Abstract
BACKGROUND
Various histological types of gastric carcinomas (GCs) differ in terms of their pathogenesis and their preexisting background, both of which could impact the tumor immune microenvironment (TIME). However, the current understanding of the immune contexture of GC is far from complete.
AIM
To clarify the tumor-host immune interplay through histopathological features and the tumor immune cycle concept.
METHODS
In total, 50 GC cases were examined (15 cases of diffuse GC, 31 patients with intestinal-type GC and 4 cases of mucinous GC). The immunophenotype of GC was assessed and classified as immune desert (ID), immune excluded (IE) or inflamed (Inf) according to CD8+ cell count and spatial pattern. In addition, CD68+ and CD163+ macrophages and programmed death-ligand 1 (PD-L1) expression were estimated.
RESULTS
We found that GCs with different histological differentiation demonstrated distinct immune contexture. Most intestinal-type GCs had inflamed TIMEs rich in both CD8+ cells and macrophages. In contrast, more aggressive diffuse-type GC more often possessed ID characteristics with few CD8+ lymphocytes but abundant CD68+ macrophages, while mucinous GC had an IE-TIME with a prevalence of CD68+ macrophages and CD8+ lymphocytes in the peritumor stroma. PD-L1 expression prevailed mostly in intestinal-type Inf-GC, with numerous CD163+ cells observed. Therefore, GCs of different histological patterns have specific mechanisms of immune escape. While intestinal-type GC was more often related to PD-L1 expression, diffuse and mucinous GCs possessing more aggressive behavior demonstrated low immunogenicity and a lack of tumor antigen recognition or immune cell recruitment into the tumor clusters.
CONCLUSION
These data help to clarify the links between tumor histogenesis and immunogenicity for a better understanding of GC biology and more tailored patient management.
Keywords: Gastric carcinoma, Tumor immune microenvironment, Tumor infiltrating lymphocytes, Tumor associated macrophages
Core Tip: In this study, we investigated the tumor-host interplay in gastric carcinoma (GC) through the tumor immune cycle concept. Histologically different GCs vary in immunogenicity and differ in tumor-infiltrating lymphocyte and macrophage densities. Intestinal GC demonstrated predominantly inflamed tumor immune microenvironment and frequent programmed death-ligand 1 expression. In contrast, more aggressive diffuse and mucinous GCs possessed low immunogenicity with a lack of cancer antigen recognition and trafficking. These data help to clarify the links between tumor histogenesis and immunogenicity, offering a better understanding of GC biology and the ability to provide more tailored patient management.
INTRODUCTION
Gastric carcinoma (GC) is one of the leading fatal malignancies worldwide[1]. Currently, GC prognosis and patient management are based on the UICC/American Joint Committee on Cancer (AJCC) tumour-node-metastasis staging system[2]. When assessing tumour prognosis, GC histopathological features are also significant[3]. The Laurén pathohistological classification is the most widely used in clinical practice because it reflects the GC morphology, specifies the tumour biology and behaviour and is helpful for treatment decisions[3,4]. However, GC patients with the same TNM stages and similar histopathological features may differ in outcomes[5]. Therefore, the current approach provides incomplete information for patient prognostic stratification and treatment decisions[5]. Recent large-scale studies, including genomic, transcriptomic and proteomic investigations, defined several molecular classifications for GC[6]. The Cancer Genome Atlas Research Network Group[7] suggested a molecular classification that divides GC into four main subtypes, including Epstein-Barr virus (EBV)-positive, microsatellite unstable (MSI), genomically stable and GC with chromosomal instability[8]. At the same time, the Asian Cancer Research Group established another classification with the following types: MSI, microsatellite stable (MSS) with epithelial-mesenchymal transition, MSS/TP53+ and MSS/TP53−[9]. However, unlike breast cancer, GC molecular classifications have not yet been connected to treatment options[10].
A growing body of evidence has demonstrated the clinical importance of immune cells for tumour prognosis and immunotherapy sensitivity[11]. Tumour-infiltrating immune cells, including neutrophils, macrophages, dendritic cells and different types of T cells, are major contributors to the tumour immune microenvironment (TIME), which modulates tumour development and progression[11]. Previous studies have demonstrated a correlation between the number of tumour-infiltrating lymphocytes (TILs) and the survival of patients with GC[12]. In addition, tumour-associated macrophages (TAMs) are known as alternative regulators of tumour cell proliferation, angiogenesis and tissue remodelling, thereby facilitating metastasis formation in GC[13]. A promising approach is to consider the tumour-host interplay through the mechanisms of tumour immunology and steps of tumour antigen recognition, T-cell activation and tumour cell killing[14].
However, the connections between the histopathological patterns and immune contexture of GC are not well established. This study aims to clarify the interplay among tumour-host immunity, histopathological features and the tumour immune cycle.
MATERIALS AND METHODS
Case selection and patient characteristics
This paper presents a retrospective study that includes 50 cases of primary gastric adenocarcinoma naïve to preoperative chemotherapy or radiotherapy (Table 1). We excluded patients who suffered from other primary malignancies or had a primary tumour of unclear pathological types as well as patients with a known familial or hereditary history of GC. There were 29 males (58%) and 21 females (42%) aged 55.1 ± 3.76 and 45.7 ± 3.59 years, respectively.
Table 1.
Clinical-pathological features of the patients
|
Characteristics
|
Intestinal GC
|
Mucinous GC
|
Diffuse GC
|
Total
|
P
value
|
| Number | 31 | 4 | 15 | 50 | |
| Age | 56.3 11.2 | 51.3 7.37 | 46.0 18.3 | 52.8 13.7 | 0.174 |
| Sex, n (%) | 0.738 | ||||
| Males | 18 (58.1) | 3 (75) | 8 (53.5) | 29 (58) | |
| Females | 13 (41.9) | 1 (25) | 7 (46.7) | 21 (42) | |
| Stage, n (%) | 0.33 | ||||
| Stage 2 | 2 (6.5) | 0 | 0 | 2 (4) | |
| Stage 3 | 13 (41.9) | 1 (25) | 6 (40) | 20 (40) | |
| Stage 4 | 16 (51.6) | 3 (75) | 9 (60) | 28 (56) | |
| Grade, n (%) | - | - | |||
| G1 | 5 (16.1) | ||||
| G2 | 11 (35.5) | ||||
| G3 | 15 (48.4) | ||||
| MMR status | |||||
| MSI | 2 | - | - | 2 | |
| MSS | 29 | 4 | 15 | 48 |
GC: Gastric carcinoma; MMR: Mismatch repair; MSI: Microsatellite unstable; MSS: Microsatellite stable.
The parameters evaluated for each patient included sex, age, tumor location, depth of invasion, grade of differentiation, and tumor staging. Tumor staging was based on the 8th AJCC criteria[15]. Tumor histological type was assessed according to Lauren’s classification[3,16]. Among the enrolled cases, there were 15 cases of diffuse GC and 35 patients with intestinal-type GC. The group with intestinal GC included 4 cases of mucinous GC and 31 nonmucinous adenocarcinomas. As mucinous GCs are rare and have a poor prognosis, we assessed them separately.
Tissue processing and immunohistochemistry
The tissues taken after surgery were fixed in 10% neutral buffered formalin and processed. Histological slides were stained by hematoxylin and eosin, as well as by PAS with Alcian blue. For each tumor, the histological subtype and grade were assessed. For immunohistochemistry (IHC), serial sections 4 μm in thickness were used. Tissues were deparaffinized and hydrated. Endogenous peroxidase activity was blocked using 3% methanol in hydrogen peroxide. Next, antigen retrieval in a water bath at 98 °C was performed using Tris EDTA or citrate buffer (pH 6), followed by incubation with primary antibodies. After washing, labeled polymer secondary antibodies (Envision Detection System, Dako) were added to the slides. Peroxidase activity was detected using diaminobenzidine (DAB)–tetrahydrochloride liquid plus Chromogen System (Dako) substrate. The reaction was stopped with distilled water, after which sections were counterstained with hematoxylin and mounted in Richard-Allan Scientific Mounting Medium (Thermo Fisher).
The following antibodies were used for IHC: CD3 (DAKO; polyclonal) for the entire lineage of T lymphocytes; CD8 (DAKO; Clone C8/144B) for cytotoxic T cells as effector cells of cell-mediated antitumor immunity; and CD68 (DAKO, Clone KP1) and CD163 (Cell Marque, Clone MRQ-26) to visualize TAMs of M1 and M2 phenotypes. In addition, mismatch repair (MMR) deficiency was assessed using antibodies against MLH1 (Clone ES05, DAKO), MSH2 (Clone FE11, DAKO), PMS2 (Clone EP51, DAKO), and MSH6 (Clone EP49, DAKO). Tumors with a lack of MMR enzyme expression in the tumor cell nuclei were defined as MMR deficient or microsatellite unstable (MSI). Staining of stromal and immune cell nuclei was considered a positive control. Additionally, programmed death-ligand 1 (PD-L1) [PDL Cell Signaling, clone E1L3N(R)] expression was detected immunohistochemically. Human tonsil tissue was used as a positive control.
Methodology of tumor-host immunity assessment
To interpret the tumour-host interplay and mechanisms of immune escape, we assessed the TIME according to the immune cycle concept[17]. The following types of TIME were considered: Immune desert, immune-excluded and inflamed. The immune desert (ID) type demonstrates a lack of pre-existing immunity and a few T-cells inside and around the tumour. The immune-excluded (IE) type shows prominent peritumour infiltration but sparse intratumour T lymphocytes. The inflamed (Inf) TIME possesses high lymphocyte infiltration, thus reflecting the activation of antitumour T cells with improper functioning.
When assessing different immune cells, we examined their number and spatial distribution. The primary histopathological assessment was performed microscopically by two independent pathologists. In addition, digital images of the sections were captured using a digital slide scanner (3DHISTECH, Budapest, Hungary) and assessed blindly. The density of CD8+ cells was counted inside the tumour clusters (TCs) and in the peritumour stroma (TS) in 10 visual fields corresponding to “hot spots”, with further quantification per 1 mm2.
The number of immunopositive cells was assessed as both continuous and dichotomized variables using cut-off values (84 cells per mm2 as a median) to separate low-density from high-density results. The ID type of TIME was defined in cases with low CD8+ infiltration in both the TC and TS. The cases of the IE immunophenotype included tumours with a low CD8+ quantity in the TC compartment and high CD8+ infiltration in the TS compartment. GC with high CD8+ cell infiltration in the TC and TS was classified as belonging to the Inf immunophenotype. The same approach was used to evaluate the TAM distribution by counting CD68+ and CD163+ cells in the TC and TS. Immune cells in vessels, submucosal lymphatic areas, and necrosis/necrosis adjacent areas were not counted in this study. The expression of PD-L1 was evaluated using the combined positive score (CPS) according to the percentage of stained tumour or immune (lymphocytes and macrophages) cells, with a positive score defined at CPS ≥ 1%[16].
Statistical analysis
The descriptive statistics included count and frequencies for categorical variables and the mean ± SEM with 95% confidence interval (CI) for continuous variables. Normally distributed continuous variables were analysed with unpaired sample t-tests. Nonnormally distributed continuous variables were assessed with nonparametric Mann-Whitney U-tests. Chi-square or Fisher's exact test was used to compare categorical variables, whereas one-way analysis of variance was used to compare the continuous variables. Correlations were assessed using the Pearson correlation coefficient. Statistical assessment of the data was carried out using the MedCalc software package. Statistical significance was accepted at P < 0.05.
RESULTS
We did not find sex-related differences in the frequency of various histological types of GCs. However, significant differences were observed in the age of patients with diffuse and intestinal GC types, which were 41.1 ± 6.51 (95%СІ: 25.2-57.1) and 53.3 ± 3.07 (46.8-59.8) years old, respectively. However, age differences were not observed between patients with mucinous and nonmucinous intestinal GC.
TIL and TAM densities varied in GC of different histological types
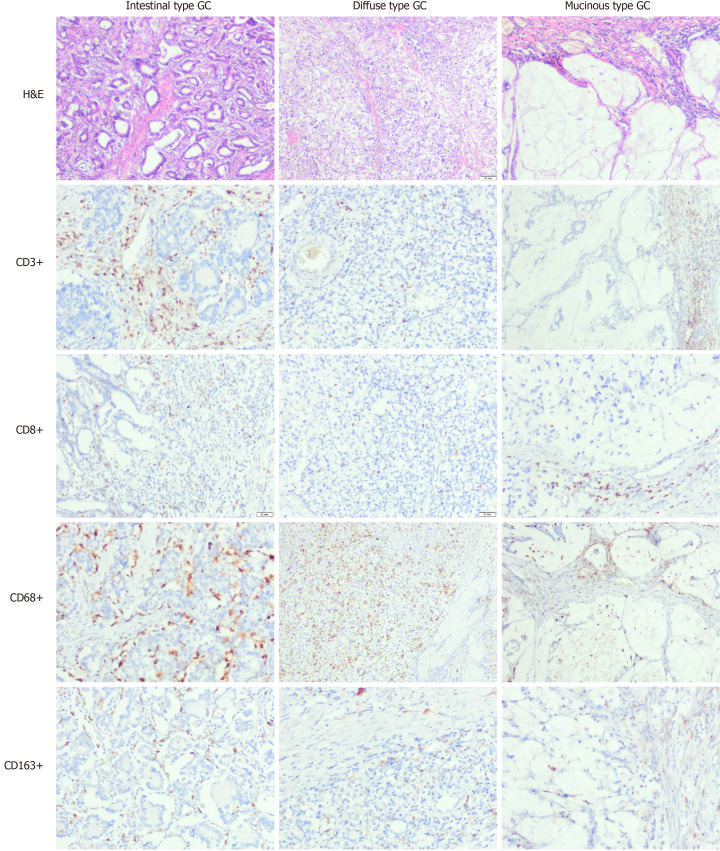
The assessment of GC immune profiles revealed that CD3+ and CD8+ lymphocyte counts varied in GCs with different histological types. However, CD8+ cell density did not correlate with the tumour grade (P = 0.669) or stage (P = 0.560). GCs of various histological differentiation types differed in the density of TILs (Figure 1 and Table 2). The number of CD3+ and CD8+ lymphocytes in intestinal-type GC was significantly higher than that in diffuse and mucinous GCs (P < 0.001). Importantly, mucinous GCs demonstrated prominent heterogeneity of immune cell infiltration, with few cells within tumour clusters and a higher density around them.
Figure 1.
Density of immune cells in gastric carcinoma of different histological types. H&E and immunohistochemistry (× 100). Figures demonstrate differences in infiltration of various histological type gastric carcinoma by immune cells, including entire population of T-lymphocytes (CD3), T-cytotoxic cells (CD8), M1 and M2 macrophages (CD68 and CD163 respectively). GC: Gastric carcinoma.
Table 2.
Immune cells number in gastric carcinoma of different histological types and tumor immune microenvironment
| Characteristics |
CD8+ cells
|
CD68+ cells
|
CD163+ cells
|
|||
|
TC
|
TS
|
TC
|
TS
|
TC
|
TS
|
|
| Histological type | ||||||
| Intestinal | 214 ± 44.9 | 202 ± 14.7 | 303 ± 21.7 | 244 ± 12.4 | 173 ± 17.3 | 170 ± 9.67 |
| 124-305 | 173-232 | 259-347 | 219-269 | 138-208 | 151-190 | |
| Diffuse | 49.5 ± 6.63 | 66.0 ± 6.03 | 339 ± 23.6 | 225 ± 23.8 | 51.7 ± 8.60 | 55.5 ± 8.94 |
| 36.0-63.1 | 43.6-98.3 | 291-388 | 176-274 | 34.1-69.4 | 37.2-73.9 | |
| Mucinous | 7.53 ± 2.50 | 52.5 ± 37.5 | 101 ± 59.0 | 219 ± 16.2 | 12.5 ± 11.8 | 62.5 ± 12.5 |
| 2.26-34.3 | 23.9-82.9 | 64-125 | 126-264 | 6.32-78.2 | 36-121 | |
| P = 0.045 | P = 0.059 | P = 0.071 | P = 0.471 | P = 0.032 | P = 0.011 | |
| TIME type | ||||||
| ID | 43.7 ± 4.05 | 43.2 ± 5.93 | 367.5 ± 36.9 | 213.1 ± 35.9 | 72.5 ± 18.3 | 48.7 ± 10.5 |
| 35.1-52.3 | 30.6-55.7 | 288-446 | 135-290 | 33.2-111 | 26.2-71.2 | |
| IE | 19.4 ± 3.43 | 112 ± 15.2 | 157.3 ± 29.6 | 266.6 ± 25.8 | 55.5 ± 13.6 | 119.5 ± 16.3 |
| 12.2-26.7 | 80.1-144 | 94.5-220 | 211-321 | 26.5-84.4 | 84.9-154.1 | |
| Inflamed | 229 ± 44.9 | 190 ± 16.5 | 352.8 ± 15.6 | 232.6 ± 13.7 | 165.6 ± 18.2 | 151.3 ± 12.1 |
| 138-319 | 156-223 | 321-384 | 204-260 | 128-202 | 126-175 | |
| P = 0.042 | P = 0.004 | P = 0.674 | P = 0.060 | P = 0.024 | P = 0.011 | |
| Status PD-L1 expression | ||||||
| Positive | 341 ± 72.3 | 204 ± 52.7 | 430 ± 27.7 | 200 ± 8.19 | 231 ± 54.3 | 135 ± 29.5 |
| 66.3-488 | 58.4-350 | 341-518 | 174-226 | 58.4-404 | 41.3-229 | |
| Negative | 49.0 ± 13.1 | 116 ± 20.7 | 261 ± 41.4 | 279 ± 37.9 | 96.3 ± 28.0 | 131 ± 28.2 |
| 20.1-77.9 | 72.0-159 | 169-352 | 195-362 | 34.6-157 | 69.5-193 | |
| P = 0.010 | P = 0.075 | P = 0.252 | P = 0.715 | P = 0.032 | P = 0.260 | |
TC: Tumor clusters; TS: Peritumor stroma; TIME: Tumor immune microenvironment; PD-L1: Programmed death-ligand 1; ID: Immune desert; IE: Immune-excluded.
Compared with the common concept that TAMs correspond to M2 macrophages, in GCs, we found that M1 macrophages prevailed over the M2 type. Significant differences were not observed in CD68+ macrophage infiltration in the peritumour stroma with regard to the histological subtypes of GC (P = 0.471). However, the number of CD68+ cells within the tumour cluster was higher in intestinal and diffuse GC than in mucinous GC (P < 0.001). Moreover, M1 macrophages were more numerous than CD8+ cells in both intra- and peritumour areas of most observed tumours, and they were even more conspicuous in diffuse and mucinous GC. In contrast, CD163+ cells were less frequent and demonstrated specific associations with distinct histological types. M2 macrophages were few in mucinous and diffuse tumours; however, CD163+ cells were much more numerous in intestinal-type GCs (P = 0.032). Notably, the M2 macrophage number was comparable to the CD8+ cell count in all GC types. Notably, GCs with a poor prognosis (mucinous and diffuse type) demonstrated a proinflammatory macrophage phenotype in which the number of CD68+ cells was several times higher than that of M2-type macrophages (P < 0.001).
Therefore, CD68+ macrophages were the predominant type of immune cells in GCs of all histological types. The density of TILs demonstrated a relationship with the distinct immune contexture. Intestinal-type GCs were rich in CD68+, CD8+ and CD163+ cells. Diffuse GCs were heavily infiltrated by CD68+ cells but low in CD8+-and CD163+ cells. In mucinous GCs, CD68+ cells were the most abundant and prevailed over CD8+ and CD163+ cells, although the immune cell number was significantly lower within the TC than in the peritumor stroma.
Immunophenotyping GC of different histological types
The histological types of GCs correlated with distinct immunophenotypes (P < 0.001) assessed in line with the immune cycle concept (Table 3). There were 10 cases of ID (20%), 13 tumors with IE (26%) and 27 cases with Inf-TIME (54%). Notably, the majority of intestinal-type GCs had an inflammatory TIME (25 of 31 patients; 80.7%). The rest of the patients with intestinal-type GCs showed ID (1 of 31; 3.2%) or IE TIME (5 of 31; 16.1%). More than half of diffuse-type GCs had ID TIME (8 of 15 patients; 53.3%). The rest were IE (5 of 15; 33.3%) or had an inflammatory TIME (2 of 15; 13.3%). Most mucinous GCs (3 of 4; 75%) were IE TIME, and one case represented the ID immunophenotype. We did not find any statistically significant relationship between TIME and tumor grade (P = 0.523) or stage (P = 0.756). There were two intestinal-type GCs with MMR deficiency. Both cases were of inflamed-TIME and did not differ significantly in terms of immune cell patterns from other intestinal-type GCs. Therefore, GCs of different histological types illustrated the prevalence of distinct immunophenotypes. Inflamed TIME was more common for intestinal GCs, IE TIME prevailed in mucinous adenocarcinomas, and ID TIME was more typical for diffuse-type GC.
Table 3.
Relationship between immunophenotype and histological pattern of gastric carcinoma
|
Characteristics
|
ID TIME
|
IE TIME
|
Inf TIME
|
Total number
|
| Histological types of GC, n (%) | ||||
| Intestinal | 1 (3.2) | 5 (16.1) | 25 (80.7) | 31 (62) |
| Diffuse | 8 (53.3) | 5 (33.3) | 2 (13.3) | 15 (30) |
| Mucinous | 1 (25) | 3 (75) | 0 | 4 (8) |
| P < 0.001 | ||||
| PD-L1 expression status, n (%) | ||||
| PD-L1 positive GCs | 1 (5.6) | 0 | 8 (94.4) | 9 (18) |
| PD-L1 negative GCs | 9 (21.9) | 13 (31.8) | 19 (46.3) | 41 (82) |
| Total number | 10 | 13 | 27 | 50 |
| P < 0.001 | ||||
TIME: Tumor immune microenvironment; PD-L1: Programmed death-ligand 1; GC: Gastric carcinoma; ID: Immune desert; IE: Immune-excluded.
Tumor-infiltrating macrophages in GCs with different TIMEs
Naturally, ID-type GCs demonstrated a low number of CD8+ cells with a similar rate of M2 macrophages. At the same time, the count of CD68+ cells was several times higher than the lymphocyte density (P < 0.001). In contrast, GCs with the IE immunophenotype had a centrifugal pattern of immune cell distribution with a twofold lower CD68+ cell count inside tumour clusters compared to peritumour areas (P < 0.05). Although CD68+ macrophages were the most abundant immune cells in both the intratumour and peritumour compartments of IE-GCs, their number was twice as low as that in carcinomas with ID and inflammatory TIME (P < 0.001). Notably, GCs of the inflamed TIME demonstrated the highest density of CD8+ lymphocytes and CD163+ macrophages in both the tumour cluster and peritumour stroma compared to GCs with other immunophenotypes (Table 2).
We did not find any tight association between the number of CD68+ cells and the T lymphocyte infiltration pattern. GCs of ID and inflamed TIME demonstrated the highest number of intratumor CD68+ cells with a smaller amount in peritumor stroma (Figure 2). In contrast, the CD163+ cell number was more tightly associated with GC morphology and TIL patterns. M2 macrophages were the most numerous in intestinal-type GC with an inflamed TIME (Table 2). In addition, their amount was associated with PD-L1 expression.
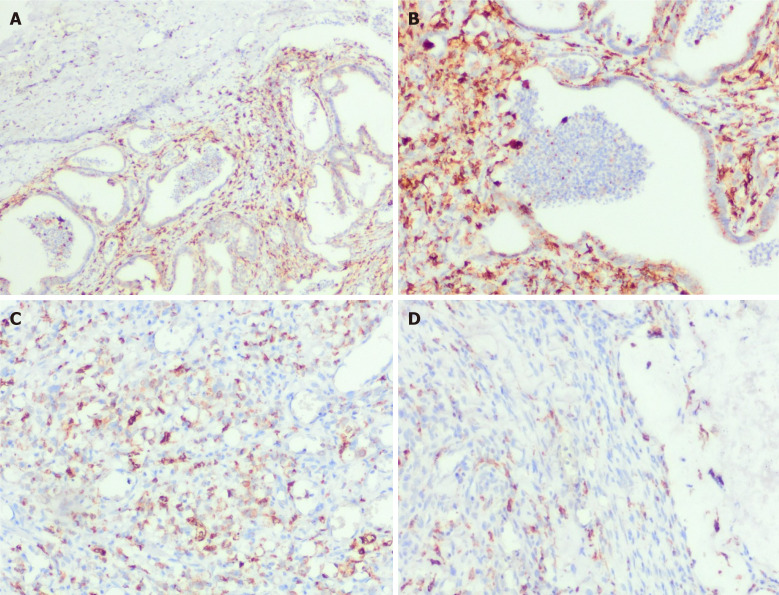
Figure 2.
Density and spatial distribution of macrophages in gastric carcinoma of different histological types. Immunohistochemistry for CD68. A and B: Intestinal type gastric carcinoma (GC) of inflamed tumor immune microenvironment (TIME) (A) with high count of CD68+ cells forming dense meshwork (B) inside the tumor. (A: × 100, B: × 200); C: High number of intratumor CD68+ cells of diffuse type GC of ID TIME, × 200; D: Prevalence of CD68+ cells in peritumor stroma of mucinous GC of IE TIME, × 200.
PD-L1 expression in GCs with different immunophenotypes
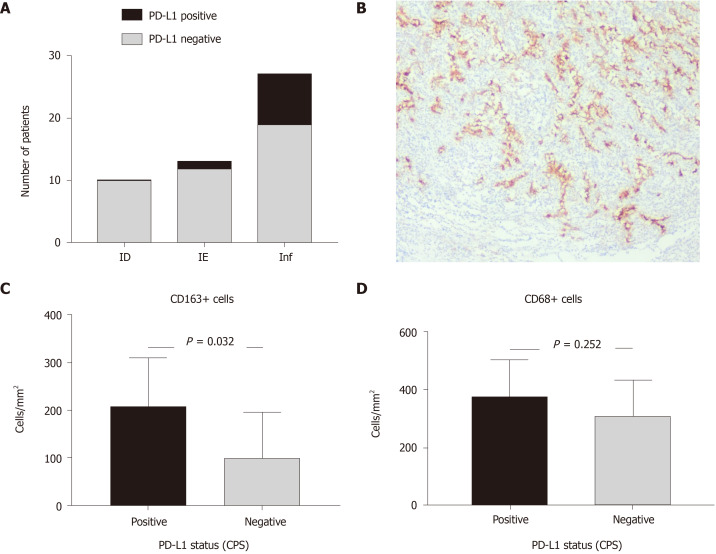
To clarify the potential mechanisms of GC immune evasion and its relationship with the TIME, we evaluated PD-L1 expression. There were 9 PD-L1-positive GCs of 50 observed cases (18%), and 8 of them were intestinal-type GCs (8 of 9). We did not find a significant relationship between PD-L1 expression and variables such as age, sex, pathological stage and GC histological type and grade. However, PD-L1 expression was related to TIME type (P = 0.05). Most cases of PD-L1 positivity were associated with an inflamed TIME (Table 3 and Figure 3). For tumour-infiltrating immune cells, PD-L1 positivity correlated with high CD8+ cell counts in TCs (P = 0.005) and CD163+ macrophages (P = 0.032) but not CD68+ density. Additionally, there were two intestinal-type GCs with Inf-TIME demonstrating MMR deficiency, and one of those tumours was PD-L1 positive. Thus, PD-L1 expression was found mostly in GC with an inflamed TIME, intestinal histology and a high density of CD8+ and CD163+ cells. Preexisting chronic inflammation and M2 macrophage polarization may be related to the activation of immune escape mechanisms in GC with an inflamed TIME.
Figure 3.
Relationship between programmed death-ligand 1 expression, gastric carcinoma immunophenotype and number of M1 and M2 macrophages. A: Frequency of programmed death-ligand 1 (PD-L1) expression in gastric carcinoma (GC) of different tumor immune microenvironment (TIME); B: PD-L1 expression in tumor cells of Inflamed TIME GC, immunohistochemistry for PD-L1, × 50; C and D: number of M1 (C) and M2 (D)-macrophages in GC regarding PD-L1 expression. PD-L1: Programmed death-ligand 1; CPS: Combined positive score.
DISCUSSION
The results of the study showed a close relationship between GC histological differentiation and immunophenotype. GCs of distinct histological types differed in terms of the density and spatial patterning of lymphocytes and macrophages, thus reflecting the divergence of the TIME among GCs. Several years ago, three broad classes of the TIME (namely, ID, IE, and Inflamed), representing alterations in consequent steps of the tumour immune cycle, were proposed[17]. According to this concept, ID TIME is characterized by a low number of CD8+ cells caused by poor tumour immunogenicity. This type was typical for diffuse carcinoma and could be due to low mutational load or a lack of antigen release. Alternatively, dendritic cell insufficiency or disrupted costimulatory interactions can diminish antigen presentation and T cell activation[12]. The immune-excluded phenotype, which is more common for mucinous GCs, was associated with the abundance of CD8+ cells in the peritumour stroma; however, T-cytotoxic cells were not able to reach tumour cells. Such a failure can be due to altered mechanisms of T lymphocyte recruitment inside the tumour, which can be caused by the lack of chemokines or low levels of chemokine receptors[18,19]. In contrast, the inflammatory TIME predominated in intestinal-type GCs and was rich in lymphocytes infiltrating tumour clusters. Although these tumours demonstrated an "immune hot" phenotype, it seems that the functioning of TILs was improper[20,21]. In addition, the elevated expression of immunosuppressive molecules, such as VEGF and TGF-β, can make antitumour immunity ineffective[14,21]. Therefore, cancer cells of various differentiation types use specific strategies of immune escape and can block either the early steps of the antitumour response or the final mechanisms of cancer cell killing[22,23].
Why mucinous and diffuse GCs exhibit more invasive behavior remains unclear. It might be related to low cell differentiation, which is associated with a higher rate of proliferation, cell motility and cancer dispersion[24]. However, there is an alternative explanation based on immune-mediated mechanisms. Papadopoulos et al[25] stated that mucin interferes with the inflammatory response and immunological recognition of tumor cells[25]. Our study supports this hypothesis, demonstrating the disruption of either antigen-presenting cell functioning or CD8+ cell trafficking into the tumor in diffuse and mucinous GCs. As there are no molecular targets identified for diffuse and mucinous GCs, it seems that elucidation of their immunosuppressive potential is a promising issue for further investigation.
In contrast, intestinal-type GC demonstrated a predominantly inflamed TIME that looks natural because it arose from preexisting immunological impairments[26,27]. The chronic inflammatory response is believed to be required for a sequence of epithelial transformations called the Correa cascade, which includes multifocal atrophic gastritis, intestinal metaplasia, dysplasia, and cancer[28-31]. Despite the high density of TILs, the inflamed TIME is often associated with the arrest of antitumour immunity[17,32], which can be provoked by various factors, such as the lack of a major histocompatibility complex in tumour cells, the impact of immunosuppressive immune cells (T-regulatory lymphocytes and myeloid-derived suppressor cells), or the expression of immune checkpoints[33]. Previously, an association between PD-L1 expression and MSI and EBV molecular subtypes was noticed[18]. In this study, we found a link between PD-L1 expression and inflamed TIME predominating in intestinal-type GCs. It is also worth noting that most PD-L1-positive cases were related to the high density of M2 macrophages, which could reflect the role of M2 macrophages in immune escape mechanisms.
Finally, this study revealed the prevalence of CD68+ macrophages in the TIME of GCs. Macrophages are thought to be key players in the innate immune response that could modulate chronic inflammatory responses and carcinogenesis. After monocyte recruitment and transformation, macrophages adjust to particular environmental conditions and adopt either proinflammatory (M1) or anti-inflammatory (M2) phenotypes[34]. It is widely accepted that cancer-associated macrophages are represented by the M2 type, which contributes to the suppression of adaptive immunity, tissue remodelling, angiogenesis and tumour growth[33]. In contrast, in this study, we found that GCs were associated with the prevalence of M1 macrophages possessing proinflammatory features. CD68+ cells were the predominant cell type of the TIME and were much more numerous than lymphocytes, particularly in diffuse GCs. On the one hand, this finding demonstrates the role of innate vs adaptive immune cell balance in carcinomas of various aggressiveness levels. On the other hand, it conflicts with the concept of the high proinflammatory and antitumour activity of M1 macrophages because a high number of these macrophages can improve patient outcomes. However, in diffuse GC, the loss of cadherin expression in cancer cells combined with M1 macrophage activity (reactive oxygen species, causing additional DNA damage and mutagenesis, and matrix-degrading enzymes) could build an effective strategy against adverse biological cancer behaviour. This hypothesis motivates a greater understanding of the role of M1 macrophages in gastric cancer prognostication.
Our study is limited by its retrospective design and descriptive nature as well as the small number of cases for each tumour subtype, and these limitations preclude multivariate analyses of the immunologic variables and clinical outcomes. In addition, the cloning of antibodies used in the study to evaluate PD-L1 expression is not currently used in clinical practice to select gastric cancer patients for immunotherapy.
CONCLUSION
In this study, we found a relationship between the histological and immunological features of GC. Various histological types of GC demonstrate distinct immunophenotypes that could be related to different pathways defining tumor immunogenicity and mechanisms of immune evasion. Diffuse and mucinous GCs have low immunogenicity, which is associated with a lack of antitumor immunity activation or T cell recruitment but dense infiltration by CD68+ macrophages. In contrast, intestinal-type GCs exhibit a predominantly inflamed TIME associated with M2 macrophage polarization and PD-L1 expression.
ARTICLE HIGHLIGHTS
Research background
Various histological types of gastric carcinomas (GCs) differ in terms of their pathogenesis and preexisting background, both of which could impact the tumour immune microenvironment (TIME). However, the current understanding of the immune contexture of GC is far from complete.
Research motivation
The data can help to clarify the links between tumor histogenesis and immunogenicity, offering a better understanding of GC biology and the ability to provide more tailored patient management.
Research objectives
This retrospective study aimed to clarify the tumor-host immune interplay through histopathological features and the tumor immune cycle concept.
Research methods
Of 50 GC cases were examined (15 cases of diffuse GC, 31 patients with intestinal-type GC and 4 cases of mucinous GC). The immunophenotype of GC was assessed and classified as immune desert (ID), immune excluded (IE) or inflamed (Inf) according to CD8+ cell count and spatial pattern. In addition, CD68+ and CD163+ macrophages and programmed death-ligand 1 (PD-L1) expression were estimated.
Research results
GCs with different histological differentiation demonstrated distinct immune contexture. Most intestinal-type GCs had inflamed TIMEs rich in both CD8+ cells and macrophages. In contrast, more aggressive diffuse-type GC more often possessed ID characteristics with few CD8+ lymphocytes but abundant CD68+ macrophages, while mucinous GC had an IE-TIME with a prevalence of CD68+ macrophages and CD8+ lymphocytes in the peritumor stroma. PD-L1 expression prevailed mostly in intestinal-type Inf-GC, with numerous CD163+ cells observed.
Research conclusions
GCs of different histological patterns have specific mechanisms of immune escape. While intestinal-type GC was more often related to PD-L1 expression, diffuse and mucinous GCs possessing more aggressive behavior demonstrated low immunogenicity and a lack of tumor antigen recognition or immune cell recruitment into the tumor clusters.
Research perspectives
Assessment of tumor immune microenvironment in complex with the molecular landscape will provide a deeper understanding of the links between tumor histogenesis and immunogenicity for a better understanding of GC biology and more tailored patient management.
Footnotes
Institutional review board statement: The study protocol was consistent with the guidelines outlined in the Declaration of Helsinki and approved by the Ethics Committee of Medical Laboratory CSD. The only patients who provided written informed consent for participation in research before their surgery were included in the study.
Informed consent statement: Patients were not required to give informed consent to the study because the analysis used anonymous clinical data that were obtained after each patient agreed to treatment by written consent.
Conflict-of-interest statement: We have no financial relationships to disclose.
Manuscript source: Invited manuscript
Peer-review started: March 3, 2021
First decision: April 17, 2021
Article in press: August 9, 2021
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Ukraine
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Carneiro F S-Editor: Fan JR L-Editor: A P-Editor: Li JH
Contributor Information
Artem Mashukov, Department of Oncology, Odessa National Medical University, Odessa 65082, Ukraine.
Dmytro Shapochka, Department of Molecular Pathology and Genetics, Medical Laboratory CSD, Kyiv 03022, Ukraine.
Oleksii Seleznov, Department of Pathology, Medical Laboratory CSD, Kyiv 03022, Ukraine.
Nazarii Kobyliak, Department of Pathology, Medical Laboratory CSD, Kyiv 03022, Ukraine; Department of Endocrinology, Bogomolets National Medical University, Kyiv 01601, Ukraine. nazariikobyliak@gmail.com.
Tetyana Falalyeyeva, Biomedicine, Educational-Scientific Center, "Institute of Biology and Medicine" Taras Shevchenko National University of Kyiv, Kyiv 01601, Ukraine.
Stanislav Kirkilevsky, Department of Thoracic Tumors, National Cancer Institute, Kyiv 03022, Ukraine.
Roman Yarema, Department of Oncology and Medical Radiology, Danylo Halytsky Lviv National Medical University, Lviv 79010, Ukraine.
Oksana Sulaieva, Department of Pathology, Medical Laboratory CSD, Kyiv 03022, Ukraine.
Data sharing statement
No additional data are available.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7–34. doi: 10.3322/caac.21551. [DOI] [PubMed] [Google Scholar]
- 2.Ji X, Bu ZD, Yan Y, Li ZY, Wu AW, Zhang LH, Zhang J, Wu XJ, Zong XL, Li SX, Shan F, Jia ZY, Ji JF. The 8th edition of the American Joint Committee on Cancer tumor-node-metastasis staging system for gastric cancer is superior to the 7th edition: results from a Chinese mono-institutional study of 1663 patients. Gastric Cancer. 2018;21:643–652. doi: 10.1007/s10120-017-0779-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lauren P. The two histological main types of gastric carcinoma: Diffuse and so-called intestinal-type carcinoma. An attempt at a histo-clinical classification. Acta Pathol Microbiol Scand. 1965;64:31–49. doi: 10.1111/apm.1965.64.1.31. [DOI] [PubMed] [Google Scholar]
- 4.Chen YC, Fang WL, Wang RF, Liu CA, Yang MH, Lo SS, Wu CW, Li AF, Shyr YM, Huang KH. Clinicopathological Variation of Lauren Classification in Gastric Cancer. Pathol Oncol Res. 2016;22:197–202. doi: 10.1007/s12253-015-9996-6. [DOI] [PubMed] [Google Scholar]
- 5.Giraldo NA, Peske JD, Sautès-Fridman C, Fridman WH. Integrating histopathology, immune biomarkers, and molecular subgroups in solid cancer: the next step in precision oncology. Virchows Arch. 2019;474:463–474. doi: 10.1007/s00428-018-02517-1. [DOI] [PubMed] [Google Scholar]
- 6.Liu X, Meltzer SJ. Gastric Cancer in the Era of Precision Medicine. Cell Mol Gastroenterol Hepatol. 2017;3:348–358. doi: 10.1016/j.jcmgh.2017.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ahn S, Lee SJ, Kim Y, Kim A, Shin N, Choi KU, Lee CH, Huh GY, Kim KM, Setia N, Lauwers GY, Park DY. High-throughput Protein and mRNA Expression-based Classification of Gastric Cancers Can Identify Clinically Distinct Subtypes, Concordant With Recent Molecular Classifications. Am J Surg Pathol. 2017;41:106–115. doi: 10.1097/PAS.0000000000000756. [DOI] [PubMed] [Google Scholar]
- 8.Liu Y, Sethi NS, Hinoue T, Schneider BG, Cherniack AD, Sanchez-Vega F, Seoane JA, Farshidfar F, Bowlby R, Islam M, Kim J, Chatila W, Akbani R, Kanchi RS, Rabkin CS, Willis JE, Wang KK, McCall SJ, Mishra L, Ojesina AI, Bullman S, Pedamallu CS, Lazar AJ, Sakai R Cancer Genome Atlas Research Network. Thorsson V, Bass AJ, Laird PW. Comparative Molecular Analysis of Gastrointestinal Adenocarcinomas. Cancer Cell. 2018;33:721–735.e8. doi: 10.1016/j.ccell.2018.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang Q, Liu G, Hu C. Molecular Classification of Gastric Adenocarcinoma. Gastroenterology Res. 2019;12:275–282. doi: 10.14740/gr1187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chivu-Economescu M, Matei L, Necula LG, Dragu DL, Bleotu C, Diaconu CC. New therapeutic options opened by the molecular classification of gastric cancer. World J Gastroenterol. 2018;24:1942–1961. doi: 10.3748/wjg.v24.i18.1942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yang S, Liu T, Cheng Y, Bai Y, Liang G. Immune cell infiltration as a biomarker for the diagnosis and prognosis of digestive system cancer. Cancer Sci. 2019;110:3639–3649. doi: 10.1111/cas.14216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chung HW, Lim JB. Role of the tumor microenvironment in the pathogenesis of gastric carcinoma. World J Gastroenterol. 2014;20:1667–1680. doi: 10.3748/wjg.v20.i7.1667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tuccitto A, Shahaj E, Vergani E, Ferro S, Huber V, Rodolfo M, Castelli C, Rivoltini L, Vallacchi V. Immunosuppressive circuits in tumor microenvironment and their influence on cancer treatment efficacy. Virchows Arch. 2019;474:407–420. doi: 10.1007/s00428-018-2477-z. [DOI] [PubMed] [Google Scholar]
- 14.Moehler M, Göpfert K, Lenz HJ. Outlook: Immunotherapy in Gastrointestinal Carcinoma - Innovative Strategies. Oncol Res Treat. 2018;41:313–315. doi: 10.1159/000489047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Amin MB, Gress DM, Vega LRM, Edge SB, Greene FL . , Byrd DR, Brookland RK, Washington MK, Compton CC. AJCC Cancer Staging Manual. 8th ed. Springer, 2017. [DOI] [PubMed] [Google Scholar]
- 16.Savic Prince S, Bubendorf L. Predictive potential and need for standardization of PD-L1 immunohistochemistry. Virchows Arch. 2019;474:475–484. doi: 10.1007/s00428-018-2445-7. [DOI] [PubMed] [Google Scholar]
- 17.Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39:1–10. doi: 10.1016/j.immuni.2013.07.012. [DOI] [PubMed] [Google Scholar]
- 18.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 19.Horton BL, Fessenden TB, Spranger S. Tissue Site and the Cancer Immunity Cycle. Trends Cancer. 2019;5:593–603. doi: 10.1016/j.trecan.2019.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Binnewies M, Roberts EW, Kersten K, Chan V, Fearon DF, Merad M, Coussens LM, Gabrilovich DI, Ostrand-Rosenberg S, Hedrick CC, Vonderheide RH, Pittet MJ, Jain RK, Zou W, Howcroft TK, Woodhouse EC, Weinberg RA, Krummel MF. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat Med. 2018;24:541–550. doi: 10.1038/s41591-018-0014-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wang M, Li Z, Peng Y, Fang J, Fang T, Wu J, Zhou J. Identification of immune cells and mRNA associated with prognosis of gastric cancer. BMC Cancer. 2020;20:206. doi: 10.1186/s12885-020-6702-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Choi JS, Kim MA, Lee HE, Lee HS, Kim WH. Mucinous gastric carcinomas: clinicopathologic and molecular analyses. Cancer. 2009;115:3581–3590. doi: 10.1002/cncr.24422. [DOI] [PubMed] [Google Scholar]
- 23.Perrot-Applanat M, Vacher S, Pimpie C, Chemlali W, Derieux S, Pocard M, Bieche I. Differential gene expression in growth factors, epithelial mesenchymal transition and chemotaxis in the diffuse type compared with the intestinal type of gastric cancer. Oncol Lett. 2019;18:674–686. doi: 10.3892/ol.2019.10392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Perez RO, Bresciani BH, Bresciani C, Proscurshim I, Kiss D, Gama-Rodrigues J, Pereira DD, Rawet V, Cecconnello I, Habr-Gama A. Mucinous colorectal adenocarcinoma: influence of mucin expression (Muc1, 2 and 5) on clinico-pathological features and prognosis. Int J Colorectal Dis. 2008;23:757–765. doi: 10.1007/s00384-008-0486-0. [DOI] [PubMed] [Google Scholar]
- 25.Papadopoulos VN, Michalopoulos A, Netta S, Basdanis G, Paramythiotis D, Zatagias A, Berovalis P, Harlaftis N. Prognostic significance of mucinous component in colorectal carcinoma. Tech Coloproctol. 2004;8 Suppl 1:s123–s125. doi: 10.1007/s10151-004-0131-z. [DOI] [PubMed] [Google Scholar]
- 26.Shi J, Qu YP, Hou P. Pathogenetic mechanisms in gastric cancer. World J Gastroenterol. 2014;20:13804–13819. doi: 10.3748/wjg.v20.i38.13804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Piazuelo MB, Riechelmann RP, Wilson KT, Algood HMS. Resolution of Gastric Cancer-Promoting Inflammation: A Novel Strategy for Anti-cancer Therapy. Curr Top Microbiol Immunol. 2019;421:319–359. doi: 10.1007/978-3-030-15138-6_13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Abnet CC, Freedman ND, Kamangar F, Leitzmann MF, Hollenbeck AR, Schatzkin A. Non-steroidal anti-inflammatory drugs and risk of gastric and oesophageal adenocarcinomas: results from a cohort study and a meta-analysis. Br J Cancer. 2009;100:551–557. doi: 10.1038/sj.bjc.6604880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fox JG, Wang TC. Inflammation, atrophy, and gastric cancer. J Clin Invest. 2007;117:60–69. doi: 10.1172/JCI30111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Correa P, Haenszel W, Cuello C, Tannenbaum S, Archer M. A model for gastric cancer epidemiology. Lancet. 1975;2:58–60. doi: 10.1016/s0140-6736(75)90498-5. [DOI] [PubMed] [Google Scholar]
- 31.Lee JS, Won HS, Sun S, Hong JH, Ko YH. Prognostic role of tumor-infiltrating lymphocytes in gastric cancer: A systematic review and meta-analysis. Medicine (Baltimore) 2018;97:e11769. doi: 10.1097/MD.0000000000011769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD. Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol. 2002;3:991–998. doi: 10.1038/ni1102-991. [DOI] [PubMed] [Google Scholar]
- 33.Noy R, Pollard JW. Tumor-associated macrophages: from mechanisms to therapy. Immunity. 2014;41:49–61. doi: 10.1016/j.immuni.2014.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mills CD. M1 and M2 Macrophages: Oracles of Health and Disease. Crit Rev Immunol. 2012;32:463–488. doi: 10.1615/critrevimmunol.v32.i6.10. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No additional data are available.