Abstract
A 51-year-old man with a history of renal cell carcinoma presented with sudden aphasia, right hemiparesis, and dysesthesia. MRA showed left middle cerebral artery occlusion, and he was diagnosed with acute ischemic stroke and treated with intravenous recombinant tissue plasminogen activator and endovascular thrombectomy. The pathological diagnosis of the retrieved thrombus was consistent with the already-known pathological findings of the primary renal cell carcinoma. Therefore, a diagnosis of cerebral embolism caused by tumor cells was made. The pathological findings of the retrieved thrombi were important in determining the cause of ischemic stroke.
Keywords: cerebral infarction, cancer, thrombectomy, thrombus
Introduction
The pathophysiological mechanisms of stroke in patients with cancer include hypercoagulability, therapeutic or diagnostic interventions, and infection (1). However, in clinical practice, the precise diagnosis of whether stroke is caused by the cancer itself, by treatment for cancer, or by general causes of stroke remains still challenging. Tumor embolization can directly result in cerebral embolism, but there have been few reports of pathologically diagnosed tumor embolization in patients with ischemic stroke (2-5).
Case Report
A 51-year-old man with a history of primary renal cell carcinoma, pleural metastasis, and cancerous pleural effusion presented with sudden aphasia, right hemiparesis, and dysesthesia. He underwent left nephrectomy for primary renal cell carcinoma at 46 years old and subsequently received chemotherapy and radiotherapy for metastases to the adrenal gland, mediastinal lymph nodes, pleura, bone, and a carcinomatous pleural effusion. The baseline National Institutes of Health Stroke Scale (NIHSS) score was 7.
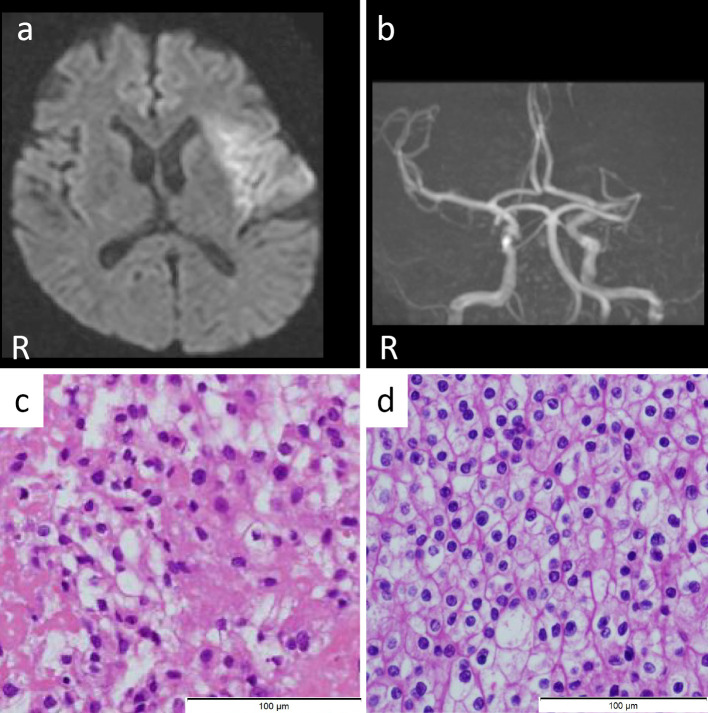
Diffusion-weighted magnetic resonance imaging (MRI) showed a hyperintense area in the left insular cortex and the left middle cerebral artery territory (Figure a). Magnetic resonance angiography (MRA) showed left middle cerebral artery occlusion (Figure b). The D-dimer level was 7.93 μg/dL. The diagnosis was acute ischemic stroke. He had diffusion-weighted imaging-fluid-attenuated inversion recovery mismatch, which indicated that the stroke had occurred within the past 4.5 hours (2), suggesting the presence of salvageable brain tissue.
Figure.
(a) Axial diffusion-weighted image (DWI, 1.5 T; B value 1,000 s/mm2, TR 6,000 ms, TE 100 ms) shows a hyperintense lesion in the left insular cortex and middle cerebral artery cortex. (b) MRA shows left middle cerebral artery occlusion. (c) Retrieved thrombus (×200). Hematoxylin and Eosin staining shows a solid mass growing in the fibrin-based thrombus. (d) Microscopic image of the renal cell carcinoma (×200). The pathological findings are closely consistent with the retrieved thrombus.
He was treated with intravenous recombinant tissue-type plasminogen activator and endovascular thrombectomy. Using a Penumbra System (Penumbra, Alameda, USA) and Solitaire (Medtronic, Dublin, Ireland), a red and white combined ‘clot’ was retrieved. Successful recanalization (Thrombolysis in Cerebral Infarction Grade 2b) was achieved, and the NIHSS score improved to 1 after treatment. The time course for this patient was 102 minutes from the onset to door, 176 minutes from the onset to needle, and 257 minutes from the onset to recanalization.
The retrieved thrombus was 3 mm×1 mm×1 mm in size and consisted mainly of clotted tissue with fibrin precipitation. The histopathological examination of the retrieved thrombus showed degenerated tumor cells with clear cytoplasm (Figure c), resembling the previous findings of the renal cell carcinoma operated on five years earlier (Figure d). His 24-hour electrocardiogram, transthoracic echocardiogram, and carotid ultrasonography findings were normal. Chest CT showed pleural effusion due to the renal cell carcinoma metastasis. There was no mass lesion in the left atrium on CT. The final diagnosis was cerebral embolism due to tumor cells based on the pathological diagnosis of the retrieved thrombi and primary renal cell carcinoma.
Because the thrombi contained fibrin, subcutaneous heparin therapy was selected for secondary prevention, as for cancer-associated stroke. The patient was discharged home on the 15th day without assistance. He died two months after discharge from the hospital due to exacerbation of cancerous pleural effusion and respiratory failure associated with renal cell carcinoma. He and his family were able to achieve a satisfactory end of life.
Discussion
In the present case, the pathological findings of the mechanically retrieved thrombi were extremely important for determining the cause of ischemic stroke. This proved a major point, with tumor embolization from known cancer resulting in embolic stroke based on the pathologically confirmed findings of the retrieved thrombi.
There have been a few reports of mechanical tumor embolectomy performed for acute ischemic stroke in patients with cancer (3-8). The previous reports of tumor cells retrieved by endovascular thrombectomy for acute ischemic stroke are shown in Table. Those reports suggest that tumor cells can enter the blood stream through direct invasion to the pulmonary vein or left heart, although there have been no autopsy reports to prove this. In the present case, body CT showed no evidence of gross pulmonary venous invasion, but there was pleural dissemination. The tumor cells invaded the intersegmental and interlobar veins within the lung. It appears that the tumor cells infiltrated microscopically into the subpleural small pulmonary veins in the pleural disseminated lesions and extended to the larger pulmonary vein. Even in cases of subdiaphragmatic carcinoma, pleural disseminated lesions can cause cerebral embolism.
Table.
Characteristics of Patients with Tumor Cells in Retrieved Thrombi Seen on Pathology during Endovascular Thrombectomy for Acute Ischemic Stroke.
| Age (years)/ Sex |
Occluded artery | Baseline NIHSS score | Post-treatment NIHSS score | Primary cancer | Metastatic | Tumor invasion | Suspected stroke mechanism | |
|---|---|---|---|---|---|---|---|---|
| Present case | 51 M | Left M1 portion of the MCA | 7 | 1 | Renal cell carcinoma | Adrenal gland, mediastinal lymph node, pleural, bone | Suspected invasion lesion in the pulmonary veins | Emboli mixed with thrombi and tumor cells flowed into the left atrium from the pulmonary vein and caused occlusion of the intracranial artery |
| (2) | 62 F | Supraclinoid left ICA | 19 | Not available | Breast cancer | Endobronchial mass, Bilateral hilar and mediastinal lymphadenopathy, Multiple pulmonary nodules | Suspected invasion lesion in the pulmonary veins | Tumor growth into the pulmonary veins; occasionally fragments may break off and become lodged in the cerebral circulation |
| (3) | 22 F | Left M1 portion of the MCA | 4 | 1 | Malignant melanoma | Brain, both upper arms, chest walls, heart, right axillary, right supraclavicular, and right renal hilar lymph nodes | Cardiac metastasis on PET | Tumor embolization of metastatic cardiac melanomas |
| (4) | 69 M | Right M1 portion of the MCA | 16 | 30 | Adenocarcinoma of the lung | No evidence of metastasis on PET CT | Not available | Embolism due to metastatic lung adenocarcinoma |
| (5) | 56 M | Basilar artery and ICA | Not available | Not available | Right pulmonary hilum tumor | Left atrium, pulmonary veins | Left atrium and pulmonary vein on contrast CT | Embolism from an invasive extracardiac tumor |
| (6) | 80 M | Basilar artery | 19 | Not available | Large-cell, high-grade neuroendocrine lung cancer | Not available | Suspected invasion lesion in the pulmonary veins | Embolus detached into the bloodstream from the lung after intruding into a pulmonary vein |
| (7) | 66 M | Right M1 portion of the MCA | 14 | Not available | Pleomorphic lung carcinoma | Bronchial invasion | Suspected invasion lesion in the pulmonary veins | Embolus migrated from an invasive lesion in the pulmonary vein |
This list excludes cases of primary cardiac tumor.
ICA: internal carotid artery, MCA: middle cerebral artery, PET: positron emission tomography
The stroke mechanism in the present patient was likely as follows: disseminated tumor cells infiltrated microscopically into the small intersegmental intrapulmonary vein and then extended to the larger interlobar vein and denatured to form thrombi. Thereafter, detached thrombi with collapsed tumor cells flowed into the left atrium from the pulmonary vein and then caused occlusion of the intracranial artery.
Conclusions
Prompt reperfusion therapy for cancer-associated stroke should be considered in some cases to maintain patients' quality of life during their limited lifetime, even if their cancers are advanced, as long as each patient's condition permits.
The authors state that they have no Conflict of Interest (COI).
Acknowledgement
The authors are deeply grateful to Dr. Hiroki Sasamori, Dr. Masataka Torii, and Dr. Eishi Sato (Kyorin University Hospital) for their clinical support. They also express their sincere gratitude to Dr. Takehito Naruoka (The Jikei University Daisan Hospital) for providing pathological information.
References
- 1.Dardiotis E, Aloizou AM, Markoula S, et al. Cancer-associated stroke: pathophysiology, detection and management (Review). Int J Oncol 54: 779-796, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Aoki J, Kimura K, Iguchi Y, et al. FLAIR can estimate the onset time in acute ischemic stroke patients. J Neurol Sci 293: 39-44, 2010. [DOI] [PubMed] [Google Scholar]
- 3.Bhatia S, Ku A, Pu C, et al. Endovascular mechanical retrieval of a terminal internal carotid artery breast tumor embolus. J Neurosurg 112: 572-574, 2010. [DOI] [PubMed] [Google Scholar]
- 4.Kim CS, Jung HR, Cho KH, et al. Forced-suction thrombectomy of an arterial tumor embolism due to metastatic melanoma. Arch Neurol 69: 272-273, 2012. [DOI] [PubMed] [Google Scholar]
- 5.Hughes SE, Hunter A, Campbell J, et al. Extraction of tumour embolus following perioperative stroke. J Neurol Sci 353: 172-174, 2015. [DOI] [PubMed] [Google Scholar]
- 6.Pop R, Mihoc D, Manisor M, et al. Mechanical thrombectomy for repeated cerebral tumor embolism from a thoracic sarcomatoid carcinoma. BMJ Case Rep 2017: 013092, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Goddard JK, Nussbaum ES, Madison M, et al. Endovascular aspiration to treat acute ischemic stroke caused by embolic carcinoma. Interv Neuroradiol 25: 403-406, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yoshikawa S, Kamide T, Kasakura S, et al. A case of cerebral infarction due to pleomorphic carcinoma of the lung. Surg Neurol Int 11: 217, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]