Graphical abstract

Keywords: Daphne mucronata extract, DPPH, ABTS, Antioxidants, Silymarin, Phytochemicals, Liver biomarkers, Kidney biomarkers
Abbreviations: ABTS, 2, 2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); ALT, alanine aminotransferase; ALP, alkaline phosphatase; AST, aspartate aminotransferase; D. mucronata, Daphne mucronata; DPPH, 2, 2-Diphenyl,1,picrylhydrazyl; GSH, reduced glutathione; MDA, malondialdehyde; Met. Ext, Methanolic extract; NAPQI, N-acetyl-p-benzo-quineimine; OECD, Organisation for Economic Co-operation and Development; p.o., Per oral; SEM, Standard error mean; TAC, total antioxidant capacity; TFC, Total flavonoid content; TPC, Total phenolic content
Abstract
The paracetamol-induced injuries of liver and kidneys in animals are mostly used to screen out the hepato and nephroprotective effect of extract or other therapeutic agents. In the present study total phenolic and flavonoid contents, in vitro antioxidant, and in vivo hepato/nephroprotective (on paracetamol-induced intoxication in experimental rabbits) potentials of the Daphne mucronata leaves methanolic extract were determined. For the identification of possible phytochemicals, HPLC (high performance liquid chromatography) analysis was carried out and a total of eight phenolic compounds; malic acid, gallic acid, chlorogenic acid, epigallocatechin gallate, quercetin, morin, ellagic acid, and rutin were identified. D. mucronata extract at doses of 250 and 500 mg/kg body weight were given for eight days to paracetamol intoxicated rabbits and the observed results were compared with standard Silymarin. The level of liver enzymes like aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, serum triglyceride, serum cholesterol, serum bilirubin, and kidneys biomarkers like serum urea, uric acid, and creatinine, as well as lipid peroxidation malondialdehyde contents were increased while the antioxidant enzymes like reduced glutathione and total antioxidant capacity were decreased. Furthermore, histopathological analysis of the liver and kidney tissues of control and treated groups also confirmed the hepatoprotective and nephroprotective effect of the D. mucronata which was most probably due to its high antioxidant phenolic and flavonoid phytoconstituents.
1. Introduction
In human body, the liver is the major organ that regulates the body homeostasis and control the metabolism of carbohydrates, proteins and fats. In other words liver has a role in almost all body functions in terms of biochemical and metabolic pathways, defence against various diseases, supply of nutrients, involved in reproduction and provision of energy (Rui, 2014, Thorne et al., 2020). In spite of the mentioned functions liver also acts as a storehouse of many valuable biochemical required for different metabolic pathways. In today's world the liver diseases are considered lethal diseases due to causalities caused every year around the globe. In developing countries, the main causes of liver diseases are; hepatitis viruses, environmental poisons, drug, and alcohol habits. Some medicines like antibiotics, and other chemotherapeutics (paracetamol etc) also causes hepatotoxicity (Saleem et al., 2010).
Paracetamol, an analgesic and antipyretic drug, is widely used as a hepatotoxic drug in many experimental animals as it causes moderate to severe lobular necrosis. High doses of paracetamol (200 mg/kg and above) is a major cause of liver toxicity and is also a cause of nephrotoxicity (Rehman et al., 2013). Paracetamol overdoses in 10–40% of patients, causes severe liver necrosis and in <2% patients causes renal failure. Its ingestion above therapeutic levels causes acute toxicity in liver, as 90% of it is converted into sulphate and glucuronoid conjugates while the remainder is converted to N-acetyl-p-benzo-quineimine (NAPQI) through cytochrome P450; a highly reactive and toxic intermediate (Kassem et al., 2013). In normal condition it is detoxified through conjugation with hepatic glutathione. Over doses causes the depletion of glutathione (GSH) pool which is an antioxidant enzyme and is involved in detoxification function of the body. Liver and kidney toxicity is mediated by high levels of NAPQI (Jaeschke and Ramachandran, 2020).
The kidneys are also the most important organ of the human body that effectively filters the toxins and waste products from the blood. Kidneys play an important role to regulate our endocrine, acid-base balance, and blood pressure. The renal failure can be acute or chronic depending on exposure to toxins. In chronic state, the kidneys suffer slowly and gradually that last for years that is why renal failure is also referred to as “silent killer”. Different free radicals (hydroxyl radicals, hydrogen peroxide, superoxide anions, nitric oxide, nascent oxygen and lipid oxides) produced during metabolic processes are the main causes of liver and kidneys disorders and in cases where glutathione pool is depleted the hazardous effects are more drastic (Levey et al., 2007).
Modern synthetic drugs used to ameliorate hepatotoxicity have minor therapeutic effects and are mostly associated with renal failure. Medicinal plants on the other hands due to its compatibility with human life are more effective in such cases and are associated with no or low side effects (Nazir et al., 2020a, Nazir et al., 2020b). Many medicinal plants have shown hepatoprotective and nephroprotective effect in liver and renal lesions induced by paracetamol, gentamicin, profenofos, D galactosamine, chronic stress, cytotoxic drugs, diabetic nephropathy, chemically induced nephrolithiasis by inflammatory mediators and oxidative stress (Vargas-Pozada and Muriel, 2020, Nazir et al., 2020a, Nazir et al., 2020b).
The genus Daphne belongs to the family Thymelaeaceae, comprising of almost 90 species, found in Europe, North Africa, and Asia. Mostly these species are used in traditional Chinese and other plant based medication systems for the treatment of various diseases. Experimentally, they have demonstrated a variety of biological and pharmacological effects like antioxidant, antifungal, antibacterial, analgesic, anti-inflammatory, cytotoxic, antiviral, along with abortive and haemostatic effects (Moshiashvili et al., 2020). D. mucronata Royle is a wild shrub and is a member of this genus that is widely found in the northern regions of Pakistan and Iran (Katayoun et al., 2003). D. mucronata is considered as essential medicinal plant because of its ethnomedicinal and pharmacological uses. It is used as an active ingredient in folkloric medicines to cure various diseases (Rasool et al., 2009). The plant woody bark is recommended to cure bone diseases, relieve eye pain, while the fruits and leaves are used for the treatment of rheumatism (Ghasemi et al., 2012). The extract of the plant has traditionally been used for the treatment of infections related to skin, allergies, and cancer (Malik et al., 2019). Leaves and root extracts of D. mucronata have been used in traditional Chinese medicine to treat toothache, ulcers, rheumatism, and as purgative and abortive agent (Katayoun et al., 2003). Its boiled leaves and in ointment form they are used to treat wound infections, constipation, menstrual problems, infertility, and gynaecological complications (Mosaddegh et al., 2012). As mentioned before, extracts from different parts of the Daphne species have a wide range of pharmacological and biological activities which are due to the presence of secondary metabolites in them, suggesting that these plants could serve as a source of active ingredients that could be effectively used in the pharmaceutical, cosmetic, and food industries (Can et al., 2020).
Taking into account the importance of selected plant as depicted by various in vitro and in vivo studies along with traditional application, the present research study was designed to evaluate the hepatoprotective and nephroprotective effects of the methanolic leaf extract of D. mucronata against hepato and nephrotoxicity induced by paracetamol in rabbits. The observed effects were correlated with phenolic and flavonoid contents. In addition HPLC profiling was done to identify possible phenolic and flavonoid compounds where malic acid, gallic acid, chlorogenic acid, epigallocatechin gallate, quercetin, morin, ellagic acid, and rutin were identified through comparison with standards and those reported in literature.
2. Material and methods
2.1. Chemicals and reagents
Antioxidant chemicals such as DPPH, ABTS, ascorbic acid, antioxidant standards, Folin-Ciocalteu regent, and silymarin, were purchased from Sigma-Aldrich (St. Louis, MO, USA); while normal saline solution (Utsoka Pharma, Las Bela Baluchistan, Pakistan), liver profile test kits (Human, Hamburg, Germany), and kidney profile test kits (Biomed: Germany; diagnostic) of the mentioned firms (in brackets) were used in this study. Methanol was purchased from Merck, Darmstadt, Germany.
2.2. Preparation of D. mucronata methanolic extract
The plant leaves were collected from local area and after collection, they were cleaned and kept on a clean paper for 20 days. The shade dried leaves were chopped into small size pieces through mortar and pestle then crushed to fine powder by mechanical grinder. Approximately 8 kg of powder sample was subjected to maceration in 80% methanol for 14 days with periodical shaking. Filtration was carried out through muslin cloth followed by filtration through Whattman filter paper. The filtrates were concentrated into a semisolid mass using rotary evaporator (Heidolph Laborota 4000, Schwabach, Germany) under reduced pressure and then completely dried through lyophiliser (Nazir et al., 2018a, Nazir et al., 2018b). The dried mass obtained was 280 g.
2.3. Assessment of total phenolic content
The total phenolic contents (TPC) in the methanolic extract (Met. Ext) were determined using previously reported method (Shirazi et al., 2014). Where Met. Ext (100 µL), distilled water (500 µL), Folin-Ciocalteu reagent (100 µL) and 7% sodium carbonate (1000 µL) were mixed thoroughly and after 90 min absorbance was recorded at 760 nm using UV-Spectrophotometer. TPC in plant samples were measured from the calibration curve of standard Gallic acid and expressed as mg of GAE/gm of dry sample.
2.4. Assessment of total flavonoid contents (TFC)
The TFC were also estimated in D. mucronata leaves Met. Ext using previously reported assay (Shirazi et al., 2014). Quercetin was used as a standard and the enumerated contents were expressed as mg of Quercetin equivalent (mg QE/g) per gram of dry sample of leaves extract. Different dilutions of quercetin were used to draw a calibration curve. Extract (100 µL) mixed with distilled water (500 µL), 5% sodium nitrate (100 µL), 10% aluminium chloride (150 µL) and 1 M sodium hydroxide (200 µL) were mixed thoroughly and after 5 min absorbance at 510 nm was recorded using UV Spectrophotometer.
2.5. Phytochemical profiling of extract by HPLC-UV analysis
For HPLC analysis, about 1 g leaves powder was mixed with water and methanol (20 mL; 1:1 v/v) and then kept in a water bath for 1 h at 70 °C. Finally the sample was cooled and centrifugation was carried out for 10 min at 4000 rpm. The supernatants were then filtered through Whattman filter paper. About 2 mL of it was taken in a properly labelled HPLC vial. Agilent zorbax eclipse (XDB-C18) column was used for the separation of phytochemicals and the resultant chromatogram was compared with that of the reference standards run on the same column (Zeb, 2015). Quantification of phytochemicals was carried out using the following single point calibration formula:
| (1) |
where: Cx = sample concentration; As = standard peak area; Ax = sample peak area; Cs = standard concentration (0.09 µg/ml).
2.6. In vitro DPPH (2, 2-diphenyle-1-picrylhyzyl) free radical scavenging potential
DPPH scavenging potential of D. mucronata leaves Met. Ext was measured using Brand-Williams method (Brand-Williams et al., 1995). DPPH (0.039 g/100 mL in methanol) solution was prepared and incubated for 30 min in dark. The stock solution of extract (1 mg/mL) was also prepared in methanol from which serial dilutions in the concentration range of 1000, 500, 250, 125, 62.5 and 31.05 µg/mL were then prepared. About 0.1 mL of each working dilution was mixed with 3 mL of DPPH solution and incubated for 30 min at 25 °C. Finally the absorbance of mixture was recorded using UV-spectrophotometer at 517 nm. Same procedure was used for ascorbic acid that was used as standard. %DPPH scavenging potential was measured using the following formula;
| (2) |
2.7. In vitro ABTS (2, 2-azinobis [3-ethylebenzthiazoline]-6-sulfonic acid) scavenging potential
According to the reported procedure (Re et al., 1999), the ABTS free radical scavenging potential was determined as well. ABTS (7 mM) and potassium per sulphate (2.45 mM) solutions were mixed to gather and incubated overnight in dark. About 300 µL of D. mucronata Met. Ext working dilutions and 3 mL of ABTS solution were mixed thoroughly and incubated for about 6 min and the absorbance of resulting mixture was recorded via double beam spectrophotometer at 745 nm. Ascorbic acid was used as positive control. The %ABTS free radical scavenging potential of extract was measured using the Eq. (2).
2.8. Animal experiments
The in vivo experiments were carried out on forty (40) healthy adults’ male rabbits (Oryctolagus cuniculus) having initial weight from 1.6 to 2.6 kg. The animals were harboured with 12 h of light–dark cycle at a constant temperature of about 25 ± 3.5 °C and relative humidity of 55 ± 10%, and acclimatized for one week prior to the experiment. The animals were provided free access to water in a well-ventilated room at animal house, University of Malakand, Khyber Pakhtunkhwa, Pakistan. The animals were fed on green vegetables along with fresh grasses. The animals were provided standard food obtainable as ad libitum (Nazir et al., 2018a, Nazir et al., 2018b). All the animal procedures were conducted according to the ARRIVE guidelines and Animal Scientific Procedure Act; UK (1986) and the approval for the current experimental protocol was taken from the Departmental Animal Ethical Committee (DAEC/2019/1).
2.9. Acute toxicity analysis of D. mucronata Met. Ext
The acute oral toxicity study on the D. mucronata leaves Met. Ext was carried out according to the Organization for Economic Co-operation and Development (OECD) guideline 423. Three animals were treated orally with a single dose of 2000 mg/kg body weight of Met. Ext, while other three with distilled water (10 mL/kg) to evaluate the toxic effects if any, in the experimental animals. Immediately after dosing, the rabbits were observed continuously for 2 h for any symptoms of toxicity (convulsions, loss of righting reflex, motor activity, muscle spasm, tremors, lacrimation, sedation, hypnosis, diarrhoea, and salivation). Animals were observed up to 14 days for any signs of toxicity or mortality. The Met. Ext remained safe and nontoxic up to the dose range of 2000 mg/kg body weight. After that the use of an additional upper dose level of ≥ 2000 mg/kg body weight (5000 mg/kg, body weight) was tested but no toxic signs were observed in the animals. Therefore, according to the guidelines of OECD, Met. Ext at dose 250 and 500 mg/kg body weight (maximum dose) that was 1/10th of 1500 and 5000 mg/kg dose were given to experimental animals in subsequent experiments to assess the hepatoprotective and nephroprotective effect of the extract (Nazir et al., 2021).
2.10. Animal grouping and dosing
The total study population that was about forty (40) rabbits were grouped into five groups of 8 animals each (Rehman et al., 2015). Each group rabbits were tagged separately for the purpose of identification. Animals of Group I were administered normal saline, p.o., for 8 days, served as normal control group. Group II: Animals were administered with paracetamol 2 g/kg body weight for 8 day. Group III served as standard control group, received paracetamol along with silymarin; a well-known standard hepatoprotective drug (50 mg/ kg p.o) for 8 days. Group IV and V were given D. mucronata Met. Ext (250 and 500 mg/kg p.o.) for 8 days along with paracetamol. Immediately after Paracetamol dosing animals were observed for 24 h for signs of toxicity (Rehman et al. 2015). The doses of tested Met. Ext solutions and animals experimental design have been presented in Table 1.
Table 1.
Experimental design for Daphne mucronata Met. Ext tretament groups used in the study.
| Group | Group category | Treatment given | Route/times of administration |
|---|---|---|---|
| I | Normal control | Normal saline (8 mL/kg) | p.o/at 8:00 am and at 8:00 pm (Twice a day) |
| II | Paracetamol control | Paracetamol (2 g/kg) | p.o/at 8:00 am and at 8:00 pm (Twice a day) |
| III | Standard control | Paracetamol (2 g/kg b.w) + Silymarin (50 mg/kg b.w) | p.o/at 8:00 am and at 8:00 pm (Twice a day) |
| IV | Extract treated group | Paracetamol (2 g/kg b.w) + Met. Ext (250 mg/kg) | p.o/at 8:00 am and at 8:00 pm (Twice a day) |
| V | Extract treated group | Paracetamol (2 g/kg b. w) + Met. Ext (500 mg/kg) | p.o/at 8:00 am and at 8:00 pm (Twice a day) |
Met.Ext, Methanolic extract, p.o., Per oral, b.w., body weight.
2.11. Hematological and serological profile of the test rabbits
After dosage completion (24 h later on day 9th), all the animals were anaesthetised by injecting anaesthetic combinations (ketamine/xylazine, IM), and euthanized by injecting sodium pentobarbital (100 mg/kg body weight, IV). Onset of sedation was achieved and surgery was performed successfully of all rabbits. Administration of sodium pentobarbital is one of the few methods of euthanasia that is listed as acceptable in laboratory rodents. The blood samples (3 mL) were taken by cardiac puncture, transferred to EDTA and non-EDTA containing tubes. The serum was separated by centrifugation for 10 min at 3,000 rpm and 37 °C, stored at 4 °C till determination of biochemical parameters (Cicero et al., 2018).
2.12. Assessment of biochemical parameters
Aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), triglyceride, cholesterol, and bilirubin levels in serum were measured using Biochemistry analyser (PS-520; Shenzhen Procan Electronics, China) as per companies’ instructions using diagnostics kits (Reactivos, GPL Barcelona, Spain). Kidneys biomarkers like serum urea, uric acid, and creatinine were measured by ROCHE Diagnostic (Reflotron® Plus instrument) kits.
2.13. Assessment of lipid peroxidation and antioxidant biomarkers
The hepatic and renal tissues collected from each rabbit group were excised, cleaned, and immediately perfused with cold saline. The tissues were homogenized in cold phosphate buffer saline (pH 7.4, 0.1 M). Then, the homogenates were filtered and centrifuged (at 3,000 rpm for 20 min). The supernatant was then stored at −80 °C until use for further biochemical analysis of lipid peroxidation and antioxidant biomarkers. Lipid peroxidation was evaluated through measurement of malondialdehyde (MDA) content in the tissues. Oxidative status was assessed by evaluation of the nonenzymatic antioxidant marker; reduced glutathione (GSH) and total antioxidant capacity (TAC) according to the reported assays (Abdel-Daim and Ghazy, 2015).
2.14. Histopathology
For histopathological examination the liver and kidney portions were immediately removed, washed thoroughly with normal saline solution to remove the blood and were processed following the standard protocols (Rehman et al., 2015). The liver and kidney tissues were fixed in formalin (10%), dehydrated with ethanol-xylene mixtures and fixed with paraffin. Tissue blocks were sectioned from 4.5 to 6 μm thickness by Microtome (ACCU-Cut® SRM™; 200 Sakura). Slides were stained with hematoxylin and eosin (H & E) dye using automatic slide stainer (Sakura Tissue-Tek® DRS™ 2000, Japan). The stained slides were cleaned properly and observed under microscope to see alteration in hepatic and kidney tissues architecture of paracetamol intoxicated and treated (Silymarin/extract) groups.
2.15. Statistical analysis
All in vitro and in vivo experiments were performed in three replicates. All results have been presented as Mean ± SEM. The Student’s t-test and one way ANOVA followed by Dunnett’s post hoc multiple comparison test was used to evaluate significance of the data obtained. P ≤ 0.05 were considered as significant.
3. Results
3.1. Total phenolic/flavonoid content
Results of TPC and TFC of D. mucronata leaves extract are presented in Fig. 1. The TPC of D. mucronata Met. Ext at various concentrations are presented in Fig. 1A. A calibration curve was drawn for gallic acid (standard) using the dilutions; 1000, 500, 250, 125 and 62.5 µg/mL to assess the TPC while standard quercetin regression curve was made for the estimation of TFC (Fig. 1C) in D. mucronata leaves extract.
Fig. 1.
Total phenolic and flavonoids content of Met. Ext of Daphne mucronata and their linear correlation with DPPH and ABTS inhibition potentials. {(A) Total Phenolic contents in Met. Ext and free radicals (DPPH & ABTS) inhibition potential at various concentration (B) Linear correlation of TPC in Met. Ext vs. % DPPH and %ABTS inhibition (C) Total flavonoids content in Met. Ext and free radicals (DPPH & ABTS) inhibition potential at various concentration (D) TFC vs. % DPPH and %ABTS inhibition}.
3.2. Correlation between TPC and TFC versus %DPPH and ABTS inhibition activity
Correlation between TPC and TFC versus antioxidant activities have been presented in Fig. 1 B & D. The highest regression coefficient value (R2 = 0.9303 and 0.953) was obtained when TPC was plotted against %DPPH and ABTS inhibition (Fig. 1B) while, the correlation coefficient for TFC against % inhibition of DPPH and ABTS were 0.9598 and 0.969 (Fig. 1D) respectively. Regression line for DPPH and ABTS also goes parallel with TPC and TFC indicating a good correlation between the contents and observed inhibition potentials.
3.3. Identification and quantification of possible phytochemicals
A typical HPL-UV chromatogram of Daphne mucronata methanolic extract (Met. Ext) is shown in Fig. 2. A total of eight antioxidant phytochemicals were identified. The detailed identification of each antioxidant with their respective peak position in chromatogram and retention time (Rt) are given in Table 2. Malic acid, gallic acid, chlorogenic acid, epigallocatechin gallate, quercetin, morin, ellagic acid, and rutin were eluted at retention time of 2.7, 4.3, 6.0, 8.0, 10.3, 12.0, 16.6, and 22.7 min with the concentration values of 1172.98, 204.91, 70.46, 708.78, 148.04, 45.0, 7.88, and 106.63 µg/ml respectively (Table 2).
Fig. 2.
HPLC chromatogram of Daphne mucronata methanolic extract (Met. Ext).
Table 2.
Possible phytochemicals identified in Daphne mucronata methanolic extract.
| Extract | Peak no | Retention time (min) | Detected phenolic compounds | Sample peak area | Standard peak area | Concentration (µg/ml) |
|---|---|---|---|---|---|---|
| Met. Ext | 1 | 2.7 | Malic acid | 4727.11 | 40.32 | 1172.98 |
| 2 | 4.3 | Gallic acid | 4004.04 | 195.4 | 204.91 | |
| 3 | 6.0 | Chlorogenic acid | 90.9 | 12.9 | 70.46 | |
| 4 | 8.0 | Epigallocatechin gallate | 5145.81 | 72.6 | 708.78 | |
| 5 | 10.3 | Quercetin | 1345.72 | 90.9 | 148.04 | |
| 6 | 12.0 | Morin | 9.0 | 2.02 | 45.0 | |
| 7 | 16.6 | Ellagic acid | 251.57 | 319.24 | 7.88 | |
| 8 | 22.7 | Rutin | 238.86 | 22.4 | 106.63 |
Met.Ext, Methanolic extract.
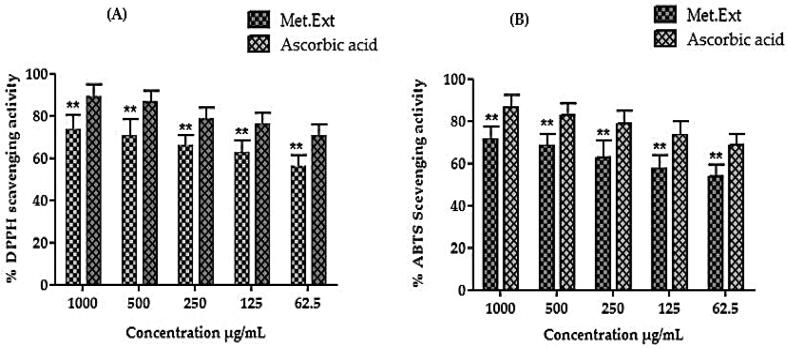
3.4. In vitro DPPH free radical scavenging potential
The antioxidant potential of D. mucronata Met. Ext displayed a dose dependent response against DPPH free radical. The Met. Ext showed 75 ± 0.55, 71 ± 0.34, 68 ± 0.99, and 63 ± 0.72, and 57 ± 0.79% inhibitions at concentrations of 1000, 500, 250, 125 and 62.5 μg/mL (Table S1 & Fig. 3A) respectively. While standard ascorbic acid caused 90 ± 0.33, 87 ± 0.42, 81 ± 0.36, 77 ± 0.41 and 71 ± 0.75% inhibitions at 1000, 500, 250, 125 and 62.5 µg/mL concentration against DPPH radical respectively.
Fig. 3.
% Free radical (DPPH and ABTS) scavenging potentials of Met. Ext of Daphne mucronata Royle. {(A) %DPPH scavenging activity (B) %ABTS scavenging activity (** indicates that values were significantly different, p < 0.001)}.
3.5. In vitro ABTS free radical scavenging potential
The result of %ABTS inhibition potential of D. mucronata leaves Met. Ext are shown in Table S1 & Fig. 3B. The highest observed %inhibition of Met. Ext was 72 ± 0.51 at 1000 μg/mL concentration. Ascorbic acid was used as a standard.
3.6. Hepatoprotective effect of D. mucronata leaves methanolic extract
Hepatotoxicity can be inferred from the increase levels of the biochemical parameter such as serum ALT, AST, ALP, triglyceride, cholesterol and bilirubin. Treatment of rabbits with paracetamol resulted in severe damage to liver which is apparent from high levels of serum ALT, AST, ALP, triglyceride, cholesterol and bilirubin as shown in Table 3 and Fig. 4. Pre-treatment of rabbits with silymarin standard, significantly (***:P < 0.001) reduced the elevated levels of serum ALT, AST, ALP, triglyceride, cholesterol and bilirubin (46 ± 1.5, 82 ± 3.7, 69 ± 1.2, 44 ± 4.0, 34 ± 1.7, 0.5 ± 0.1% respectively) that were induced by paracetamol (Table 3). D. mucronata Met. Ext displayed significant (**: p < 0.01, ***: p < 0.001) decrease (36 ± 6.3, 66 ± 6.5, 67 ± 4.7, 50 ± 3.7, 40 ± 4.1, and 0.6 ± 0.2) in all the tested parameters (serum ALT, AST, ALP, triglyceride, cholesterol and bilirubin level, respectively) at 500 mg/kg dose (Fig. 4 A, B, C, D, E, and F) as compared to the paracetamol-treated group. Pronounced dose dependency has been observed for the tested doses of selected plant extract which were comparable to the animal group treated with standard silymarin.
Table 3.
Effect of Met. Ext of Daphne mucronata on the level of biochemical parameters in paracetamol-induced hepatotoxicity in rabbits.
| Groups | Dose |
Liver-related parameters with % change values |
|||||
|---|---|---|---|---|---|---|---|
| ALT (U/L) | AST (U/L) | ALP(U/L) | Serum Triglyceride (mg/dL) | Serum Cholesterol(mg/dL) | Serum bilirubin (mg/dL) | ||
| Normal control | 8.0 (mL/kg) | 38 ± 2.4*** | 78 ± 1.2*** | 66 ± 0.5*** | 40 ± 0.8*** | 32 ± 2.0*** | 0.4 ± 0.2*** |
| Paracetamol control | 2.0 (gm/Kg) | 94 ± 6.4 | 124 ± 2.3 | 185 ± 1.2 | 75 ± 6.3 | 68 ± 3.8 | 1.52 ± 0.3 |
| Silymarin + Paracetamol | 50 (mg/Kg) | 46 ± 1.5*** | 82 ± 3.7*** | 69 ± 1.2*** | 44 ± 4.0*** | 34 ± 1.7*** | 0.5 ± 0.1*** |
| Met. Ext + Paracetamol | 250 (mg/Kg) | 77 ± 3.2** | 71 ± 3.1*** | 98 ± 6.1*** | 69 ± 6.1 ns | 35 ± 1.2*** | 0.5 ± 0.1*** |
| 500 (mg/Kg) | 36 ± 6.3*** | 66 ± 6.5*** | 67 ± 4.7*** | 50 ± 3.7** | 40 ± 4.1*** | 0.6 ± 0.2*** | |
Each value is presented as mean ± SEM, n = 8 animals. Comparisons have been made between Normal control versus Paracetamol control using student t-test (***p < 0.001) and significant differences (*p < 0.05**, p < 0.01***, p < 0.001) were found between the treated groups (Silymarin/Met. Ext) versus Paracetamol control, determined through One way ANOVA followed by Dunnett’s posthoc multiple comparison test.
Fig. 4.
Effects of Met. Ext of Daphne mucronata on the level of liver serum biochemical parameters in paracetamol-induced hepatotoxicity in rabbits {(A) serum level of ALT, (B) serum level of AST, (C) serum level of ALP, (D) serum level of Triglyceride, (E) serum level of cholesterol (F), and serum level of bilirubin in paracetamol-intoxicated rabbits model: Values are presented as mean ± SEM, n = 8. Significant difference (***p < 0.001) between the normal control versus paracetamol control have been determined using student t-test and **p < 0.01 and ***p < 0.001 indicates that values are significantly different from that of the treated groups (Silymarin/Met. Ext) in comparison to paracetamol control that have been estimated through One way ANOVA followed by Dunnett's posthoc test}.
3.7. Nephroprotective effect of D. mucronata leaves methanolic extract
Results of nephroprotective effects of D. mucronata Met. Ext on paracetamol intoxicated rabbits are presented in Table 4, and in Fig. 5. A significant increase was observed in kidneys biomarkers like serum urea, uric acid and creatinine (Fig. 5A,B,C) in paracetamol intoxicated animals as compared to control (normal saline) and treated groups (silymarin/extract). Pre-treatment with silymarin (50 mg/kg body weight) reduced the elevated levels of serum urea, uric acid and creatinine. The most effective nephroprotective results were observed for Met. Ext at dose of 500 mg/kg body weight where a significant (**P < 0.01, ***P < 0.001) reduction was observed in serum urea (42 ± 1.2**), uric acid (3.2 ± 4.0***), and creatinine (0.7 ± 0.1 ***) levels.
Table 4.
Effects of Met. Ext of Daphne mucronata on the level of kidney serum biochemical parameters in paracetamol-induced hepatotoxicity in rabbits.
| Groups | Dose |
Kidney related parameters with % change values |
||
|---|---|---|---|---|
| Serum urea (mg/dL) | Serum uric acid (mg/dL) | Serum creatinine (mg/dL) | ||
| Normal control | 8.0 (mL/kg) | 37 ± 0.5*** | 3.2 ± 1.1*** | 0.4 ± 0.1*** |
| Paracetamol control | 2.0 (gm/Kg) | 92 ± 2.3 | 8.2 ± 6.3 | 2.5 ± 0.3 |
| Silymarin + Paracetamol | 50 (mg/Kg) | 39 ± 3.8*** | 3.7 ± 3.7*** | 0.6 ± 0.1*** |
| Met. Ext + Paracetamol | 250 (mg/Kg) | 61 ± 4.4* | 4.8 ± 6.3** | 0.8 ± 0.2*** |
| 500 (mg/Kg) | 42 ± 1.2** | 3.2 ± 4.0*** | 0.7 ± 0.1 *** | |
Each value has been presented as mean ± SEM, n = 8 animals. Comparisons have been made between normal control versus paracetamol control groups using student t-test (***p < 0.001) while significant differences (* p < 0.05,** p < 0.01, ***p < 0.001) were found between the treated groups (Silymarin/Met. Ext) in comparison to paracetamol control group determined through One way ANOVA followed by Dunnett’s posthoc multiple comparison test.
Fig. 5.
Effects of Met. Ext of Daphne mucronata on the level of kidney serum biochemical parameters in paracetamol-induced nephrotoxicity in rabbits. {(A) serum level of urea (B), serum level of uric acid, and (C), serum level of creatinine in paracetamol-intoxicated rabbits model: Values are presented as mean ± SEM, n = 8. Significant difference (***p < 0.001) between the normal control group versus paracetamol control group have been observed that were determined using student t-test. *p < 0.05, **p < 0.01, and ***p < 0.001 indicates that values are significant and have been determined for treated groups (Silymarin/Met. Ext) versus paracetamol control group using One way ANOVA followed by Dunnett’s posthoc multiple comparison test}.
3.8. Hepatic lipid peroxidation and antioxidant status
Treatment of rabbits with paracetamol significantly (P ≤ 0.001) increased the hepatic lipid peroxidation marker; MDA level as compared to the normal control group (Fig. 6A). Moreover, paracetamol significantly (P ≤ 0.001) reduced the liver antioxidant capacity as indicated by declines in GSH (Fig. 6B) and TAC levels (Fig. 6C) compared to the normal control group. Treatment with silymarin (50 mg/Kg) and Met. Ext (250 & 500 mg/kg) significantly (**P ≤ 0.01; ***P ≤ 0.001) reinstated the antioxidant capacity in the rabbit liver homogenate to the normal level, that restored the GSH and TAC levels comparable to that of the control group values. In the silymarin (50 mg/Kg) and Met. Ext (250 & 500 mg/kg) treated groups, the MDA level was significantly (P ≤ 0.001) decreased, while that of the antioxidant markers like GSH and TAC were significantly (**P ≤ 0.01; ***P ≤ 0.001) high in comparison with the paracetamol treated group. These results indicates the synergistic hepatoprotective and antioxidant effects of D. mucronata leaf extract (Fig. 6).
Fig. 6.
Effects of Daphne mucronata leaf Met. Ext and silymarin on liver and kidney lipid peroxidation and antioxidant biomarkers in paracetamol-induced toxicity in rabbits. {Lipid peroxidation and antioxidant biomarkers in liver: (A) MDA level (B) GSH level (C) TAC level. Lipid peroxidation and antioxidant biomarkers in kidney: (D) MDA level (E) GSH level (F) TAC level. Values are presented as mean ± SEM, n = 8. Significant differences (***p < 0.001) between the normal control and paracetamol treated groups were observed which were estimated using student t-test. **p < 0.01 and ***p < 0.001 indicated that values were significantly different for the treated groups (silymarin/Met. Ext) in comparison to paracetamol control group determined through One way ANOVA followed by Dunnett’s posthoc multiple comparison test}.
3.9. Renal lipid peroxidation and antioxidant status
The effects of paracetamol, silymarin (50 mg/kg), and Met. Ext (250 & 500 mg/kg) supplementation on the renal lipid peroxidation and antioxidant activities are demonstrated in Fig. 6. Parallel to results of hepatic MDA and oxidative status, paracetamol intoxication significantly (***P ≤ 0.001) elevated the renal MDA level (Fig. 6A) and reduced GSH (Fig. 6B) and TAC levels (Fig. 6C) as compared to the normal control group. In the same manner, daily administration of silymarin (50 mg/Kg) and Met. Ext at the dose of 250 mg/kg and 500 mg/kg, significantly (***P ≤ 0.001) restored the antioxidant activities to the normal levels in liver. Silymarin and Met. Ext (250 & 500 mg/kg) significantly (***P ≤ 0.001) reduced MDA level and increased GSH and TAC levels significantly (P ≤ 0.01; P ≤ 0.001) as compared to the paracetamol treated group.
3.10. Histopathological evaluation of the liver tissues
Histopathological slides of the animals liver tissues treated with D. mucronata leaves Met. Ext and paracetamol are presented in Fig. 7. Animals of Group I (control group) shows normal liver parenchyma and the endothelia linings of central veins with normal morphology and no evidence of necrosis. The liver sections in group II animals treated with paracetamol, reveals necrosis, inflammation, bile duct proliferation, and cholestasis. Group III liver histopathology slides shows that the animals treated with standard drug silymarin shows a normal liver architecture. Histopathology of group IV animals, treated with Met. Ext (250 mg/kg) shows a mild bile duct ingestion and scattered inflammatory cells. Improved and normal histological architecture were seen in animals of group V treated with Met. Ext (500 mg/kg).
Fig. 7.
Effects of Met. Ext of Daphne mucronata on histopathology studies in paracetamol-induced hepatotoxicity in rabbits. {(A) Liver of control group displaying normal liver parenchyma and the endothelia linings of central veins had normal morphology with no evidence of necrosis, (B) Section of liver parenchyma from a rabbit of Group II treated with paracetamol reveals necrosis, inflammation, and bile duct proliferation, (C) Histopathology of a liver from Group III treated with standard Silymarin shows almost recovery of the normal architecture with normal hepatocytes arrangement, (D) Histopathology of a liver from Group IV treated with Met.Ext (250 mg/kg) exhibiting with mild bile duct ingestion, scattered inflammatory cells are seen, (E) Histopathology of a Liver from Group V of rabbits treated with Met. Ext (500 mg/kg) shows normal histological appearance and Improved architecture is observed in groups; (H & E staining; 40X & 100X, scale bar = 100 μm)}.
3.11. Histopathological evaluation of the kidney tissues
Protective effects of D. mucronata leaves Met. Ext on paracetamol induced nephrotoxicity are presented in Fig. 8. Histopathological slide of Rabbit’s kidney of Group I (normal saline) shows normal glomeruli and flat epithelium lining glomerular capsule with distinct capsular space. Animals of Group II, treated with paracetamol shows somewhat necrosis and inflammation. Histopathology of kidney from Group III animals treated with standard silymarin shows normal parenchyma with no significant changes. Group IV animals, treated with Met. Ext. (250 mg/kg) shows normal renal parenchymal tubules with the normal lumen, while for Group V rabbits treated with Met. Ext (500 mg/kg) normal renal parenchyma with no significant pathology were observed.
Fig. 8.
Effects of Met. Ext of Daphne mucronata on histopathology studies in paracetamol-induced nephrotoxicity in rabbits. {(A) Histopathological slide of Rabbit’s kidney of Group I showing normal glomeruli and flat epithelium lining glomerular capsule with distinct capsular space, (B) Histopathological slide of Rabbit’s kidney of Group II, treated with paracetamol shows somewhat necrosis and inflammation, (C) Histopathology of a kidney from Group III treated with standard Silymarin shows normal parenchyma with no significant changes, (D) Histopathology of a kidney from Group IV, treated with Met. Ext. (250 mg/kg) shows normal renal parenchymal tubules with the normal lumen, (E) Improved architecture is observed in groups of rabbits treated with Met. Ext (500 mg/kg) indicating normal renal parenchyma with no significant pathology; (H &E staining; 40X & 100X, scale bar = 100 μm)}.
4. Discussion
In the current study, hepatotoxicity is evident from the high levels of biochemical parameters such as ALT, AST, ALP, serum triglycerides, cholesterol and bilirubin in paracetamol treated groups as compared to the standard silymarin group. The paracetamol treatment increases the level of the mentioned liver biomarkers that results in excessive damage to liver cells. The plant extracts reinstated/restored the damage caused by paracetamol which has been observed in animal models for different plant extracts. Similar results have been observed in this study as well. Previous studies on D. mucronata phytochemical analysis have shown that it contains coumarins, flavonoids, triterpenoids, diterpene, sterols, lignin cumarinolignans, glucosides, daphnecin, aquillochin, daphnine and umbelliferone (Al-Snafi et al., 2019). Other biologically active compounds isolated from this plant previously includes; 5, 7, 3′, 4′-tetrahydroxyflavone and 5, 3′, 4′-trihydroxyflavone 7-O-β-D-glycopyranoside that have exhibited antioxidant potentials as well (Rasool et al., 2009). The plants phenols, diterpenes, and coumarins are associated with significant antioxidant activities (Shah et al., 2018). There is a close relationship between antioxidant activities and phenolic compounds (Yener et al., 2020). In the current study HPLC profiling of D. mucronata leaf extract showed the presence of phenolic and flavonoid compounds like malic acid, gallic acid, chlorogenic acid, epigallocatechin gallate, quercetin, morin, ellagic acid, and rutin. Among them, chlorogenic acid, epigallocatechin gallate, quercetin, and ellagic acid have previously exhibited strong antioxidant and hepatoprotective activities (Zhang et al., 2014, Afifi et al., 2018, Ashraf et al., 2018, Aljelehawy et al., 2020). Plants have antioxidant properties depending on the content of phenol and flavonoids, due to which they may play a role in scavenging free radicals thus exhibiting protective effects in oxidative stress and related complications in animal models (Nazir et al., 2021).
Since paracetamol toxicity demonstrates itself through oxidation pathways, ROS and MDA levels (as oxidative markers), GSH and TAC levels (as antioxidant indices) are evaluated in liver and kidney tissues is such situations. In the current study, an increase in lipid peroxidation have caused a significant increase in MDA level and decrease in the levels of antioxidants such as GSH and TAC that indicates the oxidative stress, which is in agreement with the other reported studies in literature (Sohrabinezhad et al., 2019, Ashfaq et al., 2020). Studies have shown that the enzymatic and non-enzymatic systems that maintain cellular homeostasis, as well as the level of lipid peroxidation, are markedly influenced by paracetamol (Belinskaia et al., 2020). The severity of the damage in terms hepatotoxicity and nephrotoxicity induced by paracetamol was evident from an increase in the serum levels of the lipid peroxidation indicator; malondialdehyde and decreased levels of GSH and TAC (which serves as indices of antioxidant status). Significant decreases in GSH and TAC levels have also been observed in the liver, kidney, and blood of patients with liver disease compared to controls in a reported study (Afsharinasab et al., 2020). In recent years, a large number of plants have been tested to eradicate the liver damage caused by paracetamol in animal models, and the bioactive compounds responsible for alleviating oxidative stress have been isolated (Meharie et al., 2020). The predominant and responsible metabolite of paracetamol; NAPQI that is responsible for liver damage, in high concentration causes a decrease in GSH and TAC levels in hepatocytes. Overproduction of NAPQI causes a break down of the SH group present in macromolecules such as proteins, nucleic acids, and membranes leading to liver damage. Under reduced GSH level, excessive NAPQI covalently bound to vital proteins sandwitched between lipid bilayer in membranes of hepatocytes, resulting in lipid peroxidation and increased MDA levels. The improvement in GSH levels in the liver alleviates both the initiation and the progression of liver damage caused by paracetamol (Jaeschke et al., 2020).
The kidney controls plasma levels of ions like sodium, potassium, calcium, magnesium, and chloride and are importantly involved in the removal of nitrogenous waste products such as urea, uric acid, and creatinine from the body. Increase in the levels of kidney parameters like serum urea, uric acid and creatinine are usually monitored to assess the drug induced nephrotoxicity in man and also in animal models (Aziz, 2019). Our results are in agreement with the reported studies where pre-treatment with silymarin (50 mg/kg) standard significantly decline the elevated level of serum creatinine, urea and plasma ions induced by Paracetamol (Kassem at al., 2013).
The observed effects in terms of biochemical parameters were further supported by histopathological examination. Histological architecture of the liver sections of paracetamol intoxicated rabbits displayed a significant necrosis, inflammation, bile duct proliferation, and cholestasis. Paracetamol treated rabbits showed cuboidal epithelial cell necrosis in proximal convoluted tubules which are also in line with the reported studies (Rehman et al., 2015). The histopathological examination revealed that D. mucronata extract protect liver and kidneys of intoxicated animals. The standard silymarin (A standard hepatoprotective drug) greatly reduced the cellular impairment caused by paracetamol due to its strong antioxidant potential (Nazir et al., 2018a, Nazir et al., 2018b), and thus reduces the hepatotoxicity and lipid peroxidation caused by a variety of other agents as well. In the current study the protective effects of D. mucronata Met. Ext were comparable to that of the standard silymarin.
Medicinal plants and their metabolites are of great importance in the present modern world and are in use from centuries. However, before use as therapeutic agent, it is needed to assess their pharmacological and toxicological effects scientifically (Jothy et al., 2012). Keeping in view the importance of medicinal plant; D. mucronata, the current study was carried out to assess the hepatoprotective and nephroprotective effect intoxicated rabbits. Literature studies revealed that plants contain valuable phytoconstituents which are responsible for the observed hepatoprotective and nephroprotective activities (Akomolafe et al., 2014, Domitrović et al., 2014, Nehal et al., 2018, Ghosia et al., 2019, Bhatia et al., 2019, Kuzu et al., 2019, Prasad and Prasad, 2019). The results of this study suggests that possible mechanism of this protection might be due the scavenging capabilities of phytoconstituents (mostly phenolics) present that have effectively scavenged the free radicals produced in intoxicated animals. However further studies are required to evaluate the exact mechanism of action.
To the best of our knowledge, there was no reported hepatoprotective and nephroprotective studies on D. mucronata. Therefore, this study was designed to report the contents of phenolic acids and flavonoids which most probably were responsible in vitro antioxidant activity and in vivo hepatoprotective and nephroprotective effects. D. mucronata leaves Met. Ext ameliorated paracetamol intoxicated acute liver and kidney injuries in rabbits, evidenced by the outcomes of both biochemical and histological studies.
5. Conclusion
The results of the current study revealed that leaves extract of D. mucronata have in vitro antioxidant and in vivo hepatoprotective, and nephroprotective potentials and is capable of decreasing elevated levels of liver biochemical parameters like AST, ALT, ALP, serum triglyceride, serum cholesterol, serum bilirubin, and kidneys biomarkers like serum urea, serum uric acid, and serum creatinine levels in paracetamol intoxicated rabbits. D. mucronata extract also inhibited the physiological and histological changes in liver and kidney in addition to decreasing effect on lipid peroxidation MDA content. The extract has also increased the level of antioxidant enzymes like GSH and TAC, and therefore, it can be inferred that the extract might be capable of preventing hepatic and renal toxicity through enhancing hepatic and kidney tissue oxidant/antioxidant balance. These effects might be due to the presence of phenolic and flavonoids phytoconstituents present in the methanolic extract of D. mucronata. Anti-oxidative therapy, especially the use of natural antioxidants, is an affordable therapeutic approach for the prevention of oxidative stress related diseases of liver and kidney. Keeping in view the high medicinal importance of D. mucronata it could be recommended for reducing hepatic impairments and related diseases of liver and kidney and could also be used as bioceutical or dietary supplement in controlling liver and kidney diseases. However, further experiments are needed to assess its permissible doses and toxicities in other animal models along with isolation of responsible compounds.
Declarations
Ethics approval
All procedures related to the animal activities have been approved by the Departmental Animal Ethical Committee of the University of Malakand (DAEC/2019/1) and were conducted according to the ARRIVE guidelines and UK: Animal Scientific Procedure Act (1986). These guidelines were in accordance with the internationally documented principles for laboratory used and care.
Consent for publication
Not applicable for this submission.
Availability of data and materials
The data presented in this manuscript belong to the research work done by Mr. Jabran Muhammad under the supervision of Dr. Nausheen Nazir and has not been deposited in any repository yet. However, the materials are available to the researchers upon request.
Authors’ contributions
JM & NN carried out plants collection and experimental work. NN carried out literature search and manuscript preparation and supervised the research project. NN, RG & JM carried out in vivo experimental work. Statistical analysis was done by FU. RU and AA carried out formal analysis. Corrections and editing of the manuscript was done by NN, AA, MZ, and MN. MZ revised the paper. All the authors have read and finally approved the manuscript.
Funding
This research was funded by Deanship of Scientific Research at Princess Nourah bint Abdulrahman University Riyadh Saudi Arabia.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at Princess Nourah bint Abdulrahman University Riyadh Saudi Arabia for funding this work through the Fast-Track Research Funding Program.
Footnotes
Peer review under responsibility of King Saud University.
Supplementary data to this article can be found online at https://doi.org/10.1016/j.sjbs.2021.05.051.
Contributor Information
Nausheen Nazir, Email: nausheen.nazir@uom.edu.pk.
Riaz Ullah, Email: rullah@ksu.edu.sa.
Amal Alotaibi, Email: amaalotaibi@pnu.edu.sa.
Appendix A. Supplementary material
The following are the Supplementary data to this article:
References
- Abdel-Daim M.M., Ghazy E.W. Effects of Nigella sativa oil and ascorbic acid against oxytetracycline-induced hepato-renal toxicity in rabbits. Iran J. Basic Med. Sci. 2015;18:221–227. [PMC free article] [PubMed] [Google Scholar]
- Al-Snafi A., Mousa H., Majid W. Medicinal plants possessed hepatoprotective activity. IOSR J. Pharm. 2019;9:26–56. [Google Scholar]
- Afifi N.A., Ibrahim M.A., Galal M.K. Hepatoprotective influence of quercetin and ellagic acid on thioacetamide-induced hepatotoxicity in rats. Can. J. Physiol. Pharmacol. 2018;96(6):624–629. doi: 10.1139/cjpp-2017-0651. [DOI] [PubMed] [Google Scholar]
- Akomolafe S.F., Akinyemi A.J., Anadozie S.O. Phenolic Acids (Gallic and Tannic Acids) Modulate Antioxidant Status and Cisplatin Induced Nephrotoxicity in Rats. Int. Sch. Res. Notices. 2014;2014 doi: 10.1155/2014/984709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashraf I., Zubair M., Rizwan K., Rasool N., Jamil M., Khan S.A., Tareen R.B., Ahmad V.U., Mahmood A., Riaz M., Zia-Ul-Haq M., Jaafar H.Z. Chemical composition, antioxidant and antimicrobial potential of essential oils from different parts of Daphne mucronata Royle. Chem. Cent. J. 2018;12:135. doi: 10.1186/s13065-018-0495-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Afsharinasab M., Mohammad-Sadeghipour M., Hajizadeh M.R., Khoshdel A., Mirzaiey V., Mahmoodi M. The effect of hydroalcoholic Berberis integerrima fruits extract on the lipid profile, antioxidant parameters and liver and kidney function tests in patients with nonalcoholic fatty liver disease. Saudi J. Biol. Sci. 2020;27(8):2031–2037. doi: 10.1016/j.sjbs.2020.04.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashfaq F., Butt M.S., Bilal A., Awan K.A., Suleria H.A. Assessing the therapeutic potential of red cabbage (Brassica oleracea var. capitata) to modulate renal and cardiac oxidative stress biomarkers in hypercholesterolemic rabbits. Curr. Bioact. Compd. 2020;16(4):521–527. [Google Scholar]
- Aljelehawy Q., Karimi N., Alavi M. Comparison of antibacterial and cytotoxic activities of phytosynthesized ZnONPs by leaves extract of Daphne mucronata at different salt sources. Mater. Technol. 2020;36(6):1–13. [Google Scholar]
- Aziz R. Effects of date extract on paracetamol induced nephrotoxicity in rabbits. Pakistan J. Medical Health Sci. 2019;13:510. [Google Scholar]
- Brand-Williams W., Cuvelier M., Berset C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995;28(1):25–30. [Google Scholar]
- Belinskaia D.A., Voronina P.A., Shmurak V.I., Vovk M.A., Batalova A.A., Jenkins R.O., Goncharov N.V. The universal soldier: enzymatic and non-enzymatic antioxidant functions of serum albumin. Antioxidants. 2020;9(10):966. doi: 10.3390/antiox9100966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhatia D., Bansal M., Bhangu S., Singh G. Hepatoprotective effects of epigallocatachin gallate via mitochondrial permeability transition pore in paracetamol induced hepatotoxicity. Plant Arch. 2019;19:2162–2167. [Google Scholar]
- Can T.H., Tufekci E.F., Altunoglu Y.C., Baloglu M.C., Llorent-Martínez E.J., Stefanucci A. Chemical characterization, computational analysis and biological views on Daphne gnidioides Jaub. & Spach extracts: Can a new raw material be provided for biopharmaceutical applications? Comput. Biol. Chem. 2020;87 doi: 10.1016/j.compbiolchem.2020.107273. [DOI] [PubMed] [Google Scholar]
- Cicero L., Fazzotta S., Palumbo V.D., Cassata G., Lo Monte A.I. Anesthesia protocols in laboratory animals used for scientific purposes. Acta Biomed. 2018;89:337–342. doi: 10.23750/abm.v89i3.5824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Domitrović R., Cvijanović O., Šušnić V., Katalinić N. Renoprotective mechanisms of chlorogenic acid in cisplatin-induced kidney injury. Toxicol. 2014;324:98–107. doi: 10.1016/j.tox.2014.07.004. [DOI] [PubMed] [Google Scholar]
- Ghasemi Pirbalouti A., Momeni M., Bahmani M. Ethnobotanical study of medicinal plants used by Kurd tribe in Dehloran and Abdanan Districts, Ilam Province. Iran. Afr. J. Tradit. Complement. Altern. Med. 2012;10:368–385. doi: 10.4314/ajtcam.v10i2.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosia L., Asma S., Kafeel A., Jamila H. Phytochemical screening, antioxidant and antibacterial properties of daphne mucronata. J. Tradit. Chin. Med. 2019;39:764–771. [PubMed] [Google Scholar]
- Jaeschke H., Ramachandran A. The role of oxidant stress in acetaminophen-induced liver injury. Curr. Opin. Toxicol. 2020;20–21:9–14. doi: 10.1016/j.cotox.2020.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jothy S.L., Aziz A., Chen Y., Sasidharan S. Antioxidant activity and hepatoprotective potential of polyalthia longifolia and cassia spectabilis leaves against paracetamol-induced liver injury. Evid. Based Complement. Altern. Med. 2012;2012 doi: 10.1155/2012/561284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katayoun J., Ramin M., Rahim B.N., Narjess K.J. A preliminary study on the biological activity of daphne mucronata royle. DARU J. Pharm. Sci. 2003;11:28–31. [Google Scholar]
- Kuzu M., Yıldırım S., Kandemir F.M., Küçükler S., Çağlayan C., Türk E. Protective effect of morin on doxorubicin-induced hepatorenal toxicity in rats. Chem. Biol. Interact. 2019;308:89–100. doi: 10.1016/j.cbi.2019.05.017. [DOI] [PubMed] [Google Scholar]
- Kassem F., Alqasoumi S., Sallam S., Bekhit A., Al-shaer N., Farraj A. Evaluation of the hepatoprotective, nephroprotective and anti-malarial activities of different parts of Bauhinia purpurae and Tipuana speciosa grown in Egypt. J. Med. Plant Res. 2013;7:1190–1200. [Google Scholar]
- Levey A.S., Atkins R., Coresh J., Cohen E.P., Collins A.J., Eckardt K.U., Nahas M.E., Jaber B.L., Jadoul M., Levin A., Powe N.R., Rossert J., Wheeler D.C., Lameire N., Eknoyan G. Chronic kidney disease as a global public health problem: Approaches and initiatives - a position statement from Kidney Disease Improving Global Outcomes. Kidney Int. 2007;10:1–5. doi: 10.1038/sj.ki.5002343. [DOI] [PubMed] [Google Scholar]
- Meharie B.G., Amare G.G., Belayneh Y.M. Evaluation of hepatoprotective activity of the crude extract and solvent fractions of Clutia abyssinica (Euphorbiaceae) leaf against CCl4-induced hepatotoxicity in mice. J. Exp. Pharmacol. 2020;12:137–141. doi: 10.2147/JEP.S248677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moshiashvili G., Tabatadze N., Mshvildadze V. The genus Daphne: A review of its traditional uses, phytochemistry and pharmacology. Fitoterapia. 2020;143 doi: 10.1016/j.fitote.2020.104540. [DOI] [PubMed] [Google Scholar]
- Malik, K., Ahmad, M., Zafar, M., Ullah, R., Mahmood, H.M., Parveen, B., Rashid, N., Sultana, S., Shah, S.N., Lubna., 2019. An ethnobotanical study of medicinal plants used to treat skin diseases in northern Pakistan. BMC Complement. Altern. Med. 19(1), 210. [DOI] [PMC free article] [PubMed]
- Mosaddegh M., Naghibi F., Moazzeni H., Pirani A., Esmaeili S. Ethnobotanical survey of herbal remedies traditionally used in Kohghiluyeh va Boyer Ahmad province of Iran. J. Ethnopharmacol. 2012;141:80–95. doi: 10.1016/j.jep.2012.02.004. [DOI] [PubMed] [Google Scholar]
- Nazir N., Zahoor M., Nisar M. A review on traditional uses and pharmacological importance of genus elaeagnus species. Bot. Rev. 2020;86:247–280. [Google Scholar]
- Nazir N., Zahoor M., Nisar M., Karim N., Latif A., Ahmad S., Uddin Z. Evaluation of neuroprotective and anti-amnesic effects of Elaeagnus umbellata Thunb. On scopolamine-induced memory impairment in mice. BMC Complement. Med. Ther. 2020;20:143. doi: 10.1186/s12906-020-02942-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nazir N., Zahoor M., Nisar M., Khan I., Karim N., Abdel-Halim H. Phytochemical analysis and antidiabetic potential of Elaeagnus umbellata (Thunb.) in streptozotocin-induced diabetic rats: pharmacological and computational approach. BMC Complement. Altern. Med. 2018;18(1):332. doi: 10.1186/s12906-018-2381-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nazir N., Nisar M., Zahoor M., Uddin F., Ullah S., Ullah R. Phytochemical analysis, in vitro anticholinesterase, antioxidant activity and in vivo nootropic effect of ferula ammoniacum (Dorema ammoniacum) D. Don. in scopolamine-induced memory impairment in mice. Brain Sci. 2021;11:259. doi: 10.3390/brainsci11020259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nazir N., Karim N., Abdel-Halim H., Khan I., Wadood S.F., Nisar M. Phytochemical analysis, molecular docking and antiamnesic effects of methanolic extract of Silybum marianum (L.) Gaertn seeds in scopolamine induced memory impairment in mice. J. Ethnopharmacol. 2018;210:198–208. doi: 10.1016/j.jep.2017.08.026. [DOI] [PubMed] [Google Scholar]
- Nehal, A., Marwa, A., Ibrahim, A., Mona. K., Galal., 2018. Hepatoprotective influence of quercetin and ellagic acid on thioacetamide-induced hepatotoxicity in rats. Can. J. Physiol. Pharmacol. 96(6), 624-629. [DOI] [PubMed]
- Prasad R., Prasad S. A review on the chemistry and biological properties of Rutin, a promising nutraceutical agent. Asian J. Pharm. 2019;5(S1):1–20. [Google Scholar]
- Rui L. Energy metabolism in the liver. Compr. Physiol. 2014;4:177–197. doi: 10.1002/cphy.c130024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rehman J.N., Saqib N., Akhtar M., Jamshaid H., Asif S., Sultana R. Hepatoprotective activity of aqueous-methanolic extract of suaeda fruticosa in paracetamol-induced hepatotoxicity in rabbits. Bangladesh J. Pharmacol. 2013;8(4):378–381. [Google Scholar]
- Rasool M.A., Imran M., Nawaz H., Malik A., Kazmi S.U. Phytochemical studies on Daphne mucronata. J. Chem. Soc. Pak. 2009;31:845–850. [Google Scholar]
- Re R., Pellegrini N., Proteggente A., Pannala A., Re Y.M. Antioxidant activity applying an improved FBTS radical cation decolorization assay. Free Rad. Biol. Med. 1999;26(9/10):7–1231. doi: 10.1016/s0891-5849(98)00315-3. [DOI] [PubMed] [Google Scholar]
- Rehman J., Akhtar N., Khan M., Ahmad K., Ahmad M., Sultana S. Phytochemical Screening and Hepatoprotective Effect of Alhagi maurorum Boiss (Leguminosae) Against Paracetamol-Induced Hepatotoxicity in Rabbits. Tropical. J. Pharm. Res. 2015;14:1029–1034. [Google Scholar]
- Shirazi U., Khattak K., Shukri M., Nasyriq N. Determination of total phenolic, flavonoid content and free radical scavenging activities of common herbs and spices. J. Pharm. Phytochem. 2014;3(3):104–108. [Google Scholar]
- Shah A., Lutfullah G., Ahmad K., Khalil A.T., Maaza M. Daphne mucronata-mediated phytosynthesis of silver nanoparticles and their novel biological applications, compatibility and toxicity studies. Green Chem. Lett. Rev. 2018;11:318–333. [Google Scholar]
- Saleem T.S.M., Chetty S.M., Ramkanth S., Rajan V.S.T., Kumar K.M., Gauthaman K. Hepatoprotective herbs: A review. Int. J. Res. Pharm. Sci. 2010;1:1–5. [Google Scholar]
- Sohrabinezhad Z., Dastan D., Asl S.S., Nili-Ahmadabadi A. Allium Jesdianum extract improve acetaminophen-induced hepatic failure through inhibition of oxidative/nitrosative stress. J. Pharmacopunct. 2019;22(4):239–247. doi: 10.3831/KPI.2019.22.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorne A.M., Ubbink R., Brüggenwirth I.M.A., Nijsten M.W., Porte R.J., Meijer V.E. Hyperthermia-induced changes in liver physiology and metabolism: a rationale for hyperthermic machine perfusion. Am. J. Physiol. Gastrointest. Liver Physiol. 2020;319:G43–G50. doi: 10.1152/ajpgi.00101.2020. [DOI] [PubMed] [Google Scholar]
- Vargas-Pozada E.E., Muriel P. Herbal medicines for the liver: from bench to bedside. Eur. J. Gastroenterol. Hepatol. 2020;32(2):148–15832. doi: 10.1097/MEG.0000000000001485. [DOI] [PubMed] [Google Scholar]
- Yener I., Kocakaya S.O., Ertas A., Erhan B., Kaplaner E., Oral E.V. Selective in vitro and in silico enzymes inhibitory activities of phenolic acids and flavonoids of food plants: Relations with oxidative stress. Food Chem. 2020;327 doi: 10.1016/j.foodchem.2020.127045. [DOI] [PubMed] [Google Scholar]
- Zeb A. Reversed phase HPLC-DAD method for the determination of phenolic compounds in plant leaves. Anal. Methods. 2015;7:7753–7757. [Google Scholar]
- Zhang X., Wu Z., Weng P. Antioxidant and hepatoprotective effect of (−)-epigallocatechin 3-O-(3-O-methyl) gallate (EGCG3 ″Me) from Chinese oolong tea. J. Agric. Food Chem. 2014;62(41):10046–10054. doi: 10.1021/jf5016335. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.