Abstract
The present study aimed to evaluate the effects of 2 mg drospirenone (DRSP) and 1 mg 17β-estradiol (E2) hormone therapy (HT) in combination with rehabilitation therapy for postmenopausal women with Meniere's disease (MD). The combined drug hormone treatment was denoted as DRSP/E2. A total of 65 postmenopausal female patients with MD and severe distress were enrolled in the present prospective study. A total of 31 women comprised the study group (group A), undergoing HT and rehabilitation therapy, whereas 34 women who opted for rehabilitation therapy alone comprised the control group (group B). Vestibular function and distress associated with MD were assessed by stabilometry and the Dizziness Handicap Inventory (DHI) questionnaire, respectively. The data were collected at baseline and during the 3- and 6-month follow-up visits. The areas of the stabilometric ellipses exhibited a higher reduction in group A compared with group B with regard to the baseline values at both follow-up assessments (P<0.001). At baseline, both groups exhibited severe self-perceived discomfort, with similar DHI scores of 72.3±3.7 (group A) and 70.6±3.9 (group B; P=0.07). At the 3-month follow-up, both groups underwent a gradual improvement, which was significantly higher in group A (47.5±3.7) compared with in group B (64.2±3.3; P<0.001). At the 6-month follow-up, the DHI scores were improved in group A (43.4±3.4) compared with in group B (58.5±3.1; P<0.001). Therefore, DRSP/E2 HT was effective in reducing the fluid overload, which is characteristic of MD. The findings of the present study demonstrated that integrated therapy based on the administration of DRSP/E2 HT and rehabilitation may be more effective compared with rehabilitation alone for the management of postmenopausal exacerbation of MD.
Keywords: drospirenone, 17β-estradiol, hormone therapy, Meniere's disease, menopause, stabilometry
Introduction
Postmenopausal women can experience symptoms that negatively affect their quality of life. These symptoms may develop several years prior to menopause (1) and comprise mainly vasomotor symptoms (VMS), such as hot flushes and night sweats, palpitations, headache, shoulder stiffness, insomnia, depression, nervousness, anxiety, fatigue, irritability and dizziness (2). Balance disturbances may be one of the most common morbidities experienced by postmenopausal women and are defined as Meniere's disease (MD) (3). These are characterized by vertigo, fluctuating hearing loss and tinnitus, and may be attributable to the inability of the cochlea to regulate endolymph absorption, production and/or circulation (4,5).
The prevalence of MD is 34-190 per 100,000 individuals. The age of onset ranges from the third to the seventh decades of life, with a female predominance (6). It has been recognized that the specific reduction in estrogen levels in perimenopausal women may cause microcirculatory disruption in the inner ear, promoting the development of MD or contributing to the deterioration of the symptoms (7). The peak range of the development of MD is estimated at the age of 40-60 years in women (8).
In conclusion, postmenopausal women with MD exhibit lower estrogen levels, increased pure-tone threshold, elevated I-V interpeak latency and higher incidence of unilateral caloric weakness. Changes in estrogen levels may contribute to the deteriorating symptoms of MD (low-frequency hearing loss, episodic symptoms of vertigo, aural fullness and tinnitus) in postmenopausal women to a certain extent.
Hormone therapy (HT) is considered as the most effective therapy for VMS and is currently the first choice of treatment (9). A continuous HT combining 2 mg drospirenone and 1 mg 17β-estradiol (E2), denoted as DRSP/E2, may be used to treat VMS in postmenopausal women. Unlike other synthetic progestins, DRSP exhibits a similar physiological profile to that of natural progesterone. DRSP is derived from spirolactone and inhibits the effects of aldosterone and androgen (10). Moreover, its antimineralocorticoid activity reduces salt and water retention and exerts diuretic and anti-edema effects (11,12). Notably, DRSP/E2 provides symptomatic relief in women with VMS and improves genitourinary atrophy, while it protects against endometrial hyperplasia and reduces the risk of osteoporosis (13).
Taking into consideration the existing evidence regarding the pathophysiology of vertigo, the present study aimed to investigate the effects of DRSP/E2 HT combined with rehabilitation therapy (study group) and compare them with those of rehabilitation therapy alone (control group) for postmenopausal women with MD.
Materials and methods
Patients
The current prospective study was performed at the Gynecology Clinic of the Department of General Surgery and Medical Surgical Specialties and at the Otorhinolaryngology Clinic of the Department of Medical Surgical Sciences and Advanced Technologies, University of Catania, Catania, Italy. The study conformed to the ethical guidelines of the 2013 Helsinki Declaration. Written informed consent was obtained from each participant upon enrollment. The present study was not advertised and no remuneration was offered.
A total of 74 postmenopausal women (age range 49-56 years) with MD and severe distress were recruited at the Otorhinolaryngology Clinic and invited to participate in the study. The time period of subject recruitment was from to April 2017 to October 2019. All women had their last spontaneous menstrual bleeding >6 months earlier. The plasma follicle-stimulating hormone levels were >40 mIU/ml (pre-menopausal normal range, 3,4-20 mUI/ml) and the serum E2 levels were <30 pg/ml (pre-menopausal normal range, 40-400 pg/ml). Each participant underwent general medical and gynecological examination.
Women with a history of thromboembolic and liver disease who had used steroid treatment in the previous 3 months or had received phytoestrogens within 1 month prior to the initiation of the study, were excluded. Furthermore, women with a body mass index of 35 kg/m2 or higher, with endometrial thickness ≥4 mm measured by transvaginal ultrasound prior to the study initiation, and/or presenting with abnormal uterine bleeding, hormone-dependent malignancies, receiving antihypertensive drugs, or suffering from diabetes or other chronic medical conditions, were also excluded from the study. The otolaryngological exclusion criteria included previous vestibular diseases, low compliance due to physical disabilities, or treatment with vestibular-suppressor drugs.
Instruments
The modified Kupperman index (KI) was used to evaluate menopausal symptoms by 13 items (14), including somatic (hot flushes/night sweats, paresthesia, dizziness, arthralgia/myalgia, headache, palpitations and formication), psychological (insomnia/sleep disturbance, depression, irritability and fatigue) and urogenital (urinary infection and sexual complaints) symptoms. The scoring system used was as follows: 0=none, 1=mild, 2=moderate and 3=severe. The following score range system was used to rate the degree of severity as follows: 0-6 (none), 7-15 (mild), 16-30 (moderate) and >30 (severe). The range was from 0 to 63 points.
The participants underwent Semont's maneuver (15) to assess the characteristics of positional vertigo (16). During this assessment, the women were sat upright in the middle of a stretcher. The head was rotated by the therapist towards the healthy ear; this rotation was maintained throughout the maneuver. Subsequently, the therapist lowered the body of the patient sideways onto the stretcher so that she was allowed to lie on the side of the affected ear, with her nose pointing upwards. This position was maintained for 3 min and the therapist instructed the woman to adopt an upright position without straightening her head, which was subsequently lowered to her opposite side with the nose pointing downwards. Finally, after maintaining this position for 3 min, the woman was slowly brought back to an upright position and her head was rotated back to the normal position.
Stabilometry was used to evaluate and measure the balance of each patient through a platform connected to a computer by three transducers. The platform was sensitive to the pressure applied on it due to the movements of each patient (17). The computer recorded the changes of the position at the center of pressure (CoP). The movements on the CoPs of the platform were presented on the monitor as an ellipsoid graphic. The recorded movements indicated that the patients may have moved on the medial-lateral (x-axis, right-left), or anterior-posterior (y-axis, back-forth) direction (18). Each subject performed the test sequentially with her eyes open, closed and the retroflex, and then open again. The displacements on the platform and the surface of the ellipse were calculated (mm2).
Finally, the Dizziness Handicap Inventory (DHI) questionnaire was used for the self-assessment of severe discomfort due to vertigo (19). This questionnaire is based on 25 items investigating physical, functional and emotional aspects of dizziness and unsteadiness. The scores ranged between 0, indicating no discomfort, and 100, indicating significant self-perceived discomfort. Each instrument was used at baseline (T0) and at the 3-month (T1) and 6-month (T2) follow-up visits.
Statistical analysis
The demographic non categorical variables, stabilometric and DHI intergroup data were compared by one-way analysis of variance with Bonferroni as a post hoc test. The χ2 was used to compare the categorical demographic data. Intragroup analysis was performed to compare baseline stabilometry and DHI values at follow-up by paired Student's t-test. All values are shown as mean ± SD. P<0.05 was considered to indicate statistically significant differences. Statistical analysis was carried out using software package for Windows 95 (Grantz SA; Primer of Biostatistics).
Results
Clinicopathological parameters
A total of 9 women refused to participate in the study; consequently, 65 women were finally included in the analysis, of whom 31 (47.7%) consented to receiving HT and rehabilitation. These patients comprised the study group (group A). A total of 34 women (52.3%) refused to receive HT and comprised the control group (group B), which selected rehabilitation alone. A total of 6 (17.6%) of the 34 study subjects did not come to the appointment T1, whereas 28 (82.4%) completed the study.
Table I indicates the baseline demographic characteristics of the two groups. The vertiginous crises suffered by each woman lasted for 20 sec, and had paroxysmal onset and rotatory character, mainly during the morning and night. At baseline, the women were affected by menopausal symptoms, including severe dizziness, associated with mild hot flushes, night sweats, sleep disturbances and fatigue [group A (32±7.3) vs. group B (30±6.1; P=0.2)].
Table I.
Demographic characteristics at baseline of women on 2 mg drospirenone and 1 mg 17β-estradiol hormone therapy and rehabilitation (Study group) or on rehabilitation (Control group) for the Maniere's disease.
| Variable | Study group (n=31) | Control group (n=34) | P-value |
|---|---|---|---|
| Age range, years | 49-56 | 50-56 | 0.9 |
| Mean age, years (mean ± SD) | 52.6±3.1 | 53.1±3.3 | 0.5 |
| BMI, kg/m2 (mean ± SD) | 26.3±4.1 | 25.5±5.1 | 0.4 |
| Age at menopause, years (mean ± SD) | 50.5±6.1 | 51.3±2.8 | 0.4 |
| Years from menopause (mean ± SD) | 5.0±3.4 | 6.0±4.1 | 0.2 |
| Parity, n (%) | |||
| One | 5 (16.1) | 6 (17.6) | 0.8 |
| Two or more children | 26 (83.9) | 28 (82.4) | 0.8 |
| Kupperman index (mean ± SD) | 32.0±7.3 | 30.0±6.1 | 0.2 |
| Cigarette smoking, n (%) | |||
| Non-smoker | 28 (90.3) | 29 (85.3) | 0.9 |
| Current smoker | 3 (9.7) | 5 (14.7) | 0.8 |
| Systolic blood pressure, mmHg (mean ± SD) | 132.5±11.6 | 134.2±15.4 | 0.6 |
| Diastolic blood pressure, mmHg (mean ± SD) | 83.8±8.3 | 82.7±7.5 | 0.5 |
| Heart rate, bpm (mean ± SD) | 65.8±10.3 | 67.4±12.2 | 0.5 |
P-values were obtained by one-way analysis of variance with Bonferroni post hoc test for non-categorical variables, and a χ2 test was used for categorical demographic variables.
Following Semont's maneuver, the subjects in group A exhibited an improvement at T1 (26/31, 83.8%; P<0.001) and at T2 (31/31, 100%; P<0.001) compared with T0. In group B, 7/28 (25%) patients exhibited an improvement at T1 (P<0.03) with regard to Semont's maneuver alone, whereas 10 (35.7%) women exhibited an improvement at T2 (P<0.01) compared with T0. A total of 11 (39.3%) subjects reported no benefit. The women of group A exhibited an improvement compared with those of group B at both follow-up visits when intergroup comparison was performed (P<0.001).
Stabilometry and DHI measurements
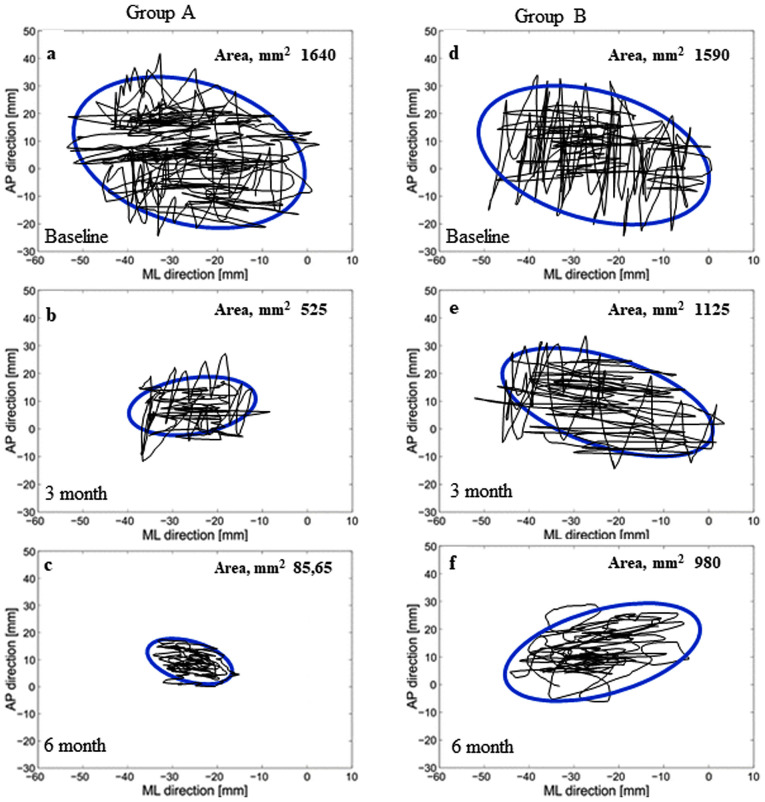
The changes in the stabilometric area of the ellipses of both groups were assessed (Fig. 1). The intragroup stabilometric evaluation indicated improved normalization in group A (T1 and T2 vs. T0, P<0.001) compared with group B (T1 vs. T0, P<0.01; T2 vs. T0, P<0.002). The intergroup statistical comparison indicated an improvement in group A compared with group B at each follow-up assessment (P<0.001; Table II).
Figure 1.
Stabilometric values expressed by ellipse areas in female patients with Meniere's disease treated with 2 mg drospirenone and 1 mg 17β-estradiol hormone therapy for 3 and 6 months combined with rehabilitation (Group A; a-c), or rehabilitation alone (Group B; d-f). Intragroup stabilometric analysis indicated improved normalization in group A [(b) 3 and (c) 6 month vs. (a) baseline; P<0.001)] compared with group B [(e) 3 month vs. (d) baseline; P<0.01; (f) 6 month vs. (d) baseline; P<0.002). The intergroup statistical comparison indicated an improvement in group A compared with group B at each follow-up assessment (b vs. e, c vs. f; P<0.001). AP, anterior-posterior direction; ML, medial-lateral direction.
Table II.
Stabilometry of female patients treated with 2 mg drospirenone and 1 mg 17β-estradiol hormone therapy combined with rehabilitation (group A), or rehabilitation alone (group B), for Meniere's disease.
| Stabilometric values | Baseline (T0) | 3 months (T1) | 6 months (T2) | P-valuea (T1 vs. T0) | P-valuea (T2 vs. T0) |
|---|---|---|---|---|---|
| Group A | n=31; 1,640±655 | n=31; 525±125 | n=31; 85.65±25 | 0.001 | 0.001 |
| Group B | n=34; 1,590±590 | n=28; 1,125±880 | n=28; 980±660 | 0.016 | 0.002 |
| P-valueb | 0.747 | 0.001 | 0.001 |
aPaired Student's t-test.
bOne way ANOVA. Values are presented as mm2 ± SD. Intra- and intergroup statistical evaluation at 3 (T1) and 6 (T2) months.
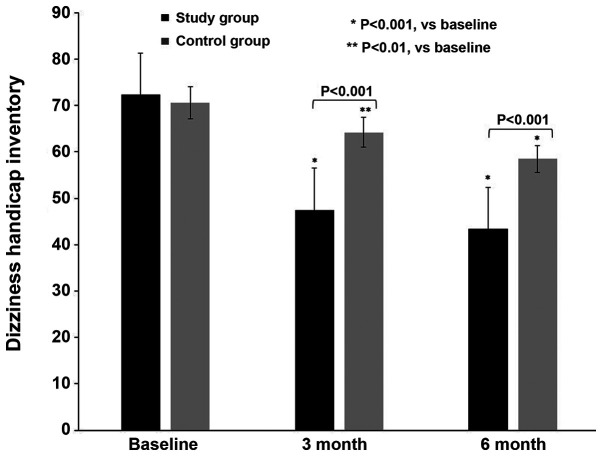
The DHI scores of both groups were assessed during the study (Fig. 2). Table III summarizes the DHI intra- and intergroup statistical evaluation. At T0, both groups exhibited severe self-perceived discomfort, with similar DHI scores of 72.3±3.7 (group A) and 70.6±3.9 (group B; P=0.07). At T1, both groups exhibited a gradual improvement, which was more prominent in group A (47.5±3.7) compared with group B (64.2±3.3; P<0.001). At T2, the DHI scores had improved in group A (43.4±3.4) compared with those in group B (58.5±3.1; P<0.001).
Figure 2.
Dizziness Handicap Inventory of female patients with Meniere's disease treated with 2 mg drospirenone and 1 mg 17β-estradiol hormone therapy for 3 and 6 months combined with rehabilitation (group A), or rehabilitation alone (group B). *P<0.001; **P<0.01 vs. baseline.
Table III.
DHI of female patients with Meniere's disease.
| DHI | Baseline (T0) | 3 months (T1) | 6 months (T2) | P-valuea (T1 vs. T0) | P-valuea (T2 vs. T0) | P-valuea (T2 vs. T1) |
|---|---|---|---|---|---|---|
| Group A | 72.3±3.7 | 47.5±3.7 | 43.4±3.4 | 0.001 | 0.001 | 0.001 |
| Group B | 70.6±3.9 | 64.2±3.3 | 58.5±3.1 | 0.010 | 0.001 | 0.004 |
| P-valueb | 0.077 | 0.001 | 0.001 |
aPaired Student's t-test;
bTwo-sided t-test for independent samples by ANOVA. Intra- and intergroup statistical evaluation of female patients treated with 2 mg drospirenone and 1 mg 17β-estradiol hormone therapy combined with rehabilitation (group A), or rehabilitation alone (group B), for 3 (T1) and 6 (T2) months. DHI, Dizziness Handicap Inventory.
Discussion
To the best of our knowledge, the present study was the first to evaluate the therapeutic effects of DRSP/E2 HT on postmenopausal women affected by severe MD associated with mild VMS. Initially, women on DRSP/E2 exhibited an improvement of VMS. Moreover, they exhibited a more notable improvement of the vertiginous crisis and of DHI compared with women on rehabilitation alone. The improvement of DHI observed in group B was less pronounced compared with that in group A, which may be attributed to the effects of Semont's maneuver. Of note, stabilometric evaluation indicated an optimal normalization with a more evident reduction of the areas of the ellipses in women on DRSP/E2 than in those who refused to receive HT. These data indicated that the women acquired more balance skills following treatment initiation.
The treatment usually recommended for women with MD exacerbations includes restriction of dietary salt intake and the use of diuretics (20). In the present study, an HT containing DRSP was selected to regulate the anti-aldosterone and antimineralocorticoid activities of progestogen, which, in turn, decreased salt and water retention (21).
The effects of sex on medical care and behavioral therapies have not been examined following treatment of women with MD (22). MD is most prevalent between the ages of 40 and 60 years, with a peak onset at 40-50 years (23,24). It also exhibits a slightly higher prevalence in women than in men (23,24). A correlation has been observed between VMS and vertigo exacerbation (3). However, that study did not record differences in vertigo prevalence between users and non-users of HRT (3). This could depend on the composition and regimen of the HT used to treat symptomatic postmenopausal women. DRSP derived from 17a-spironolactone is a progestin with an important antimineralocorticoid activity, which is able to reduce salt and water retention and to exert diuretic and anti-edema effects (11).
Moreover, it has been shown that the low estrogen levels noted in postmenopausal women may cause microcirculatory disorders in the inner ear, leading to the development of MD (7). Consequently, the present study indicated that the benefits obtained by postmenopausal women with MD using DRSP/E2 treatment may be due to the type, dosage, combination and regimen of progestogen and E2. Furthermore, the use of an oral hormonal contraceptive containing DRPS and ethinylestradiol was effective in improving the exacerbation of MD in fertile women affected by premenstrual syndrome (25).
Estrogen receptors are expressed in various tissues and their activation is implicated in several biological functions. In addition, progesterone receptors are expressed at variable levels, and their effects display different intensities or, occasionally, are unknown, in different tissues (26). Although estradiol increases neuronal excitability, progesterone metabolites enhance the inhibitory effects of gamma-amino butyric acid (GABA) (27). In women, electroencephalography and auditory processes may change during the menstrual cycle and postmenopausally (28,29). Of note, estrogen fluctuations during the menstrual cycle may affect neuronal plasticity and the metabolic levels of neurotransmitters and, consequently, the neuronal conduction time into the audiological system. This was observed by studying the auditory brainstem response via auditory-evoked potentials (30).
It has been shown that psychological stress and plasma levels of antidiuretic hormones may exacerbate MD symptoms (31). The cochlea and central auditory system have receptors for both α- and β-estrogen sex hormones, and the female cyclical hormonal changes may influence the inner ear anatomy and function. Moreover, complex genomic and non-genomic mechanisms may affect MD symptoms by regulating the auditory system pathways, GABA, serotonin, dopamine and glutamate levels, as well as the levels of other sex hormones, including testosterone (32).
In addition, olfactory sensitivity in postmenopausal women was found to be enhanced during treatment with DRSP/E2 HT: The antimineralocorticoid activity of DRSP may decrease nasal edema, promoting efficient interaction between odorous substances and receptors (33), further emphasizing the role of sex steroids on non-genital targets. Moreover, the protective activities against osteoporosis and cardiovascular diseases in postmenopausal women are widely known (34). Finally, the risk factors of vertigo include vitamin D deficiency, decreased ionized Ca levels and osteoporosis (35,36). Moreover, low estrogen levels predispose to bone loss following menopause and HT is considered the primary preventative method for the development of osteoporosis (37). The risk of fractures can be reduced in postmenopausal women affected by MD. HT, such as DRSP/E2, can reduce both MD exacerbation and the risk of osteoporosis, thereby reducing the risk of fractures. In conclusion, DRSP, which is the main component of HT, can reduce fluid overload, which serves as the main trigger for MD, and may prove effective for improving the symptoms of this disease.
Despite its significant findings, the present study comes with certain limitations. The therapeutic regimen included DRSP/E2 and was not compared with HTs containing different progestins other than DRSP. Moreover, the present study did not compare women with naturally occurring or surgically induced menopause using estrogen replacement therapy alone, in order to verify whether estrogen was sufficient to improve vertigo, as previously reported (38). Therefore, future studies should aim to address these issues.
Acknowledgements
Not applicable.
Funding Statement
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
CMG, LM and SC contributed to protocol and project development, follow-up/examination/treatment of the patients, data collection or management, data analysis and manuscript writing/editing. AMCR, GP and GC contributed to conception and design, follow-up/examination/treatment of the patients, data collection or management and manuscript writing/editing. SC and LM confirm the authenticity of all the raw data. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The authors declare that the study protocol conformed to the ethical guidelines of the 2013 Helsinki Declaration. The approval of the institutional review board of the department research committees was obtained (Ethics Committee-Catania 1, Azienda Ospedaliero-Universitaria Policlinico ‘G. Rodolico-San Marco’ Catania, Italy; reference number: 0033012/TMP/10-2015). Written informed consent was obtained directly from all women.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Hale GE, Zhao X, Hughes CL, Burger HG, Robertson DM, Fraser IS. Endocrine features of menstrual cycles in middle and late reproductive age and the menopausal transition classified according to the staging of reproductive Aging workshop (STRAW) staging system. J Clin Endocrinol Metab. 2007;92:3060–3067. doi: 10.1210/jc.2007-0066. [DOI] [PubMed] [Google Scholar]
- 2.National Institute of Health State-of-the-Science Conference statement: Management of menopause-related symptoms. Ann Intern Med. 2005;142:1003–1013. National Institutes of Health. [PubMed] [Google Scholar]
- 3.Ekblad S, Bergendabl A, Enler P, Ledin T, Möllen C, Hammar M. Disturbances in postural balance are common in postmenopausal women with vasomotor symptoms. Climacteric. 2000;3:192–198. doi: 10.1080/13697130008500097. [DOI] [PubMed] [Google Scholar]
- 4.Muncie HL, Sirmans SM, James E. Dizziness: Approach to evaluation and management. Am Fam Physician. 2017;95:154–162. [PubMed] [Google Scholar]
- 5.Bruderer SG, Bodmer D, Stohler NA, Jick SS, Meier CR. Population-based study on the epidemiology of Ménière's disease. Audiol Neurootol. 2017;22:74–82. doi: 10.1159/000475875. [DOI] [PubMed] [Google Scholar]
- 6.Lopez-Escamez JA, Carey J, Chung WH, Goebel JA, Magnusson M, Mandalà M, Newman-Toker DE, Strupp M, Suzuki M, Trabalzini F, et al. Diagnostic criteria for Menière's disease. J Vestib Res. 2015;25:1–7. doi: 10.3233/VES-150549. [DOI] [PubMed] [Google Scholar]
- 7.Jian H, Yu G, Chen G, Lin N, Wang H. Correlation between auditory-vestibular functions and estrogen levels in postmenopausal patients with Meniere's disease. J Clin Lab Anal. 2019;33(e22626) doi: 10.1002/jcla.22626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shojaku H, Watanabe Y, Fujisaka M, Tsubota M, Kobayashi K, Yasumura S, Mizukoshi K. Epidemiologic characteristics of definite Meniere's disease in Japan. A long-term survey of Toyama and Niigata prefectures. ORL J Otorhinolaryngol Relat Spec. 2005;67:305–309. doi: 10.1159/000089413. [DOI] [PubMed] [Google Scholar]
- 9.Baber RJ, Panay N, Fenton A. 2016 IMS recommendations on women's midlife health and menopause hormone therapy. Climacteric. 2019;19:109–150. doi: 10.3109/13697137.2015.1129166. IMS Writing Group. [DOI] [PubMed] [Google Scholar]
- 10.Foidart JM. Added benefits of drospirenone for compliance. Climacteric 8 Suppl. 2005;3:S28–S34. doi: 10.1080/13697130500330309. [DOI] [PubMed] [Google Scholar]
- 11.Sitruk-Ware R. Pharmacology of different progestogens: The special case of drospirenone. Climacteric. 2005;8 (Suppl 3):S4–S12. doi: 10.1080/13697130500330382. [DOI] [PubMed] [Google Scholar]
- 12.Palacios S, Foidart JM, Genazzani AR. Advances in hormone replacement therapy with drospirenone, a unique progestogen with aldosterone receptor antagonism. Maturitas. 2006;55:297–307. doi: 10.1016/j.maturitas.2006.07.009. [DOI] [PubMed] [Google Scholar]
- 13.Archer DF. Drospirenone and estradiol: A new option for the postmenopausal woman. Climacteric. 2007;10 (Suppl 1):S3–S10. doi: 10.1080/13697130601114859. [DOI] [PubMed] [Google Scholar]
- 14.Schneider HP, Heinemann LA, Rosemeier HP, Potthoff P, Behre HM. The menopause rating scale (MRS): Comparison with Kupperman index and quality-of-life scale SF-36. Climacteric. 2000;3:50–58. doi: 10.3109/13697130009167599. [DOI] [PubMed] [Google Scholar]
- 15.Semont A, Freyss G, Vitte E. Curing the BPPV with a liberatory maneuver. Adv Otorhinolaryngol. 1988;42:290–293. doi: 10.1159/000416126. [DOI] [PubMed] [Google Scholar]
- 16.Zhang X, Qian X, Lu L, Chen J, Liu J, Lin C, Gao X. Effects of Semont maneuver on benign paroxysmal positional vertigo: A meta-analysis. Acta Otolaryngol. 2017;137:63–70. doi: 10.1080/00016489.2016.1212265. [DOI] [PubMed] [Google Scholar]
- 17.Kapteyn TS, Bles W, Njiokiktjien CJ, Kodde L, Massen CH, Mol JM. Standardization in platform stabilometry being a part of posturography. Agressologie. 1983;24:321–326. [PubMed] [Google Scholar]
- 18.Prieto TE, Myklebust JB, Hoffmann RG, Lovett EG, Myklebust BM. Measures of postural steadiness: Differences between healthy young and elderly adults. IEEE Trans Biomed Eng. 1996;43:956–966. doi: 10.1109/10.532130. [DOI] [PubMed] [Google Scholar]
- 19.Jacobson GP, Newman CW. The development of the Dizziness handicap inventory. Arch Otolaryngol Head Neck Surg. 1990;116:424–427. doi: 10.1001/archotol.1990.01870040046011. [DOI] [PubMed] [Google Scholar]
- 20.Foster CA. Optimal management of Ménière's disease. Ther Clin Risk Manag. 2015;11:301–307. doi: 10.2147/TCRM.S59023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Miyashita T, Inamoto R, Fukuda S, Hoshikawa H, Hitomi H, Kiyomoto H, Nishiyama A, Mori N. Hormonal changes following a low-salt diet in patients with Ménière's disease. Auris Nasus Larynx. 2017;44:52–57. doi: 10.1016/j.anl.2016.03.001. [DOI] [PubMed] [Google Scholar]
- 22.Pullen RL Jr. Navigating the challenges of Meniere disease. Nursing. 2017;47:38–45. doi: 10.1097/01.NURSE.0000520504.06428.ce. [DOI] [PubMed] [Google Scholar]
- 23.Watanabe I. Ménière's disease in males and females. Acta Otolaryngol. 1981;91:511–514. doi: 10.3109/00016488109138535. [DOI] [PubMed] [Google Scholar]
- 24.Basura GJ, Adams ME, Monfared A, Schwartz SR, Antonelli PJ, Burkard R, Bush ML, Bykowski J, Colandrea M, Derebery J, et al. Clinical practice guideline: Ménière's disease. Otolaryngol Head Neck Surg. 2020;162 (Suppl 2):S1–S55. doi: 10.1177/0194599820909438. [DOI] [PubMed] [Google Scholar]
- 25.Caruso S, Mauro D, Maiolino L, Grillo C, Rapisarda AMC, Cianci S. Effects of combined oral contraception containing drospirenone on premenstrual exacerbation of Meniere's disease: Preliminary study. Eur J Obstet Gynecol Reprod Biol. 2018;224:102–107. doi: 10.1016/j.ejogrb.2018.03.015. [DOI] [PubMed] [Google Scholar]
- 26.Farage MA, Neill S, MacLean AB. Physiological changes associated with the menstrual cycle: A review. Obstet Gynecol Surv. 2009;64:58–72. doi: 10.1097/OGX.0b013e3181932a37. [DOI] [PubMed] [Google Scholar]
- 27.Smith MJ, Keel JC, Greenberg BD, Adams LF, Schmidt PJ, Rubinow DA, Wassermann EM. Menstrual cycle effects on cortical excitability. Neurology. 1999;53:2069–2072. doi: 10.1212/wnl.53.9.2069. [DOI] [PubMed] [Google Scholar]
- 28.Al-Mana D, Ceranic B, Djahanbakhch O, Luxon LM. Hormones and the auditory system: A review of physiology and pathophysiology. Neuroscience. 2008;153:881–900. doi: 10.1016/j.neuroscience.2008.02.077. [DOI] [PubMed] [Google Scholar]
- 29.Caruso S, Cianci A, Grasso D, Agnello C, Galvani F, Maiolino L, Serra A. Auditory brainstem response in postmenopausal women treated with hormone replacement therapy: A pilot study. Menopause. 2000;7:178–183. doi: 10.1097/00042192-200007030-00008. [DOI] [PubMed] [Google Scholar]
- 30.Caruso S, Maiolino L, Agnello C, Garozzo A, Di Mari L, Serra A. Effects of patch or gel estrogen therapies on auditory brainstem response in surgically postmenopausal women: A prospective, randomized study. Fertil Steril. 2003;79:556–561. doi: 10.1016/s0015-0282(02)04763-5. [DOI] [PubMed] [Google Scholar]
- 31.Aoki M, Asai M, Nishihori T, Mizuta K, Ito Y, Ando K. The relevance of an elevation in the plasma vasopressin levels to the pathogenesis of Meniere's attack. J Neuroendocrinol. 2007;19:901–906. doi: 10.1111/j.1365-2826.2007.01601.x. [DOI] [PubMed] [Google Scholar]
- 32.Canlon B, Frisina RD. Sex hormones and hearing: A pioneering area of enquiry. Hear Res. 2009;252:1–4. doi: 10.1016/j.heares.2009.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Caruso S, Serra A, Grillo C, De Leo V, Maiolino L, Agnello C, Cianci A. Prospective study evaluating olfactometric and rhinomanometric outcomes in postmenopausal women on 1 mg 17beta-estradiol and 2 mg drospirenone HT. Menopause. 2008;15:967–972. doi: 10.1097/gme.0b013e31816be973. [DOI] [PubMed] [Google Scholar]
- 34.Nelson HD, Humphrey LL, Nygren P, Teutsch SM, Allan JD. Postmenopausal hormone replacement therapy. Scientific review. JAMA. 2002;288:872–881. doi: 10.1001/jama.288.7.872. [DOI] [PubMed] [Google Scholar]
- 35.Kim SY, Han SH, Kim YH, Park MH. Clinical features of recurrence and osteoporotic changes in benign paroxysmal positional vertigo. Auris Nasus Larynx. 2016;44:156–161. doi: 10.1016/j.anl.2016.06.006. [DOI] [PubMed] [Google Scholar]
- 36.Yu S, Liu F, Cheng Z, Wang Q. Association between osteoporosis and benign paroxysmal positional vertigo: A systematic review. BMC Neurol. 2014;14(110) doi: 10.1186/1471-2377-14-110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vignozzi L, Malavolta N, Villa P, Mangili G, Migliaccio S, Lello S. Consensus statement on the use of HRT in postmenopausal women in the management of osteoporosis by SIE, SIOMMMS and SIGO. J Endocrinol Invest. 2019;42:609–618. doi: 10.1007/s40618-018-0978-x. [DOI] [PubMed] [Google Scholar]
- 38.Liu DH, Kuo CH, Wang CT, Chiu CC, Chen TJ, Hwang DK, Kao CL. Age-related increases in benign paroxysmal positional vertigo are reversed in women taking estrogen replacement therapy: A population-based study in Taiwan. Front Aging Neurosci. 2017;9(404) doi: 10.3389/fnagi.2017.00404. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.