Abstract
Not all persons recovering from severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection develop SARS-CoV-2–specific antibodies. We show that nonseroconversion is associated with younger age and higher reverse transcription PCR cycle threshold values and identify SARS-CoV-2 viral loads in the nasopharynx as a major correlate of the systemic antibody response.
Keywords: COVID-19, coronavirus disease, SARS-CoV-2, severe acute respiratory syndrome coronavirus 2, viruses, respiratory infections, zoonoses, nonseroconversion, RT-PCR, cycle threshold, nasopharyngeal viral loads, humoral response, serological nonresponders
Coronavirus disease (COVID-19) is typically diagnosed by reverse transcription PCR (RT-PCR) amplification of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) RNA from nasopharyngeal fluids (1). RT-PCR yields cycle threshold (Ct) values that are inversely correlated with viral loads (2) and thus provide an estimate of the number of SARS-CoV-2 RNA copies in the sample. Serologic assays complement COVID-19 diagnosis by documenting past infections. In most persons, binding and neutralizing antibodies develop within 1–3 weeks after onset of symptoms (3), and titers correlate with disease severity (4).
Initial serosurveys identified antibodies in nearly 100% of persons with RT-PCR–confirmed SARS-CoV-2 infection (5). However, more recent studies have shown that seroconversion rates are surprisingly variable (6–10). For example, a multicenter study from Israel reported that 5% of participants remained seronegative despite a positive test result on a nasal swab specimen (6). In contrast, a seroprevalence study from New York found that 20% of persons with a positive RT-PCR test result did not seroconvert (8). Another study from Germany reported that 85% of confirmed infected COVID-19 contacts failed to develop antibodies (9). To examine the reasons for these differences, we investigated the relationship between seroconversion and demographic, clinical, and laboratory data in a convenience sample of convalescent persons recruited at the University of Alabama at Birmingham (Birmingham, Alabama, USA) in 2020.
The Study
We studied 72 persons, all of whom had a previous positive RT-PCR test but were symptom-free for >3 weeks before blood was collected for testing (Table). Only 2 persons (3%) reported no symptoms, whereas 13 (18%) persons reported mild disease, 48 (67%) reported moderate disease, and 9 (12%) reported severe disease (Appendix Table 1).
Table. Demographic, clinical, and laboratory characteristics of serologic responders and nonresponders after SARS-CoV-2 infection*.
| Characteristic | SARS-CoV-2 antibody positive, n = 46 | SARS-CoV-2 antibody negative, n = 26 | p value† |
|---|---|---|---|
| Age, y, median (IQR) |
49 (37–63) |
35 (30–46) |
0.03 |
| Sex | 0.17 | ||
| M | 30 (65) | 10 (38) | |
| F |
16 (35) |
16 (62) |
|
| Race/ethnicity | 1.00 | ||
| White | 28 (61) | 20 (77) | |
| Black | 7 (15) | 3 (12) | |
| Asian | 7 (15) | 3 (12) | |
| Latinx |
4 (9) |
0 |
|
| RT-PCR of nasal swabs | |||
| DFOS, d, median (IQR) | 5 (3–11) | 5 (4–8) | 0.95 |
| Ct value, median (IQR)‡ |
24.5 (22–27) |
36 (34–77) |
<0.00001 |
| Symptoms§ | 45 (98) | 25 (96) | 0.21 |
| Severity 0 | 1 (2) | 1 (4) | |
| Severity 1 | 5 (11) | 8 (31) | |
| Severity 2 | 33 (72) | 15 (58) | |
| Severity 3 |
7 (15) |
2 (8) |
|
| Hospitalization |
6 (13) |
2 (8) |
1.00 |
| Serologic analyses | |||
| DFOS of T1, d, median (IQR) |
34 (26–46) |
33 (22–43) |
0.74 |
| Binding antibodies (positive¶) | |||
| Spike protein (IgG)# | 46 (100) | 0 | |
| Spike protein (IgA)# | 43 (93) | 0 | |
| RBD (IgG)** | 44 (96) | 0 | |
| RBD (IgM)** | 38 (83) | 0 | |
| Nucleocapsid protein (IgG)†† |
43 (93) |
0 |
|
| Neutralizing antibodies (positive¶) | 45 (98) | 0 |
*Participants were a convenience sample recruited at the University of Alabama at Birmingham (Birmingham, AL, USA) during March–May 2020. Values are no. (%) unless otherwise indicated. Ct, cycle threshold; DFOS, days following onset of symptoms; IQR, interquartile range; RBD, receptor binding domain; RT-PCR, reverse transcription PCR; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; T1, time of first serologic test. †Calculated using a likelihood ratio test for a logistic regression predicting seropositivity for the category indicated after Bonferroni correction for multiple comparisons, except for RT-PCR and serologic DFOS, for which p-values were calculated using a Welch’s 2-sample t-test. ‡Ct values were only available for a subset of seropositive (n = 34) and seronegative (n = 25) persons Appendix Table 1. §Symptom severity was self-reported, with 0 indicating no symptoms, 1 indicating mild symptoms with little impact on daily activities, 2 indicating moderate symptoms with noticeable impact on daily activities, and 3 indicating severe symptoms with a substantial reduction in quality of life (Appendix Table 1). ¶Above assay detection limits (Appendix Table 2 details midpoint and endpoint titers). #ELISA detection of IgG and IgA binding antibodies to a prefusion stabilized Wuhan-Hu-1 spike protein. **ELISA detection of IgM and IgG binding antibodies to RBD of the Wuhan-Hu-1 spike protein. ††Detection of IgG binding antibodies to the nucleocapsid protein by the Abbott Architect assay.
We tested plasma samples (n = 144) collected at enrollment and follow-up visits for antibodies to the spike protein by using a validated ELISA (Appendix). Only 46 of the 72 participants had detectable IgG responses, IgA responses, or both (Table); reciprocal endpoint titers ranged from 182 to >312,500 (Appendix Table 2). Analysis of the same samples for receptor-binding domain (RBD) and nucleocapsid (N) antibodies yielded very similar results (Appendix Figure 1). All persons with spike protein antibodies also had detectable RBD (IgG, IgM, or both) or N (IgG) protein responses, except for 1 participant whose spike protein endpoint titers were very low (Appendix Table 2). In contrast, 26 participants remained seronegative, despite the testing of up to 3 samples per person for IgA, IgM, and IgG against multiple antigens as well as neutralizing antibodies. Thus, 36% of our cohort represented serologic nonresponders.
To investigate potential reasons for the lack of seroconversion, we examined available demographic, clinical, and laboratory data. Comparing race/ethnicity, sex, and symptom severity, we failed to find a significant association with serostatus (Table), although we did observe a trend for increasing antibody positivity with increasing symptom severity (Appendix Figure 2). We also found no significant differences in seroconversion between patients reporting or not reporting various symptoms, including symptoms characteristic of COVID-19 (Appendix Figure 3). However, seronegative persons were on average 10 (95% CI 3–17) years younger than seropositive persons (Figure 1, panel A) and exhibited RT-PCR Ct values that were 11 (95% CI 8–14) cycles higher (Figure 1, panel B). Moreover, logistic regression showed a precipitous decline in the probability of seroconversion at higher Ct values (Figure 2). For example, a Ct of 35 predicted only a 15% (95% CI 5%–37%) probability of seroconversion, which decreased further with increasing Ct values. Thus, low nasopharyngeal viral loads seem insufficient to elicit a systemic antibody response.
Figure 1.
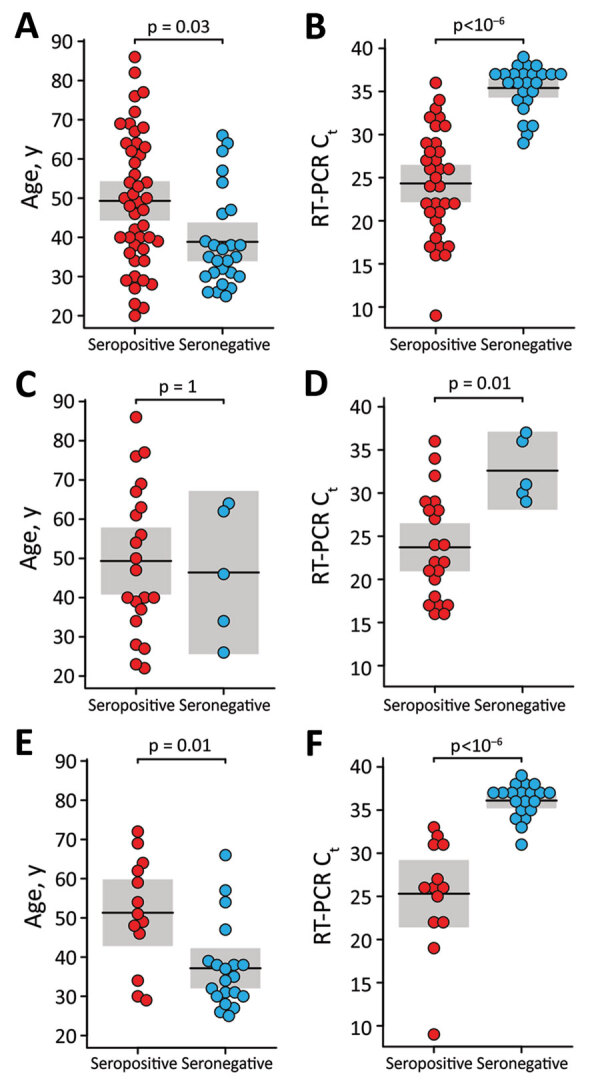
Relationship of age and nasopharyngeal viral loads with SARS-CoV-2 serostatus among convalescent persons after SARS-CoV-2 infection. Participants were a convenience sample of convalescent SARS-CoV-2–infected persons recruited at the University of Alabama at Birmingham, Birmingham, Alabama, USA, 2020. Age (panels A, C, and E) and RT-PCR Ct values (panels B, D, and F) are plotted for seropositive (red) and seronegative (blue) persons. Panels show comparisons of persons tested at all sites (panels A, B), the Assurance Scientific Laboratories site (panels B, C), and the University of Alabama at Birmingham Fungal Reference Laboratory and Children’s of Alabama Diagnostic Virology Laboratory sites (panels E, F). The mean (horizontal line) and corresponding 95% CI (shading) are shown; p-values indicate the results of a likelihood ratio test after Bonferroni correction for multiple comparisons. Ct, cycle threshold; RT-PCR, reverse transcription PCR; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
Figure 2.
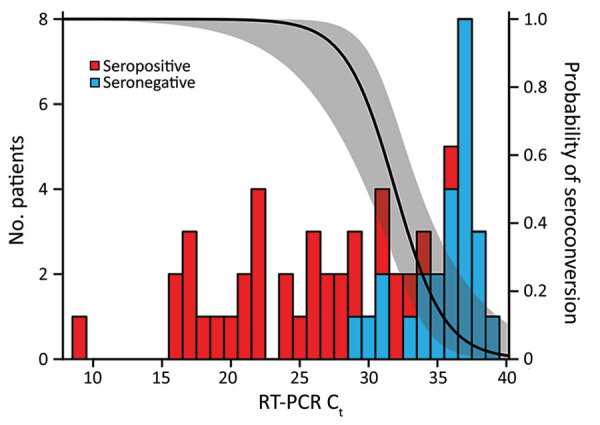
Decreasing probability of SARS-CoV-2 seroconversion with increasing RT-PCR Ct values among persons recovered from SARS-CoV-2 infection. Participants were a convenience sample of convalescent SARS-CoV-2–infected persons recruited at the University of Alabama at Birmingham, Birmingham, Alabama, USA, 2020. The number of serologic responders (red bars) and nonresponders (blue bars) is shown for varying RT-PCR Ct values. A logistic regression was used to estimate the probability of seroconversion for a given Ct (line) and its 95% CI (shaded). Ct, cycle threshold; RT-PCR, reverse transcription PCR; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
For control, we plotted Ct values of serologic responders and nonresponders against the times of RT-PCR and antibody testing relative to symptom onset (Appendix Figure 4). In both cases, the distributions of sampling times were similar for the 2 groups, thus excluding the possibility that seronegative persons had higher Ct values because they were tested too late or that they lacked antibodies because they were tested too early. We also examined remnants of purified RNA used for the initial diagnosis for the presence of SARS-CoV-2 sequences. By analyzing 12 available samples (Appendix Table 1), we were able to amplify full-length intact spike genes from 4 specimens, including 2 from seronegative persons with high Ct values (Appendix Figure 5).
Finally, we asked whether the relationship between seroconversion, age and Ct values was dependent on the diagnostic laboratory. We found that 2 sites with highly sensitive RT-PCR tests (University of Alabama at Birmingham Fungal Reference Laboratory and Children’s of Alabama Diagnostic Virology Laboratory in Birmingham) were 6 (95% CI 2–30) times more likely to identify serologic nonresponders than a third site with a less sensitive test (Assurance Scientific Laboratories in Birmingham) (Appendix Methods). However, this difference did not change the relationship between Ct values and seroconversion because seronegative persons had higher Ct values than seropositive persons regardless of the test site (Figure 1, panels D, F). In contrast, we observed little association between age and seroconversion at the Assurance Scientific Laboratories site (Figure 1, panel C), and the difference observed at the other sites was largely driven by young persons who also had high Ct values (Figure 1, panel E). Thus, nasopharyngeal viral loads represent a major correlate of the systemic antibody response, whereas age seems to have only a minor effect.
Conclusions
In summary, we show that patients with low SARS-CoV-2 viral loads in their respiratory tract are less likely to mount a systemic antibody response. Although we cannot formally exclude false-positive RT-PCR results in some participants, PCR contamination is highly unlikely as an explanation for our findings (Appendix). We also show that clinical illness does not guarantee seroconversion and that laboratories with highly sensitive RT-PCR assays are more likely to detect serologic nonresponders. These results provide an explanation for the puzzling variability of seroconversion in different cohorts.
The fact that a considerable fraction of RT-PCR positive persons fail to seroconvert has practical implications. Such persons remain undetected in seroprevalence studies, including in vaccine studies that assess protection from asymptomatic infection by measuring antibodies to antigens not included in the vaccine. Seroconverters and nonseroconverters will probably also respond differently to vaccination. Recent studies revealed that seropositive persons have a heightened antibody response after the first, but not the second, dose of an mRNA vaccine, suggesting that a single dose is sufficient (11–13; Samanovic et al., unpub. data, https://doi.org/10.1101/2021.02.07.21251311). Serologic nonresponders might not exhibit a similarly heightened anamnestic response, but resemble SARS-CoV-2 naive persons, as was observed for 1 previously infected vaccinee who never seroconverted (14). Finally, RT-PCR positive persons who experienced COVID-19 symptoms might be less inclined to seek vaccination, believing they are protected, but our results caution against this assumption.
Additional information about predictors of nonseroconversion after SARS-CoV-2 infection.
Acknowledgments
This work was supported by grants from the National Institutes of Health (R01 AI050529, R37 AI150590, P30 AI045008, R01 AI110553, R01 AI36082, P01 AI110657, U19 AI082630). A.N.S. is supported by a training grant (T32 GM07170).
Biography
Dr. Liu is a virologist at the University of Pennsylvania. His primary research interests include the evolutionary history and biology of zoonotic pathogens.
Footnotes
Suggested citation for this article: Liu W, Russell RM, Bibollet-Ruche F, Skelly AN, Sherrill-Mix S, Freeman DA, et al. Predictors of nonseroconversion after SARS-CoV-2 infection. Emerg Infect Dis. 2021 Sep [date cited]. https://doi.org/10.3201/eid2709.211042
These first authors contributed equally to this article.
Current affiliation: MBMicrobio Labs, Birmingham, Alabama, USA.
References
- 1.Kevadiya BD, Machhi J, Herskovitz J, Oleynikov MD, Blomberg WR, Bajwa N, et al. Diagnostics for SARS-CoV-2 infections. Nat Mater. 2021;20:593–605. 10.1038/s41563-020-00906-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pinninti SG, Pati S, Poole C, Latting M, Seleme MC, Yarbrough A, et al. Virological characteristics of hospitalized children with SARS-CoV-2 infection. Pediatrics. 2021;147:e2020037812; Epub ahead of print. 10.1542/peds.2020-037812 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sette A, Crotty S. Adaptive immunity to SARS-CoV-2 and COVID-19. Cell. 2021;184:861–80. 10.1016/j.cell.2021.01.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gaebler C, Wang Z, Lorenzi JCC, Muecksch F, Finkin S, Tokuyama M, et al. Evolution of antibody immunity to SARS-CoV-2. Nature. 2021;591:639–44. 10.1038/s41586-021-03207-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fafi-Kremer S, Bruel T, Madec Y, Grant R, Tondeur L, Grzelak L, et al. Serologic responses to SARS-CoV-2 infection among hospital staff with mild disease in eastern France. EBioMedicine. 2020;59:102915. 10.1016/j.ebiom.2020.102915 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Oved K, Olmer L, Shemer-Avni Y, Wolf T, Supino-Rosin L, Prajgrod G, et al. Multi-center nationwide comparison of seven serology assays reveals a SARS-CoV-2 non-responding seronegative subpopulation. EClinicalMedicine. 2020;29:100651. 10.1016/j.eclinm.2020.100651 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Masiá M, Telenti G, Fernández M, García JA, Agulló V, Padilla S, et al. SARS-CoV-2 seroconversion and viral clearance in patients hospitalized with COVID-19: viral load predicts antibody response. Open Forum Infect Dis. 2021;8:b005; Epub ahead of print. 10.1093/ofid/ofab005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pathela P, Crawley A, Weiss D, Maldin B, Cornell J, Purdin J, et al. Seroprevalence of SARS-CoV-2 following the largest initial epidemic wave in the United States: findings from New York City, May 13–July 21, 2020. J Infect Dis. 2021 Apr 9 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wellinghausen N, Plonné D, Voss M, Ivanova R, Frodl R, Deininger S. SARS-CoV-2-IgG response is different in COVID-19 outpatients and asymptomatic contact persons. J Clin Virol. 2020;130:104542. 10.1016/j.jcv.2020.104542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Thiruvengadam R, Chattopadhyay S, Mehdi F, Desiraju BK, Chaudhuri S, Singh S, et al. ; DBT India Consortium for COVID 19 Research. Longitudinal serology of SARS-CoV-2-infected individuals in India: a prospective cohort study. Am J Trop Med Hyg. 2021;tpmd210164; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Krammer F, Srivastava K, Alshammary H, Amoako AA, Awawda MH, Beach KF, et al. Antibody responses in seropositive persons after a single dose of SARS-CoV-2 mRNA vaccine. N Engl J Med. 2021;384:1372–4. 10.1056/NEJMc2101667 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stamatatos L, Czartoski J, Wan YH, Homad LJ, Rubin V, Glantz H, et al. mRNA vaccination boosts cross-variant neutralizing antibodies elicited by SARS-CoV-2 infection. Science. 2021;eabg9175; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Saadat S, Rikhtegaran Tehrani Z, Logue J, Newman M, Frieman MB, Harris AD, et al. Binding and neutralization antibody titers after a single vaccine dose in health care workers previously infected with SARS-CoV-2. JAMA. 2021;325:1467–9. 10.1001/jama.2021.3341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Reynolds CJ, Pade C, Gibbons JM, Butler DK, Otter AD, Menacho K, et al. ; UK COVIDsortium Immune Correlates Network. UK COVIDsortium Investigators. Prior SARS-CoV-2 infection rescues B and T cell responses to variants after first vaccine dose. Science. 2021;372:1418–23; [Epub ahead of print]. 10.1126/science.abh1282 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional information about predictors of nonseroconversion after SARS-CoV-2 infection.


