Abstract
Background
Patients with breast cancer who overexpress the human epidermal growth factor receptor 2 (HER2) and subsequently develop brain metastasis (BM) typically experience poor quality of life and low survival. We conducted a comprehensive literature review to identify prognostic factors for BM and predictors of survival after developing BM, and the effects of therapies with different mechanisms of action among patients with HER2+ breast cancer (BC).
Methods
A prespecified search strategy was used to identify research studies investigating BM in patients with HER2+ BC published in English during January 1, 2009–to June 25, 2021. Articles were screened using a two-phase process, and data from selected articles were extracted.
Results
We identified 25 published articles including 4097 patients with HER2+ BC and BM. Prognostic factors associated with shorter time to BM diagnosis after initial BC diagnosis included younger age, hormone receptor negative status, larger tumor size or higher tumor grade, and lack of treatment with anti-HER2 therapy. Factors predictive of longer survival after BM included having fewer brain lesions (< 3 or a single lesion) and receipt of any treatment after BM, including radiosurgery, neurosurgery and/or systemic therapy. Patients receiving combination trastuzumab and lapatinib therapy or trastuzumab and pertuzumab therapy had the longest median survival compared with other therapies assessed in this review.
Conclusions
More research is needed to better understand risk factors for BM and survival after BM in the context of HER2+ BC, as well as the assessment of new anti-HER2 therapy regimens that may provide additional therapeutic options for BM in these patients.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12885-021-08708-5.
Keywords: HER2-positive, HER2 + , Brain metastasis, Prognostic factors, Survival, Anti-HER2 therapy
Background
Approximately 15 to 20% of patients with breast cancer (BC) have tumors with elevated levels of human epidermal growth factor receptor 2 (HER2), which are associated with an aggressive clinical phenotype and poor prognosis [1, 2]. Up to 50% of patients with HER2-positive (HER2+) metastatic BC will develop brain metastasis (BM) during the course of the disease, often leading to worse morbidity and shorter survival [3]. Current treatment strategies for BM in patients with HER2+ BC incorporate local therapies and systemic therapies. Local therapies include surgery, whole-brain radiotherapy (WBRT), and stereotactic radiosurgery [4–6]. Systemic therapies include chemotherapy (e.g., docetaxel, capecitabine) and anti-HER2 therapies, which can encompass monoclonal antibodies such as trastuzumab (approved in the U.S. in 1998 [7]) and pertuzumab (2012 [8]), antibody-drug conjugates such as trastuzumab emtansine (2013 [9]), and small molecule tyrosine kinase inhibitors (TKIs) such as lapatinib (2007 [10]) and neratinib (2017 [11]) [4–6]. Recent evidence suggests that lapatinib and neratinib can penetrate the blood-brain barrier (BBB), and therefore these drugs and similar HER2-targeting TKIs may be promising therapeutic options for patients [12, 13].
We conducted a literature review to assess the epidemiology of patients with HER2+ metastatic or advanced BC with BM by describing prognostic factors for developing BM and factors predictive of survival among patients with BM. Additionally, differences in survival and time to progression by HER2-targeting treatment drug classes were explored based on the drug mechanism of action.
Methods
Search design
Electronic searches were conducted in PubMed and Embase. A predefined search strategy (Online Resource 1 and Online Resource 2) was used to identify research studies investigating BM in patients with HER2+ BC. The search was restricted to studies published in English. The original search was restricted to studies published during the period of January 1, 2009 to July 30, 2019. However, the protocol was amended to expand the search to June 25, 2021 to capture the most recent published literature. Titles and abstracts identified from the electronic databases were exported to an Excel (Microsoft Corporation; Redmond, Washington) file for screening. Systematic literature reviews and meta-analyses relevant to the study objectives were not themselves included within the scope of this literature review, but the bibliographies were reviewed to identify potential additional publications.
Screening and extraction
Articles were screened in a two-level process. In Level 1 screening, one researcher reviewed the titles and abstracts of the identified articles according to the literature review inclusion and exclusion criteria (Table 1) and selected articles for further review. In Level 2 screening, the full text of articles selected at Level 1 were reviewed by one researcher using the same set of inclusion and exclusion criteria. If there was any uncertainty about the inclusion of articles, the Level 2 reviewer discussed the article with a second researcher to confirm, by consensus, whether the article met the study inclusion and exclusion criteria. Data were extracted according to prespecified data fields using the full-text articles, including study population, country, observation period, sample size, and select sample characteristics. A checklist was used to assess the quality of the studies.
Table 1.
Inclusion and Exclusion Criteria for Level 1 (Titles and Abstracts) and Level 2 (Full-Text) Screening
| Criterion | Included | Excluded |
|---|---|---|
| Study design |
▪ Randomized controlled trials ▪ Single-arm studies ▪ Observational research studies (e.g., prospective cohort study, retrospective database study, cohort study, case-control study) ▪ Literature reviews and meta-analysesa ▪ Natural history studies ▪ Incidence and prevalence studies ▪ Prognostic factor studies |
▪ Consensus reports ▪ Preclinical studies ▪ Nonsystematic reviews ▪ Case reports ▪ Case studies/series ▪ Editorials ▪ Commentaries ▪ Letters ▪ Guideline or position statements ▪ Economic analyses ▪ Animal or other nonhuman (e.g., bench) studies ▪ Study of < 25 patients |
| Population | ▪ Patients with diagnosis of metastatic, recurrent, advanced, incurable, or unresectable HER2+ breast cancer (stages 3–4) with BM, either at the time of breast cancer diagnosis or after breast cancer diagnosis |
▪ Aged under 18 y ▪ Only patients with HER2− or stage 1 or 2 breast cancer ▪ Only patients with HER2+ breast cancer without BM |
| Treatment |
▪ Evaluation of chemotherapy by mechanism of action ▪ Nonpharmacological studies |
▪ Specific regimens of chemotherapy (not mechanism of action) ▪ Patients receiving surgical or radiation intervention in place of chemotherapy ▪ Patients receiving CDK4/6 inhibitors |
| Evaluation |
▪ Incidence or prevalence ▪ Prognostic and/or predictive factors ▪ Treatment outcomes (safety or effectiveness) |
▪ PK/PD of treatments |
BM brain metastasis; CDK4/6 cyclin-dependent kinase 4 and 6; HER2 human epidermal growth factor receptor 2; PD pharmacodynamics; PK pharmacokinetics
a Literature reviews and meta-analyses were not be included in the review but were used to identify primary studies not previously identified
Results
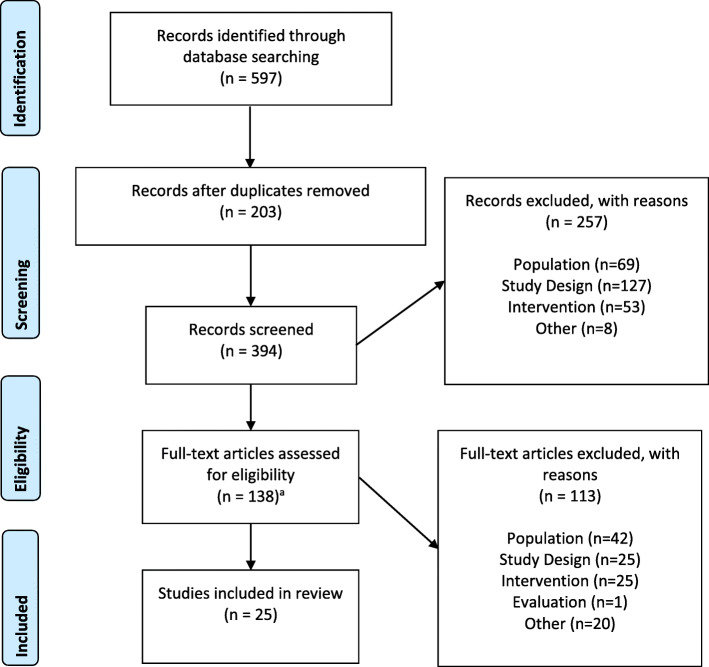
The number of studies included and excluded at each stage of screening was documented in a PRISMA diagram (Fig. 1) [14]. The original search strategy yielded 232 records for Level 1 screening and the expanded search added 162 records for Level 1 screening, for a total of 394 records retrieved. The bibliographies of 8 systematic reviews/meta-analyses were reviewed and yielded one additional study to be included for full-text screening for a total of 138 articles that were included in the Level 2 full-text screening. A total of 25articles met the inclusion criteria described in Table 1 and were selected for data extraction.
Fig. 1.
PRISMA Diagram. aThis includes one additional article identified from the review of the bibliography of a systematic review/meta-analysis after initial record screening, hence there appears to be one extra article in this diagram; PRISMA = Preferred Reporting Items for Systematic Reviews and Meta-Analyses
Study and patient population characteristics
Online Resource 3 presents the study and patient characteristics of the 25studies included in this review. Overall, the studies covered more than 10,000 patients, of which 4097 patients had HER2+ BC with BM. Across all studies, the observation/enrollment period started as early as 1988 [15] and ended as late as 2020 [16]. Geographically, studies were conducted in Europe (n = 11), North America (n = 7), the Asia-Pacific region (n = 7), and Turkey (n = 1).
Among studies reporting the median age of patients at the time of their initial BC diagnosis (n = 19 [76%]), the median age range was 43 to 55 years. The sex distribution was typically not reported, although three studies did report including 100% females [17–19]. Only two studies reported on race, which included 67% white patients [15] and 58% white patients [20].
Twelve studies (48%) included only patients with HER2+ status [18, 20–28], while the remaining studies (n = 13 [52%]) reported on patients in whom a subset were HER2+, ranging from 10.1% [29] to 58.2% [30]. Among the seven studies that enrolled patients with or without BM [21–23, 26, 29, 31, 32], the prevalence of BM in patients with HER2+ BC ranged from 7.8% [29] to 56.0% [23]. Twenty-one studies (84%) provided information on hormone receptor (HR) status among patients with HER2+ BC, reporting a range of 24.2% [33] to 71.0% [26] of patients with HR+ status (i.e., estrogen receptor positive and/or progesterone receptor positive), while four studies (16%) did not report on HR status [16, 30, 34, 35].
Prognostic factors for developing brain metastasis
Table 2 presents information on time to first BM diagnosis (TTBM) from initial HER2+ BC diagnosis and prognostic factors associated with a shorter TTBM. Across the 20 studies that reported information on TTBM, the shortest reported median TTBM was 10.8 months [31] and the longest was 76.2 months [26]. Among the 10 studies that reported on prognostic factors for BM diagnosis [15, 17, 18, 21–23, 26, 29–31], the most commonly assessed prognostic factors included age, HR status, receipt of anti-HER2 therapy, and tumor grade.
Table 2.
Prognostic Factors Associated with Developing Brain Metastasis Among Patients with HER2+ Breast Cancer
| Citation | Prognostic Factors for Shorter Time to BM | |||||||
|---|---|---|---|---|---|---|---|---|
| HER2+ Group | Sample Size, n | Median Time to BMa, mo | Age | HR Status | Anti-HER2 Therapy | Tumor Grade | Other | |
| Ahn et al., 2013 [17] | Without trastuzumab | 39 | 32.1 | NR | NR | No association | NR | ▪ NR |
| With trastuzumab | 47 | 35.4 | ||||||
| Anders et al., 2011 [15] | HR+ | 21 | 49.8 (95% CI, 10.2–54.5) | NR | HR- vs. HR+ (suggestive association) | NR | NR | ▪ NR |
| HR- | 18 | 19.8 (95% CI, 13.6–36.2) | ||||||
| Berghoff et al., 2012 [30] | All | 102 | 18 (95% CI, 14.5–21.5)b | NR | ER- vs. ER+ | No association | NR | ▪ NR |
| ER+ | NR | NR | NR | NA | NR | NR | ▪ Did not receive palliative endocrine therapy | |
| Braccini et al., 2013 [36] | All | 109 | 36 (range, 0–287) | NR | NR | NR | NR | ▪ NR |
| Brufsky et al., 2011 [31] | All | 377 | 10.8 | < 50 y vs. ≥50 y | HR- vs. HR+ | No trastuzumab vs. trastuzumab | NR | ▪ ≥2 vs. < 2 metastatic sites |
| Duchnowska et al., 2012 [21] | All | 142 | 13 (95% CI, 9–18) | No association | No association | NR | Tumor grade 3 vs. grade 1–2 |
▪ Higher H2T levels (≥50 RF/mm2)c ▪ Time to nonbrain progressiond ▪ HER-2 gene amplifications as defined by the HER-2/CEP17 ratio (no association) ▪ Menopausal status (no association) |
| Duchnowska et al., 2009 e [22] | All | 264 | 15 (range, 0–81)b | No association | No association | No association | NR | ▪ Time to distant relapse ≤2 y vs. > 2 y |
| Duchnowska et al., 2015 [23] | Cohort A (discovery) | 83f | 36 (range, 2–141) | NR | ER- vs. ER + e | No trastuzumab vs. trastuzumab | No association |
▪ Visceral site of first distant relapse ▪ 3-gene classifierg |
| Cohort B (validation) | 75 | 40 (range, 0.33–125) | NR | ER- vs. ER+ | No trastuzumab vs. trastuzumab | Grade high vs. low e | ▪ Visceral site of first distant relapse | |
| Gori et al., 2019 [24] | All | 154 | 39.1 (IQR, 20.3–62.4) | NR | NR | NR | NR | ▪ NR |
| Hayashi et al., 2015 [25] | All | 432 | 33.5 | NR | NR | NR | NR | ▪ NR |
| Heitz et al., 2009 e [29] | All | 245 | 30 | No association | No association | No association | No association |
▪ Pathological tumor size category 3/4 vs. category 1/2 ▪ TNM classification of metastatic (M) status at diagnosis is 1 vs. 0 |
| Jang et al., 2011 [34] | All | 137 | 31.6 (95% CI, 27.3–35.9) | NR | NR | NR | NR | ▪ NR |
| Kuba et al., 2014 [35] | All | 26 | 15.6 (range, 0–52.8) | NR | NR | NR | NR | ▪ NR |
| Maurer et al., 2018 [26] | All | 483 | 76.2 | ≤40 y vs. > 40 y | No association | No association | No association |
▪ No surgery vs. surgery for primary BC ▪ Larger tumor size ▪ Nodal involvement ▪ Received adjuvant endocrine treatment ▪ Received no anthracyclines + taxanes as (neo) adjuvant chemotherapy |
| Morikawa et al., 2018 [27] | All | 100 | 34.6 (range, 0–176) | NR | NR | NR | NR | ▪ NR |
| Mounsey et al., 2018 [20] | All | 123 | 34.6 (95% CI, 26.6–41.0) | NR | NR | NR | NR | ▪ NR |
| Sperduto et al., 2013 [37] | HR+ | 98 | 47.4 (IQR, 26.3–70.5) | NR | NR | NR | NR | ▪ NR |
| HR- | 119 | 35.8 (IQR, 13.4–69.2) | NR | NR | NR | NR | ▪ NR | |
| Witzel et al., 2018 [16] | All | 732 | 32.4 (95% CI, 29.6–36.1) | NR | NR | NR | NR | ▪ NR |
| Yap et al., 2012 [18] | All | 280 | 30.1 (95% CI, 25.0–32.7) | NR | NR | No anti-HER2 treatment vs. anti-HER2 treatment | NR | ▪ NR |
| Zhang et al., 2016 [28] | All | 60 | 12 (range, 1–94) | NR | NR | NR | NR | ▪ NR |
BC breast cancer; BM brain metastasis; CI confidence interval; ER estrogen receptor; H2T the quantitative HER2 level as measured by the HERmark® Breast Cancer Assay; HER-2/CEP17 HER-2/centromeric probe for chromosome 17 ratio > 2.0; HR hormone receptor; IQR interquartile range; NA not applicable; NR not reported; RF relative fluorescence; TNM TNM staging system (T tumor size and spread, N nodal involvement, M = metastatic status) developed by the American Joint Committee on Cancer
a From the time of breast cancer diagnosis
b From the time of diagnosis of metastatic disease
c H2T is the quantitative HER-2 level as measured by the HERmark® Breast Cancer Assay (i.e., The VeraTag™ proximity-based assay; Monogram Biosciences, Inc., South San Francisco, California). The assay enables precise quantitative measurements of total HER-2 expression in formalin-fixed, paraffin-embedded tissue specimens. Higher H2T levels modeled as a continuous variable or as a categorical variable were associated with a shorter time to BM
d Time from initiation of trastuzumab therapy to nonbrain progression. The direction of the effect was not specified in the article
e Based on univariable analyses only
f 83 of the 84 patient samples were analyzable
g 3-gene classifier (including hepatoma-derived growth factor [HDGF], RAD51 homolog [RAD51], and translocated promoter region [TPR]) as a predictive model representing a 13-gene profile, which was associated with early (≤ 36 months) vs. late (> 36 months) BM and included the 3 genes in the 3-gene classifier and the following 11 genes: cyclin-dependent kinase 4 (CDK4), cyclin C (CCNC), focal adhesion kinase (protein tyrosine kinase 2, PTK2), v-myc avian myelocytomatosis viral oncogene homolog (MYC), breast cancer 1 [BRCA1] associated RING domain 1 (BARD1), Fanconi anemia group G (FANCG), proliferating cell nuclear antigen (PCNA), papillary renal cell carcinoma-translocation associated (PRCC), cortactin (CTTN), and desmoplakin (DSP)
In three studies, age was not found to be associated with TTBM [21, 22, 29], while two studies reported an association between younger age at BC diagnosis (aged ≤40 or < 50 years in Maurer et al. [26] and Brufsky et al. [31], respectively) and shorter TTBM [26, 31]. Hormone receptor status was assessed as a prognostic factor for TTBM in eight studies; four studies reported no association between HR status and TTBM [21, 22, 26, 29], while four other studies reported that patients with HR- status had a shorter TTBM compared with those who were HR+ [15, 23, 30, 31]. Among the eight studies that assessed trastuzumab use and TTBM, five reported no association between receipt of trastuzumab and TTBM [17, 22, 26, 29, 30], while three reported that patients who received trastuzumab had a longer TTBM compared with those who did not [18, 23, 31]. Two studies reported an association between higher tumor grade and shorter TTBM [21, 23], while two studies reported no association with tumor grade but did report an association between larger tumor size and shorter TTBM [26, 29].
Other treatment-related factors and TTBM were also assessed. In one study, longer TTBM was reported among those receiving treatment with endocrine therapy versus no endocrine therapy in the palliative care setting [30]. Conversely, another study reported that patients receiving adjuvant endocrine treatment had a shorter TTBM [26]. Longer TTBM was reported among patients who had surgery for their BC and among those treated with (neo) adjuvant anthracyclines + taxanes [26].
Other reported prognostic factors for shorter TTBM included higher disease burden (i.e., ≥ 2 metastatic sites) [31], higher HER2 levels (measured by the HERmark® Breast Cancer Assay) [21], distant relapse in ≤2 years [22], first distant relapse at a visceral site [23], and nodal involvement [26]. One study reported on genetic factors of TTBM, but the reported associations in the discovery set were not observed in the validation set [23].
Predictors of survival after brain metastasis among patients with HER2+ breast cancer
Table 3 presents median time to death (i.e., survival) after BM diagnosis and predictive factors associated with shorter survival after BM diagnosis. Across the 23 studies that reported information on median survival after BM, the shortest reported median survival was 5.2 months [34] and the longest was 28 months [21].
Table 3.
Predictors of Survival Among Patients with HER2+ Breast Cancer and Brain Metastasis
| Citation | Predictors for Shorter Time to Death (i.e., Survival) After BM Diagnosis | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| HER2+ Group | Sample Size, n | Median Time to Death After BM Diagnosis, mo | Age | HR Status | No. of Brain Lesions | Anti-HER2 Therapy | Any Systemic Treatment | Other | |
| Ahn et al., 2013 [17] | Without trastuzumab | 39 | 19.1a | NR | NR | NR | No trastuzumab vs. trastuzumab | NR | NR |
| With trastuzumab | 47 | 26.9a | |||||||
| Anders et al., 2011 [15] | HR- | 18 | 14.3 (95% CI, 3.2–36.2) | No association | No association | NR | NR | NR | Race (no association) |
| HR+ | 21 | 15.2 (95% CI, 7.8–40.4) | |||||||
| Anwar et al., 2021 [32] | All | 39 | 13.93 (95% CI 10.53–20.67)b | NR | No association | NR | NR | NR |
▪ Number of prior lines of therapy (no association) ▪ Liver metastasis (no association) ▪ Lung metastasis (no association) ▪ Bone metastasis (no association) |
| Bergen et al., 2021 [19] | All | 252 |
Before 2000: 12 2000–2010: 11 After 2010: 22 |
Included in DS-GPA but not reported independently | Included in DS-GPA but not reported independently | NR |
▪ No HER2-targeted therapy vs. therapy with trastuzumab + pertuzumab, or trastuzumab alone, or lapatinib alone, or T-DM1 alone ▪ Other HER2-targeted therapy vs. trastuzumab + pertuzumab ▪ No HER2-targeted therapy vs. trastuzumab + lapatinib (no association) |
NR |
▪ DS-GPA ▪ Time period (year) of initial BC diagnosis (< 2000, 2000–2010, > 2010) (no association) |
| Berghoff et al., 2012 [30] | All | 102 | 7 (95% CI, 4.3–969) | NR | NR | NR | ▪ Local therapyc alone vs. trastuzumab-based therapy after local therapy | NR | NR |
| Braccini et al., 2013 [36] | All | 109 | 11.9 (95% CI, 8.7–15.5) | NR | No association | NR |
▪ No anti-HER2 therapy vs. anti-HER2 therapy ▪ Trastuzumab alone or lapatinib alone vs. trastuzumab + lapatinib (sequentially) |
NR | NR |
| Brufsky et al., 2011 [31] | All | 377 | 13.0 (range, 0.1–55.5)d | No association | No association | NR | ▪ No trastuzumab vs. trastuzumab | ▪ No chemotherapy vs. chemotherapy |
▪ No surgery vs. surgery ▪ Radiotherapy – no association ▪ ECOG PS ≥ 2 vs. 0 or 1 ▪ CNS disease at mBC diagnosis vs. no CNS disease at mBC diagnosis |
| Duchnowska et al., 2012 [21] | All | 142 | 28 (95% CI, 16–32) | NR | NR | NR | ▪ NR | NR | NR |
| Gori et al., 2019 [24] | All | 154 | 24.5 | ≥ 60 y vs. < 60 y at BM diagnosise | No association | > 3 vs. 1–3 BMse | ▪ Systemic therapy without HER2-targeted agents or no systemic therapy vs. HER2-targeted agents | NR |
▪ WBRT or no local treatment vs. surgery and/or SRS ▪ KPS ≤ 7 0 vs. > 70 ▪ Presence of neurologic symptoms |
| Hayashi et al., 2015 [25] | ER+ | 162 | 16.5 (95% CI, 11.9–21.1) | NR | No association | > 3 vs. ≤ 3 BMs |
▪ Neither trastuzumab nor lapatinib vs. at least one of these after BM diagnosis ▪ Either trastuzumab alone, lapatinib alone, or no HER2-targeting agent vs. trastuzumab and lapatinib after BM diagnosis |
NR | NR |
| ER- | 270 | 11.5 (95% CI, 9.1–13.8) | |||||||
| Heitz et al., 2009 f [29] | All | 245 | 11 | NR | NR | NR | ▪ NR | NR | ▪ NR |
| Jang et al., 2011 [34] | All | 137 | 5.2 (95% CI, 3.6–6.8) | NR | NR | NR | ▪ NR | NR | ▪ NR |
| Kaplan et al., 2012 [33] | ER−/PR- | 102 | 11.04 (95% CI, 6.18–15.90) | ≥ 46 y vs. < 46 y at BM diagnosise (suggestive association in multivariable analyses) | No association | > 3 vs. ≤ 3 BMse (suggestive association in multivariable analyses) |
▪ Trastuzumab- or lapatinib-based therapy alone vs. trastuzumab- and lapatinib-based therapy (sequential) ▪ Trastuzumab-based therapy alone vs. lapatinib-based therapy alone |
NR |
▪ KPS ≤ 70 vs. > 70 ▪ Tumor grade 3 vs. grade 1–2 ▪ ≥ 2 vs. < 2 metastatic sites outside the brain ▪ No neurosurgery vs. neurosurgery ▪ No radiosurgery vs. radiosurgery |
| Luminal Bf | 113 | 9.99 (95% CI, 4.99–14.98) | |||||||
| Kuba et al., 2014 [35] | All | 26 | 23 (95% CI, 14–31) | No association | NR | No association | ▪ NR | NR |
▪ PS ≥ 2 vs. 0/1 ▪ Undergoing surgery or SRS (no association) |
| Martin et al., 2017 [38] | HR+ | 136 | 21 (IQR: 6-not reached)g | NR | NR | NR | ▪ NR | NR | ▪ NR |
| HR- | 106 | 10 (IQR: 4–27)g | |||||||
| Maurer et al., 2018 [26] | All | 483 | 20.8 (IQR: 5.36-not reached) | NR | NR | NR | ▪ No association | NR | ▪ CNS symptomsh vs. no CNS symptoms at BM diagnosis |
| Morikawa et al., 2018 [27] | All | 100 | 19.4 (95% CI, 15.5–26.6) | No association | No association | Multiple lesions vs. single lesion |
▪ No anti-HER2 use vs. anti-HER2 use after BM diagnosis ▪ No anti-HER2 use vs. lapatinib use after BM diagnosis |
NR |
▪ KPS < 70 vs. ≥ 70 ▪ Neurologic symptoms vs. no neurologic symptoms ▪ Uncontrolled extracranial disease vs. controlled |
| Mounsey et al., 2018 [20] | All | 123 | 18.1 (95% CI, 14.9–24.6) | NR | NR | NR | ▪ No HER2-targeted therapy vs. HER2-targeted therapy after BM diagnosis | NR | ▪ NR |
| Niwinska et al., 2010 [39] | All | 109 | 9 (range, 0.6–3.4) | NR | NR | NR | ▪ No systemic therapy or chemotherapy without trastuzumab vs. chemotherapy with trastuzumab | WBRT alone vs. systemic therapyi after WBRT |
▪ KPS < 70 vs. ≥ 70 ▪ RPA RTOG Prognostic class III vs. class I/II ▪ Visceral metastasis vs. no visceral metastasis |
| Sperduto et al., 2013 [37] | HR+ | 98 | 22.9 (95% CI, 16.1–29.5) | NR | NR | NR | ▪ NR | NR | ▪ NR |
| HR- | 119 | 17.9 (95% CI, 13.4–22.9) | |||||||
| Witzel et al., 2018 [16] | All | 732 | 11.6 (95% CI, 10.0–13.4) | NR | NR | NR | ▪ NR | NR | ▪ NR |
| Yap et al., 2012 [18] | All | 280 | 10.9 (95% CI, 9.0–11.9) | Older age vs. younger age at BM | NR | Multiple lesions vs. single lesion |
▪ No anti-HER2 treatment vs. anti-HER2 treatment after BM diagnosis ▪ No anti-HER2 treatment or trastuzumab alone vs. lapatinib alone after BM diagnosis ▪ No anti-HER2 treatment vs. trastuzumab alone after BM diagnosis ▪ Anti-HER2 therapy before BM (no association with survival after BM) |
▪ No chemotherapy vs. receipt of chemotherapy after BM diagnosis ▪ No hormonal therapy vs. receipt of hormonal therapy after BM |
▪ NR |
| Zhang et al., 2016 [28] | All | 60 | 12 (range, 1–94) | < 50 y vs. ≥ 50 y at BM diagnosise | No association | Multiple lesions vs. single lesione |
▪ No anti-HER2 therapy after WBRT vs. anti-HER2 therapy after WBRT ▪ No systemic therapy, anti-HER2 therapy alone, or chemotherapy alone after WBRT vs. both anti-HER2 therapy and chemotherapy after WBRT |
[See “Anti-HER2 therapy” column] |
▪ Uncontrolled extracranial metastasis vs. controlled ▪ KPS < 70 vs. ≥ 70e ▪ Total dose radiotherapy (no association)e ▪ Time from BC diagnosis to BM diagnosis (no association)e |
BC breast cancer; BM brain metastasis; CI confidence interval; CNS central nervous system; DS-GPA diagnosis specific graded prognostic assessment
Score (includes BC subtype, age < 60 or > 60 years, Karnofsky performance status), ECOG Eastern Cooperative Oncology Group; ER estrogen receptor; HER2 human epidermal growth factor receptor 2; HR hormone receptor; IQR interquartile range; KPS Karnofsky performance score; mBC metastatic breast cancer; NR not reported; PR progesterone receptor; PS performance status; RPA RTOG recursive partitioning analysis of Radiation Therapy Oncology Group prognostic class; SRS stereotactic radiosurgery; T-DM1 trastuzumab emtansine; WBRT whole-brain radiotherapy
a Time from diagnosis of distant metastasis
b Represents median time to death after start of pyrotinib therapy
c Local therapy for BM including surgery and/or radiotherapy
d Overall survival after BM for all patients diagnosed with BM, including patients who presented with BM at the time of their mBC diagnosis (n = 75 [19.9%]; overall survival after diagnosis was 20.3 months [range, 1.0–55.5]) and patients who were diagnosed with BM after their mBC diagnosis (n = 302 [80.1%]; overall survival after BM diagnosis was 9.6 months [range, 0.1–54.5])
e Based on univariable analyses only
f Luminal B subtype is defined as HER2+ status with ER+ and/or PR+
g Survival defined as the time between BC diagnosis and death
h The most common symptoms were headaches (50.0%), nausea and vomiting (25.0%), confusion and memory impairment (18.2%), paresis (18.2%), aphasia and dysarthria (6.8%), and seizures (6.8%)
i Includes chemotherapy, endocrine therapy, and HER2-targeted therapy
The most commonly assessed predictive factors for shorter survival after BM diagnosis included age, HR status, number of brain lesions, receipt of anti-HER2 therapy, and receipt of any systemic therapy. Four studies reported no association between age at BM diagnosis and survival [15, 27, 31, 35], three reported shorter survival among older patients [18, 24, 33], and one reported shorter survival among younger patients [28]. The nine studies that observed HR status reported no association between HR status and survival after BM diagnosis [15, 24, 25, 27, 28, 31–33, 36]. Six studies reported that the presence of a higher number brain lesions compared with fewer brain lesions was associated with shorter survival after BM diagnosis [18, 24, 25, 27, 28, 33]. Other reported predictors of shorter survival after BM diagnosis included the presence of neurologic symptoms [24, 26, 27], tumor grade 3, two or more extracranial metastatic sites [33], central nervous system disease at BC diagnosis [31], uncontrolled extracranial metastases [27, 28], visceral metastases, and Radiation Therapy Oncology Group recursive partitioning analysis prognostic class 3 versus class 1/2 [39].
While specific regimens were not assessed in this literature review, data were included from studies that did not assign treatment and for which any anti-HER2 treatment was captured within typical clinical practice. All 13 studies that assessed treatment with anti-HER2 therapy and survival after BM diagnosis reported an association between anti-HER2 therapy and survival. Twelve studies reported that patients who received anti-HER2 therapy after their BM diagnosis had a longer survival compared with patients who did not receive anti-HER2 therapy [17–20, 24, 25, 27, 28, 30, 31, 36, 39]. Four studies [18, 25, 33, 36] found that patients receiving both trastuzumab and lapatinib after their BM diagnosis had longer survival than those receiving either agent alone or no anti-HER2 therapy. One study found that patients receiving trastuzumab and pertuzumab after their BM diagnosis had longer survival than those receiving other HER2-targeted therapies or no HER2-targeted therapy [19].
Four studies [18, 31, 35, 39] assessed non-HER2-targeted therapies and survival after BM diagnosis and reported that shorter survival was associated with no chemotherapy versus chemotherapy [18, 31], no hormonal therapy versus hormonal therapy [18], WBRT alone versus any systemic therapy after WBRT [39], and no systemic therapy versus any systemic therapy [35]. In addition, three studies reported that patients receiving surgery or stereotactic radiosurgery had longer survival than those not receiving these treatments [24, 31, 33], while one study reported no association [35]. One study reported no association between the total dose of radiotherapy and survival after BM diagnosis [28].
Treatment mechanism of action and outcomes after brain metastasis
HER2-targeted monoclonal antibodies
Table 4 presents information on anti-HER2 treatment type by mechanism of action, and disease progression and survival after BM. Four studies evaluated survival among patients receiving a HER2-targeted monoclonal antibody (i.e., trastuzumab) after BM diagnosis and reported that overall survival (OS) was longer in patients who received trastuzumab after local therapy compared with patients who did not receive trastuzumab [17, 30, 31, 39]. In Ahn et al. [17], Berghoff et al. [30], and Brufsky et al. [31], the difference in median OS after BM between trastuzumab users and nonusers was 7.8 months, 10 months, and 13.8 months, respectively. Niwinska et al. [39] reported that among patients with HR+ status, trastuzumab users had a 11-month longer median OS compared with nonusers (P < 0.001), and among patients with HR- status, trastuzumab users had a 6-month longer median OS compared with nonusers (P = 0.004). One study evaluated survival among patients receiving HER2-targeted monoclonal antibody combination therapy (i.e., trastuzumab + pertuzumab) and reported that OS was longer in patients who received trastuzumab + pertuzumab (44 months) compared to those who received other HER2-targeted therapy (17 months) or no HER2-targeted therapy (3 months) [19].
Table 4.
Effect of Treatment Mechanisms of Action on Survival, Tumor Response, Time to Progression
| Citation | Therapy After BM | Outcome Assessed |
|---|---|---|
| HER2-targeted monoclonal antibodies | ||
| Ahn et al., 2013 [17] | Trastuzumab | OS after diagnosis of distant metastasis: trastuzumab, 26.9 mo; no trastuzumab, 19.1 mo; P = 0.020 |
| Berghoff et al., 2012 [30] | Trastuzumab | OS after BM diagnosis, 7 mo (95% CI, 4.3–969); trastuzumab-based therapy after completion of local therapy for BM (surgery, radiotherapy), 14 mo (95% CI, 7.22–20.78); vs. not, 4 mo (95% CI, 2.40–5.61) |
| Brufsky et al., 2011 [31] | Trastuzumab | OS after BM diagnosis, 13.0 mo (range, 0.1–55.5); trastuzumaba, 17.5 mo; vs. no trastuzumab, 3.7 mo; adjusted hazard ratio = 0.33 (95% CI, 0.25–0.46) |
| Niwinska et al., 2010 [39] | Trastuzumab |
OS after BM diagnosis ▪ HER2+/HR+: trastuzumab + chemotherapy after WBRT, 13 mo; chemotherapy alone after WBRT, 8 mo; no systemic treatment after WBRT, 2 mo; P < 0.001 ▪ HER2+/HR-: trastuzumab + chemotherapy after WBRT, 10 mo; chemotherapy alone after WBRT, 8 mo; no systemic treatment after WBRT, 4 mo; P = 0.004 |
| Bergen et al., 2021 [19] | Trastuzumab + Pertuzumab |
OS after BM diagnosis, trastuzumab + pertuzumab, 44 mo; other HER2-targeted therapy, 17 mo; no HER2-targeted therapy, 3 mo (P < 0.001) Overall intracranial CBR, trastuzumab + pertuzumab as systemic first-line therapy after diagnosis of BM, 100% Overall intracranial ORR, trastuzumab + pertuzumab as systemic first-line therapy after diagnosis of BM, 92.9% |
| HER2-targeted non-monoclonal antibodies | ||
| Anwar et al., 2021 [32] | Pyrotinib |
OS after pyrotinib initiation, pyrotinib + surgery/radiation, 20.7 mo; pyrotinib only, 12.4 mo (P = 0.021) PFS after pyrotinib initiation, pyrotinib + surgery/radiation, 10.0 mo; pyrotinib only, 7.7 mo (P = 0.19) CBR after pyrotinib initiation, pyrotinib + surgery/radiation, 58.6%; pyrotinib only, 41.4% ORR after pyrotinib initiation pyrotinib + surgery/radiation, 24.1%; pyrotinib only, 31.0% |
| HER2-targeted monoclonal antibodies + Tyrosine kinase inhibitors | ||
| Braccini et al., 2013 [36] | Trastuzumab + Lapatinib | OS after BM diagnosis, trastuzumab and lapatinib (sequential), 25.7 mo (95% CI, 17.1–33.3); only 1 of the 2 targeted therapies, 9.6 mo (95% CI, 8.2–12.8); P < 0.001 |
| Kaplan et al., 2012 [33] | Trastuzumab + Lapatinib | OS after BM diagnosis, trastuzumab- and lapatinib-based therapy (sequential), 23.6 mo; only 1 of the 2 targeted therapies, 14.6 mo; P = 0.023 |
| Hayashi et al., 2015 [25] | Trastuzumab + Lapatinib | OS after BM diagnosis, trastuzumab and lapatinib had a longer survivalb than trastuzumab alone, lapatinib alone, or no HER2-targeting agent; P < 0.001 |
| Tyrosine kinase inhibitors + other anti-HER2 therapies (not otherwise specified) | ||
| Morikawa et al., 2018 [27] | Anti-HER2 therapy + Lapatinib | OS from BM diagnosis, 19.4 mo (95% CI, 15.5–26.6); anti-HER2 therapy with lapatinib vs. no use, adjusted hazard ratio = 0.26 (95% CI, 0.13–0.52); anti-HER2 therapy without lapatinib vs. no use, adjusted hazard ratio = 0.32 (95% CI, 0.18–0.59) |
| Anti-HER2 therapies (not otherwise specified) | ||
| Braccini et al., 2013 [36] | Anti-HER2 therapy |
▪ OS after BM diagnosis, 11.9 mo (95% CI, 8.7–15.5); anti-HER2 therapy, 15.2 mo (95% CI, 11.5–19.4); without anti-HER2 therapy, 3.4 mo (95% CI, 1.4–6) ▪ Cerebral progression-free survival, anti-HER2 therapy, 6.3 mo (95% CI, 7.8–11.5); without anti-HER2 therapy, 5.5 mo (95% CI, 1.2–6.7) |
| Kaplan et al., 2012 [33] | HER2-targeted therapy (includes all patients receiving trastuzumab, lapatinib, or both) | OS after BM diagnosis; HER2-targeted therapy, 16.7 mo; without HER2-targeted therapy, 4.7 mo; P < 0.001 |
| Gori et al., 2019 [24] | Anti-HER2 therapy | OS after BM diagnosis, 24.5 mo; HER2-targeted therapy (27.5 mo) vs. without anti-HER2 therapy (13.8 mo) (hazard ratio = 0.44 [95% CI, 0.25–0.78]) vs. no systemic therapy (2.1 mo) (hazard ratio = 0.09 [95% CI, 0.05–0.16]) |
| Maurer et al., 2018 [26] | Anti-HER2 treatment | No impact on the development of a second CNS event or on OS. OS, 20.8 mo (IQR, 5.36-not reached) |
| Mounsey et al., 2018 [20] | HER2-targeted therapy (includes trastuzumab, lapatinib, pertuzumab, and T-DM1) |
▪ Mortality after BM, receipt of HER2-targeted therapy after BM diagnosis, adjusted hazard ratio = 0.61 (95% CI, 0.39–0.97) ▪ OS after BM diagnosis, 18.1 mo (95% CI, 14.9–24.6); HER2-targed therapy (62% of patients), 25.3 mo (95% CI, 18.6–31.2); without HER2-targeted therapy, 7.8 mo (95% CI, 4.56–15.0) |
| Yap et al., 2012 [18] | Anti-HER2 therapy (includes trastuzumab alone, lapatinib alone, or trastuzumab and lapatinib combined) | OS after BM diagnosis, 10.9 mo (95% CI, 9.0–11.9); anti-HER2 therapy, 18.5 mo; no anti-HER2 therapy, 5.7 mo; adjusted hazard ratio = 0.62 (95% CI, 0.43–0.89) |
| Zhang et al., 2016 [28] | Anti-HER2 therapy (includes trastuzumab alone, lapatinib alone, or trastuzumab and lapatinib combined) | OS after BM diagnosis, 12 mo (range, 1–94); anti-HER2 therapy after WBRT, 21 mo, no anti-HER2 therapy after WBRT, 9 mo; P = 0.002 |
| Bergen et al., 2021 [19] | HER2-targeted therapy, or no HER2-targeted therapy | OS after BM diagnosis, other HER2-targeted therapy, 17 mo; no HER2-targeted therapy, 3 mo |
BM brain metastasis; CBR clinical benefit rate; CI confidence interval; CNS central nervous system; HER2 Human Epidermal Growth Factor 2; HR hormone receptor; IQR interquartile range; ORR overall response rate; OS overall survival; T-DM1 ado-trastuzumab emtansine; WBRT whole-brain radiotherapy
a 27.5% of patients who received trastuzumab after BM diagnosis also received lapatinib (mostly after trastuzumab). No patients received only lapatinib after BM diagnosis
b Survival months not reported
HER2-targeted non-monoclonal antibodies
One study evaluated survival among patients receiving a HER2-targeted non-monoclonal antibody (i.e., pyrotinib) with or without surgery/radiation, and reported that OS was longer in those with combination HER2-targeted non-monoclonal antibody and surgery/radiation [32]. OS was 20.7 months in those that received both a HER2-targeted non-monoclonal antibody (i.e., pyrotinib) and surgery/radiation, compared with 12.4 months in those who received only a HER2-targed non-monoclonal antibody (i.e., pyrotinib) [P = 0.021] [32].
Tyrosine kinase inhibitor combination therapies
Four studies evaluated the association between patients receiving lapatinib in combination with either trastuzumab or another anti-HER2 therapy, not otherwise specified [25, 27, 33, 36]. Braccini et al. [36] and Kaplan et al. [33] reported that patients receiving both lapatinib and trastuzumab had longer median OS (25.7 months and 23.6 months, respectively) after BM compared with those receiving either lapatinib alone or trastuzumab alone (9.6 months [P < 0.001] and 14.6 months [P = 0.023], respectively). Hayashi et al. [25] reported longer OS after BM in patients receiving lapatinib and trastuzumab compared with patients receiving only one of the two targeted therapies or no HER2-targeting therapy (P < 0.001). Similarly, Morikawa et al. [27] reported that patients receiving lapatinib in combination with another anti-HER2 therapy, not otherwise specified, had lower mortality compared with those not receiving the combination therapy (adjusted hazard ratio = 0.26; 95% confidence interval [CI], 0.13–0.52).
Any anti-HER2 therapy
The use of anti-HER2 therapy, not otherwise specified, after BM diagnosis was associated with an increase in OS in all studies except one [26]. Among the seven studies that reported longer survival in patients treated with anti-HER2 therapy after BM diagnosis [18–20, 24, 28, 33, 36], the median OS among those receiving anti-HER2 therapy ranged from 15.2 to 44 months compared with the median OS among those not receiving anti-HER2 therapy, which ranged from 3 to 13.8 months. Among patients receiving anti-HER2 therapy after BM diagnosis, median OS ranged from 11.8 [36] to 17.5 months [20] longer after their BM diagnosis compared with those not receiving anti-HER2 therapy. Conversely, Maurer et al. [26] reported no association between anti-HER2 therapy after BM diagnosis and OS.
Two studies reported on cerebral disease progression after BM diagnosis and treatment with anti-HER2 therapy [26, 36]. Braccini et al. [36] reported longer cerebral progression-free survival in patients treated with anti-HER2 therapy than in patients not receiving anti-HER2 therapy (6.3 months [95% CI, 7.8–11.5] vs. 5.5 months [95% CI, 1.2–6.7]), while Maurer et al. [26] reported no association between anti-HER2 therapy and a second central nervous system event.
Quality of studies
Quality assessment of included studies was conducted using the Good Research for Comparative Effectiveness (GRACE) checklist [40, 41]. This 11-item scale contains 6 items related to quality of data and 5 items related to methodology. For each question, the quality is assessed based on “fit for purpose”, and the quality is considered sufficient if the data or information provided per item is adequate for study purposes. The quality assessment revealed that all studies were eligible to be included in this review, even though 5 (20%) studies have some limitations in the scientific methods (See Table 5).
Table 5.
Quality of Studies Included
| Citation | Data Quality a | Scientific Method b | Overall Quality Rating |
|---|---|---|---|
| Anders et al., 2011 [15] | Sufficient | Sufficient | Sufficient |
| Witzel et al., 2018 [16] | Sufficient | Sufficient | Sufficient |
| Ahn et al., 2013 [17] | Sufficient | Insufficient | Sufficient |
| Yap et al., 2012 [18] | Sufficient | Sufficient | Sufficient |
| Mounsey et al., 2018 [20] | Sufficient | Sufficient | Sufficient |
| Duchnowska et al., 2012 [21] | Sufficient | Insufficient | Sufficient |
| Duchnowska et al., 2009 [22] | Sufficient | Insufficient | Sufficient |
| Duchnowska et al., 2015 [23] | Sufficient | Sufficient | Sufficient |
| Gori et al., 2019 [24] | Sufficient | Sufficient | Sufficient |
| Hayashi et al., 2015 [25] | Sufficient | Sufficient | Sufficient |
| Maurer et al., 2018 [26] | Sufficient | Sufficient | Sufficient |
| Morikawa et al., 2018 [27] | Sufficient | Sufficient | Sufficient |
| Zhang et al., 2016 [28] | Sufficient | Sufficient | Sufficient |
| Heitz et al., 2009 e [29] | Sufficient | Sufficient | Sufficient |
| Berghoff et al., 2012 [30] | Sufficient | Sufficient | Sufficient |
| Brufsky et al., 2011 [31] | Sufficient | Sufficient | Sufficient |
| Kaplan et al., 2012 [33] | Sufficient | Sufficient | Sufficient |
| Jang et al., 2011 [34] | Sufficient | Insufficient | Sufficient |
| Kuba et al., 2014 [35] | Sufficient | Sufficient | Sufficient |
| Braccini et al., 2013 [36] | Sufficient | Sufficient | Sufficient |
| Niwinska et al., 2010 [39] | Sufficient | Sufficient | Sufficient |
| Sperduto et al., 2013 [37] | Sufficient | Insufficient | Sufficient |
| Martin et al., 2017 [38] | Insufficient | Sufficient | Sufficient |
| Anwar et al., 2021 [32] | Sufficient | Sufficient | Sufficient |
| Bergen et al., 2021 [19] | Sufficient | Sufficient | Sufficient |
a - Data attributes of exposure, outcomes (recording, objective measurement, validation) and important covariates/co-founders were assessed using 6 item checklist
b – Scientific methods were assessed using 5 item checklist and focused on the following areas
- New initiators of treatment
- Concurrent comparators
- Control of covariates/confounders/effect modifier
- Control of immortal time bias
- Analyses to evaluate the potential for bias for biased assessment
Discussion
This literature review included 25 published articles that assessed a total of 4097patients with HER2+ BC with BM. Among these patients, prognostic factors of BM development and predictive factors of survival after BM diagnosis were assessed. Prognostic factors associated with shorter TTBM among patients with HER2+ BC included younger age at BC diagnosis, HR- versus HR+ status [15, 23, 30, 31], no receipt of trastuzumab versus receipt of trastuzumab [18, 23, 31], and higher tumor grade versus lower grades [21, 23]. While these associations were observed across multiple studies, six studies found no association with TTBM and these prognostic factors [17, 21, 22, 26, 29, 30]. Some studies reported longer TTBM in patients receiving trastuzumab or anti-HER2 therapy, not otherwise specified, while some studies found no association.
Overall survival after BM diagnosis was shorter in patients with a higher number of lesions, was unaffected by HR status, and was variably affected by age at diagnosis. Treatment-related factors predictive of longer survival after BM included receipt of any systemic therapy. Studies that assessed treatment with anti-HER2 therapy after BM diagnosis and survival (n = 13) reported that patients who received anti-HER2 therapy had longer survival after BM compared with patients who did not receive anti-HER2 therapy. Among studies that assessed survival differences between trastuzumab-based and lapatinib-based therapies [18, 25, 33, 36], patients receiving both trastuzumab and lapatinib after BM diagnosis had longer survival than those receiving either agent alone or no anti-HER2 therapy. One study found that patients receiving both trastuzumab and pertuzumab therapy after BM diagnosis had longer survival than those receiving other HER2-targeted therapy or no HER2-targeted therapy [19]. While trastuzumab has limited capability to cross the intact BBB, elevated concentrations of trastuzumab in the cerebrospinal fluid have been demonstrated when the BBB is impaired via radiotherapy and/or meningeal carcinomatosis [42]. As described in this review, trastuzumab in combination with lapatinib, which has been shown to cross the BBB in the BM setting [43], is favorable for survival in patients with BM. Lapatinib-based therapies may be an alternative therapeutic option for patients with BM and trastuzumab resistance [44]. The studies in this review also reported that survival after BM was improved with other therapies, including surgery or radiosurgery [24, 31, 33] and other systemic therapies [18, 31, 39]; however, the improvements in survival in patients receiving anti-HER2 therapy superseded survival in those receiving one of these other therapies alone [24, 28, 39].
Contemporary patients with HER2+ BC have better OS compared with patients with HER2+ status who received treatment for BC 20 years ago [45]. As more patients with HER2+ BC are living longer, with more opportunity to develop distant metastases, new investigational agents are needed to treat patients with HER2+ BC with BM. One such drug is tucatinib, a next-generation small molecule TKI that is currently under assessment for improving outcomes specifically among patients with HER2+ BC with BM (ClinicalTrials.gov: NCT02614794 and NCT03975647) [46]. A recent randomized controlled trial found the addition of tucatinib compared with placebo to trastuzumab and capecitabine regimens improved progression-free survival and OS [47]. Trastuzumab deruxtecan, an antibody-drug conjugate, demonstrated encouraging antitumor activity during a phase 2 trial that evaluated patients with HER2+ metastatic BC, including 24 patients with BM [48]. Neratinib, a pan-HER TKI, showed efficacy in combination with capecitabine for treatment of refractory HER2+ BC with BM [49]. Treatment with anti-HER2 therapies, including current investigational and newly approved therapies, may provide additional options for patients with HER2+ BC with BM.
This review includes some limitations, such as the search was limited to the past 10 years and to English-language articles only. Only studies that assessed patients with BM are included in this review. Clinical trials often exclude patients with any BM or enroll only patients with clinically stable BM [50], limiting the ability to comprehensively assess the predictors of survival in patients with BM.
Conclusion
In this literature review, we describe the epidemiology of patients with HER2+ metastatic or advanced BC with BM, including prognostic factors for developing BM, factors predictive of survival among patients with BM, and differences in survival and time to progression by HER2-targeting drug class based on drug mechanism of action. Prognostic factors associated with shorter TTBM included younger age, HR- status, no receipt of trastuzumab or anti-HER2 therapy, higher tumor grade, and larger tumor size. Predictors of longer OS after BM included receipt of anti-HER2 therapy or any systemic therapy, and the presence of fewer brain lesions (< 3 or a single lesion). Trastuzumab and lapatinib combination therapy after BM diagnosis was associated with longer OS after BM compared with other treatments assessed in this review. More research is needed to better understand risk factors for BM and treatments that may improve outcomes.
Supplementary Information
Acknowledgements
The authors thank Brian Samsell of RTI Health Solutions for medical writing assistance.
Abbreviations
- BBB
Blood-brain barrier
- BC
Breast cancer
- BM
Brain metastasis
- HER2
Human epidermal growth factor receptor 2
- HR
Hormone receptor
- OS
Overall survival
- TKI
Tyrosine kinase inhibitor
- TTBM
Time to first brain metastasis diagnosis
- WBRT
Whole-brain radiotherapy
Authors’ contributions
MDH and MS: Conceptualized, designed the study, interpreted the data and substantively revised the manuscript. MH and EW: Reviewed, designed the updated study, interpreted the data and revised the manuscript. HED, AL and MER: Acquired and analyzed data, drafted the manuscript. NT and ZI: Designed the study, interpreted the data and substantively revised the manuscript. All authors read and approved the submitted version the manuscript and agreed both to be personally accountable for the author’s own contributions and the accuracy or integrity of any part of the work.
Funding
Financial support for the study was provided by Daiichi Sankyo, Inc. RTI Health Solutions received funding under a research contract with Daiichi Sankyo, Inc. to conduct this study and provide editorial support in the form of manuscript writing, styling, and submission.
Availability of data and materials
Data has been made available as electronic supplementary material.
Declarations
Ethics approval and consent to participate
The article does not contain any studies with human participants or animals performed by any of the authors, and the protocol required waiver from IRB. As this study is based on a publicly available database without identifying patient information, informed consent was not required.
Consent for publication
Not applicable.
Competing interests
MS, MDH, MH, NT, and ZI are Daiichi Sankyo, Inc. employees. EW is an employee of Rutgers University and is contracted to Daiichi Sankyo, Inc. HED, AL, and MER are employees of RTI Health Solutions, an independent nonprofit research organization that performs contracted project work for medical device and pharmaceutical companies.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Howlader N, Altekruse SF, Li CI, Chen VW, Clarke CA, Ries LAG, et al. US incidence of breast cancer subtypes defined by joint hormone receptor and HER2 status. J Natl Cancer Inst. 2014;106(5). 10.1093/jnci/dju055. [DOI] [PMC free article] [PubMed]
- 2.Goddard KA, Weinmann S, Richert-Boe K, Chen C, Bulkley J, Wax C. HER2 evaluation and its impact on breast cancer treatment decisions. Public Health Genomics. 2012;15(1):1–10. doi: 10.1159/000325746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Leone JP, Lin NU. Systemic therapy of central nervous system metastases of breast cancer. Curr Oncol Rep. 2019;21(6):49. doi: 10.1007/s11912-019-0802-6. [DOI] [PubMed] [Google Scholar]
- 4.National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology (NCCN guidelines): breast Cancer. 2019. Available at: https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf. Accessed: 1 Jul 2019.
- 5.Ramakrishna N, Temin S, Lin NU. Recommendations on disease management for patients with advanced human epidermal growth factor receptor 2-positive breast cancer and brain metastases: ASCO clinical practice guideline update summary. J Oncol Pract. 2018;14(8):505–507. doi: 10.1200/JOP.18.00291. [DOI] [PubMed] [Google Scholar]
- 6.Cardoso F, Senkus E, Costa A, Papadopoulos E, Aapro M, André F, Harbeck N, Aguilar Lopez B, Barrios CH, Bergh J, Biganzoli L, Boers-Doets CB, Cardoso MJ, Carey LA, Cortés J, Curigliano G, Diéras V, el Saghir NS, Eniu A, Fallowfield L, Francis PA, Gelmon K, Johnston SRD, Kaufman B, Koppikar S, Krop IE, Mayer M, Nakigudde G, Offersen BV, Ohno S, Pagani O, Paluch-Shimon S, Penault-Llorca F, Prat A, Rugo HS, Sledge GW, Spence D, Thomssen C, Vorobiof DA, Xu B, Norton L, Winer EP. 4th ESO-ESMO international consensus guidelines for advanced breast Cancer (ABC 4)dagger. Ann Oncol. 2018;29(8):1634–1657. doi: 10.1093/annonc/mdy192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.US Food and Drug Administration. Herceptin prescribing information. 2018. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/103792s5345lbl.pdf. Accessed: 4 Nov 2019.
- 8.US Food and Drug Administration. Perjeta prescribing information. 2018. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/125409s123lbl.pdf. Accessed: 4 Nov 2019.
- 9.US Food and Drug Administration. Kadcyla prescribing information. 2019. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/125427s105lbl.pdf. Accessed: 28 Jun 2019.
- 10.US Food and Drug Administration. Tykerb prescribing information. 2018. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/022059s024lbl.pdf. Accessed: 14 Nov 2019.
- 11.US Food and Drug Administration. Nerlynx prescribing information. 2017. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/208051s000lbl.pdf. Accessed: 11 Jul 2019.
- 12.Kotecki N, Gombos A, Awada A. Adjuvant therapeutic approaches of HER2-positive breast cancer with a focus on neratinib maleate. Expert Rev Anticancer Ther. 2019;19(6):447–454. doi: 10.1080/14737140.2019.1613892. [DOI] [PubMed] [Google Scholar]
- 13.Laakmann E, Muller V, Schmidt M, Witzel I. Systemic treatment options for HER2-positive breast cancer patients with brain metastases beyond trastuzumab: a literature review. Breast Care (Basel) 2017;12(3):168–171. doi: 10.1159/000467387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Moher D, Liberati A, Tetzlaff J, Altman DG, Group P Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Anders CK, Deal AM, Miller CR, Khorram C, Meng H, Burrows E, Livasy C, Fritchie K, Ewend MG, Perou CM, Carey LA. The prognostic contribution of clinical breast cancer subtype, age, and race among patients with breast cancer brain metastases. Cancer. 2011;117(8):1602–1611. doi: 10.1002/cncr.25746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Witzel I, Laakmann E, Weide R, et al. Treatment and outcomes of patients in the brain metastases in breast Cancer Network registry. Eur J Cancer. 2018;102:1. doi: 10.1016/j.ejca.2018.07.004. [DOI] [PubMed] [Google Scholar]
- 17.Ahn HK, Park YH, Lee SJ, Park S, Maeng CH, Park W, Choi DH, Hur SJ, Ahn JS, Im YH. Clinical implication of time to brain metastasis (TTBM) according to breast cancer subtypes. Springerplus. 2013;2(1):136. doi: 10.1186/2193-1801-2-136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yap YS, Cornelio GH, Devi BC, et al. Brain metastases in Asian HER2-positive breast cancer patients: anti-HER2 treatments and their impact on survival. Br J Cancer. 2012;107(7):1075–1082. doi: 10.1038/bjc.2012.346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bergen ES, Binter A, Starzer AM, et al. Favourable outcome of patients with breast cancer brain metastases treated with dual HER2 blockade of trastuzumab and pertuzumab. Ther Adv Med Oncol. 2021;13:17588359211009002. doi: 10.1177/17588359211009002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mounsey LA, Deal AM, Keith KC, Benbow JM, Shachar SS, Zagar T, Dees EC, Carey LA, Ewend MG, Anders CK. Changing natural history of HER2-positive breast cancer metastatic to the brain in the era of new targeted therapies. Clin Breast Cancer. 2018;18(1):29–37. doi: 10.1016/j.clbc.2017.07.017. [DOI] [PubMed] [Google Scholar]
- 21.Duchnowska R, Biernat W, Szostakiewicz B, Sperinde J, Piette F, Haddad M, Paquet A, Lie Y, Czartoryska-Arłukowicz B, Wysocki P, Jankowski T, Radecka B, Foszczyńska-Kłoda M, Litwiniuk M, Dȩbska S, Weidler J, Huang W, Buyse M, Bates M, Jassem J. Correlation between quantitative HER-2 protein expression and risk for brain metastases in HER-2+ advanced breast cancer patients receiving trastuzumab-containing therapy. Oncologist. 2012;17(1):26–35. doi: 10.1634/theoncologist.2011-0212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Duchnowska R, Dziadziuszko R, Czartoryska-Arlukowicz B, et al. Risk factors for brain relapse in HER2-positive metastatic breast cancer patients. Breast Cancer Res Treat. 2009;117(2):297–303. doi: 10.1007/s10549-008-0275-z. [DOI] [PubMed] [Google Scholar]
- 23.Duchnowska R, Jassem J, Goswami CP, Dundar M, Gökmen-Polar Y, Li L, Woditschka S, Biernat W, Sosińska-Mielcarek K, Czartoryska-Arłukowicz B, Radecka B, Tomasevic Z, Stępniak P, Wojdan K, Sledge GW, Jr, Steeg PS, Badve S. Predicting early brain metastases based on clinicopathological factors and gene expression analysis in advanced HER2-positive breast cancer patients. J Neuro-Oncol. 2015;122(1):205–216. doi: 10.1007/s11060-014-1704-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gori S, Puglisi F, Moroso S, Fabi A, la Verde N, Frassoldati A, Tarenzi E, Garrone O, Vici P, Laudadio L, Cretella E, Turazza M, Foglietta J, Leonardi V, Cavanna L, Barni S, Galanti D, Russo A, Marchetti F, Valerio M, Lunardi G, Alongi F, Inno A. The HERBA study: a retrospective multi-institutional Italian study on patients with brain metastases from HER2-positive breast cancer. Clin Breast Cancer. 2019;19(4):e501–e510. doi: 10.1016/j.clbc.2019.05.006. [DOI] [PubMed] [Google Scholar]
- 25.Hayashi N, Niikura N, Masuda N, Takashima S, Nakamura R, Watanabe KI, Kanbayashi C, Ishida M, Hozumi Y, Tsuneizumi M, Kondo N, Naito Y, Honda Y, Matsui A, Fujisawa T, Oshitanai R, Yasojima H, Yamauchi H, Saji S, Iwata H. Prognostic factors of HER2-positive breast cancer patients who develop brain metastasis: a multicenter retrospective analysis. Breast Cancer Res Treat. 2015;149(1):277–284. doi: 10.1007/s10549-014-3237-7. [DOI] [PubMed] [Google Scholar]
- 26.Maurer C, Tulpin L, Moreau M, Dumitrescu C, de Azambuja E, Paesmans M, Nogaret JM, Piccart MJ, Awada A. Risk factors for the development of brain metastases in patients with HER2-positive breast cancer. ESMO Open. 2018;3(6):e000440. doi: 10.1136/esmoopen-2018-000440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Morikawa A, Wang R, Patil S, Diab A, Yang J, Hudis CA, McArthur HL, Beal K, Seidman AD. Characteristics and prognostic factors for patients with HER2-overexpressing breast cancer and brain metastases in the era of HER2-targeted therapy: an argument for earlier detection. Clin Breast Cancer. 2018;18(5):353–361. doi: 10.1016/j.clbc.2017.12.009. [DOI] [PubMed] [Google Scholar]
- 28.Zhang Q, Chen J, Yu X, Cai G, Yang Z, Cao L, Hu C, Guo X, Sun J, Chen J. Survival benefit of anti-HER2 therapy after whole-brain radiotherapy in HER2-positive breast cancer patients with brain metastasis. Breast Cancer. 2016;23(5):732–739. doi: 10.1007/s12282-015-0631-x. [DOI] [PubMed] [Google Scholar]
- 29.Heitz F, Harter P, Lueck HJ, Fissler-Eckhoff A, Lorenz-Salehi F, Scheil-Bertram S, Traut A, Bois A. Triple-negative and HER2-overexpressing breast cancers exhibit an elevated risk and an earlier occurrence of cerebral metastases. Eur J Cancer. 2009;45(16):2792–2798. doi: 10.1016/j.ejca.2009.06.027. [DOI] [PubMed] [Google Scholar]
- 30.Berghoff A, Bago-Horvath Z, De Vries C, et al. Brain metastases free survival differs between breast cancer subtypes. Br J Cancer. 2012;106(3):440–446. doi: 10.1038/bjc.2011.597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Brufsky AM, Mayer M, Rugo HS, Kaufman PA, Tan-Chiu E, Tripathy D, Tudor IC, Wang LI, Brammer MG, Shing M, Yood MU, Yardley DA. Central nervous system metastases in patients with HER2-positive metastatic breast cancer: incidence, treatment, and survival in patients from registHER. Clin Cancer Res. 2011;17(14):4834–4843. doi: 10.1158/1078-0432.CCR-10-2962. [DOI] [PubMed] [Google Scholar]
- 32.Anwar M, Chen Q, Ouyang D, Wang S, Xie N, Ouyang Q, Fan P, Qian L, Chen G, Zhou E, Guo L, Gu X, Ding B, Yang X, Liu L, Deng C, Xiao Z, Li J, Wang Y, Zeng S, Hu J, Zhou W, Qiu B, Wang Z, Weng J, Liu M, Li Y, Tang T, Wang J, Zhang H, Dai B, Tang W, Wu T, Xiao M, Li X, Liu H, Li L, Yi W. Pyrotinib treatment in patients with HER2-positive metastatic breast Cancer and brain metastasis: exploratory final analysis of real-world. Multicenter Data Clin Cancer Res. 2021;27(16):4634–4641. doi: 10.1158/1078-0432.CCR-21-0474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kaplan MA, Isikdogan A, Koca D, Kucukoner M, Gumussoy O, Yildiz R, Dayan A, Demir L, Geredeli C, Kocer M, Arslan UY, Inal A, Unal OU, Mert AG, Bilici M, Ozkan M, Elkiran ET, Yaman S, Durnali AG, Suner A, Alici S, Tarhan MO, Boruban C, Urakci Z, Buyukberber S. Biological subtypes and survival outcomes in breast cancer patients with brain metastases (study of the Anatolian Society of Medical Oncology) Oncology. 2012;83(3):141–150. doi: 10.1159/000338782. [DOI] [PubMed] [Google Scholar]
- 34.Jang G, Lee SS, Ahn JH, Jung KH, Lee H, Gong G, Kim HH, Ahn SD, Son BH, Ahn SH, Kim SB. Clinical features and course of brain metastases in triple-negative breast cancer: comparison with human epidermal growth factor receptor 2-positive and other type at single institution in Korea. Breast Cancer Res Treat. 2011;128(1):171–177. doi: 10.1007/s10549-011-1526-y. [DOI] [PubMed] [Google Scholar]
- 35.Kuba S, Ishida M, Nakamura Y, Yamanouchi K, Minami S, Taguchi K, Eguchi S, Ohno S. Treatment and prognosis of breast cancer patients with brain metastases according to intrinsic subtype. Jpn J Clin Oncol. 2014;44(11):1025–1031. doi: 10.1093/jjco/hyu126. [DOI] [PubMed] [Google Scholar]
- 36.Braccini AL, Azria D, Thezenas S, Romieu G, Ferrero JM, Jacot W. Prognostic factors of brain metastases from breast cancer: impact of targeted therapies. Breast. 2013;22(5):993–998. doi: 10.1016/j.breast.2013.05.011. [DOI] [PubMed] [Google Scholar]
- 37.Sperduto PW, Kased N, Roberge D, Chao ST, Shanley R, Luo X, Sneed PK, Suh J, Weil RJ, Jensen AW, Brown PD, Shih HA, Kirkpatrick J, Gaspar LE, Fiveash JB, Chiang V, Knisely JPS, Sperduto CM, Lin N, Mehta M. The effect of tumor subtype on the time from primary diagnosis to development of brain metastases and survival in patients with breast cancer. J Neuro-Oncol. 2013;112(3):467–472. doi: 10.1007/s11060-013-1083-9. [DOI] [PubMed] [Google Scholar]
- 38.Martin AM, Cagney DN, Catalano PJ, Warren LE, Bellon JR, Punglia RS, Claus EB, Lee EQ, Wen PY, Haas-Kogan DA, Alexander BM, Lin NU, Aizer AA. Brain metastases in newly diagnosed breast cancer: a population-based study. JAMA Oncol. 2017;3(8):1069–1077. doi: 10.1001/jamaoncol.2017.0001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Niwinska A, Murawska M, Pogoda K. Breast cancer brain metastases: differences in survival depending on biological subtype, RPA RTOG prognostic class and systemic treatment after whole-brain radiotherapy (WBRT) Ann Oncol. 2010;21(5):942–948. doi: 10.1093/annonc/mdp407. [DOI] [PubMed] [Google Scholar]
- 40.Dreyer NA, Bryant A, Velentgas P. The GRACE checklist: a validated assessment tool for high quality observational studies of comparative effectiveness. J Manag Care Spec Pharm. 2016;22(10):1107–1113. doi: 10.18553/jmcp.2016.22.10.1107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Dreyer NA, Velentgas P, Westrich K, Dubois R. The GRACE checklist for rating the quality of observational studies of comparative effectiveness: a tale of Hope and caution. J Manag Care Pharm. 2014;20(3):301–308. doi: 10.18553/jmcp.2014.20.3.301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Stemmler HJ, Schmitt M, Willems A, Bernhard H, Harbeck N, Heinemann V. Ratio of trastuzumab levels in serum and cerebrospinal fluid is altered in HER2-positive breast cancer patients with brain metastases and impairment of blood-brain barrier. Anti-Cancer Drugs. 2007;18(1):23–28. doi: 10.1097/01.cad.0000236313.50833.ee. [DOI] [PubMed] [Google Scholar]
- 43.Saleem A, Searle GE, Kenny LM, Huiban M, Kozlowski K, Waldman AD, Woodley L, Palmieri C, Lowdell C, Kaneko T, Murphy PS, Lau MR, Aboagye EO, Coombes RC. Lapatinib access into normal brain and brain metastases in patients with her-2 overexpressing breast cancer. EJNMMI Res. 2015;5(1):30. doi: 10.1186/s13550-015-0103-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lin NU, Dieras V, Paul D, et al. Multicenter phase II study of lapatinib in patients with brain metastases from HER2-positive breast cancer. Clin Cancer Res. 2009;15(4):1452–1459. doi: 10.1158/1078-0432.CCR-08-1080. [DOI] [PubMed] [Google Scholar]
- 45.Pernas S, Tolaney SM. HER2-positive breast cancer: new therapeutic frontiers and overcoming resistance. Ther Adv Med Oncol. 2019;11:1758835919833519. doi: 10.1177/1758835919833519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Murthy R, Borges VF, Conlin A, Chaves J, Chamberlain M, Gray T, Vo A, Hamilton E. Tucatinib with capecitabine and trastuzumab in advanced HER2-positive metastatic breast cancer with and without brain metastases: a non-randomised, open-label, phase 1b study. Lancet Oncol. 2018;19(7):880–888. doi: 10.1016/S1470-2045(18)30256-0. [DOI] [PubMed] [Google Scholar]
- 47.Murthy RK, Loi S, Okines A, et al. Tucatinib, Trastuzumab, and Capecitabine for HER2-positive metastatic breast Cancer. N Engl J Med. 2019. [DOI] [PubMed]
- 48.Modi S, Saura C, Yamashita T, et al. Trastuzumab Deruxtecan in previously treated HER2-positive breast Cancer. N Engl J Med. 2019. [DOI] [PMC free article] [PubMed]
- 49.Freedman RA, Gelman RS, Anders CK, Melisko ME, Parsons HA, Cropp AM, Silvestri K, Cotter CM, Componeschi KP, Marte JM, Connolly RM, Moy B, van Poznak CH, Blackwell KL, Puhalla SL, Jankowitz RC, Smith KL, Ibrahim N, Moynihan TJ, O’Sullivan CC, Nangia J, Niravath P, Tung N, Pohlmann PR, Burns R, Rimawi MF, Krop IE, Wolff AC, Winer EP, Lin NU, on behalf of the Translational Breast Cancer Research Consortium TBCRC 022: a phase II trial of Neratinib and Capecitabine for patients with human epidermal growth factor receptor 2-positive breast Cancer and brain metastases. J Clin Oncol. 2019;37(13):1081–1089. doi: 10.1200/JCO.18.01511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kim ES, Bruinooge SS, Roberts S, Ison G, Lin NU, Gore L, Uldrick TS, Lichtman SM, Roach N, Beaver JA, Sridhara R, Hesketh PJ, Denicoff AM, Garrett-Mayer E, Rubin E, Multani P, Prowell TM, Schenkel C, Kozak M, Allen J, Sigal E, Schilsky RL. Broadening eligibility criteria to make clinical trials more representative: American Society of Clinical Oncology and friends of Cancer research joint research statement. J Clin Oncol. 2017;35(33):3737–3744. doi: 10.1200/JCO.2017.73.7916. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data has been made available as electronic supplementary material.