Abstract
Purpose
To evaluate the association between body mass index (BMI) and clinical outcomes other than death in patients hospitalised and intubated with COVID-19.
Methods
This is a single-centre cohort study of adults with COVID-19 admitted to New York Presbyterian Hospital-Weill Cornell Medicine from 3 March 2020 through 15 May 2020. Baseline and outcome variables, as well as lab and ventilatory parameters, were generated for the admitted and intubated cohorts after stratifying by BMI category. Linear regression models were used for continuous, and logistic regression models were used for categorical outcomes.
Results
The study included 1337 admitted patients with a subset of 407 intubated patients. Among admitted patients, hospital length of stay (LOS) and home discharge was not significantly different across BMI categories independent of demographic characteristics and comorbidities. In the intubated cohort, there was no difference in in-hospital events and treatments, including renal replacement therapy, neuromuscular blockade and prone positioning. Ventilatory ratio was higher with increasing BMI on days 1, 3 and 7. There was no significant difference in ventilator free days (VFD) at 28 or 60 days, need for tracheostomy, hospital LOS, and discharge disposition based on BMI in the intubated cohort after adjustment.
Conclusions
In our COVID-19 population, there was no association between obesity and morbidity outcomes, such as hospital LOS, home discharge or VFD. Further research is needed to clarify the mechanisms underlying the reported effects of BMI on outcomes, which may be population dependent.
Keywords: COVID-19, ARDS
Key messages.
Is obesity as defined by body mass index associated with different clinical outcomes, such as hospital length of stay, home discharge, ventilator free days and need for tracheostomy, in patients hospitalised and intubated with COVID-19?
Body mass index (BMI) was not associated with ventilator free days, need for tracheostomy, hospital length of stay, and discharge disposition in intubated patients with COVID-19 after adjustment for demographic characteristics and comorbidities.
In our population, although obesity was a risk factor for intubation, obesity was not linked with other adverse outcomes in hospitalised or intubated individuals with COVID-19. The lack of association indicates that the effect of BMI on COVID-19 severity may be mediated through other comorbidities, and further research is needed to clarify the mechanisms underlying the reported effects of BMI on outcomes, which may be population dependent.
Introduction
The prevalence of obesity (body mass index (BMI) >30 kg/m2) in the USA has been reported to be 42.2% in adults according to 2017–2018 data from the Centers for Disease Control and is expected to rise over time.1 Higher BMIs have been associated with negative respiratory complications such as increased risk for bacterial and viral pneumonias, and in particular with the development of acute respiratory distress syndrome (ARDS).2–4 The ongoing COVID-19 pandemic magnifies the public health impact of obesity on respiratory and critical illness as studies suggest its association with elevated risks for hospitalisation, intubation and death, particularly among patients with BMI >40 kg/m2 infected with the SARS-CoV-2 virus.5–11
ARDS represents a final common pathway of lung inflammation and injury from multiple aetiologies and is characterised by the acute onset of hypoxaemia with bilateral lung opacities that cannot be explained by cardiac failure or hydrostatic pulmonary oedema.12 ARDS development has emerged as a prominent reason for intubation and intensive care unit (ICU) admission in COVID-19.13 14 Prior to this pandemic, a few studies have suggested that rising BMI is associated with increased risk of ARDS,4 15 as well as increased duration of mechanical ventilation, ICU and hospital lengths of stay, and the likelihood of subsequent discharge to a rehabilitation or skilled nursing facility.4 16 17 On the other hand, meta-analyses and observational studies have failed to show that patients with obesity have reduced survival compared with normal weight ICU patients raising the possibility of an obesity paradox.16–18 For COVID-19, several reports have identified obesity as a risk factor for hospitalisation, critical illness and intubation, as well as, contrary to ARDS from other etiologies, death.6 7 9–11 19–22 Our group previously studied the association between obesity, respiratory failure and death among a diverse cohort of persons hospitalised with confirmed COVID-19 at 2 New York City hospitals and showed that obesity was an independent risk factor for respiratory failure but not for in-hospital mortality.5
While more evidence for the negative effects of obesity in patients with COVID-19 is surfacing, few studies describe the impact of BMI on morbidity, hospital course and clinical outcomes other than death in these patients. Examples of these outcomes include the duration of mechanical ventilation, the development of chronic respiratory failure requiring tracheostomy, kidney injury requiring renal replacement therapy, length of hospital stay, and discharge disposition. Studies in patients with ARDS have suggested that increasing BMI is associated with longer duration of mechanical ventilation, lengths of stay, and likelihood of discharge to a rehabilitation or skilled nursing facility, reflecting worse hospital outcomes.4 16 Obesity causes alterations in respiratory mechanics that can affect mechanisms of lung injury,23 such as atelectrauma, as well as treatments shown to change outcomes in ARDS, such as prone positioning. Obesity has also been associated with the release of inflammatory mediators, such as tumour necrosis factor α and interleukin 6, from adipose tissues predisposing to a pro-inflammatory state and oxidative stress,24 and with altered mitochondrial bioenergetics in lung epithelial cells increasing the risk of acute lung injury.25 We therefore hypothesised that higher BMI would be linked to longer ICU and hospital lengths of stay, higher need for tracheostomy, and less frequent discharge to home in patients with COVID-19. In the present study, we aim to examine the association between BMI and clinical outcomes other than death, which could affect quality of life, in patients hospitalised and intubated with COVID-19.
Methods
This single-centre cohort study examined patients older than 18 years old with a positive reverse transcription PCR for SARS-CoV-2 admitted to New York Presbyterian Hospital-Weill Cornell Medicine (NYP-WCM) from 3 March 2020 through 15 May 2020. Admitted patients were defined as any patient that was originally admitted directly from the emergency department or transferred to NYP-WCM from an outside hospital. Intubated patients included patients that were admitted directly and intubated at any point and patients that were transferred and intubated at any point before, on, or after arrival to NYP-WCM. Patients were excluded if they did not have height and weight recorded.
Patients were classified into the following BMI groups: <18.5 kg/m2 (underweight), 18.5–24.9 kg/m2 (normal), 25–29.9 kg/m2 (overweight), 30–39.9 kg/m2 (obese) and ≥40 kg/m2 (morbidly obese).26 We excluded women who were pregnant at the time of admission. For patients with recurring records/admissions, we used records that provided information for outcome variables such as mortality status and ventilation length. For all other such patients, their earliest record was used. Care of the patients was at the discretion of the treating physicians. Data were extracted from the electronic medical record. For ventilatory parameters set fraction of inspired oxygen, plateau pressure, extrinsic positive end expiratory pressure (PEEP), set tidal volume, and minute ventilation were recorded. In patients treated with pressure-targeted ventilation, the distending pressure was used to estimate a plateau pressure. Volumetric capnography was not available; therefore, a surrogate of dead space, called the ventilatory ratio, was used.27 The ventilatory ratio is an independent predictor of survival in ARDS.27 28 The first ventilatory parameter in each 24-hour window is reported. For lab parameters, the worst value in the first 48 hours after hospitalisation and intubation was recorded. The sequential organ failure assessment (SOFA) score was calculated for the first 3 days of intubation. Given that institutional protocols, clinical practice, and effects of social distancing may have varied over the study period, we examined 7-day rolling averages of admissions and first intubations stratified by BMI, where the absolute count for a particular day was right aligned compared with the remaining rolling window of observations.
Outcomes of interest were hospital length of stay (LOS) and discharge disposition among admitted and intubated patients; in addition, ventilator free days at 28 (VFD28) and 60 days (VFD60) and need for tracheostomy were examined in intubated patients. Patients who died while intubated were assigned a value of zero VFD. Patients that were extubated prior to death were assigned the respective number of days they were extubated until death. Discharge disposition was categorised as home versus other: death, acute and subacute rehabilitation facility, skilled nursing facility, hospice, shelter and transfers out to another hospital. The discharge disposition of interest was primarily discharge to home; this was selected as a surrogate for ability to self-care and acceptable functional status at the time of discharge. For LOS, analyses were repeated with restriction to survivors in order to eliminate any effect of death on these outcomes. We performed a sensitivity analysis for the outcomes in the intubated cohort excluding the underweight group due to the small sample size within that group.
Statistical analysis
The analytic sample included 1337 admitted patients and 407 intubated patients, which were a subset of the admitted cohort. Descriptive tables containing baseline and outcome variables, as well as lab and ventilator parameters, were generated for the admitted and intubated cohorts after stratifying by BMI category. Linear regression models were fitted for continuous outcomes, while logistic regression models were used for categorical outcomes. Covariates for the various models were chosen based on prior review of the literature and clinical judgement, focusing on variables that might be expected to confound the BMI-risk relationship. Models were initially adjusted for age, sex, and race/ethnicity, and subsequently additionally adjusted for diabetes, hypertension, chronic obstructive pulmonary disease (COPD), asthma, end stage renal disease, coronary artery diseases, heart failure, cerebrovascular accident (CVA) and cancer. Adjusted p values using Benjamini & Hochberg’s correction method were obtained to account for multiple comparisons in the analyses.29
The Strengthening the Reporting of Observational Studies in Epidemiology cohort reporting guidelines were used.30
Patient and public involvement
Patients or the public were not involved in the design, or conduct, or reporting, or dissemination plans of our research.
Results
Patient cohort
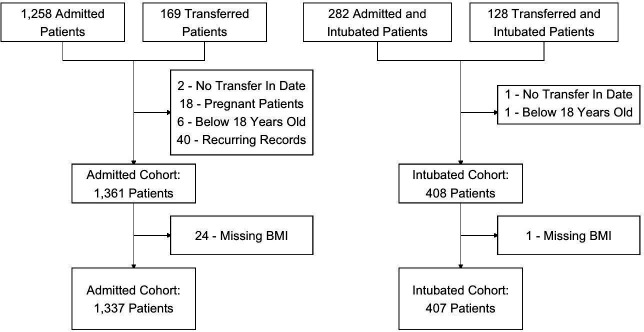
There were 1427 admissions with positive SARS-CoV-2 PCR to NYP-WCM during the study period. Two transfer patients from our admitted cohort and one transfer patient in our intubated cohort were excluded because they did not have a transfer in date. Eighteen patients were excluded due to pregnancy, 6 patients were younger than 18 years old, and 40 had recurring records. In addition, 24 patients in our admitted cohort and 1 patient in our intubated cohort did not have height and weight recorded. Our descriptive analyses included 1337 admitted patients, 165 of whom were transferred to NYP-WCM. A total of 407 patients were in the intubated subset, 127 of whom were transfers (figure 1). Patients were followed until death, discharge or transfer out date. The last data pull for our analysis was on 24 March 2021. There were no missing outcomes data, except for need for tracheostomy in 17 patients in the intubated cohort.
Figure 1.
Cohort selection. BMI, body mass index.
Characteristics of hospitalised patients
In the total cohort, the median age was 65.7 years (IQR, 53.3–75.9 years) and 61.4% were male. The median BMI was 27.4 kg/m2 (IQR, 23.8–31.4 kg/m2). Among all admitted patients, obese and morbidly obese were younger, more likely to be female and have a history of asthma, and less likely to be Asian, have active cancer and a history of CVA (table 1). There was no difference in outpatient prescription of ACE inhibitors, non-steroidal anti-inflammatory drugs or statins among BMI classes. During hospitalisation, no difference was observed in thromboembolic events or need for renal replacement therapy. Remdesivir, tocilizumab, antibiotic and steroid therapy was similar among groups, but we observed a difference among BMI classes in hydroxychloroquine and vasopressor administration with a lower percentage of underweight patients receiving either. The median hospital LOS was 8.5 days (IQR, 3.9–20.8 days), 241 patients (18 %) died, 1071 were discharged (80.1 %), 15 were transferred out to another hospital (1.1 %) and 10 (0.7 %) remained hospitalised. Hospital LOS was longer for patients who required intubation (31.3 days, IQR, 16–52.1 days vs 5.8 days, IQR, 3–10.6 days, p<0.001), but it did not drastically change across BMI categories (table 1). Results of LOS estimates across BMI categories were similar when the analysis was restricted to survivors. We observed a difference in discharge disposition with higher percentage of overweight, obese and morbidly obese individuals being discharged home (table 1).
Table 1.
Characteristics of patients admitted with COVID-19 by obesity class
| Characteristic | Underweight (BMI <18.5 kg/m2) n=40 | Normal weight (18.5–24.9 kg/m2) n=405 | Overweight (25.0–29.9 kg/m2) n=455 | Obese (30.0–39.9 kg/m2) n=366 | Morbidly obese (>40.0 kg/m2) n=71 |
| Age (Years) | 78 (56, 87) | 71 (59, 82) | 65 (54, 74) | 62 (50, 70) | 53 (41, 62) |
| Female | 16 (40%) | 152 (38%) | 155 (34%) | 152 (41%) | 41 (58%) |
| Race | |||||
| Black | 7 (18%) | 42 (10%) | 54 (12%) | 60 (16%) | 19 (27%) |
| White | 16 (40%) | 157 (39%) | 177 (39%) | 139 (38%) | 26 (37%) |
| Asian | 11 (28%) | 67 (17%) | 59 (13%) | 18 (4.9%) | 4 (5.6%) |
| Other | 3 (7.5%) | 93 (23%) | 102 (22%) | 89 (24%) | 12 (17%) |
| Not specified | 3 (7.5%) | 46 (11%) | 63 (14%) | 60 (16%) | 10 (14%) |
| Outside transfer | 4 (10%) | 51 (13%) | 64 (14%) | 39 (11%) | 7 (9.9%) |
| Smoking status | |||||
| Active smoker | 2 (5.0%) | 11 (2.7%) | 18 (4.0%) | 14 (3.8%) | 2 (2.8%) |
| Former smoker | 9 (22%) | 91 (22%) | 121 (27%) | 77 (21%) | 21 (30%) |
| No | 29 (72%) | 303 (75%) | 315 (69%) | 277 (75%) | 48 (68%) |
| Comorbidities | |||||
| Diabetes mellitus | 13 (32%) | 108 (27%) | 151 (33%) | 118 (32%) | 27 (38%) |
| Hypertension | 25 (62%) | 218 (54%) | 254 (56%) | 218 (60%) | 44 (62%) |
| COPD | 2 (5.0%) | 25 (6.2%) | 21 (4.6%) | 13 (3.6%) | 9 (13%) |
| Asthma | 2 (5.0%) | 29 (7.2%) | 26 (5.7%) | 48 (13%) | 19 (27%) |
| ESRD | 5 (12%) | 34 (8.4%) | 26 (5.7%) | 17 (4.6%) | 3 (4.2%) |
| CAD | 10 (25%) | 66 (16%) | 81 (18%) | 52 (14%) | 13 (18%) |
| Heart failure | 8 (20%) | 45 (11%) | 31 (6.8%) | 24 (6.6%) | 8 (11%) |
| Active cancer | 5 (12%) | 49 (12%) | 28 (6.2%) | 22 (6.0%) | 3 (4.2%) |
| CVA | 5 (12%) | 49 (12%) | 30 (6.6%) | 16 (4.4%) | 4 (5.6%) |
| Cirrhosis | 0 (0%) | 8 (2.0%) | 2 (0.4%) | 5 (1.4%) | 0 (0%) |
| History of transplant | 1 (2.5%) | 20 (4.9%) | 17 (3.7%) | 16 (4.4%) | 3 (4.2%) |
| Immunocompromised | 1 (2.5%) | 24 (5.9%) | 11 (2.4%) | 9 (2.4%) | 1 (1.4%) |
| In-Hospital events | |||||
| Myocardial infarction | 3 (7.5%) | 26 (6.4%) | 21 (4.6%) | 21 (5.7%) | 4 (5.6%) |
| Arrythmia | 0 (0%) | 50 (12%) | 55 (12%) | 45 (12%) | 8 (11%) |
| VTE | 1 (2.5%) | 42 (10%) | 44 (9.7%) | 46 (13%) | 6 (8.5%) |
| Acute kidney injury | 6 (15%) | 74 (18%) | 80 (18%) | 72 (20%) | 11 (15%) |
| Renal replacement | 3 (7.5%) | 38 (9.4%) | 37 (8.1%) | 45 (12%) | 7 (9.9%) |
| Home medications | |||||
| NSAID | 12 (30%) | 118 (29%) | 127 (28%) | 102 (28%) | 21 (30%) |
| ACE inhibitor | 8 (20%) | 114 (28%) | 130 (29%) | 110 (30%) | 26 (37%) |
| Statins | 13 (32%) | 153 (38%) | 171 (38%) | 138 (38%) | 23 (32%) |
| Inpatient therapies | |||||
| Antibiotics | 24 (60%) | 225 (56%) | 252 (56%) | 208 (57%) | 35 (49%) |
| Steroids | 8 (20%) | 115 (28%) | 137 (30%) | 124 (34%) | 26 (37%) |
| Hydroxychloroquine | 15 (38%) | 238 (59%) | 310 (68%) | 260 (71%) | 46 (65%) |
| Tocilizumab | 0 (0%) | 19 (4.7%) | 26 (5.7%) | 23 (6.3%) | 3 (4.2%) |
| Vasopressors | 6 (15%) | 102 (25%) | 140 (31%) | 134 (37%) | 22 (31%) |
| Remdesivir | 1 (2.5%) | 23 (5.7%) | 38 (8.4%) | 31 (8.5%) | 10 (14%) |
| IVIG | 1 (2.5%) | 3 (0.7%) | 5 (1.1%) | 7 (1.9%) | 0 (0%) |
| Outcomes | |||||
| Discharge to home | 15 (38%) | 216 (53%) | 275 (60%) | 217 (59%) | 53 (75%) |
| Length of stay | 5.9 (3.1, 13.3) | 8.0 (3.8, 18.6) | 9.0 (4.2, 22.2) | 8.6 (4.0, 24.9) | 7.3 (3.9, 16.6) |
| Length of stay (survivors only) | 6.1 (4.2, 14.8) | 7.7 (3.8, 16.6) | 8.5 (3.9, 22.0) | 8.5 (4.0, 26.8) | 7.0 (3.8, 16.0) |
| Death | 15 (38%) | 98 (24%) | 62 (14%) | 56 (15%) | 10 (14%) |
Baseline values are presented as median (25–75th percentile) for continuous variables and N (%) for categorical variables.
ACE, angiotensin converting enzyme; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; ESRD, end-stage renal disease; IVIG, intravenous immunoglobulin; NSAID, non steroidal anti-inflammatory drug; VTE, venous thromboembolic event.
Characteristics of intubated patients
Our group has previously shown that the HR of intubation progressively increased with higher BMI.5 In this study, we examined the timing of intubation across BMI groups and found that the rolling weekly average number of intubations in the obese and morbidly obese peaked earlier during the Spring surge compared with normal weight and overweight individuals (online supplemental figure S1). Similar to the total admitted cohort, in the intubated cohort, obese and morbidly obese were younger, but no major difference in race and sex was observed among BMI categories. Diabetes was more frequently observed with rising BMI (table 2). Asthma was not different among BMI categories. There was no difference in days 1, 2 and 3 SOFA scores based on BMI (table 2). There were no differences in laboratory values, which included inflammatory markers (table 3 and S1). There was also no difference in in-hospital events and treatments, including renal replacement therapy, neuromuscular blockade and prone positioning (table 2).
Table 2.
Characteristics of intubated patients with COVID-19 by obesity class
| Characteristic | Underweight (BMI <18.5 kg/m2) n=5 |
Normal weight (BMI 18.5–24.9 kg/m2) n=104 |
Overweight (BMI 25.0–29.9 kg/m2) n=138 |
Obese (BMI 30.0–39.9 kg/m2) n=137 |
Morbidly obese (BMI >40.0 kg/m2) n=23 |
| Age | 54 (50, 78) | 71 (59, 77) | 66 (55, 74) | 63 (52, 70) | 57 (38, 64) |
| Female | 1 (20%) | 30 (29%) | 40 (29%) | 41 (30%) | 10 (43%) |
| Race | |||||
| Black | 1 (20%) | 6 (5.8%) | 11 (8.0%) | 17 (12%) | 3 (13%) |
| White | 3 (60%) | 36 (35%) | 38 (28%) | 54 (39%) | 13 (57%) |
| Asian | 1 (20%) | 26 (25%) | 28 (20%) | 12 (8.8%) | 2 (8.7%) |
| Other | 0 (0%) | 26 (25%) | 40 (29%) | 32 (23%) | 3 (13%) |
| Not specified | 0 (0%) | 10 (9.6%) | 21 (15%) | 22 (16%) | 2 (8.7%) |
| Outside transfer | 0 (0%) | 31 (30%) | 54 (39%) | 36 (26%) | 6 (26%) |
| Smoking status | |||||
| Active smoker | 0 (0%) | 0 (0%) | 5 (3.6%) | 6 (4.4%) | 1 (4.3%) |
| Former smoker | 2 (40%) | 18 (17%) | 37 (27%) | 36 (26%) | 7 (30%) |
| No | 3 (60%) | 86 (83%) | 96 (70%) | 95 (69%) | 15 (65%) |
| Comorbidities | |||||
| Diabetes mellitus | 0 (0%) | 25 (24%) | 48 (35%) | 51 (37%) | 11 (48%) |
| Hypertension | 3 (60%) | 52 (50%) | 82 (59%) | 85 (62%) | 14 (61%) |
| COPD | 0 (0%) | 6 (5.8%) | 9 (6.5%) | 7 (5.1%) | 4 (17%) |
| Asthma | 0 (0%) | 10 (9.6%) | 10 (7.2%) | 19 (14%) | 3 (13%) |
| ESRD | 0 (0%) | 5 (4.8%) | 4 (2.9%) | 6 (4.4%) | 0 (0%) |
| CAD | 1 (20%) | 17 (16%) | 21 (15%) | 26 (19%) | 7 (30%) |
| Heart failure | 2 (40%) | 4 (3.8%) | 9 (6.5%) | 10 (7.3%) | 2 (8.7%) |
| Active cancer | 1 (20%) | 8 (7.7%) | 7 (5.1%) | 10 (7.3%) | 0 (0%) |
| CVA | 0 (0%) | 15 (14%) | 7 (5.1%) | 3 (2.2%) | 1 (4.3%) |
| Cirrhosis | 0 (0%) | 2 (1.9%) | 0 (0%) | 3 (2.2%) | 0 (0%) |
| History of transplant | 0 (0%) | 6 (5.8%) | 3 (2.2%) | 5 (3.6%) | 0 (0%) |
| Immunocompromised | 1 (20%) | 3 (2.9%) | 3 (2.2%) | 5 (3.6%) | 0 (0%) |
| SOFA score | |||||
| SOFA score (Day 1) | 10.00 (8.00, 15.00) | 11.75 (10.00, 13.00) | 12.00 (10.00, 13.00) | 11.00 (10.00, 13.00) | 11.00 (10.00, 12.50) |
| SOFA score (Day 2) | 10.00 (6.00, 11.00) | 11.00 (10.00, 13.00) | 11.00 (9.50, 12.00) | 11.00 (10.00, 13.00) | 11.00 (9.00, 12.25) |
| SOFA score (Day 3) | 10.00 (8.25, 11.25) | 11.00 (9.25, 12.75) | 11.00 (9.00, 12.25) | 11.00 (9.00, 13.00) | 11.00 (8.50, 13.00) |
| In-hospital events | |||||
| Myocardial infarction | 0 (0%) | 13 (12%) | 14 (10%) | 16 (12%) | 4 (17%) |
| Arrythmia | 0 (0%) | 28 (27%) | 43 (31%) | 38 (28%) | 5 (22%) |
| VTE | 1 (20%) | 24 (23%) | 27 (20%) | 32 (23%) | 5 (22%) |
| Renal replacement | 0 (0%) | 21 (20%) | 25 (18%) | 37 (27%) | 5 (22%) |
| Acute kidney injury | 2 (40%) | 53 (51%) | 70 (51%) | 66 (48%) | 10 (43%) |
| Home medications | |||||
| NSAIDs | 1 (20%) | 37 (36%) | 33 (24%) | 44 (32%) | 8 (35%) |
| ACE inhibitor | 3 (60%) | 26 (25%) | 42 (30%) | 41 (30%) | 7 (30%) |
| Statins | 2 (40%) | 36 (35%) | 52 (38%) | 51 (37%) | 9 (39%) |
| Hospital interventions | |||||
| Antibiotics | 3 (60%) | 94 (90%) | 130 (94%) | 134 (98%) | 21 (91%) |
| Steroids | 2 (40%) | 76 (73%) | 93 (67%) | 89 (65%) | 17 (74%) |
| Hydroxychloroquine | 4 (80%) | 89 (86%) | 123 (89%) | 125 (91%) | 17 (74%) |
| Tocilizumab | 0 (0%) | 11 (11%) | 24 (17%) | 23 (17%) | 3 (13%) |
| Vasopressors | 5 (100%) | 98 (94%) | 133 (96%) | 131 (96%) | 22 (96%) |
| Remdesivir | 0 (0%) | 11 (11%) | 16 (12%) | 17 (12%) | 7 (30%) |
| IVIG | 0 (0%) | 3 (2.9%) | 2 (1.4%) | 5 (3.6%) | 0 (0%) |
| Rocuronium | 1 (100%) | 26 (96%) | 39 (91%) | 56 (88%) | 10 (100%) |
| Cisatracurium | 0 (0%) | 1 (3.7%) | 5 (12%) | 9 (14%) | 0 (0%) |
| Prone positioning | 2 (40%) | 38 (37%) | 64 (46%) | 74 (54%) | 12 (52%) |
| Outcomes | |||||
| Discharge to home | 1 (20%) | 21 (20%) | 31 (22%) | 42 (31%) | 10 (43%) |
| Length of stay | 15.2 (10.5, 18.3) | 26.5 (15.5, 52.1) | 34.0 (20.6, 50.9) | 33.9 (15.6, 54.2) | 21.3 (14.3, 48.1) |
| Length of stay (survivors only) | 18.7 (16.9, 20.4) | 48.8 (29.2, 69.9) | 40.9 (26.1, 56.5) | 41.9 (26.7, 66.1) | 27.1 (17.4, 57.3) |
| Tracheostomy | 0 (0%) | 32 (32%) | 47 (36%) | 51 (39%) | 5 (22%) |
| Vent-free (28) days | 0 (0, 19) | 0 (0, 9) | 0 (0, 14) | 0 (0, 12) | 0 (0, 17) |
| Vent-free (60) days | 0 (0, 51) | 0 (0, 39) | 26 (0, 46) | 23 (0, 44) | 23 (0, 49) |
| Death | 3 (60%) | 49 (47%) | 42 (30%) | 40 (29%) | 8 (35%) |
Baseline values are presented as median (25–75th percentile) for continuous variables and N (%) for categorical variables.
CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; ESRD, end stage renal disease; IVIG, intravenous immunoglobulin; NSAID, non steroidal anti-inflammatory drug; SOFA, Sequential Organ Failure Assessment; VTE, venous thromboembolic event.
Table 3.
Laboratory parameters of intubated patients with COVID-19 by obesity class
| Lab value (highest value) |
Underweight (BMI <18.5 kg/m2) n=5 |
Normal weight (BMI 18.5–24.9 kg/m2) n=101 |
Overweight (BMI 25.0–29.9 kg/m2) n=130 |
Obese (BMI 30.0–39.9 kg/m2) n=129 |
Morbidly obese (BMI >40.0 kg/m2) n=20 |
| White blood cell (103/μL) | 10 (8–15) | 14 (9–19) | 13 (10–18) | 13 (9–16) | 12 (9–14) |
| Platelet (103/μL) | 202 (139–306) | 282 (193–358) | 297 (228–374) | 278 (217–350) | 262 (211–302) |
| Creatinine (mg/dL) | 1.14 (1.01–2.82) | 1.31 (0.88–2.16) | 1.40 (0.87–2.49) | 1.35 (0.92–2.63) | 1.37 (1.08–2.31) |
| Alanine aminotransferase (U/L) | 29 (29–274) | 46 (23–82) | 49 (28–86) | 47 (31–82) | 71 (51–93) |
| Aspartate aminotransferase (U/L) | 86 (59–272) | 62 (40–94) | 64 (40–98) | 57 (42–94) | 89 (70–180) |
| Bilirubintotal (mg/dL) | 1.10 (0.60–1.20) | 0.70 (0.50–1.50) | 0.80 (0.50–1.15) | 0.70 (0.50–1.08) | 1.10 (0.60–1.50) |
| Glucose level (mg/dL) | 170 (149–183) | 186 (144–229) | 176 (139–242) | 170 (133–236) | 162 (126–301) |
| Ferritin level (ng/mL) | 2393 (1294–3832) | 1447 (875–1990) | 1535 (884–2023) | 1260 (802–1838) | 875 (807–1242) |
| Creatine kinase (U/L) | 1143 (1143–1143) | 160 (65–291) | 223 (95–455) | 138 (62–360) | 460 (166–1028) |
| D-dimer (ng/mL) | 2150 (961–3195) | 1147 (550–3674) | 1087 (531–3005) | 2127 (597–8544) | 909 (451–3718) |
| Lactate dehydrogenase (U/L) | 951 (712–1036) | 545 (422–784) | 574 (432–812) | 592 (443–788) | 647 (465–915) |
| C reactive protein (mg/dL) | 20 (12–22) | 22 (15–29) | 25 (16–31) | 24 (17–30) | 27 (23–32) |
| Sedimentation rate (mm/hour) | 79 (43–104) | 78 (62–100) | 89 (63–120) | 85 (61–108) | 91 (84–102) |
| Fibrinogen (mg/dL) | NA (NA - NA) | 601 (430–754) | 680 (454–861) | 677 (578–868) | 638 (585–699) |
| Procalcitonin (ng/mL) | 1 (0–6) | 1 (0–8) | 1 (0–2) | 1 (0–2) | 0 (0–4) |
| Lab value (lowest value) | |||||
| White blood cell (103/μL) | 8.0 (7.5–12.2) | 9.9 (6.4–13.5) | 9.9 (7.1–12.4) | 8.9 (6.8–12.1) | 8.1 (7.5–9.6) |
| Absolute lymphocyte count (103/μL) | 0.80 (0.40–0.80) | 0.58 (0.30–0.83) | 0.80 (0.50–1.10) | 0.70 (0.48–0.90) | 0.90 (0.60–1.10) |
| Platelet 103/μL | 201 (139–242) | 217 (147–295) | 250 (182–319) | 229 (178–286) | 212 (178–256) |
Statistics presented: median (25–75th percentile, IQR).
bmjresp-2021-000970supp001.pdf (198.2KB, pdf)
Ventilator parameters were available for 260 patients, all directly admitted to WCM-NYP (table 4). Tidal volume over predicted body weight was higher in the obese and morbidly obese and higher than the recommended 6 ml/kg of ideal body weight on day 1. The trend remained but improved on days 3 and 7 with volumes closer to 6 ml/kg. PEEP was set higher with increasing BMI on days 1, 3 and 7. There was no difference in plateau pressure, driving pressure or respiratory system compliance between the BMI groups, but conclusions should be limited due to the high rate of missing data (online supplemental table S2). Ventilatory ratio was higher with rising BMI on days 1, 3 and 7. There was no difference in VFD28 or VFD60, need for tracheostomy, hospital LOS, and discharge disposition based on BMI in the intubated cohort and the results remained the same after a sensitivity analysis that excluded the low number of underweight individuals (table 2 and online supplemental table S3).
Table 4.
Ventilator parameters of intubated patients with COVID-19 by obesity class
| Values (days 1, 3 and 7) |
Underweight (BMI <18.5 kg/m2) n=5 |
Normal weight (BMI 18.5–24.9 kg/m2) n=66 |
Overweight (BMI 25.0–29.9 kg/m2) n=76 |
Obese (BMI 30.0–39.9 kg/m2) n=97 |
Morbidly obese (BMI >40.0 kg/m2) n=16 |
| PEEP (cm H2O) | |||||
| PEEP 1 | 10.0 (8.0–10.0) | 10.0 (8.0–10.0) | 10.0 (10.0–12.0) | 10.0 (10.0–12.0) | 14.0 (10.0–15.0) |
| PEEP 3 | 8.0 (8.0–9.0) | 10.0 (8.0–12.0) | 12.0 (8.0–12.0) | 12.0 (10.0–14.0) | 14.0 (10.0–15.0) |
| PEEP 7 | 8.0 (7.0–9.0) | 10.0 (8.0–13.0) | 10.0 (8.0–12.0) | 12.0 (8.0–14.0) | 12.0 (10.0–14.5) |
| Plateau pressure (cm H2O) | |||||
| Plateau pressure 1 | 24.5 (21.2–27.8) | 23.5 (20.0–27.0) | 24.0 (21.0–28.0) | 26.0 (21.0–30.0) | 30.0 (27.0–30.0) |
| Plateau pressure 3 | 23 (22–24) | 24 (19–27) | 24 (20–28) | 25 (22–29) | 27 (23–33) |
| Plateau pressure 7 | 44.0 (44.0–44.0) | 27.0 (22.5–30.8) | 26.0 (22.0–28.0) | 24.0 (22.5–28.5) | 27.0 (22.0–30.0) |
| PaO2/FiO2 ratio | |||||
| PaO2/FiO2 ratio 1 | 188 (152–207) | 116 (84–160) | 108 (91–146) | 115 (84–151) | 96 (62–129) |
| PaO2/FiO2 ratio 3 | 134 (124–144) | 128 (105–180) | 148 (110–174) | 140 (110–168) | 103 (83–147) |
| PaO2/FiO2 ratio 7 | 200 (200–200) | 141 (93–172) | 138 (109–160) | 142 (116–179) | 154 (112–186) |
| Tidal volume per predicted body weight (PBW; ml/kg) | |||||
| Tidal volume/PBW 1 | 5.53 (5.29–5.95) | 6.74 (6.26–7.76) | 6.97 (6.15–8.38) | 7.34 (6.42–8.21) | 8.02 (7.43–9.66) |
| Tidal volume/PBW 3 | 5.29 (5.17–5.62) | 6.21 (5.95–6.82) | 6.68 (6.06–7.13) | 6.57 (6.03–7.03) | 7.02 (6.26–7.73) |
| Tidal volume/PBW 7 | 5.05 (5.05–5.05) | 6.43 (5.97–7.02) | 6.70 (6.15–7.49) | 6.83 (6.27–7.60) | 6.97 (6.76–7.30) |
| Static compliance (mL/cm H2O) | |||||
| Static compliance 1 | 29 (20–36) | 30 (22–39) | 29 (23–40) | 28 (22–38) | 29 (27–36) |
| Static compliance 3 | 25 (24–29) | 30 (24–38) | 29 (23–39) | 31 (22–39) | 31 (23–35) |
| Static compliance 7 | 11 (11–11) | 25 (20–33) | 31 (21–39) | 28 (24–37) | 25 (22–36) |
| Driving pressure (cm H2O) | |||||
| Driving pressure 1 | 9.5 (8.8–10.2) | 12.2 (10.0–16.0) | 14.0 (11.0–18.0) | 14.0 (11.0–19.0) | 15.0 (14.0–17.0) |
| Driving pressure 3 | 14.0 (13.0–15.0) | 12.0 (10.0–16.0) | 12.0 (10.0–15.2) | 13.0 (9.5–16.0) | 15.5 (12.0–18.2) |
| Driving pressure 7 | 34.0 (34.0–34.0) | 15.0 (12.2–17.5) | 13.0 (11.0–17.0) | 13.0 (11.2–17.0) | 14.0 (8.0–16.0) |
| Ventilatory ratio | |||||
| Ventilatory ratio 1 | 1.27 (1.22–1.44) | 1.63 (1.24–2.18) | 1.88 (1.64–2.23) | 1.87 (1.54–2.49) | 2.21 (1.84–2.59) |
| Ventilatory ratio 3 | 1.44 (1.17–1.44) | 1.72 (1.43–2.10) | 2.03 (1.62–2.60) | 2.17 (1.73–2.65) | 2.22 (1.96–2.51) |
| Ventilatory ratio 7 | 0.99 (0.99–0.99) | 1.80 (1.52–2.34) | 2.19 (1.68–2.63) | 2.26 (1.88–2.64) | 2.39 (1.98–3.03) |
Statistics presented: median (25–75th percentile, IQR).
PEEP, plateau pressure, extrinsic positive end expiratory pressure.
Regression models
For the main outcomes, in the total hospitalised cohort, our unadjusted model showed no differences in LOS between the BMI groups (online supplemental table S4). There were higher odds of being discharged home for patients with morbid obesity (online supplemental table S4). After adjusting for sociodemographic variables (online supplemental table S5) and comorbidities, we found no significant differences in the outcomes between the BMI groups (table 5). In the intubated cohort, our unadjusted models showed no differences in VFD28, VFD60, LOS, discharge home, or need for tracheostomy (online supplemental table S6); similar results were seen after adjusting for sociodemographic variables (online supplemental table S7) and comorbidities (table 6).
Table 5.
Adjusted outcomes by obesity class in admitted cohort*
| BMI | Normal | Underweight | Overweight | Obese | Morbidly obese |
| Discharge to home | |||||
| OR | — | 0.54 | 1.1 | 0.81 | 1.4 |
| 95% CI | — | 0.25 to 1.1 | 0.79 to 1.4 | 0.59 to 1.1 | 0.73 to 2.7 |
| Q value† | 0.19 | 0.8 | 0.31 | 0.45 | |
| Length of stay (days) | |||||
| Estimate | — | −6.9 | 2.6 | 4.3 | 0.58 |
| 95% CI | — | −15 to 1.2 | −0.81 to 6.0 | 0.57 to 8.0 | −6.1 to 7.2 |
| Q value | 0.62 | 0.63 | 0.24 | 0.91 | |
*Adjusted for age, sex, race, diabetes, hypertension, chronic obstructive pulmonary disease, asthma, end-stage renal disease, coronary artery diseases, heart failure, cerebrovascular accident and cancer.
†Q-value: false discovery rate correction for multiple testing.
Table 6.
Adjusted outcomes by obesity class in intubated cohort*
| BMI | Normal | Underweight | Overweight | Obese | Morbidly obese |
| VFD (60 days) | |||||
| Estimate | — | 0.15 | 3.4 | 3.5 | 1.1 |
| 95% CI | — | −19 to 19 | −2.1 to 8.8 | −2.2 to 9.2 | −9.0 to 11 |
| Q value† | — | 0.99 | 0.54 | 0.54 | 0.97 |
| VFD (28 days) | |||||
| Estimate | — | 2.6 | 0.93 | 0.32 | 1.2 |
| 95% CI | — | −4.8 to 10.0 | −1.2 to 3.1 | −1.9 to 2.5 | −2.7 to 5.1 |
| Q value | — | 0.76 | 0.76 | 0.92 | 0.79 |
| Length of stay (days) | |||||
| Estimate | — | −23 | 4 | 1.5 | −7.6 |
| 95% CI | — | −55 to 9.5 | −5.2 to 13 | −8.1 to 11 | −25 to 9.5 |
| Q value | — | 0.67 | 0.72 | 0.93 | 0.72 |
| Discharge to home | |||||
| OR | — | 0.86 | 0.94 | 1.5 | 2.1 |
| 95% CI | — | 0.04 to 8.0 | 0.47 to 1.9 | 0.78 to 3.1 | 0.64 to 6.5 |
| Q value | — | >0.99 | >0.99 | 0.7 | 0.7 |
| Tracheostomy | |||||
| OR | — | 0 | 1.2 | 1.4 | 0.6 |
| 95% CI | — | 0.68 to 2.2 | 0.79 to 2.7 | 0.17 to 1.8 | |
| Q value | — | >0.99 | 0.95 | 0.95 | 0.95 |
*Adjusted for age, sex, race, diabetes, hypertension, chronic obstructive pulmonary disease, asthma, end stage renal disease, coronary artery diseases, heart failure, cerebrovascular accident and cancer.
†Q-value: False discovery rate correction for multiple testing.
VFD, ventilator free days.
To account for the possibility of death affecting our LOS analyses, we restricted our models to just survivors for both the hospitalised and intubated cohorts. For our hospitalised cohort, LOS point estimates in our model restricted to survivors were similar to those in our main adjusted model and no significant differences were found in any of the BMI groups compared with the referent normal BMI group (online supplemental table S8). For our intubated cohort, LOS point estimates in our model restricted to survivors were slightly lower than those in our main adjusted model, but no significant differences were found in any of the BMI groups compared with the referent normal BMI group (online supplemental table S9).
Discussion
This study examines the relationship between BMI and outcomes other than death in a large cohort of adults hospitalised with COVID-19 in a New York City academic hospital. We found no association between obesity and longer hospital LOS or home discharge in our overall cohort of patients. Focusing on the intubated cohort, we did not find significant differences in ventilator free days, need for tracheostomy, hospital LOS, and discharge disposition based on BMI independent of demographic characteristics and comorbid conditions. This is in accordance with a previous study by our group that examined this population as part of a larger cohort, which showed that obesity was an independent risk factor for intubation but not for in hospital mortality both in the overall and the intubated population admitted with COVID-19.5
Observational evidence have long suggested that obese patients may be at greater risk of developing ARDS.4 15 17 On the other hand, several meta-analyses and studies have failed to show that obese patients have higher mortality compared with normal weight ICU patients,16–18 31 32 even though rising BMI has been associated with other morbidity indices such as longer duration of mechanical ventilation, ICU and hospital LOS and discharge to a rehabilitation or skilled nursing facility.4 16 Influenza A H1N1 infection, another cause of ARDS, has been an exception, in the setting of which obesity has been linked to not only increased risk of ICU admission, but also death.3 Similarly during the Spring surge of COVID-19, multiple reports have emerged of obesity being a risk factor for mechanical ventilation, critical illness and death from COVID-19.6 7 10 11 22 33 However, much less has been written about other outcomes that have significant impact on patients’ recovery and well-being as well as notable societal implications, such as hospital LOS, tracheostomy and need for longer term mechanical ventilation, and discharge disposition. This study sought to examine these factors that could be coupled with pre-existing mortality studies to improve our comprehensive understanding of the trajectory of obese patients who suffer from severe COVID-19.
In agreement with a previous study from our group that did not find increased mortality in obese adults hospitalised with COVID-19,5 we were not able to link obesity with other adverse outcomes in hospitalised or intubated individuals with COVID-19. LOS was evaluated to gauge the impact of obesity on the duration of severe disease requiring hospitalisation, and discharge destination as a surrogate of functional status; in particular, we chose discharge to home as it more likely reflects the ability for self-care. Obese individuals experience a number of changes in their physiology that could affect their susceptibility to ARDS, their presentation and outcomes.34 Patients who are obese have considerable atelectasis that may result in decreased lung compliance and impaired gas exchange leading to an earlier presentation with respiratory failure compared with lean individuals, even in the setting of similar severity of underlying infection.34 In support of this, we did observe that the rolling weekly average number of admissions and intubations in the obese and morbidly obese peaked earlier in the pandemic compared with normal weight and overweight individuals.
Moreover in agreement to another study of adults hospitalised with severe SARS-CoV-2,6 higher BMI was not associated with higher levels of non specific inflammatory biomarkers on admission or higher SOFA scores after intubation, indicating that higher rates of respiratory failure were not necessarily associated with heightened inflammation or more severe overall organ failure. Atelectasis can be more readily resolved by the application of PEEP35 than acute lung injury, providing perhaps a more easily reversible cause of respiratory failure in obese patients. Indeed, higher PEEP was applied with rising BMI in our study population. The temporal variation in the rolling weekly average of intubations by BMI category may have also been affected by the use of non invasive mechanical ventilation modalities that were avoided at the start of the pandemic, such as high flow nasal cannula, which may prevent subsequent intubation with the application of PEEP, especially if atelectasis is a contributing factor to the acute respiratory failure.
Medical management and resource availability is also pivotal for successful outcomes. Studies in COVID-19 have shown that treatments and supportive therapies may vary considerably among hospitals and that patients admitted to hospitals with fewer ICU beds have a higher risk of death.9 We are a quaternary academic medical centre and, although our hospital was significantly challenged similarly to other institutions, ICU bed capacity was quickly and successfully expanded.36 Careful titration of mechanical ventilation to avoid ventilator induced lung injury is a mainstay of treatment in ARDS and even more important in patients with increased BMI and altered respiratory system physiology. The reduction of tidal volume over predicted body weight from days 1 to 3 along with the use of higher PEEP in the higher BMI categories indicates that, despite the increased workload, medical professionals and respiratory therapists were committed to lung protective ventilation.
A notable observation in our cohort is that although ventilatory ratio seemed to be positively associated with BMI, we did not observe worse outcomes in the groups with the higher ventilatory ratio. Ventilatory ratio has been shown to correlate well with the pulmonary dead space fraction (VD/VT) in ARDS and higher values have been associated with increased risk of adverse outcomes.28 Obesity has been linked with increased resting metabolic rate and minute ventilation,37–39 and the ventilatory ratio is influenced by pulmonary dead space as well as carbon dioxide production/elimination via minute ventilation. It is therefore not surprising that its predictive value of ARDS outcomes may be skewed in the setting of obesity.
Our study has several limitations. Our cohort includes some patients that were transferred from outside hospitals, but the majority were direct admissions, and may not be representative of the overall New York City population. Racial and socioeconomic disparities in the infections, hospitalisations and deaths have now been well documented in COVID-19.40 41 Our hospital serves the Upper East Side (UES) of New York City. In 2018, 75.9% of the population identified as white, only 3.2% identified as black, 6.7% Hispanic and 11.4% as Asian.42 Median household income in 2018 was 91% more than citywide median household income with a much lower poverty rate.42 In addition, 11% of UES residents are obese, 4% are diabetic, and 15% have high blood pressure compared with the citywide averages of 24%, 11% and 28% respectively.43
Moreover, our cohort included a lower percentage of individuals with obesity and morbid obesity compared with prior studies linking obesity, and mostly morbid obesity (BMI≥40 kg/m2), with worse outcomes.6 7 33 Furthermore, the small number of patients in the underweight and morbid obesity class, especially in the intubated subgroup, may have limited power to detect important differences. Obesity’s associations with other chronic conditions such as diabetes, hypertension and cardiac conditions, is well described, but the interaction of these risk factors for adverse outcomes in the setting of COVID-19 has not been disentangled. In a recent study using Mendelian randomisation, researchers found that genetically increased BMI was a risk factor for COVID-19 susceptibility and severity, but this effect was abolished after adjusting for the genetic effect of type 2 diabetes, suggesting that type 2 diabetes may mediate the causal association of BMI with COVID-19.44 We did not notice significant differences in cardiometabolic comorbid conditions across BMI categories, perhaps explaining the lack of association of BMI with worse outcomes, if adverse effects are mediated through these metabolic consequences. Comorbid conditions, however, were identified from the electronic medical record, which may be incomplete due to the overwhelming workload for healthcare professionals during that period. Moreover, the large number of missing laboratory data allows us to use them only as indicators and cannot support strong conclusions. Finally, we did not aim to interpret these relationships between BMI and outcomes as causal because of the well described collider bias effect, which hinders identification of risk factors in COVID-19 hospitalised cohorts like ours.45 Nonetheless, our highly detailed report of associations between BMI, baseline and temporal characteristics, and final outcomes in a hospitalised cohort may have important implications for the clinical management of patients with obesity and morbid obesity and may alert clinicians to practices that are fundamental for the best outcome in this patient population, such as lung protective mechanical ventilation.
In conclusion, in our COVID-19 population, there was no association between obesity and home discharge or longer hospital LOS among admitted patients, as well as no significant differences in VFD28 and VFD60 period, need for tracheostomy, home discharge and hospital LOS with higher BMI in the intubated subgroup independent of demographic characteristics and comorbid conditions. We do not interpret our results to represent an ‘obesity paradox’, but rather a conjecture that the effect of BMI on COVID-19 susceptibility and severity may be mediated through other comorbidities, and further research is needed to clarify the mechanisms underlying the reported effects of BMI on outcomes, which may be population dependent. More in-depth understanding is necessary to inform decisions on public policies tailored to at risk populations.
Footnotes
Contributors: MP had full access to all the data in the study and takes responsibility for the integrity of the data, the accuracy of the data analysis and the content of the manuscript. MP, DP, PG, MMS and EJS contributed substantially to the study design, data interpretation, and the writing of the manuscript. KH and JMKC contributed substantially to the study design and data analysis and interpretation. HH contributed substantially to data acquisition, analysis and interpretation and manuscript preparation.
Funding: This work was supported by the Weill Cornell DOM Pre Career Award (to MP).
Competing interests: No conflicts exist for MP, DP, KLH, JMKC, HH and EJS. Dr. Goyal has received personal fees for medicolegal consulting on heart failure. Dr Safford receives salary support from Amgen for investigator-initiated research.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request.
Ethics statements
Patient consent for publication
Not required.
Ethics approval
The study was approved by the Institutional Review Board at WCM with a waiver of informed consent (no. 20-04021909).
References
- 1.Hales CM, Carroll MD, Fryar CD, et al. Prevalence of obesity and severe obesity among adults: United States, 2017-2018. NCHS Data Brief 2020;360:1–8. [PubMed] [Google Scholar]
- 2.Mertz D, Kim TH, Johnstone J, et al. Populations at risk for severe or complicated influenza illness: systematic review and meta-analysis. BMJ 2013;347:f5061. 10.1136/bmj.f5061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fezeu L, Julia C, Henegar A, et al. Obesity is associated with higher risk of intensive care unit admission and death in influenza A (H1N1) patients: a systematic review and meta-analysis. Obes Rev 2011;12:653–9. 10.1111/j.1467-789X.2011.00864.x [DOI] [PubMed] [Google Scholar]
- 4.Gong MN, Bajwa EK, Thompson BT, et al. Body mass index is associated with the development of acute respiratory distress syndrome. Thorax 2010;65:44–50. 10.1136/thx.2009.117572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Goyal P, Ringel JB, Rajan M, et al. Obesity and COVID-19 in New York City: a retrospective cohort study. Ann Intern Med 2020;173:855–8. 10.7326/M20-2730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Anderson MR, Geleris J, Anderson DR. Body mass index and risk for intubation or death in SARS-CoV-2 infection: a retrospective cohort study. Ann Intern Med 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tartof SY, Qian L, Hong V, et al. Obesity and mortality among patients diagnosed with COVID-19: results from an integrated health care organization. Ann Intern Med 2020;173:773–81. 10.7326/M20-3742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nakeshbandi M, Maini R, Daniel P, et al. The impact of obesity on COVID-19 complications: a retrospective cohort study. Int J Obes 2020;44:1832–7. 10.1038/s41366-020-0648-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gupta S, Hayek SS, Wang W, et al. Factors associated with death in critically ill patients with coronavirus disease 2019 in the US. JAMA Intern Med 2020;180:1436. 10.1001/jamainternmed.2020.3596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kompaniyets L, Goodman AB, Belay B, et al. Body Mass Index and Risk for COVID-19-Related Hospitalization, Intensive Care Unit Admission, Invasive Mechanical Ventilation, and Death - United States, March-December 2020. MMWR Morb Mortal Wkly Rep 2021;70:355–61. 10.15585/mmwr.mm7010e4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gao M, Piernas C, Astbury NM, et al. Associations between body-mass index and COVID-19 severity in 6·9 million people in England: a prospective, community-based, cohort study. Lancet Diabetes Endocrinol 2021;9:350–9. 10.1016/S2213-8587(21)00089-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ranieri VM, Rubenfeld GD, et al. , ARDS Definition Task Force . Acute respiratory distress syndrome: the Berlin definition. JAMA 2012;307:2526–33. 10.1001/jama.2012.5669 [DOI] [PubMed] [Google Scholar]
- 13.Guan W-J, Ni Z-Y, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020;382:1708–20. 10.1056/NEJMoa2002032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fan E, Beitler JR, Brochard L, et al. COVID-19-associated acute respiratory distress syndrome: is a different approach to management warranted? Lancet Respir Med 2020;8:816–21. 10.1016/S2213-2600(20)30304-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Anzueto A, Frutos-Vivar F, Esteban A, et al. Influence of body mass index on outcome of the mechanically ventilated patients. Thorax 2011;66:66–73. 10.1136/thx.2010.145086 [DOI] [PubMed] [Google Scholar]
- 16.Morris AE, Stapleton RD, Rubenfeld GD, et al. The association between body mass index and clinical outcomes in acute lung injury. Chest 2007;131:342–8. 10.1378/chest.06-1709 [DOI] [PubMed] [Google Scholar]
- 17.Stapleton RD, Suratt BT. Obesity and nutrition in acute respiratory distress syndrome. Clin Chest Med 2014;35:655–71. 10.1016/j.ccm.2014.08.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Martino JL, Stapleton RD, Wang M, et al. Extreme obesity and outcomes in critically ill patients. Chest 2011;140:1198–206. 10.1378/chest.10-3023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Docherty AB, Harrison EM, Green CA, et al. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ 2020;369:m1985. 10.1136/bmj.m1985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lighter J, Phillips M, Hochman S, et al. Obesity in patients younger than 60 years is a risk factor for COVID-19 hospital admission. Clin Infect Dis 2020;71:896–7. 10.1093/cid/ciaa415 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Petrilli CM, Jones SA, Yang J, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ 2020;369:m1966. 10.1136/bmj.m1966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Simonnet A, Chetboun M, Poissy J, et al. High prevalence of obesity in severe acute respiratory syndrome Coronavirus‐2 (SARS‐CoV‐2) requiring invasive mechanical ventilation. Obesity 2020;28:1195–9. 10.1002/oby.22831 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dixon AE, Peters U. The effect of obesity on lung function. Expert Rev Respir Med 2018;12:755–67. 10.1080/17476348.2018.1506331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ellulu MS, Patimah I, Khaza'ai H, et al. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci 2017;13:851–63. 10.5114/aoms.2016.58928 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Plataki M, Fan L, Sanchez E, et al. Fatty acid synthase downregulation contributes to acute lung injury in murine diet-induced obesity. JCI Insight 2019;4. 10.1172/jci.insight.127823 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Physical status: the use and interpretation of anthropometry. Report of a WHO expert Committee. World Health Organ Tech Rep Ser 1995;854:1–452. [PubMed] [Google Scholar]
- 27.Sinha P, Fauvel NJ, Singh S, et al. Ventilatory ratio: a simple bedside measure of ventilation. Br J Anaesth 2009;102:692–7. 10.1093/bja/aep054 [DOI] [PubMed] [Google Scholar]
- 28.Sinha P, Calfee CS, Beitler JR, et al. Physiologic analysis and clinical performance of the ventilatory ratio in acute respiratory distress syndrome. Am J Respir Crit Care Med 2019;199:333–41. 10.1164/rccm.201804-0692OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Haynes W. Benjamini-Hochberg method. In: Encyclopedia of systems biology, 2013. [Google Scholar]
- 30.von Elm E, Altman DG, Egger M, et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Int J Surg 2014;12:1495–9. 10.1016/j.ijsu.2014.07.013 [DOI] [PubMed] [Google Scholar]
- 31.Pickkers P, de Keizer N, Dusseljee J, et al. Body mass index is associated with hospital mortality in critically ill patients: an observational cohort study. Crit Care Med 2013;41:1878–83. 10.1097/CCM.0b013e31828a2aa1 [DOI] [PubMed] [Google Scholar]
- 32.Zhi G, Xin W, Ying W, et al. “Obesity paradox” in acute respiratory distress syndrome: asystematic review and meta-analysis. PLoS One 2016;11:e0163677. 10.1371/journal.pone.0163677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hendren NS, de Lemos JA, Ayers C, et al. Association of body mass index and age with morbidity and mortality in patients hospitalized with COVID-19: results from the American heart association COVID-19 cardiovascular disease registry. Circulation 2021;143:135–44. 10.1161/CIRCULATIONAHA.120.051936 [DOI] [PubMed] [Google Scholar]
- 34.Hibbert K, Rice M, Malhotra A. Obesity and ARDS. Chest 2012;142:785–90. 10.1378/chest.12-0117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fumagalli J, Santiago RRS, Teggia Droghi M, et al. Lung recruitment in obese patients with acute respiratory distress syndrome. Anesthesiology 2019;130:791–803. 10.1097/ALN.0000000000002638 [DOI] [PubMed] [Google Scholar]
- 36.Griffin KM, Karas MG, Ivascu NS, et al. Hospital preparedness for COVID-19: a practical guide from a critical care perspective. Am J Respir Crit Care Med 2020;201:1337–44. 10.1164/rccm.202004-1037CP [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Busetto L, Perini P, Giantin V, et al. Relationship between energy expenditure and visceral fat accumulation in obese women submitted to adjustable silicone gastric banding (ASGB). Int J Obes Relat Metab Disord 1995;19:227–33. [PubMed] [Google Scholar]
- 38.Littleton SW. Impact of obesity on respiratory function. Respirology 2012;17:43–9. 10.1111/j.1440-1843.2011.02096.x [DOI] [PubMed] [Google Scholar]
- 39.Parameswaran K, Todd DC, Soth M. Altered respiratory physiology in obesity. Can Respir J 2006;13:203–10. 10.1155/2006/834786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Belanger MJ, Hill MA, Angelidi AM, et al. Covid-19 and disparities in nutrition and obesity. N Engl J Med 2020;383:e69. 10.1056/NEJMp2021264 [DOI] [PubMed] [Google Scholar]
- 41.Laurencin CT, McClinton A. The COVID-19 pandemic: a call to action to identify and address racial and ethnic disparities. J Racial Ethn Health Disparities 2020;7:398–402. 10.1007/s40615-020-00756-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.NYU Furman Center . State of New York City’s housing & neighborhoods. Upper east side MN08.
- 43.NYC Health . Upper east side, 2018. Available: nyc.gov
- 44.Leong A, Cole J, Brenner LN, et al. Cardiometabolic risk factors for COVID-19 susceptibility and severity: a Mendelian randomization analysis. medRxiv 2020 10.1101/2020.08.26.20182709 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Griffith GJ, Morris TT, Tudball MJ, et al. Collider bias undermines our understanding of COVID-19 disease risk and severity. Nat Commun 2020;11:5749. 10.1038/s41467-020-19478-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjresp-2021-000970supp001.pdf (198.2KB, pdf)
Data Availability Statement
Data are available upon reasonable request.