Abstract
Coronavirus disease 2019 (COVID-19), as a severe respiratory disease, affects various tissues and organs. The specific SARS-CoV-2 receptor, angiotensin-converting enzyme 2 (ACE2), is highly expressed in male gonads. Thus, male reproductive tissues could be a potential target for virus colonization. We performed a comprehensive search in PubMed and Google Scholar to retrieve relevant articles published till 15 April 2021. The keywords used were: male fertility, male reproductive health, semen parameters, sex hormones, SARS-CoV-2, and COVID-19. Validated evidence about the adverse effects of the SARS-CoV-2 infection on the male reproductive system is limited and few studies have reported semen analysis results or presence of viral RNA in semen samples of infected men. Nevertheless, alterations in reproductive hormones such as decreased level of testosterone (T) with raised luteinizing hormone (LH) have been reported in some patients. Although the impact of SARS-CoV-2 infection on the male reproduction health remains unclear, evidence suggests that male gonads may be potentially vulnerable to SARS-CoV-2 infection. In this article, we discussed the possible impacts of COVID-19 on male gonads, sex hormones, and semen quality and suggested preventive solutions.
Keywords: COVID-19, Fertility, Male Reproduction, SARS-CoV-2, Semen
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has led to a public health crisis affecting the world population (1). Coronaviruses are a group of human pathogenic viruses originated from animals. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection symptoms are fever, cough, shortness of breath and/or gastrointestinal disorders. While almost 80% of SARS-CoV-2 infections are mild, near 5 % are critical and death may occur in up to 3%. Older age and underlying disorders are major risk factors for severe SARS-CoV-2 infection (2). In addition to the abovementioned disorders, COVID-19 epidemic can also affect reproductive health (3).
SARS-CoV-2 enters the host cells through the cellular receptors including angiotensin-converting enzyme 2 (ACE2), and the trans-membrane serine protease (TMPRSS) (4). Wang and colleagues reported that ACE2 is expressed in spermatogonia, Leydig and Sertoli cells, while TMPRSS2 is expressed more in Sertoli cells compared to spermatogonia cells (5). Also, Hikmet et al. (6) reported high expression levels of ACE2 in Leydig and seminiferous ducts cells. Expression of other coronavirus-associated receptors including basigin (BSG), alanyl aminopeptidase (ANPEP) and dipeptidyl peptidase 4 (DPP4) in spermatogonia stem cell (SSC) and co-expression of ANPEP and DPP4 in prostate gland have been reported recently (7). These findings suggested that male gonads could be vulnerable following SARS-CoV-2 infection which might consequently result in spermatogenesis impairment. Previous studies reported that certain viruses such as human immunodeficiency virus (HIV), hepatitis B and C viruses, papilloma, mumps, and SARS coronavirus (SARS-CoV) cause orchitis and leading to germ cell destruction and sterility (8, 9). However, there are few clinical reports on the association of SARS-CoV-2 infection and orchitis. In addition, studies suggested that coronavirus might have indirect effects on testicular function through immunological and inflammatory responses disturbing the hypothalamic-pituitary-testis axis (10). Understanding whether SARS-CoV-2 can infect testicular tissue, is important for assessing its possible adverse effects on male fertility. Therefore, the goal of this review is to discuss probable adverse impact of COVID-19 on male reproduction and show the possibility of fertility complications. Accordingly, we searched for the following keywords in Google Scholar and PubMed: COVID-19, SARS-CoV-2, or severe acute respiratory syndrome coronavirus 2 AND male fertility, testes, semen, testosterone (T), assisted reproductive technology (ART), or reproductive system.
Cellular and molecular aspects of COVID-19 effects in the testis
ACE2 is expressed in various human organs such as kidneys and gastrointestinal tract (11). In addition, ACE2 is highly expressed in the testis (2), particularly on Leydig cells. SARS-CoV-2 could effect on the renin angiotensin system (RAS) via ACE2. The RAS controls cascade of hormones and receptors in the multiple organ systems and balances blood pressure. The pathological processes in COVID-19 leading to overreaction of the renin-angiotensin-aldosterone along with clinical abnormalities such as adverse cardiovascular and respiratory in infected patients (12). Accordingly, this could be important for the pathogenesis effects of SARS-CoV-2 in men patients. Studies confirmed high levels of ACE2 transcripts in spermatogonia cells, Sertoli and Leydig cells and a lower expression of ACE2 in late spermatocytes and spermatids (5). Shen et al. (13) showed that the positive rate of ACE2 expression in infertile men was higher than normal by single-cell RNA-sequencing. This indicates that men with reproductive disorder may be simply infected by this virus, however there isn’t any study which directly prove this subject. In addition, Pan and colleagues showed a correlation between ACE2 expression and severe impairment in spermatogenesis. In addition, it has been reported that 18% of infected patients had a scrotal discomfort (14). Moreover, several studies have demonstrated the effects of SARS-CoV-2 infection on the testis including orchitis with fibrin micro-thrombi (15), mild lymphocytic infiltration, decreased the percentage of Leydig cells, and seminiferous tubular damage (16, 17). Therefore, high expression of ACE2 in the testis could facilitate viral entry and colonization, which may have negative impact on male fertility and possible reproductive impairments (18). TMPRSS2 is strongly expressed in both early and late SSCs as well as Sertoli cells (7). TMPRSS2 is also highly expressed in the prostate, seminal vesicles and epididymis, where its aberrant expression is associated with tumorigenesis (14). Singh et al. (7) experimentally showed that other receptors such as ANPEP (SARS-CoV), DPP4 (MERS-CoV) and BGS (an alternate receptor for both SARS-CoV and SARS-CoV-2) facilitate SARS-CoV-2 entry. Also, the highest transcript levels of coronavirus-associated receptors factors )SCARFs( have been showed in SSC and SPG. Taken together, these observations indicated that spermatogonial cells may be highly prone to SARS-CoV-2 infection.
SARS-CoV-2 infection and male gonad function
Several studies reported low levels of T but high levels of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) in male patients who infected with different viruses. In fact, viral infections could disrupt normal function in Leydig cells and decrease T to LH ratio (T/LH). These studies suggested that viruses might have an indirect impact on testicular function via modulation of the hypothalamic-pituitary-testis axis (19).
Recently, the effect of SARS-CoV-2 infection was evaluated on gonadal function and male sex hormones; based on the results, LH level in serum was increased and ratios of FSH/LH and T/LH were decreased in recovered patients compared to the healthy men. However, other studies showed lower levels of total T and dihydrotestosterone in the patients with severe COVID-19 (20). Rastrelli et al. (20) reported that inflammatory cytokine storm in SARS-CoV-2 infection may be a potential risk factor for Leydig cells and would result in lower T levels, subsequently. Increased LH and decreased T/LH ratio may have occurred due to damage to Leydig cells and dysfunction of steroidogenic pathways after viral infection (21).
It seems that leukocytes and immune cells can pass the blood-testis barrier (BTB) and produce interferons that inhibit steroidogenesis and T production in the infected testis. Importantly, all above-mentioned symptoms are clinical phenotypes of primary hypogonadism. This phenomenon increases the risk of male infertility (22).
Epidemiological data showed higher COVID-19 mortality rate in men compared to women, which may suggest an important role for T in determining gender-disparity in SARS-CoV-2 infection (3, 23). Previous studies showed that certain moods have adverse effects on sperm quality parameters such as motility, concentration and DNA integrity (23, 24).
Accordingly, the outbreak of COVID-19 can be associated with depression and posttraumatic stress disorder (PTSD), which lead to activation of the central stress response system via the hypothalamic-pituitary-adrenal (HPA) axis (25). Continuous stimulation of the HPA axis by stressors might lead to dysregulation of this axis. Because of the dynamic interactions between the HPA axis and the hypothalamic-pituitary-gonadal (HPG) axis, any alteration in this regard could induce abnormalities in the reproductive health (26). Wdowiak et al. (27) showed that depression and anxiety are associated with decreased level of sex hormone-binding globulin (SHBG), but increased secretion of cortisol and prolactin in sub-fertile males.
It has been reported that SARS-CoV-2 infection can interfere with the production of FSH, T, and LH. Accordingly, serum LH level was higher in the COVID-19 patients and also the ratios of T: LH and FSH: LH were decreased in them compared to the control group (28). Ruan et al. (29) demonstrated that semen quality declined, while hormonal profiles remained normal in SARS-CoV-2 patients. Temiz et al. (30) evaluated sex hormone levels between COVID-19 patients before and after treatment and control group. Patients before treatment had significantly lower serum FSH, LH and T levels compared to the controls, which it could be consistent with the patient stress due to COVID-19.
SARS-CoV-2 infection and semen quality
Until now, only Li et al. (31) reported SARS-CoV-2 RNA detection in 15.8% of semen samples from COVID-19 patients after recovery. Moreover, Pan et al. (14) reported 19% of patients had scrotal discomfort at the time of COVID-19 diagnosis. Holtmann et al. (32) showed that in COVID-19 patients with moderate symptoms, sperm quality decreased. In addition, Ma and colleagues evaluated the sex-hormones and semen characteristics in 12 infected men. They didn’t find any SARS-CoV-2 in semen samples. Although they observed a higher serum LH and a lower ratio of T to LH in the COVID-19 patients compared to the control group. However, 8 out of 12 patients had normal semen quality (28). Temiz et al. (30) also evaluated the effects of SARS-CoV-2 infection on semen parameters and serum male sexual hormones. They reported that SARS-CoV-2 was not detected in the semen samples of infected patients and also all the semen parameters and hormone levels were normal except sperm morphology in some patients compared to the control. However, the levels of the median serum FSH, LH and T showed significant decreased in the COVID-19 patients. It seems that SARS-CoV-2 can increase apoptosis rate in spermatogonia cells following increased oxidative stress and reactive oxygen species (ROS) production which lead to reduced semen quality. Furthermore, Gacci et al. (33) showed that despite the absence of viral RNA in the semen, 25% of the 43 infected men were oligocrypto-azoospermic, whereas8 were azoospermic and 3 were oligospermic after recovery from COVID-19. Moreover, 33 out of 43 patients (76.7%) showed pathological levels of IL-8 in their semen, although the semen quality of these men were unknown before infection. The previous studies in terms of the detection of the SARS-CoV-2 virus in semen showed conflicting results (Table 1), thus further studies are needed to understand sexual transmissibility of the SARS-CoV-2 and its impact on sperm quality (34).
Table 1.
SARS-CoV-2 infection and semen quality
|
| ||||
|---|---|---|---|---|
| Patients (n) | Viral RNA in semen | Reproductive system symptoms | Semen quality | Reference |
|
| ||||
| 34 | Negative | Scrotal discomfort (19%) | Not assessed | (14) |
| 20 | Negative | Not reported | Decreased sperm concentration and motility | (32) |
| 38 | Positive (15.8%) | Not reported | Not assessed | (25) |
| 38 | Positive (26.7% in the acute stage of infection), Positive (8.7% in recovery) | Not reported | Not assessed | (31) |
| 12 | Negative | Not reported | 33.3% (low sperm motility with higher DFI) | (28) |
| 74 | Negative | Scrotal discomfort in one patient | Decreased semen quality | (29) |
| 30 | Negative | Not reported | Decreased sperm morphology | (30) |
| 43 | Negative | Not reported | 5% of the men were oligo-crypto-azoospermic, 8 were azoospermic and 3 were oligospermic | (33) |
|
| ||||
SARS-CoV-2; Severe acute respiratory syndrome coronavirus 2 and DFI; DNA fragmentation index.
SARS-CoV-2 infection and inflammatory responses
Previous studies have reported that SARS-CoV-2 could damage the male reproductive system by inflammatory caused by cytokine storm (35). The potential mechanisms of COVID-19-induced immunopathology in terms of immune cells including higher levels of neutrophils, lower levels of eosinophils, basophils, and monocytes. Depletion and exhaustion of lymphocytesincreased production of certain cytokines including IL-1β, IL-6, and IL-10 are other possible mechanisms of COVID-19 induced immunopathology.
In addition, SARS-CoV-2 damages lymphatic organs, such as the spleen and lymph nodes, leading to lymphopenia (36). Based on the recent reports, inflammatory responses in SARS-CoV-2 infection could be classified as primary and secondary responses. Primary inflammatory responses happen after viral infection, prior to the advent of neutralizing antibodies (NAbs). Secondary inflammatory responses start with the generation of adaptive immunity and NAbs. In fact, the virus-NAb complex causes FcR-mediated inflammatory responses, skewing macrophage responses, cellular damages and acute lung injury (37).
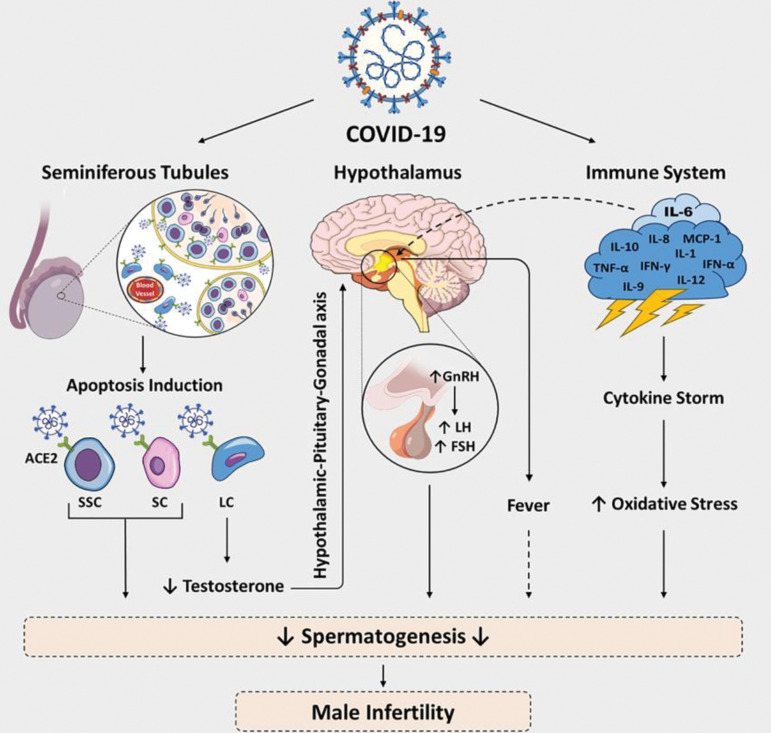
The main cause of COVID-19 severity is the cytokine storm which is associated with increasing serum levels of interleukin-6 (IL-6), IL-7, IL-8 and tumor necrosis factor alpha (TNF-α) (38, 39). Systemic viral infections such as influenza can indirectly interfere with male reproduction. Their detrimental effects can be exerted by fever, immune cell activation, and elevated inflammatory mediators (cytokines and interferons), which may have inhibitory effects on spermatogenesis and steroidogenesis (Fig .1) (8).
Fig.1.
Risk factors, mechanisms and pathophysiology of COVID-19 on the male reproduction. ACE2; Angiotensin-converting enzyme 2, SSC; Spermatogonia stem cell, LC; Leydig cell, SC; Sertoli cell, GnRH; Gonadotrophin-releasing hormone, LH; Luteinizing hormone, FSH; Follicle-stimulating hormone, and IL; Interleukin.
Furthermore, increased IgG precipitation and leukocyte infiltration were shown in testicular interstitial tissue following viral infection (7). High levels of inflammatory cytokines produced by leukocytes, T cells and macrophages following viral infections, can deteriorate spermatogenesis and steroidogenesis (22). For other coronaviruses such as SARS-CoV, reproductive system damage was observed in males as well. Accordingly, Xu and colleagues showed IgG deposition in seminiferous epithelium and degenerated germ cells and Sertoli cells in SARS-CoV patients. However, they did not observe SARS-CoV related mRNA in the testicular tissue (8). The observed effect could be due to an immune-mediated response to SARS-CoV infection.
Moreover, the gene ontology (GO) analysis showed that genes related to immune reaction were increased in ACE2- positive Leydig and Sertoli cells, while reproductive-related genes were down-regulated (6). It was suggested that increased endothelial cell death and vascular permeability could promote production of pro-inflammatory cytokines, which can lead to dysfunction of the renin-angiotensin system (RAS), and further exacerbate the inflammation (40). In addition, inflammation caused by SARS-CoV-2 infection can increase immune cell recruitment in testicular tissue and affect the BTB integrity (22). Epididymitis was reported using color Doppler ultrasound in 42.3% of mild-to-moderate COVID-19 men, which 54.5% of them presenting with enlarged epididymis head, and 19.2% had bilateralism. It could be an indirect deleterious effect of inflammation that targeted the epididymis (41). Further studies are needed to provide enough evidence about the role of the inflammatory responses in male infertility under SARS-CoV-2 infection.
COVID-19 and male fertility
Documented information about the adverse effect of SARS-CoV-2 infection on male fertility is limited. Similar to other coronaviruses, several mechanisms may be involved in testicular dysfunction and male infertility caused by SARS-CoV-2 infection. It was shown that some members of the coronavirus family can cause orchitis and testicular dysfunctions. Pathological findings from SARS-CoV-infected patients showed that this virus induces apoptosis in spermatogonia cells and, increases leukocyte infiltration (8). Influenza viruses promote oxidative stress (42) that negatively influences spermogram parameters including motility and viability (39). In this context, it can be assumed that SARS-CoV-2 may affect sperm function through systemic inflammation leading to enhanced oxidative stress. Only one study reported that SARS-CoV-2 can decrease sperm concentration and motion parameters in infected patients (43), while other studies recommended assessing the adverse effects of SARS-CoV-2 on semen quality in infected and recovering patients. Fever that associated with viral infections causes spermatogenesis impairment, e.g. SARS, leads to increased apoptosis in testicular germ cells (44) and disrupted spermatogenesis (45). The percentage of asymptomatic infection in the children under 10 years old is about 16% and negative effects of infection may be a serious challenge for their future reproductive health. Therefore, long-term side effects of high fever on male fertility should be considered following SARS-CoV-2 infection. SARS-CoV-2 may cause testicular dysfunction by two distinct effects, direct or indirect, due to the presence of receptors such as ACE2 in the testis or immunological and inflammatory responses and dysregulated male sex hormones. However, supportive data for direct adverse effects are controversial (Fig .1) (37).
The effect of COVID-19 treatment on male fertility
Currently, there are no highly effective therapeutic solutions to control SARS-CoV-2. However, antiviral drugs such as interferons, ribavirin, lopinavir/ritonavir, antibiotics such as moxifloxacin and azithromycin, and glucocorticoids are recommended for COVID-19 treatment (2, 25) .Several studies noted that some of the mentioned drugs have adverse effects on male reproductive health (46). Almasry et al. (47) have indicated that ribavirin decreased T levels and impaired spermatogenesis in animal models. Additionally, sperm abnormalities and decreased sperm count were observed in rats treated with ribavirin (48, 49). Lopinavir/ritonavir could also impair spermatogenesis in male rats (50). Glucocorticoids could destroy cell junctions in spermatogenic epithelium and the BTB, which cause germ cell apoptosis in testicular tissue (46). Moreover, it was reported that excessive use of disinfectants could lead to a decrease in sperm count and quality in mice (51). Side effects of drugs used for the treatment of COVID-19 on male reproductive health should be considered cautiously.
Managing the possible effects of COVID-19 on male reproduction
Management of SARS-CoV-2 possible adverse effects on male fertility is complex due to the underlying pathophysiology and associated co-morbidities. It appears that SARS-CoV-2 may have indirect impacts on the male reproductive system by inducing inflammatory storm (25) and infected male fertility remains at risk due to the increased oxidative stress level. Therefore, immune response modulation and inflammation management were suggested for the disease management (52-54). Treatment with various antioxidants including vitamin E, C and D as well as selenium, and zinc, which positively modulate inflammation and oxidative stress, may be warranted (55- 57). In addition, it was reported that supplementation with omega-3 fatty acids is associated with better testicular function (58). Antioxidant treatment can reduce the risk of DNA damage in sperms and improve fertility through improving semen parameters (55, 56). Therefore, administration of antioxidants and immunosuppressants could be a novel therapeutic strategy against possible male subfertility induced by COVID-19. However, currently there is no approved guideline for nutrition in the male patients. In addition, because of global financial crisis during SARS-CoV-2 infection many fertile couples decide to postpone the time of parenthood. Therefore, it may cause primary or secondary infertility in the future.
Additionally, selective infecundity may cause biological infertility which might be linked to short and mid-term increase in global infertility (59). Generally, there are a few proposals for individualized clinical management in ART services to alleviate the adverse effect of the coronavirus pandemic. The global guidelines such as the American Society for Reproductive Medicine (ASRM) and the Society for Assisted Reproductive Technology (SART) recommended caution to couples planning natural pregnancy or assisted reproduction (60). ART treatment should be carried out for infertile patients who need immediate treatment. Anthological services such as fertility preservation should be available during any treatment. Patients and healthcare providers should use proper personal protective equipment. It seems training ART staff, following social distancing protocols, psychological support, and prioritizing of COVID-19 patients are necessary (61). In addition, because of global financial crisis during SARS-CoV-2 infection many fertile couples decide to postpone the time of parenthood. Therefore, it may cause primary or secondary infertility in the future. Additionally, selective infecundity may cause biological infertility which might be linked to short and mid-term increase in global infertility (59).
Conclusion
So far, there is limited evidence about SARS-CoV-2 pathogenicity on male reproductive health. ACE2 protein as a functional receptor for SARS-CoV-2 is highly expressed on testicular cells. TMPRSS2 receptor also has a high expression level in Sertoli cells. Thus, male gonads may be potentially vulnerable to SARS-CoV-2 infection. Although limited data is currently available, detection of SARS-CoV-2 virus in semen of some patients may indicates the possibility of sexual transmission in COVID-19. Follow-up studies are required to investigate this possibility. Moreover, DNA fragmentation and defective sperm function might be occurred during SARS-CoV-2 infection because of inflammation responses and increased oxidative stress in the patients. DNA damage in sperm can lead to a lower fertility rate or idiopathic infertility. On the other hand, recent reports revealed that all the parameters in the semen samples were similar between the COVID-19 patients and the healthy control group and the only significant reduction in the percentage of the sperms with normal morphology has been observed in the COVID-19 patients, which could be the result of fever in the infected men. In addition, epididymitis could be as an indirect effect of inflammation in COVID-19 men, thus semen analysis and testicular evaluation are recommended in the patients after recovery. Some antiviral drugs have adverse impact on the male reproductive health system. Hence, further studies should investigate in terms of a safe treatment strategy for male fertility following SARS-CoV-2 infection. Furthermore, psychological consultation and clinical information should be provided to couples during the outbreak of COVID-19 and contraception could be suggested for the patients who were COVID-19 positive and received antiviral treatment. Nonetheless, further research is needed for assessing the impact of SARS-CoV-2 infection on testicular tissue, sex hormones and sperm quality.
Acknowledgements
We thank Dr. Amiri Amiri-Yekta for his wonderful comments. The authors declare that there is no financial support and conflict of interest in this study.
Authors’ Contributions
M.H.; Drafted the manuscript, which was revised by A.Sh. and M.V. B.Sh.; Designed the figure of the manuscript and contributed to conception and design. A.Sh., P.T.; Contributed to the design of the study. M.H.N.-E; Provided critical revision of the manuscript. All authors read and approved the final manuscript.
References
- 1.Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, et al. A novel Coronavirus from patients with Pneumonia in China, 2019. N Engl J Med. 2020;382(8):727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jin Y, Yang H, Ji W, Wu W, Chen S, Zhang W, et al. Virology, epidemiology, pathogenesis, and control of COVID-19. Viruses. 2020;12(4):372–372. doi: 10.3390/v12040372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Anifandis G, Messini CI, Daponte A, Messinis IE. COVID-19 and fertility: a virtual reality. Reprod Biomed Online. 2020;41(2):157–159. doi: 10.1016/j.rbmo.2020.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li SR, Tang ZJ, Li ZH, Liu X. Searching therapeutic strategy of new coronavirus pneumonia from angiotensin-converting enzyme 2: the target of COVID-19 and SARS-CoV. Eur J Clin Microbiol Infect Dis. 2020;39(6):1021–1026. doi: 10.1007/s10096-020-03883-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang Z, Xu X. scRNA-seq profiling of human testes reveals the presence of the ACE2 receptor, a target for SARS-CoV-2 infection in spermatogonia, leydig and sertoli cells. Cells. 2020;9(4):920–920. doi: 10.3390/cells9040920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hikmet F, Méar L, Edvinsson Å, Micke P, Uhlén M, Lindskog C. The protein expression profile of ACE2 in human tissues. Mol Syst Biol. 2020;16(7):e9610–e9610. doi: 10.15252/msb.20209610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Singh M, Bansal V, Feschotte C. A single-cell RNA expression map of human coronavirus entry factors. Cell Rep. 2020;32(12):108175–108175. doi: 10.1016/j.celrep.2020.108175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Xu J, Qi L, Chi X, Yang J, Wei X, Gong E, et al. Orchitis: a complication of severe acute respiratory syndrome (SARS)1. Biol Reprod. 2006;74(2):410–416. doi: 10.1095/biolreprod.105.044776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Liu W, Han R, Wu H, Han D. Viral threat to male fertility. Andrologia. 2018;50(11):e13140–e13140. doi: 10.1111/and.13140. [DOI] [PubMed] [Google Scholar]
- 10.Abobaker A, Raba AA. Does COVID-19 affect male fertility? World J Urol. 2021;39(3):975–976. doi: 10.1007/s00345-020-03208-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Khoshdel-Rad N, Zahmatkesh E, Shpichka A, Timashev P, Vosough M. Outbreak of chronic renal failure: will this be a delayed heritage of COVID-19? J Nephrol. 2020;34(1):3–5. doi: 10.1007/s40620-020-00851-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wiese OJ, Allwood BW, Zemlin AE. COVID-19 and the renin-angiotensin system (RAS): a spark that sets the forest alight? Med Hypotheses. 2020;144:110231–110231. doi: 10.1016/j.mehy.2020.110231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shen Q, Xiao X, Aierken A, Yue W, Wu X, Liao M, et al. The ACE2 expression in Sertoli cells and germ cells may cause male reproductive disorder after SARS-CoV-2 infection. J Cell Mol Med. 2020;24(16):9472–9477. doi: 10.1111/jcmm.15541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pan F, Xiao X, Guo J, Song Y, Li H, Patel DP, et al. No evidence of severe acute respiratory syndrome-coronavirus 2 in semen of males recovering from coronavirus disease 2019. Fertil Steril. 2020;113(6):1135–1139. doi: 10.1016/j.fertnstert.2020.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Duarte-Neto AN, Monteiro RAA, da Silva LFF, Malheiros DMAC, de Oliveira EP, Theodoro-Filho J, et al. Pulmonary and systemic involvement in COVID-19 patients assessed with ultrasound-guided minimally invasive autopsy. Histopathology. 2020;77(2):186–197. doi: 10.1111/his.14160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Teixeira TA, Bernardes FS, Oliveira YC, Hsieh MK, Esteves SC, Duarte-Neto AN, et al. SARS-CoV-2 and multi-organ damage - what men’s health specialists should know about the COVID-19 pathophysiology. Int Braz J Urol. 2021;47(3):637–646. doi: 10.1590/S1677-5538.IBJU.2020.0872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Teixeira TA, Oliveira YC, Bernardes FS, Kallas EG, Duarte-Neto AN, Esteves SC, et al. Viral infections and implications for male reproductive health. Asian J Androl. 2021;23(4):335–347. doi: 10.4103/aja.aja_82_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lukassen S, Chua RL, Trefzer T, Kahn NC, Schneider M, Muley T, et al. SARS-CoV-2 receptor ACE2 and TMPRSS2 are primarily expressed in bronchial transient secretory cells. EMBO J. 2020;39(10):e105114–e105114. doi: 10.15252/embj.20105114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pozzilli P, Lenzi A. Commentary: testosterone, a key hormone in the context of COVID-19 pandemic. Metabolism. 2020;108:154252–154252. doi: 10.1016/j.metabol.2020.154252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rastrelli G, Di Stasi V, Inglese F, Beccaria M, Garuti M, Di Costanzo D, et al. Low testosterone levels predict clinical adverse outcomes in SARS-CoV-2 pneumonia patients. Andrology. 2021;9(1):88–98. doi: 10.1111/andr.12821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Salonia A, Corona G, Giwercman A, Maggi M, Minhas S, Nappi RE, et al. SARS-CoV-2, testosterone and frailty in males (PROTEGGIMI): a multidimensional research project. Andrology. 2021;9(1):88–98. doi: 10.1111/andr.12811. [DOI] [PubMed] [Google Scholar]
- 22.Verma S, Saksena S, Sadri-Ardekani H. ACE2 receptor expression in testes: implications in coronavirus disease 2019 pathogenesis. Biol Reprod. 2020;103(3):449–451. doi: 10.1093/biolre/ioaa080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Evans-Hoeker EA, Eisenberg E, Diamond MP, Legro RS, Alvero R, Coutifaris C, et al. Major depression, antidepressant use, and male and female fertility. Fertil Steril. 2018;109(5):879–887. doi: 10.1016/j.fertnstert.2018.01.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Vellani E, Colasante A, Mamazza L, Minasi MG, Greco E, Bevilacqua A. Association of state and trait anxiety to semen quality of in vitro fertilization patients: a controlled study. Fertil Steril. 2013;99(6):1565–1572. doi: 10.1016/j.fertnstert.2013.01.098. [DOI] [PubMed] [Google Scholar]
- 25.Li R, Yin T, Fang F, Li Q, Chen J, Wang Y, et al. Potential risks of SARS-CoV-2 infection on reproductive health. Reprod Biomed Online. 2020;41(1):89–95. doi: 10.1016/j.rbmo.2020.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Joseph DN, Whirledge S. Stress and the HPA Axis: balancing homeostasis and fertility. Int J Mol Sci. 2017;18(10):2224–2224. doi: 10.3390/ijms18102224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wdowiak A, Bien A, Iwanowicz-Palus G, Makara-Studzińska M, Bojar I. Impact of emotional disorders on semen quality in men treated for infertility. Neuro Endocrinol Lett. 2017;38:50–58. [PubMed] [Google Scholar]
- 28.Ma L, Xie W, Li D, Shi L, Ye G, Mao Y, et al. Evaluation of sexrelated hormones and semen characteristics in reproductive-aged male COVID-19 patients. J Med Virol. 2021;93(1):456–462. doi: 10.1002/jmv.26259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ruan Y, Hu B, Liu Z, Liu K, Jiang H, Li H, et al. No detection of SARS-CoV-2 from urine, expressed prostatic secretions, and semen in 74 recovered COVID-19 male patients: a perspective and urogenital evaluation. Andrology. 2021;9(1):99–106. doi: 10.1111/andr.12939. [DOI] [PubMed] [Google Scholar]
- 30.Temiz MZ, Dincer MM, Hacibey I, Yazar RO, Celik C, Kucuk SH, et al. Investigation of SARS-CoV-2 in semen samples and the effects of COVID-19 on male sexual health by using semen analysis and serum male hormone profile: a cross-sectional, pilot study. Andrologia. 2021;53(2):e13912–e13912. doi: 10.1111/and.13912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Li D, Jin M, Bao P, Zhao W, Zhang S. Clinical characteristics and results of semen tests among men with coronavirus disease 2019. JAMA Netw Open. 2020;3(5):e208292–e208292. doi: 10.1001/jamanetworkopen.2020.8292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Holtmann N, Edimiris P, Andree M, Doehmen C, Baston-Buest D, Adams O, et al. Assessment of SARS-CoV-2 in human semen-a cohort study. Fertil Steril. 2020;114(2):233–238. doi: 10.1016/j.fertnstert.2020.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gacci M, Coppi M, Baldi E, Sebastianelli A, Zaccaro C, Morselli S, et al. Semen impairment and occurrence of SARS-CoV-2 virus in semen after recovery from COVID-19. Hum Reprod. 2021;36(6):1520–1529. doi: 10.1093/humrep/deab026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Perry MJ, Arrington S, Neumann LM, Carrell D, Mores CN. It is currently unknown whether SARS-CoV-2 is viable in semen or whether COVID-19 damages spermatozoa. Andrology. 2021;9(1):30–32. doi: 10.1111/andr.12831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tian Y, Zhou LQ. Evaluating the impact of COVID-19 on male reproduction. Reproduction. 2021;161(2):R37–R44. doi: 10.1530/REP-20-0523. [DOI] [PubMed] [Google Scholar]
- 36.Yang L, Liu S, Liu J, Zhang Z, Wan X, Huang B, et al. COVID-19: immunopathogenesis and Immunotherapeutics. Signal Transduct Target Ther. 2020;5(1):128–128. doi: 10.1038/s41392-020-00243-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fu Y, Cheng Y, Wu Y. Understanding SARS-CoV-2-mediated inflammatory responses: from mechanisms to potential therapeutic tools. Virol Sin. 2020;35(3):266–271. doi: 10.1007/s12250-020-00207-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Baradaran Ghavami SH, Shahrokh SH, Hossein-Khannazer N, Shpichka A, Asadzadeh Aghdaei H, Timashev P, et al. IBD patients could be silent carriers for novel coronavirus and less prone to its severe adverse events: true or false? Cell J. 2020;22(Suppl 1):151–154. doi: 10.22074/cellj.2020.7603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Agarwal A, Rana M, Qiu E, AlBunni H, Bui AD, Henkel R. Role of oxidative stress, infection and inflammation in male infertility. Andrologia. 2018;50(11):e13126–e13126. doi: 10.1111/and.13126. [DOI] [PubMed] [Google Scholar]
- 40.Imai Y, Kuba K, Penninger JM. The discovery of angiotensinconverting enzyme 2 and its role in acute lung injury in mice. Exp Physiol. 2008;93(5):543–548. doi: 10.1113/expphysiol.2007.040048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Carneiro F, Teixeira TA, Bernardes FS, Pereira MS, Milani G, Duarte-Neto AN, et al. Radiological patterns of incidental epididymitis in mild-to-moderate COVID-19 patients revealed by colour Doppler ultrasound. Andrologia. 2021;53(4):e13973–e13973. doi: 10.1111/and.13973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Khomich OA, Kochetkov SN, Bartosch B, Ivanov AV. Redox biology of respiratory viral infections. Viruses. 2018;10(8):392–392. doi: 10.3390/v10080392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hamatani T. Spermatozoal RNA profiling towards a clinical evaluation of sperm quality. Reprod Biomed Online. 2011;22(2):103–105. doi: 10.1016/j.rbmo.2010.12.001. [DOI] [PubMed] [Google Scholar]
- 44.Jung A, Schuppe HC. Influence of genital heat stress on semen quality in humans. Andrologia. 2008;39(6):203–215. doi: 10.1111/j.1439-0272.2007.00794.x. [DOI] [PubMed] [Google Scholar]
- 45.Carlsen E, Andersson AM, Petersen JH, Skakkebaek NE. History of febrile illness and variation in semen quality. Hum Reprod. 2003;18(10):2089–2092. doi: 10.1093/humrep/deg412. [DOI] [PubMed] [Google Scholar]
- 46.Drobnis EZ, Nangia AK. Antivirals and male reproduction. Adv Exp Med Biol. 2017;1034:163–178. doi: 10.1007/978-3-319-69535-8_11. [DOI] [PubMed] [Google Scholar]
- 47.Almasry SM, Hassan ZA, Elsaed WM, Elbastawisy YM. Structural evaluation of the peritubular sheath of rat’s testes after administration of ribavirin: a possible impact on the testicular function. Int J Immunopathol Pharmacol. 2017;30(3):282–296. doi: 10.1177/0394632017726261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Narayana K, D’Souza UJA, Rao KP. Effect of ribavirin on epididymal sperm count in rat. Indian J Physiol Pharmacol. 2002;46(1):97–101. [PubMed] [Google Scholar]
- 49.Narayana K, D’Souza UJ, Seetharama Rao KP. Ribavirin-induced sperm shape abnormalities in Wistar rat. Mutat Res. 2002;513(1- 2):193–196. doi: 10.1016/s1383-5718(01)00308-4. [DOI] [PubMed] [Google Scholar]
- 50.Adaramoye OA, Akanni OO, Adewumi OM, Owumi SE. Lopinavir/ ritonavir, an antiretroviral drug, lowers sperm quality and induces testicular oxidative damage in rats. Tokai J Exp Clin Med. 2015;40(2):51–57. [PubMed] [Google Scholar]
- 51.Sakamoto KQ, Ishizuka M, Kazusaka A, Fujita S. Iodine intake as a possible cause of discontinuous decline in sperm counts: a reevaluation of historical and geographic variation in semen quality. Jpn J Vet Res. 2004;52(2):85–94. [PubMed] [Google Scholar]
- 52.Hossein-Khannazer N, Shokoohian B, Shpichka A, Asadzadeh Aghdaei H, Timashev P, Vosough M. Novel therapeutic approaches for treatment of COVID-19. J Mol Med (Berl) 2020;98(6):789–803. doi: 10.1007/s00109-020-01927-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ramezankhani R, Solhi R, Memarnejadian A, Nami F, Hashemian SMR, Tricot T, et al. Therapeutic modalities and novel approaches in regenerative medicine for COVID-19. Int J Antimicrob Agents. 2020;56(6):106208–106208. doi: 10.1016/j.ijantimicag.2020.106208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hossein-Khannazer N, Shokoohian B, Shpichka A, Aghdaei HA, Timashev P, Vosough M. An update to “novel therapeutic approaches for treatment of COVID-19”. J Mol Med (Berl) 2021;99(2):303–310. doi: 10.1007/s00109-020-02027-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gharagozloo P, Gutiérrez-Adán A, Champroux A, Noblanc A, Kocer A, Calle A, et al. A novel antioxidant formulation designed to treat male infertility associated with oxidative stress: promising preclinical evidence from animal models. Hum Reprod. 2016;31(2):252–262. doi: 10.1093/humrep/dev302. [DOI] [PubMed] [Google Scholar]
- 56.Giahi L, Mohammadmoradi S, Javidan A, Sadeghi MR. Nutritional modifications in male infertility: a systematic review covering 2 decades. Nutr Rev. 2016;74(2):118–130. doi: 10.1093/nutrit/nuv059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Abbasihormozi Sh, Kouhkan A, Alizadeh AR, Shahverdi AH, NasrEsfahani MH, Sadighi Gilani MA, et al. Association of vitamin D status with semen quality and reproductive hormones in Iranian subfertile men. Andrology. 2017;5(1):113–118. doi: 10.1111/andr.12280. [DOI] [PubMed] [Google Scholar]
- 58.Esmaeili V, Shahverdi AH, Moghadasian MH, Alizadeh AR. Dietary fatty acids affect semen quality: a review. Andrology. 2015;3(3):450–461. doi: 10.1111/andr.12024. [DOI] [PubMed] [Google Scholar]
- 59.Trinchant RM, Cruz M, Marqueta J, Requena A. Infertility and reproductive rights after COVID-19 pandemic. Reprod Biomed Online. 2020;41(2):151–153. doi: 10.1016/j.rbmo.2020.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Khalili MA, Leisegang K, Majzoub A, Finelli R, Panner Selvam MK, Henkel R, et al. Male fertility and the COVID-19 pandemic: systematic review of the literature. World J Mens Health. 2020;38(4):506–520. doi: 10.5534/wjmh.200134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Alviggi C, Esteves SC, Orvieto R, Conforti A, La Marca A, Fischer R, et al. COVID-19 and assisted reproductive technology services: repercussions for patients and proposal for individualized clinical management. Reprod Biol Endocrinol. 2020;18(1):45–45. doi: 10.1186/s12958-020-00605-z. [DOI] [PMC free article] [PubMed] [Google Scholar]