Abstract
The 4th Universal Definition of Myocardial Infarction has stimulated considerable debate since its publication in 2018. The intention was to define the types of myocardial injury through the lens of their underpinning pathophysiology. In this review, we discuss how the 4th Universal Definition of Myocardial Infarction defines infarction and injury and the necessary pragmatic adjustments that appear in clinical guidelines to maximize triage of real-world patients.
Keywords: Troponin, Myocardial infarction, Myocardial injury
1. Introduction
The measurement of Cardiac troponin concentration in systemic venous blood has become a core component of the assessment of patients with acute—and chronic—cardiovascular disease. This is enshrined in the Universal Definition of Myocardial Infarction (UDMI),1,2 now in its fourth iteration—with the aim to (i) guide the clinician through the numerous differential diagnoses that result in cardiac troponin elevation, and (ii) provide classification and naming conventions to assist a structured approach. However, the 4th UDMI has stimulated considerable debate.3–5 Our previous review6 covered the basic biology of cardiac troponin, the physiology underlying its release from the heart, the analytic science enabling its detection in the blood, and its use in the diagnosis of myocardial infarction according to the 3rd UDMI.7 The purpose of this current review is to discuss the pathophysiology that underpins the 4th UDMI and how it is translated into clinical guidelines and practice—with a specific focus on the challenges encountered ‘at the coalface’ of acute cardiovascular care.
2. Summary of the 4th UDMI
The 4th UDMI is based on sound pathophysiological concepts which are then used to classify everyday cardiovascular events in patients with or without diagnostic ST-elevation on their presenting ECG. Such patients are almost always first identified by a troponin concentration in a venous blood draw exceeding the ‘normal’ range; defined by the 99th centile upper reference limit (URL). For reasons that are practical, rather than rational, patients in whom myocardial infarction is extremely unlikely will still have their troponin measured. Although, such overuse of the troponin assays seems benign, from a Bayesian perspective it means that the pre-test probability of myocardial infarction is very low. Depending on the specific healthcare environment studied, this results in wide variation in prevalence of myocardial infarction—from 5% to 20% in published studies across the world.8,9
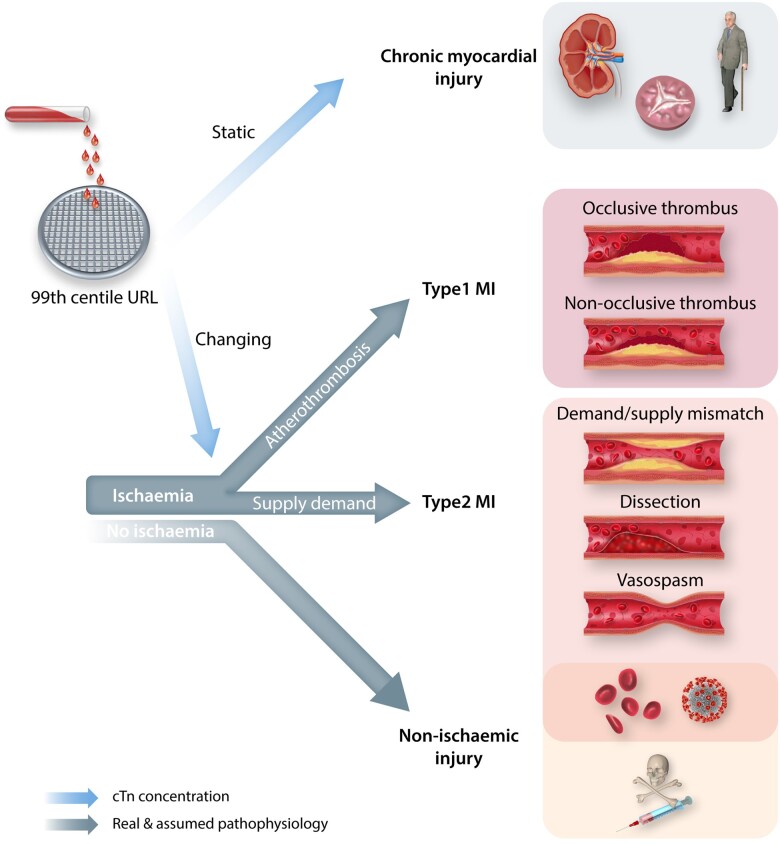
The 4th UDMI therefore follows a ‘surgical sieve’ approach in an attempt to filter these heterogeneous patient cohorts towards their correct classification. This sieve applies three questions in series (see Figure 1): (i) Is the concentration of cTn above or below the 99th centile URL? (ii) Is the cTn concentration static or changing? (iii) Is there evidence of myocardial ischaemia?
Figure 1.
Schematic of the 4th Universal Definition of Myocardial Infarction (UDMI). The blood sample is from a patient with suspected non-ST elevation acute coronary syndrome. The sieve represents the cardiac troponin concentration cut-point at the 99th centile upper reference limit. In patients with troponin above this concentration, the UDMI recommends a differentiation of myocardial infarction from injury, and emphasizes the use of the best imaging techniques available to define aetiology of myocardial injury (preferably cardiac magnetic resonance imaging).1 See text for further details.
All patients with a cardiac Troponin concentration above the URL are defined as having ongoing myocardial damage—in those with a dynamic rise/fall the damage is considered to be acute/unstable—in those with more static concentrations the damage is considered to be chronic/stable. Those patients with acute damage are defined as having either acute myocardial infarction (AMI) or acute myocardial (non-ischaemic) injury, with the dichotomy between these conditions driven by the presence or absence of cardiac ischaemia, respectively. The following are considered indications of cardiac ischaemia:
signs (ECG) or symptoms of myocardial ischaemia,
new loss of viable myocardium,
evidence of coronary thrombus, and
the context of the cTn elevation (spontaneous, peri-procedural).
The 4th UDMI then subclassifies AMI based on aetiology—with Type 1 MI referring to events triggered by atherosclerotic plaque rupture/erosion. Type 2 MI can be seen as a hypernym capturing all the events and processes that lead to myocardial ischaemia not caused by acute atherosclerotic plaque rupture. These include excessive myocardial demand and/or reductions in myocardial supply (such as coronary artery vasospasm, microvascular dysfunction, coronary embolus, or spontaneous coronary artery dissection) in the presence or absence of stable obstructive atherosclerosis. The other subclassifications of AMI are more straightforward—Type 3 MI occurs in patients who suffer cardiac death due to likely myocardial ischaemia. Type 4a MI is myocardial infarction occurring in the context of percutaneous coronary intervention (PCI), Type 4b MI is due to stent/scaffold thrombosis following prior PCI, Type 4c MI is due to restenosis (within stent or in the native coronary artery following plain old balloon angioplasty); finally Type 5 MI occurs in the setting of coronary artery bypass graft (CABG) surgery. For types 4 and 5 AMI, a cTn threshold much higher than the 99th URL is used to reduce the incidence of trivial AMI and just highlight events that maybe clinically meaningful. The use of a higher cTn concentration for procedural vs. spontaneous AMI is controversial and has recently been reviewed elsewhere (see E.H.J. Bulluck et al.,2 in review).
Although this framework to filter patients towards their final diagnosis is logical and straightforward, its translation into clinical practice is confounded by the reality of cardiovascular pathophysiology. Below we discuss these key confounders in the order that they arise in Figure 1.
2.1 What has changed from the 3rd UDMI?
The 4th UDMI has introduced novel concepts: Most importantly, there is now a clear distinction between myocardial infarction and injury, with the latter also affecting the classification of peri-procedural injury. New sections include the description of Takotsubo syndrome and Myocardial Infarction with Non-Obstructive Coronary Arteries (MINOCA). The document further touches on the use of non-invasive imaging techniques to assist in defining aetiology of injury (CMR) and anatomy in the context of infarction (CT coronary angiography). Several concepts have seen an update, as described above; most importantly, sex-specific 99th centile thresholds (which are assay-specific) received a clear recommendation where hs-cTn assays are used.
3. Overview of troponin structure and risk of false-positives
The structure of the sarcomere as the contractile unit of the heart has been discussed previously (see Chapters 2.1 and 2.2 of Park et al.6). In brief, the different subunits of cardiac troponin10, specifically troponin T and I which are targeted by the monoclonal antibodies used in modern cTn assays, were first described and classified by Greaser and Gergely in 1973:11 the inhibitory fraction (cTnI) limits activity of the actomyosin ATPase, while the cTnT fraction binds to tropomyosin and serves as a mechanical link. Modern hs-cTn assays specifically quantify cardiac isoforms of the troponin subunits, but there is evidence to suggest that re-expression of foetal isoforms can occur in the context of pathologies affecting the skeletal muscle.12 These have been shown to cross-react with the (cardiac) monoclonal antibodies, thus yielding a true false-positive result in a rare set of conditions.13 Assay interferences from endogenous sources comprise (rare) endogenous antibodies against either the cTn complex or the exogenous, usually murine, antibodies used for detection and/or capture cTn.14 Biotin in over-the-counter dietary supplements can interfere with biotin–streptavidin based assays: depending on the assay format (competitive vs. sandwich method), this can result in false-negative or false-positive results.15
4. Release mechanisms
Experimental evidence supports the thesis that release of cTn is exclusively due to irreversible cell death.16–18 It was postulated that myocardial ischaemia alone could result in cTn release without evidence of necrosis—potentially with only ‘reversible’ cell damage or through the release of membranous blebs; neither mechanism is substantiated to date.19–21 For an in-depth review of these mechanisms, we refer the reader to Chapter 6.1 of Park et al.6 Overall, there appears to be a spectrum of ischaemia/reperfusion injury—some mild enough to remain sub-clinical—but it appears unlikely that there is no myocyte death when there is cTn detection.22 After all, the latest hs-cTn assays operate in the femtomolar range, thus providing greater resolution and precision than modern imaging techniques.23,24 Furthermore, depending on the timing and success of revascularization following myocardial infarction, evidence suggests extended elevation of cTn for 12–20 days.25
5. The 99th centile URL
The 99th centile is the threshold to distinguish between ‘normal’ and ‘abnormal’ cTn concentrations. The dichotomy is artificial since the mode, median, mean, range, and skewness of the distribution of troponin concentrations in a given ‘healthy normal’ population depends on the inclusion criteria used to define ‘normal’. As Apple et al.16 point out, these criteria vary between assay manufacturers—whereas some define healthy based on age (<30 years) and apparent lack of comorbidity, others screen with blood tests (e.g. for natriuretic peptides to exclude sub-clinical cardiac dysfunction) and group according to race and sex. These inclusion criteria are not standardized and they each ‘shift’ the 99th centile, which increases with age, male sex, serum creatinine (renal dysfunction), and a range of other cardiovascular risk factors.26 One ramification of ill-defined 99th centiles was highlighted by Shah et al., amongst others, who compared a combined 99th centile to sex-specific thresholds [which are almost two-fold higher in men than in women for hs-cTnI (Architect)], demonstrating that the combined threshold would result in an under-diagnosis of AMI in women.16,27–29 But the use of sex-specific thresholds for the 99th centile is not without debate—and has demonstrated significant variation depending on the cTn assay in use. In a systematic review published by Kimenai et al.,30 hs-cTnI was subject to much higher variation (80%) with respect to the 99th centile thresholds than hs-cTnT (29.4%). Nevertheless, the female-specific URL was consistently lower than the uniform decision limits for any hs-cTn assay. In a large cluster-randomized controlled trial involving >48 000 patients, hs-cTnI with sex-specific threshold identified 5 times more women than men with any myocardial injury, however without an improvement in outcomes; the latter possibly due to undertreatment.31 Interestingly, in a retrospective analysis of hs-cTnT data collected as part of a prospective diagnostic multicenter study, the use of sex-specific thresholds did not lead to a significant diagnostic reclassification.32 Overall, the variation appears to affect hs-cTnI assays more than hs-cTnT, with a risk/benefit assessment favouring the use of sex-specific thresholds—likely a contributing factor to the 4th UDMI’s endorsement of their use.1
With respect to standardization, Apple et al.33 summarized recommendations for defining an assay-specific 99th centile on behalf of the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) Task Force on Clinical Applications of Cardiac Bio-Markers (TF-CB). Hopefully, these recommendations will increase uniformity of the 99th centile URL across assay platforms. In addition to population selection, the assay method and specimen type can also influence the 99th centile. Thus, the minimum recommended sample size for derivation is ‘300 male and 300 female subjects’.33 As per the IFCC recommendations, a hs-cTn assay ought to (i) achieve an imprecision defined by the coefficient of variation (CV) ≤10% at the 99th centile and (ii) quantify cTn in ≥50% of healthy subjects. Both requirements encourage the use of less stringent inclusion criteria for normal studies; which will skew the concentration vs. frequency distribution to the right and increase the 99th centile URL.
Crucially, the population of patients undergoing troponin testing on clinical grounds includes many individuals who would have been excluded from the ‘healthy’ normal population used to derive the 99th centile URL.34 As a direct consequence, the prevalence of cTn concentrations above the URL increases from the expected 1% to as much as 40%. This very high ‘false-positive’ prevalence of myocardial injury is a major challenge for the clinical implementation of high-sensitivity assays and the way in which this liability is mitigated is discussed further below.
6. Static or changing troponin?
The cTn concentration measured in a healthy stable person varies overtime due to a combination of measurement imprecision (analytical variance) and true biological variation. cTnI concentrations appear to vary randomly over a 24-h period: the coefficient of variation within-subject (CVI) is constant at 8–9% for all time intervals and is unaffected by the underlying renal function.35,36 cTnT on the other hand follows a marked diurnal variation in healthy volunteers, with the concentration in the morning on an average 4 ng/L (approximately 1/3rd of URL) higher than in the evening.36
As discussed, much more than 1% of the population presenting to emergency departments will have a cTn concentration above the 99th centile URL even when they were stable, outside hospital, and going about their daily activities. This is because they are older, have more cardiovascular risk factors, and worse renal function than the healthy reference population used to define the 99th centile URL. It is therefore necessary to distinguish people with chronic/stable elevations in cTn concentration from those with acute elevations related to a medical event triggering their presentation. Those with chronically elevated troponins may have absolute cTn concentrations which are similar to those in patients with acute, but minor, myocardial injury; so, magnitude alone cannot be used to differentiate between these scenarios. As the concentration of cTn increases the chances of an underlying acute event rise too. Nonetheless, cTn concentration alone is poor at discriminating acute from chronic myocardial damage, particularly with concentrations of cTn modestly above the 99th centile URL. The two groups can also be distinguished by identifying a change in concentration over time that exceeds the ‘noise’ expected in stable individuals due to a combination of natural biological variation and the analytic variance of the assay. The need to identify that the cTn concentration is either rising and/or falling is an absolute requirement for the diagnosis of myocardial infarction that is endorsed in all the major guidelines.37,38 The most usual way to set the dichotomy limit between static or changing cTn is to compare concentration measurements from two blood draws separated by a defined time interval in patients in whom the final diagnosis has been rigorously classified. The change in the second troponin concentration relative to the first can either be expressed as an absolute difference or as a percentage. Reichlin et al.39 compared these different reference change strategies in a well-classified cohort and found absolute change to be diagnostically superior.
The guideline-recommended reference (or delta) change values are unexpectedly small [e.g. 3 ng/L for hs-cTnT (Elecsys), 2 ng/L for hs-cTnI (Architect)], however, these are optimized/calibrated to achieve a high sensitivity: As outlined, small fluctuations in cTn concentration occur over a 24-h period as well as in a diurnal pattern,35,36 and a number of assays demonstrate a substantial within-subject coefficient of variation when comparing short-term repeats.40–42 Consequently, diurnal and/or individual variation can result in false-positives for acute myocardial injury that compound the false-positives for any form of myocardial injury set by a 99th centile URL derived from a healthy cohort.
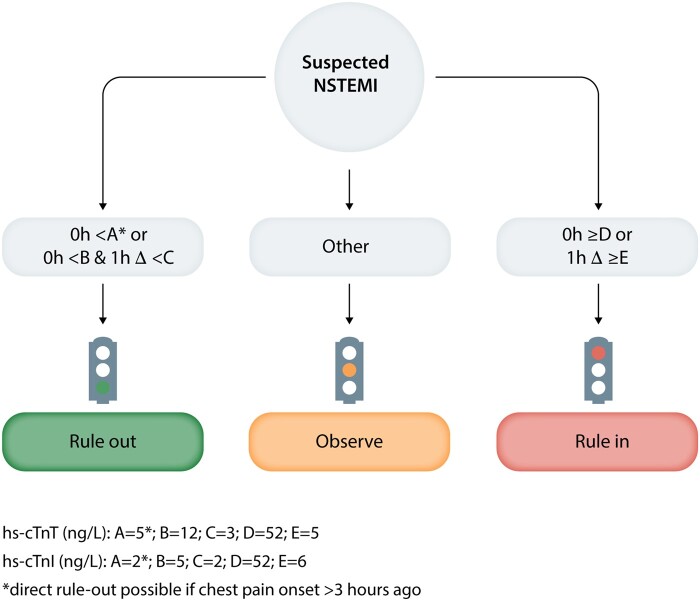
How do these considerations impact medical care? For this, we reference Figure 2 (2015 ESC guidelines for the management of ACS38) which outlines how clinicians in the Emergency Department may assess patients for an acute coronary syndrome, in the absence of ST segment elevation on the ECG. As is evident from the flowchart, the decision cTn concentrations for immediate rule-out or rule-in of ACS on a single blood draw taken at presentation (0 h)—at least 3 h after chest pain onset for rule-out—are widely spaced around the 99th centile URLs of the commercial assays [14 ng/L for hs-cTnT (Elecsys), 34 ng/L in men and 16 ng/L in women for hs-cTnI (Architect)]. For those patients with cTn concentrations between these widely spaced limits a further blood draw is necessary after 1 h (other side of the ‘or’ in Figure 2). The absolute deltas in cTn concentration between first and second blood-draw, that are used to categorize patients are relatively low, only very minimal changes will allow the rule-out of an ACS. A greater change (≥5 ng/L for hs-cTnT, ≥6 ng/L for hs-cTnI) will categorize the patient as rule-in for ACS and likely lead to treatment for Type 1 MI with antiplatelet and anti-thrombotic medication and probable invasive coronary angiography with a view to PCI (balloon angioplasty and stent). In addition, evident from the flowchart is the intermediate (amber) group of patients that neither rule-in nor rule-out—who are confined to an ‘observe’ category. This accounts for the inherent uncertainty that occurs between the extremes of the immediate and 0/1-h decision thresholds and reflects the spectrum of human pathobiology. In fact, the decision thresholds are optimized for high sensitivity at the rule-out threshold, and high specificity at the rule-in threshold. As a consequence, 24–50% of patients assessed with the ESC 0/1 h algorithm remain in the ‘observe’ zone after a second blood draw9,43–63 and require further assessment.
Figure 2.
ESC 0/1 h rule-in and rule-out algorithms using high-sensitivity cardiac troponins (hs-cTn) assays in patients presenting with suspected non-ST-elevation myocardial infarction (NSTEMI) to the emergency department; concept as per Roffi et al.38
6.1 Imaging adjuncts to refine risk stratification in the grey zones of hs-cTn pathways
The form of the assessment for those patients left in the observe zone has not yet been similarly protocolized and tends to be tailored to the individual patients. However, patients in the observe zone tend to be categorized further based on repeat cTn measurement, serial ECGs, and non-invasive imaging including CT coronary angiography (CTCA) and stress echocardiography. Only few studies to date have investigated the use of ECG-gated CTCA to refine the population with indeterminate hs-cTn concentrations—a majority of the evidence base stems from the era of sensitive cardiac troponin assays and is thus not translatable to modern chest pain protocols. Of those employing CTCA in conjunction with hs-cTn, the BEACON trial demonstrated no reduction in length of stay nor 30-day revascularization;64 Smulders et al.65 demonstrated a reduction of the use of invasive coronary angiography with similar outcomes when used in patients with hs-cTn concentrations above the 99th centile; the VERDICT trial evaluated the use of CTCA in an observational component of a trial comparing very early to standard invasive coronary angiography, demonstrating comparable diagnostic accuracy to rule-out significant coronary artery disease.66 Most recently, the 2020 ESC guidelines for the management of acute coronary syndromes recommend the use of CT as an alternative to invasive angiography ‘when there is low-to-intermediate likelihood of CAD’ and biomarkers are ‘normal or inconclusive’.67
Importantly, many of the patients in observe zone, as well as some patients in the Rule-In zone (up to 25%), will receive a final diagnosis of Type 2 MI or ‘chronic myocardial injury’. Latest data, adjudicated on the 4th UDMI, demonstrates that both are prognostically relevant and meaningful. But whilst there is compelling evidence that myocardial injury has a prognostic impact on individuals, in the presence or the absence of an acute event, the management of patients with chronic elevations is poorly defined and it is often unclear how their risk can be modified.68–72 We hence discuss the aetiology of chronic myocardial injury in greater detail below.
7. Chronic myocardial injury
The release of cTn is agnostic to the cause of myocardial damage. Why the majority of the healthy population has quantifiable cTn in their peripheral blood (based on high-sensitivity assays) remains an enigma. With the IFCC’s definition of high-sensitivity cardiac Troponin, it follows that a majority of individuals investigated at Emergency Departments have a cTn result above the LOD, and many above the URL.33,73 As such, the distinction of acute vs. chronic myocardial injury lies in both the magnitude of the cTn concentration and its temporality of change—this is illustrated by the derivation and validation of the cTn concentration cut points that underpin Figure 2. Below we summarize the chronic pathophysiological processes that are known to elevate cTn and may help shed light on why chronic cTn concentration correlates with long-term prognosis, even below the 99th centile URL.74,75
In the absence of a preceding acute insult, apoptosis can lead to chronic cTn elevation, in particular in the context of a failing heart.76,77 Hibernating myocardium might play a role in the chronic elevation of cTn but is both poorly understood and unproven. Inflammatory cytokines or increased plasma membrane permeability of injured cells might also play a role.78 Myocardial cell stretch-related mechanisms in viable and non-injured cardiomyocytes mediated by integrin signalling could further lead to constant cTn release.79
In the recovery period following an acute insult, remodelling of the injured myocardium ensues over weeks to months leading to a variable degree of tissue repair, compensatory hypertrophy, and replacement-fibrosis—which could explain cTn release that persists following MI.80 Left ventricular wall strain, interstitial changes reducing capillary density and resulting subendocardial ischaemia (due to lower coronary reserve), as well as activation of vasoconstrictive neurohormones could all contribute to cTn release under this circumstance.81–86
An interesting subset is the patient with stable coronary artery disease—recently coined ‘chronic coronary syndrome’ by the ESC87 as a juxtaposition to the acute syndrome causing infarction. At this stage, it is likely that a combination of different mechanisms leads to chronically elevated cTn concentrations in this cohort:86,88–90 apoptosis, cardiomyocyte turnover, myocardial strain, increased cardiac mass, and subclinical plaque rupture are all thought to contribute. The extent of coronary atherosclerosis and high-risk plaque phenotypes (based on intravascular ultrasound) also associate with elevated circulating cTn concentrations.91
Response to normal everyday physiological stress further adds to the pathological potpourri contributing to chronic myocardial injury. Numerous studies have detected elevated cTn concentrations in otherwise healthy individuals following physical exertion.92–100 Intriguingly, the magnitude of biomarker change poorly correlates with conventional cardiac risk factors. However, younger age,101,102 increased intensity of exercise,97,99,102 and lower baseline fitness92,101 are all significantly associated with the magnitude of cTn elevation post-exercise. Whilst there is no evidence of structural myocardial damage on cardiac MRI,95,103–105 this technique routinely lacks the resolution to identify the small volume myocardial damage that can cause significant cTn elevation—although, future applications using 7T and imaging optimization might be able to overcome these shortfalls.24,106 It was hypothesized that the cTn release during exercise could result from the release of cytosolic pools of cTn due to increased cellular permeability without irreversible myocardial injury.93,98,100 However, this concept is difficult to prove since cTn is the most sensitive marker of myocardial injury at our disposal and there is no alternative technique to arbitrate the presence or absence of reversibility.
8. The quest for ischaemia
The distinction between acute and chronic myocardial injury, based on whether cTn concentration is static or changing, is logical; albeit more complicated in clinical practice than predicted by pathological theory. The next key question to the right of the sieve in Figure 1 is whether a cTn concentration above the 99th centile which is changing is due to myocardial injury or myocardial infarction? The latter mandates the presence of myocardial ischaemia at some point during the patient’s presenting illness. Unlike, the questions of whether cTn concentrations are changing or static, the dichotomy between acute myocardial injury and infarction is ephemeral.
Clinicians rely on relatively crude tools to detect the presence of ischaemia in the acute setting—chiefly, the pattern of chest pain symptoms and/or the occurrence of ST-segment changes on the surface ECG. The differential diagnosis of chest pain is very wide and includes common pathologies unrelated to the heart (e.g. musculoskeletal and oesophageal). Whilst the ECG—in the absence of ST elevation—has a sensitivity for AMI of less than 50%107 and poor specificity. Tools for the detection of stress-induced ischaemia are available, but these are designed and validated to quantify ischaemia in patients with stable coronary artery disease87 and cannot be used to detect ischaemia after a spontaneous event.
The gold-standard in the assessment of the coronary arterial tree—invasive coronary angiography—detects luminal narrowing, but cannot reliably identify or exclude an atherosclerotic plaque as the culprit of an acute coronary syndrome.108,109 The best intravascular imaging modalities use either intravascular ultrasound (IVUS) or optical coherence tomography (OCT). The former, in its conventional form, can reliably quantify lumen area, plaque burden, and vascular remodelling, but due to its axial resolution (70–200 µm) it cannot define plaque morphology.110 Near-infrared spectroscopy-IVUS (NIRS-IVUS) enhances conventional IVUS through its ability to further characterize a plaque’s lipid core.111 OCT provides higher resolution (10 µm) and has been used extensively for plaque characterization.112–114 Serial intravascular imaging studies have confirmed the development of atherosclerotic plaque occurs over several years, and morphological features such as cap thinning, plaque burden115 and microcalcifications together with shear stress—rather than luminal narrowing—determine the chance of plaque rupture.109 Therein lies the challenge of detection, as these vulnerable plaques often remain clinically silent due to their non-obstructive nature, until sudden plaque rupture and subsequent localized thrombosis leads to an acute coronary syndrome.
9. Evidence is required—of plaque rupture and atherothrombosis
In ST-segment elevation myocardial infarction, it is common to find thrombus (at least partially) occluding an epicardial coronary vessel on invasive coronary angiography.116 In only very few additional cases, the ‘evidence’ is as obvious—the ischaemic insult can trigger ventricular fibrillation, which leads to cardiac arrest and—unless promptly resuscitated—death.117 In patients with non-ST-elevation myocardial infarction (NSTEMI), and as outlined above, the ‘trail of evidence’ is much less clear. Most individuals will have suffered prolonged (>20 min) ischaemic chest pain at rest, but reveal a normal cardiovascular examination and unhelpful ECG.38,118 Beyond patient demographics and the assessment of cardiac biomarkers, there is little that can refine the clinician’s pre-test probability before proceeding to an invasive assessment of the coronary anatomy. The invasive coronary angiogram (ICA) can demonstrate localized (non-occlusive) thrombus, but more often demonstrates only luminal narrowing. Features suggestive of acute plaque rupture are intraluminal filling defects (consistent with thrombus), plaque ulceration (contrast holdup and hazy contour extending beyond the vessel lumen), irregularity of the plaque, dissection, or impaired flow.38,119–121 As many of these features are subject to intra-observer variability, identifying the culprit lesion remains a frequently encountered challenge in NSTEMI presentations. Intravascular imaging can improve the precision, but adds time, cost, and risk to the procedure which has to be carefully weighed against the potential benefits.
10. Demand and supply imbalance, chronic overload, and cellular injury
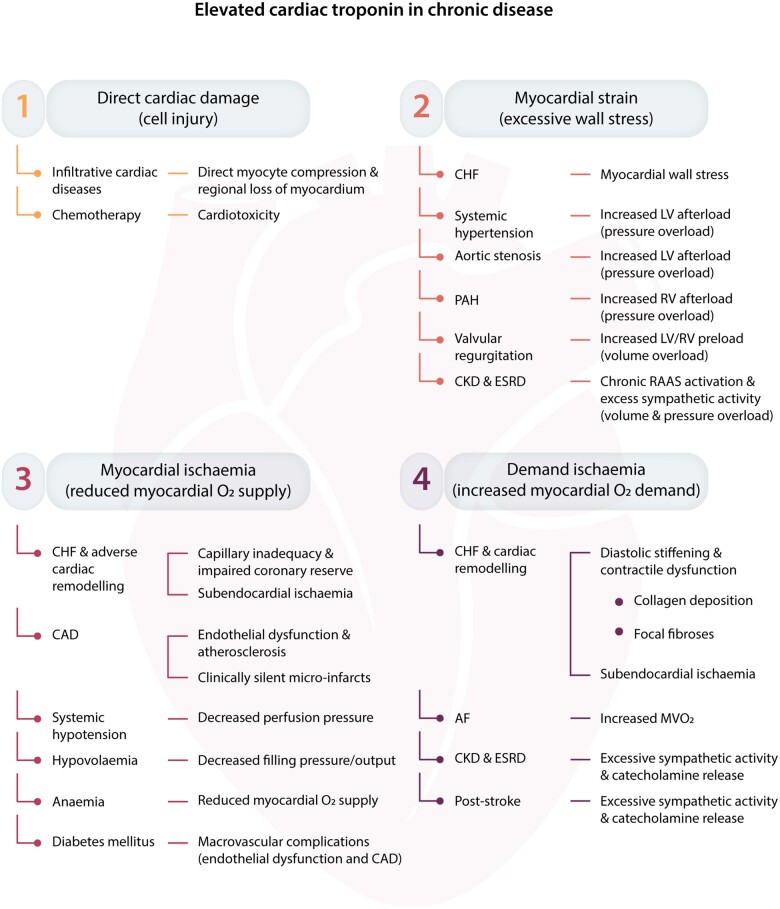
Any transient insult to the myocardium will result in an acute biomarker release—depending on the circumstance, this might be an acute on chronic release when there are other factors contributing to myocardial strain. Figure 3 highlights the many patterns of increased demand, ischaemia, strain, or direct cardiac damage which can lead to cardiac Troponin release.6 From a pathophysiological standpoint, there is significant overlap between conditions that cause chronic and acute cTn release and indeed those mechanisms have to be recognized as a continuous spectrum rather than an easily dichotomized disease entity.
Figure 3.
An outline of the different mechanisms contributing to an elevated cardiac Troponin concentration in chronic disease; from Park et al.6
11. Acute myocardial injury
There is one final disease classification that captures conditions with a dynamic cTn rise/fall above the 99th centile: acute myocardial injury. This might arise as a consequence of conditions such as myocarditis or non-cardiac entities such as renal failure—the implication being that there is no evidence of ischaemia leading to cellular damage and in some cases necrosis. The suggested mechanisms mirror those causing chronic myocardial injury, but due to the ‘acuity’ of the extra-cardiac insult, the heart is affected too—through mechanical stretch, physiological stress, apoptosis, increased turnover of cardiomyocytes, or cellular release of cTn degradation products.1,122–124 Given the challenges with reliable detection of ischaemia, the hypothesized margins between acute cTn release due to acute heart failure (acute myocardial injury) and severe hypertension (T2MI) certainly blur—as both would cause an increase in myocardial strain and affect myocardial wall stress, the pathophysiological differences are all but clear.
12. Why are clinicians unhappy?
The 4th UDMI takes an approach centred around the aetiology of cardiac Troponin elevation to classify disease entities. The challenge lies in the real-world application of these criteria—which require a post hoc analysis of all available clinical information to distinguish between categories such as Type 1 and Type 2 MI. And here is where the water becomes increasingly muddied—how does the clinician distinguish between a plaque-rupture event causing non-occlusive thrombus leading to distal embolization and subsequent cardiomyocyte necrosis125,126—Type 1 MI; and an oxygen supply/demand imbalance due to atherosclerosis—Type 2 MI—after the fact? Only a few milligrams of tissue are required to undergo necrosis to elevate the systemic cardiac Troponin level above the 99th centile,24 but the best available imaging modalities—cardiac magnetic resonance imaging—does not have the spatial resolution to discern supply/demand mismatch from distal embolization affecting less than 1 g of myocardium.127
Further complications arise from the mixed entities summarized as leading to Type 2 MI. Whilst non-atherosclerotic coronary dissection is classified as Type 2 MI, the process by which this causes myocardial necrosis is partially comparable to atherosclerotic plaque rupture resulting in obstructive thrombus formation (Type 1 MI): the lack of distal coronary blood flow for a transient period of time leads to necrosis, and the clinical management until the point of diagnosis remains similar—consideration of antiplatelet medication to limit an assumed pro-thrombotic state, and invasive coronary angiography to establish the exact cause of the clinical and biochemical syndromes.
The various pathologies which can be summarized under Type 2 MI yield an additional challenge: whilst an update of the International Classification of Diseases (10th edition) has introduced a code for Type 2 MI,128 the heterogeneity of the classification makes future research incredibly challenging. De Lemos et al.3 have highlighted that at least three entities (SCAD, coronary embolism, and vasospasm) are acute processes that require management similar to Type 1 MI. As such, their recommendation was to re-classify the above entities under a sub-group of Type 1 MI—to allow a distinction between atherosclerotic events and acute coronary obstruction for other reasons. Whilst treatment for these diagnoses are different, the investigation of choice to discern atherothrombotic from other coronary disease is the same—an ICA. Operationally, many might favour a colocation of subtypes of myocardial infarction that streamlines (i) the investigative path and (ii) subsequent management which is inherently dependent on (i).
13. Clinical scenario
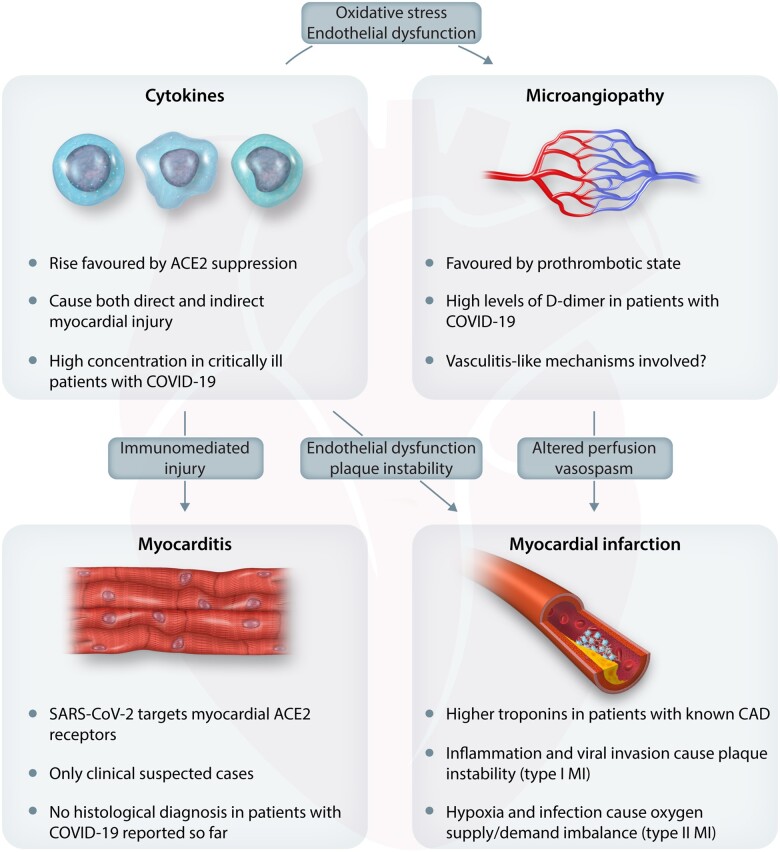
Imagine the following scenario: a 60-year-old male with a past medical history of hypertension and Type 2 diabetes mellitus is admitted to hospital in April 2020 with breathing difficulty, low oxygen saturation, and relative hypotension at 90/60 mmHg. The patient is noted to be tachycardic and on clinical examination is severely breathless at rest. The chest radiograph is supportive of a diagnosis of COVID-19 pneumonitis, but the differential diagnosis includes pulmonary congestion from acute heart failure, potentially triggered by an acute coronary syndrome. An electrocardiogram obtained at admission is not diagnostic—there are lateral T-wave changes which could be in keeping with left-ventricular hypertrophy or myocardial ischaemia; laboratory parameters demonstrate a low lymphocyte count, high d-dimers and fibrinogen levels, and a high-sensitivity cTnT of 60 ng/L (URL 14 ng/L); renal function demonstrates an acute kidney injury. Symptomatically, the patient describes pain on deep inspiration, but no classic symptoms suggestive of ACS. There is no evidence of ongoing ischaemia through presence of regional wall motion abnormalities on echocardiography. The patient is appropriately treated in a critical care environment and a cardiologist is asked to interpret the elevated cTn level, to guide further management. The patient is pre-disposed to a chronically elevated cTn concentration: systemic hypertension causes an increase in LV afterload. Diabetes mellitus affects micro- and macrovasculature, and the combination of endothelial dysfunction and (previously) stable coronary artery disease in the context of relative hypotension would lead to Type 2 MI. In the context of this patient’s illness, an acute kidney injury could contribute to acute myocardial injury. Even with invasive coronary angiography, the clinician might not be able to rule-out Type 1 MI as a unifying explanation for the elevated cTn concentration in a pro-thrombotic state such as during acute COVID-19. Figure 4 highlights the various mechanisms contributing to this patient’s illness.
Figure 4.
From Tersalvi et al.129
Irrespective of logistics, subjecting a critically ill individual to an invasive procedure does not come without its own risks. Expecting the treating cardiologist to ascribe an elevated biomarker level to a single unifying diagnosis does not only appear to be unduly challenging, given our knowledge of the multi-facetted pathophysiological contributors to acute and chronic troponin elevations but also operationally untenable. The imagined pathophysiological basis of the 4th UDMI often cannot be made to fit clinical reality.
14. Does it matter?
The case presented above highlights the uncertainties in everyday acute cardiovascular care—whilst the concepts of the 4th UDMI are pathophysiologically sound, they are challenging to define operationally. What is needed are evolutions in clinical practice that will enable patients to be placed within the pathological framework described in the 4th UDMI. This is necessary for treatments to target the relevant and dominant causative pathology, or pathologies, contributing to the cTn elevation. The future is bright and clinical landscape is changing. Powerful diagnostic techniques such as CTCA and CMR are increasingly available. What is clear is that whatever the cause of elevation cTn indicates prognosis, we just have to learn how to better answer its siren call!
Conflict of interest: T.E.K. and B.A. have no conflict of interest to declare. M.M. has interest to declare. M.M. is named as an inventor on a patent held by King’s College London for the detection of cardiac myosin-binding protein C as a biomarker of myocardial injury.
Funding
This work was supported by grants from the Medical Research Council (London, UK) (G1000737), Guy’s and St Thomas’ Charity (London, UK; R060701, R100404), British Heart Foundation (Birmingham, London; TG/15/1/31518, FS/15/13/31320), and the UK Department of Health through the National Institute for Health Research Biomedical Research Centre award to Guy’s & St Thomas’ National Health Service Foundation Trust. Dr Kaier is funded through an National Institute for Health Research clinical lectureship (CL-2019-17-006).
References
- 1.Thygesen K, Alpert JS, Jaffe AS, Chaitman BR, Bax JJ, Morrow DA, White HD.. Fourth universal definition of myocardial infarction (2018). J Am Coll Cardiol 2018;72:2231–2264. [DOI] [PubMed] [Google Scholar]
- 2.E.H.J. Bulluck et al., 10.1016/j.jacc.2020.08.009 [Google Scholar]
- 3.De Lemos J, Newby LK, Mills NL.. A proposal for modest revision of the definition of type 1 and type 2 myocardial infarction. Circulation 2019;140:1773–1775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alpert JS.The fourth edition of the universal definition of myocardial infarction. Am J Med 2018;131:1265–1266. [DOI] [PubMed] [Google Scholar]
- 5.Valentine CM, Tcheng JE, Waites T.. Translating the translation. J Am Coll Cardiol 2018;72:2668–2670. [DOI] [PubMed] [Google Scholar]
- 6.Park KC, Gaze DC, Collinson PO, Marber MS.. Cardiac troponins: from myocardial infarction to chronic disease. Cardiovasc Res 2017;113:1708–1718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD.. Third universal definition of myocardial infarction. Circulation 2012;126:2020–2035. [DOI] [PubMed] [Google Scholar]
- 8.Chapman AR, Lee KK, McAllister DA, Cullen L, Greenslade JH, Parsonage W, Worster A, Kavsak PA, Blankenberg S, Neumann J, Sörensen NA, Westermann D, Buijs MM, Verdel GJE, Pickering JW, Than MP, Twerenbold R, Badertscher P, Sabti Z, Mueller C, Anand A, Adamson P, Strachan FE, Ferry A, Sandeman D, Gray A, Body R, Keevil B, Carlton E, Greaves K, Korley FK, Metkus TS, Sandoval Y, Apple FS, Newby DE, Shah ASV, Mills NL.. Association of high-sensitivity cardiac troponin I concentration with cardiac outcomes in patients with suspected acute coronary syndrome. Jama 2017;318:1913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Alaour B, Liew F, Kaier TE.. Cardiac troponin – diagnostic problems and impact on cardiovascular disease. Ann Med 2018;50:655–665. [DOI] [PubMed] [Google Scholar]
- 10.ErikssonS, , HellmanJ, , Pettersson K.. Autoantibodies against Cardiac Troponins. N Engl J Med 2005;352:98–100. [DOI] [PubMed] [Google Scholar]
- 11.Greaser ML, Gergely J.. Purification and properties of the components from troponin. J Biol Chem 1973;248:2125–2133. [PubMed] [Google Scholar]
- 12.Jaffe AS, Vasile VC, Milone M, Saenger AK, Olson KN, Apple FS.. Diseased skeletal muscle. J Am Coll Cardiol 2011;58:1819–1824. [DOI] [PubMed] [Google Scholar]
- 13.Schmid J, Liesinger L, Birner-Gruenberger R, Stojakovic T, Scharnagl H, Dieplinger B, Asslaber M, Radl R, Beer M, Polacin M, Mair J, Szolar D, Berghold A, Quasthoff S, Binder JS, Rainer PP.. Elevated cardiac troponin T in patients with skeletal myopathies. J Am Coll Cardiol 2018;71:1540–1549. [DOI] [PubMed] [Google Scholar]
- 14.Adamczyk M, Brashear RJ, Mattingly PG.. Prevalence of autoantibodies to cardiac troponin T in healthy blood donors. Clin Chem 2009;55:1592–1593. [DOI] [PubMed] [Google Scholar]
- 15.Willeman T, Casez O, Faure P, Gauchez AS.. Evaluation of biotin interference on immunoassays: new data for troponin I, digoxin, NT-Pro-BNP, and progesterone. Clin Chem Lab Med (CCLM) 2017;55:e226–e229. [DOI] [PubMed] [Google Scholar]
- 16.Apple FS, Collinson PO, IFCC Task Force on Clinical Applications of Cardiac Biomarkers. Analytical characteristics of high-sensitivity cardiac troponin assays. Clin Chem 2012;58:54–61. [DOI] [PubMed] [Google Scholar]
- 17.Katus H.Enzyme linked immuno assay of cardiac troponin T for the detection of acute myocardial infarction in patients. J Mol Cell Cardiol 1989;21:1349–1353. [DOI] [PubMed] [Google Scholar]
- 18.Cummins B, Auckland ML, Cummins P.. Cardiac-specific troponin-l radioimmunoassay in the diagnosis of acute myocardial infarction. Am Heart J 1987;113:1333–1344. [DOI] [PubMed] [Google Scholar]
- 19.Wu AHB, Ford L.. Release of cardiac troponin in acute coronary syndromes: ischemia or necrosis? Clin Chim Acta 1999;284:161–174. [DOI] [PubMed] [Google Scholar]
- 20.Möhlenkamp S, Leineweber K, Lehmann N, Braun S, Roggenbuck U, Perrey M, Broecker-Preuss M, Budde T, Halle M, Mann K, Jöckel K-H, Erbel R, Heusch G.. Coronary atherosclerosis burden, but not transient troponin elevation, predicts long-term outcome in recreational marathon runners. Basic Res Cardiol 2014;109:391. [DOI] [PubMed] [Google Scholar]
- 21.Jaffe AS, Wu AHB.. Troponin release—reversible or irreversible injury? Should we care? Clin Chem 2012;58:148–150. [DOI] [PubMed] [Google Scholar]
- 22.White HD.Pathobiology of troponin elevations. J Am Coll Cardiol 2011;57:2406–2408. [DOI] [PubMed] [Google Scholar]
- 23.Mahajan VS, Jarolim P.. How to interpret elevated cardiac troponin levels. Circulation 2011;124:2350–2354. [DOI] [PubMed] [Google Scholar]
- 24.Marjot J, Kaier TE, Martin ED, Reji SS, Copeland O, Iqbal M, Goodson B, Hamren S, Harding SE, Marber MS.. Quantifying the release of biomarkers of myocardial necrosis from cardiac myocytes and intact myocardium. Clin Chem 2017;63:990–996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Katus HA, Remppis A, Scheffold T, Diederich KW, Kuebler W.. Intracellular compartmentation of cardiac troponin T and its release kinetics in patients with reperfused and nonreperfused myocardial infarction. Am J Cardiol 1991;67:1360–1367. [DOI] [PubMed] [Google Scholar]
- 26.Venge P, Lindahl B.. Cardiac troponin assay classification by both clinical and analytical performance characteristics: a study on outcome prediction. Clin Chem 2013;59:976–981. [DOI] [PubMed] [Google Scholar]
- 27.Shah ASV, Griffiths M, Lee KK, McAllister DA, Hunter AL, Ferry AV, Cruikshank A, Reid A, Stoddart M, Strachan F, Walker S, Collinson PO, Apple FS, Gray AJ, Fox KAA, Newby DE, Mills NL.. High sensitivity cardiac troponin and the under-diagnosis of myocardial infarction in women: prospective cohort study. BMJ 2015;350:g7873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Apple FS, Ler R, Murakami MM.. Determination of 19 cardiac troponin I and T assay 99th percentile values from a common presumably healthy population. Clin Chem 2012;58:1574–1581. [DOI] [PubMed] [Google Scholar]
- 29.Mueller T, Egger M, Peer E, Jani E, Dieplinger B.. Evaluation of sex-specific cut-off values of high-sensitivity cardiac troponin I and T assays in an emergency department setting – results from the Linz Troponin (LITROP) study. Clin Chim Acta 2018;487:66–74. [DOI] [PubMed] [Google Scholar]
- 30.Kimenai DM, Janssen EBNJ, Eggers KM, Lindahl B, den RH, Bekers O, Appelman Y, Meex SJR.. Sex-specific versus overall clinical decision limits for cardiac troponin I and T for the diagnosis of acute myocardial infarction: a systematic review. Clin Chem 2018;64:1034–1043. [DOI] [PubMed] [Google Scholar]
- 31.Lee KK, Ferry AV, Anand A, Strachan FE, Chapman AR, Kimenai DM, Meex SJR, Berry C, Findlay I, Reid A, Cruickshank A, Gray A, Collinson PO, Apple FS, McAllister DA, Maguire D, Fox KAA, Newby DE, Tuck C, Keerie C, Weir CJ, Shah ASV, Mills NL, Mills NL, Strachan FE, Tuck C, Shah ASV, Anand A, Ferry AV, Lee KK, Chapman AR, Sandeman D, Adamson PD, Stables CL, Vallejo CA, Tsanasis A, Marshall L, Stewart SD, Fujisawa T, Hautvast M, McPherson J, McKinlay L, Mills NL, Newby DE, Fox KAA, Berry C, Walker S, Weir CJ, Ford I, Mills NL, Newby DE, Gray A, Fox KAA, Berry C, Walker S, Collinson PO, Apple FS, Reid A, Cruikshank A, Findlay I, Amoils S, McAllister DA, Maguire D, Stevens J, Norrie J, Weir C, Shah ASV, Anand A, Chapman AR, Lee KK, Andrews JPM, Adamson PD, Moss A, Anwar MS, Hung J, Mills NL, Walker S, Malo J, Reid A, Cruikshank A, Collinson PO, Fischbacher CM, Croal BL, Leslie SJ, Keerie C, Parker RA, Walker A, Harkess R, Tuck C, Wackett T, Weir C, Armstrong R, Flood M, Stirling L, MacDonald C, Sadat I, Finlay F, Charles H, Linksted P, Young S, Alexander B, Duncan C.. Sex-specific thresholds of high-sensitivity troponin in patients with suspected acute coronary syndrome. J Am Coll Cardiol 2019;74:2032–2043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rubini Giménez M, Twerenbold R, Boeddinghaus J, Nestelberger T, Puelacher C, Hillinger P, Wildi K, Jaeger C, Grimm K, Heitzelmann K-F, Sabti Z, Badertscher P, Cupa J, Honegger U, Schaerli N, Kozhuharov N, Du Fay de Lavallaz J, Lopez B, Salgado E, Miró Ò, Martín-Sánchez FJ, Adrada ER, Morawiec B, Parenica J, Ganovska E, Neugebauer C, Rentsch K, Lohrmann J, Osswald S, Reichlin T, Mueller C.. Clinical effect of sex-specific cutoff values of high-sensitivity cardiac troponin T in suspected myocardial infarction. JAMA Cardiol 2016;1:912. [DOI] [PubMed] [Google Scholar]
- 33.Apple FS, Jaffe AS, Collinson P, Mockel M, Ordonez-Llanos J, Lindahl B, Hollander J, Plebani M, Than M, Chan MHM.. IFCC educational materials on selected analytical and clinical applications of high sensitivity cardiac troponin assays. Clin Biochem 2015;48:201–203. [DOI] [PubMed] [Google Scholar]
- 34.Mariathas M, Allan R, Ramamoorthy S, Olechowski B, Hinton J, Azor M, Nicholas Z, Calver A, Corbett S, Mahmoudi M, Rawlins J, Simpson I, Wilkinson J, Kwok CS, Cook P, Mamas MA, Curzen N.. True 99th centile of high sensitivity cardiac troponin for hospital patients: prospective, observational cohort study. BMJ 2019;364:l729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Linden NVD, Hilderink JM, Cornelis T, Kimenai DM, Klinkenberg LJJ, Doorn WV, Litjens EJR, Suijlen JV, Loon LV, Bekers O, Kooman JP, Meex SJR.. Twenty-four-hour biological variation profiles of cardiac troponin I in individuals with or without chronic kidney disease. Clin Chem 2017;63:1655–1656. [DOI] [PubMed] [Google Scholar]
- 36.Klinkenberg LJJ, Wildi K, Linden NVD, Kouw IWK, Niens M, Twerenbold R, Rubini Giménez M, Puelacher C, Daniel Neuhaus J, Hillinger P, Nestelberger T, Boeddinghaus J, Grimm K, Sabti Z, Bons JAP, Suijlen JV, Tan FES, Kate JT, Bekers O, Loon LV, Dieijen-Visser MV, Mueller C, Meex SJR.. Diurnal rhythm of cardiac troponin: consequences for the diagnosis of acute myocardial infarction. Clin Chem 2016;62:1602–1611. [DOI] [PubMed] [Google Scholar]
- 37.Amsterdam EA, Wenger NK, Brindis RG, Casey DE, Ganiats TG, Holmes DR, Jaffe AS, Jneid H, Kelly RF, Kontos MC, Levine GN, Liebson PR, Mukherjee D, Peterson ED, Sabatine MS, Smalling RW, Zieman SJ, Cardiology AC of, Guidelines AHATF on P, Interventions S for CA and, Surgeons S of T, Chemistry AA for C. 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2014;64:e139-228. [DOI] [PubMed] [Google Scholar]
- 38.Roffi M, Patrono C, Collet J-P, Mueller C, Valgimigli M, Andreotti F, Bax JJ, Borger MA, Brotons C, Chew DP, Gencer B, Hasenfuss G, Kjeldsen K, Lancellotti P, Landmesser U, Mehilli J, Mukherjee D, Storey RF, Windecker S.. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J 2016;37:267–315. [DOI] [PubMed] [Google Scholar]
- 39.Reichlin T, Irfan A, Twerenbold R, Reiter M, Hochholzer W, Burkhalter H, Bassetti S, Steuer S, Winkler K, Peter F, Meissner J, Haaf P, Potocki M, Drexler B, Osswald S, Mueller C.. Utility of absolute and relative changes in cardiac troponin concentrations in the early diagnosis of acute myocardial infarction. Circulation 2011;124:136–145. [DOI] [PubMed] [Google Scholar]
- 40.Klinkenberg LJJ, Dijk JV, Tan FES, Loon LV, Dieijen-Visser MV, Meex SJR.. Circulating cardiac troponin T exhibits a diurnal rhythm. J Am Coll Cardiol 2014;63:1788–1795. [DOI] [PubMed] [Google Scholar]
- 41.Dupuy A-M, Lozano C, Badiou S, Bargnoux A-S, Kuster N, Cristol J-P.. Biological variability of hs-cardiac troponin T on the Roche Cobas 8000/e602® immunoanalyzer. Clin Chim Acta 2013;425:62–63. [DOI] [PubMed] [Google Scholar]
- 42.Vasile VC, Saenger AK, Kroning JM, Jaffe AS.. Biological and analytical variability of a novel high-sensitivity cardiac troponin T assay. Clin Chem 2010;56:1086–1090. [DOI] [PubMed] [Google Scholar]
- 43.Reichlin T, Twerenbold R, Wildi K, Gimenez MR, Bergsma N, Haaf P, Druey S, Puelacher C, Moehring B, Freese M, Stelzig C, Krivoshei L, Hillinger P, Jager C, Herrmann T, Kreutzinger P, Radosavac M, Weidmann ZM, Pershyna K, Honegger U, Wagener M, Vuillomenet T, Campodarve I, Bingisser R, Miro O, Rentsch K, Bassetti S, Osswald S, Mueller C.. Prospective validation of a 1-hour algorithm to rule-out and rule-in acute myocardial infarction using a high-sensitivity cardiac troponin T assay. CMAJ 2015;187:E243–E252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Marjot J, Kaier TE, Henderson K, Hunter L, Marber MS, Perera D.. A single centre prospective cohort study addressing the effect of a rule-in/rule-out troponin algorithm on routine clinical practice. Eur Heart J Acute Cardiovasc Care 2017;1:2048872617746850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pickering JW, Greenslade JH, Cullen L, Flaws D, Parsonage W, Aldous S, George P, Worster A, Kavsak PA, Than MP.. Assessment of the European Society of Cardiology 0 hour/1 hour algorithm to rule out and rule in acute myocardial infarction. Circulation 2016;134:1532–1541. [DOI] [PubMed] [Google Scholar]
- 46.Reichlin T, Schindler C, Drexler B, Twerenbold R, Reiter M, Zellweger C, Moehring B, Ziller R, Hoeller R, Rubini Giménez M, Haaf P, Potocki M, Wildi K, Balmelli C, Freese M, Stelzig C, Freidank H, Osswald S, Mueller C.. One-hour rule-out and rule-in of acute myocardial infarction using high-sensitivity cardiac troponin T. Arch Intern Med 2012;172:1211–1218. [DOI] [PubMed] [Google Scholar]
- 47.Mueller C, Giannitsis E, Christ M, Ordóñez-Llanos J, deFilippi C, McCord J, Body R, Panteghini M, Jernberg T, Plebani M, Verschuren F, French J, Christenson R, Weiser S, Bendig G, Dilba P, Lindahl B, Twerenbold R, Katus HA, Popp S, Santalo-Bel M, Nowak RM, Horner D, Dolci A, Zaninotto M, Manara A, Menassanch-Volker S, Jarausch J, Zaugg C.. Multicenter evaluation of a 0-hour/1-hour algorithm in the diagnosis of myocardial infarction with high-sensitivity cardiac troponin T. Ann Emerg Med 2016;68:76–87.e4. [DOI] [PubMed] [Google Scholar]
- 48.Rubini Giménez M, Twerenbold R, Jaeger C, Schindler C, Puelacher C, Wildi K, Reichlin T, Haaf P, Merk S, Honegger U, Wagener M, Druey S, Schumacher C, Krivoshei L, Hillinger P, Herrmann T, Campodarve I, Rentsch K, Bassetti S, Osswald S, Mueller C.. One-hour rule-in and rule-out of acute myocardial infarction using high-sensitivity cardiac troponin I. Am J Med 2015;128:861–870.e4. [DOI] [PubMed] [Google Scholar]
- 49.Jaeger C, Wildi K, Twerenbold R, Reichlin T, Rubini Gimenez M, Neuhaus JD, Grimm K, Boeddinghaus J, Hillinger P, Nestelberger T, Singeisen H, Gugala M, Pretre G, Puelacher C, Wagener M, Honegger U, Schumacher C, Moreno Weidmann Z, Kreutzinger P, Krivoshei L, Freese M, Stelzig C, Dietsche S, Ernst S, Rentsch K, Osswald S, Mueller C.. One-hour rule-in and rule-out of acute myocardial infarction using high-sensitivity cardiac troponin I. Am Heart J Elsevier Inc 2016;171:92–102.e5. [DOI] [PubMed] [Google Scholar]
- 50.Twerenbold R, Badertscher P, Boeddinghaus J, Nestelberger T, Wildi K, Puelacher C, Sabti Z, Rubini Gimenez M, Tschirky S, Fay de Lavallaz JD, Kozhuharov N, Sazgary L, Mueller D, Breidthardt T, Strebel I, Flores Widmer D, Shrestha S, Miró Ò, Martín-Sánchez FJ, Morawiec B, Parenica J, Geigy N, Keller DI, Rentsch K, Eckardstein AV, Osswald S, Reichlin T, Mueller C.. 0/1-Hour triage algorithm for myocardial infarction in patients with renal dysfunction. Circulation 2018;137:436–451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.van der Linden N, Wildi K, Twerenbold R, Pickering JW, Than M, Cullen L, Greenslade J, Parsonage W, Nestelberger T, Boeddinghaus J, Badertscher P, Rubini Giménez M, Klinkenberg LJJ, Bekers O, Schöni A, Keller DI, Sabti Z, Puelacher C, Cupa J, Schumacher L, Kozhuharov N, Grimm K, Shrestha S, Flores D, Freese M, Stelzig C, Strebel I, Miró Ò, Rentsch K, Morawiec B, Kawecki D, Kloos W, Lohrmann J, Richards AM, Troughton R, Pemberton C, Osswald S, van Dieijen-Visser MP, Mingels AM, Reichlin T, Meex SJR, Mueller C.. Combining high sensitivity cardiac troponin I and cardiac troponin T in the early diagnosis of acute myocardial infarction. Circulation 2018;138:989–999. [DOI] [PubMed] [Google Scholar]
- 52.Kaier TE, Twerenbold R, Puelacher C, Marjot J, Imambaccus N, Boeddinghaus J, Nestelberger T, Badertscher P, Sabti Z, Gimenez MR, Wildi K, Hillinger P, Grimm K, Loeffel S, Shrestha S, Widmer DF, Cupa J, Kozhuharov N, Miró Ò, Martin-Sanchez FJ, Morawiec B, Rentsch K, Lohrmann J, Kloos W, Osswald S, Reichlin T, Weber E, Marber M, Mueller C.. Direct comparison of cardiac myosin-binding protein C with cardiac troponins for the early diagnosis of acute myocardial infarction. Circulation 2017;136:1495–1508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Druey S, Wildi K, Twerenbold R, Jaeger C, Reichlin T, Haaf P, Gimenez MR, Puelacher C, Wagener M, Radosavac M, Honegger U, Schumacher C, Delfine V, Kreutzinger P, Herrmann T, Weidmann ZM, Krivoshei L, Freese M, Stelzig C, Isenschmid C, Bassetti S, Rentsch K, Osswald S, Mueller C.. Early rule-out and rule-in of myocardial infarction using sensitive cardiac Troponin I. Int J Cardiol 2015;195:163–170. [DOI] [PubMed] [Google Scholar]
- 54.Nestelberger T, Wildi K, Boeddinghaus J, Twerenbold R, Reichlin T, Gimenez MR, Puelacher C, Jaeger C, Grimm K, Sabti Z, Hillinger P, Kozhuharov N, de Lavallaz Du FJ, Pinck F, Lopez B, Salgado E, Miró Ò, Bingisser R, Lohrmann J, Osswald S, Mueller C.. Characterization of the observe zone of the ESC 2015 high-sensitivity cardiac troponin 0h/1h-algorithm for the early diagnosis of acute myocardial infarction. Int J Cardiol 2016;207:238–245. [DOI] [PubMed] [Google Scholar]
- 55.Shortt C, Ma J, Clayton N, Sherbino J, Whitlock R, Pare G, Hill SA, McQueen M, Mehta SR, Devereaux PJ, Worster A, Kavsak PA.. Rule-in and rule-out of myocardial infarction using cardiac troponin and glycemic biomarkers in patients with symptoms suggestive of acute coronary syndrome. Clin Chem 2017;63:403–414. [DOI] [PubMed] [Google Scholar]
- 56.Body R, Carley S, Mcdowell G, Jaffe AS, France M, Bs MB, Cruickshank K, Wibberley C, Nuttall M, Mackway-Jones K, Bc H.. Rapid exclusion of acute myocardial infarction in patients with undetectable troponin using a high-sensitivity assay rapid exclusion of acute myocardial infarction in patients with undetectable troponin using a high-sensitivity assay. Jac Elsevier Inc 2011;58:1332–1339. [DOI] [PubMed] [Google Scholar]
- 57.Shiozaki M, Inoue K, Suwa S, Lee CC, Chikata Y, Ishiura J, Kimura Y, Fukuda K, Tamura H, Fujiwara Y, Sumiyoshi M, Daida H.. Utility of the 0-hour/1-hour high-sensitivity cardiac troponin T algorithm in Asian patients with suspected non-ST elevation myocardial infarction. Int J Cardiol 2017;249:32–35. [DOI] [PubMed] [Google Scholar]
- 58.Mokhtari A, Borna C, Gilje P, Tydén P, Lindahl B, Nilsson H-J, Khoshnood A, Björk J, Ekelund U.. A 1-h combination algorithm allows fast rule-out and rule-in of major adverse cardiac events. J Am Coll Cardiol 2016;67:1531–1540. [DOI] [PubMed] [Google Scholar]
- 59.Reichlin T, Cullen L, Parsonage WA, Greenslade J, Twerenbold R, Moehring B, Wildi K, Mueller S, Zellweger C, Mosimann T, Rubini Gimenez M, Rentsch K, Osswald S, Müller C.. Two-hour algorithm for triage toward rule-out and rule-in of acute myocardial infarction using high-sensitivity cardiac troponin T. Am J Med 2015;128:369–379.e4. [DOI] [PubMed] [Google Scholar]
- 60.Boeddinghaus J, Reichlin T, Cullen L, Greenslade JH, Parsonage WA, Hammett C, Pickering JW, Hawkins T, Aldous S, Twerenbold R, Wildi K, Nestelberger T, Grimm K, Rubini Giménez M, Puelacher C, Kern V, Rentsch K, Than M, Mueller C.. Two-hour algorithm for triage toward rule-out and rule-in of acute myocardial infarction by use of high-sensitivity cardiac troponin I. Clin Chem 2016;62:494–504. [DOI] [PubMed] [Google Scholar]
- 61.Eggers KM, Aldous S, Greenslade JH, Johnston N, Lindahl B, Parsonage WA, Pickering JW, Than M, Cullen L.. Two-hour diagnostic algorithms for early assessment of patients with acute chest pain – implications of lowering the cardiac troponin I cut-off to the 97.5th percentile. Clin Chim Acta 2015;445:19–24. [DOI] [PubMed] [Google Scholar]
- 62.Lindahl B, Jernberg T, Badertscher P, Boeddinghaus J, Eggers KM, Frick M, Giménez Linder RM, Ljung R, Martinsson L, Melki A, Nestelberger D, Rentsch T, Reichlin K, Sabti T, Schubera Z, Svensson M, Twerenbold P, Wildi R, Mueller K.. C. An algorithm for rule-in and rule-out of acute myocardial infarction using a novel troponin I assay. Heart Br Card Soc 2017;103:125–131. [DOI] [PubMed] [Google Scholar]
- 63.McRae AD, Innes G, Graham M, Lang E, Andruchow JE, Yang H, Ji Y, Vatanpour S, Southern DA, Wang D, Seiden-Long I, DeKoning L, Kavsak P.. Comparative evaluation of 2-hour rapid diagnostic algorithms for acute myocardial infarction using high-sensitivity cardiac troponin T. Can J Cardiol the Authors 2017;33:1006–1012. [DOI] [PubMed] [Google Scholar]
- 64.Dedic A, Lubbers MM, Schaap J, Lammers J, Lamfers EJ, Rensing BJ, Braam RL, Nathoe HM, Post JC, Nielen T, Beelen D, Cocq d’Armandville M-CL, Rood PPM, Schultz CJ, Moelker A, Ouhlous M, Boersma E, Nieman K.. Coronary CT angiography for suspected ACS in the era of high-sensitivity troponins. J Am Coll Cardiol 2016;67:16–26. [DOI] [PubMed] [Google Scholar]
- 65.Smulders MW, Kietselaer BLJH, Wildberger JE, Dagnelie PC, Brunner-La Rocca H-P, Mingels AMA, Cauteren YV, Theunissen RALJ, Post MJ, Schalla S, Kuijk SV, Das M, Kim RJ, Crijns HJGM, Bekkers SCAM.. Initial imaging-guided strategy versus routine care in patients with non-ST-segment elevation myocardial infarction. J Am Coll Cardiol 2019;74:2466–2477. [DOI] [PubMed] [Google Scholar]
- 66.Linde JJ, Kelbæk H, Hansen TF, Sigvardsen PE, Torp-Pedersen C, Bech J, Heitmann M, Nielsen OW, Høfsten D, Kühl JT, Raymond IE, Kristiansen OP, Svendsen IH, Vall-Lamora MHD, Kragelund C, de Knegt M, Hove JD, Jørgensen T, Fornitz GG, Steffensen R, Jurlander B, Abdulla J, Lyngbæk S, Elming H, Therkelsen SK, Jørgensen E, Kløvgaard L, Bang LE, Hansen PR, Helqvist S, Galatius S, Pedersen F, Abildgaard U, Clemmensen P, Saunamäki K, Holmvang L, Engstrøm T, Gislason G, Køber LV, Kofoed KF.. Coronary CT angiography in patients with non-ST-segment elevation acute coronary syndrome. J Am Coll Cardiol 2020;75:453–463. [DOI] [PubMed] [Google Scholar]
- 67.Collet J-P, Thiele H, Barbato E, Barthélémy O, Bauersachs J, Bhatt DL, Dendale P, Dorobantu M, Edvardsen T, Folliguet T, Gale CP, Gilard M, Jobs A, Jüni P, Lambrinou E, Lewis BS, Mehilli J, Meliga E, Merkely B, Mueller C, Roffi M, Rutten FH, Sibbing D, Siontis GCM, Kastrati A, Mamas MA, Aboyans V, Angiolillo DJ, Bueno H, ESC Scientific Document Group. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J 2020. doi:10.1093/eurheartj/ehaa575. [DOI] [PubMed] [Google Scholar]
- 68.Neumann JT, Twerenbold R, Ojeda F, Sörensen NA, Chapman AR, Shah ASV, Anand A, Boeddinghaus J, Nestelberger T, Badertscher P, Mokhtari A, Pickering JW, Troughton RW, Greenslade J, Parsonage W, Mueller-Hennessen M, Gori T, Jernberg T, Morris N, Liebetrau C, Hamm C, Katus HA, Münzel T, Landmesser U, Salomaa V, Iacoviello L, Ferrario MM, Giampaoli S, Kee F, Thorand B, Peters A, Borchini R, Jørgensen T, Söderberg S, Sans S, Tunstall-Pedoe H, Kuulasmaa K, Renné T, Lackner KJ, Worster A, Body R, Ekelund U, Kavsak PA, Keller T, Lindahl B, Wild P, Giannitsis E, Than M, Cullen LA, Mills NL, Mueller C, Zeller T, Westermann D, Blankenberg S.. Application of high-sensitivity troponin in suspected myocardial infarction. N Engl J Med 2019;380:2529–2540. [DOI] [PubMed] [Google Scholar]
- 69.Chapman AR, Adamson PD, Shah ASV, Anand A, Strachan FE, Ferry AV, Ken Lee K, Berry C, Findlay I, Cruikshank A, Reid A, Gray A, Collinson PO, Apple F, McAllister DA, Maguire D, Fox KAA, Vallejos CA, Keerie C, Weir CJ, Newby DE, Mills NL, Tuck C, Bularga A, Wereski R, Sandeman D, Stables CL, Tsanasis A, Marshall L, Stewart SD, Fujisawa T, Hautvast M, McPherson J, McKinlay L, Walker S, Ford I, Walker S, Amoils S, Stevens J, Norrie J, Andrews J, Adamson P, Moss A, Anwar M, Hung J, Walker S, Malo J, Fischbacher C, Croal B, Leslie SJ, Parker R, Walker A, Harkess R, Tuck C, Wackett T, Armstrong R, Flood M, Stirling L, MacDonald C, Sadat I, Finlay F, Charles H, Linksted P, Young S, Alexander B, Duncan C.. High-sensitivity cardiac troponin and the universal definition of myocardial infarction. Circulation 2020;141:161–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hartikainen TS, Sörensen NA, Haller PM, Goßling A, Lehmacher J, Zeller T, Blankenberg S, Westermann D, Neumann JT.. Clinical application of the 4th universal definition of myocardial infarction. Eur Heart J 2020;41:2209–2216. [DOI] [PubMed] [Google Scholar]
- 71.Chapman AR, Shah ASV, Lee KK, Anand A, Francis O, Adamson P, McAllister DA, Strachan FE, Newby DE, Mills NL.. Long-term outcomes in patients with type 2 myocardial infarction and myocardial injury. Circulation 2018;137:1236–1245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.DeFilippis AP, Chapman AR, Mills NL, Lemos JD, Arbab-Zadeh A, Newby LK, Morrow DA.. Assessment and treatment of patients with type 2 myocardial infarction and acute non-ischemic myocardial injury. Circulation 2019;140:1661–1678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Apple FS, Sandoval Y, Jaffe AS, Ordonez-Llanos J; IFCC Task Force on Clinical Applications of Cardiac Bio-Markers. Cardiac troponin assays: guide to understanding analytical characteristics and their impact on clinical care. Clin Chem 2017;63:73–81. [DOI] [PubMed] [Google Scholar]
- 74.Than MP, Aldous SJ, Troughton RW, Pemberton CJ, Richards AM, Frampton CMA, Florkowski CM, George PM, Bailey S, Young JM, Cullen L, Greenslade JH, Parsonage WA, Everett BM, Peacock WF, Jaffe AS, Pickering JW.. Detectable high-sensitivity cardiac troponin within the population reference interval conveys high 5-year cardiovascular risk: an observational study. Clin Chem 2018;64:1044–1053. [DOI] [PubMed] [Google Scholar]
- 75.Sandoval Y, Bielinski SJ, Daniels LB, Blaha MJ, Michos ED, DeFilippis AP, Szklo M, deFilippi C, Larson NB, Decker PA, Jaffe AS.. Atherosclerotic cardiovascular disease risk stratification based on measurements of troponin and coronary artery calcium. J Am Coll Cardiol 2020;76:357–370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Olivetti G, Abbi R, Quaini F, Kajstura J, Cheng W, Nitahara JA, Quaini E, Di Loreto C, Beltrami CA, Krajewski S, Reed JC, Anversa P.. Apoptosis in the failing human heart. N Engl J Med 1997;336:1131–1141. [DOI] [PubMed] [Google Scholar]
- 77.Narula J, Haider N, Virmani R, DiSalvo TG, Kolodgie FD, Hajjar RJ, Schmidt U, Semigran MJ, Dec GW, Khaw B-A.. Apoptosis in myocytes in end-stage heart failure. N Engl J Med 1996;335:1182–1189. [DOI] [PubMed] [Google Scholar]
- 78.Kociol RD, Pang PS, Gheorghiade M, Fonarow GC, O’Connor CM, Felker GM.. Troponin elevation in heart failure: prevalence, mechanisms, and clinical implications. J Am Coll Cardiol 2010;56:1071–1078 [DOI] [PubMed] [Google Scholar]
- 79.Hessel MHM, Atsma DE, Valk EVD, Bax WH, Schalij MJ, Laarse AVD.. Release of cardiac troponin I from viable cardiomyocytes is mediated by integrin stimulation. Pflugers Arch 2008;455:979–986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Sutton M, Sharpe N.. Left ventricular remodeling after myocardial infarction: pathophysiology and therapy. Circulation 2000;101:2981–2988. [DOI] [PubMed] [Google Scholar]
- 81.Vatner SF.Reduced subendocardial myocardial perfusion as one mechanism for congestive heart failure. Am J Cardiol 1988;62:94E–98E. [DOI] [PubMed] [Google Scholar]
- 82.Packer M.The neurohormonal hypothesis: a theory to explain the mechanism of disease progression in heart failure. J Am Coll Cardiol 1992;20:248–254. [DOI] [PubMed] [Google Scholar]
- 83.Tan LB, Jalil JE, Pick R, Janicki JS, Weber KT.. Cardiac myocyte necrosis induced by angiotensin II. Circ Res 1991;69:1185–1195. [DOI] [PubMed] [Google Scholar]
- 84.Mann DL, Kent RL, Parsons B, Cooper G.. Adrenergic effects on the biology of the adult mammalian cardiocyte. Circulation 1992;85:790–804. [DOI] [PubMed] [Google Scholar]
- 85.Schaper J, Froede R, Hein S, Buck A, Hashizume H, Speiser B, Friedl A, Bleese N.. Impairment of the myocardial ultrastructure and changes of the cytoskeleton in dilated cardiomyopathy. Circulation 1991;83:504–514. [DOI] [PubMed] [Google Scholar]
- 86.Logeart D, Beyne P, Cusson C, Tokmakova M, Leban M, Guiti C, Bourgoin P, Solal AC.. Evidence of cardiac myolysis in severe nonischemic heart failure and the potential role of increased wall strain. Am Heart J 2001;141:247–253. [DOI] [PubMed] [Google Scholar]
- 87.Knuuti J, Wijns W, Saraste A, Capodanno D, Barbato E, Funck-Brentano C, Prescott E, Storey RF, Deaton C, Cuisset T, Agewall S, Dickstein K, Edvardsen T, Escaned J, Gersh BJ, Svitil P, Gilard M, Hasdai D, Hatala R, Mahfoud F, Masip J, Muneretto C, Valgimigli M, Achenbach S, Bax JJ, Neumann F-J, Sechtem U, Banning AP, Bonaros N, Bueno H, Bugiardini R, Chieffo A, Crea F, Czerny M, Delgado V, Dendale P, Flachskampf FA, Gohlke H, Grove EL, James S, Katritsis D, Landmesser U, Lettino M, Matter CM, Nathoe H, Niessner A, Patrono C, Petronio AS, Pettersen SE, Piccolo R, Piepoli MF, Popescu BA, Räber L, Richter DJ, Roffi M, Roithinger FX, Shlyakhto E, Sibbing D, Silber S, Simpson IA, Sousa-Uva M, Vardas P, Witkowski A, Zamorano JL, Achenbach S, Agewall S, Barbato E, Bax JJ, Capodanno D, Cuisset T, Deaton C, Dickstein K, Edvardsen T, Escaned J, Funck-Brentano C, Gersh BJ, Gilard M, Hasdai D, Hatala R, Mahfoud F, Masip J, Muneretto C, Prescott E, Saraste A, Storey RF, Svitil P, Valgimigli M, Windecker S, Aboyans V, Baigent C, Collet J-P, Dean V, Delgado V, Fitzsimons D, Gale CP, Grobbee D, Halvorsen S, Hindricks G, Iung B, Jüni P, Katus HA, Landmesser U, Leclercq C, Lettino M, Lewis BS, Merkely B, Mueller C, Petersen S, Petronio AS, Richter DJ, Roffi M, Shlyakhto E, Simpson IA, Sousa-Uva M, Touyz RM, Benkhedda S, Metzler B, Sujayeva V, Cosyns B, Kusljugic Z, Velchev V, Panayi G, Kala P, Haahr-Pedersen SA, Kabil H, Ainla T, Kaukonen T, Cayla G, Pagava Z, Woehrle J, Kanakakis J, Tóth K, Gudnason T, Peace A, Aronson D, Riccio C, Elezi S, Mirrakhimov E, Hansone S, Sarkis A, Babarskiene R, Beissel J, Maempel AJC, Revenco V, de Grooth GJ, Pejkov H, Juliebø V, Lipiec P, Santos J, Chioncel O, Duplyakov D, Bertelli L, Dikic AD, Studenčan M, Bunc M, Alfonso F, Bäck M, Zellweger M, Addad F, Yildirir A, Sirenko Y, Clapp B, ESC Scientific Document Group. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J 2020;41:407–477. [DOI] [PubMed] [Google Scholar]
- 88.Brown AJ, Shah ASV, West NEJ, Costopoulos C, Orzalkiewicz M, Newby DE, Bennett MR, Mills NL, Calvert PA.. High-sensitivity troponin I is associated with high-risk plaque and MACE in stable coronary artery disease. JACC Cardiovasc Imaging 2017;10:1200–1203. [DOI] [PubMed] [Google Scholar]
- 89.Omland T, Lemos JD, Sabatine MS, Christophi CA, Rice MM, Jablonski KA, Tjora S, Domanski MJ, Gersh BJ, Rouleau JL, Pfeffer MA, Braunwald E.. A sensitive cardiac troponin T assay in stable coronary artery disease. N Engl J Med 2009;361:2538–2547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Beatty AL, Ku IA, Christenson RH, DeFilippi CR, Schiller NB, Whooley MA.. High-sensitivity cardiac troponin t levels and secondary events in outpatients with coronary heart disease from the heart and soul study. JAMA Intern Med 2013;173:763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Oemrawsingh RM, Cheng JM, García-García M, Kardys I, Schaik RV, Regar E, Geuns R-JV, Serruys PW, Boersma E, Akkerhuis KM.. High-sensitivity troponin T in relation to coronary plaque characteristics in patients with stable coronary artery disease; results of the ATHEROREMO-IVUS study. Atherosclerosis 2016;247:135–141. [DOI] [PubMed] [Google Scholar]
- 92.Neilan TG, Januzzi JL, Lee-Lewandrowski E, Ton-Nu TT, Yoerger DM, Jassal DS, Lewandrowski KB, Siegel AJ, Marshall JE, Douglas PS, Lawlor D, Picard MH, Wood MJ.. Myocardial injury and ventricular dysfunction related to training levels among nonelite participants in the Boston Marathon. Circulation 2006;114:2325–2333. [DOI] [PubMed] [Google Scholar]
- 93.Shave R, Baggish A, George K, Wood M, Scharhag J, Whyte G, Gaze D, Thompson PD.. Exercise-induced cardiac troponin elevation: evidence, mechanisms, and implications. J Am Coll Cardiol 2010;56:169–176. [DOI] [PubMed] [Google Scholar]
- 94.Shave R, George KP, Atkinson G, Hart E, Middleton N, Whyte G, Gaze D, Collinson PO.. Exercise-induced cardiac troponin T release: a meta-analysis. Med Sci Sports Exerc 2007;39:2099–2106. [DOI] [PubMed] [Google Scholar]
- 95.Mousavi N, Czarnecki A, Kumar K, Fallah-Rad N, Lytwyn M, Han S-Y, Francis A, Walker JR, Kirkpatrick IDC, Neilan TG, Sharma S, Jassal DS.. Relation of biomarkers and cardiac magnetic resonance imaging after marathon running. Am J Cardiol 2009. [DOI] [PubMed] [Google Scholar]
- 96.Eijsvogels TMH, Hoogerwerf MD, Oudegeest-Sander MH, Hopman MTE, Thijssen DHJ.. The impact of exercise intensity on cardiac troponin I release. Int J Cardiol 2014;171:e3–e4. [DOI] [PubMed] [Google Scholar]
- 97.Skadberg Ø, Kleiven Ø, Bjørkavoll-Bergseth M, Melberg T, Bergseth R, Selvåg J, Auestad B, Greve OJ, Dickstein K, Aarsland T, Ørn S.. Highly increased troponin I levels following high-intensity endurance cycling may detect subclinical coronary artery disease in presumably healthy leisure sport cyclists: the North Sea Race Endurance Exercise Study (NEEDED) 2013. Eur J Prev Cardiol 2017;1:2047487317693130. [DOI] [PubMed] [Google Scholar]
- 98.Gresslien T, Agewall S.. Troponin and exercise. Int J Cardiol 2016;221:609–621. [DOI] [PubMed] [Google Scholar]
- 99.Legaz-Arrese A, George K, Carranza-García LE, Munguía-Izquierdo D, Moros-García T, Serrano-Ostáriz E.. The impact of exercise intensity on the release of cardiac biomarkers in marathon runners. Eur J Appl Physiol 2011;111:2961–2967. [DOI] [PubMed] [Google Scholar]
- 100.White HD, Tonkin A, Simes J, Stewart R, Mann K, Thompson P, Colquhoun D, West M, Nestel P, Sullivan D, Keech AC, Hunt D, Blankenberg S.. Association of contemporary sensitive troponin i levels at baseline and change at 1 year with long-term coronary events following myocardial infarction or unstable angina: results from the LIPID study (Long-Term Intervention with Pravastatin in Ischaemic Disease). J Am Coll Cardiol 2014;63:345–354. [DOI] [PubMed] [Google Scholar]
- 101.Fortescue EB, Shin AY, Greenes DS, Mannix RC, Agarwal S, Feldman BJ, Shah MI, Rifai N, Landzberg MJ, Newburger JW, Almond CSD.. Cardiac troponin increases among runners in the Boston marathon. Ann Emerg Med 2007;49:137–143.e1. [DOI] [PubMed] [Google Scholar]
- 102.Eijsvogels TMH, Hoogerwerf MD, Maessen MFH, Seeger JPH, George KP, Hopman MTE, Thijssen DHJ.. Predictors of cardiac troponin release after a marathon. J Sci Med Sport 2015;18:88–92. [DOI] [PubMed] [Google Scholar]
- 103.Hanssen H, Keithahn A, Hertel G, Drexel V, Stern H, Schuster T, Lorang D, Beer AJ, Schmidt-Trucksäss A, Nickel T, Weis M, Botnar R, Schwaiger M, Halle M.. Magnetic resonance imaging of myocardial injury and ventricular torsion after marathon running. Clin Sci 2011;120:143–152. [DOI] [PubMed] [Google Scholar]
- 104.O’Hanlon R, Wilson M, Wage R, Smith G, Alpendurada FD, Wong J, Dahl A, Oxborough D, Godfrey R, Sharma S, Roughton M, George K, Pennell DJ, Whyte G, Prasad SK.. Troponin release following endurance exercise: is inflammation the cause? A cardiovascular magnetic resonance study. J Cardiovasc Magn Reson 2010;12: 38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Scharhag J, Urhausen A, Schneider G, Herrmann M, Schumacher K, Haschke M, Krieg A, Meyer T, Herrmann W, Kindermann W.. Reproducibility and clinical significance of exercise-induced increases in cardiac troponins and N-terminal pro brain natriuretic peptide in endurance athletes. Eur J Prev Cardiol 2006;13:388–397. [DOI] [PubMed] [Google Scholar]
- 106.Schelbert EB, Hsu L-Y, Anderson SA, Mohanty BD, Karim SM, Kellman P, Aletras AH, Arai AE.. Late gadolinium-enhancement cardiac magnetic resonance identifies postinfarction myocardial fibrosis and the border zone at the near cellular level in ex vivo rat heart. Circ Cardiovasc Imaging 2010;3:743–752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.McManus DD, Gore J, Yarzebski J, Spencer F, Lessard D, Goldberg RJ.. Recent trends in the incidence, treatment, and outcomes of patients with STEMI and NSTEMI. Am J Med 2011;124:40–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Toutouzas K, Benetos G, Karanasos A, Chatzizisis YS, Giannopoulos AA, Tousoulis D.. Vulnerable plaque imaging: updates on new pathobiological mechanisms. Eur Heart J 2015;36:3147–3154. [DOI] [PubMed] [Google Scholar]
- 109.Stone GW, Maehara A, Lansky AJ, B de B, Cristea E, Mintz GS, Mehran R, McPherson J, Farhat N, Marso SP, Parise H, Templin B, White R, Zhang Z, Serruys PW.. A prospective natural-history study of coronary atherosclerosis. N Engl J Med 2011;364:226–235. [DOI] [PubMed] [Google Scholar]
- 110.Costopoulos C, Brown AJ, Teng Z, Hoole SP, West NEJ, Samady H, Bennett MR.. Intravascular ultrasound and optical coherence tomography imaging of coronary atherosclerosis. Int J Cardiovasc Imaging 2016;32:189–200. [DOI] [PubMed] [Google Scholar]
- 111.Kilic ID, Caiazzo G, Fabris E, Serdoz R, Abou-Sherif S, Madden S, Moreno PR, Goldstein J, Di Mario C.. Near-infrared spectroscopy-intravascular ultrasound: scientific basis and clinical applications. Eur Heart J Cardiovasc Imaging 2015;16:1299–1306. [DOI] [PubMed] [Google Scholar]
- 112.Uemura S, Ishigami K, Soeda T, Okayama S, Sung JH, Nakagawa H, Somekawa S, Takeda Y, Kawata H, Horii M, Saito Y.. Thin-cap fibroatheroma and microchannel findings in optical coherence tomography correlate with subsequent progression of coronary atheromatous plaques. Eur Heart J 2012;33:78–85. [DOI] [PubMed] [Google Scholar]
- 113.Tearney GJ, Regar E, Akasaka T, Adriaenssens T, Barlis P, Bezerra HG, Bouma B, Bruining N, Cho J-M, Chowdhary S, Costa MA, de Silva R, Dijkstra J, Di Mario C, Dudeck D, Falk E, Feldman MD, Fitzgerald P, Garcia H, Gonzalo N, Granada JF, Guagliumi G, Holm NR, Honda Y, Ikeno F, Kawasaki M, Kochman J, Koltowski L, Kubo T, Kume T, Kyono H, Lam CCS, Lamouche G, Lee DP, Leon MB, Maehara A, Manfrini O, Mintz GS, Mizuno K, Morel M-A, Nadkarni S, Okura H, Otake H, Pietrasik A, Prati F, Räber L, Radu MD, Rieber J, Riga M, Rollins A, Rosenberg M, Sirbu V, Serruys PWJC, Shimada K, Shinke T, Shite J, Siegel E, Sonada S, Suter M, Takarada S, Tanaka A, Terashima M, Troels T, Uemura S, Ughi GJ, van Beusekom HMM, van der Steen AFW, van Es G-A, van Soest G, Virmani R, Waxman S, Weissman NJ, Weisz G.. Consensus standards for acquisition, measurement, and reporting of intravascular optical coherence tomography studies. J Am Coll Cardiol 2012;59:1058–1072. [DOI] [PubMed] [Google Scholar]
- 114.Prati F, Regar E, Mintz GS, Arbustini E, Di Mario C, Jang I-K, Akasaka T, Costa M, Guagliumi G, Grube E, Ozaki Y, Pinto F, Serruys PWJ, for the Expert’s OCT Review Document. Expert review document on methodology, terminology, and clinical applications of optical coherence tomography: physical principles, methodology of image acquisition, and clinical application for assessment of coronary arteries and atherosclerosis. Eur Heart J 2010;31:401–415. [DOI] [PubMed] [Google Scholar]
- 115.Tian J, Ren X, Vergallo R, Xing L, Yu H, Jia H, Soeda T, McNulty I, Hu S, Lee H, Yu B, Jang I-K.. Distinct morphological features of ruptured culprit plaque for acute coronary events compared to those with silent rupture and thin-cap fibroatheroma. J Am Coll Cardiol 2014;63:2209–2216. [DOI] [PubMed] [Google Scholar]
- 116.Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, Bueno H, Caforio ALP, Crea F, Goudevenos JA, Halvorsen S, Hindricks G, Kastrati A, Lenzen MJ, Prescott E, Roffi M, Valgimigli M, Varenhorst C, Vranckx P, Widimský P, Collet J-P, Kristensen SD, Aboyans V, Baumbach A, Bugiardini R, Coman IM, Delgado V, Fitzsimons D, Gaemperli O, Gershlick AH, Gielen S, Harjola V-P, Katus HA, Knuuti J, Kolh P, Leclercq C, Lip GYH, Morais J, Neskovic AN, Neumann F-J, Niessner A, Piepoli MF, Richter DJ, Shlyakhto E, Simpson IA, Steg PG, Terkelsen CJ, Thygesen K, Windecker S, Zamorano JL, Zeymer U, Windecker S, Aboyans V, Agewall S, Barbato E, Bueno H, Coca A, Collet J-P, Coman IM, Dean V, Delgado V, Fitzsimons D, Gaemperli O, Hindricks G, Iung B, Jüni P, Katus HA, Knuuti J, Lancellotti P, Leclercq C, McDonagh T, Piepoli MF, Ponikowski P, Richter DJ, Roffi M, Shlyakhto E, Simpson IA, Zamorano JL, Chettibi M, Hayrapetyan HG, Metzler B, Ibrahimov F, Sujayeva V, Beauloye C, Dizdarevic-Hudic L, Karamfiloff K, Skoric B, Antoniades L, Tousek P, Terkelsen PJ, Shaheen SM, Marandi T, Niemelä M, Kedev S, Gilard M, Aladashvili A, Elsaesser A, Kanakakis IG, Merkely B, Gudnason T, Iakobishvili Z, Bolognese L, Berkinbayev S, Bajraktari G, Beishenkulov M, Zake I, Lamin HB, Gustiene O, Pereira B, Xuereb RG, Ztot S, Juliebø V, Legutko J, Timóteo AT, Tatu-Chiţoiu G, Yakovlev A, Bertelli L, Nedeljkovic M, Studenčan M, Bunc M, García de Castro AM, Petursson P, Jeger R, Mourali MS, Yildirir A, Parkhomenko A, Gale CP, ESC Scientific Document Group. 2017 ESC guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J 2018;39:119–177.28886621 [Google Scholar]
- 117.Larsen JM, Ravkilde J.. Acute coronary angiography in patients resuscitated from out-of-hospital cardiac arrest—A systematic review and meta-analysis. Resuscitation 2012;83:1427–1433. [DOI] [PubMed] [Google Scholar]
- 118.Savonitto S.Prognostic value of the admission electrocardiogram in acute coronary syndromes. Jama 1999;281:707. [DOI] [PubMed] [Google Scholar]
- 119.Ambrose JA, Winters SL, Stern A, Eng A, Teichholz LE, Gorlin R, Fuster V.. Angiographie morphology and the pathogenesis of unstable angina pectoris. J Am Coll Cardiol 1985;5:609–616. [DOI] [PubMed] [Google Scholar]
- 120.Kerensky RA, Wade M, Deedwania P, Boden WE, Pepine CJ.. Revisiting the culprit lesion in non-Q-wave myocardial infarction. J Am Coll Cardiol 2002;39:1456–1463. [DOI] [PubMed] [Google Scholar]
- 121.Goldstein JA, Demetriou D, Grines CL, Pica M, Shoukfeh M, O’Neill WW.. Multiple complex coronary plaques in patients with acute myocardial infarction. N Engl J Med 2000;343:915–922. [DOI] [PubMed] [Google Scholar]
- 122.Weil BR, Suzuki G, Young RF, Iyer V, Canty JM.. Troponin release and reversible left ventricular dysfunction after transient pressure overload. J Am Coll Cardiol 2018;71:2906–2916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Turer AT, Addo TA, Martin JL, Sabatine MS, Lewis GD, Gerszten RE, Keeley EC, Cigarroa JE, Lange RA, Hillis LD, Lemos J. D.. Myocardial ischemia induced by rapid atrial pacing causes troponin T release detectable by a highly sensitive assay. J Am Coll Cardiol 2011;57:2398–2405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Siriwardena M, Campbell V, Richards AM, Pemberton CJ.. Cardiac biomarker responses to dobutamine stress echocardiography in healthy volunteers and patients with coronary artery disease. Clin Chem 2012;58:1492–1494. [DOI] [PubMed] [Google Scholar]
- 125.Bentzon JF, Otsuka F, Virmani R, Falk E.. Mechanisms of plaque formation and rupture. Circ Res 2014;114:1852–1866. [DOI] [PubMed] [Google Scholar]
- 126.Falk E, Nakano M, Bentzon JF, Finn AV, Virmani R.. Update on acute coronary syndromes: the pathologists’ view. Eur Heart J 2013;34:719–728. [DOI] [PubMed] [Google Scholar]
- 127.Hundley WG, Bluemke DA, Finn JP, Flamm SD, Fogel MA, Friedrich MG, Ho VB, Jerosch-Herold M, Kramer CM, Manning WJ, Patel M, Pohost GM, Stillman AE, White RD, Woodard PK, Writing Committee Members. ACCF/ACR/AHA/NASCI/SCMR 2010 Expert consensus document on cardiovascular magnetic resonance: a report of the American College of Cardiology Foundation Task Force on Expert Consensus Documents. Circulation 2010;121:2462–2508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Goyal A, Gluckman TJ, Tcheng JE.. What’s in a name?: The new ICD-10 (10th Revision of the International Statistical Classification of Diseases and Related Health Problems) codes and type 2 myocardial infarction. Circulation 2017;136:1180–1182. [DOI] [PubMed] [Google Scholar]
- 129.Tersalvi G, Vicenzi M, Calabretta D, Biasco L, Pedrazzini G, Winterton D.. Elevated troponin in patients with coronavirus disease 2019: possible mechanisms. J Card Fail 2020;26:470–475. [DOI] [PMC free article] [PubMed] [Google Scholar]