Abstract
COVID-19, the disease caused by the SARS-CoV-2 virus, can progress to multisystem organ failure and viral sepsis characterized by respiratory failure, arrhythmias, thromboembolic complications, and shock with high mortality. Autopsy and preclinical evidence implicate aberrant complement activation in endothelial injury and organ failure. Erythrocytes express complement receptors and are capable of binding immune complexes; therefore, we investigated complement activation in patients with COVID-19 using erythrocytes as a tool to diagnose complement activation. We discovered enhanced C3b and C4d deposition on erythrocytes in COVID-19 sepsis patients and non-COVID sepsis patients compared with healthy controls, supporting the role of complement in sepsis-associated organ injury. Our data suggest that erythrocytes may contribute to a precision medicine approach to sepsis and have diagnostic value in monitoring complement dysregulation in COVID-19-sepsis and non-COVID sepsis and identifying patients who may benefit from complement targeted therapies.
Keywords: complement, COVID-19, erythrocyte, SARS-CoV-2, sepsis
INTRODUCTION
Despite the current availability of vaccines against SARS-CoV-2, COVID-19 continues to inflict high morbidity and mortality (1). Given the concerns of highly infectious emerging variants, new strategies are urgently needed to rapidly identify and treat patients, especially those at risk for respiratory failure. Clinical and epidemiologic studies demonstrate that vulnerability to organ failure is the greatest after viral clearance from the upper airway, suggesting that dysregulation of the host immune response is a critical mediator of clinical deterioration and death (2). Autopsy and preclinical evidence implicate aberrant complement activation in endothelial injury and organ failure, and recent epidemiologic studies have found an association between complement dysregulation and acute respiratory distress syndrome (ARDS) (3–6). One potential therapeutic strategy is based on case reports of successful treatment of COVID-19 with inhibitors of complement (7, 8). However, this approach requires a careful balance between the host protective and potential injurious effects of complement activation, and biomarkers to identify the optimal timing and candidates for therapy are lacking.
The complement system provides a frontline defense against invading microorganisms and is a highly coordinated system vital for the innate immune response to pathogens, the removal of dead cells, and the maintenance of homeostasis. The three pathways of complement activation can be engaged by distinct initiators, including antigen-antibody complexes (classical pathway), lectins (lectin pathway), and spontaneous C3 hydrolysis (alternative pathway). Although the complement system promotes clearance of pathogens through opsonization, inflammation, and cytolysis, dysregulated complement activation can lead to cellular injury, microvascular thrombosis, and organ failure. Animal models with complement regulator deficiencies or mutations are prone to complement-mediated tissue injury, coagulopathy, and thrombotic microangiopathy (9). The complement system is also implicated in CoV-mediated tissue injury (6).
Complement-containing immune complexes bind to cells through specific receptors, including complement receptor 1 (CR1), which recognizes complement activation products C3b and iC3b. Given the sheer number of red blood cells (RBCs) in circulation, it is not surprising that RBCs account for the majority of CR1 expression in mammals. Immune adherence, the binding of antigen-antibody-complement complex to RBCs first described half a century ago, is one mechanism by which RBCs modulate innate responses (10, 11). Here, we report complement deposition on circulating erythrocytes from hospitalized COVID-19 patients using flow cytometry. These findings suggest that novel erythrocyte-based diagnostics can identify patients with dysregulated complement activation.
METHODS
SARS-CoV-2/COVID-19 Cohort and Blood Processing
Whole blood was obtained in EDTA tubes (BD Bioscience) from inpatient subjects positive for SARS-CoV-2 enrolled in the Molecular Epidemiology of SepsiS in the ICU-COVID (MESSI-COVID) study at the University of Pennsylvania, except for those samples used for ELISA, which were collected in sodium citrate. Studies involving human subjects were approved by the University of Pennsylvania Institutional Review Board (12). Subjects were screened and approached for informed consent during the first 3 days of hospitalization. Written informed consent, in accordance with protocols approved by the regional ethical research boards and the Declaration of Helsinki, was given by subjects or their proxies. Samples were processed within 3 h of collection. Whole blood was centrifuged for 15 min at 3,000 rpm, and plasma was collected.
ELISA
Immulon 4 HBx 96-well plates were coated with recombinant SARS-CoV-2 spike protein (2 µg/mL, BEI Resources) for 2 h, washed three times with 0.1% Tween-20 in PBS (PBS/T, wash buffer), then blocked for 1 h with 3% nonfat milk in PBS/T. Plasma from patient with COVID-19 diluted 1:20 with 1% nonfat milk in PBS/T was added for 1 h, then plates were washed three times. Complement deposition was detected by adding C3/C3b/iC3b/C3d antibody (0.3 µg/mL, BioLegend) in 1% nonfat milk in PBS/T for 30 min. After subsequent washes, peroxidase-conjugated goat anti-mouse IgG (0.1 µg/mL) was added for 30 min, then washed plates were developed with TMB substrate (Seracare). After 5 min, the enzymatic reaction was stopped with 1 M HCl, and optical density (OD) at 450 nm was measured on a Synergy 2 plate reader (BioTek Instruments).
Red Blood Cell Isolation
Whole blood was obtained from healthy donors (HD) in EDTA tubes. Blood from HD and patients was centrifuged for 5 min at 800 g, and the remaining plasma and buffy coat were removed. Red blood cells (RBCs) were then diluted at 1 µL RBC/1 mL PBS for flow cytometry and microscopy. All work was conducted in the biosafety cabinet under enhanced BSL2 conditions.
Flow Cytometry
Diluted RBCs (100 µL) were incubated with anti-C3/C3b/iC3b/C3d (BioLegend, 1 µg/mL), anti-C4d (Immunoquest, 1 mg/mL), or isotype (BioLegend, 1 ≤ g/mL) antibody for 45 min followed by two washes in PBS. Cells were resuspended in 100 µL of phycoerythrin (PE) goat-anti-mouse IgG secondary antibody (BioLegend, 2 µg/mL) for 45 min before two washes in PBS and fixation in electron microscopy (EM)-grade glutaraldehyde (Polysciences, 2.5%) for 15 min. After subsequent washes, data were acquired using the LSR Fortessa, and analysis was performed using FlowJo software.
Microscopy
Diluted RBCs were fixed in 0.05% glutaraldehyde for 10 min before two washes in FACS buffer (2% FBS in PBS) followed by resuspension in FACS (1 µL RBC/1 mL buffer). Hundred microliters of diluted RBCs were incubated with anti-C3/C3b/iC3b/C3d or anti-SARS-CoV spike (GeneTex, 1:200) antibodies overnight at 4°C. Cells were washed twice in FACS and incubated with AlexaFluor488 goat-anti-mouse IgG secondary antibody (Thermofisher, 2 µg/mL) for 45 min. After subsequent washes, cells were fixed in 2.5% glutaraldehyde, washed, and mounted on slides with fluoromount G. Slides were imaged using Nikon eclipse microscope, and images were analyzed using ImageJ software. Three to five fields/sample were counted for quantification.
Statistics
Clinical correlations were analyzed by Stata (StataCorp LLC), and all other statistical analyses were performed with SigmaPlot (SyStat Software Inc.)
RESULTS AND DISCUSSION
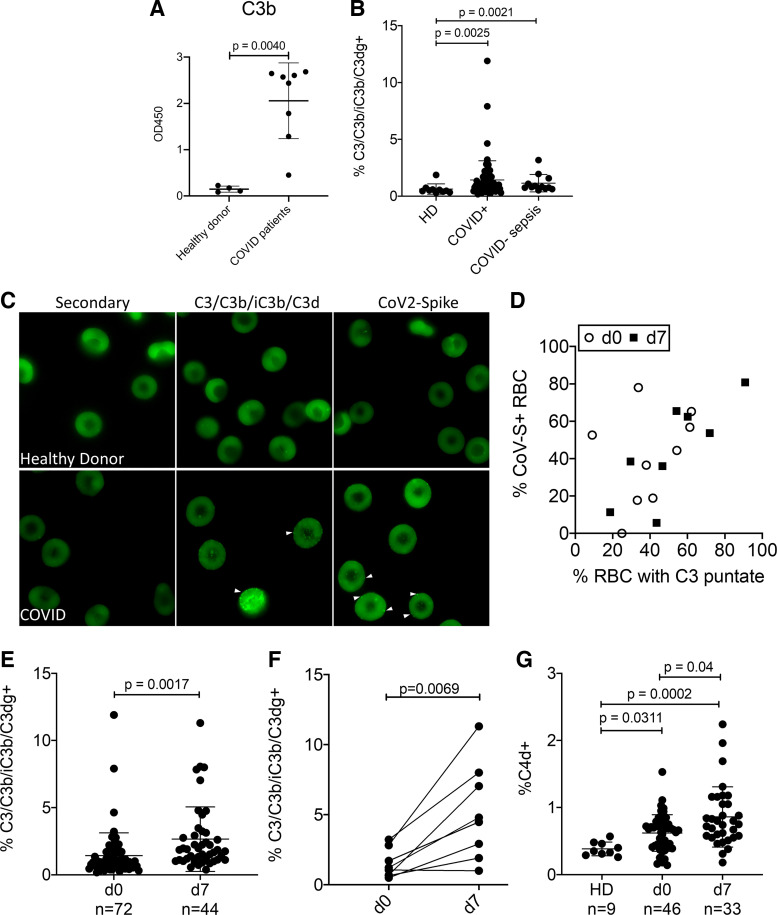
To detect complement activation in patients with COVID-19, we performed ELISA to detect the deposition of C3b by patient plasma on immobilized spike protein (Fig. 1A). Patient plasma deposited significantly more C3b on spike-coated plates than did plasma from healthy subjects (P < 0.004); this would suggest that SARS-CoV-2 infection led to complement activation in vivo. Then, we measured erythrocyte-bound C3b/iC3b/C3dg and C4d on healthy donors, hospitalized COVID patients, and COVID-negative septic patients using flow cytometry (patient characteristics are described in Table 1). The percentage of RBCs with bound C3b/iC3b/C3dg and C4d was markedly elevated in hospitalized sepsis, and patients with COVID-19 were admitted to the ICU compared with healthy donors, and these percentages increased even further by day 7 in patients with COVID-19 (Fig. 1, B and E–G). Immunofluorescence demonstrated that patient erythrocytes bind C3 fragments and bound viral spike protein, suggesting activation of complement and immune complex deposition on the RBCs (Fig. 1, C and D). Together, these data indicate that complement activation products and viral antigens are present on RBCs in patients with COVID-19.
Figure 1.
Complement activation and deposition on RBCs during COVID-sepsis and non-COVID sepsis. A: ELISA for C3/C3b/iC3b deposition on immobilized spike protein. Plasma from healthy donors and patients with COVID-19 was tested, n = 4–9 subjects, P < 0.004. B: C3 deposition on RBCs from healthy donors (HD, n = 10), patients hospitalized with COVID-19-associated organ injury (n = 72), and COVID-negative septic patients (n = 11) evaluated on day (d) 0 of ICU admission (P = 0.0025 and P = 0.0021, one-way ANOVA with Dunn’s post hoc; HD vs. COVID and HD vs. non-COVID sepsis). C: C3 and CoV spike protein detection on RBCs, bright green punctae indicate positive staining, RBCs from a healthy donor are shown for control. Results were quantified and correlations between d0 and d7 are shown in D, d0: r2 = 0.59, P = 0.01; d7: r2 = 0.78, P = 0.014. E: C3 deposition on RBCs in patients with COVID. Comparison of C3 on RBCs at d0 and d7 of study enrollment, P = 0.0017, t test. F: RBC-bound C3 from individual patients on d0 and d7, P = 0.0069, paired t test. G: C4d deposition on RBCs from patients with COVID-19 (one-way ANOVA, Dunn’s post hoc). CoV-S, coronavirus-spike protein; ICU, intensive care unit; OD, optical density; RBCs, red blood cells.
Table 1.
Baseline characteristics of COVID-19 and non-COVID-19 sepsis patients
| COVID-19 Patients (n = 72) | Non-COVID-19 Sepsis (n = 11) | |
|---|---|---|
| Age, yr | 60 ± 14 | 57 ± 14 |
| Male, n (%) | 42 (58) | 3 (27) |
| Race, n (%) | ||
| Black | 51 (71) | 4 (36) |
| White | 17 (24) | 6 (55) |
| Asian | 4 (5) | 1 (9) |
| Ventilator/oxygen support, n (%) | ||
| Invasive ventilation | 25 (35) | 5 (45) |
| Noninvasive ventilation* | 13 (18) | 2 (18) |
| Nasal cannula oxygen | 22 (31) | 3 (27) |
| No support | 12 (17) | 1 (9) |
| ARDS, n (%)† | 24 (33) | 2 (18) |
| Septic shock, n (%) | 16 (22) | 6 (55) |
| Clotting complication, n (%) | 15 (21) | 4 (36) |
| Mortality, n (%) | 13 (18) | 2 (18) |
*Noninvasive ventilation includes noninvasive mask ventilation, helmet ventilation, and high flow nasal cannula oxygenation. †Acute respiratory distress syndrome (ARDS) defined by the Berlin criteria with the added requirement of invasive mechanical ventilation.
Although the small sample size precludes drawing associations between RBC-bound complement and clinical outcomes, several clinically relevant implications arise from our findings. First, our findings may support a potential role for complement-mediated pathophysiology in patients with COVID-19, which is consistent with recently published studies (3, 4). Second, because plasma-based assays of individual complement components or total complement activity may fail to reflect the levels of complement activation on cell surfaces responsible for tissue injury, flow cytometry of RBCs may serve as a readily accessible, sensitive, and more relevant measure of complement activation. Third, an increase in RBC-bound complement over time might serve as a biomarker for severe COVID-19 disease and help depict disease trajectory and identify potential patients for clinical trials of anticomplement therapy. Although immune adherence may serve to immobilize and transport pathogens to immune cells, deposition of immune complexes and complement on RBCs may alter their rheology and promote intravascular stasis and thrombosis, contributing to the pathogenesis of virus-associated lung injury.
In sum, our findings suggest that RBC-immune adherence occurs in patients with COVID-19, and flow cytometric analysis of RBCs provides a clinically feasible means to help identify patients who are susceptible to complement-mediated host injury and might therefore benefit from intervention with complement inhibitors. The role of RBC immune adherence in delivering viral antigen to immune cells has yet to be elucidated. However, exploiting this innate immune mechanism to detect pathologic complement activation may hold one key to preventing organ injury in infectious and autoimmune systemic disorders characterized by complement dysregulation.
DATA AVAILABILITY
All data are available in the main text.
GRANTS
The research was supported by National Institutes of Health (National Heart, Lung, and Blood Institute Grants HL126788 to N.S.M., HL137006 to N.J.M., and HL128895 and HL142122 to D.B.C.) and the Penn Center for Precision Medicine (N.S.M).
DISCLOSURES
E.J.W. is a member of the Parker Institute for Cancer Immunotherapy. E.J.W. has consulting agreements with and/or is on the scientific advisory board for Merck, Roche, Pieris, Elstar, and Surface Oncology. E.J.W. is a founder of Surface Oncology and Arsenal Biosciences. E.J.W. has a patent licensing agreement on the PD-1 pathway with Roche/Genentech. J.D.L. is the founder of Amyndas Pharmaceuticals, which is developing complement inhibitors for therapeutic purposes and is the inventor of patents or patent applications that describe the use of complement inhibitors for therapeutic purposes some of which are developed by Amyndas. J.D.L. is also the inventor of the compstatin technology licensed to Apellis Pharmaceuticals. N.J.M has received grant funding to her institution from Athersys, Inc., Biomarck, Inc., Quantum Leap Healthcare Consortium, and the Marcus Foundation for work unrelated to manuscript under consideration. W.-C.S. is a cofounder and a consultant to Kira Pharmaceuticals and Aevitas Therapeutics from which he receives research grants. None of the other authors has any conflicts of interest, financial or otherwise, to disclose.
AUTHOR CONTRIBUTIONS
L.K.M.L., D.B.C., N.S.M., J.P.R., and A.H.R. conceived and designed research; L.K.M.L., E.J.W., W.-C.S., N.S.M., J.P.R., A.H.R., S.J.M., L.K.-C., A.R.W., C.A.G.I., M.B.P., and M.R.B. performed experiments; L.K.M.L., W.-C.S., J.D.L., N.J.M., D.B.C., N.S.M., J.P.R., A.H.R., A.R.W., C.A.G.I., M.B.P., and M.R.B. analyzed data; L.K.M.L., J.D.L., N.J.M., N.S.M., J.P.R., A.H.R., and M.B.P. interpreted results of experiments; L.K.M.L., N.S.M., J.P.R., and A.H.R. prepared figures; L.K.M.L., D.B.C., N.S.M., J.P.R., and A.H.R. drafted manuscript; L.K.M.L., E.J.W., W.-C.S., J.D.L., N.J.M., D.B.C., N.S.M., J.P.R., A.H.R., S.J.M., L.K.-C., A.R.W., C.A.G.I., M.B.P., and M.R.B. edited and revised manuscript; L.K.M.L., D.B.C., N.S.M., J.P.R., and A.H.R. approved final version of manuscript.
ACKNOWLEDGMENTS
We thank the nurses and staff at the Hospital of the University of Pennsylvania for procuring patient samples. We also thank Peggy Zhang for excellent technical assistance.
Preprint is available at https://doi.org/10.1101/2020.05.20.20104398.
REFERENCES
- 1.Anesi GL, Jablonski J, Harhay MO, Atkins JH, Bajaj J, Baston C, et al. Characteristics, outcomes, and trends of patients with COVID-19-related critical illness at a learning health system in the United States. Ann Intern Med 174: 613–621, 2021. doi: 10.7326/M20-5327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liu Y, Yan L-M, Wan L, Xiang T-X, Le A, Liu J-M, Peiris M, Poon LLM, Zhang W. Viral dynamics in mild and severe cases of COVID-19. Lancet Infect Dis 20: 656–657, 2020. doi: 10.1016/S1473-3099(20)30232-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Holter JC, Pischke SE, de Boer E, Lind A, Jenum S, Holten AR, Tonby K, Barratt-Due A, Sokolova M, Schjalm C, Chaban V, Kolderup A, Tran T, Gjolberg TT, Skeie LG, Hesstvedt L, Ormasen V, Fevang B, Austad C, Muller KE, Fladeby C, Holberg-Petersen M, Halvorsen B, Muller F, Aukrust P, Dudman S, Ueland T, Andersen JT, Lund-Johansen F, Heggelund L, Dyrhol-Riise AM, Mollnes TE. Systemic complement activation is associated with respiratory failure in COVID-19 hospitalized patients. Proc Natl Acad Sci USA 117: 25018–25025, 2020. doi: 10.1073/pnas.2010540117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Magro C, Mulvey JJ, Berlin D, Nuovo G, Salvatore S, Harp J, Baxter-Stoltzfus A, Laurence J. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res 220: 1–13, 2020. doi: 10.1016/j.trsl.2020.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gao T, Hu M, Zhang X, Li H, Zhu L, Liu H, et al. Highly pathogenic coronavirus N protein aggravates lung injury by MASP-2-mediated complement over-activation (Preprint). medRxiv, 2020. doi: 10.1101/2020.03.29.20041962. [DOI] [Google Scholar]
- 6.Wilk CM. Coronaviruses hijack the complement system. Nat Rev Immunol 20: 350–350, 2020. doi: 10.1038/s41577-020-0314-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Diurno F, Numis FG, Porta G, Cirillo F, Maddaluno S, Ragozzino A, De Negri P, Di Gennaro C, Pagano A, Allegorico E, Bressy L, Bosso G, Ferrara A, Serra C, Montisci A, D’Amico M, Lo Morello SS, Di Costanzo G, Tucci AG, Marchetti P, Di Vincenzo U, Sorrentino I, Casciotta A, Fusco M, Buonerba C, Berretta M, Ceccarelli M, Nunnari G, Diessa Y, Cicala S, Facchini G. Eculizumab treatment in patients with COVID-19: preliminary results from real life ASL Napoli 2 Nord experience. Eur Rev Med Pharmacol Sci 24: 4040–4047, 2020. doi: 10.26355/eurrev_202004_20875. [DOI] [PubMed] [Google Scholar]
- 8.Mastaglio S, Ruggeri A, Risitano AM, Angelillo P, Yancopoulou D, Mastellos DC, Huber-Lang M, Piemontese S, Assanelli A, Garlanda C, Lambris JD, Ciceri F. The first case of COVID-19 treated with the complement C3 inhibitor AMY-101. Clin Immunol 215: 108450, 2020. doi: 10.1016/j.clim.2020.108450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ueda Y, Mohammed I, Song D, Gullipalli D, Zhou L, Sato S, Wang Y, Gupta S, Cheng Z, Wang H, Bao J, Mao Y, Brass L, Zheng XL, Miwa T, Palmer M, Dunaief J, Song W-C. Murine systemic thrombophilia and hemolytic uremic syndrome from a factor H point mutation. Blood 129: 1184–1196, 2017. doi: 10.1182/blood-2016-07-728253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nelson RA Jr.The immune-adherence phenomenon; an immunologically specific reaction between microorganisms and erythrocytes leading to enhanced phagocytosis. Science 118: 733–737, 1953.doi: 10.1126/science.118.3077.733. [DOI] [PubMed] [Google Scholar]
- 11.Anderson HL, Brodsky IE, Mangalmurti NS. The evolving erythrocyte: red blood cells as modulators of innate immunity. J Immunol 201: 1343–1351, 2018. doi: 10.4049/jimmunol.1800565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kuri-Cervantes L, Pampena MB, Meng W, Rosenfeld AM, Ittner CAG, Weisman AR, Agyekum RS, Mathew D, Baxter AE, Vella LA, Kuthuru O, Apostolidis SA, Bershaw L, Dougherty J, Greenplate AR, Pattekar A, Kim J, Han N, Gouma S, Weirick ME, Arevalo CP, Bolton MJ, Goodwin EC, Anderson EM, Hensley SE, Jones TK, Mangalmurti NS, Prak ETL, Wherry EJ, Meyer NJ, Betts MR. Comprehensive mapping of immune perturbations associated with severe COVID-19. Sci Immunol 5: eabd7114, 2020. doi: 10.1126/sciimmunol.abd7114. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data are available in the main text.