Pneumomediastinum is a rare complication of acute respiratory distress syndrome (ARDS), when air leaks into the mediastinum. An increased pneumomediastinum incidence, of up to 5–13%, was reported during the coronavirus disease 2019 (COVID-19) pandemic [1, 2] and even occurred spontaneously without a history of mechanical ventilation [3], similarly to the previous severe acute respiratory syndrome (SARS)-1 [4]. Almost half of the 30 consecutive COVID-19 patients who had prolonged invasive mechanical ventilation had full-thickness tracheal lesions and pneumomediastinum [5]. Although pronation and high positive end-expiratory pressure levels were presumed to be the putative causes of pneumomediastinum [5], the mechanism of this COVID-19 complication remains unknown. We hypothesise that sonic hedgehog (SHH) and Wnt5a signalling, crucial pathways in tracheal morphogenesis, and repair/regeneration of cartilage lesions in adulthood [6, 7], could play a role in pneumomediastinum-related COVID-19 tracheal lesions.
Short abstract
Pneumomediastinum is a rare complication of ARDS but is more common during #COVID19. The fibrous hyaline degeneration of the tracheal rings seen in this autoptic series is an original observation that has not been previously described in COVID-19 patients. https://bit.ly/3vxTQde
To the Editor:
Pneumomediastinum is a rare complication of acute respiratory distress syndrome (ARDS), when air leaks into the mediastinum. An increased pneumomediastinum incidence, of up to 5–13%, was reported during the coronavirus disease 2019 (COVID-19) pandemic [1, 2] and even occurred spontaneously without a history of mechanical ventilation [3], similarly to the previous severe acute respiratory syndrome (SARS)-1 [4]. Almost half of the 30 consecutive COVID-19 patients who had prolonged invasive mechanical ventilation had full-thickness tracheal lesions and pneumomediastinum [5]. Although pronation and high positive end-expiratory pressure levels were presumed to be the putative causes of pneumomediastinum [5], the mechanism of this COVID-19 complication remains unknown. We hypothesise that sonic hedgehog (SHH) and Wnt5a signalling, crucial pathways in tracheal morphogenesis, and repair/regeneration of cartilage lesions in adulthood [6, 7], could play a role in pneumomediastinum-related COVID-19 tracheal lesions.
This study assessed the pneumomediastinum characteristics in hospitalised COVID-19 patients through a retrospective analysis of a prospectively collected COVID-19 database to identify the clinical–radiological and pathological signatures associated with severe SARS-coronavirus-2 viral infection.
This observational study was carried out from 10 March 2020 to 28 February 2021 at the University Hospital of Trieste and was approved by the local Ethical Committee (CEUR#2020-Os-148). The patient or next of kin provided written informed consent.
Inclusion criteria were: 1) hospitalised patients with pneumonia positive for COVID-19 confirmed by a positive reverse-transcription-PCR analysis from nasopharyngeal or oropharyngeal swabs [8]; 2) aged ≥18 years; 3) chest computed tomography (CT) scan confirmed pneumomediastinum. Volumetric unenhanced CT images were acquired (Revolution EVO GE Healthcare) and reconstructed with a slice thickness of 1 mm and lung sharp kernels. All CT images were evaluated by three radiologists aware of the COVID-19 status, but blinded to other clinical information. Clinical data were obtained from the local electronic clinical records. Three pathologists blinded to patient identification performed a masked pathological study on the autoptic trachea specimens from patients who died of pneumomediastinum. The patients’ haematoxylin and eosin slides were reviewed separately and anonymously. Immunohistochemistry (IHC) was performed on formalin-fixed and paraffin-embedded (4-Gm-thick) sections using antibodies directed against SHH (Abcam; clone EP1190Y, rabbit monoclonal anti-human, 1:100) and Wnt5a (ThermoFisher; clone 3D10, mouse anti-human monoclonal, 1:200). IHC assays were assessed by a semi-quantitative ranking score, ranging from 0, for no labelling, to 4 for intense labelling, in the slices of cartilage ring remodelling [9].
Statistical analysis were performed using the software R (rel. 4.0.2). The Gaussian distribution of continuous variables was assessed by the Shapiro–Wilk test. The study population characteristics were assessed by mean±sd or median (interquartile range), as appropriate. Continuous variables were compared by t-tests or Mann–Whitney tests.
A total of 1098 consecutive patients with COVID-19 pneumonia were reviewed and 54 (4.9%) out of 1098 had an unenhanced CT scan of the thorax and met the eligibility criteria. There was a 44.6% pneumomediastinum-associated mortality rate, making it a severe complication of the disease, whilst the study population mortality rate of hospitalised patients was 22.8% (p-value=0.002).
The median (interquartile range) age of patients with pneumomediastinum was 73 (64–77) years, 40 (74.1%) were males (29.2% smokers), and most patients (n=43, 81.1%) had comorbidities (cardiopathy 52.8%, hypertension 50.9%, obesity 31.4%, diabetes 30.2%, cancer 17.3%, COPD 16.6%, immune depression 3.2%). Pneumomediastinum alone was detected in 40 (74%) patients, while the remaining 14 patients had associated pneumothorax (10 mono-lateral partial, three mono-lateral complete, one bi-lateral pneumothorax). The prevalent lung parenchyma CT scan pattern was ground glass in 20 (37%) patients, alveolar filling in 11 (20.4%) patients, mixed in 10 (18.5%) patients, crazy paving in seven (13%) patients, and reticular/fibrotic pattern in six (11.1%) patients. Spontaneous pneumomediastinum occurred in four (7.4%) patients, one (1.8%) patient received high-flow nasal cannula while they had Pneumomediastinum, 30 (55.6%) patients received noninvasive ventilation/continuous positive airway pressure, and 19 (35.2%) patients were invasively ventilated.
We observed no statistically significant differences regarding clinical characteristics, co-morbidity, and CT scan features between deceased and surviving patients. Only invasive mechanical ventilation was significantly associated with death.
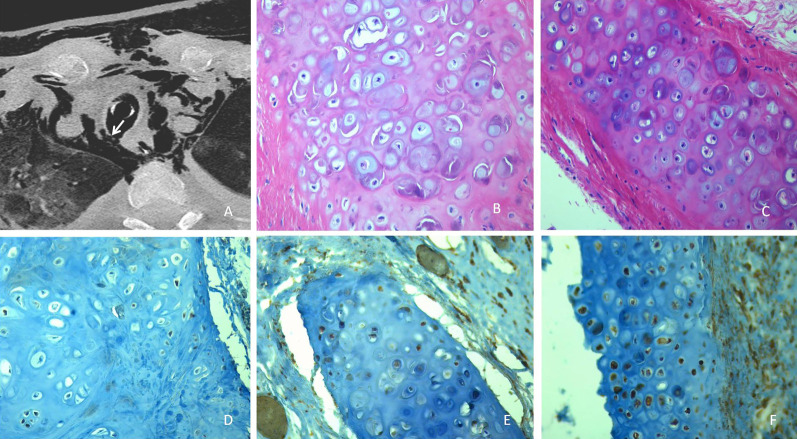
There was a 9 day median duration (IQR 3–13 day) of mechanical ventilation from intubation to pneumomediastinum. An autopsy was carried out on the 23 patients with pneumomediastinum who died found all of them had tracheal/large airway lesions. A control group of eight patients who died of non-COVID-19-ARDS were also pathologically studied. Autopsy showed that most of the COVID-19 pneumomediastinum patients had full thickness tracheal/large airway lesions (29 (53.7%) patients) and two had tracheal fistula (figure 1a), while the remaining 16 pneumomediastinum patients had tracheal mucosal ulcers and/or cartilage lesions. All the autopsies showed peculiar findings of the tracheal/airways cartilage, specifically fibrous-hyaline degeneration (figure 1b and c), that were not present in the trachea and main bronchi of control ARDS subjects (figure 1d). The IHC staining evidenced a strong Wnt5a expression (labelling score 3.69±0.55) and a weak SHH expression (labelling score 2.21±0.90) in the cartilage cells of the tracheal/bronchial rings of the COVID-19 patients with pneumomediastinum (figure 1e). Controls were negative for both Wnt5a and SHH for cartilage regeneration biomarkers (labelling score 0.25±0.46 for both SHH and Wnt5a, p=0.0001).
FIGURE 1.
a) Axial chest computed tomography scan showing a full-thickness tracheal lesion with fistula. b) Tracheal cartilage micro-cracks in a patient with coronavirus disease 2019 (COVID-19) and pneumomediastinum (Haematoxylin and eosin ×40). c) Basophilic cartilage matrix and intracellular fracture with chondrocyte apoptosis (Haematoxylin and eosin ×20). d) Control non-COVID-acute respiratory distress syndrome with absent Sonic Hedgehog (Immunohistochemistry ×20). e) Weak positive Sonic Hedgehog tracheal chondrocytes in a COVID-19 patient (Immunohistochemistry ×20). f) Strong expression of Wnt5a in the same patient (Immunohistochemistry ×20).
Both spontaneous and mechanical ventilation-related pneumomediastinum was observed in our series of COVID-19 patients. Moreover, pneumomediastinum was more isolated than associated to pneumothorax. Autopsies of patients with pneumomediastinum evidenced diffuse tracheal/large airway cartilage lesions with fibrous-hyaline degeneration. Although some of these lesions could represent an iatrogenic effect of mechanical ventilation and a superimposed bacterial infection, no association with intubation or bacterial pneumonia was observed. Therefore, they more likely represent an intrinsic feature of COVID-19.
Other authors already observed an increased pneumomediastinum incidence which was not explainable by barotrauma or high transpulmonary pressure. Our findings are consistent with cartilage remodelling during COVID-19 and ARDS, without a complete post-injury regenerative process [10]. Indeed, two major pathways of chrondrocyte regeneration had a significantly different expression in our autoptic samples, i.e. aberrantly expressed Wnt5a and weakly expressed SHH in the injured cartilage tissue. This is quite different to the usual molecular crosstalk involving Wnt5a and SHH during tracheal development and adult tissue regeneration [7]. In our pneumomediastinum patients tracheal and large airways were frequently injured and an abnormal regenerative process with cartilage tissue remodelling occurred as a pathologic signature, absent in non-COVID-ARDS controls. The fibrous-hyaline degeneration of the tracheal rings seen in our autoptic series is an original observation which has not been previously described in COVID-19 patients.
Acknowledgements
The authors would like to thank Barbara Wade, contract professor at the University of Turin, for her linguistic advice.
Footnotes
Conflict of interest: E. Baratella has nothing to disclose.
Conflict of interest: R. Bussani has nothing to disclose.
Conflict of interest: F. Zanconati has nothing to disclose.
Conflict of interest: C. Marrocchio has nothing to disclose.
Conflict of interest: G. Fabiola has nothing to disclose.
Conflict of interest: L. Braga has nothing to disclose.
Conflict of interest: S. Maiocchi has nothing to disclose.
Conflict of interest: G. Berlot has nothing to disclose.
Conflict of interest: M.C. Volpe has nothing to disclose.
Conflict of interest: E. Moro has nothing to disclose.
Conflict of interest: P. Confalonieri has nothing to disclose.
Conflict of interest: M.A. Cova has nothing to disclose.
Conflict of interest: M. Confalonieri has nothing to disclose.
Conflict of interest: F. Salton has nothing to disclose.
Conflict of interest: B. Ruaro has nothing to disclose.
Submitted article, peer reviewed.
References
- 1.Mart MF, Norfolk SG, Flemmons LN, et al. . Pneumomediastinum in acute respiratory distress syndrome from COVID-19. Am J Respir Crit Care Med 2021; 203: 237–238. doi: 10.1164/rccm.202008-3376IM [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lemmers DHL, Hilal MA, Bnà C, et al. . Pneumomediastinum and subcutanneous emphysema in COVID-19: barotrauma or lung frailty? ERJ Open Res 2020; 6: 00385-2020. doi: 10.1183/23120541.00385-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Diaz A, Patel D, Sayedy N, et al. . COVID-19 and spontaneous pneumomediastinum: a case series. Heart Lung 2021; 50: 202–205. doi: 10.1016/j.hrtlng.2020.12.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chu CM, Leung YY, Hui JYH, et al. . Spontaneous pneumomediastinum in patients with severe acute respiratory syndrome. Eur Respir J 2004; 23: 802–804. doi: 10.1183/09031936.04.00096404 [DOI] [PubMed] [Google Scholar]
- 5.Fiacchini G, Tric D, Ribechini A, et al. . Evaluation of the incidence and potential mechanisms of tracheal complications in patients with COVID-19. JAMA Otolaryngol Head Neck Surg 2021; 147: 70–76. doi: 10.1001/jamaoto.2020.4148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Frank DB, Morrisey EE. Hedgehog and Wnt signaling hubs in tracheal morphogenesis. Am J Respir Crit Care Med 2019; 200: 1202–1204. doi: 10.1164/rccm.201907-1285ED [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kiyokawa H, Morimoto M. Molecular crosstalk in tracheal development and its recurrence in adult tissue regeneration. Dev Dyn 2021; in press [ 10.1002/dvdy.345]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Geri P, Salton F, Zuccatosta L, et al. . Limited role for bronchoalveolar lavage to exclude COVID-19 after negative upper respiratory tract swabs: a multicentre study. Eur Respir J 2020; 56: 2001733. doi: 10.1183/13993003.01733-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fedchenko N, Reifenrath J. Different approaches for interpretation and reporting of immunohistochemistry analysis results in the bone tissue – a review. Diagn Pathol 2014; 9: 221. doi: 10.1186/s13000-014-0221-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nasr T, Holderbaum AM, Chaturvedi P, et al. . Disruption of a Hedgehog-Foxf1-Rspo2 signaling axis leads to tracheomalacia and loss of Sox9+ tracheal chondrocytes. Dis Mod Mech 2021; 14: dmm046573. doi: 10.1242/dmm.046573 [DOI] [PMC free article] [PubMed] [Google Scholar]