Supplemental Digital Content is available in the text.
Keywords: coronavirus disease 2019, dysregulated immune response, hyperinflammatory syndrome, plasma exchange, rescue therapy
Abstract
IMPORTANCE:
Recent evidence suggests a multilevel inflammatory syndrome as a driving factor in some of the most severely ill coronavirus disease 2019 patients with overlapping features to other hyperinflammatory or autoimmune diseases. Therefore, plasma exchange is considered as potential therapy in these patients.
OBJECTIVES:
We characterize the longitudinal therapeutic efficacy and safety profile of plasma exchange in critically ill patients with clinical and laboratory evidences of coronavirus disease 2019–related immunopathology.
DESIGN, SETTING, AND PARTICIPANTS:
A retropsective case-control study of critically ill coronavirus disease 2019 patients treated with plasma exchange at Heidelberg University Hospital between March and December 2020. Plasma exchange–treated patients were compared with coronavirus disease 2019 patients on standard therapy matched for age, gender, disease severity, and features of hyperinflammatory syndrome.
MAIN OUTCOME AND MEASURES:
Mortality rate and course of clinical and laboratory parameters in response to plasma exchange were assessed in coronavirus disease 2019 patients and in patients on standard care. A plasma volume of 50 mL per kg body weight or a maximum of 4 L was exchanged.
RESULTS:
In total, 28 critically ill coronavirus disease 2019 patients were treated with a median of three plasma exchange procedures per patient. No relevant complications occurred during plasma exchange therapy. Inflammatory and biochemical markers of end-organ damage and endothelial activation were significantly reduced following plasma exchange together with normalization of body temperature, improved pulmonary function, and reduced vasopressor demand. Most importantly, these improvements were maintained after the last plasma exchange. In contrast, no such effects were observed in the control group, although baseline clinical and laboratory parameters were comparable. Kaplan-Meier analysis showed improved 30-day survival in the plasma exchange group compared with the control group (67.9% vs 42.9%; p = 0.044). In a multivariable analysis, the hazard ratio for death was 0.27 (95% CI, 0.11–0.68; p = 0.005) with plasma exchange versus standard care.
CONCLUSIONS AND RELEVANCE:
Our data provide further evidence for plasma exchange as a novel therapeutic strategy in a subset of critically ill coronavirus disease 2019 patients by potentially reversing the complex coronavirus disease 2019 immunopathology. Randomized controlled trials are underway to confirm these positive results.
The severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2)–related pandemic is a heavy burden for healthcare providers and ICUs around the globe. Resulting coronavirus disease 2019 (COVID-19) ranges from mild or even asymptomatic infection to critical illness with acute respiratory distress syndrome (ARDS), multiple organ failure, and death (1, 2). In particular, the presence of acute kidney injury (AKI) and the need for mechanical ventilation substantially increase the risk for death with mortality rates ranging from 50% to 67% (3–5). In these critically ill patients, a dysregulated hyperinflammatory syndrome that follows the initial phase of viral replication is discussed as a potential disease-exacerbating driver (6, 7). Underlying mechanisms are thought to be a dysregulated innate and adaptive immune response, formation of (prothrombotic) autoantibodies, a hyperinflammatory cytokine storm-like state, and an endothelial and complement dysfunction with microcirculatory clot formation (6, 8–11). Plasma exchange (PE) is discussed as potential rescue therapy for severe COVID-19 since it is removing inflammatory mediators, balancing the hypercoagulable state, stabilizing endothelial membranes, and potentially eliminating harmful autoantibodies (11–13). Furthermore, PE has been proven safe and beneficial to other hyperinflammatory conditions such as sepsis, hemophagocytic lymphohistiocytosis, and influenza (14–16).
In this context, we and others have recently reported the first data on improved disease outcomes in response to PE treatment, showing effective reduction of inflammatory markers associated with reversal of organ failure and the first evidence of improved survival (17–19). However, there exists still controversy as to whether PE is associated only with an artificial reduction of laboratory parameters (“treating the numbers”) without any clinical relevance, but a possible impairment of the adaptive immune response by removing protective immunoglobulins, or whether PE is reversing the inflammatory syndrome with improvement of clinical outcomes (12, 13, 20). To address these valid objections, we have now analyzed the disease courses and outcomes of all critically ill COVID-19 patients treated with PE at our center and compared them with COVID-19 patients on standard care.
MATERIAL AND METHODS
Study Design and Patient Population
All critically ill COVID-19 patients who gave their written informed consent were included in a prospective register study. The study was approved by the local Ethics Committee of the Medical Faculty of Heidelberg (S148/2020). Severe COVID-19 cases were considered for PE in the presence of the criteria of invasive mechanical ventilation and ARDS, and/or vasopressor-dependent circulatory shock, and clinical and laboratory features of a COVID-19-associated immunopathology with elevated d-dimers and ferritin levels, and persistent and refractory fever greater than or equal to 38.5°C without conclusive pathogenic evidence and despite anti-infectious treatment. All COVID-19 patients treated with PE on the ICUs of Heidelberg University Hospital between March and December 2020 were retrospectively evaluated for the present analyses and compared with a control group of patients with severe COVID-19 on standard care matched for age, gender, disease severity (Sequential Organ Failure Assessment [SOFA] score and extent of AKI), and features of COVID-19-associated hyperinflammatory syndrome. Two reasons led to patients in the control group not being treated with PE. First, patients or their legal representatives agreed to participate in the registry study but did not give consent for PE therapy. Second, we first started PE treatments in patients who already had a central venous catheter in place (need for renal replacement therapy [RRT]), which caused us to miss patients with a comparable disease severity and symptom complex around that time. SARS-CoV-2 infection was detected by a positive nasopharyngeal swab reverse transcription polymerase chain reaction. Prophylactic antibiotic and antifungal therapies were initiated in all patients according to the center’s standards and subsequently adjusted for current pathogen evidence. PE was performed using a “Comtech” centrifuge (Fresenius Medical Care AG & Co. KGaA, Bad Homburg, Germany) and against fresh frozen plasma, exchanging a plasma volume of 50 mL/kg of body weight or a maximum of 4,000 mL per treatment. The “Comtech” is a continuous-flow centrifugation blood cell separator with blood flow rates ranging from 45 to 55 mL/min, resulting in a plasma flow rate between 30 and 34 mL/min. The blood cells are separated from the plasma by gravity based on the different densities of the blood components. Regional citrate anticoagulation was used as standard anticoagulation. Heparin was used only (as add-on) in patients with a preexisting indication for systematic anticoagulation. All PE procedures were performed in accordance with the German Medical Devices Act (“Medizinproduktegesetz”) and the corresponding “Conformité Européenne” certificate of the “Comtech” centrifuge.
Data Collection and Analysis
Patient characteristics and presented laboratory and clinical parameters were obtained from medical records. The individual laboratory parameters were taken from daily routine blood results. Changes in laboratory and clinical parameters related to PE treatment were evaluated before the first, after the first, after the third, and after the last PE procedure. To assess PE-related long-term effects, the same parameters were evaluated 3 and 5 days after the last PE. Observation of laboratory and clinical parameters in the control group was started when patients showed the same potential indicators of a dysregulated COVID-19-related immune response. In the control group, laboratory and clinical parameters were observed for 7 consecutive days. All laboratory parameters were measured in the accredited Central Laboratory of the Heidelberg University Hospital.
Statistical Analysis
Statistical analyses were performed using SPSS Statistics 25 (IBM, Armonk, NY) and Graph Pad Prism 9 (GraphPad Software, La Jolla, CA). Continuous variables are presented as median (interquartile range); categorical data are presented as absolute numbers (percentages). Mann-Whitney U test was used for group comparisons. Categorical variables were analyzed using chi-square test. Kruskal-Wallis test was used for multiple group comparisons. Survival was evaluated by the Kaplan-Meier method. Multivariable Cox regression models were performed to determine the hazard ratios (HRs) and 95% CIs of individual factors on death within 15 and 30 days after ICU admission. Correlations were assessed by using Spearman correlation analysis. Two-tailed p values equal to or less than 0.05 were considered statistically significant.
RESULTS
Patient Characteristics
A total of 28 patients with severe COVID-19 were treated with PE in our ICUs and compared with 21 critically ill COVID-19 patients on standard therapy. Patient characteristics are described in Table 1. In both groups, patients showed the typical comorbidities associated with severe courses of COVID-19 such as hypertension, diabetes, coronary heart disease, chronic kidney disease (CKD), and obesity. Except for CKD, preexisting kidney failure, and peripheral vascular disease in the PE group, underlying comorbidities, SOFA score, and vasopressor demand were comparable between both groups. All patients were mechanically ventilated and suffered from multiple organ failure at baseline. Twenty-seven of 28 patients (96.4%) received corticosteroids in the PE group and 19 of 21 patients (90.5%) in the control group. Remdesivir was given in nine patients (32.1%) in the PE group and two patients (9.5%) in the control group. Tocilizumab was administered in one patient (3.6%) in the PE group and three patients (14.3%) in the control group. According to the center standard, all patients with ARDS were empirically treated with piperacillin-tazobactam and received a prophylactic antifungal therapy with caspofungin. Anti-infective therapy was adjusted according to microbiological evidence.
TABLE 1.
Patient Characteristics and Outcome
| Baseline Characteristics | COVID-19 Patients PE Group (n = 28) | COVID-19 Patients Control Group (n = 21) | p |
|---|---|---|---|
| Age (yr) | 66.0 (57.3–72.8) | 66.0 (59.5–77.5) | > 0.9999 |
| Male gender, n (%) | 22 (78.6) | 14 (66.7) | 0.350 |
| BMI (kg/m2) | 31.3 (25.7–39.5) | 29.4 (27.3–33.3) | 0.976 |
| Underlying comorbidity, n (%) | |||
| Obesity (BMI > 30 kg/m2) | 16 (57.1) | 9 (42.9) | 0.458 |
| Diabetes | 12 (42.9) | 7 (33.3) | 0.498 |
| Hypertension | 20 (71.4) | 16 (76.2) | 0.709 |
| Congestive heart failure | 2 (7.1) | 2 (9.5) | 0.763 |
| Coronary heart disease | 12 (42.8) | 5 (23.8) | 0.166 |
| Peripheral vascular disease | 5 (17.9) | 0 (0.0) | 0.041 |
| Chronic kidney disease | 11 (39.3) | 2 (9.5) | 0.020 |
| Preexisting kidney failure | 6 (21.4) | 2 (9.5) | < 0.0001 |
| Chronic obstructive pulmonary disease | 1 (3.4) | 0 (0.0) | 0.382 |
| Cancer | 3 (10.7) | 2 (9.5) | 0.892 |
| Sequential Organ Failure Assessment score before start of PE or observation, value (range) | 13.5 (12.0–15.8) | 12.0 (10.0–14.0) | 0.122 |
| Time between ICU admission and start of PE or observation, d (range) | 6.0 (1.3–9.0) | 5.0 (2.5–7.0) | 0.665 |
| Vasopressors before start of PE or observation, n (%) | 20 (71.4) | 14 (66.7) | 0.720 |
| Invasive mechanical ventilation, n (%) | 28 (100) | 21 (100) | > 0.9999 |
| Acute kidney injury, n (%) | 20 (71.4) | 16 (76.2) | 0.513 |
| KDIGO stage 0 | 2 (7.1) | 3 (14.3) | 0.513 |
| KDIGO stage 1 | 0 (0) | 1 (4.8) | 0.276 |
| KDIGO stage 2 | 2 (7.1) | 2 (9.5) | 0.877 |
| KDIGO stage 3 | 18 (67.9) | 13 (61.9) | 0.470 |
| Renal replacement therapy, n (%) | 20 (71.4) | 12 (57.1) | 0.299 |
| Length of ICU stay (days) | 26 (14.5–54.0) | 18 (9.5–33.5) | 0.031 |
BMI = body mass index, COVID-19 = coronavirus disease 2019, KDIGO = Kidney Disease Improving Global Outcomes, n = number, PE = plasma exchange.
Results are given as number (percentage) or median (interquartile range). Bold values are statistically significant for p ≤ 0.05.
Plasma Exchange and Therapy-Associated Complications
In total, 98 PE procedures were performed in 28 patients, corresponding to an average of 3.0 procedures (2.0–5.0) per patient. Overall, PE treatments were well tolerated. Therapy-associated complications were mainly mild reductions of ionized calcium levels, which were immediately corrected throughout the PE procedure. No serious complications such as anaphylactic shock, cardiac arrhythmias, transfusion reactions, or bleeding occurred in treated patients. PE therapy-related information is summarized in Table 2.
TABLE 2.
Plasma Exchange and Associated Complications
| PE-Related Parameters | Values Recorded |
|---|---|
| Total number of PE treatments (n) | 98 |
| Number of PE procedures per patient (n) | 3.0 (2.0–5.0) |
| Treatment-free days between PE procedures | 1.0 (0.0–2.0) |
| Exchanged plasma volume per treatment (L) | 3.6 (3.2–3.9) |
| PE treatments with reported complications, n (%) | 85 (86.7) |
| Anaphylactic reactions | 0 (0) |
| Transfusion reactions | 0 (0) |
| Hypotension | 0 (0) |
| Hypocalcemia | 85 (86.7) |
| Min. ionized serum calcium (mmol/L) | 1.06 (1.01–1.12) |
| Cardiac arrhythmia | 0 (0) |
| Bleeding | 0 (0) |
n = number, PE = plasma exchange.
Results are given as number (percentage) or median (interquartile range).
Disease Courses in Patients on Standard Care Versus Patients With Additional Plasma Exchange
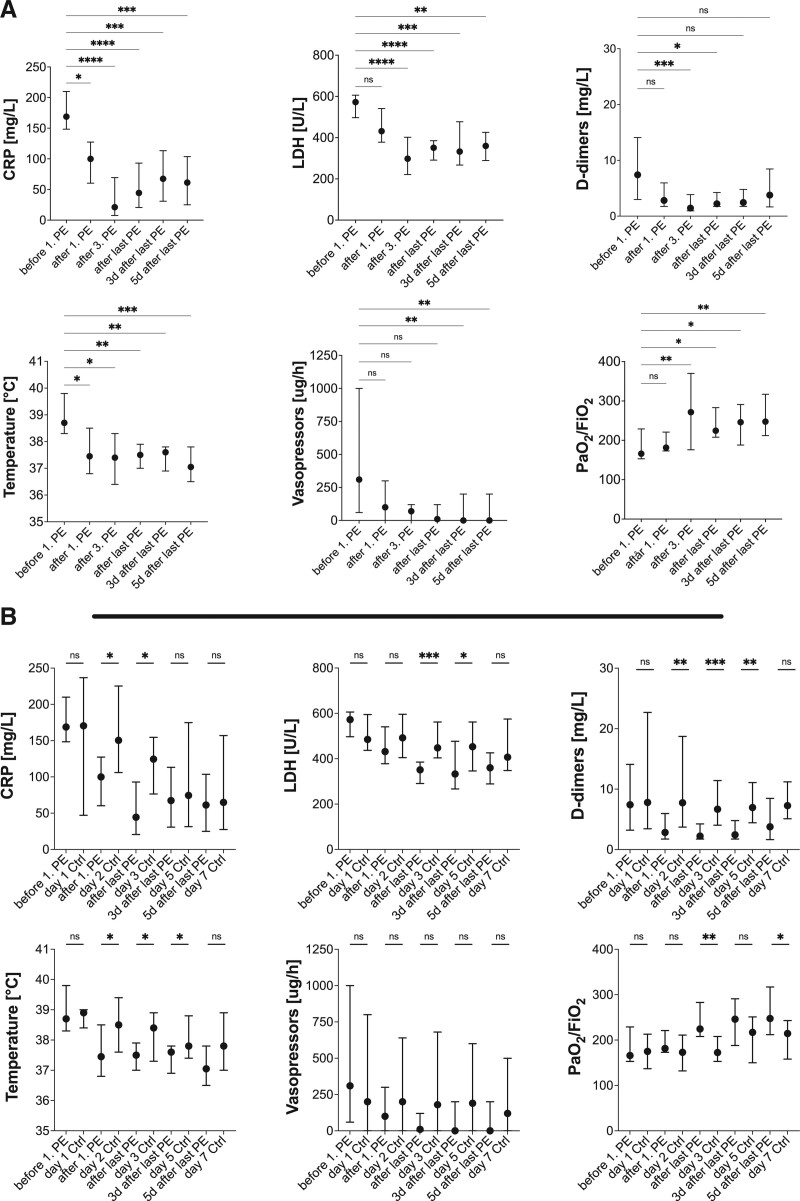
At baseline, patients in both groups showed typical laboratory and clinical signs of a severe systemic inflammatory immune response such as elevated levels of C-reactive protein (CRP), leukocytes and ferritin, refractory fever, and vasopressor-dependent circulatory shock. The time between ICU admission and first PE treatment or start of observation was comparable with 6.0 and 5.0 days (Table 1), respectively. The clinical and laboratory parameters measured were comparable at the time of PE initiation or start of observation in the respective groups (Supplemental Fig. S1, http://links.lww.com/CCX/A760). All patients suffered from ARDS, which was associated with a reduced Pao2/Fio2 ratio (Fig. 1A; Supplemental Fig. S1, http://links.lww.com/CCX/A760). d-dimers, a prognostic marker in COVID-19 and an indicator of ongoing coagulation and endothelial activation, were elevated along with the lactate dehydrogenase (LDH) and troponin T (TnT) levels in both groups (Fig. 1A; Supplemental Fig. S2, http://links.lww.com/CCX/A760). Upon PE initiation, serum levels of CRP, ferritin, LDH, and d-dimers were significantly reduced over time, whereas leukocyte count and TnT levels were not significantly affected (Fig. 1A; Supplemental Fig. S2, http://links.lww.com/CCX/A760). Remarkably, the observed laboratory changes in the PE group were accompanied by clinical improvement in the form of significantly reduced vasopressor demand and body temperature, and improved pulmonary function with an increased Pao2/Fio2 ratio. These significant laboratory and clinical improvements persisted several days beyond the last PE procedure, as shown in Figure 1A. Accordingly, CRP, LDH, and d-dimers levels after the last PE correlated negatively with future Pao2/Fio2 levels, demonstrating an association between improvement of these biomarkers by PE treatment and subsequent pulmonary improvement (Supplemental Table S1, http://links.lww.com/CCX/A760). A comparable pattern was observed for vasopressor demand after the last PE and future Pao2/Fio2 ratio at days 3 and 5 after the last PE (Supplemental Table S1, http://links.lww.com/CCX/A760). In contrast, the same biochemical and clinical parameters did not significantly improve over 7 days compared to baseline in the control group (Supplemental Fig. S2, http://links.lww.com/CCX/A760). Only an improvement in body temperature and CRP level was observed in the controls; the latter, however, did not reach statistical significance. Furthermore, direct comparison of laboratory and clinical parameters between the PE and control groups showed significant improvement in CRP, LDH, ferritin, d-dimers, temperature, and Pao2/Fio2 after the last PE compared with the same time-matched parameters in the control group (Fig. 1B; Supplemental Fig. S3, http://links.lww.com/CCX/A760). These improvements persisted after the last PE and compared with controls for LDH, d-dimers, body temperature, and Pao2/Fio2, once again indicating an improved condition of the patient in response to PE compared with controls. We also observed a trend toward reduced vasopressor requirements compared with controls 3 and 5 days after the last PE, barely missing statistical significance (Fig. 1B; Supplemental Fig. S3, http://links.lww.com/CCX/A760).
Figure 1.
Longitudinal analysis of laboratory and clinical parameters. Course of laboratory and clinical parameters in relation to plasma exchange procedures (A) and time-matched comparison of parameters between patients treated with plasma exchange and controls on standard care (B). Results are given as median and 95% CI. Kruskal-Wallis test was used for statistical analyses. Not significant: p > 0.05, *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001, ****p ≤ 0.0001. CRP = C-reactive protein, LDH = lactate dehydrogenase, PE = plasma exchange.
Outcomes in Patients on Standard Care and Patients With Additional Plasma Exchange
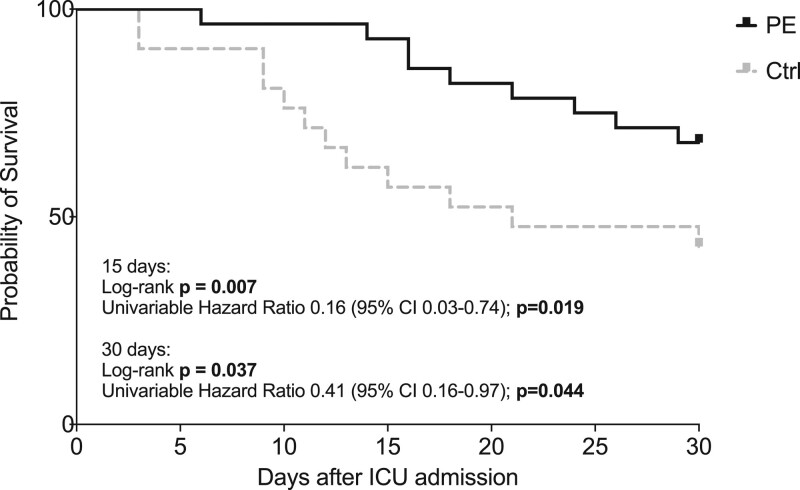
AKI severity and frequency of RRT were comparable between both groups. Two (7.1%) and nine (32.1%) patients in the PE group and eight (38.1%) and 12 (57.1%) in the control group had died by days 15 and 30, respectively, after ICU admission. As a result of the higher mortality, the length of ICU stay was significantly shorter in the control group. Kaplan-Meier analysis revealed a survival benefit within 15 and 30 days after ICU admission for patients treated with PE compared with standard care (Fig. 2). Univariable HR for death within 15 or 30 days after ICU admission in the PE group was 0.16 (95% CI, 0.03–0.74; p = 0.019) and 0.41 (95% CI, 0.16–0.97; p = 0.044), respectively. Even after adjustment for gender, age, and disease severity (SOFA score), risk for death was lower in PE-treated patients than in patients receiving standard care (Table 3).
Figure 2.
Kaplan-Meier curves for survival at days 15 and 30 after ICU admission in the plasma exchange and control group. Bold values are statistically significant for p ≤ 0.05. Ctrl = control group, PE = plasma exchange group.
TABLE 3.
Multivariable Cox Regression Analysis for Death in Patients With Severe Coronavirus Disease 2019 Treated With Plasma Exchange Versus Control Group
| Variables | Multivariable Model 15-d Mortality | Multivariable Model 30-d Mortality | ||
|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | |
| Plasma exchange treatment | 0.09 (0.02–0.44) | 0.003 | 0.27 (0.11–0.68) | 0.005 |
| Male gender | 6.37 (0.74–54.49) | 0.091 | 1.99 (0.70–5.66) | 0.198 |
| Age ≥ 70 yr | 1.75 (0.48–6.38) | 0.091 | 1.46 (0.60–3.55) | 0.405 |
| Sequential Organ Failure Assessment score | 1.43 (1.02–2.01) | 0.040 | 1.26 (1.04–1.52) | 0.016 |
HR = hazard ratio.
Bold values are statistically significant for p ≤ 0.05.
DISCUSSION
Here, we report critically ill patients with severe COVID-19 in whom PE was considered as add-on therapy in cases with clinical and laboratory evidences of dysregulated immune response syndrome. Patients treated with PE were compared with COVID-19 patients on standard care with similar clinical and laboratory features. Inflammatory markers such as CRP and ferritin, as well as indicators of end-organ damage and endothelial activation, more specifically LDH and d-dimers, were significantly reduced during PE. At the same time, these laboratory changes were accompanied by striking clinical improvement with normalization of body temperature, improved pulmonary function, and a reduced vasopressor demand. Improved levels of CRP and LDH persisted for several days after the last PE, along with persistent normalization of body temperature, reduction in vasopressor demand, and reduced Fio2/Pao2 ratio. Furthermore, reduced CRP, LDH, and d-dimer levels after the last PE correlated highly with future improvement of pulmonary function 3 and 5 days after the last PE. Most importantly, no relevant complications occurred during PE procedures. At the same time, relevant clinical and laboratory parameters did not improve within a 7-day observation period compared to a control cohort matched for age, sex, and disease severity. Furthermore, PE treatment resulted in a survival benefit within 15 and 30 days after ICU admission compared with standard treatment, and risk for death was significantly lower in PE-treated patients even after adjustment for age, gender, and disease severity.
In summary, these observations suggest a beneficial and persistent effect of PE on COVID-19 pathomechanisms in critically ill patients, which seems to go beyond a mere artificial reduction of laboratory parameters, as speculated by some authors (20).
Great efforts have been made in numerous clinical trials to develop new therapeutic strategies to improve outcomes in patients with severe COVID-19 (5). They include specific antiviral therapy in the form of remdesivir, nonspecific immunomodulatory, or immunosuppressive therapies such as hydroxychloroquine, corticosteroids, and specific cytokine blockade such as tocilizumab. Although corticosteroids or treatment with remdesivir or the combination of remdesivir and baricitinib has revealed some beneficial effects by reducing the risk of death in ARDS patients or shortening the median time to recovery, other therapeutics have provided inconsistent results or were potentially related to adverse events as with hydroxychloroquine (5, 21). Meanwhile, mortality rates in mechanically ventilated patients remain as high as 50–67%, and the chances for death are further aggravated by the presence of severe AKI (HR, 9.8; 95% CI, 5.5–17.7; p < 0.001), highlighting the need for new therapeutic strategies for severe COVID-19 cases (22–25). In this context, there is increasing evidence that intensivists are dealing with a complex immunopathology rather than a classical viral disease (6, 7). The observed inflammatory syndrome shares overlapping features with other hyperinflammatory or autoimmune diseases such as viral-induced hemophagocytic lymphohistiocytosis, thrombotic microangiopathies, and antiphospholipid syndrome (6, 8, 15, 26). A cytokine storm-like state with dysregulated innate immune response and endothelial and complement dysfunctions together with a thrombogenic milieu with formation of microcirculatory clots dominates the clinical picture and leads to ARDS and multiple organ failure (6, 8–10). Theoretically, PE provides benefits to all of these pathogenic levels by restoring deficient blood components, eliminating harmful (auto)antibodies and inflammatory mediators, and counter acting exuberant release of coagulation factors and endothelial activation markers (12). The complexity of these deregulated processes is obvious from the example of COVID-19-related coagulopathy. A very diverse set of pathogenic drivers has been identified in this context including a potential deficiency of antithrombin and disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS-13), increased levels of von Willebrand factor multimer and factor VIII, and formation of prothrombotic antibodies (anti-ADAMTS-13, antiphospholipid) (8, 27–30). Thus, the overall multidimensional nature of COVID-19-related immunopathology may further explain why single-target therapies such as tocilizumab are less effective in COVID-19 compared with that, for example, in cytokine release syndrome after chimeric antigen receptor-T cell therapy with specified pathogenesis and significantly higher interleukin (IL)-6 levels (31–33). In addition, it is noteworthy that little is known about chemokines that regulate the distribution and activity of effector immune cell populations in COVID-19 patients (34). The demonstrated elevation of immunosuppressive cytokines in these patients, such as IL-10, may therefore be a rationale for opposing therapeutic approaches that restore immune cell paralysis rather than hampering the innate and adaptive immune systems through cytokine blockade or removal (34). Among others, this immunosuppressive state in COVID-19 patients is associated with decreased lymphocyte count and impaired type 1 interferon response (6, 34). Since autoantibodies against type I interferons have recently been detected in patients with life-threatening COVID-19, PE could also be a rational therapeutic approach under these circumstances by eliminating anti-interferon antibodies and immunosuppressive cytokines (11). In line with this, Faqihi et al (35) recently reported improved lymphocyte numbers in response to PE. However, their findings may be limited by the fact that they studied lymphocyte counts only in survivors and within a disproportionately large time period (days 2 vs 35 after ICU admission). This excludes patients with the potentially lowest lymphocyte counts from their analysis and leaves room for other underlying mechanisms besides PE to influence lymphocyte population. In contrast to the study by Faqihi et al (35), we could not demonstrate a clear increase of lymphocytes around the time when PE was applied (Supplemental Figs. S2 and S3, http://links.lww.com/CCX/A760).
The use of PE in COVID-19, however, remains highly controversial, and one of the most legitimate objections is that functional immune response may be attenuated by removing protective antibodies (20). We therefore initiated PE rather “late” in the presence of ARDS and COVID-19-related immune response syndrome despite standard care. Interestingly, however, large randomized controlled trials (RCTs) have recently failed to show any improvements in survival or other prespecified outcomes in ventilated patients by administration of convalescent plasma containing large amounts of anti-SARS-CoV-2 antibody. This may indicate a dominant role of COVID-19-related immunopathology as the main disease-exacerbating driver, which cannot be sufficiently influenced by antibodies directed against SARS-CoV-2 at this disease stage (36). The latter observation also weakens the claim for the use of convalescent plasma for PE therapy.
Based on our longitudinal data, we now strengthen the concept of PE as therapeutic approach in a large cohort of critically ill COVID-19 patients by showing beneficial effects on survival and that laboratory and clinical improvements persist beyond the last PE treatment. Most importantly, we have shown that PE is a safe procedure in ventilated COVID-19 patients with evidence of hyperinflammatory syndrome. Taken together, our data suggest that pathogenic immunological drivers are more likely to be disrupted by PE than antibody-mediated immune responses in critically ill COVID-19 patients. This also contradicts an earlier criticism that PE reduces only inflammatory mediators and levels of LDH, and d-dimers based on their molecular weight, implying an artificial reduction rather than a true improvement in the patient’s condition. In addition, our results are in agreement with a smaller retrospective study by Gucyetmez et al (19), in which the authors demonstrated a beneficial effect of PE by showing reduced mortality rates in patients with d-dimers greater than or equal to 2 mg/L (16.7%) on PE therapy when compared with patients with d-dimers greater than or equal to 2 mg/L without PE (45.7%). Despite the higher disease severity in our cohort, our data further support this idea of improved survival via PE by showing a 30-day mortality rate of only 32.1% in PE-treated patients compared with 57.1% under standard care. Finally, another large study showed a trend toward improved survival with the use of PE in critically ill COVID-19 patients with features of a hyperinflammatory syndrome but did not reach statistical significance (35). The study differed from ours, however, in an earlier PE start in patients with lower disease severity (SOFA score and RRT) and the absence of clinical criteria such as fever for PE initiation.
Nonetheless, based on current evidence, PE cannot be recommended as standard therapeutic approach in critically ill patients but may represent a therapeutic alternative in the most severe COVID-19 cases with ARDS, kidney failure, and features of hyperinflammatory syndrome. In addition, though our data show beneficial effects of PE on the disease course in a subset of critically ill COVID-19 patients, it is still unclear whether PE therapy may impair the functionality of antiviral immune response by eliminating protective antibodies in earlier disease phases.
Some limitations of our study must be noted. Remdesivir was used to a greater extent in the PE group than in the control group and, therefore, may be partially responsible for the observed differences in outcomes. However, we consider this very unlikely, as no comparable effect was shown in the numerous RCTs on remdesivir, and the only study that included a relevant number of ventilated patients could not find any beneficial therapeutic effect compared to the placebo group (37). The main limitation of our presented analyses is the retrospective nature and sample size. The latter, however, can be explained by the fact that PE indication was made only in a specific subset of critically ill COVID-19 patients, and restrictive criteria for PE initiation were used. Finally, the sample size did not allow meaningful use of methodological approaches (e.g., propensity score matching) to control for confounding by indication, and other unmeasured confounders may have influenced the observed results. Nevertheless, it is worth noting that outcomes were superior in the PE group compared with the control group, despite the tendency for greater disease severity in the PE group.
To address the limitations mentioned above and to further evaluate the efficacy and safety of PE as a novel therapeutic strategy for severe COVID-19 cases with features of dysregulated immune response, we recently initiated an RCT, the “RescuE pLAsma eXchange in Severe COVID-19 trial” (NCT04685655).
CONCLUSIONS
In conclusion, our data suggest PE once more as a promising and safe therapeutic approach for a subset of patients with severe COVID-19 courses by reversing the multilevel inflammatory syndrome. Further research is urgently needed to improve our understanding of ongoing pathomechanisms in these patients.
Supplementary Material
Footnotes
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal’s website (http://journals.lww.com/ccejournal).
Dr. Nusstag is funded by the Physician Scientist Programme of Heidelberg Faculty of Medicine. The remaining authors have disclosed that they do not have any potential conflicts of interest.
REFERENCES
- 1.Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet. 2020; 395:1054–1062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Suleyman G, Fadel RA, Malette KM, et al. Clinical characteristics and morbidity associated with coronavirus disease 2019 in a series of patients in metropolitan Detroit. JAMA Netw Open. 2020; 3:e2012270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cheng Y, Luo R, Wang K, et al. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020; 97:829–838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chan L, Chaudhary K, Saha A, et al. AKI in hospitalized patients with COVID-19. J Am Soc Nephrol. 2020; 32:151–160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huang C, Soleimani J, Herasevich S, et al. Clinical characteristics, treatment, and outcomes of critically ill patients with COVID-19: A scoping review. Mayo Clin Proc. 2021; 96:183–202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gustine JN, Jones D. Immunopathology of hyperinflammation in COVID-19. Am J Pathol. 2021; 191:4–17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cao X. COVID-19: Immunopathology and its implications for therapy. Nat Rev Immunol. 2020; 20:269–270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zuo Y, Estes SK, Ali RA, et al. Prothrombotic autoantibodies in serum from patients hospitalized with COVID-19. Sci Transl Med. 2020; 12:eabd3876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.McFadyen JD, Stevens H, Peter K. The emerging threat of (micro)thrombosis in COVID-19 and its therapeutic implications. Circ Res. 2020; 127:571–587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mehta P, McAuley DF, Brown M, et al. ; HLH Across Speciality Collaboration, UK. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet. 2020; 395:1033–1034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bastard P, Rosen LB, Zhang Q, et al. ; HGID Lab; NIAID-USUHS Immune Response to COVID Group; COVID Clinicians; COVID-STORM Clinicians; Imagine COVID Group; French COVID Cohort Study Group; Milieu Intérieur Consortium; CoV-Contact Cohort; Amsterdam UMC Covid-19 Biobank; COVID Human Genetic Effort. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science. 2020; 370:eabd4585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Keith P, Day M, Perkins L, et al. A novel treatment approach to the novel coronavirus: An argument for the use of therapeutic plasma exchange for fulminant COVID-19. Crit Care. 2020; 24:128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tabibi S, Tabibi T, Conic RRZ, et al. Therapeutic plasma exchange: A potential management strategy for critically ill COVID-19 patients. J Intensive Care Med. 2020; 35:827–835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Knaup H, Stahl K, Schmidt BMW, et al. Early therapeutic plasma exchange in septic shock: A prospective open-label nonrandomized pilot study focusing on safety, hemodynamics, vascular barrier function, and biologic markers. Crit Care. 2018; 22:285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nusshag C, Morath C, Zeier M, et al. Hemophagocytic lymphohistiocytosis in an adult kidney transplant recipient successfully treated by plasmapheresis: A case report and review of the literature. Medicine (Baltimore). 2017; 96:e9283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Patel P, Nandwani V, Vanchiere J, et al. Use of therapeutic plasma exchange as a rescue therapy in 2009 pH1N1 influenza A—An associated respiratory failure and hemodynamic shock. Pediatr Crit Care Me. 2011; 12:e87–e89 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Morath C, Weigand MA, Zeier M, et al. Plasma exchange in critically ill COVID-19 patients. Crit Care. 2020; 24:481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fernandez J, Gratacos-Ginès J, Olivas P, et al. Plasma exchange: An effective rescue therapy in critically ill patients with coronavirus disease 2019 infection. Crit Care Med. 2020; 48:e1350–e1355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gucyetmez B, Atalan HK, Sertdemir I, et al. ; COVID-19 Study Group. Therapeutic plasma exchange in patients with COVID-19 pneumonia in intensive care unit: A retrospective study. Crit Care. 2020; 24:492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Honore PM, Barreto Gutierrez L, Kugener L, et al. Plasma exchange in critically ill COVID-19 patients improved inflammation, microcirculatory clot formation, and hypotension, thereby improving clinical outcomes: Fact or fiction? Crit Care. 2020; 24:551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kalil AC, Patterson TF, Mehta AK, et al. ; ACTT-2 Study Group Members. Baricitinib plus remdesivir for hospitalized adults with Covid-19. N Engl J Med. 2021; 384:795–807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang Y, Lu X, Li Y, et al. Clinical course and outcomes of 344 intensive care patients with COVID-19. Am J Respir Crit Care Med. 2020; 201:1430–1434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Arentz M, Yim E, Klaff L, et al. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State. JAMA. 2020; 323:1612–1614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Myers LC, Parodi SM, Escobar GJ, et al. Characteristics of hospitalized adults with COVID-19 in an integrated health care system in California. JAMA. 2020; 323:2195–2198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020; 180:934–943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Merrill JT, Erkan D, Winakur J, et al. Emerging evidence of a COVID-19 thrombotic syndrome has treatment implications. Nat Rev Rheumatol. 2020; 16:581–589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lippi G, Henry BM, Sanchis-Gomar F. Plasma antithrombin values are significantly decreased in coronavirus disease 2019 (COVID-19) patients with severe illness. Semin Thromb Hemost. 2021; 47:460–462 [DOI] [PubMed] [Google Scholar]
- 28.Doevelaar AAN, Bachmann M, Hölzer B, et al. von Willebrand factor multimer formation contributes to immunothrombosis in coronavirus disease 2019. Crit Care Med. 2021; 49:e512–e520 [DOI] [PubMed] [Google Scholar]
- 29.Goshua G, Pine AB, Meizlish ML, et al. Endotheliopathy in COVID-19-associated coagulopathy: Evidence from a single-centre, cross-sectional study. Lancet Haematol. 2020; 7:e575–e582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Doevelaar AAN, Bachmann M, Hölzer B, et al. Generation of inhibitory autoantibodies to ADAMTS13 in coronavirus disease 2019. Medrxiv. Preprint posted online March 20, 2021. doi: 2021.03.18.21253869 [Google Scholar]
- 31.Sinha P, Matthay MA, Calfee CS. Is a “cytokine storm” relevant to COVID-19? JAMA Intern Med. 2020; 180:1152–1154 [DOI] [PubMed] [Google Scholar]
- 32.Veiga VC, Prats JAGG, Farias DLC, et al. Effect of tocilizumab on clinical outcomes at 15 days in patients with severe or critical coronavirus disease 2019: Randomised controlled trial. BMJ. 2021; 372:n84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Stone JH, Frigault MJ, Serling-Boyd NJ, et al. ; BACC Bay Tocilizumab Trial Investigators. Efficacy of tocilizumab in patients hospitalized with Covid-19. N Engl J Med. 2020; 383:2333–2344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Remy KE, Brakenridge SC, Francois B, et al. Immunotherapies for COVID-19: Lessons learned from sepsis. Lancet Respir Med. 2020; 8:946–949 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Faqihi F, Alharthy A, Abdulaziz S, et al. Therapeutic plasma exchange in patients with life-threatening COVID-19: A randomised controlled clinical trial. Int J Antimicrob Agents. 2021; 57:106334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Abani O, Abbas A, Abbas F, et al. Convalescent plasma in patients admitted to hospital with COVID-19 (RECOVERY): A randomised controlled, open-label, platform trial. Lancet. 2021; 397:2049–2059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of Covid-19 — final report. New Engl J Med. 2020; 383:1813–1826 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.