | 0.46 (0.25-0.84) | .012 |
| Hypopharynx/larynx | 0.98 (0.75-1.27) | .856 | 0.65 (0.47-0.89) | .008 |
| AJCC 6/7 pathologic tumor category | ||||
| T1/T2 | 1 [Reference] | 1 [Reference] | ||
| T3/T4 | 1.89 (1.49-2.41) | <.001 | 1.74 (1.30-2.34) | <.001 |
| AJCC 6/7 pathologic nodal category | —a | |||
| N1 | Reference | |||
| N2 | 1.63 (1.16-2.29) | .005 | ||
| N3 | 3.26 (1.80-5.92) | <.001 | ||
| Lymphovascular invasion | —a | |||
| Absent | 1 [Reference] | |||
| Present | 1.13 (0.88-1.44) | .348 | ||
| No. of metastatically involved nodes | ||||
| 1 | 1 [Reference] | 1 [Reference] | ||
| 2 | 1.19 (0.80-1.78) | .380 | 1.06 (0.68-1.65) | .795 |
| ≥3 | 1.98 (1.44-2.73) | <.001 | 1.62 (1.14-2.31) | .007 |
| Time to PORT | —a | |||
| ≤42 days | 1 [Reference] | |||
| >42 days | 1.20 (0.92-1.56) | .183 | ||
| Total package time | ||||
| <87 days | 1 [Reference] | 1 [Reference] | ||
| ≥87 days | 1.45 (1.07-1.96) | .015 | 1.11 (0.77-1.58) | .583 |
Abbreviations: AJCC, American Joint Committee on Cancer; CoC, Commission on Cancer; CRT, chemoradiation therapy; HPV, human papillomavirus; HR, hazard ratio; PORT, postoperative radiation therapy; RT, radiation therapy.
Dropped out of final multivariable model.
A sensitivity analysis excluding patients who received targeted therapy (eg, cetuximab) concurrent with adjuvant RT was performed (see Supplemental Table 2 in the online version of the article). Adjuvant CRT was still not associated with improved OS on univariable (HR, 0.92; 95% CI, 0.69-1.24) or multivariable (aHR, 0.85; 95% CI, 0.60-1.20) analysis (see Supplemental Table 3 in the online version of the article).
Discussion
The prognostic significance and therapeutic implications of ENEmi for patients with HPV-negative HNSCC remain poorly characterized. We used a robust nationally representative data set with a precisely defined cohort of patients with surgically treated HPV-negative HNSCC to show that (1) ENEmi is associated with worse OS than ENE-negative disease and (2) adding chemotherapy concurrent with adjuvant RT is not associated with improved OS for patients with ENEmi.
Prognostic Significance of ENEmi
Prior studies evaluating the prognostic significance of ENEmi for HPV-negative HNSCC have been mixed in their findings.10-12,16 Our data showing that ENEmi is associated with worse OS than node-positive HNSCC without ENE are in accordance with those of de Almeida et al,10 who also used the AJCC/CAP definition of ENEmi (≤2 mm). Interestingly, they found no difference among DFS, locoregional control, and distant control between patients with and without ENEmi (oncologic end points not available for analysis in the NCDB). Brasilino de Carvalho,16 however, demonstrated that ENEmi (defined as microscopically, but not grossly, identifiable ENE) was not associated with increased recurrence relative to pN+ without ENE. Similarly, Wreesmann et al12 showed that ENEmi (defined as ≤1.7 mm) was not associated with worse disease-specific survival than pN+ without ENE. Finally, Prabhu et al,11 using a 0 to 4 grading scale of ENE derived from patients with HPV-related OPSCC, showed that grade 1 to 3 ENE was not associated with worse OS than no ENE (grade 0).14 The inconsistent relationship of ENEmi with prognosis across these retrospective cohort studies may be explained in part by the varied definitions of ENEmi as well as the complexity of ENE and its association with other factors reflective of metastatic nodal burden such as size, number, density, and anatomic location (level in the neck).6,7,31 The current CAP and AJCC standardization for the collecting and reporting of ENEmi will hopefully improve consistency across future study designs and clarify the prognostic significance of ENEmi.
Concurrent Chemoradiation for ENEmi
The results of 2 landmark randomized controlled trials demonstrated a benefit in terms of OS (EORTC 22931) and recurrence-free survival (RFS) (RTOG 9501) for adjuvant CRT with high-dose bolus cisplatin relative to RT for patients with ENE,6,7,9 although the benefit for RFS and locoregional control did not persist in the long-term follow-up of RTOG 9501.32 In addition, some have raised concerns about methodological issues with the post hoc pooled analysis of EORTC 22931 and RTOG 9501.33 Nevertheless, these collective trials established adjuvant CRT as standard of care for patients with ENE per National Comprehensive Cancer Network guidelines and the basis of high-risk surgically resected HNSCC for subsequent cooperative group trials.8,34,35 However, because of the significant morbidity associated with cytotoxic chemotherapy concurrent with RT that was also evident in the long-term follow-up of RTOG 9501,36 there has been an intense effort to define precisely the subset of high-risk patients who benefit from CRT.34,35,37-39 Because ENE was assessed in a dichotomous fashion (present/absent) in these landmark trials, whether the oncologic benefit of adjuvant CRT applies equally regardless of the extent of ENE (microscopic or macroscopic) is not known. Our results add to the emerging body of literature suggesting that for patients with HPV-negative HNSCC with ENEmi, adding chemotherapy concurrent with adjuvant RT does not improve OS.10
In addition to refining the definition of therapeutically relevant ENE, other attempts to deliver risk-adjusted, personalized adjuvant therapy may include evaluating other adverse pathologic features, molecular profiling of tumors and/or resection margins, and immune profiling of tumor-host microenvironments.40 Data from Zumsteg et al41 suggest that adjuvant CRT relative to RT only improves OS in patients with ENE and/or positive margins when there are ≥6 positive LNs. Expanding upon the negative prognostic role that p53 expression may have on time to recurrence and OS,42 a subset analysis of the RTOG 0234 assessing the effect of TP53 mutational status on response to adjuvant CRT demonstrated that patients with low-risk molecular profiles achieved improved survival with adjuvant docetaxel-based CRT vs cisplatin-based CRT.43
Although we found that patients with ENEmi do not have an OS benefit from adjuvant CT over RT, our findings do not suggest deescalation from the current standard of clinical care (cisplatin-based chemotherapy concurrent with adjuvant RT).8 Rather, our findings, which were derived from a retrospective, nonrandomized study design, are hypothesis generating and may assist future design of clinical trials and large-scale multicentric studies. Examination of gradations of traditional high-risk factors through the additional post hoc analyses of prior randomized controlled trials may also help define intermediate- vs high-risk populations as well as clarify the oncologic benefit of different adjuvant therapy regimens for patients with ENEmi.
Limitations
Although this was a methodologically rigorous study using a precisely defined cohort in a national sample of patients to address an ongoing area of clinical uncertainty, it has numerous limitations. A large proportion of potentially eligible patients (45%) were excluded due to missing data regarding extent of ENE within the NCDB. In addition, studies have shown substantial inter- and intrarater reliability of grading extent of ENE between head and neck pathologists.44 Variable and discordant grading of ENE likely exists within the NCDB as well as in multi-institutional studies without central pathologic review. In addition, because data for this study were collected prior to the initiation of AJCC 8, the reliability of ENEmi grading is less clear, and a high proportion of patients without extent of ENE reported (45% of our potential sample) were excluded from the sample. Future studies with a higher proportion of patients staged using AJCC 8 will hopefully result in fewer exclusions due to missing data. The nonrandomized allocation of subjects to RT or CRT introduces a significant source of bias for interpreting the finding that CRT does not improve OS. Although we attempted to control for this bias by adjusting for potential confounding variables, it is likely that residual unmeasured confounding exists, which could explain the lack of improved OS seen in the adjuvant CRT arm. Because of the importance of high-quality RT to oncologic outcomes, we limited our secondary analysis to patients with ≥60 Gy data to ensure optimal RT dosing as prescribed by cooperative group trials.34 However, data regarding specific intensity-modulated radiation therapy fields are lacking in our study, are known to have a strong association with oncologic outcomes, and could contribute to systematic differences between the treatment groups.45 Furthermore, the type of cytotoxic systemic therapy is not reported in the NCDB. We performed a sensitivity analysis to exclude patients receiving targeted therapy (eg, cetuximab) after 2013 (when anti–epidermal growth factor receptor [EGFR]–based therapies began to be classified separately from cytotoxic chemotherapies in the NCDB30); however, patients with concurrent EGFR- and RT-based therapy were likely included in our cohort for patients diagnosed prior to 2013 and may have contributed to the observed lack of OS benefit seem with CRT.27-29 The amount of cisplatin that patients receive is also not reported in the NCDB. Given the strong association between cisplatin doses ≥200 mg/m2 and survival, it is possible that the lack of survival benefit seen in the CRT arm is due to inadequate cisplatin dosing.46,47 Although we tested for effect modification of the relationship between CRT and OS by key prognostic variables to evaluate whether the association of CRT with OS differed in subgroups of patients with ENEmi, variables such as smoking history, performance status, and perineural invasion are not available in the NCDB, may moderate the impact of CRT on oncologic outcomes, and should be explored in future studies. In addition, “close” negative margin status was not routinely identified, limiting interpretation margin status on survival. All patients were treated at CoC-accredited institutions; however, we did not control for treatment volume, which is known to be associated with outcomes.41 Also, as tumors of the various subsites encompassed by HNSCC can behave distinctly, future studies should explore whether the prognostic significance of ENEmi and the benefit of adding concurrent chemotherapy with RT differs between subsites. Furthermore, although RTOG 9501 demonstrated a significant RFS benefit with CRT relative RT in patients with ENE or positive margins,7 we only evaluated the prognostic and therapeutic implications of ENEmi relative to OS because other survival end points are unavailable in the NCDB. It is therefore possible that the addition of chemotherapy to adjuvant therapy may improve other oncologically relevant measures (eg, DFS, disease-specific survival, patterns of failure). Future studies should explore the benefit of adjuvant CRT for ENEmi relative to these other end points. Last, beyond N-category and number of metastatically involved LNs, other factors may be reflective of regional metastatic burden. Future studies may also consider analyzing whether adjuvant CRT would be beneficial for patients with ENEmi and additional adverse features such as metastatic LNs in level 4/5 and/or high LN density.6,7,31
Conclusions
For patients with HPV-negative HNSCC, our data suggest that ENEmi is associated with worse OS than pN+ without ENE following margin-negative surgery and neck dissection. However, for patients with ENEmi, adjuvant CRT may not be associated with improved OS relative to RT. Further investigations are warranted to evaluate how to best define high-risk ENE and determine optimal adjuvant therapy paradigms.
Supplementary Material
Acknowledgments
Funding source: Evan M. Graboyes reports grant funding from the National Cancer Institute and the Doris Duke Charitable Foundation outside of the submitted work. David M. Neskey reports grant funding from the National Institute of Dental and Craniofacial Research outside of the submitted work. This work was supported by grant P30CA138313 from the National Cancer Institute to the Biostatistics Shared Resource of the Hollings Cancer Center. The funding organizations had no influence on the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Footnotes
Competing interests: John M. Kaczmar reported receiving personal fees from Regeneron outside the submitted work.
Sponsorships: None.
This article was presented at the AAO-HNSF 2020 Virtual Annual Meeting & OTO Experience, September 13-October 25, 2020.
Supplemental Material
Additional supporting information is available in the online version of the article.
References
- 1.Leemans CR, Tiwari R, Nauta JJ, van der Waal I, Snow GB. Regional lymph node involvement and its significance in the development of distant metastases in head and neck carcinoma. Cancer. 1993;71(2):452–456. [DOI] [PubMed] [Google Scholar]
- 2.Dünne AA, Müller HH, Eisele DW, Keßel K, Moll R, Werner JA. Meta-analysis of the prognostic significance of perinodal spread in head and neck squamous cell carcinomas (HNSCC) patients. Eur J Cancer. 2006;42(12):1863–1868. [DOI] [PubMed] [Google Scholar]
- 3.An Y, Park HS, Kelly JR, et al. The prognostic value of extranodal extension in human papillomavirus–associated oropharyngeal squamous cell carcinoma. Cancer. 2017;123(14):2762–2772. [DOI] [PubMed] [Google Scholar]
- 4.Sinha P, Lewis JS Jr, Piccirillo JF, Kallogjeri D, Haughey BH. Extracapsular spread and adjuvant therapy in human papillomavirus–related, p16-positive oropharyngeal carcinoma. Cancer. 2012;118(14):3519–3530. [DOI] [PubMed] [Google Scholar]
- 5.Amin MB, Edge S, Greene F, et al. AJCC Cancer Staging Manual. 8th ed. Springer International, American Joint Commission on Cancer; 2017. [Google Scholar]
- 6.Bernier J, Domenge C, Ozsahin M, et al. Postoperative irradiation with or without concomitant chemotherapy for locally advanced head and neck cancer. N Engl J Med. 2004;350(19):1945–1952. [DOI] [PubMed] [Google Scholar]
- 7.Cooper JS, Pajak TF, Forastiere AA, et al. Postoperative concurrent radiotherapy and chemotherapy for high-risk squamouscell carcinoma of the head and neck. N Engl J Med. 2004;350(19):1937–1944. [DOI] [PubMed] [Google Scholar]
- 8.National Comprehensive Cancer Network. Head and neck cancers (version 2.2020). Accessed January 8, 2020. https://www.nccn.org/professionals/physician_gls/pdf/head-and-neck_blocks.pdf
- 9.Bernier J, Cooper JS, Pajak TF, et al. Defining risk levels in locally advanced head and neck cancers: a comparative analysis of concurrent postoperative radiation plus chemotherapy trials of the EORTC (#22931) and RTOG (# 9501). Head Neck. 2005;27(10):843–850. [DOI] [PubMed] [Google Scholar]
- 10.de Almeida JR, Truong T, Khan NM, et al. Treatment implications of postoperative chemoradiotherapy for squamous cell carcinoma of the oral cavity with minor and major extranodal extension. Oral Oncol. 2020;110:104845. [DOI] [PubMed] [Google Scholar]
- 11.Prabhu RS, Hanasoge S, Magliocca KR, et al. Extent of pathologic extracapsular extension and outcomes in patients with nonoropharyngeal head and neck cancer treated with initial surgical resection. Cancer. 2014;120(10):1499–1506. [DOI] [PubMed] [Google Scholar]
- 12.Wreesmann VB, Katabi N, Palmer FL, et al. Influence of extracapsular nodal spread extent on prognosis of oral squamous cell carcinoma. Head Neck. 2016;38(suppl 1):E1192–E1199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bullock MJ, Beitler JJ, Carlson DL, et al. Data set for the reporting of nodal excisions and neck dissection specimens for head and neck tumors: explanations and recommendations of the guidelines from the International Collaboration on Cancer Reporting. Arch Pathol Lab Med. 2018;143(4):452–462. [DOI] [PubMed] [Google Scholar]
- 14.Lewis JS, Carpenter DH, Thorstad WL, Zhang Q, Haughey BH. Extracapsular extension is a poor predictor of disease recurrence in surgically treated oropharyngeal squamous cell carcinoma. Mod Pathol. 2011;24(11):1413–1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.College of American Pathologists. Protocol for the examination of specimens from patients with cancers of the lip and oral cavity. Published 2017. Accessed June 14, 2020. https://documents.cap.org/protocols/cp-headandneck-lip-oralcavity-17protocol-4001.pdf
- 16.Brasilino de Carvalho M Quantitative analysis of the extent of extracapsular invasion and its prognostic significance: a prospective study of 170 cases of carcinoma of the larynx and hypopharynx. Head Neck. 1998;20(1):16–21. [DOI] [PubMed] [Google Scholar]
- 17.American College of Surgeons. National Cancer Database. Published 2020. Accessed June 25, 2020. https://www.facs.org/quality-programs/cancer/ncdb
- 18.Janz TA, Graboyes EM, Nguyen SA, et al. A comparison of the NCDB and SEER database for research involving head and neck cancer. Otolaryngol Head Neck Surg. 2019;160(2):284–294. [DOI] [PubMed] [Google Scholar]
- 19.Fritz A International Classification of Diseases for Oncology: ICD-O. 3rd ed. World Health Organization; 2000. [Google Scholar]
- 20.Divi V, Chen MM, Nussenbaum B, et al. Lymph node count from neck dissection predicts mortality in head and neck cancer. J Clin Oncol. 2016;34(32):3892–3897. [DOI] [PubMed] [Google Scholar]
- 21.American Joint Committee on Cancer. Collaborative stage data collection system. Accessed June 25, 2020. https://cancer-staging.org/cstage/schema/Pages/version0205.aspx
- 22.Graboyes EM, Kompelli AR, Neskey DM, et al. Association of treatment delays with survival for patients with head and neck cancer: a systematic review. JAMA Otolaryngol Head Neck Surg. 2019;145(2):166–177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tribius S, Donner J, Pazdyka H, et al. Survival and overall treatment time after postoperative radio(chemo)therapy in patients with head and neck cancer. Head Neck. 2016;38(7):1058–1065. [DOI] [PubMed] [Google Scholar]
- 24.Harris JP, Chen MM, Orosco RK, Sirjani D, Divi V, Hara W. Association of survival with shorter time to radiation therapy after surgery for US patients with head and neck cancer. JAMA Otolaryngol Head Neck Surg. 2018;144(4):349–359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hosmer DW Jr, Lemeshow S, Sturdivant RX. Applied Logistic Regression. John Wiley; 2013. [Google Scholar]
- 26.Heinze G, Wallisch C, Dunkler D. Variable selection—a review and recommendations for the practicing statistician. Biometrical J. 2018;60(3):431–449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Galloway TJ, Ridge JA. Treatment of head and neck cancer—sometimes, less is more. JAMA Otolaryngol Head Neck Surg. 2019;145(12):1167–1169. [DOI] [PubMed] [Google Scholar]
- 28.Gillison ML, Trotti AM, Harris J, et al. Radiotherapy plus cetuximab or cisplatin in human papillomavirus-positive oropharyngeal cancer (NRG Oncology RTOG 1016): a randomised, multicentre, non-inferiority trial. Lancet. 2019;393(10166):40–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Saraswathula A, Chen MM, Colevas AD, Divi V. Assessing care value for older patients receiving radiotherapy with or without cisplatin or cetuximab for locoregionally advanced head and neck cancer. JAMA Otolaryngol Head Neck Surg. 2019;145(12):1160–1167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.American College of Surgeons. PUF data dictionary items. Accessed June 25, 2020. http://ncdbpuf.facs.org/node/259
- 31.Cheraghlou S, Otremba M, Kuo Yu P, Agogo GO, Hersey D, Judson BL. Prognostic value of lymph node yield and density in head and neck malignancies. Otolaryngol Head Neck Surg. 2018;158(6):1016–1023. [DOI] [PubMed] [Google Scholar]
- 32.Cooper JS, Zhang Q, Pajak TF, et al. Long-term follow-up of the RTOG 9501/intergroup phase III trial: postoperative concurrent radiation therapy and chemotherapy in high-risk squamous cell carcinoma of the head and neck. Int J Radiat Oncol Biol Phys. 2012;84(5):1198–1205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sinha P, Piccirillo JF, Kallogjeri D, Spitznagel EL, Haughey BH. The role of postoperative chemoradiation for oropharynx carcinoma: a critical appraisal of the published literature and National Comprehensive Cancer Network guidelines. Cancer. 2015;121(11):1747–1754. [DOI] [PubMed] [Google Scholar]
- 34.Harari PM, Harris J, Kies MS, et al. Postoperative chemoradiotherapy and cetuximab for high-risk squamous cell carcinoma of the head and neck: Radiation Therapy Oncology Group RTOG-0234. J Clin Oncol. 2014;32(23):2486–2495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhang QE, Wu Q, Harari PM, Rosenthal DI. Randomized phase II/III confirmatory treatment selection design with a change of survival end points: statistical design of Radiation Therapy Oncology Group 1216. Head Neck. 2019;41(1):37–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kearney A, Cavanagh PW. Acute and long-term effects of chemoradiation therapy in head and neck cancer. In: Doyle PC, ed. Clinical Care and Rehabilitation in Head and Neck Cancer. Springer International; 2019:331–349. [Google Scholar]
- 37.Yan F, Li H, Kaczmar JM, et al. Evaluating adjuvant therapy with chemoradiation vs radiation alone for patients with HPV-negative N2a head and neck cancer. JAMA Otolaryngol Head Neck Surg. 2020;146(12):1109–1119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Howard FM, Kochanny S, Koshy M, Spiotto M, Pearson AT. Machine learning-guided adjuvant treatment of head and neck cancer. JAMA Net Open. 2020;3(11):e2025881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Trifiletti DM, Smith A, Mitra N, et al. Beyond positive margins and extracapsular extension: evaluating the utilization and clinical impact of postoperative chemoradiotherapy in resected locally advanced head and neck cancer. J Clin Oncol. 2017;35(14):1550–1560. [DOI] [PubMed] [Google Scholar]
- 40.Maghami E, Koyfman SA, Weiss J. Personalizing postoperative treatment of head and neck cancers. Am Soc Clin Oncol Educ Bk. 2018;(38):515–522. [DOI] [PubMed] [Google Scholar]
- 41.Zumsteg Z, Luu M, Kim S, et al. Quantitative lymph node burden as a "very-high risk" factor identifying head and neck cancer patients benefiting from post-operative chemoradiation. Ann Oncol. 20189;30(1):76–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Shin DM, Lee JS, Lippman SM, et al. p53 Expression: predicting recurrence and second primary tumors in head and neck squamous cell carcinoma. J Natl Cancer Inst. 1996;88(8):519–529. [DOI] [PubMed] [Google Scholar]
- 43.Michikawa C, Torres-Saavedra PA, Silver NL, et al. Evolutionary action score of TP53 analysis in pathologically high-risk HPV-negative head and neck cancer from a phase II clinical trial: NRG Oncology RTOG 0234. J Clin Oncol. 2019;37(15)(suppl):6010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lewis JS Jr, Tarabishy Y, Luo J, et al. Inter- and intra-observer variability in the classification of extracapsular extension in p16 positive oropharyngeal squamous cell carcinoma nodal metastases. Oral Oncol. 2015;51(11):985–990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Peters LJ, O’Sullivan B, Giralt J, et al. Critical impact of radiotherapy protocol compliance and quality in the treatment of advanced head and neck cancer: results from TROG 02.02. J Clin Oncol. 2010;28(18):2996–3001. [DOI] [PubMed] [Google Scholar]
- 46.Pignon JP, le Maître A, Maillard E, Bourhis J. Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): an update on 93 randomised trials and 17,346 patients. Radiother Oncol. 2009;92(1):4–14. [DOI] [PubMed] [Google Scholar]
- 47.Strojan P, Vermorken JB, Beitler JJ, et al. Cumulative cisplatin dose in concurrent chemoradiotherapy for head and neck cancer: a systematic review. Head Neck. 2016;38(suppl 1):E2151–E2158. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


